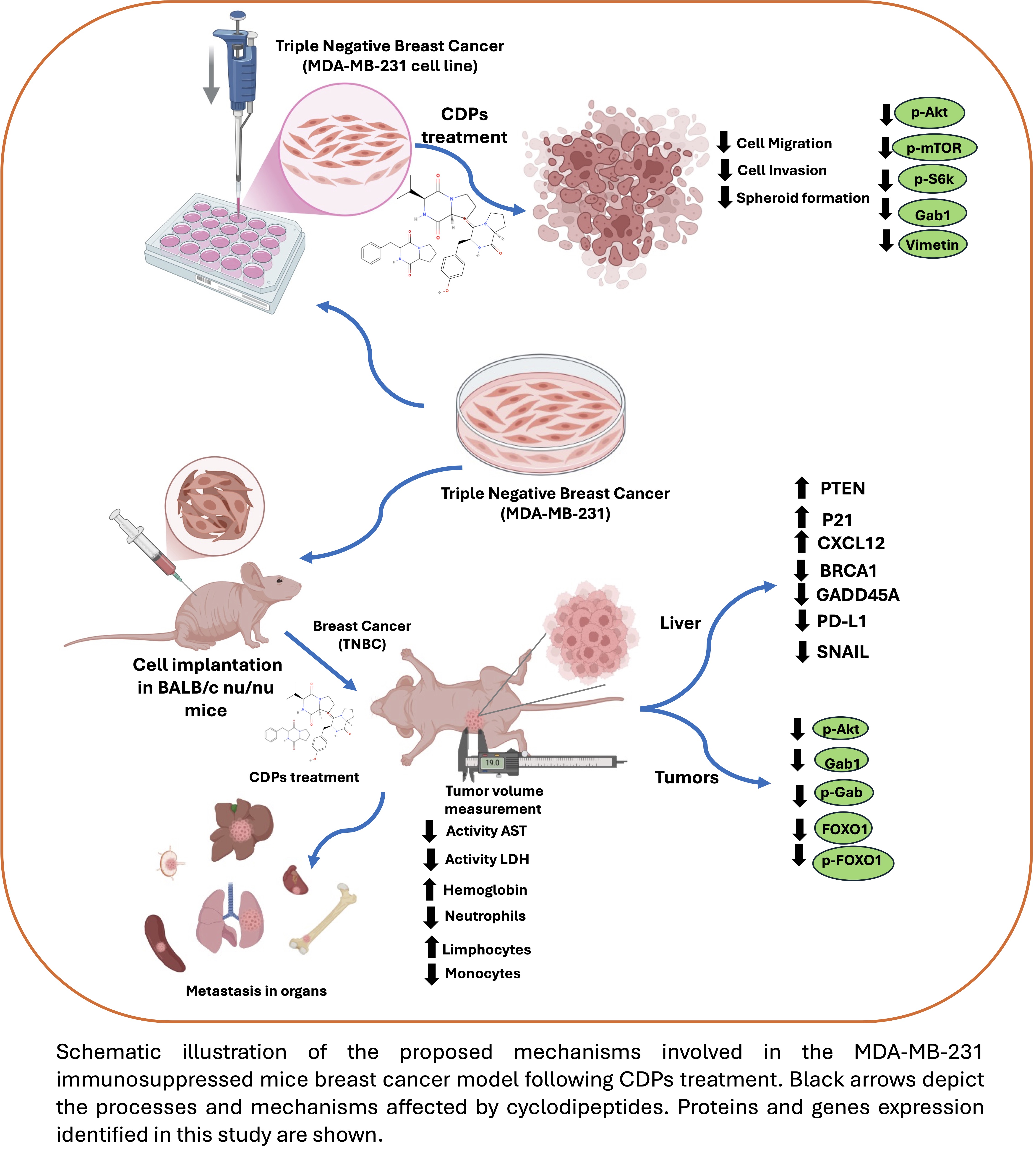
3. Discussion
CDPs have become promising options for cancer treatment because of their stability, high selectivity, and capacity to block important cancer signaling pathways [
11]. Their cytotoxic and pro-apoptotic effects have been shown in multiple cancer cell lines [
3,
5,
6,
7,
8]. Beyond their direct anti-proliferative activity, several diketopiperazines have been shown to interfere with mechanisms associated with tumor aggressiveness and therapeutic resistance. For example, fumitremorgin C analogs can overcome multidrug resistance by inhibiting the breast cancer resistance protein (BCRP), a key efflux transporter that limits intracellular drug accumulation. Similarly, the HLY838 diketopiperazine functions as an O-GlcNAc transferase (OGT) inhibitor, enhancing the anti-tumor effects of CDK9 inhibitors by downregulating c-Myc and E2F1 [
12]. Additionally, Verticillin A inhibits c-Met phosphorylation and downstream Ras/Raf/MEK/ERK signaling, leading to decreased metastatic potential in colon cancer models [
13]. Together, these studies emphasize the potential of CDPs and related diketopiperazines to act as multitarget anticancer and anti-metastatic agents. In line with this idea, earlier research in mouse melanoma showed that CDPs from Pseudomonas aeruginosa significantly reduced key EMT markers, including MMP-1, E-cadherin, N-cadherin, HIF-1α, Vimentin, and CK-1, supporting their capacity to disrupt tumor cell migration and invasion [
6,
10]. These observations support exploring the anti-invasive and metastasis-associated effects of bacterial CDPs in aggressive breast cancer subtypes such as triple-negative breast cancer.
