Submitted:
04 March 2025
Posted:
05 March 2025
You are already at the latest version
Abstract
Background/Objectives: Contrast-enhanced mammography (CEM) integrates morphological and functional imaging using contrast agents, making it a valuable tool in breast cancer (BC) diagnosis. In 2022, the Breast Imaging Reporting and Data System for CEM was introduced. This study aims to investigate the relationship between CEM morphodynamic features and the histopathological characteristics of BC. Methods: In this prospective study were consecutively enrolled, between December 2022 and May 2024, patients with BI-RADS 4 or 5 lesions undergoing CEM followed by surgical excision. Low-energy and recombined CEM images were analysed for breast composition, lesion characteristics, and enhancement patterns. Histopathological evaluation included tumor size, histotype, grade, lymphovascular invasion and immunophenotype. Spearman rank correlation and multivariable regression analysis were used to evaluate the relationship between CEM findings and histopathological characteristics. Results: Fifty female patients (mean age: 57.2 ± 13.7 years) and 50 BCs were analysed. Tumor dimensions observed on CEM showed a strong correlation with histopathological tumor size (ρ = 0.788, p < 0.001) and were associated with grade 3 lesions (p = 0.017). Not circumscribed margins on CEM were significantly associated with the Luminal-B (p = 0.001), while high lesion conspicuity was associated with Luminal-B and triple-negative (p = 0.001) and correlated with larger tumor size (ρ = 0.517, p < 0.001). Background parenchymal enhancement negatively correlated with patient age (ρ = -0.286, p = 0.049). Conclusions: CEM provides critical insights into BC morphology and biology, demonstrating significant relationship between imaging features and histopathological characteristics.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Image Acquisition
2.3. Reader Study
2.4. Histopathological Evaluation
2.5. Statistical Analysis
3. Results
3.1. Study Population
3.2. CEM Findings
3.2. Histopathological Findings
3.2. Relationship between CEM findings and histopathological characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sibylle, L.; Philip, P.; Monica, M.; et al. Breast cancer. Lancet (London, England) 2021, 397, 1750–1769.
- Jong, R.A.; Yaffe, M.J.; Skarpathiotakis, M.; Bloomquist, A.; Lockwood, G.; Plewes, D.B.; et al. Contrast-enhanced digital mammography: Initial clinical experience. Radiology 2003, 228, 842–850. [CrossRef]
- Ventura, C.; Baldassarre, S.; Cerimele, F.; Pepi, L.; Marconi, E.; Ercolani, P. et al. 2D shear wave elastography in evaluation of prognostic factors in breast cancer. Radiologia Medica 2022, 127, 1221–1227. [CrossRef]
- Lobbes, M.B.I.; Smidt, M.L.; Houwers, J.; Tjan-Heijnen, V.C.; Wildberger, J.E.; et al. Contrast-enhanced mammography: Techniques, current results, and potential indications. Clinical Radiology 2013, 68, 935–944. [CrossRef]
- Diekmann, F.; Freyer, M.; Diekmann, S.; Stengel, A.; Fischer, T.; Bick, U. Evaluation of contrast-enhanced digital mammography. European Journal of Radiology 2011, 78, 112–121. [CrossRef]
- Polat, D.S.; Evans, W.P.; Dogan, B.E.; Chen, H.L.; Burgess, M.; Paulsen, L.; et al. Contrast-enhanced digital mammography: Technique, clinical applications, and pitfalls. American Journal of Roentgenology 2020, 215, 1267–1278. [CrossRef]
- Covington, M.F.; Salmon, S.; Weaver, B.D.; Fajardo, L.L.; Rhodes, D.J. State-of-the-art for contrast-enhanced mammography. British Institute of Radiology 2024, 97, 695–704.
