Submitted:
05 March 2025
Posted:
05 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Instruments and Deceives
- Hot plat with magnetic stirring (HS-3000, S/N 0407-13, Camlab Ltd., Cambridge B24 5WE, UK) for heating and mixing as required.
- Four-digit analytical microbalance (Model SEJ-205, Taipei-Taiwan) for high accuracy weighting.
- pH meter (EUTEGH, Serial Number 897911, part of Thermos Scientific, Republic of Singapore, Singapore) for solutions pH measurement.
- Scanning electron microscope (SEM) (Inspect F50-FEI company, Eindhoven, The Netherlands) for high magnification and resolution imaging.
- Agar sputter coater instrument (Agar Scientific, Model AGB7340, Essex, UK) for specimens coating.
- Vacuum oven (JEIO TECH, MODEL OV-11, AAH13115K, Republic of Korea, Seoul) for drying under vacuum.
- Centrifugate system (HERMILE Labortechnik GmbH, Type Z 326 K, REF 311.00 V05, SN 66A1800211, Wehingen, Germany) for powder separation.
- Tube furnace (Protherm alumina tube furnace, Model PTF 12/50/450, serial No. 0907234, Protherm Inc, Ankara, Turkey) with a combined home-designed argon gas line and vacuum fitting for powder sintering.
- Digital caliper (Total, TMT 322001, Guangzhou, China) for high accuracy dimension measurements.
- Milling machine with variable speed (Changsha Tiachuang, Powder Technology Co., Changsha, China) for milling process.
- Compaction system consists of a homemade stainless-steel die integrated with a CARVER press (model 4350. L, S/N 4350-1401, CARVER, INC., Wabash, IN, USA) for powder compaction.
- Electron-Beam evaporation system (SCT-1800, SCT, System Control Technologies, sctec.com, Battle Ground, WA 98604, USA) for thin film deposition.
- UV–Visible spectrophotometer (UV-1601 (PC) S, CAT No. 206-67501-93, SERIAL No. A1077-3300310S2, Shimadzu Corporation, Tokyo, Japan) for optical properties investigation.
- Optical reflectance Spectrometer (FilmTek 3000, Scientific Computing International, Carlsbad, CA, USA) spectrometer for thin film thickness measurements.
- Fourier Transform Infrared Spectroscopy (FTIR, NEXUS, EPS-87, Thermo Fisher Scientific, Waltham, MA) in the wavenumber range of 400–4000 cm-1 for powder and thin film FTIR tests.
- X-ray diffraction (XRD) instrument (Malvern Panalytical, Aeris, monochromatic Cu kα1, 1.5406 Å, 0.02 step angle, with 2θ ranging from 10◦—60◦, Almelo, The Netherlands) for phase and unit cell investigation.
2.2. Materials
2.3. Production of Na2S Compound
2.4. Synthesis of ZnS
2.5. Sintering of ZnS Powder