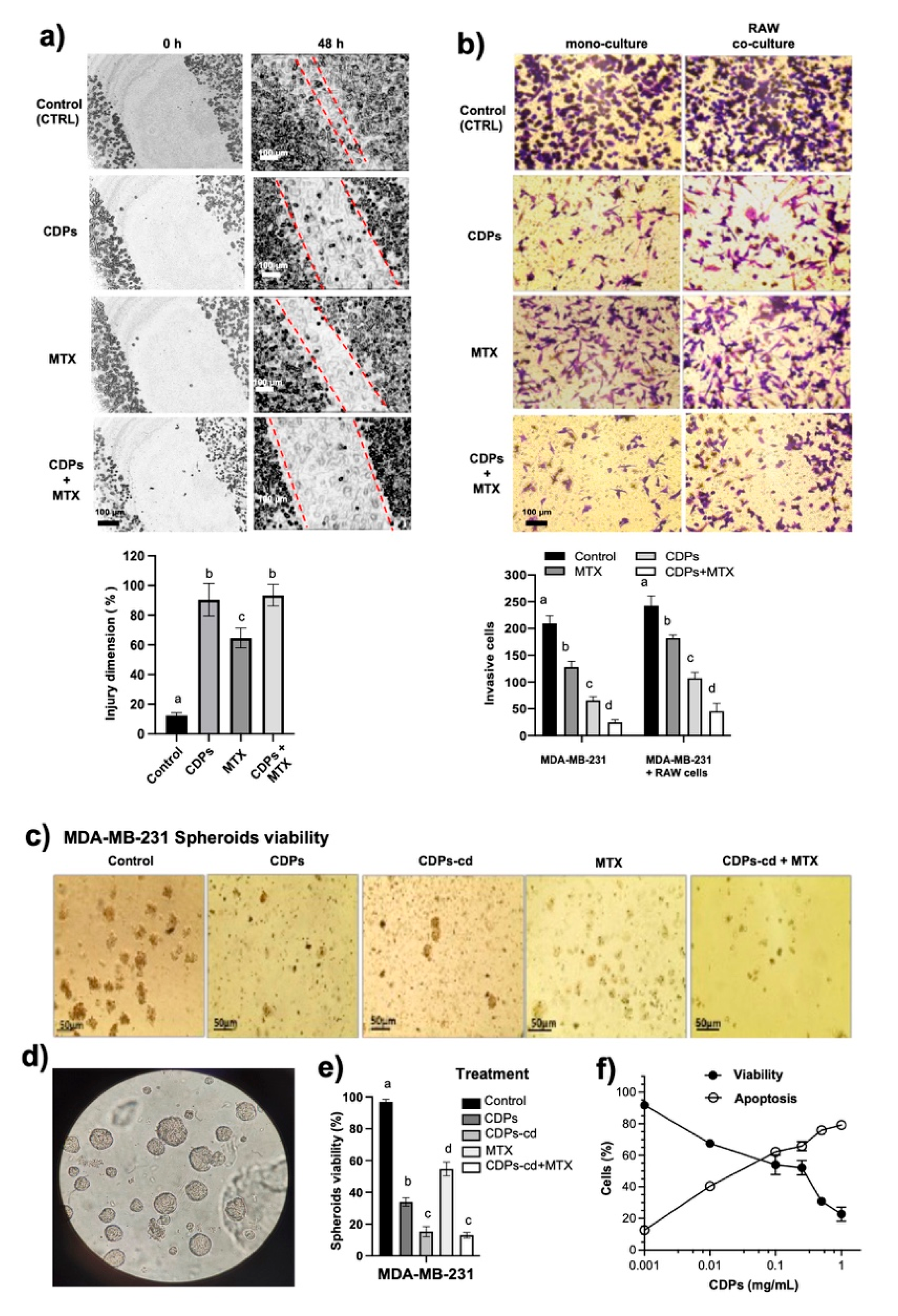
In this study, functional assays further confirm the ability of bacterial CDPs to suppress invasion and metastasis-associated behaviors in the TNBC model. Wound-healing assays demonstrated that CDPs inhibited the migratory capacity of MDA-MB-231 cells more effectively than methotrexate (MTX) (
Figure 1a). Similarly, transwell invasion assays show that CDPs significantly reduce the invasive capacity of MDA-MB-231 cells under both monoculture and macrophage co-culture conditions compared to MTX, with their combination being even more effective. Tumor-associated macrophages (TAMs) play a key role in tumor progression by secreting chemokines and growth factors that enhance cancer cell proliferation, migration, and invasiveness [
14]. Our data suggest that CDPs disrupt TAM–cancer cell interactions in the MDA-MB-231 cell line, emphasizing their dual role in promoting apoptosis and inhibiting metastasis-related processes, thereby supporting their function as inhibitors of tumor cell migration and invasiveness. This data is highly relevant because the interaction between cancer cells and macrophages, along with the release of chemokines, stimulates malignancy and the invasive capacity of cancer cells. The superior inhibition observed with the combination of CDPs and MTX (up to 90% inhibition of invasiveness) indicates synergy between these agents.
Three-dimensional spheroid models further demonstrated the anti-metastatic and anti-tumor effects of CDPs. Spheroid cultures are enriched with cancer stem cell (CSC)-like populations that show increased self-renewal and resistance to therapy [
14]. Therefore, the spheroid structure in the MDA-MB-231 lines promotes resistance to cytotoxic compounds. Our results showed that CDPs significantly reduced spheroid size and number, lowered cell viability, and increased apoptosis in the MDA-MB-231 spheroids compared to MTX-treated cultures. The effects were more pronounced when using the CDPs+MTX combination or cyclodextrins (
Figure 1c–e). Additionally, the spheroid integration assay, which mimics three-dimensional tumor growth and plasticity, supports the ability of CDPs as agents that can disrupt key stages of tumor progression and stemness-related properties. These findings emphasize the potential of CDPs to combat CSC-associated resistance mechanisms in aggressive breast cancer models.
