Submitted:
10 March 2025
Posted:
11 March 2025
You are already at the latest version
Abstract
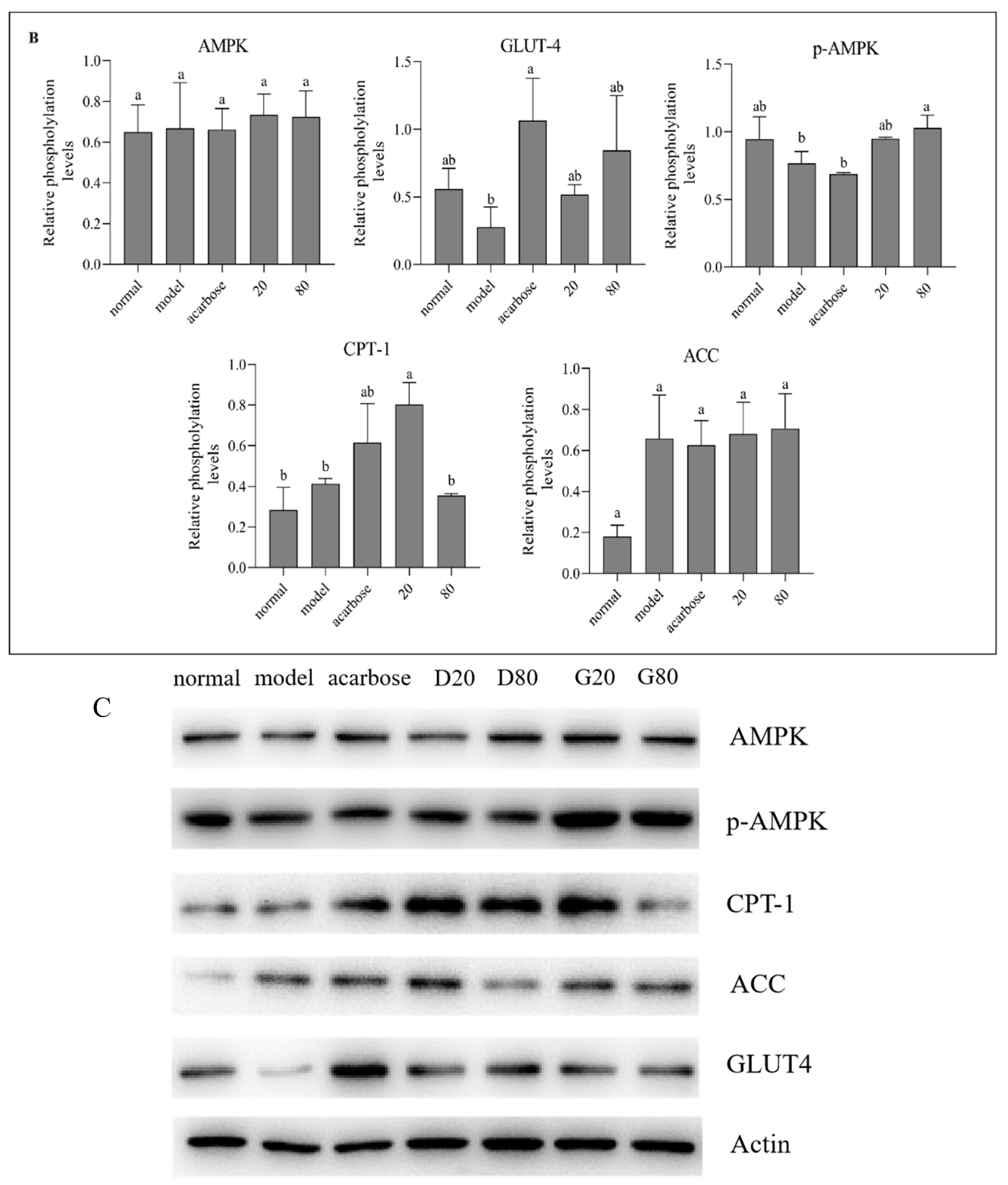
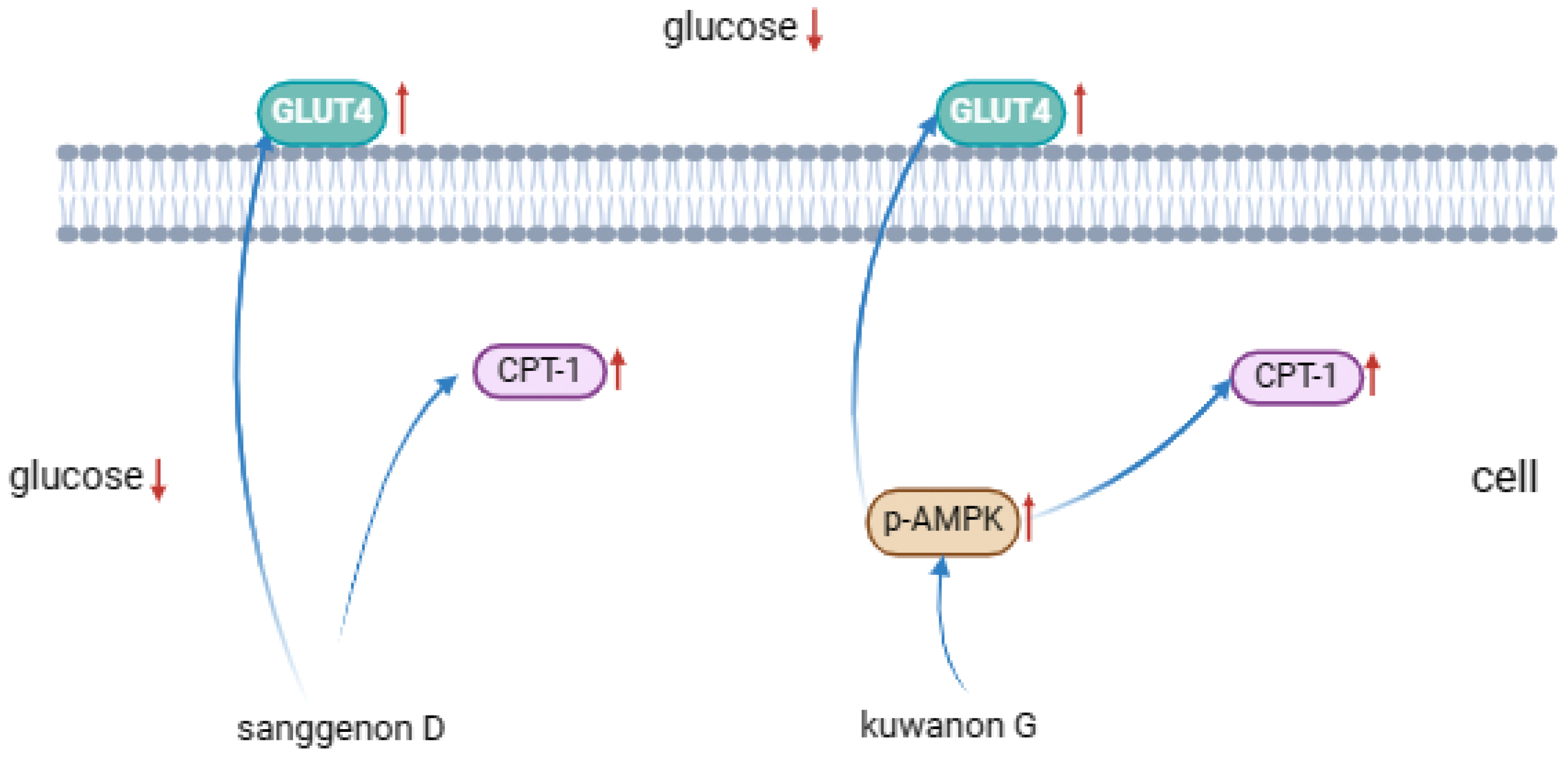
Inhibition of α-glucosidase activity is considered to be an effective treatment for type 2 diabetes. The inhibitory mechanisms of sanggenone D and kuwanon G on α-glucosidase were investigated and the pathways for hypoglycemic effects were explored in the current study. The outcomes indicated that sanggenone D (IC50: 4.51 × 10-5 mol/L) and kuwanon G (IC50: 3.83 × 10-5 mol/L) inhibited α-glucosidase activity by non-competition/anti-competition mixed inhibition and competitive inhibition, respectively. Besides, the secondary structure of α-glucosidase were altered by static quenching, and exhibited the decrease in α-helix, β-antiparallel content, while increase in β-sheet content. Furthermore, the interaction forces between sanggenone D/kuwanon G and α-glucosidase were hydrophobic interactions and hydrogen bonds as evidenced by molecular docking. The binding affinity, stability and binding energy aligned with the results of IC50. Cyclization in sanggenone D structure resulted in a decrease in the number of phenolic hydroxyl groups and thus a reduction in the formation of hydrogen bonds, which ultimately diminished the binding affinity of sanggenone D to α-glucosidase. In addition, western blot results showed that sanggenone D and kuwanon G regulated glucose metabolism by activating of GLUT4 pathway.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Inhibitory Activity
2.3. Inhibition Kinetics Assay
2.4. Multi-Fluorescence Spectroscopy
2.5. FT-IR Measurements
2.6. Molecular Docking
2.7. Cell Experiment
2.7.1. HepG2 Cell Cultivation
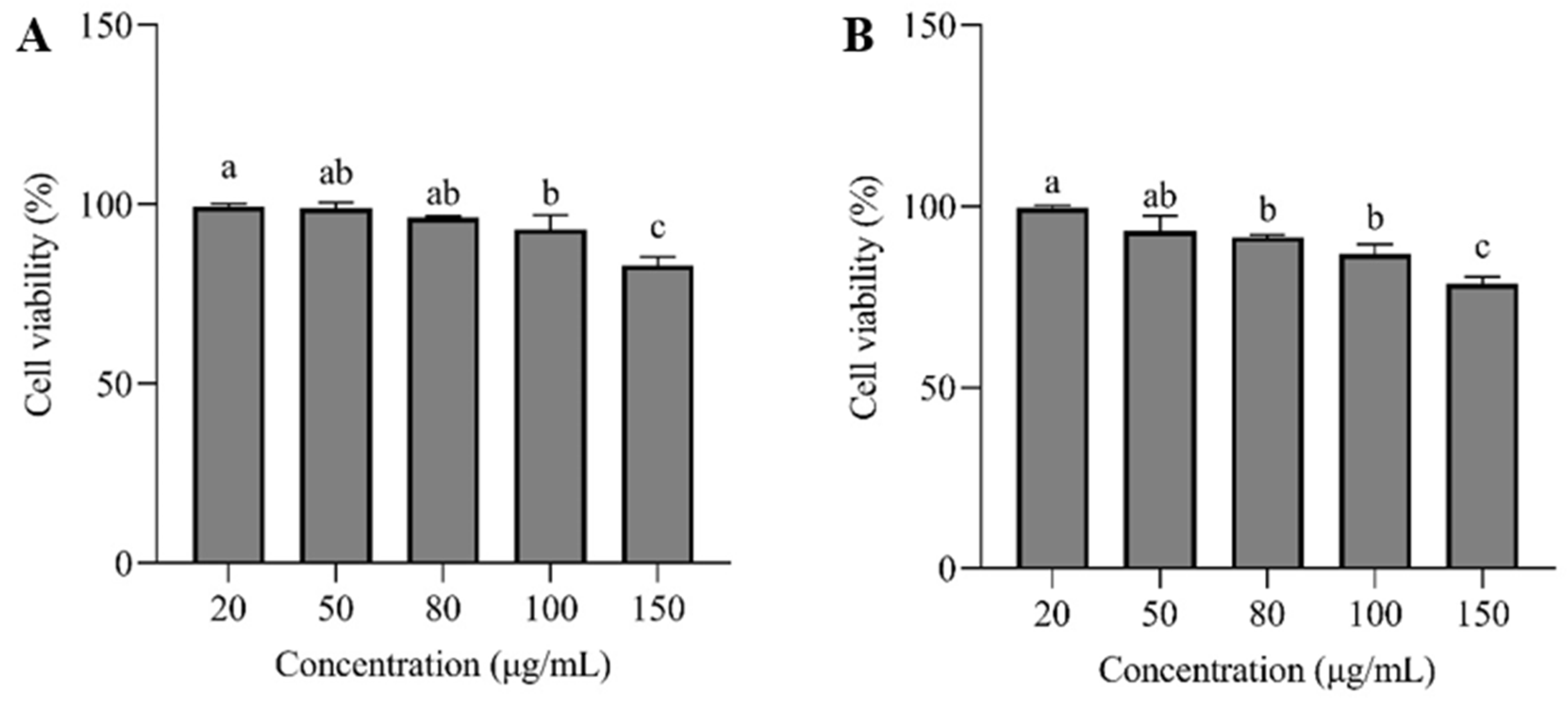
2.7.2. Toxicity Test of HepG2 Cells
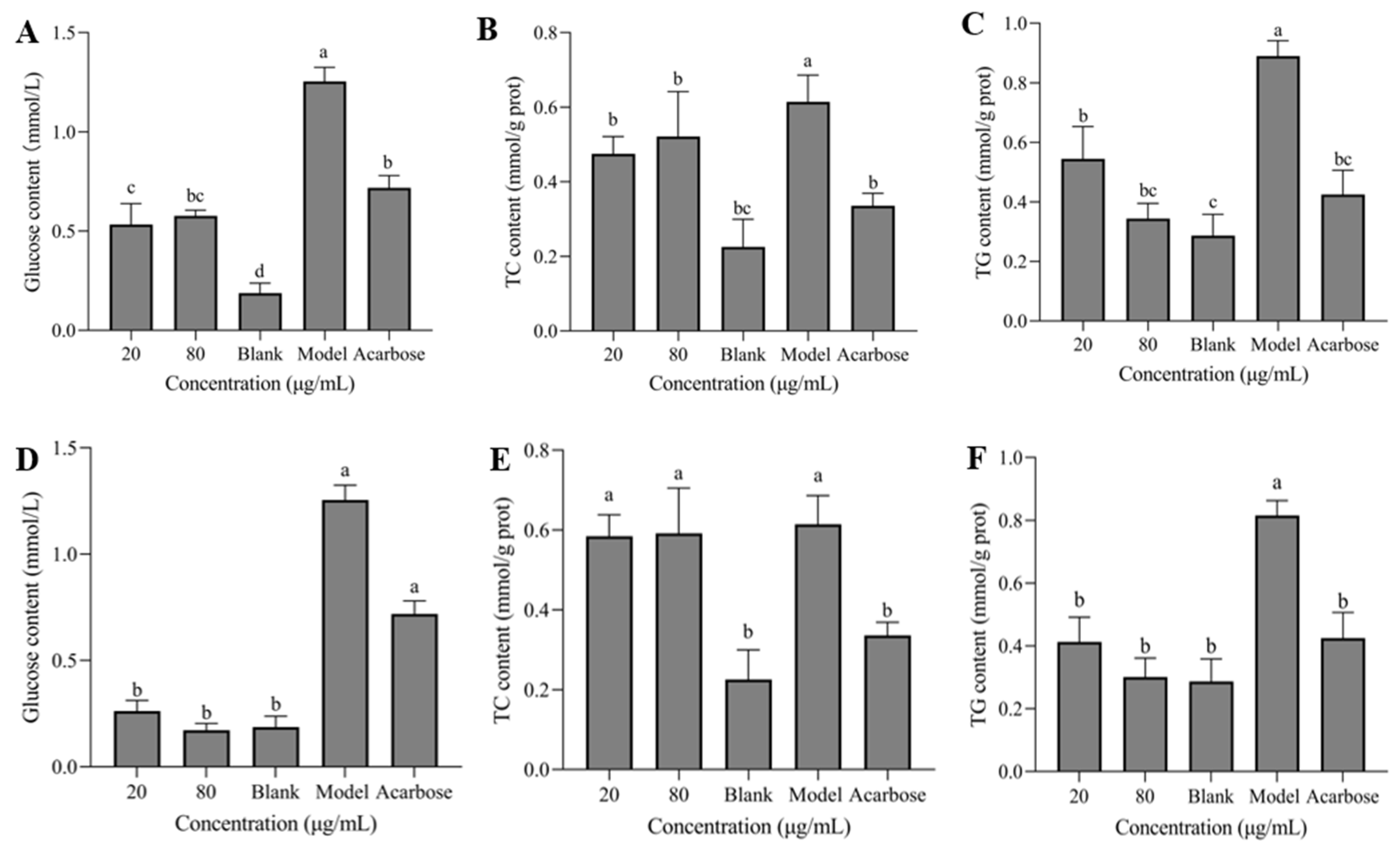
2.7.3. Determination of Cellular Glucose, TC and TG Content