2.6. Thin Film Deposition and UV–Visible Spectrophotometry
2.7. Characterizations of the Starting Powders, Synthesized ZnS, and ZnS Thin Films
3. Result and Discussion
3.1. SEM of Zn Powder, S Powder, and Synthetized ZnS Investigations
3.2. SEM Images of sintered ZnS Powder
3.3. Thin Film Microstructure at High Magnification
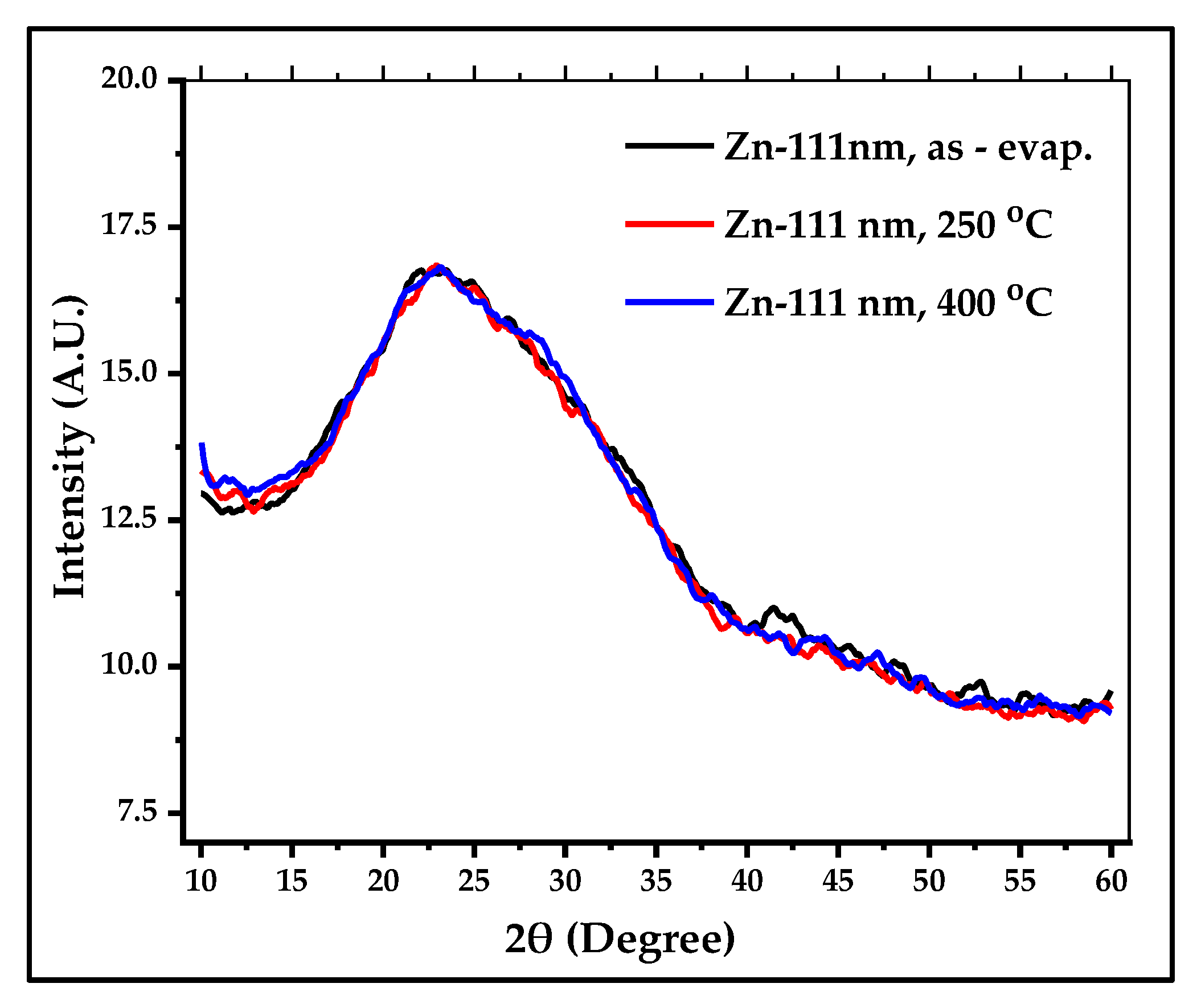
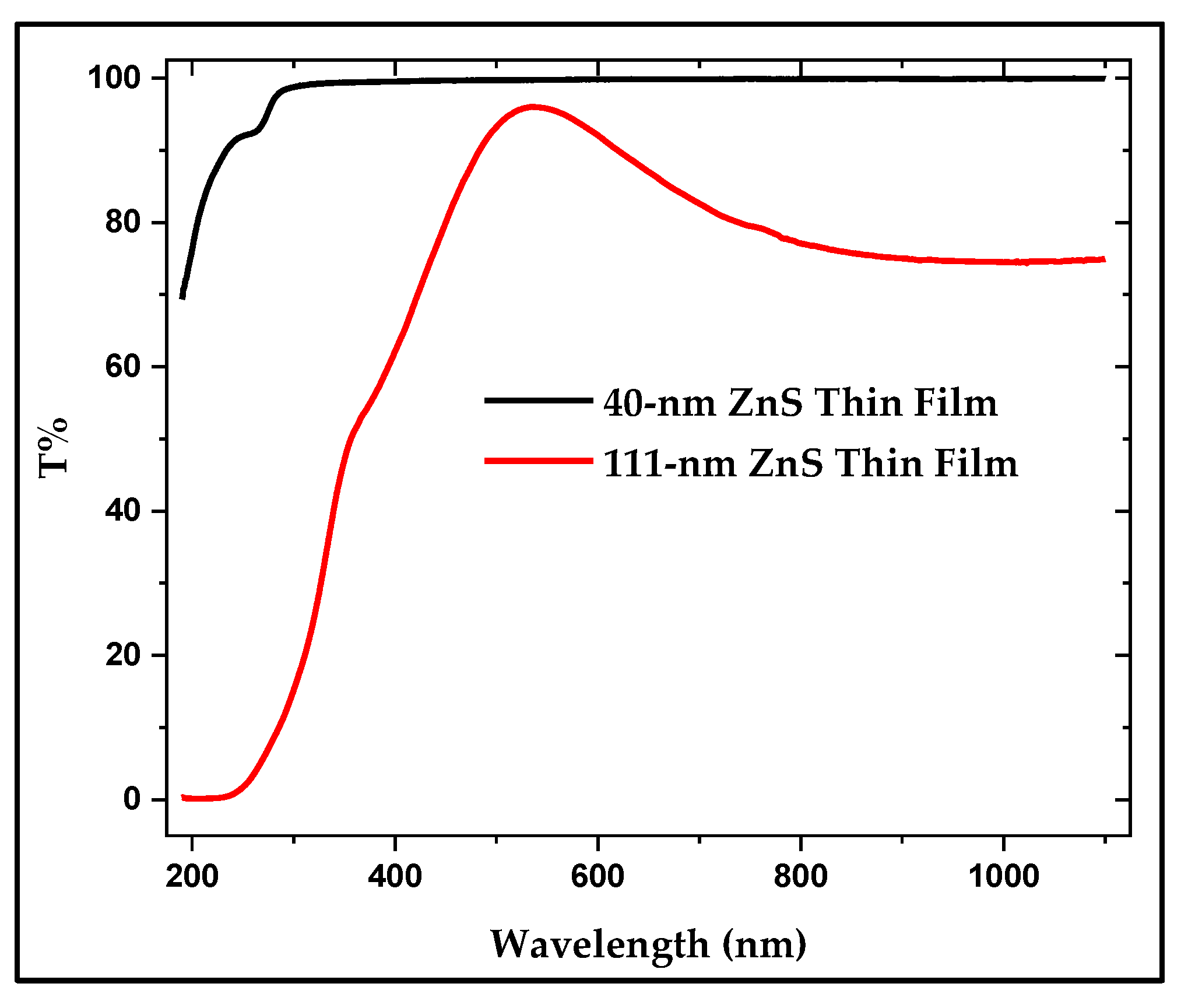
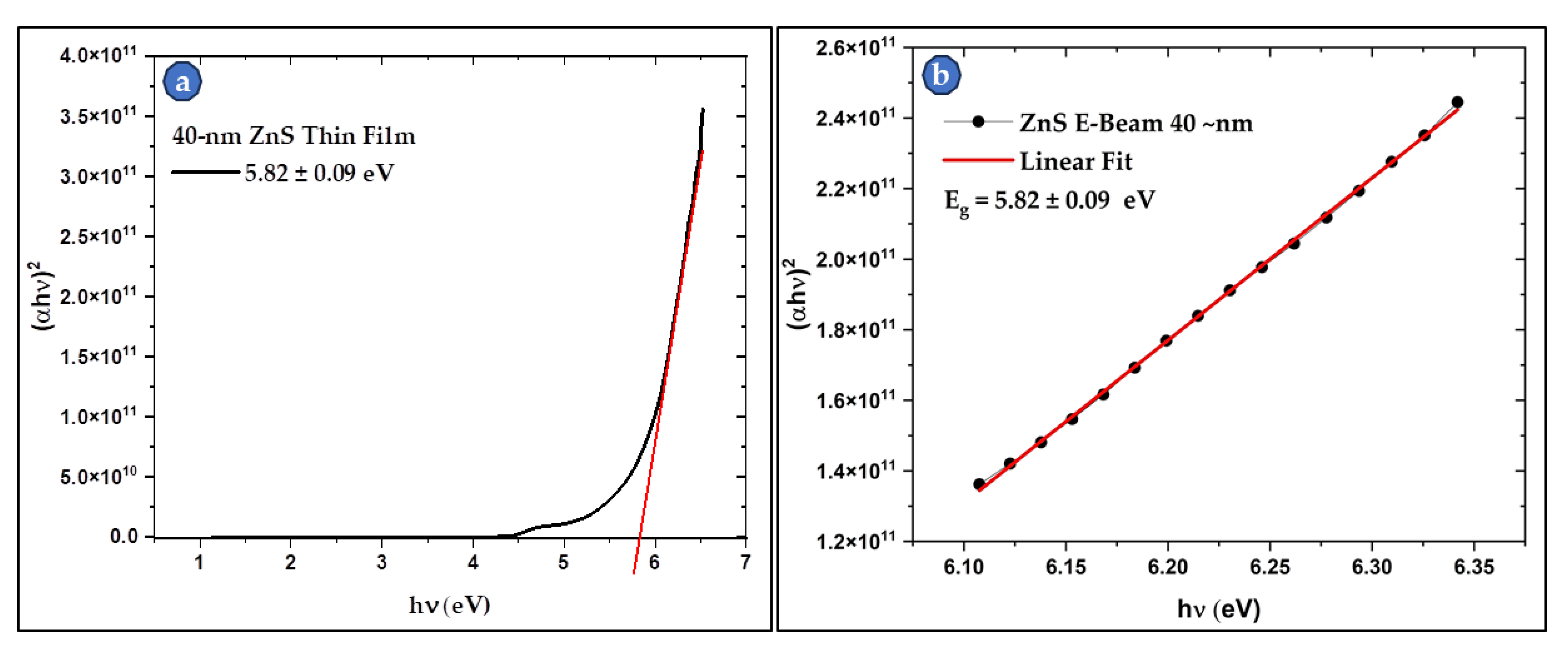
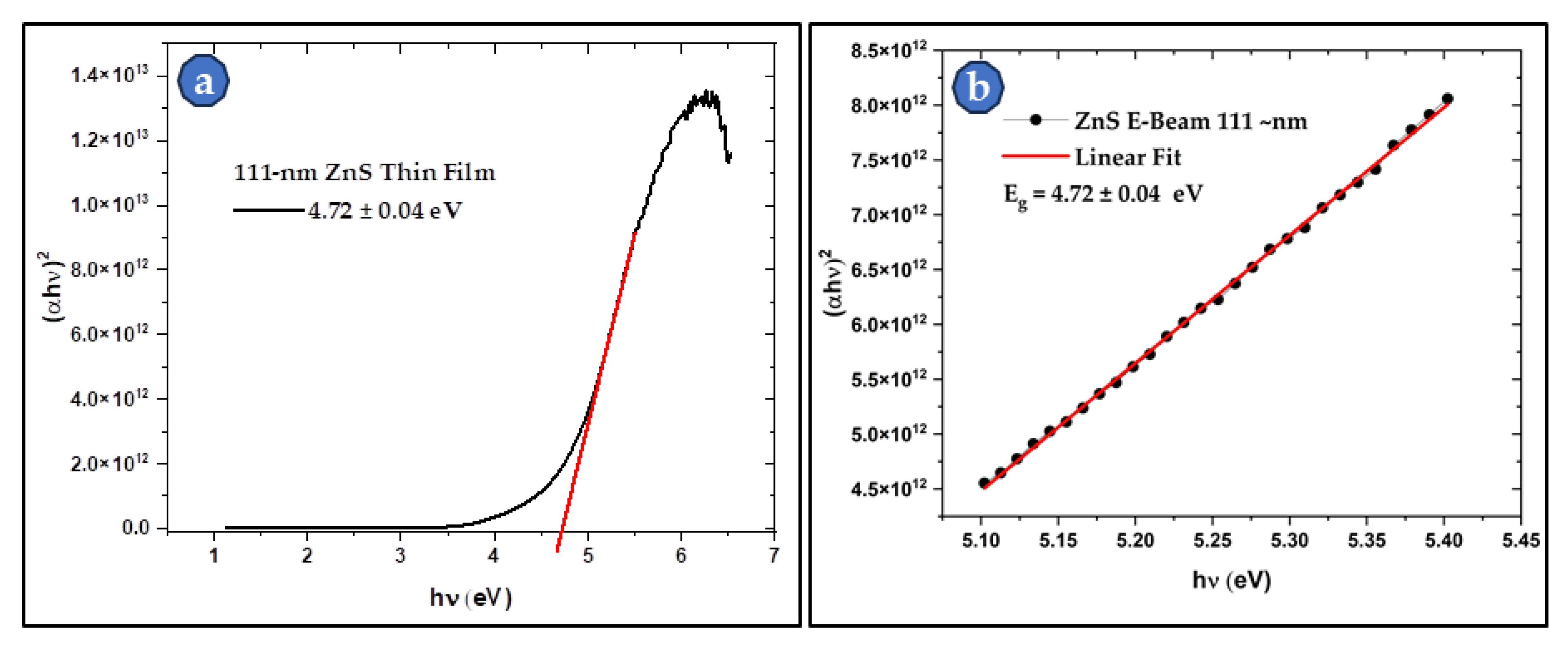
3.4. UV-Visible and FTIR Spectroscopy of the Produced ZnS
3.4.1. UV-Visible of the Thin Film Samples
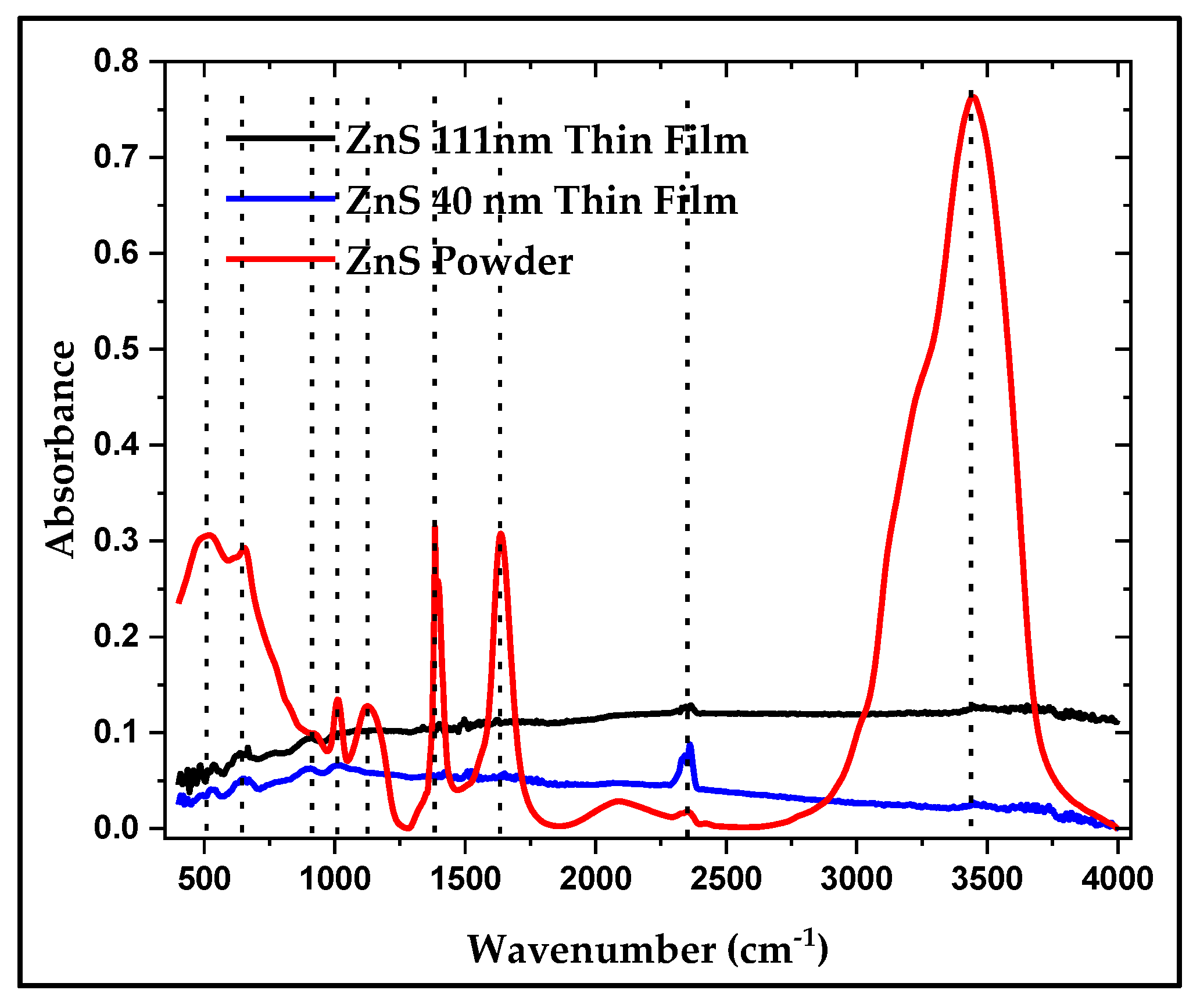
3.4.2. FTIR of ZnS Powder and its Thin Film
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability Statement
References
- Mana, P.M.; Bhujbal, P.K.; and Pathan, H.M. Fabrication and Characterization of ZnS based Photoelectrochemical Solar Cell. ES Energy Environ 2021, 12, 77–85. [Google Scholar] [CrossRef]
- Isac, L.; and Enesca, A. Recent Developments in ZnS-Based Nanostructures Photocatalysts for Wastewater Treatment. Int. J. Mol. Sci. 2022, 23, 15668. [Google Scholar] [CrossRef]
- Khan, A.U.; Tahir, K.; Albalawi, K.; Khalil, M.Y.; Almarhoon, Z.M.; Zaki, M.E.A.; Latif, S.; Hassan, H.M.A.; Refat, M.S.; and Munshi, A.M. Synthesis of ZnO and ZnS nanoparticles and their structural, optical, and photocatalytic properties synthesized via the wet chemical method. Materials Chemistry and Physics 2022, 219, 126667. [Google Scholar] [CrossRef]
- Faita, F.L.; Ersching, K.; Poffo, C.M.; Benetti, L.C.; Trichês, D.M.; Souza, S.M.; Viegas, A.D.C.; and de Lima, J. C. Structural, thermal, magnetic and optical characterization of undoped nanocrystalline ZnS prepared by solid state reaction. Journal of Alloys and Compounds 2014, 590, 176–183. [Google Scholar] [CrossRef]
- Wei, Z.; Lu, Y.; Zhao, J.; Zhao, S.; Wang, R.; Fu, N.; Li, X.; Guan, L.; and Teng, F. Synthesis and Luminescent Modulation of ZnS Crystallite by a Hydrothermal Method. ACS Omega 2018, 3, 137−143. [CrossRef]
- Kaur, H.; Kumar, S.; Kumar, P.; Ghfar, A. A.; and Bouzid, G. Tailoring ZnS nanostructures through precipitation-cum-hydrothermal synthesis for enhanced wastewater purification and antibacterial treatment. Environmental Research 2024, 259, 119534. [Google Scholar] [CrossRef]
- Lee, G.J.; and Wu, J.J. Recent developments in ZnS photocatalysts from synthesis to photocatalytic applications — A review. Powder Technology 2017, 318, 8–22. [Google Scholar] [CrossRef]
