1. Introduction
Preeclampsia (PE), in its form as hypertension combined with organ deficiency appearing during the second half of gestation, influences about 5-8 % of pregnancies. This serious disease is responsible for more than 70.000 maternal and cc. 50.000 fetal deaths annually, worldwide [
1,
2]. Despite intensive research, its treatment is practically limited to reducing high blood pressure and inducing labor [
2]. In recent decades, there has been some progress in the management by the introduction of screening tests with Acetylsalicylic acid prophylaxis and improvement in fetal condition assessment. However, recognizing preeclampsia is essentially two distinct clinical conditions may open up broad possibilities in understanding the disease and, in many cases, its treatment and even prevention.
Pioneers in breaking down the homogeneous disease concept of PE were Easterling et al. [
3] in 1990 and Xiong et al. [
4] in 2000 when, contrary to the orthodox concept of high vascular resistance / low blood volume and fetal growth restriction, they reported low resistance with high blood volume and high fetal birth weight in preeclamptic pregnancies. An important step was taken by Belfort et al. [
5], who found not only reduced but also increased cerebral blood flow in preeclamptic females in 1999. Heterogenetic origins of PE were suspected by Ness and Roberts [
6] in 1996, however, the two groups of preeclamptic patients, differing in clinical, hemodynamical, and laboratory parameters were first reported in 2003 [
7].
Evaluation regarding 44.220 relevant data of Medical Birth Registry of Norway supported further the heterogenic origin of preeclampsia revealing that prevalence not only low but also high fetal birth weights were over the average when mothers had been admitted with preeclampsia diagnosis [
8].
Today, the two-type-PE concept is fully accepted. The question, whether common reason(s) (e.g., syntitiothrophoblast stress) causes the symptoms in distinct ways, or totally independent facts lay behind the different types of PE, remains to be answered. Distinction of the subtypes is essential in many terms, especially for treatment [
9,
10]. Based on the clinical picture (fetal condition, high body mass index with high weight gain, and the appearance of edema as the first sign), this is possible (see later!). A quantitatively expressively difference can be determined through hemodynamic examinations [
7,
11,
12] and now utilizing numerous laboratory tests [13- 15].
In the case of both the placental and the much more common maternal type [
16], new aspects worth considering have emerged, which may shed new light in the realm of PE.
2. Placental Preeclampsia
2.1. Denomination
This is the “classic” type, which is also referred to in the literature as hypovolemic, low CO (cardiac output), preterm, severe, with fetal growth restriction, type I, and, most frequently early onset PE. This last term was suggested due to the fact the outcome of preeclamptic pregnancies is much worse when clinical symptoms appeare prior to the 34 weeks than those later [