- Ainakulova, A.S.; Zholdybay, Z.Z.; Kaidarova, D.R.; Baimukhanova, A.B.; Nurtayeva, S.; Tulegenov, A.; et al. Contrast-enhanced spectral mammography without and with a delayed image for diagnosing malignancy among mass lesions in dense breast. Wspolczesna Onkologia 2021, 25, 17–22. [CrossRef]
- Sorin, V.; Yagil, Y.; Yosepovich, A.; Hershkovitz, D.; Ben-Baruch, G.; Khashper, A.; et al. Contrast-enhanced spectral mammography in women with intermediate breast cancer risk and dense breasts. American Journal of Roentgenology 2018, W267–W274.
- Bernardi, D.; Vatteroni, G.; Acquaviva, A.; Monti, S.; Bariani, M.; Petrillo, A. Contrast-enhanced mammography versus MRI in the evaluation of neoadjuvant therapy response in patients with breast cancer: A prospective study. American Journal of Roentgenology 2022, 219, 884–894. [CrossRef]
- Jochelson, M.S.; Lobbes, M.B.I.; Scaranelo, A.M.; Margolies, L.R.; Gilbert, F.J.; Lee, C.H. Contrast-enhanced mammography: State of the art. Radiology 2021, 299, 36–48.
- Pötsch, N.; Vatteroni, G.; Clauser, P.; Baltzer, P.A.T.; Helbich, T.H. Contrast-enhanced mammography versus contrast-enhanced breast MRI: A systematic review and meta-analysis. Radiology 2022, 305, 94–103.
- Lee, C.H.; Phillips, J.; Sung, J.S.; Giess, C.S.; Jochelson, M.S.; Margolies, L.R. ACR BI-RADS® atlas-mammography contrast enhanced mammography (CEM). American College of Radiology 2022.
- Nicosia, L.; Battaglia, O.; Venturini, M.; Pellegrino, S.; Priola, S.; Bernardi, D. Contrast-enhanced mammography BI-RADS: A case-based approach to radiology reporting. Insights Imaging 2024, 15.
- Magni, V.; Cozzi, A.; Muscogiuri, G.; Pellegrino, S.; Nicosia, L.; De Marco, P. Background parenchymal enhancement on contrast-enhanced mammography: Associations with breast density and patient characteristics. Radiologia Medica 2024, 129, 1303–1312. [CrossRef]
- Petrillo, A.; Fusco, R.; Petrosino, T.; Bianchi, F.; Ferrara, D.; Barone, C. A multicentric study of radiomics and artificial intelligence analysis on contrast-enhanced mammography to identify different histotypes of breast cancer. Radiologia Medica 2024, 129, 864–878. [CrossRef]
- Bartolotta, T.V.; Militello, C.; Prinzi, F.; La Forgia, D.; Sarli, M.; Di Paolo, A.; et al. Artificial intelligence-based, semi-automated segmentation for the extraction of ultrasound-derived radiomics features in breast cancer: A prospective multicenter study. Radiologia Medica 2024, 129, 977–988. [CrossRef]
- Migliaro, G.; Bicchierai, G.; Valente, P.; Amato, F.; Vanzi, B.; Ventura, C. Contrast enhanced mammography (CEM) enhancing asymmetry: Single-center first case analysis. Diagnostics 2023, 13. [CrossRef]
- Li, N.; Gong, W.; Xie, Y.; Sheng, L. Correlation between the CEM imaging characteristics and different molecular subtypes of breast cancer. Radiology 2023.