- Sonawane, H.; Deore, J.; Rajshri, S.; and Chavan, P. Synthesis of ZnS Nanomaterials and Their Applications via Green Approaches: An Overview. BioNanoScience 2023, 13, 879–890. [Google Scholar] [CrossRef]
- Chakrabarti, A; and Alessandri, E. Syntheses, Properties, and Applications of ZnS-Based Nanomaterials. Appl. Nano 2024, 5, 116–142. [CrossRef]
- Lee, K.T.; Choi, B.H.; Woo, J.U.; Kang, J.S.; Paik, J.H.; Byoung-Uck Chu, B.U.; and Nahm, S. Microstructural and optical properties of the ZnS ceramics sintered by vacuum hot-pressing using hydrothermally synthesized ZnS powders. Journal of the European Ceramic Society 2018, 38(12), 4237–4244. [Google Scholar] [CrossRef]
- Sadovnikov, S.I.; Ishchenko, A.V. ; and Weinstein,I. A. Synthesis and optical properties of nanostructured ZnS and heteronanostructures based on zinc and silver sulfides. Journal of Alloys and Compounds 2020, 831, 154846. [Google Scholar] [CrossRef]
- Khan, J.A.; Ahamad, S.; Ansari, M.A.H.; Tauqeer, M.; Park, C.H.; Park, J.P.; Choi. C.H.; and Mohammad, A. State-of-the-art in ZnS-based nanoarchitects for visible-light photocatalytic degradation of antibiotics and organic dyes. Journal of Water Process Engineering 2024, 67, 106151. [CrossRef]
- Navaneethan, M.; Archana, J.; Nisha, K.D.; Hayakawa, Y.; Ponnusamy, S.; and Muthamizhchelvan, C. Temperature dependence of morphology, structural and optical properties of ZnS nanostructures synthesized by wet chemical route. Journal of Alloys and Compounds 2010, 506, 249–252. [Google Scholar] [CrossRef]
- Yin, L.; Zhang, D.; Wang, D.; Kong, X.; Huang, J.; Wang, F.; and Wu, Y. Size dependent photocatalytic activity of ZnS nanostructures prepared by a facile precipitation method. Materials Science and Engineering: B 2016, 208, 15 –21. [CrossRef]
- Shobana, T.; Venkatesan, T.; and Kathirvel, D. A Comprehensive Review on Zinc Sulphide Thin Film by Chemical Bath Deposition Techniques. J. Environ. Nanotechnol 2020, 9(1), 50–59. [Google Scholar] [CrossRef]
- Arsad, A.Z.; Zuhdi, A.W.M.; Abdullah, F.A.; Chau, C.F.; Ghazali, A.; Ahmad, I.; and Abdullah, W.S.W. Effect of Chemical Bath Deposition Variables on the Properties of Zinc Sulfide Thin Films: A Review. Molecules 2023, 28, 2780. [Google Scholar] [CrossRef]
- Ali, A.H.; Hashem, H.A.; and Elfalaky, A. Preparation, Properties, and Characterization of ZnS Nanoparticles. Eng. Proc. 2023, 31, 74. [Google Scholar] [CrossRef]