17]. This very important and groundbreaking definition unfortunately carries the potential for misunderstanding and sometimes misclassification since the difference between subgroups is not due to the gestational week when clinical symptoms appear and the notice them could be later then their manifestations. Hypovolemic or low CO expressions refer to an important pathologic feature, while the placental denomination seems the best since it refers to the origin of this type of PE.
2.2. Sequence of Symptoms
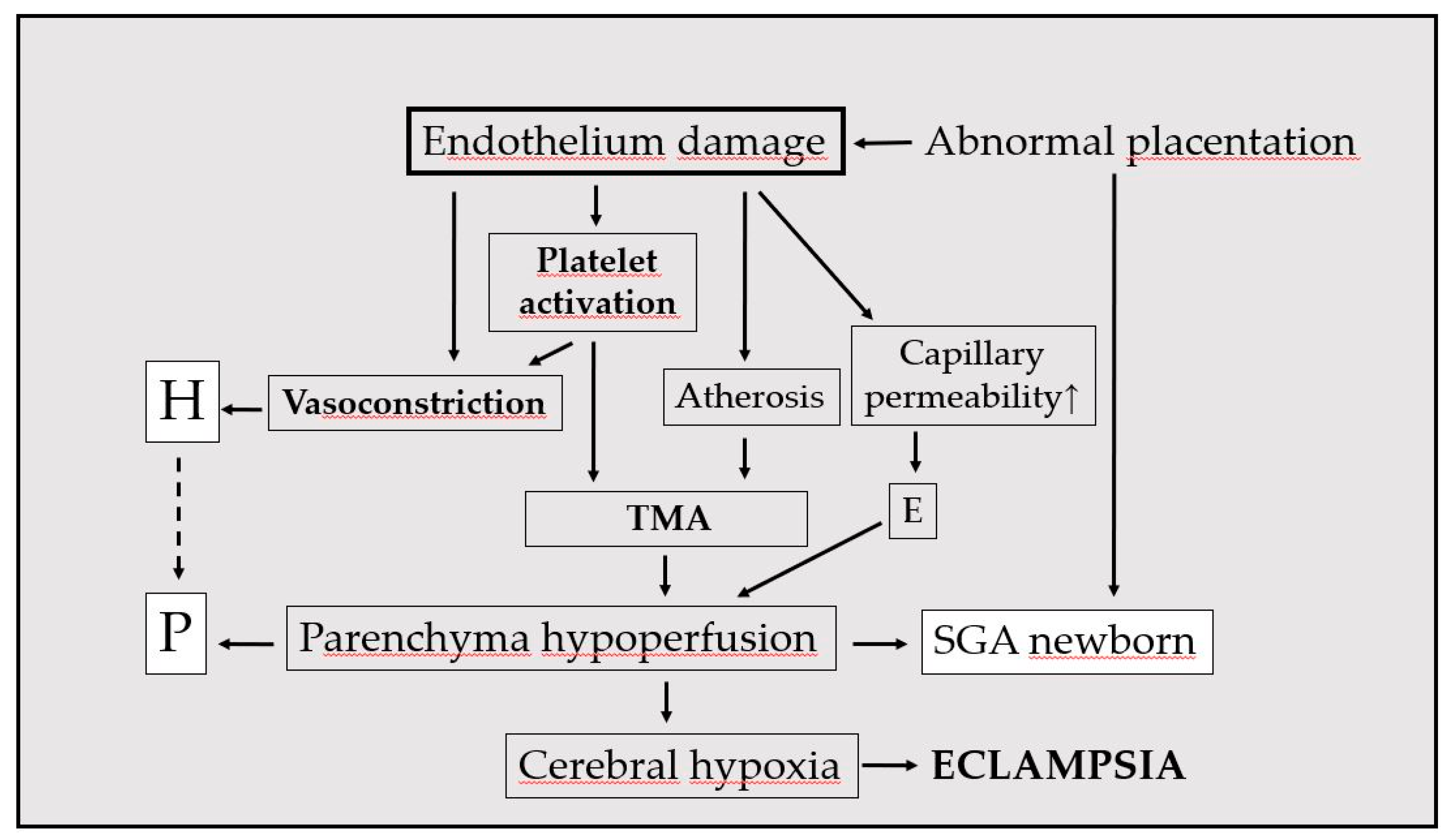
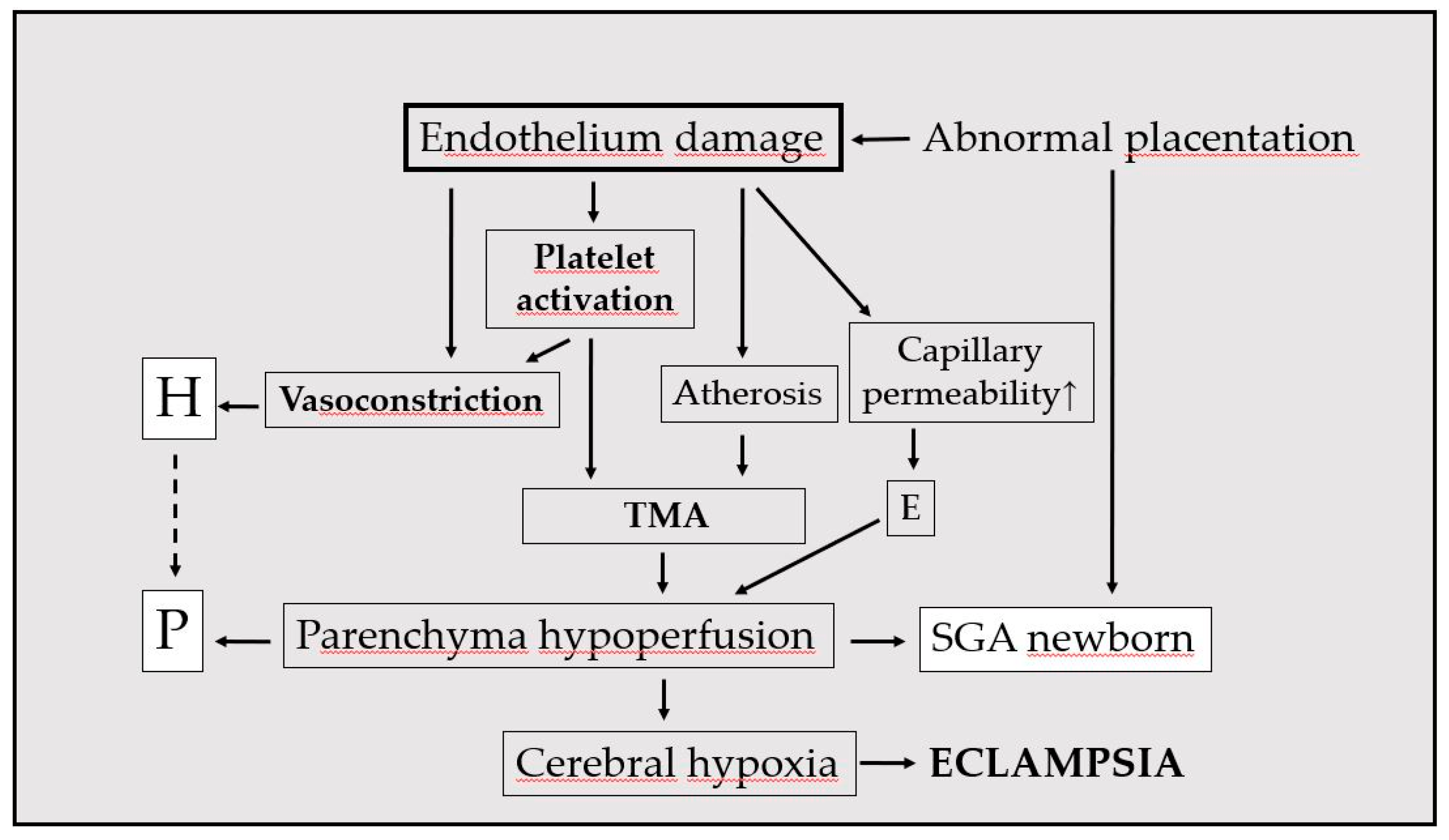
The first sign of this type of PE is an elevation in blood pressure; reaching 140 / 90 mm Hg is defined as (1) hypertension, which is significant according to the diastolic pressure as well and increases on a daily basis. Organ dysfunction may appear after several days, typically first characterized as (2) proteinuria, since kidneys are sensitive to hypoxia and detecting urinary protein is simple. Next, the indices of liver- and further kidney involvement are observed; however, lowering of platelet count can precede all (laboratory) changes [
18]. Capillary permeability accessibly increases during the final stage of placental PE and generalized, visible (3) edema appears, resulting in an acute life-threatening condition.
2.3. Hypovolemic Hypertension
This type of later preeclampsia manifests itself very early in pregnancy, and its immunological origin is now considered accepted [19, 20]. Immunological imbalance between the maternal immune system and the semi-allograft embryo is also reflected in elevated levels of anti-angiogenic agents, such as soluble fms-like tyrosine kinase 1 (sFlt-1), and soluble endoglin which diminish the functions of placental growth factor (PlGF) and transforming growth factor β during the first phase of this type of PE [
21,
22].
Agents from the under perfused placenta (e. g. antiangiogenic factors, free oxygen radicals, activated tumor necrosis factor, fetal cell debris and microparticule contents) are considered to cause the damage of the protective endothelial glycocalix layer which leads to a generalized injury of endothelial cells. Markers of endothelial damage (e.g., circulating endothelial cells, soluble vascular cell adhesion molecule 1, E-selectin and endocan) show elevated levels in placental PE [
23,
24].
Damaged endothelial cells release the lower amount of vasorelexant agents (e.g., nitrogen monoxide (NO), prostacyclin and endothelium-derived hyperpolarizing factor) and secrete increased amounts of vasoconstrictors, such as endothelin-1(ET-1) [25-27]. Additionally, activated platelets release another effective vasoconstrictor, the thromboxane A2. So, the reason of increased blood pressure in placental PE is clearly due to vasoconstriction, secondary to endothelial injury, caused by placental agents, however, hypovolemia-induced vasopressin effect may also be present [
28].