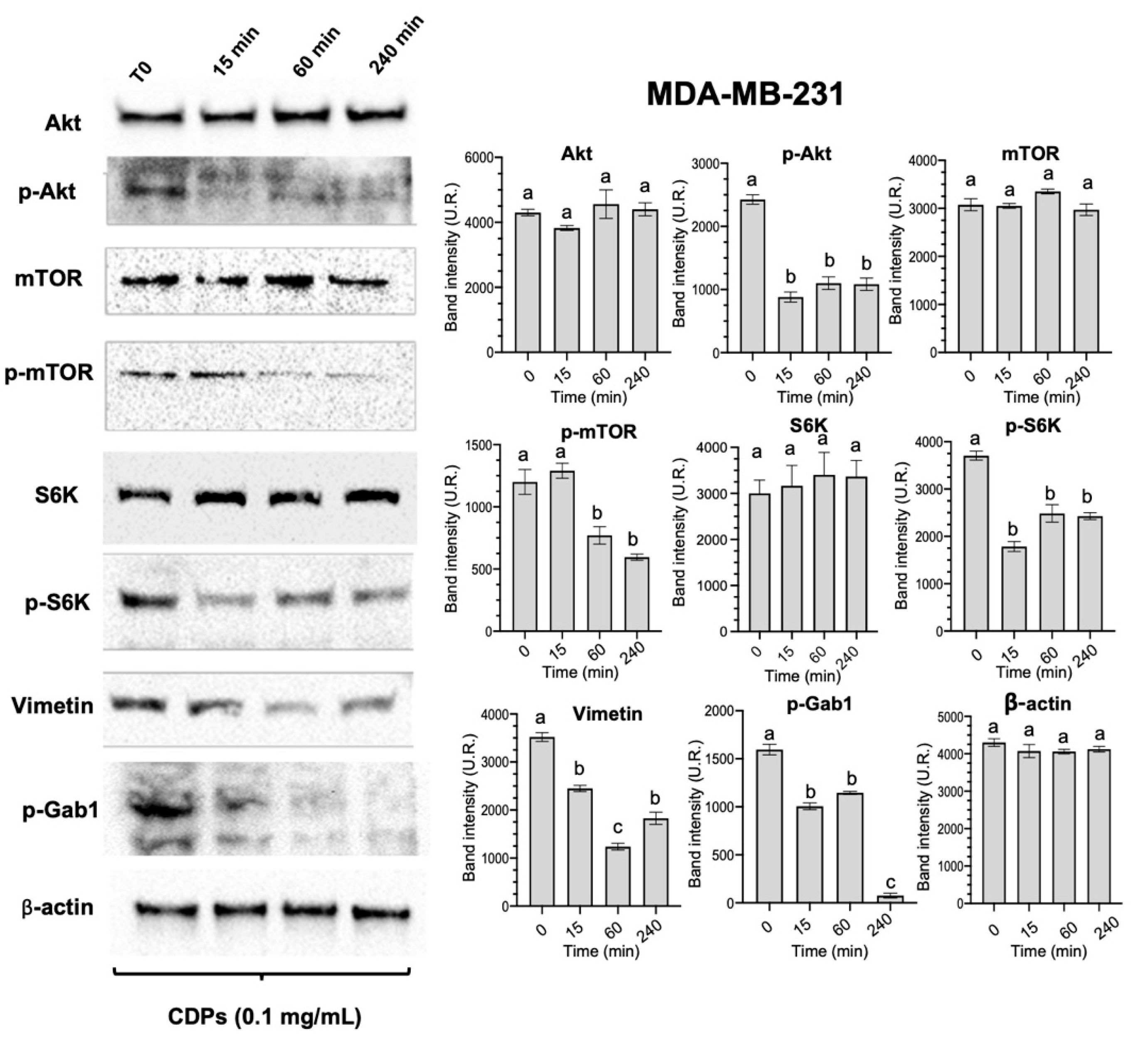
Mechanistically, our results further support previous findings that indicate CDPs act through the PI3K/Akt/mTOR pathway [
3,
5,
6,
10]. In MDA-MB-231 cells, CDPs significantly decreased phosphorylation of Akt, mTOR, and S6K (
Figure 2), as well as reducing levels of Vimentin and Gab1—two proteins closely linked to EMT-related and metastasis-associated signaling [
15]. Thus, CDPs effectively target the PI3K/Akt/mTOR signaling pathway, a key regulator of cell survival, proliferation, and growth. This aligns with the observed suppression of p-S6K phosphorylation and the decrease in Vimentin and p-Gab1 expression. Vimentin is an essential marker of EMT, a process crucial for invasiveness and metastasis. Therefore, the downregulation of Vimentin and inhibition of the PI3K/Akt/mTOR pathway are primary molecular mechanisms responsible for the reduced migration and invasiveness seen in vitro in MDA-MB-231 cells.
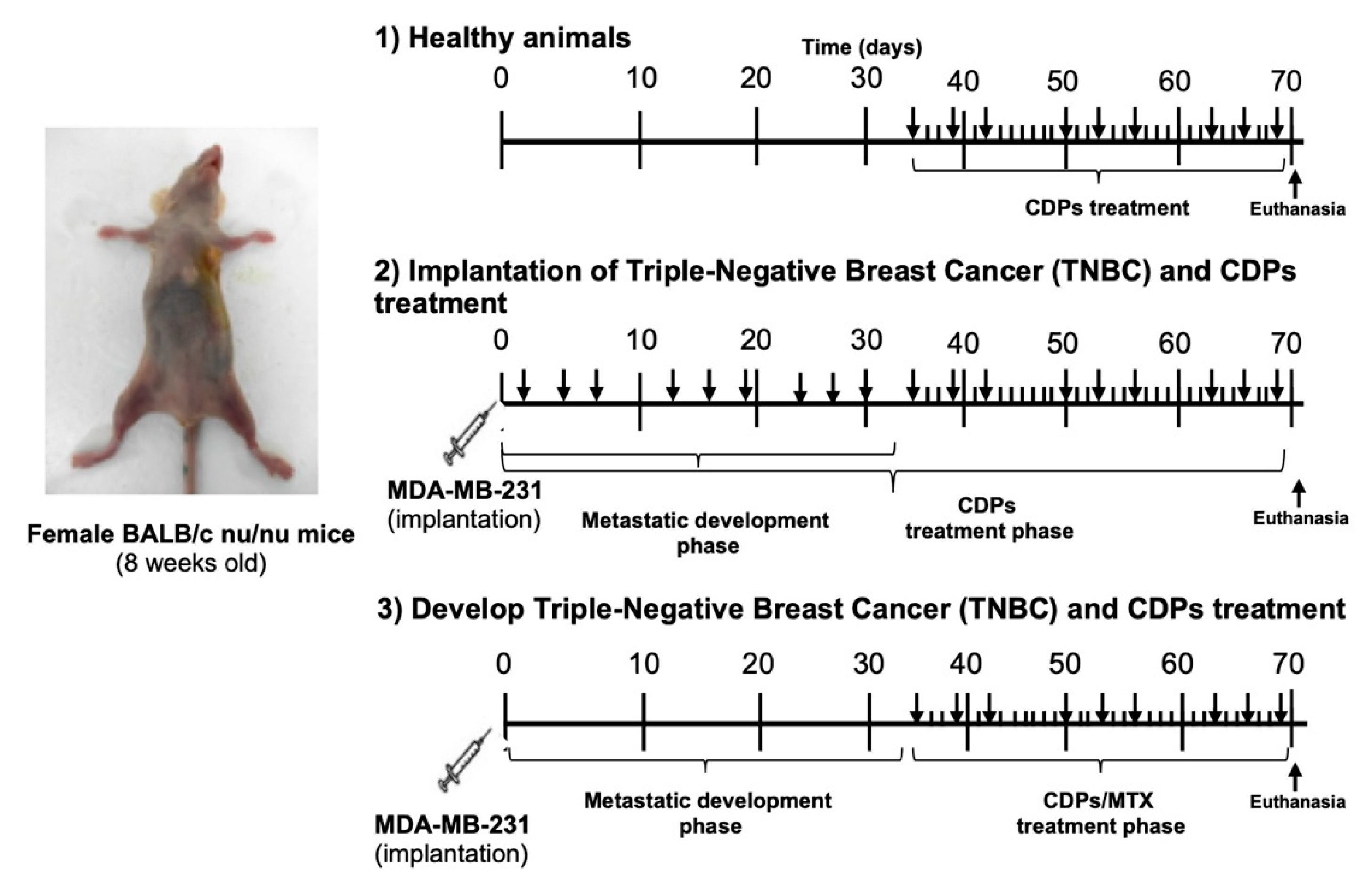
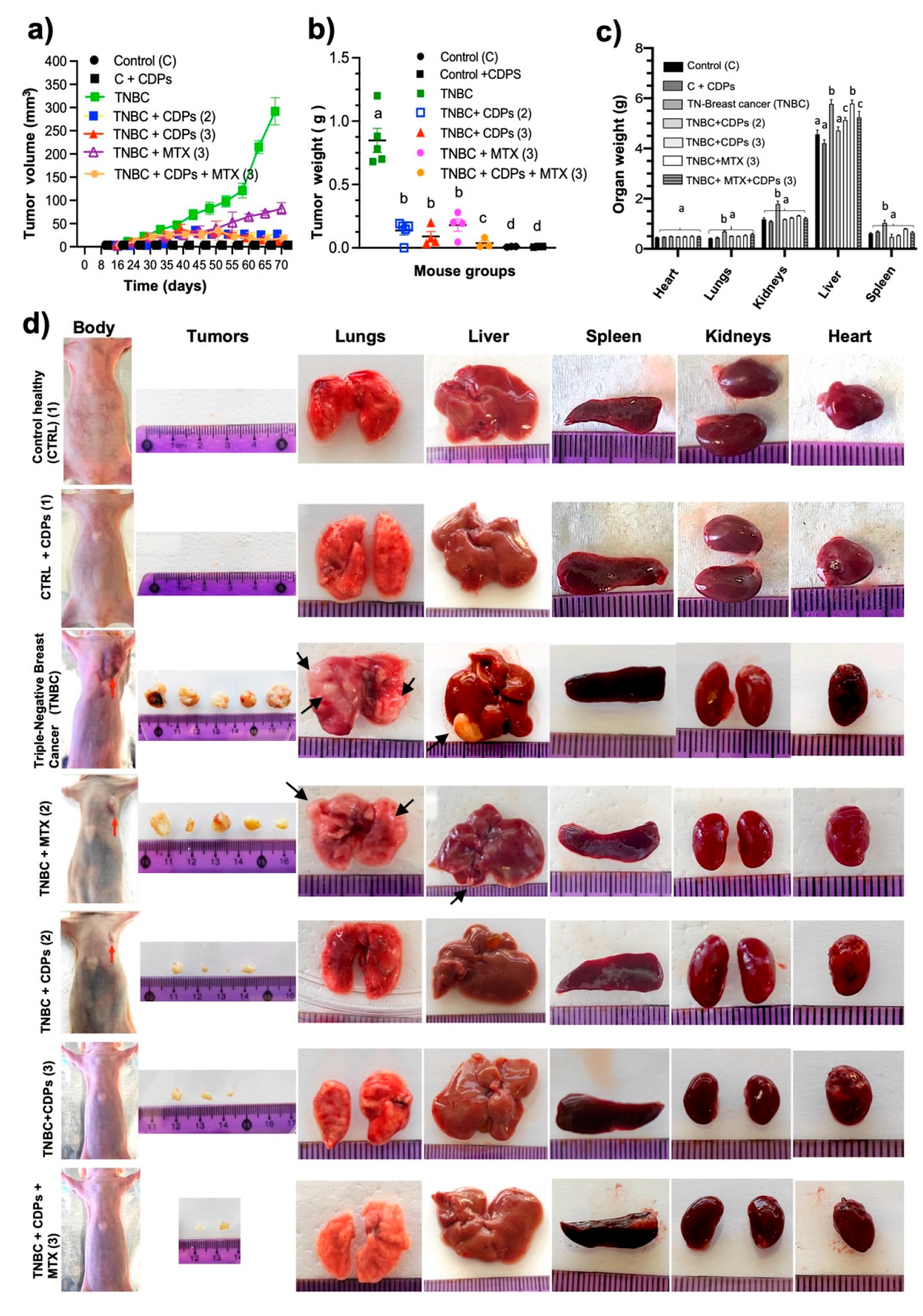