- SHAHJAHAN, M.; KABIR, R.; HOSSAIN, M.S.; HAQUE, M.A.; and Prasad PAUL, D. EFFECT OF pH ON OPTICAL PROPERTIES OF NANOSTRUCTURED Cu-DOPED ZnS THIN FILMS FOR PHOTOVOLTAIC APPLICATIONS. EUROPEAN JOURNAL OF MATERIALS SCIENCE AND ENGINEERING 2024, 8 (4). 226-239. [CrossRef]
- AlShamaileh, E.; Lahlouh, B.; AL-Masri, A.N.; Al-Qderat, M.; Wadah Mahmoud, W.; Alrbaihat, M.; and Moosa, I.S. Comparative Microstructure Characteristics of Synthesized PbS Nanocrystals and Galena. Sci 2024, 6, 61. [Google Scholar] [CrossRef]
- Ebnelwaled, A. A; Elsheikh, H.H.A; and Hamed, Z.H. Synthesis and characterization of ZnS nanoparticles by chemical precipitation method. Aswan University Journal of Environmental Studies (AUJES) 2021, 2(2), 147–154. [Google Scholar] [CrossRef]
- Nagamani, K.; Prathap, P.; Lingappa, Y.; Miles, R.W.; and Reddy, K.T.R. Properties of Al-doped ZnS Films Grown by Chemical Bath Deposition. Physics Procedia 2012, 25, 137–142. [Google Scholar] [CrossRef]
- Shakil, M.A.; Sangita Das, Rahman, M.A.; Akther, U.S.; Majumdar, M.K.H.; and Rahman, M.K. A Review on Zinc Sulphide Thin Film Fabrication for Various Applications Based on Doping Elements. Materials Sciences and Applications 2018, 9, 751–778. [CrossRef]
- Ahn, H.; and Um, Y. Post-annealing Effects on ZnS Thin Films Grown by Using the CBD Method. Journal of the Korean Physical Society 2015, 67(6), 1045–1050. [Google Scholar] [CrossRef]
- Karimi, M.; Mirkazemi, S.M.; Vahidshad, Y.; and Javadpour, J. Preparation and characterization of zinc sulfide thin film by electrostatic spray deposition of nano-colloid. Thin Solid Films 2021, 737(1), 138929. [Google Scholar] [CrossRef]
- Faita, F.L.; Ersching, K.; Poffo, C.M.; Benetti, L.C.; Trichês, D.M.; Souza, S.M.; Viegas, A.D.C.; and de Lima, J.C. Structural, thermal, magnetic and optical characterization of undoped nanocrystalline ZnS prepared by solid state reaction. Journal of Alloys and Compounds 2014, 590, 176 –183. [CrossRef]
- Krystýnová, M.; Doležal, P.; Fintová, S.; Březina, M.; Zapletal, J.; and Wasserbauer, J. Preparation and Characterization of Zinc Materials Prepared by Powder Metallurgy. Metals 2017, 7, 396. [Google Scholar] [CrossRef]
- Petersen, H.; Reichle, S.; Leiting, S.; Losch, P.; Kersten, W.; Rathmann, T.; Tseng, J.; Etter, M.; Schmidt, W.; and Weidenthaler, C. In Situ Synchrotron X-ray Diffraction Studies of the Mechanochemical Synthesis of ZnS from its Elements. Chem. Eur. J. 2021, 27, 12558–12565. [Google Scholar] [CrossRef] [PubMed]