2.4. Organ Dysfunction
Previously, vasoconstriction was considered the cause of decreased tissue blood supply. The necessary change in perspective was based on the understanding of changes in microcirculation. The diameter of flexible red blood cells is 7-8 μm, while capillary diameter is somewhere narrower. Even normal red blood cells cannot pass through a tube if its diameter is less than 2.9 μm [
29]. Once endothelial atherosis with fibrin deposition and platelet adhesions critically narrow capillary diameters, blood cells can become trapped. In our case, red blood cell deformability is reduced and their aggregability increases with damage and slowing flow [
30,
31], promoting the development of thrombotic microangiopathy (TMA) [
32,
33].
Platelets, through producing immune-modulator molecules and vasoactive agents, basically involved in blood coagulation, and even participating in infection defense, play a pivotal role in the pathogenesis of placental PE [
34,
35].
Capillary plugs obviously reduce organ perfusion and increase circulatory resistance. Some entrapped red blood cells break down augmenting lactate dehydrogenase levels; cell fragments can be detected in the bloodstream as signs of peripheral mechanical hemolysis [
36]. Adenosine-diphosphate, also released from damaged red blood cells, further enhances platelet activation, promoting the formation of a vicious cycle. Termination of pregnancy should not be delayed if oligo-anuria or signs of central nervous system involvement develops (
Figure 1).
2.5. Hemodynamics and Fetal Weight
Normally, the average 5 liters of blood increases to nearly 7 liters by the 3rd trimester; in which the corresponding CO is 6.4 – 6.8 l/min [
37,
38]. The augmentation regarding blood volume, which overtakes vasorelaxation by the 12
th week, is regulated basically by placental renin through the angiotensin - aldosterone pathway, which is also facilitated by the increased release of vasopressin in early pregnancy; blood pressure does not change significantly. The blood volume in placental PE is low. In the absence of vasodilation, pregnancy hemodilution is also absent. In addition to vasoconstriction, CO lowered even to 3.5 l/min; the deficit can be detected even before the appearance of clinical symptoms [
9,
10,
39].
Special aspect of placental impairment in this type of PE the weak trophoblast invasion, resulting in an insufficient spiral artery remodeling. These vessels remain narrow and contractable [
40]. The blood supply provided by the small, under perfused placenta is insufficient for normal fetal development; fetal somatic restriction is a sine qua non of placental PE. Critical deterioration of the fetal condition in placental PE usually precedes the development of a preterminal maternal condition
2.6. Prognosis and Prophylaxis
The placenta and the vessels are the most affected tissues in the latent phase of placental PE. Therefore, the markers of placental function [(placental growth factor - PlGF), sFlt-1, pregnancy-associated plasma protein-A, and placental protein 13 and indices of endothelial function (asymmetric dimethylarginine -ADMA), ET-1, and vascular cell adhesion molecule 1] for PE prediction all have been intensively investigated. In clinical practice, determination of sFlt-1 / PlGF ratio is frequently used [
41].
On the other hand, the assessment regarding Doppler velocimetry of flow characteristics of uterine artery (UtA) has been considered a suitable non-invasive method for evaluating placental blood supply. Examinations for PE screening are standardized for the 11 to 13 + 6-week [
42]. Abnormal UtA flow (high pulsatility and resistance index, appearance of “notching”) refers to incomplete remodeling of spiral arteries which contributes placental insufficiency characteristically aligned to placental PE [
43].
This type of PE is most threatening to those who conceive soon after initiating sexual activity, when the attenuation of the maternal immune system against future foreign proteins is not yet sufficient [
44]. Based on this, in young nulliparous women, the use of ultrasound and laboratory tests developed for the prediction of PE is entirely justified. In positive cases, high-dose acetylsalicylic acid (150 mg/day) is indicated from early pregnancy onwards, which delays and alleviates the clinical manifestation of placental PE, primarily by inhibiting platelet activation [
45].
2.7. Management
According to the simplified formula of the fundamental law regarding central hemodynamics, tissue blood supply is determined by the ratio of (blood) pressure to (circulatory) resistance (perfusion = pressure / resistance), i.e. increasing pressure or decreasing resistance improves tissue blood supply. In this sense, the initial increase in blood pressure, in the form of compensation, helps maintain the decreased perfusion due to the increasing number of capillary blocks [
46]. In our case, the question is whether the use of a vasodilator, which simultaneously reduces vasoconstriction and blood pressure, increases tissue blood supply, improves the condition of the mother or the fetus. Since there is no such data, delaying the deterioration of microcirculation (acetylsalicylic acid) has a beneficial effect, suggests blocking capillaries is more important than vasoconstriction in shaping peripheral resistance. This also implies, in terms of perfusion, vasoconstriction, by elevating blood pressure, has a more beneficial than detrimental effect. Therefore, it is not surprising the decrease of blood pressure reduces the fetal weight [
47]. A rapid and severe drop in blood pressure can lead to a critical condition for the fetus, especially since antihypertensive drugs cross through the placenta [
48]. Maternal and/or fetal tachycardia occurring with antihypertensive treatment may be a circulatory compensation to maintain blood supply rather than a side effect of the given drug.
Despite all the above, maintaining blood pressure around 140-160 / 90 mmHg is justified according to all recommendations, primarily in order to avoid sudden, critically high values. Since hypertension is caused by vasoconstriction, the first recommended drug administered is a vasodilator (e.g., nifedipine).
Attempts to improve the pathological processes in cases of fully developed disease (such as heparin, acetylsalicylic acid, fish oil, vitamin E and sedatives) have not yielded any significant results. Magnesium salt and calcium dobesilate (CAD) can be recommended as adjuvants [
49]. CAD can have a beneficial affect on microcirculation by promoting both basal and reactive NO synthesis, improving declined erythrocyte deformability, reduces platelet aggregation and vascular permeability and in gestational hypertension CAD decreases blood pressure [
50,
51]. Other promising drugs for the treatment include pravastatin, metformin, and esomeprazole; all of which reduce the antiangiogenic effect of soluble fms-like tyrosine kinase-1 (sFlt-1), which plays a key role in disturbing placental development in this type of PE [
52]. Prior to week 34, augmenting fetal pulmonary maturation by corticosteroid is indicated [
53].