In the orthotopic TNBC xenograft model, implanting MDA-MB-231 cells directly into the mammary gland of animals creates a tumor microenvironment that exhibits key features of aggressive breast cancer progression. Treatment with CDPs significantly reduced tumor volume and weight, especially when given after the tumor was established, and a large portion of treated animals showed no detectable tumors at the end of the experiment (
Figure 4). Besides suppressing primary tumor growth, CDPs treatment also reduced tumor-related systemic changes. Untreated TNBC-bearing mice had increased weights of the lungs, kidneys, liver, and spleen, along with histological changes such as metastatic foci indicating organ damage caused by the tumor (
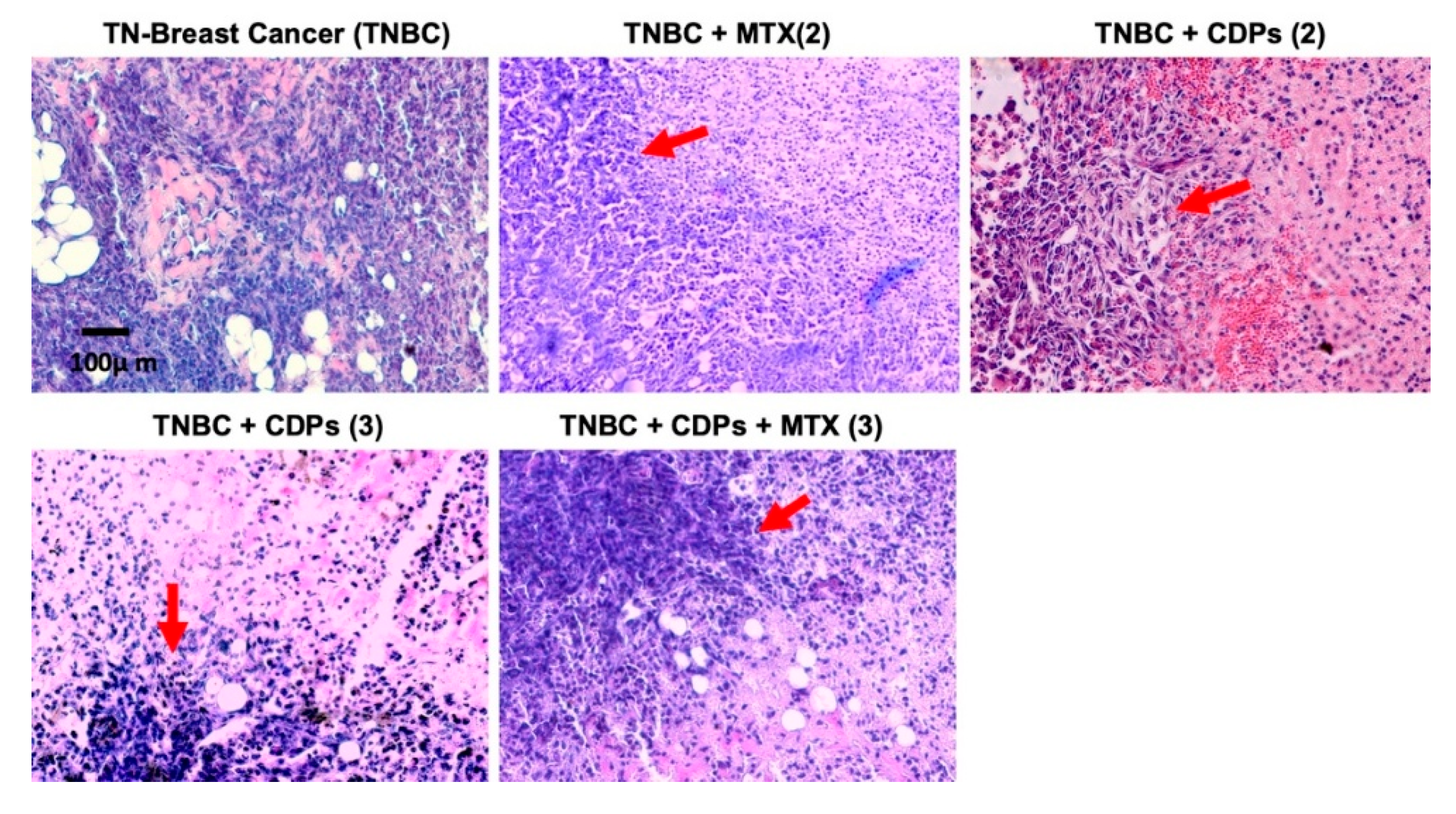
Figure 4). Conversely, groups treated with CDPs and the combination of CDPs plus MTX showed normalization of organ weights and no visible metastatic foci. Histopathological analysis of primary tumors indicated that untreated TNBC displayed extensive hyperchromasia. In contrast, CDP-treated tumors exhibited less hyperchromasia, with areas of cell aggregation observed (