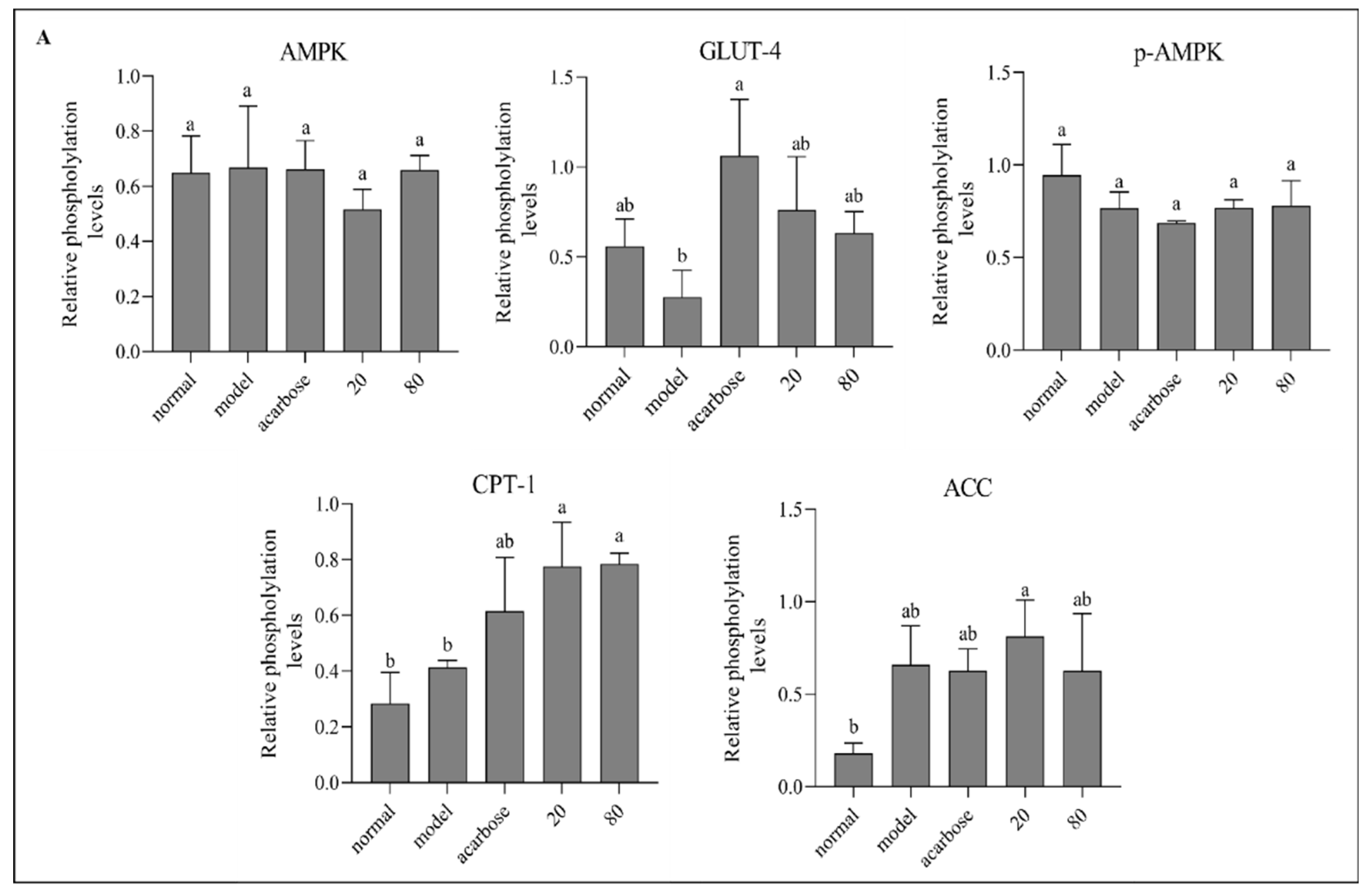
2.7.4. Western Blotting
2.8. Statistical Analysis
3. Results and Discussion
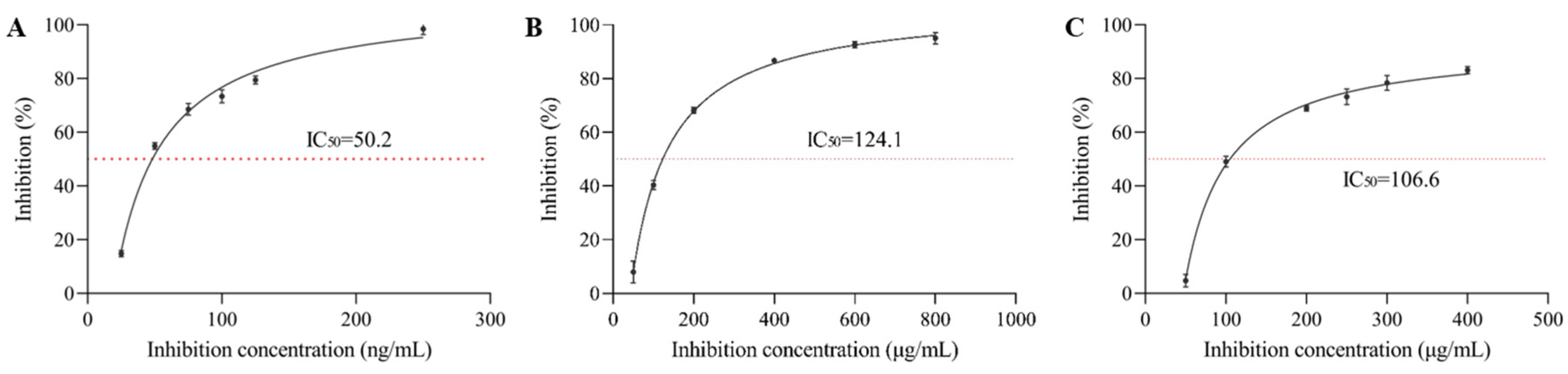
3.1. α-Glucosidase Inhibition Rate
3.2. Inhibition Type on α-Glucosidase Activity
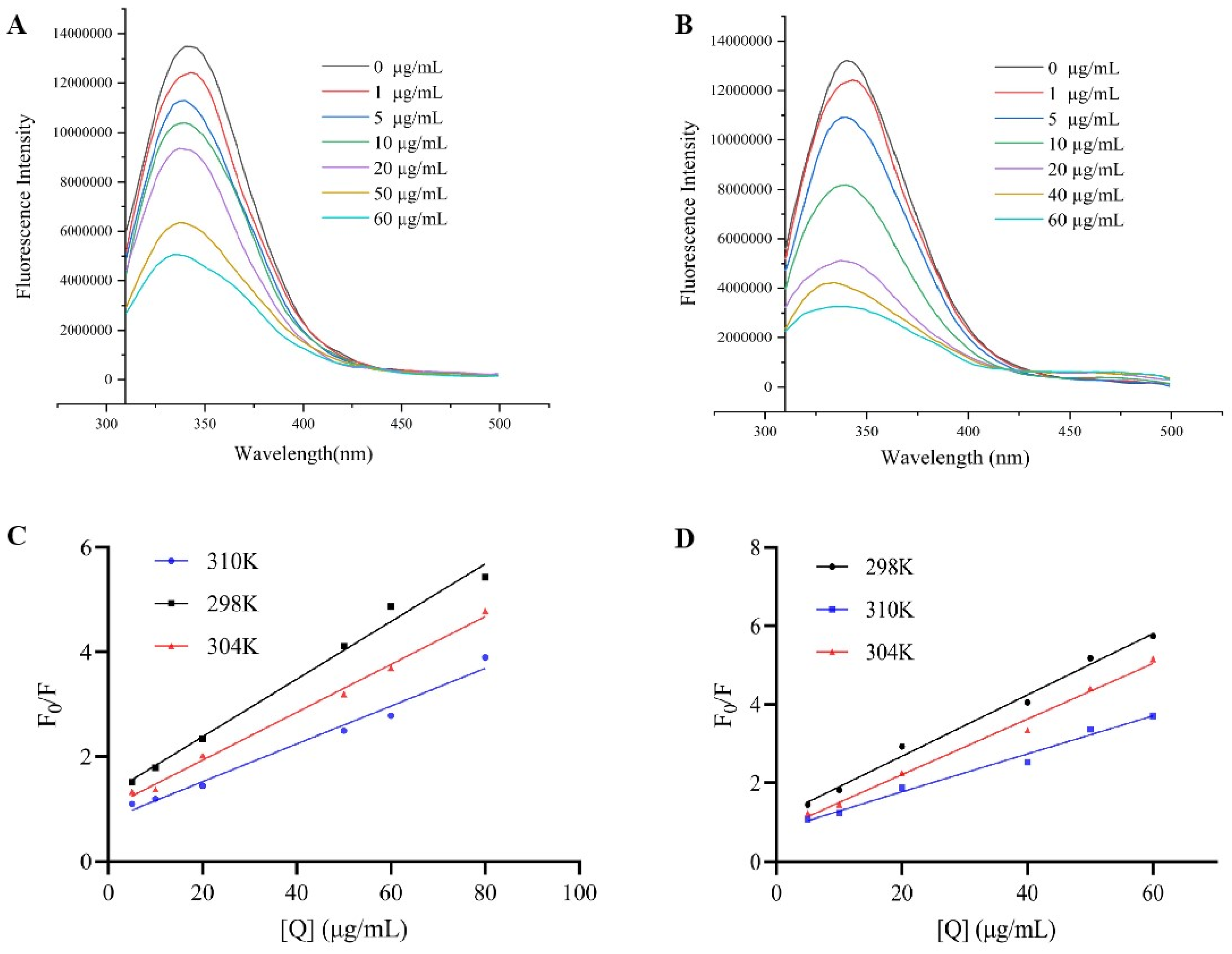
3.3. Fluorescence Spectroscopy Analysis of Binding Mechanism and Properties
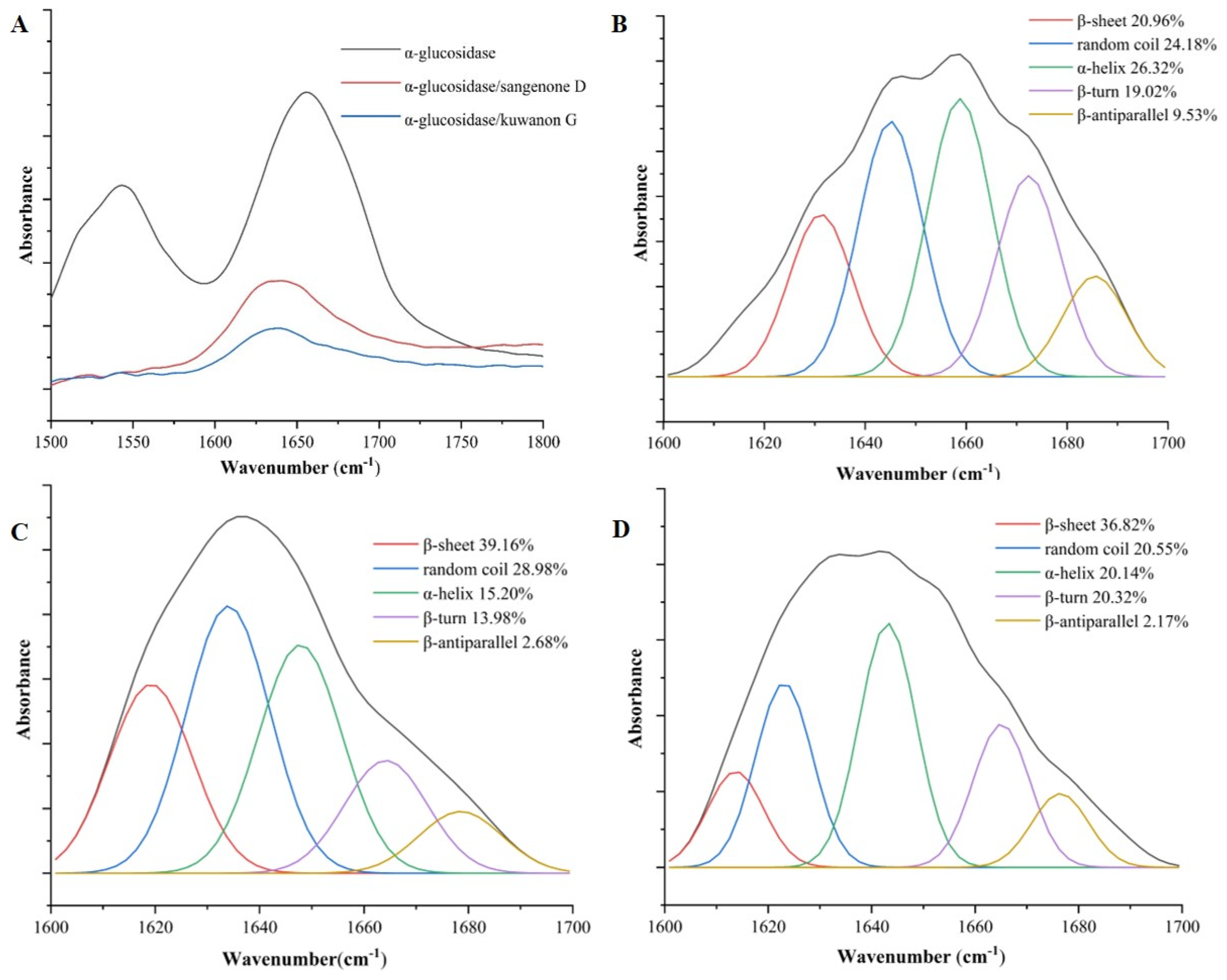
3.4. FT-IR Analysis
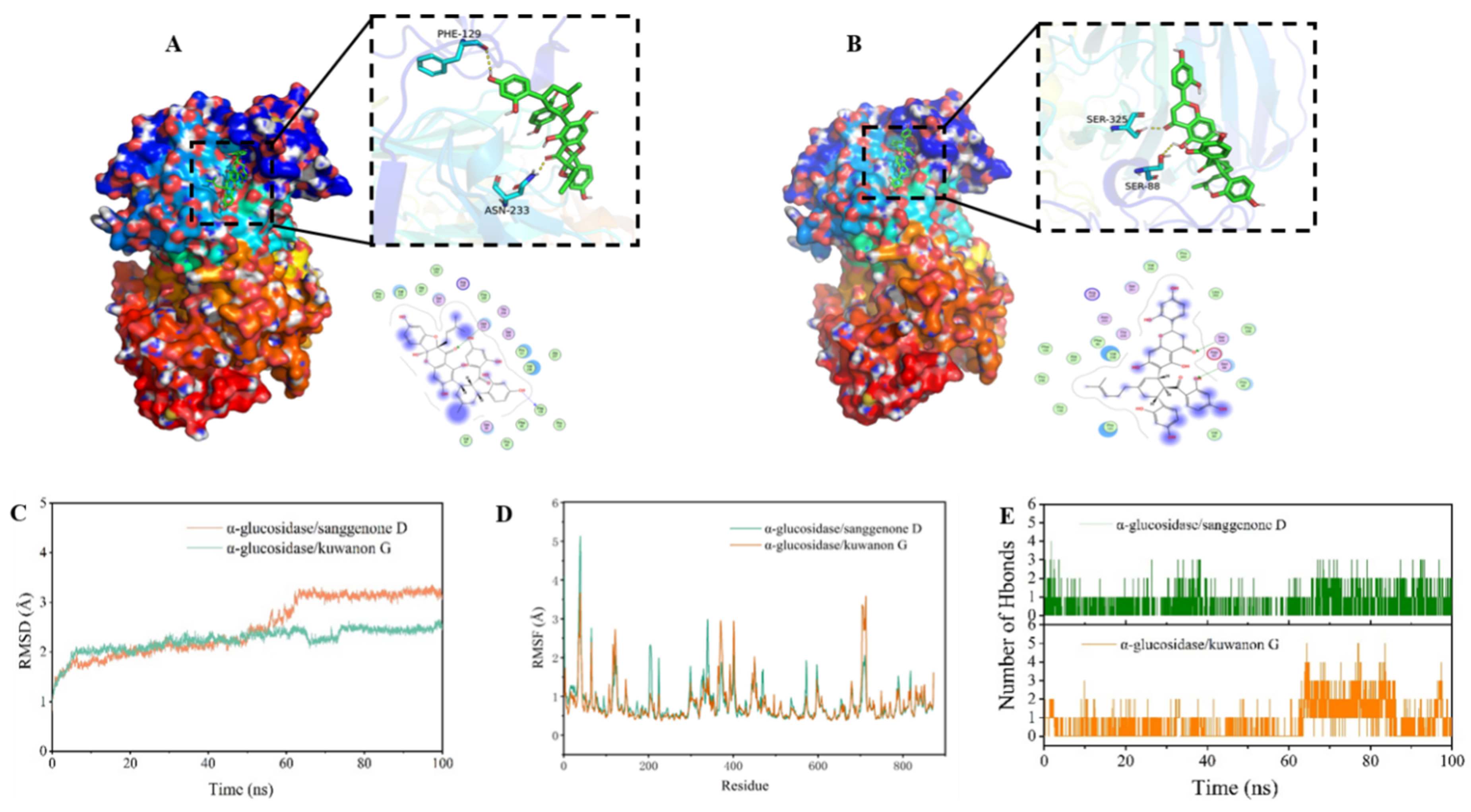
3.5. Molecular Docking
3.5.1. Docking Mode Analysis
3.5.2. MD Simulation
3.5.3. MM-GBSA Result
3.6. CCK-8 Assay
3.7. Glucose, TC, TG Contents
3.8. Western Blot Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations Used
References
- Eid, S.; Sas, K.M.; Abcouwer, S.F.; Feldman, E.L.; Gardner, T.W.; Pennathur, S.; Fort, P.E. New insights into the mechanisms of diabetic complications: role of lipids and lipid metabolism. Diabetologia 2019, 62, 1539–1549. [Google Scholar] [CrossRef]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef]
- Lee, S.-H.; Park, S.-Y.; Choi, C.S. Insulin Resistance: From Mechanisms to Therapeutic Strategies. Diabetes Metab. J. 2022, 46, 15–37. [Google Scholar] [CrossRef]
- Smirnova, I.E.; Galimova, Z.I.; Sapozhnikova, T.A.; Khisamutdinova, R.Y.; Thi, T.H.N.; Kazakova, O.B. New Dipterocarpol-Based Molecules with α-Glucosidase Inhibitory and Hypoglycemic Activity. ChemBioChem 2023, 25, e202300716. [Google Scholar] [CrossRef]
- Tan, Y.; Chang, S.K.; Zhang, Y. Comparison of α-amylase, α-glucosidase and lipase inhibitory activity of the phenolic substances in two black legumes of different genera. Food Chem. 2017, 214, 259–268. [Google Scholar] [CrossRef]
- Ma, Y.-Y.; Zhao, D.-G.; Zhou, A.-Y.; Zhang, Y.; Du, Z.; Zhang, K. α-Glucosidase Inhibition and Antihyperglycemic Activity of Phenolics from the Flowers of Edgeworthia gardneri. J. Agric. Food Chem. 2015, 63, 8162–8169. [Google Scholar] [CrossRef]