In the case of young nulliparous women, especially with a positive prognosis, regular home blood pressure monitoring is also justified, and hospital admission is recommended once elevated values are detected. Treatment of placental PE is only acceptable in an institution aptly facilitated for managing premature newborns.
3. Maternal Preeclampsia
3.1. Denomination
High CO, hypervolemic, term, type II, late onset, mild, and maternal PE actually refer to the same pathology. The latter name is perhaps the best because it sharply distinguishes this condition from the other, placental type.
3.2. Order of Symptoms
According to many decades of clinical experience, the first sign of “overfilling” is the (1) leg edema, which can develop into generalized form within a few days. Fluid retention can even raise blood pressure, (2) hypertension develops. Venous congestion with tissue edema, due to further water retention, can lead to symptoms of organ damage, such as (3) proteinuria, which is usually not severe.
3.3. Hypervolemic Hypertension
The cause of hypertension which occurs alongside hypervolemia-induced edema is also the blood volume exceeding given vascular capacity, which persists despite vasodilation in this form of PE [
9,
54].
It is known that overloaded circulation itself affects the function of endothelial cells, which in our case can also contribute to an increase in blood pressure [
55,
56]. This is also indicated by the fact the level of ADMA, which inhibits the formation of the vasodilator NO, is increased not only in early onset but also in the late onset PE when compared to normal controls [
57].
Several data refer to the crucial role of obesity (Body Mass Index - BMI ≥ 30 kg/m
2) in the development of hypertension, secondary to increased water retention, among nonpregnant women. Similarly, obesity is associated also with gestational hypertensive conditions and shows a positive correlation to edema development [
58]. Not only high pre-pregnancy weight but increased gestational weight gain is associated with the development of this type of PE [
59,
60].
Obesity is associated with elevated plasminogen activator inhibitor 1, and profibrinogen levels, of which, due to increasing blood viscosity, contribute to elevate blood pressure [
61]. Insulin resistance, a distinct characteristic of obesity, is known to enhance renal sodium- and water retention [
62]. Moreover, angiotensinogen, essential for water retention, is also produced by adipocytes [
63]
The levels of Na / K-ATPase (Na+ pump) inhibitors, including the digitalis-like marinobufagenin, are increased by salt intake [
64]. Therefore, in addition to obesity with sodium retention, the highly potent vasoconstrictor marinobufagenin, also produced by the placenta, is likely to play a role in the pathogenesis of maternal PE, suggesting a specific approach to the treatment of hypertension with hypervolemia [
65]
3.4. Organ Dysfunction
Hypervolemia, which ensures favorable tissue blood supply, can cause rapid circulatory deterioration (stasis) when venous outflow obstruction develops [
54,
66]. Increased renal venous pressure enhances aldosterone secretion, which further increases blood volume and blood pressure [
67]. Critical tissue edema can lead to ascites, placental abruption, pulmonary edema and eclampsia (
Figure 2).
3.5. Hemodynamics and Fetal Weight
As most names for this PE suggest, in maternal type blood volume is higher than the average for the normal third trimester. In addition to the abundant blood volume (CO - 7.5 l/min) and the increased pressure, fetuses usually have an above-average weight [
4,
50,
51]. The relationship between CO and fetal weight has been demonstrated in both healthy and preeclamptic pregnancies [52-54].
3.6. Prognosis and Prophylaxis
As most names for this PE suggest, in maternal type blood volume is higher than the average for the normal third trimester. In addition to the abundant blood volume (CO > 7.5 l/min) and the increased pressure, fetuses usually have an above-average
weight [
4,
8,
68]. The relationship between CO and fetal weight has been demonstrated in both healthy and preeclamptic pregnancies [
69,
71].
3.7. Management
The literature recommends using α- and/or β-blockers as the first drugs recommended in lowering blood pressure [
3,
76]. It is logical to use a diuretic to treat hypertension which occurs with an overfilled circulation. In PE, diuretics are not generally recommended since they can lead to fatal consequences in the event of hypovolemia. Previous studies have therefore been conducted with diuretics either with prophylactic purposes or administered in the postpartum period of a preeclamptic pregnancy. Collins et al. [
77] reported the results of nine studies in 1985. To prevent PE, on the whole, nearly 7.000 pregnant women received continuous administration of thiazid diuretics. This review showed stunningly diverse results and failed to elicit reliable evidence regarding the beneficial effects of diuretics on PE prevention. In another examination, twenty-one pregnant women, with elevated CO (considered as pre-hypertensive condition), received a daily administration of 20 mg furosemide, initiated between the thirteen and thirty-second weeks. An improvement of hyperdynamic circulation was achieved through the significant decrease of stroke volume and CO, however, blood pressure did not lower in control examination three weeks later [
78]. In a randomized placebo-controlled study, preeclamptic patients received 20 mg of furosemide during the postnatal five days. This medication promoted patient recovery, decreased blood pressure and, this way, antihypertensive demand [
79]. Ascarelly et al. [
80] randomly administrated 40 mg of furosemide or placebo during postpartum period in two hundred sixty-four patients with severe, mild or superimposed PE. The five days’ medication decreased blood pressure and less antihypertensive medication was required during hospitalization, and at discharge in severe preeclamptic patients than when compared with the other groups. These studies reported overall favorable results. Importantly, there were no fetal side effects, and neither diuretic treatment for gestational hypertension had a negative effect upon perinatal outcome [
81].
In consideration of increased gestational weight gain, edema, hypertension, and even proteinuria, which all can be explained by water retention beyond the given vascular capacity, the use of diuretics seems to be fully justified in hypervolemic PE. A study on direct diuretic treatment in PE was published in 2017 [
82]. In cases with high cardiac output, examined by impedance cardiography, oral furosemide 40 mg resulted in rapid and parallel reductions in CO and blood pressure. The results confirm the causal role of increased blood volume in the development of hypertension and strongly suggest the need for diuretic treatment in cases of PE associated with high blood volume.
The effectiveness of blood pressure reduction can also be improved by other factors (such as low-salt and fat, calcium-rich diet, physical activity and relaxation techniques) during pregnancy [
83]. In addition, mild diuretic treatment (e.g. phytotherapy) can be part of the practice now, without risk, in the case of edema appearing in the third trimester regarding an obese pregnant female.
Pregnancy termination in 34 – 37
th gestational weeks of hypervolemic PE is associated with better maternal yet with less favorable neonatal outcome, compared to expectant management [
84].