- Moosa. I.S.; Kazem, H.A.; and Al-Iessi, L.M.R. Production of Hydrogen via Renewable Energy and Investigation of Water Molecular Changes During Electrolysis Process. JREE: 2021, 8 (4) 19-28. [CrossRef]
- Labiadh, H.; Moualhi,Y.; Moualhi, K.; Abdelhak Othmani, A.; Zouaoui, M. Synthesis of ZnS nanoparticles and the investigation of their structural, optical and electrical properties. Euro-Mediterr J Environ Integr 2025, 10, 119–130. [CrossRef]
- Chankhanittha, T.; Watcharakitti, J.; Piyavarakorn, V.; Johnson, B.; Bushby, R.J.; Chuaicham, C.; Sasaki, K.; Nijpanich, S.; Nakajima, H.; Chanlek, N.; and Nanan, S. ZnO/ZnS photocatalyst from thermal treatment of ZnS: Influence of calcination temperature on development of heterojunction structure and photocatalytic performance. Journal of Physics and Chemistry of Solids 2023, 179, 111393. [Google Scholar] [CrossRef]
- Yang, K.; Li, B.; and Zeng, G. Effects of temperature on properties of ZnS thin films deposited by pulsed laser deposition. Superlattices and Microstructures 2019, 130, 409–415. [Google Scholar] [CrossRef]
- Al-Saqarat, B.S.; Al-Mobydeen, A.; Al-Dalahmeh, Y.; AL-Masri, A.N.; Altwaiq, A.M.; Hamadneh, I.; Abu-Afifeh, Q.; Zoubi, M.M.; Esaifan, M.; and Moosa, I.S. Study of Galena Ore Powder Sintering and Its Microstructure. Metals 2024, 14, 439. [Google Scholar] [CrossRef]
- Grayeli, A.; Sadeghi, M.; Shakoury, R.; Matos, R.S.; Filho, H.D.F; Arman, A. Structural, morphological, and optical bandgap properties of ZnS thin films: a case study on thickness dependence. Optical and Quantum Electronics 2024, 56, 1142. [Google Scholar] [CrossRef]
- Grayeli, A.; Sadeghi, M.; Shakoury, R.; Matos, R.S.; Filho, H.D.F.; and Arman, A. Structural, morphological, and optical bandgap properties of ZnS thin films: a case study on thickness dependence. Optical and Quantum Electronics 2024, 56, 1142. [Google Scholar] [CrossRef]
- Jafarov, M.A.; Nasirov, E.F.; Jahangirova, S.A.; and Jafarli, R. Nano-ZnS thin films for solar cell. NANOSYSTEMS: PHYSICS, CHEMISTRY, MATHEMATICS 2015, 6 (5), 644–649. [CrossRef]
- Choudapura, V.H.; Kapatkara, S.B.; and Rajub, A.B. STRUCTURAL AND OPTOELECTRONIC PROPERTIES OF ZINC SULFIDE THIN FILMS SYNTHESIZED BY CO-PRECIPITATION METHOD. ACTA CHEMICA IASI 2019, 2, 287–302. [Google Scholar] [CrossRef]
- Djelloul, A.; Adnane, M.; Larbah, Y.; Sahraoui, T.; Zegadi, C.; Maha, A.; and Rahal, B. Properties Study of ZnS Thin Films Prepared by Spray Pyrolysis Method. JOURNAL OF NANO- AND ELECTRONIC PHYSICS 2015, 7 (4), 04045. https://jnep.sumdu.edu.ua/download/numbers/2015/4/articles/jnep_2015_V7_04045.pdf.