- Patel, P.; Shah, D.; Bambharoliya, T.; Patel, V.; Patel, M.; Patel, D.; Bhavsar, V.; Padhiyar, S.; Patel, B.; Mahavar, A.; et al. A Review on the Development of Novel Heterocycles as α-Glucosidase Inhibitors for the Treatment of Type-2 Diabetes Mellitus. Med. Chem. 2024, 20, 503–536. [Google Scholar] [CrossRef]
- Chan, E.W.-C.; Lye, P.-Y.; Wong, S.-K. Phytochemistry, pharmacology, and clinical trials of Morus alba. Chin. J. Nat. Med. 2016, 14, 17–30. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, Q.; Zhu, S.; Liu, B.; Liu, F.; Xu, Y. Mulberry leaf (Morus alba L.): A review of its potential influences in mechanisms of action on metabolic diseases. Pharmacol. Res. 2021, 175, 106029. [Google Scholar] [CrossRef]
- Jucá, M.M.; Filho, F.M.S.C.; De Almeida, J.C.; Mesquita, D.D.S.; Barriga, J.R.D.M.; Dias, K.C.F.; Barbosa, T.M.; Vasconcelos, L.C.; Leal, L.K.A.M.; Ribeiro, J.E.; et al. Flavonoids: biological activities and therapeutic potential. Nat. Prod. Res. 2020, 34, 692–705. [Google Scholar] [CrossRef]
- Van Acker, S.A.B.E.; Van Den Berg, D.-J.; Tromp, M.N.J.L.; Griffioen, D.H.; Van Bennekom, W.P.; Van Der Vijgh, W.J.F.; Bast, A. Structural aspects of antioxidant activity of flavonoids. Free Radic. Biol. Med. 1996, 20, 331–342. [Google Scholar] [CrossRef]
- Kimura, T.; Nakagawa, K.; Kubota, H.; Kojima, Y.; Goto, Y.; Yamagishi, K.; Oita, S.; Oikawa, S.; Miyazawa, T. Food-Grade Mulberry Powder Enriched with 1-Deoxynojirimycin Suppresses the Elevation of Postprandial Blood Glucose in Humans. J. Agric. Food Chem. 2007, 55, 5869–5874. [Google Scholar] [CrossRef]
- Musabayane, C.; Bwititi, P.; Ojewole, J. Effects of oral administration of some herbal extracts on food consumption and blood glucose levels in normal and streptozotocin-treated diabetic rats. Methods Find. Exp. Clin. Pharmacol. 2006, 28, 223–8. [Google Scholar] [CrossRef]