4. Conclusions
To summarize our findings we can conclude the following statements:
Separation of (potential) preeclamptic patients in due time as placental or maternal type is essential since management is also different [
9].
Edema, especially in its generalized form is a frequent, attention-grabbing sign of imminent maternal PE; obesity means a significant risk factor [
58].
In the setting of developed placental PE, frequent and accurate assessment of fetal status is an essential part of management [
85].
It worth keeping in mind, decrease of blood pressure may influence fetal condition too, even if appropriate antihypertensive drug is used [
48].
After delivery, it is important to identify any underlying diseases, which can also help prevent diseases expected in later life, primarily cardiovascular diseases [
86].
The new approach necessitates a modification of the classification of gestational hypertension, which is already being attempted [
87].
Recognizing PE is clinically two fundamentally different conditions (they occasionally mix) explains the previous contradictory research data, greatly helps to understand the pathological events also in relation to individual cases and may create an opportunity to manage effectively maternal PE. This requires additional laboratory and clinical, bedside studies.
Author Contributions
PT conceptualization, manuscript writing, BF Data collection, JB manuscript editing and writing. All authors have read and agreed to the published version of the manuscript.
Funding
the current study received no funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is available upon request from the corresponding author.
Acknowledgments
We are thankful for the medical staff (colleagues, nurses, midwifes) of the University of pecs, Department of Obstetrics and Gynecology for their long-term support.
Conflicts of Interest
The authors declare no conflicts of interest
Abbreviations
The following abbreviations are used in this manuscript:
| PE |
Preecclampsia |
| CO |
Cardiac output |
| PlGF |
Placental growth factor |
| NO |
Nitrogen monoxide |
| TMA |
Thrombotic microangiopathy |
| ADMA |
asymmetric dimethylarginine |
| UtA |
Uterine artery |
| CAD |
Calcium dobesilate |
| BMI |
Body Mass Index |
References
- Poon, L.C.; Shenna, A.; Hyett, J.A.; et al. The International Federation of Gynecology and Obstetrics (FIGO) initiative on pre-eclampsia. Int. J. Gynaecol. Obstet. 2019, 145, 1–33. [Google Scholar] [CrossRef]
- A Magee, L.; Brown, M.A.; Hall, D.R.; Gupte, S.; Hennessy, A.; Karumanchi, S.A.; Kenny, L.C.; McCarthy, F.; Myers, J.; Poon, L.C.; et al. The 2021 International Society for the Study of Hypertension in Pregnancy classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 2022, 27, 148–169. [Google Scholar] [CrossRef] [PubMed]
- Easterling, T.R.; Bededetti, T.J.; Schmuker, B.S.; Millard, S.P. Maternal hemodynamics in normal and preeclamptic pregnancies: a longitudinal study. Obstet Gynecol. 1990, 76, 1061–1069. [Google Scholar]
- Xiong, X.; Demianczuk, N.N.; Buekens, P.; Saunders, L.D. Association of preeclampsia with high birth weight for age. Am J Obstet Gynecol. 2000, 183, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Belfort, M.A.; Grunewald, C.; Saade, G.R.; Nisell, M.V. Preeclampsia may cause both hyperperfusion and underperfusion of the brain. A cerebral perfusion based model. Acta Obstet Gynecol Scand. 1999, 78, 586–591. [Google Scholar] [CrossRef]
- Ness, R.B.; Roberts, J.M. Heterogeneous causes constituting the single syndrome of preeclampsia: A hypothesis and its implications. Am. J. Obstet. Gynecol. 1996, 175, 1465–1470. [Google Scholar] [CrossRef]
- Tamás, P.; Veszprémi, B.; Szabó, I. Dyscordant fetal weigths are associated with different pregnancy indices in preeclampsia (Hungarian with English summary). Magy Nőorv L. 2003, 66, 211–215. [Google Scholar]
- Vatten, L.J.; Skjaerven, R. Is pre-eclampsia more than one disease? BJOG 2004, 111, 298–302. [Google Scholar] [CrossRef]
- Masini GFoo, L.F.; Tay, J.; et al. Preeclampsia has two phenotypes which require different treatment strategies. Am J Obstet Gynecol. 2021, 226, S1006–S10018. [Google Scholar] [CrossRef]
- Tamás, P.; Kovács, K.; Várnagy, Á.; Farkas, B.; Wami, G.A.; Bódis, J. Preeclampsia subtypes: Clinical aspects regarding pathogenesis, signs, and management with special attention to diuretic administration. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 274, 175–181. [Google Scholar] [CrossRef]
- McLaughlin, K.; Scholten, R.R.; Kingdom, J.C.; et al. Should maternal hemodynamics guide antihypertensive therapy in preeclampsia? Hypertension 2018, 71, 550–556. [Google Scholar] [PubMed]
- Di Pasquo, E.; Ghi, T.; Dall'Asta, A.; et al. Maternal cardiac parameters can help in differentiating the clinical profile of preeclampsia and in predicting progression from mild to severe forms. Am J Obstet Gynecol. 2019, 221, 633.e1–e9. [Google Scholar] [PubMed]
- Teoh, S.S.Y.; Zhao, M.; Wang, Y.; Chen, Q.; Nie, G. Serum HtrA1 is differentially regulated between early-onset and late-onset preeclampsia. Placenta 2015, 36, 990–995. [Google Scholar] [CrossRef]