- Fogante, M.; Tagliati, C.; De Lisa, M.; Berardi, R.; Giuseppetti, G.M.; Giovagnoni, A. Correlation between apparent diffusion coefficient of magnetic resonance imaging and tumor-infiltrating lymphocytes in breast cancer. Radiologia Medica 2019, 124, 581–587. [CrossRef]
- Prochowski Iamurri, A.; Ponziani, M.; Macchini, M.; Fogante, M.; Pistelli, M.; De Lisa, M.; et al. Evaluation of multifocality and multicentricity with breast magnetic resonance imaging in each breast cancer subtype. Clinical Breast Cancer 2018, 18, e231–e235. [CrossRef]
- Soylu Boy, F.N.; Goksu, K.; Tasdelen, I. Association between lesion enhancement and breast cancer in contrast-enhanced spectral mammography. Acta Radiologica 2023, 64, 74–79. [CrossRef]
- Marzogi, A.; Baltzer, P.A.T.; Kapetas, P.; Milos, R.I.; Bernathova, M.; Helbich, T.H.; et al. Is the level of contrast enhancement on contrast-enhanced mammography (CEM) associated with the presence and biological aggressiveness of breast cancer? Diagnostics 2023, 13, 754. [CrossRef]
- Travieso-Aja, M.M.; Naranjo-Santana, P.; Fernández-Ruiz, C.; Rodríguez-Abreu, D.; García-Hernández, N. Factors affecting the precision of lesion sizing with contrast-enhanced spectral mammography. Clinical Radiology 2018, 73, 296–303. [CrossRef]
- Nicosia, L.; Bozzini, A.C.; Palma, S.; Mantini, G.; Marini, P.; Pruneri, G. Contrast-enhanced spectral mammography and tumor size assessment: A valuable tool for appropriate surgical management of breast lesions. Radiologia Medica 2022, 127, 1228–1234. [CrossRef]