- Wen, L.; Zhou, T.; Jiang, Y.; Chang, S.K.; Yang, B. Prenylated flavonoids in foods and their applications on cancer prevention. Crit. Rev. Food Sci. Nutr. 2021, 62, 5067–5080. [Google Scholar] [CrossRef]
- Sun, H.; Li, Y.; Zhang, X.; Lei, Y.; Ding, W.; Zhao, X.; Wang, H.; Song, X.; Yao, Q.; Zhang, Y.; et al. Synthesis, α-glucosidase inhibitory and molecular docking studies of prenylated and geranylated flavones, isoflavones and chalcones. Bioorganic Med. Chem. Lett. 2015, 25, 4567–4571. [Google Scholar] [CrossRef]
- Sadeghi, M.; Miroliaei, M.; Ghanadian, M.; Szumny, A.; Rahimmalek, M. Exploring the inhibitory properties of biflavonoids on α-glucosidase; computational and experimental approaches. Int. J. Biol. Macromol. 2023, 253, 127380. [Google Scholar] [CrossRef]
- Zou, Y.; Mei, C.; Liu, F.; Xing, D.; Pang, D.; Li, Q. The lipase inhibitory effect of mulberry leaf phenolic glycosides: The structure-activity relationship and mechanism of action. Food Chem. 2024, 458, 140228. [Google Scholar] [CrossRef]
- Burley, S.K.; Berman, H.M.; Kleywegt, G.J.; Markley, J.L.; Nakamura, H.; Velankar, S. Protein Data Bank (PDB): The single global macromolecular structure archive. Methods Mol. Biol. 2017, 1067, 627–641. [Google Scholar]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
- Salomon-Ferrer, R.; Case, D.A.; Walker, R.C. An overview of the Amber biomolecular simulation package. WIREs Comput. Mol. Sci. 2012, 3, 198–210. [Google Scholar] [CrossRef]
- Genheden, S.; Ryde, U. The MM/PBSA and MM/GBSA methods to estimate ligand-binding affinities. Expert Opin. Drug Discov. 2015, 10, 449–461. [Google Scholar] [CrossRef]
- Cao, M.; Wang, J.; Jiang, X.; Sun, Z.; Zhao, L.; Chen, G. Phenolic Constituents from Black Quinoa Alleviate Insulin Resistance in HepG2 Cells via Regulating IRS1/PI3K/Akt/GLUTs Signaling Pathways. J. Agric. Food Chem. 2023, 71, 18780–18791. [Google Scholar] [CrossRef]