Figure 6). These findings suggest that in tumors from animals treated with CDPs and MTX, cell proliferation is inhibited, as reflected by the size and weight of the tumors dissected from each group (
Figure 4).
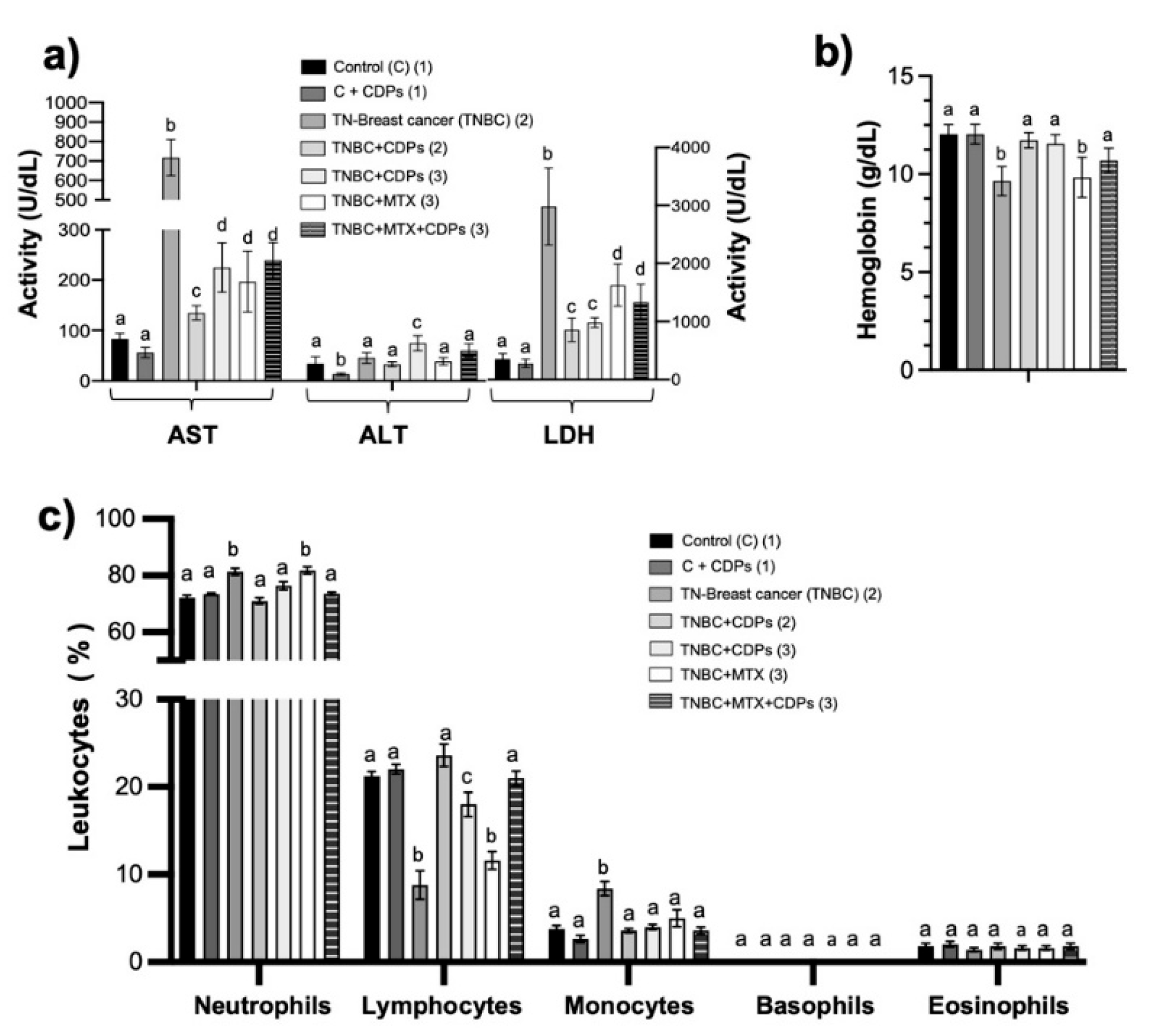
Metastatic progression in breast cancer patients is often associated with systemic alterations, including elevated levels of serum ALT, AST, gamma-glutamyl transferase (GGT), alkaline phosphatase (ALP), and LDH [
16]. In our TNBC model, untreated mice showed significant increases in AST and LDH (
Figure 5). Treatment with CDPs normalized these enzyme levels and reduced organ weight increases. Changes in leukocyte profiles, especially a higher neutrophil-to-lymphocyte ratio (NLR), are known markers of poor prognosis in breast cancer [
17]. In our study, TNBC mice showed the highest NLR, which was reduced in the CDP-treated groups, nearing control levels (
Figure 5c), indicating decreased tumor aggressiveness. Anemia, another common cancer complication, was also improved with CDP treatment. These findings suggest that CDPs alleviate tumor-related systemic and liver dysfunction, supporting a decrease in tumor aggressiveness and metastatic potential.