- NADEEM, M.Y.; and AHMED, W. Optical Properties of ZnS Thin Films. Turkish Journal of Physics 2000, 24, 5, 651 -659. https://journals.tubitak.gov.tr/physics/vol24/iss5/6.
- Alzaid, M.; Mohamed, W.S.; El-Hagary, M.; Shaaban, E.R.; and Hadia, N.M.A. Optical properties upon ZnS film thickness in ZnS/ITO/glass multilayer films by ellipsometric and spectrophotometric investigations for solar cell and optoelectronic applications. Optical Materials 2021, 118, 111228. [Google Scholar] [CrossRef]
- Jothibas, M.; Jeyakumar, S. J.; Manoharan, C.; Punithavathy, I.K.; Praveen, P.; and Richard, J.P. Structural and optical properties of zinc sulphide nanoparticles synthesized via solid state reaction method. J Mater Sci: Mater Electron 2017, 28, 1889–1894. [CrossRef]
- Mani, S.K.; Manickam, S.; Muthusamy, V.; and Thangaraj, R. Antimicrobial Activity and Photocatalytic Degradation Properties of Zinc Sulfide Nanoparticles Synthesized by Using Plant Extracts. J Nanostruct 2018, 8(2), 107–118. [Google Scholar]
- Tounsi, A.; Talantikite-Touati, D.; Merzouk, H.; Haddad, H.; and Khalfi, R. Study of the properties of La-doped ZnS thin Films synthesized by Sol-gel method. Advanced Materials Letters, 2018, 9 (3) 216-219. [CrossRef]
- Włodarski, M.; Putkonen, M.; and Norek, M. Infrared Absorption Study of Zn–S Hybrid and ZnS Ultrathin Films Deposited on Porous AAO Ceramic Support. Coatings 2020, 10, 459. [Google Scholar] [CrossRef]
- Hameed, O.M. Synthesis of Zinc Sulfide Nanocrystals by Chemical Bath Deposition Methods. E3S Web of Conferences 2023, 391, 01134. ICMED-ICMPC 2023. [CrossRef]
- Hameed, O.M. Synthesis of Zinc Sulfide Nanocrystals by Chemical Bath Deposition Methods. E3S Web of Conferences 2023, 391, 01134. [Google Scholar] [CrossRef]