- Zheng, Y.; Zheng, Y.; Tian, J.; Tian, J.; Yang, W.; Yang, W.; Chen, S.; Chen, S.; Liu, D.; Liu, D.; et al. Inhibition mechanism of ferulic acid against α-amylase and α-glucosidase. Food Chem. 2020, 317, 126346. [Google Scholar] [CrossRef]
- Xu, Z.; Hileuskaya, K.; Kraskouski, A.; Yang, Y.; Huang, Z.; Zhao, Z. Inhibition of α-glucosidase activity and intestinal glucose transport to assess the in vivo anti-hyperglycemic potential of dodecyl-acylated phlorizin and polydatin derivatives. Food Funct. 2024, 15, 4785–4804. [Google Scholar] [CrossRef]
- Sepehri, S.; Farhadi, G.; Maghbul, M.; Nasiri, F.; Faramarzi, M.A.; Mahnam, K.; Mojtabavi, S.; Mahdavi, M.; Oranj, Z.M. A new route to the synthesis of 2-hydrazolyl-4-thiazolidinone hybrids, evaluation of α-glucosidase inhibitory activity and molecular modeling insights. Heliyon 2024, 10, e36408. [Google Scholar] [CrossRef]
- Sari, S.; Barut, B.; Özel, A.; Şöhretoğlu, D. Tyrosinase inhibitory effects of Vinca major and its secondary metabolites: Enzyme kinetics and in silico inhibition model of the metabolites validated by pharmacophore modelling. Bioorganic Chem. 2019, 92, 103259. [Google Scholar] [CrossRef]
- Li, Y.Q.; Zhou, F.C.; Gao, F.; Bian, J.S.; Shan, F. Comparative Evaluation of Quercetin, Isoquercetin and Rutin as Inhibitors of α-Glucosidase. J. Agric. Food Chem. 2009, 57, 11463–11468. [Google Scholar] [CrossRef]
- Peng, X.; Zhang, G.; Zeng, L. Inhibition of α-glucosidase by vitamin D3 and the effect of vitamins B1 and B2. Food Funct. 2015, 7, 982–991. [Google Scholar] [CrossRef]
- Sun, L.; Warren, F.J.; Netzel, G.; Gidley, M.J. 3 or 3′-Galloyl substitution plays an important role in association of catechins and theaflavins with porcine pancreatic α-amylase: The kinetics of inhibition of α-amylase by tea polyphenols. J. Funct. Foods 2016, 26, 144–156. [Google Scholar] [CrossRef]