- Cozzi, A.; Schiaffino, S.; Sardanelli, F. The emerging role of contrast-enhanced mammography. Quantitative Imaging in Medicine and Surgery 2019, 9, 2012–2018.
- Bozzini, A.; Nicosia, L.; Pruneri, G.; Martella, E.; Testori, A.; Mastropasqua, M.G.; et al. Clinical performance of contrast-enhanced spectral mammography in pre-surgical evaluation of breast malignant lesions in dense breasts: A single-center study. Breast Cancer Research and Treatment 2020, 184, 723–731. [CrossRef]
- Wessam, R.; Gomaa, M.; Fouad, M.A.; Omar, W.; Mohamed, M. Added value of contrast-enhanced mammography in assessment of breast asymmetries. Clinical Radiology 2019.
- Magni, V.; Cozzi, A.; Muscogiuri, G.; Pellegrino, S.; Nicosia, L.; De Marco, P. Background parenchymal enhancement on contrast-enhanced mammography: Associations with breast density and patient characteristics. Radiologia Medica 2024, 129, 1303–1312. [CrossRef]
- Dominique, C.; Callonnec, F.; Berghian, A.; Foucher, G.; Mari, S.; Lemonnier, P.; et al. Deep learning analysis of contrast-enhanced spectral mammography to determine histoprognostic factors of malignant breast tumours. European Radiology 2022, 32, 4834–4844. [CrossRef]
- Petrillo, A.; Fusco, R.; Di Bernardo, E.; De Luca, L.; Di Giuda, D.; Petrosino, T.; et al. Prediction of breast cancer histological outcome by radiomics and artificial intelligence analysis in contrast-enhanced mammography. Cancers 2022, 14, 2132. [CrossRef]