- Khosravi, S.; Chaibakhsh, N.; S. Jafari, S.; and M. Nilkar, M. Enhanced photocatalytic activity of ZnS/TiO2 nanocomposite by nitrogen and tetrafluoromethane plasma treatments. Scientific Reports 2024, 14, 28385. [CrossRef]
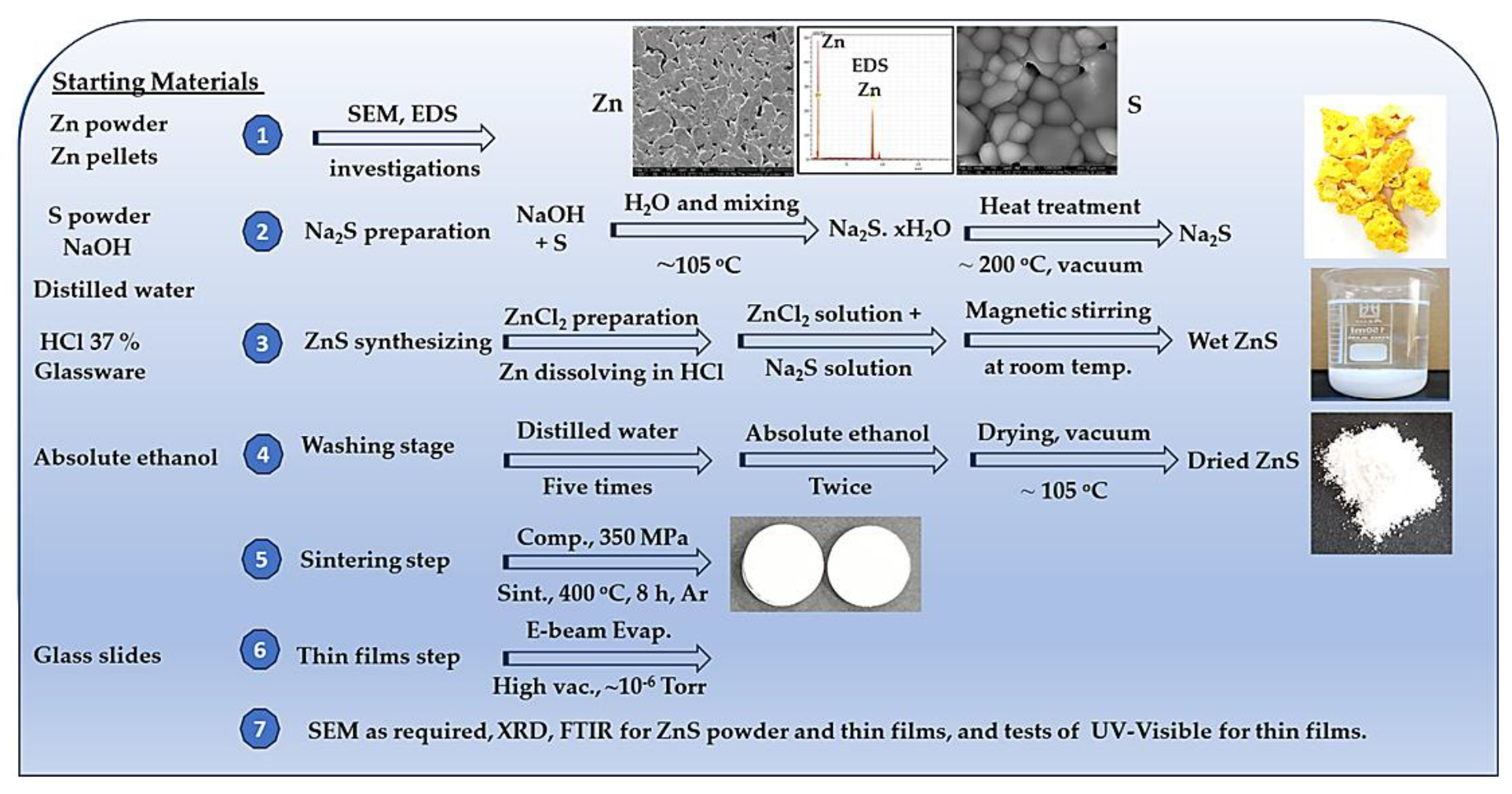


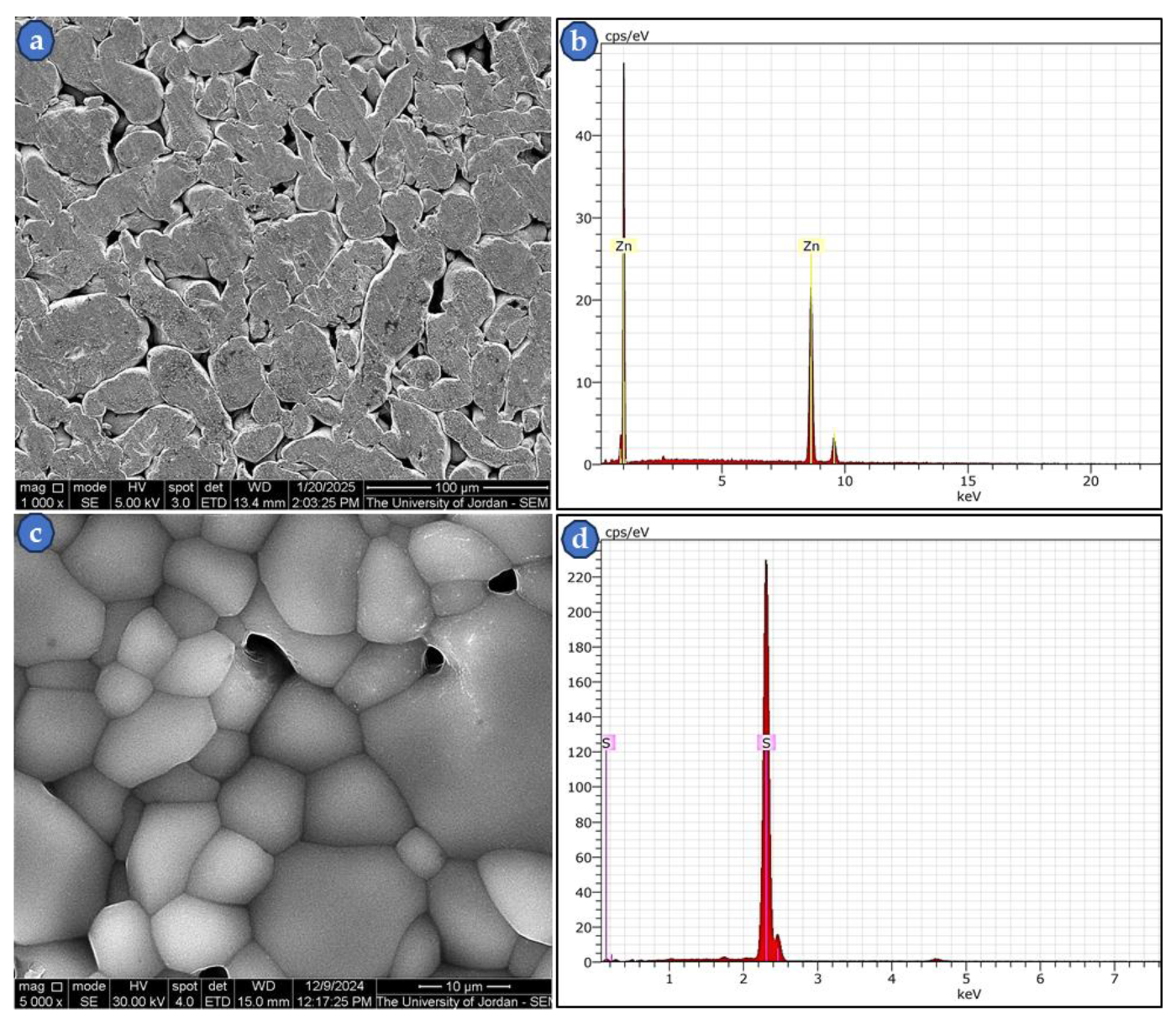
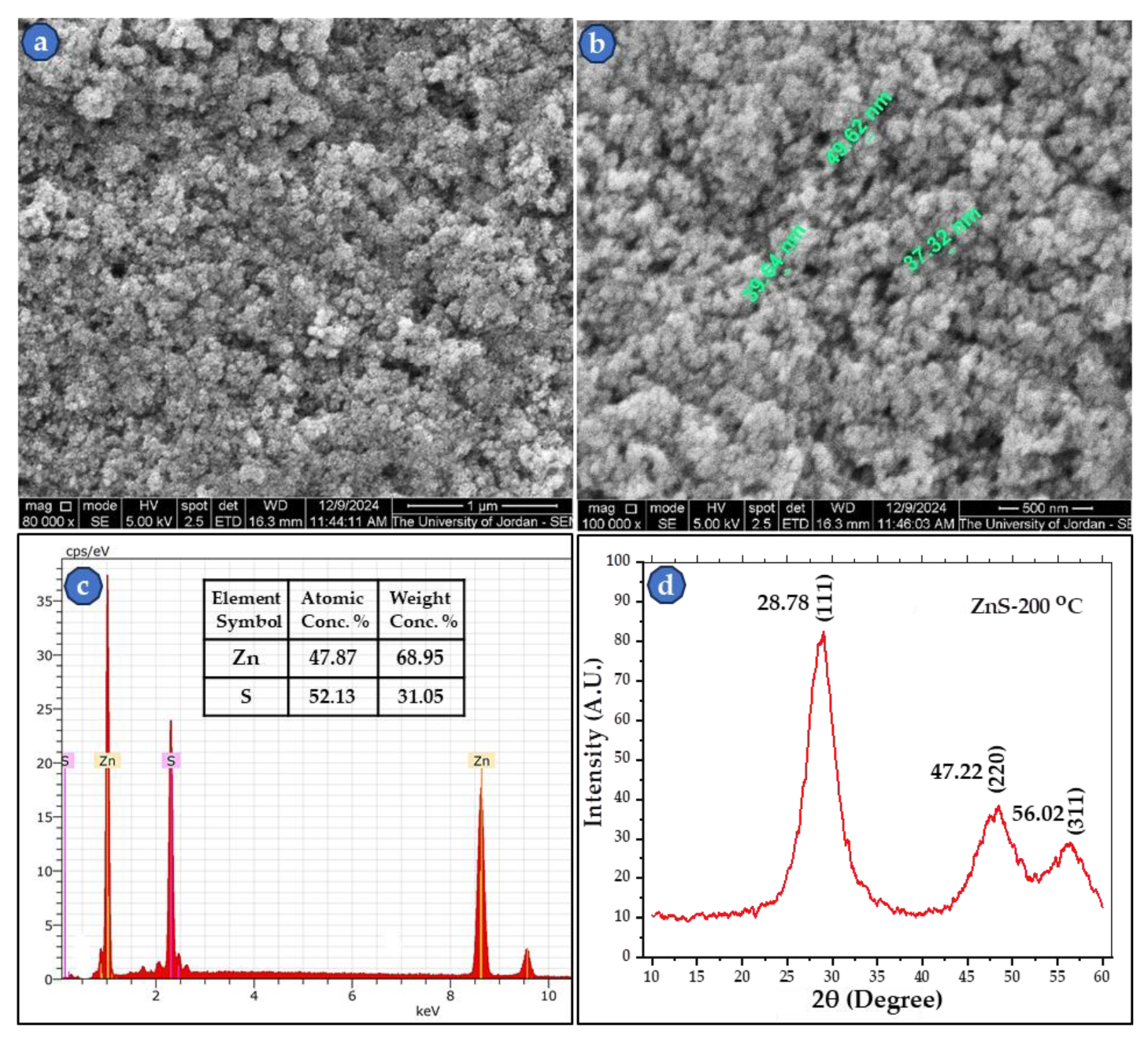
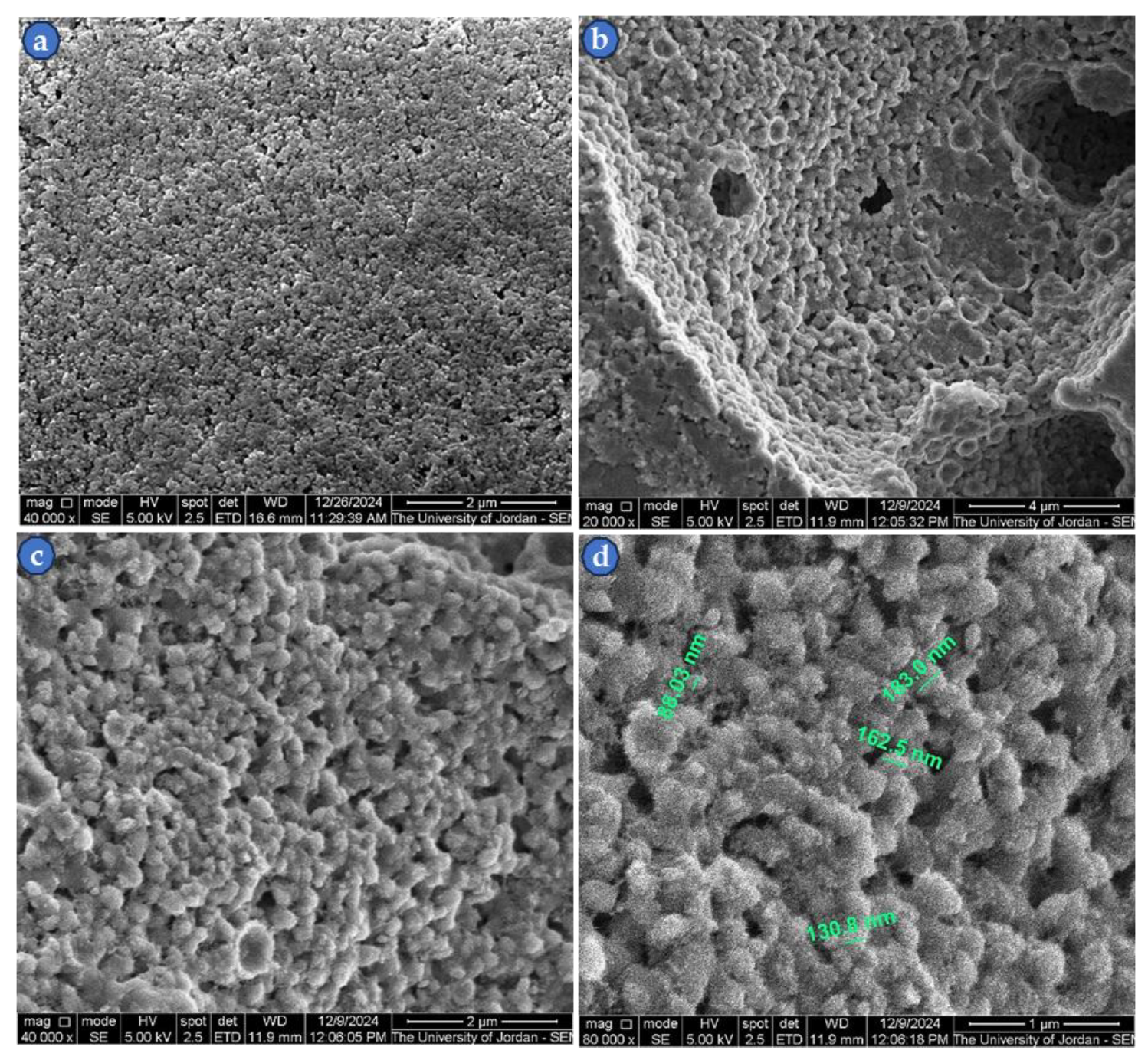

| Sample No. | ρg. (g/cm3) | ρs. (g/cm3) | Compaction and sintering conditions |
| 1 | 2.64 | 3.19 | Compaction pressure 400 MPa, sintering temp. 400 oC for 8h under an Argon atmosphere with a flow rate of approximately 1 L/min., heating rates of 5 oC/min., furnace cool. |
| 2 | 2.61 | 3.15 | |
| 3 | 2.69 | 3.32 | |
| 4 | 2.61 | 3.18 | |
| Mean Value | 2.64 | 3.21 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