- Sano, K.; Kuribara, T.; Ishii, N.; Kuroiwa, A.; Yoshihara, T.; Tobita, S.; Totani, K.; Matsuo, I. Fluorescence Quenching-based Assay for Measuring Golgi endo-α-Mannosidase. Chem. – Asian J. 2019, 14, 1965–1969. [Google Scholar] [CrossRef]
- Genovese, D.; Cingolani, M.; Rampazzo, E.; Prodi, L.; Zaccheroni, N. Static quenching upon adduct formation: a treatment without shortcuts and approximations. Chem. Soc. Rev. 2021, 50, 8414–8427. [Google Scholar] [CrossRef]
- Chen, X.; He, X.; Zhang, B.; Sun, L.; Liang, Z.; Huang, Q. Wheat gluten protein inhibits α-amylase activity more strongly than a soy protein isolate based on kinetic analysis. Int. J. Biol. Macromol. 2019, 129, 433–441. [Google Scholar] [CrossRef]
- Yang, J.; Wang, X.; Zhang, C.; Ma, L.; Wei, T.; Zhao, Y.; Peng, X. Comparative study of inhibition mechanisms of structurally different flavonoid compounds on α-glucosidase and synergistic effect with acarbose. Food Chem. 2021, 347, 129056. [Google Scholar] [CrossRef]
- Zhang, G.; Ma, Y. Mechanistic and conformational studies on the interaction of food dye amaranth with human serum albumin by multispectroscopic methods. Food Chem. 2013, 136, 442–449. [Google Scholar] [CrossRef]
- Haroun, A.A.; El Toumy, S.A. Effect of natural polyphenols on physicochemical properties of crosslinked gelatin-based polymeric biocomposite. J. Appl. Polym. Sci. 2010, 116, 2825–2832. [Google Scholar] [CrossRef]
- Wongsa, P.; Phatikulrungsun, P.; Prathumthong, S. FT-IR characteristics, phenolic profiles and inhibitory potential against digestive enzymes of 25 herbal infusions. Sci. Rep. 2022, 12, 1–11. [Google Scholar] [CrossRef]
- Nie, C.; Zou, Y.; Liao, S.; Gao, Q.; Li, Q. Molecular Targets and Mechanisms of 6,7-Dihydroxy-2,4-dimethoxyphenanthrene from Chinese Yam Modulating NF-κB/COX-2 Signaling Pathway: The Application of Molecular Docking and Gene Silencing. Nutrients 2023, 15, 883. [Google Scholar] [CrossRef]