- Sanhal, C.; Kavcar, M.C.; Yucel, A.; Erkeneklı, K.; Erkaya, S.; Uygur, D. Comparison of plasma fetuin A levels in patients with early-onset pre-eclampsia vs late-onset pre-eclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 200, 108–112. [Google Scholar] [CrossRef]
- Pillay, P.; Vatish, M.; Duarte, R.; Moodley, J.; Mackraj, I. Exosomal microRNA profiling in early and late onset preeclamptic pregnant women reflects pathophysiology. Int. J. Nanomed. 2019, 14, 5637–5657. [Google Scholar] [CrossRef]
- Lisonkova, S.; Joseph, K. Incidence of preeclampsia: risk factors and outcomes associated with early- versus late-onset disease. Am. J. Obstet. Gynecol. 2013, 209, 544.e1–544.e12. [Google Scholar] [CrossRef]
- von Dadelszen, P.; Magee, L.A. Subclassification of preeclampsia. Hypertens Pregn. 2003, 22, 143–148. [Google Scholar]
- Redman, C.W.; Bonnar, J.; Beilin, L. Early platelet consumption in pre-eclampsia. Br Med J. 1978, 25, 467–469. [Google Scholar]
- Than, N.G.; Hahn, S.; Rossi, S.W.; Szekeres-Bartho, J. Editorial: Fetal-Maternal Immune Interactions in Pregnancy. Front. Immunol. 2019, 10, 2729. [Google Scholar] [CrossRef]
- Meggyes, M.; Miko, E.; Lajko, A.; et al. Involvement of the PD-1/PD-L1 co-inhibitory pathway in the pathogenesis of the inflammatory stage of early-onset preeclampsia. Int J Mol Sci. 2019, 20, 583–592. [Google Scholar]
- Maynard, S.; Epstein, F.H.; Karumanchi, S.A. Preeclampsia and Angiogenic Imbalance. Annu. Rev. Med. 2008, 59, 61–78. [Google Scholar] [CrossRef]
- Levine, R.J.; Lam, C.; Qian, C.; Yu, K.F.; Maynard, S.E.; Sachs, B.P.; Sibai, B.M.; Epstein, F.H.; Romero, R.; Thadhani, R.; et al. Soluble Endoglin and Other Circulating Antiangiogenic Factors in Preeclampsia. New Engl. J. Med. 2006, 355, 992–1005. [Google Scholar] [CrossRef]
- Weissgerber, T.L.; Garcia-Valencia, O.; Milic, N.M.; Codsi, E.; Cubro, H.; Nath, M.C.; White, W.M.; Nath, K.A.; Garovic, V.D. Early Onset Preeclampsia Is Associated With Glycocalyx Degradation and Reduced Microvascular Perfusion. J. Am. Hear. Assoc. 2019, 8, e010647. [Google Scholar] [CrossRef]
- Yu, W.; Gao, W.; Rong, D.; Wu, Z.; Khalil, R.A. Molecular determinants of microvascular dysfunction in hypertensive pregnancy and preeclampsia. Microcirculation 2018, 26, e12508. [Google Scholar] [CrossRef]
- Boeldt, D.S.; Bird, I.M. Vascular adaptation in pregnancy and endothelial dysfunction in preeclampsia. J. Endocrinol. 2017, 232, R27–R44. [Google Scholar] [CrossRef]
- Westerberg, A.C.; Degnes, M.-H.L.; Andresen, I.J.; Roland, M.C.P.; Michelsen, T.M. Angiogenic and vasoactive proteins in the maternal-fetal interface in healthy pregnancies and preeclampsia. Am. J. Obstet. Gynecol. 2024, 231, 550.e1–550.e22. [Google Scholar] [CrossRef]
- Torres-Torres, J.; Espino-Y-Sosa, S.; Martinez-Portilla, R.; et al. A narrative rewiev on the pathophysioligy of preeclampsia. Int J Mol Sci. 2024, 25, 7569. [Google Scholar]
- Sandgren, J.A.; Deng, G.; Linggonegoro, D.W.; Scroggins, S.M.; Perschbacher, K.J.; Nair, A.R.; Nishimura, T.E.; Zhang, S.Y.; Agbor, L.N.; Wu, J.; et al. Arginine vasopressin infusion is sufficient to model clinical features of preeclampsia in mice. JCI Insight 2018, 3, e99403. [Google Scholar] [CrossRef]
- Chien, S. Red cell deformability and its relevance to blood flow. Ann Rev Physiol. 1987, 49, 177–192. [Google Scholar]
- Tamás, P.; Gresele, P.; Bódis, J.; Polidori, D.; Nenci, G.G.; Csaba, I.F. The reduced erythrocyte deformability in preeclampsia is due to altered plasma to red blood cell interaction. In: Cosmi AV, Di Renzo GC (eds): Hypertension in Pregnancy. Bologna, Monduzzi, 1991; pp. 399–402.
- Heilmann, L.; Rath, W.; Pollow, K. Hemorheological changes in women with severe preeclampsia. Clin Hemorheol Microcirc. 2004, 31, 49–58. [Google Scholar]
- Fakhouri, F.; Scully, M.; Provôt, F.; Blasco, M.; Coppo, P.; Noris, M.; Paizis, K.; Kavanagh, D.; Pène, F.; Quezada, S.; et al. Management of thrombotic microangiopathy in pregnancy and postpartum: report from an international working group. Blood 2020, 136, 2103–2117. [Google Scholar] [CrossRef]
- Vahed, S.Z.; Saadat, Y.R.; Ardalan, M. Thrombotic microangiopathy during pregnancy. Microvasc. Res. 2021, 138, 104226. [Google Scholar] [CrossRef]
- Harlow, F.H.; Brown, M.A.; Brighton, T.A.; Smith, S.L.; Trickett, A.E.; Kwan, Y.-L.; Davis, G.K. Platelet activation in the hypertensive disorders of pregnancy. Am. J. Obstet. Gynecol. 2002, 187, 688–695. [Google Scholar] [CrossRef]
- Bódis, J.; Papp, S.; Vermes, I.; et al. “Platelet-associated regulatory system” (PARS) with paticular reference to female reproduction. J Ovarian Res 2014, 7, 55. [Google Scholar] [CrossRef]
- Burwick, R.M.; Rincon, M.; Beeraka, S.S.; GuptaM; Feinberg, B. B. Eveluation of hemohysis as a severe feature of preeclampsia. Hypertension 2018, 72, 460–465. [Google Scholar] [CrossRef]
- Tamás, P.; Szilágyi, A.; Jeges, S.; Vizer, M.; Csermely, T.; Ifi, Z.; Bálint, A.; Szabó, I. Effects of maternal central hemodynamics on fetal heart rate patterns. Acta Obstet. et Gynecol. Scand. 2007, 86, 711–714. [Google Scholar] [CrossRef]