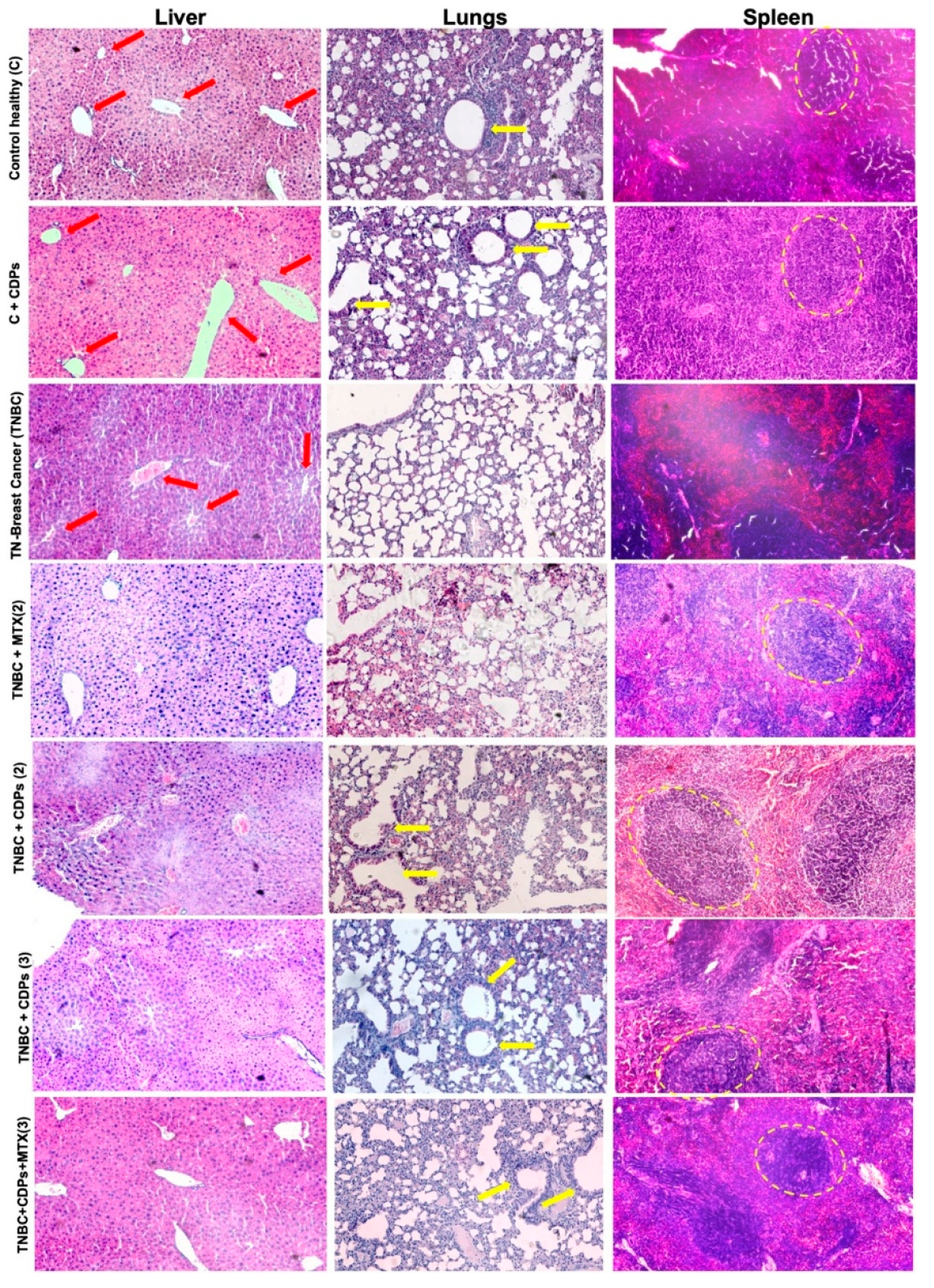
It is important to highlight the safety profile and systemic benefits. CDPs treatment did not alter the activity of liver enzymes ALT, AST, and LDH in healthy control mice (C+CDPs). Conversely, CDPs reversed the significant increase in AST and LDH activity observed in the TNBC group. These results, along with the restoration of histological structures in the lung and spleen, indicate that CDPs not only target the tumor locally but also help normalize tumor-related systemic changes. Histological analysis showed a clear reduction in vascular structures in the liver of the TNBC group. In contrast, structural damage was observed in the lung and spleen tissues, which was significantly lessened after CDPs treatment (
Figure 8). These findings further suggest that, in our model, metastasis of the MDA-MB-231 line occurred in the animals following implantation into target organs such as the liver, lungs, and spleen. The ability of CDPs to restore normal tissue architecture supports their role in reducing tumor-related organ dysfunction and lowering metastatic risk.