- Mohseni-Shahri, F.S.; Moeinpour, F.; Nosrati, M. Spectroscopy and molecular dynamics simulation study on the interaction of sunset yellow food additive with pepsin. Int. J. Biol. Macromol. 2018, 115, 273–280. [Google Scholar] [CrossRef]
- Santana, D.; Trindade, I.; Carvalho, Y.; Carvalho-Neto, A.; Silva, E.; Silva-Júnior, E.; Leite, R.; Quintans-Júnior, L.; Aquino, T.; Serafini, M.; et al. Analytical techniques to recognize inclusion complexes formation involving monoterpenes and cyclodextrins: A study case with (–) borneol, a food ingredient. Food Chem. 2021, 339, 127791. [Google Scholar] [CrossRef]
- Zhang, X.; Li, D.; Wang, K.; Xie, J.; Liu, Y.; Wang, T.; Liu, S.; Huang, Q.; Guo, Q.; Wang, H. Hyperoside inhibits pancreatic lipase activity in vitro and reduces fat accumulation in vivo. Food Funct. 2023, 14, 4763–4776. [Google Scholar] [CrossRef]
- White, S.L.; Pasupathy, D.; Sattar, N.; Nelson, S.M.; Lawlor, D.A.; Briley, A.L.; Seed, P.T.; Welsh, P.; Poston, L. Metabolic profiling of gestational diabetes in obese women during pregnancy. Diabetologia 2017, 60, 1903–1912. [Google Scholar] [CrossRef]
- Garcia, D.; Shaw, R.J. AMPK: Mechanisms of Cellular Energy Sensing and Restoration of Metabolic Balance. Mol. Cell 2017, 66, 789–800. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, P.; Gnudi, L.; Tozzo, E.; Yang, H.; Leach, F.; Kahn, B. Adipose cell hyperplasia and enhanced glucose disposal in transgenic mice overexpressing GLUT4 selectively in adipose tissue. J. Biol. Chem. 1993, 268, 22243–22246. [Google Scholar] [CrossRef]
- Nakamura, M.T.; Yudell, B.E.; Loor, J.J. Regulation of energy metabolism by long-chain fatty acids. Prog. Lipid Res. 2014, 53, 124–144. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Kang, M.; Xie, Q.; Xu, B.; Sun, C.; Chen, K.; Wu, Y. Anthocyanins from Chinese Bayberry Extract Protect β Cells from Oxidative Stress-Mediated Injury via HO-1 Upregulation. J. Agric. Food Chem. 2010, 59, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.J.; Jang, D.