- Meah, V.L.; Cockcroft, J.R.; Backx, K.; Shave, R.; Stöhr, E.J. Cardiac output and related haemodynamics during pregnancy: a series of meta-analyses. Heart 2016, 102, 518–526. [Google Scholar] [CrossRef]
- Valensise, H.; Vasapollo, B.; Gagliardi, G.; Novelli, G.P. Early and late preeclampsia: two different maternal hemodynamic states in the latent phase of the disease. Hypertension 2008, 52, 873–880. [Google Scholar] [CrossRef]
- Ives, C.W.; Sinkey, R.; Rajapreyar, I.; Tita, A.T.N.; Oparil, S.J. Preeclampsia − pathophysiology and clinical presentations: JACC State-of-the-art review. Am Coll Cardiol. 2020, 76, 1690–1702. [Google Scholar] [CrossRef]
- Verlohren, S.; Shaun PBrennecke, S.P.; Galindo, A.; et al. Clinical interpretation and implementation of the sFlt-1/PlGF ratio in the prediction, diagnosis and management of preeclampsia. Clin Hypertens. 2022, 27, 42–50. [Google Scholar] [CrossRef]
- Bhide, A.; Acharya, G.; Bilardo, C.M. ISUOG practice guidelines: use of Doppler ultrasonography in obstetrics. Ultrasound Obstet Gynecol. 2013, 41, 233–239. [Google Scholar]
- Prefumo, F.; Campbell, S.; Jauniaux, E. Maternal blood flow to the placenta. BJOG: Int. J. Obstet. Gynaecol. 2019, 126, 972–972. [Google Scholar] [CrossRef]
- Di Mascio, D.; Saccone, G.; Bellussi, F.; Vitagliano, A.; Berghella, V. Type of paternal sperm exposure before pregnancy and the risk of preeclampsia: A systematic review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 251, 246–253. [Google Scholar] [CrossRef]
- Rolnik, D.L.; Nikolaides, K.H.; Poon, L.C. Prevention of preeclampsia with aspirin. Am J Obstet Gynecol. 2022, 226, S1108–S1119. [Google Scholar]
- Tamás, P.; Bódis, J. The Possible role of Microcirculation in the Pathogenesis of Preeclampsia. Hypertens. Pregnancy 1994, 13, 215–216. [Google Scholar] [CrossRef]
- von Dadelszen, P.; Magee, L.A. Fall in Mean Arterial Pressure and Fetal Growth Restriction in Pregnancy Hypertension: An Updated Metaregression Analysis. J. Obstet. Gynaecol. Can. 2002, 24, 941–945. [Google Scholar] [CrossRef]
- Khedun, S.M.; Maharaj, B.; Moodley, J.M. Effects of antihypertensive drugs on the unborn child: what is known, and how should this influence prescribing? Pediatr Drugs. 2000, 6, 419–436. [Google Scholar] [CrossRef]
- Chiarello, D.I.; Marín, R.; Proverbio, F.; Coronado, P.; Toledo, F.; Salsoso, R.; Gutiérrez, J.; Sobrevia, L. Mechanisms of the effect of magnesium salts in preeclampsia. Placenta 2018, 69, 134–139. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Sun, D. Calcium Dobesilate and Micro-vascular diseases. Life Sci. 2019, 221, 348–353. [Google Scholar] [CrossRef]
- Tamás, P.; Csermely, T.; Ertl, T.; Vizer, M.; Szabó, I.; Prievara, F.T. Calcium Dobesilate Lowers the Blood Pressure in Mild to Moderate Midtrimester Hypertension. Gynecol. Obstet. Investig. 1999, 47, 210–213. [Google Scholar] [CrossRef]
- Burwick, R.M.; Rodriguez, M.H. Angiogenic Biomarkers in Preeclampsia. Obstet. Gynecol. 2024, 143, 515–523. [Google Scholar] [CrossRef]
- Amorim, M.M.R.; Santas, L.C.; Faunders, A. Corticosteroid therapy for prevention of respiratory distress syndrome in severe preeclampsia. Am J Obstet Gynecol 1999, 180, 1283–1288. [Google Scholar]
- Gyselaers, W. Hemodynamic pathways of gestational hypertension and preeclampsia. Am. J. Obstet. Gynecol. 2022, 226, S988–S1005. [Google Scholar] [CrossRef]
- Flythe, J.E.; Bansal, N. The relationship of volume overload and its control to hypertension in hemodialysis patients. Semin Dial. 2019, 32, 500–506. [Google Scholar]
- Mitsides, N.; Cornelis, T.; Broers, N.J.H.; Diederen, N.M.P.; Brenchley, P.; van der Sande, F.M.; Schalkwijk, C.G.; Kooman, J.P.; Mitra, S. Extracellular overhydration linked with endothelial dysfunction in the context of inflammation in haemodialysis dependent chronic kidney disease. PLOS ONE 2017, 12, e0183281. [Google Scholar] [CrossRef]
- Tamás, P.; Bódis, J.; Sulyok, E.; Kovács, G.L.; Hantosi, E.; Molnár, G.; Martens-Lobenhoffer, J.; Bode-Böger, S.M. L-arginine metabolism in early-onset and late-onset pre-eclamptic pregnancies. Scand. J. Clin. Lab. Investig. 2013, 73, 436–443. [Google Scholar] [CrossRef]
- Ruhstaller, K.E.; Bastek, J.A.; Thomas, A.; Mcelrath, T.F.; Parry, S.I.; Durnwald, C.P. The Effect of Early Excessive Weight Gain on the Development of Hypertension in Pregnancy. Am. J. Perinatol. 2016, 33, 1205–1210. [Google Scholar] [CrossRef]
- Townsend, R.; Khalil, A.; Premakumar, Y. (IPPIC Network). Prediction of preeclampsia: a review of reviews. Ultrasound Obstet Gynecol. 2019, 54, 16–27. [Google Scholar]
- Macdonald-Wallis, C.; Tilling, K.; Fraser, A.; Nelson, S.M.; Lawlor, D.A. Gestational weight gain as a risk factor for hypertensive disorders of pregnancy. Am J Obstet Gynecol. 2013, 209, e1–e17. [Google Scholar]
- Haslam, D.W.; James, W.P.T. Obesity. Lancet. 2005, 366, 1197–1209. [Google Scholar]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar]
- Poirier, P.; Giles, T.D.; Bray, G.A.; et al. Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss. Arterioscler Thromb Vasc Biol. 2006, 26, 968–976. [Google Scholar]