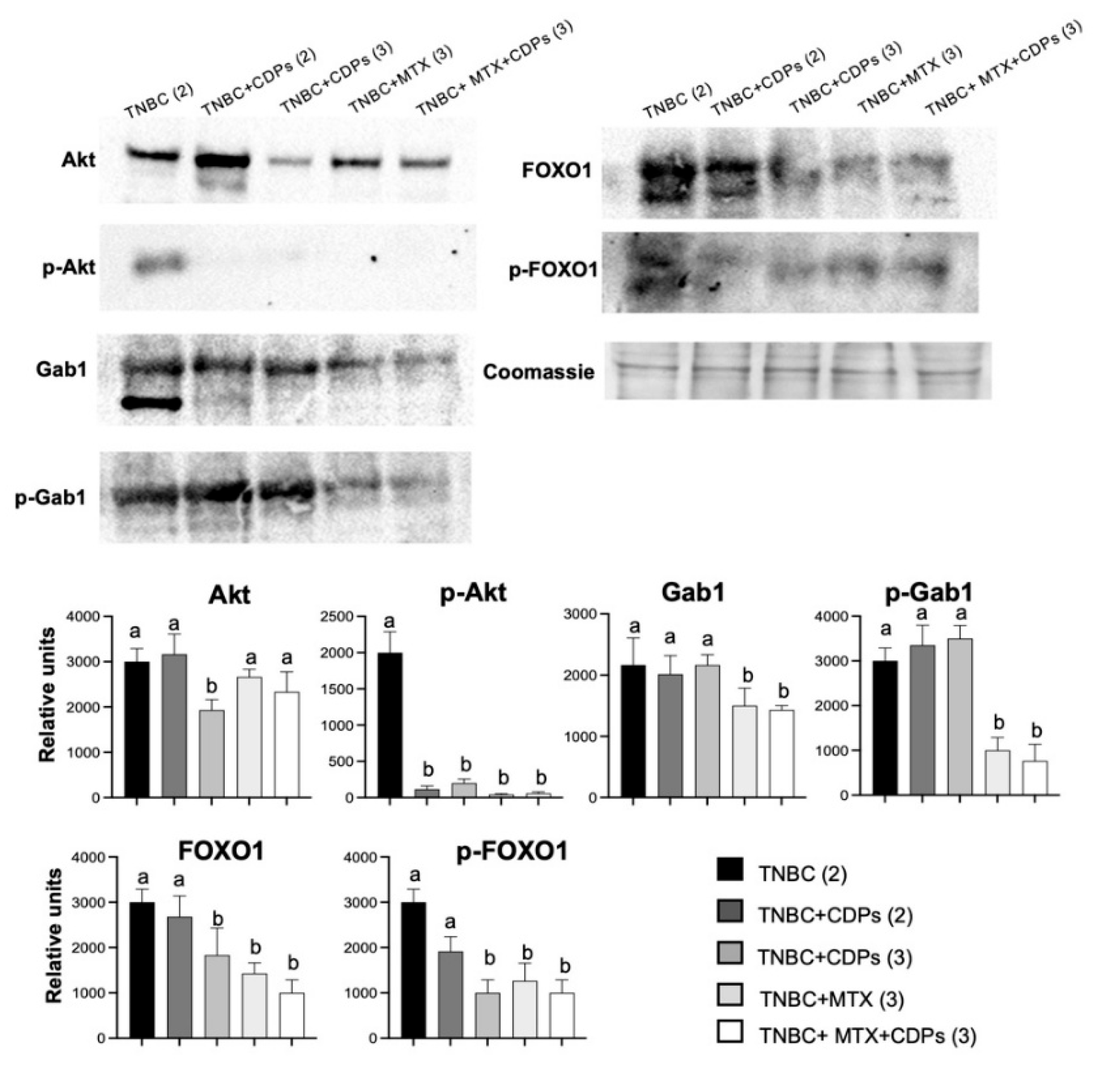
At the molecular level, protein and gene expression analyses revealed coordinated modulation of signaling pathways associated with metastasis, immune evasion, and DNA damage response. To further investigate tumor-associated signaling in TNBC, we evaluate metastasis in the tumor of the xenografted TNBC model. Molecular analyses also showed reduced p-Akt levels in tumors from CDPs-treated animals, along with decreased total Akt expression (
Figure 7), indicating inhibition of the PI3K/Akt/mTOR signaling pathway. FOXO1, a forkhead transcription factor that regulates apoptosis, autophagy, and cell cycle arrest, is negatively regulated by Akt-mediated phosphorylation [
18]. In our study, CDP-treated animals showed decreased FOXO1 phosphorylation. Although total FOXO1 protein levels were reduced, the decrease in FOXO1 phosphorylation aligns with enhanced FOXO1 activity, enabling its nuclear functions and the activation of growth-suppressive and stress-response pathways [
19]. This activation aligns with the induction of apoptotic pathways. These findings match our previous transcriptomic data in HeLa cells, showing CDPs-mediated upregulation of FOXO1-related genes such as GADD45A and SGK1 [
9].
Gab1, an adaptor protein that integrates signals for proliferation, angiogenesis, and invasion [
20], was overexpressed and phosphorylated in TNBC tumors but was downregulated by CDPs, especially when combined with MTX (
Figure 7). This suggests that inhibiting Gab1 is a key mechanism behind the suppression of invasion- and metastasis-related processes by the effect of CDPs. Protein analysis in the xenografted tumors confirmed the inhibition of the Akt pathway. Consistent with the in vitro results, the expression of phosphorylated Akt protein was reduced in the CDPs-treated groups, as Gab1, p-Gab1, FOXO1, and its phosphorylated form p-FOXO1 were markedly decreased in tumors from mice treated with CDPs, MTX, or the combination of CDPs+MTX.