-J.; Hwang, J.-T. Anti-diabetic effects of Korean red pepper via AMPK and PPAR-γ activation in C2C12 myotubes. J. Funct. Foods 2012, 4, 552–558. [Google Scholar] [CrossRef]
- Yoon, S.-A.; Kang, S.-I.; Shin, H.-S.; Kang, S.-W.; Kim, J.-H.; Ko, H.-C.; Kim, S.-J. p-Coumaric acid modulates glucose and lipid metabolism via AMP-activated protein kinase in L6 skeletal muscle cells. Biochem. Biophys. Res. Commun. 2013, 432, 553–557. [Google Scholar] [CrossRef]
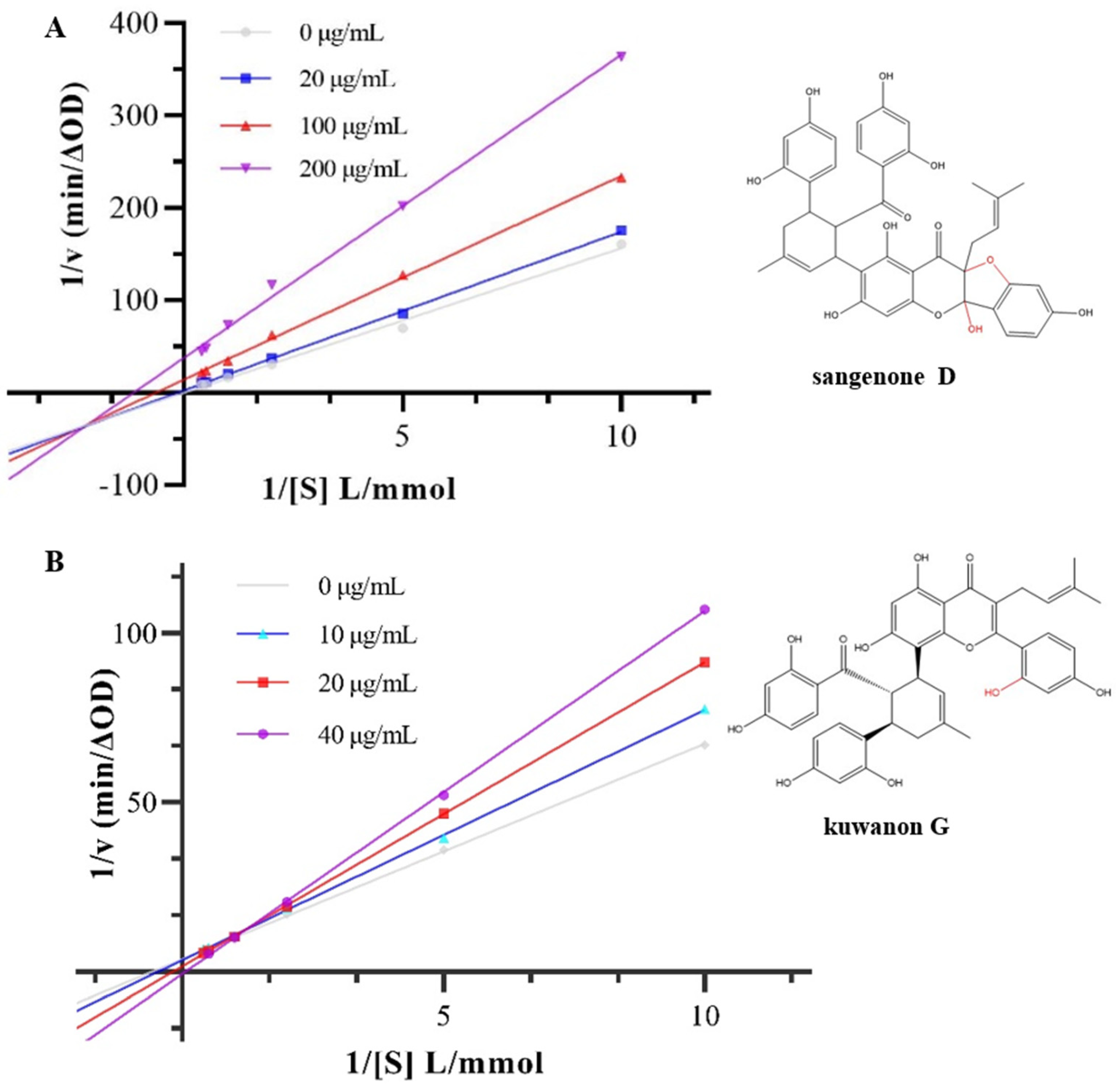
| concentration (μg/mL) | Km | Vm | Ki | Kis | |
|---|---|---|---|---|---|
| Sanggenon D | 0 | 78.49 | 5.02 | 252.95 | 9.86 |
| 20 | 17.18 | 1.00 | |||
| 100 | 1.57 | 0.071 | |||
| 200 | 0.89 | 0.027 | |||
| Kuwanon G | 0 | 1.73 | 0.27 | 83.46 | 50.89 |
| 10 | 2.17 | 0.29 | |||
| 20 | 5.68 | 0.63 | |||
| 40 | 15.98 | 1.49 |
| T(K) | KSV(×104 L/mol) |
Ra | Kq(×1012 L/mol) | Ka(×103 L/mol) |
n | Rb | |
|---|---|---|---|---|---|---|---|
| Sanggenon D | 298 | 3.90 ± 0.04 | 0.98 | 3.90 ± 0.04 | 7.28 ± 0.21 | 0.81 | 0.98 |
| 304 | 3.24 ± 0.02 | 0.99 | 3.24 ±0.02 | 16.73 ± 0.14 | 0.93 | 0.99 | |
| 310 | 2.56 ± 0.02 | 0.98 | 2.56 ±0.02 | 180.18 ± 0.30 | 1.22 | 0.98 | |
| Kuwanon G | 298 | 5.41 ± 0.01 | 0.99 | 5.41 ± 0.01 | 36.85 ± 0.17 | 0.96 | 0.99 |
| 304 | 4.91 ± 0.03 | 0.99 | 4.91 ± 0.03 | 194.98 ± 0.42 | 1.15 | 0.99 | |
| 310 | 3.35 ± 0.01 | 0.98 | 3.35 ± 0.01 | 1984.27 ± 0.33 | 1.43 | 0.98 |
| System name | α-glucosidase/sanggenon D | α-glucosidase/ kuwanon G |
| ΔEvdw | -22.55 ± 4.19 | -26.16 ± 2.43 |
| ΔEelec | 109.95 ± 4.95 | 59.73 ± 5.51 |
| ΔGGB | -94.59 ± 3.78 | -44.70 ± 6.19 |
| ΔGSA | -2.63 ± 0.51 | -2.69 ± 0.26 |
| ΔGbind | -9.83 ± 2.44 | -13.83 ± 2.94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