- Jablonski, K.L.; Fedorova, O.V.; Racine, M.L.; Geolfos, C.J.; Gates, P.E.; Chonchol, M.; Fleenor, B.S.; Lakatta, E.G.; Bagrov, A.Y.; Seals, D.R. Dietary Sodium Restriction and Association with Urinary Marinobufagenin, Blood Pressure, and Aortic Stiffness. Clin. J. Am. Soc. Nephrol. 2013, 8, 1952–1959. [Google Scholar] [CrossRef]
- Agalakova, N.I.; Kolodkin, N.I.; Adair, C.D.; Trashkov, A.P.; Bagrov, A.Y. Preeclampsia: Cardiotonic Steroids, Fibrosis, Fli1 and Hint to Carcinogenesis. Int. J. Mol. Sci. 2021, 22, 1941. [Google Scholar] [CrossRef]
- Gyselaers, W. Maternal Venous Hemodynamic Dysfunction in Proteinuric Gestational Hypertension: Evidence and Implications. J. Clin. Med. 2019, 8, 335. [Google Scholar] [CrossRef]
- Kishimoto, T.; Maekawa, M.; Abe, Y.; Yamamoto, K. Intrarenal distribution of blood flow and renin release during renal venous pressure elevation. Kidney Int. 1973, 4, 259–266. [Google Scholar] [CrossRef]
- Rasmussen S, Irgens LM. Fetal growth and body proportion in preeclampsia. Obstet Gynecol. 2003, 101, 575–583. [Google Scholar]
- Easterling, T.R.; Benedetti, T.J.; Carlson, K.C.; Brateng, D.A.; Wilson, J.; Schmucker, B.S. The effect of maternal hemodynamics on fetal growth in hypertensive pregnancies. Am. J. Obstet. Gynecol. 1991, 165, 902–906. [Google Scholar] [CrossRef]
- Nisell, H.; Lunell, N.-O. Maternal hemodynamics and impaired fetal growth in pregnancy- Tamás P, Hantosi E, Menyhárt C, Bódis J. Fetal birth weight correlates to maternal cardiac output. Fetal birth weight correlates to maternal cardiac output. J Matern Fetal Neonatal Med. 2010, 23 (Suppl. S1), 633. [Google Scholar]
- Jhee, J.H.; Lee, S.; Park, Y.; Lee, S.E.; Kim, Y.A.; Kang, S.-W.; Kwon, J.-Y.; Park, J.T. Prediction model development of late-onset preeclampsia using machine learning-based methods. PLOS ONE 2019, 14, e0221202. [Google Scholar] [CrossRef]
- Roberge, S.; Bujold, E.; Nicolaides, K.H. Aspirin for the prevention of preterm and term preeclampsia: systematic review and metaanalysis. Am. J. Obstet. Gynecol. 2018, 218, 287–293.e1. [Google Scholar] [CrossRef]
- de Wardnerer, H.E.; He, J.F.; MacGregor, G.A. Plasma sodium and hypertension. Kidney Int. 2004, 66, 2454–2466. [Google Scholar]
- Hofmeyr, G.J.; Betrán, A.P.; Singata-Madliki, M.; Cormick, G.; Munjanja, S.P.; Fawcus, S.; Mose, S.; Hall, D.; Ciganda, A.; Seuc, A.H.; et al. Prepregnancy and early pregnancy calcium supplementation among women at high risk of pre-eclampsia: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet 2019, 393, 330–339. [Google Scholar] [CrossRef]
- Tay, J.; Foo, L.; Masini, G.; Bennett, P.R.; McEniery, C.; Wilkinson, I.; Lees, C.C. Early and late preeclampsia are characterized by high cardiac output, but in the presence of fetal growth restriction, cardiac output is low: insights from a prospective study. Am J Obstet Gynecol. 2018, 218, 517.e1–517.e12. [Google Scholar] [CrossRef]
- Collins, R.; Yusuf, S.; Peto, R. Overview of randomised trials of diuretics in pregnancy. . BMJ 1985, 290, 17–23. [Google Scholar] [CrossRef]
- Carr, D.B.; Gavrila, D.; Brateng, D.; Easterling, T.R. Maternal Hemodynamic Changes Associated with Furosemide Treatment. Hypertens. Pregnancy 2007, 26, 173–178. [Google Scholar] [CrossRef]
- Metthews, G.; Gornall, R.; Saunders, N.J. A randomized placebo controlled trial of loop diuretics in moderate/severe pre-eclampsia, following delivery. J Obstet Gynaecol. 1997, 17, 30–32. [Google Scholar]
- Ascarelli, M.H.; Johnson, V.; McCreary, H.; Cushman, J.; May, W.L.; Martin, J.N. Postpartum Preeclampsia Management With Furosemide: A Randomized Clinical Trial. Obstet. Gynecol. 2005, 105, 29–33. [Google Scholar] [CrossRef]
- Sibai, B.M.; Grossman, R.A.; Grossman, H.G. Effects of diuretics on plasma volume in pregnancies with long-term hypertension. Am. J. Obstet. Gynecol. 1984, 150, 831–835. [Google Scholar] [CrossRef]
- Tamás, P.; Hantosi, E.; Farkas, B.; Ifi, Z.; Betlehem, J.; Bódis, J. Preliminary study of the effects of furosemide on blood pressure during late-onset pre-eclampsia in patients with high cardiac output. Int. J. Gynecol. Obstet. 2016, 136, 87–90. [Google Scholar] [CrossRef]
- Varma, N.N.; Rastogi, S.; Chia, Y.-C.; et al. Non-pharmacological management of hypertension. J Clin Hypertens 2021, 23, 1275–128. [Google Scholar] [CrossRef]
- Chapell, L.C.; Brocklehurst, P.; Green, M.E.; Hunter, R.; Hardy, P.; Juszczak, E.; et al. (PHOENIX Study Group). Planned early delivery or expectant management for late preterm pre-eclampsia (PHOENIX): a randomized controlled trial. Lancet 2019, 394, 1181–1190.86. [Google Scholar] [CrossRef]
- Bertholdt, C.; Dap, M.; Beaumont, M.; Duan, J.; Morel, O. New insights into human functional ultrasound imaging. Placenta 2022, 117, 5–12. [Google Scholar] [CrossRef]
- Yang, C.; Baker, P.N.; Granger, J.P.; Davidge, S.T.; Tong, C. Long-Term Impacts of Preeclampsia on the Cardiovascular System of Mother and Offspring. Hypertension 2023, 80, 1821–1833. [Google Scholar] [CrossRef]
- Tamás, P.; Koppán, M. Suggestion for modified classification of hypertension durng pregnancy. Int J Gynecol Pract. 2020, 7, 152. [Google Scholar]
- induced hypertension. Obstet Gynecol. 1988, 71, 163–166.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).