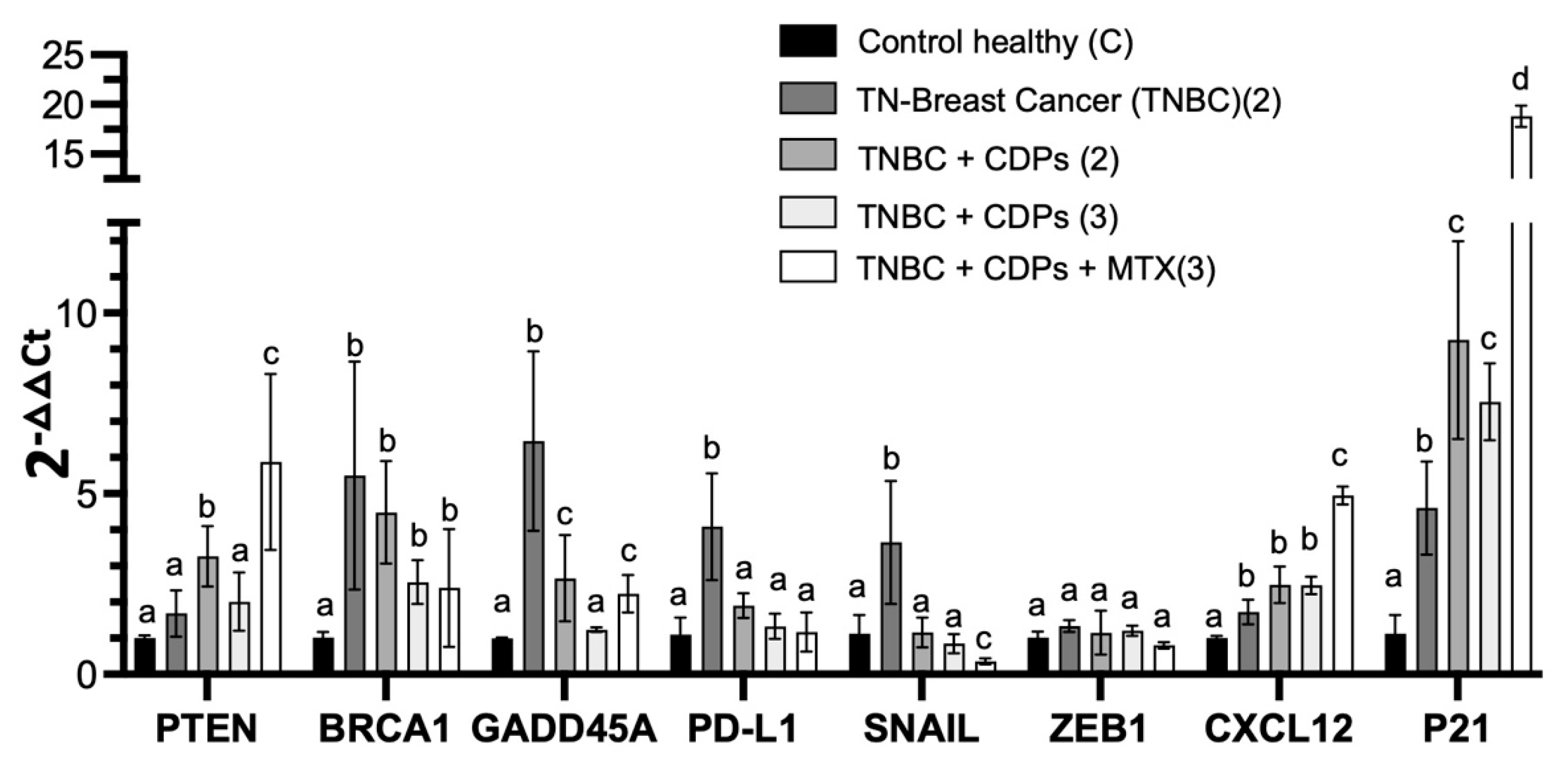
The study of gene expression in the liver provided essential data on tumor-associated systemic alterations modulated by the CDPs. To further investigate the metastasis-related signaling pathways targeted by the CDPs, we examined the hepatic expression of related gene markers linked to several pathways involved in tumor progression, immune regulation, and cellular plasticity (
Figure 9). RT-qPCR analysis revealed that CDPs treatment significantly reversed the overexpression of key genes seen in the TNBC group, including SNAIL and ZEB1, which are associated with promoting EMT, invasiveness, metastasis, and drug resistance. Among immune checkpoint components, such as PD-L1, which plays a role in immune evasion, overexpression was observed in the TNBC group and returned to normal levels following CDPs treatment, approaching expression levels seen in healthy controls. Regarding genes involved in DNA damage/repair and the cell cycle—such as BRCA1 and GADD45A, which showed a marked increase in the TNBC group consistent with tumor-induced systemic stress and genomic instability—these were normalized by CDPs treatment. Conversely, genes like PTEN, CXCL12, and CDKN1A (p21) were overexpressed in the livers of animals treated with both CDPs and MTX compared to untreated healthy mice, indicating activation of compensatory tumor-suppressive and cell cycle–regulatory mechanisms linked to EMT, invasiveness, metastasis, and drug resistance, reflecting an adaptive hepatic response to reinforce immune regulation, growth suppression, and homeostasis in the presence of tumor burden. Taken together, these findings suggest that bacterial CDPs in the TNBC model modulate tumor-associated systemic gene expression programs related to DNA damage responses, immune regulation, and cellular stress, thereby influencing molecular mechanisms associated with tumor aggressiveness and metastatic progression. This modulation targets both tumor cell–intrinsic signaling pathways and tumor-associated systemic responses. The inhibition of Akt/mTOR/S6K signaling, suppression of EMT markers, modulation of immune-related genes, and favorable safety profile collectively position CDPs as promising therapeutic agents. Moreover, the enhanced efficacy observed when CDPs are combined with MTX supports the potential of combination strategies to improve treatment outcomes in aggressive breast cancer subtypes.
In summary, in the TNBC mouse model, CDPs administration significantly reversed primary tumor growth in the mammary glands, with greater effectiveness when com-bined with MTX. Beyond local tumor control, CDPs treatment reduced tumor-associated systemic changes in distant organs, like liver, which are commonly involved in metastatic progression. The treatment with CDPs lowered metastatic foci in organs such as the liver. At the molecular level, CDPs modulated the expression of genes linked to tumor ag-gressiveness, immune regulation, and stress responses, such as SNAIL, GADD45A, and PD-L1, while increasing the expression of growth-suppressing and homeostatic regulators including PTEN, CXCL12, and CDKN1A (p21). The data provide new insights into the molecular mechanisms behind the anti-metastatic and anti-invasive effects of CDPs, showing that these effects are connected to genes involved in EMT, invasiveness, and metastasis. This indicates that the signal transduction mechanism involves inhibition of phosphorylation in the Akt/mTOR/S6K pathway and of metastasis markers like Vimentin, Gab1, and FOXO1. The findings suggest that combining CDPs with MTX amplifies the anti-metastatic effect in a TNBC xenograft model using the MDA-MB-231 line, indicating that bacterial CDPs could be a promising therapeutic agent. In conclusion, bacterial cyclic peptides represent a promising multi-target therapy for TNBC, showing greater efficacy than MTX in vivo. Their mechanism involves blocking the PI3K/Akt/mTOR/FOXO1 pathway and suppressing EMT, resulting in strong anti-tumor and anti-metastatic effects with a good safety profile.