- Piccolo, C.L.; Sarli, M.; Pileri, M.; Costa, F.; Chiarenza, M.; Tagliati, C. Radiomics for predicting prognostic factors in breast cancer: Insights from contrast-enhanced mammography (CEM). Journal of Clinical Medicine 2024, 13, 6486. [CrossRef]
- La Forgia, D.; Fanizzi, A.; Campobasso, F.; Di Palma, D.; Di Giorgio, L.; Rizzi, F.; et al. Radiomic analysis in contrast-enhanced spectral mammography for predicting breast cancer histological outcome. Diagnostics 2020, 10, 0708. [CrossRef]
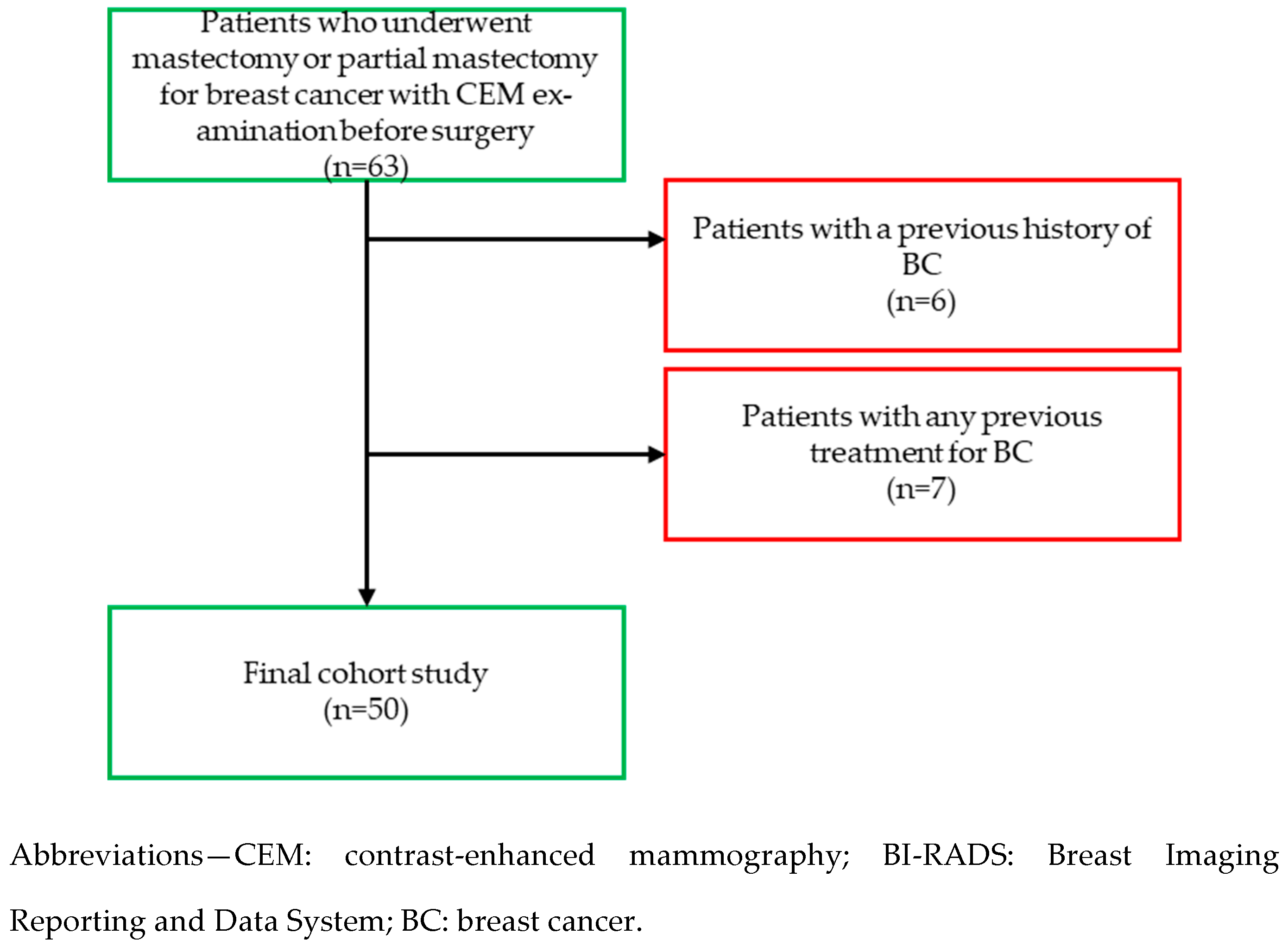
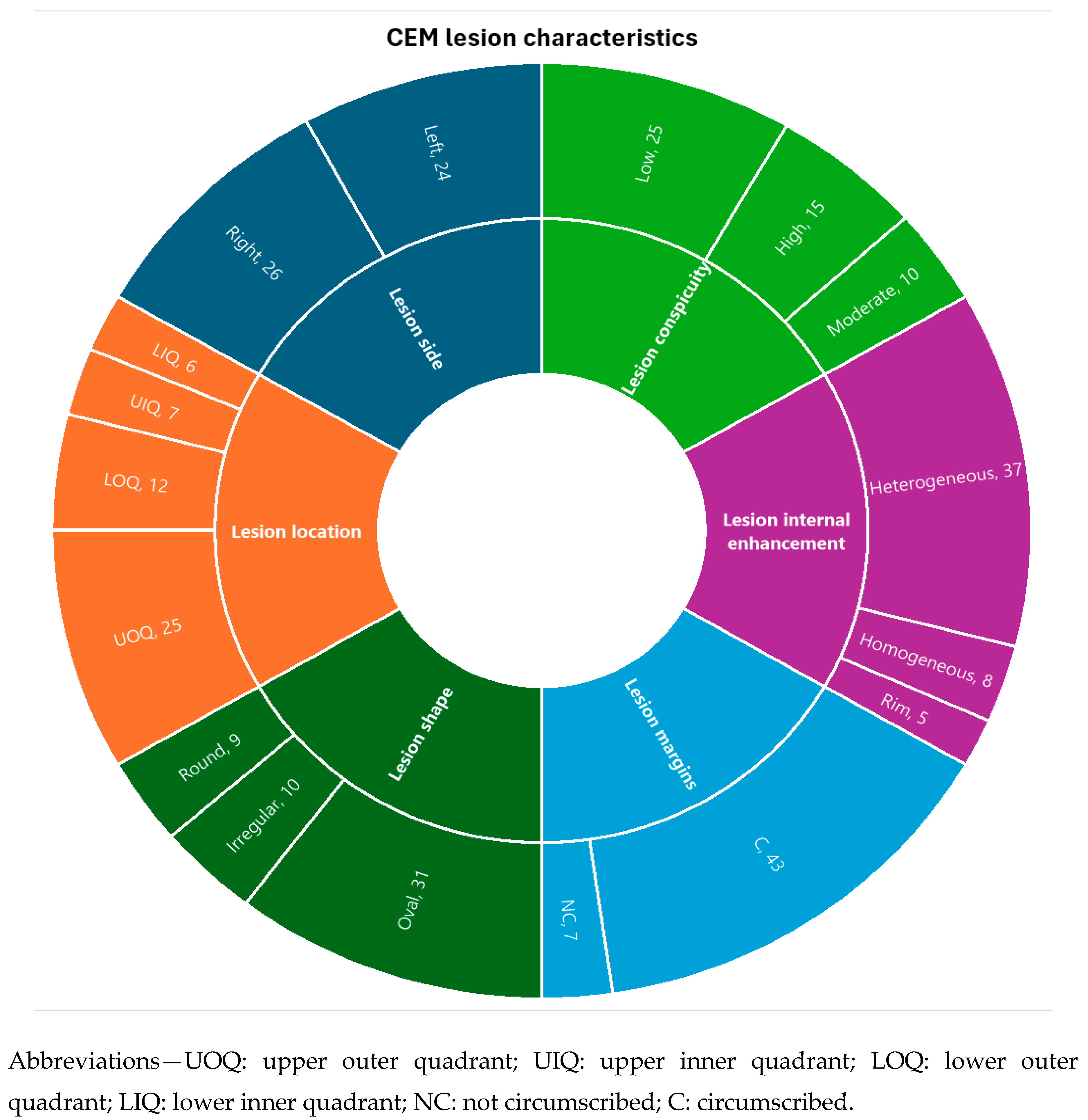
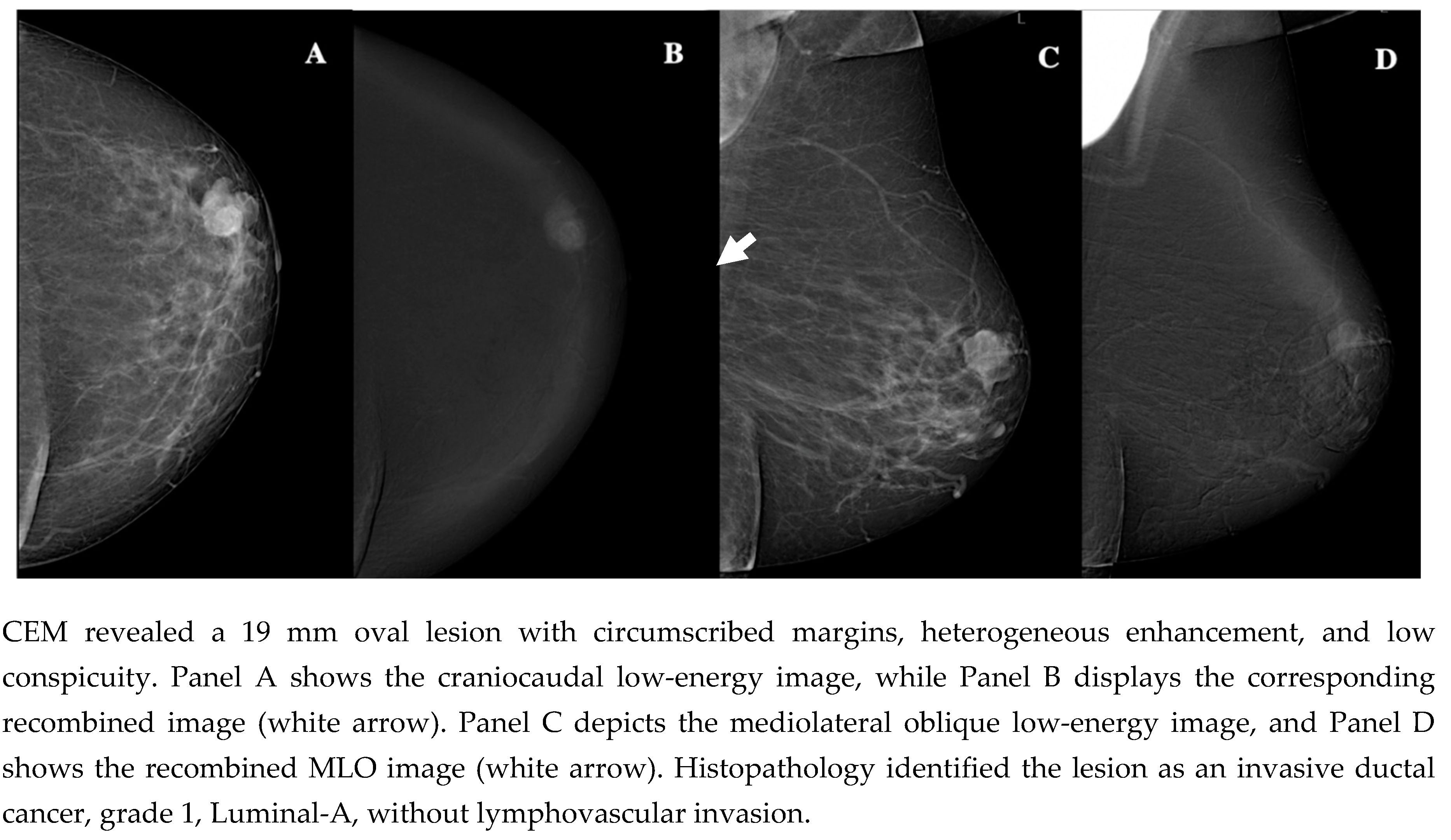
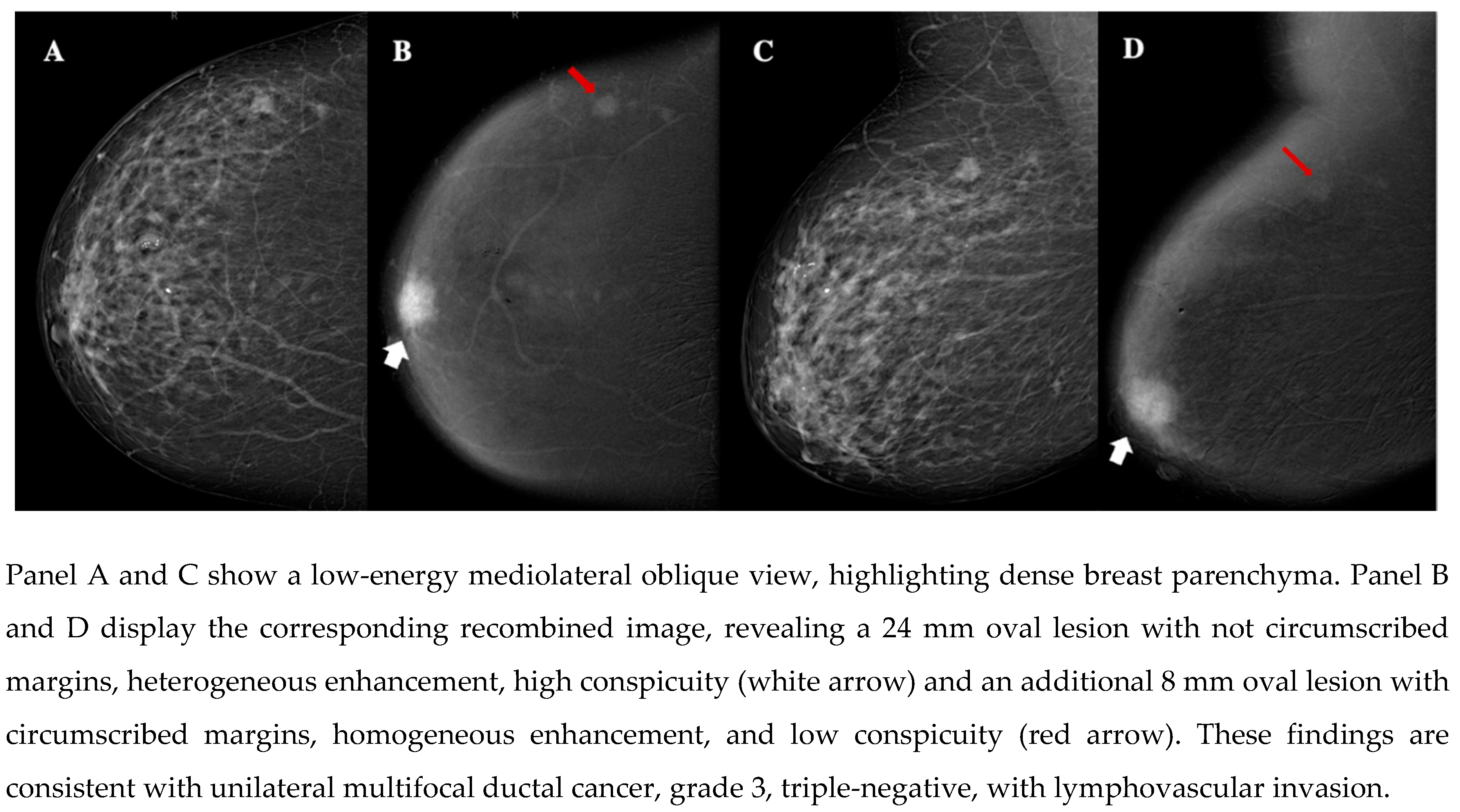
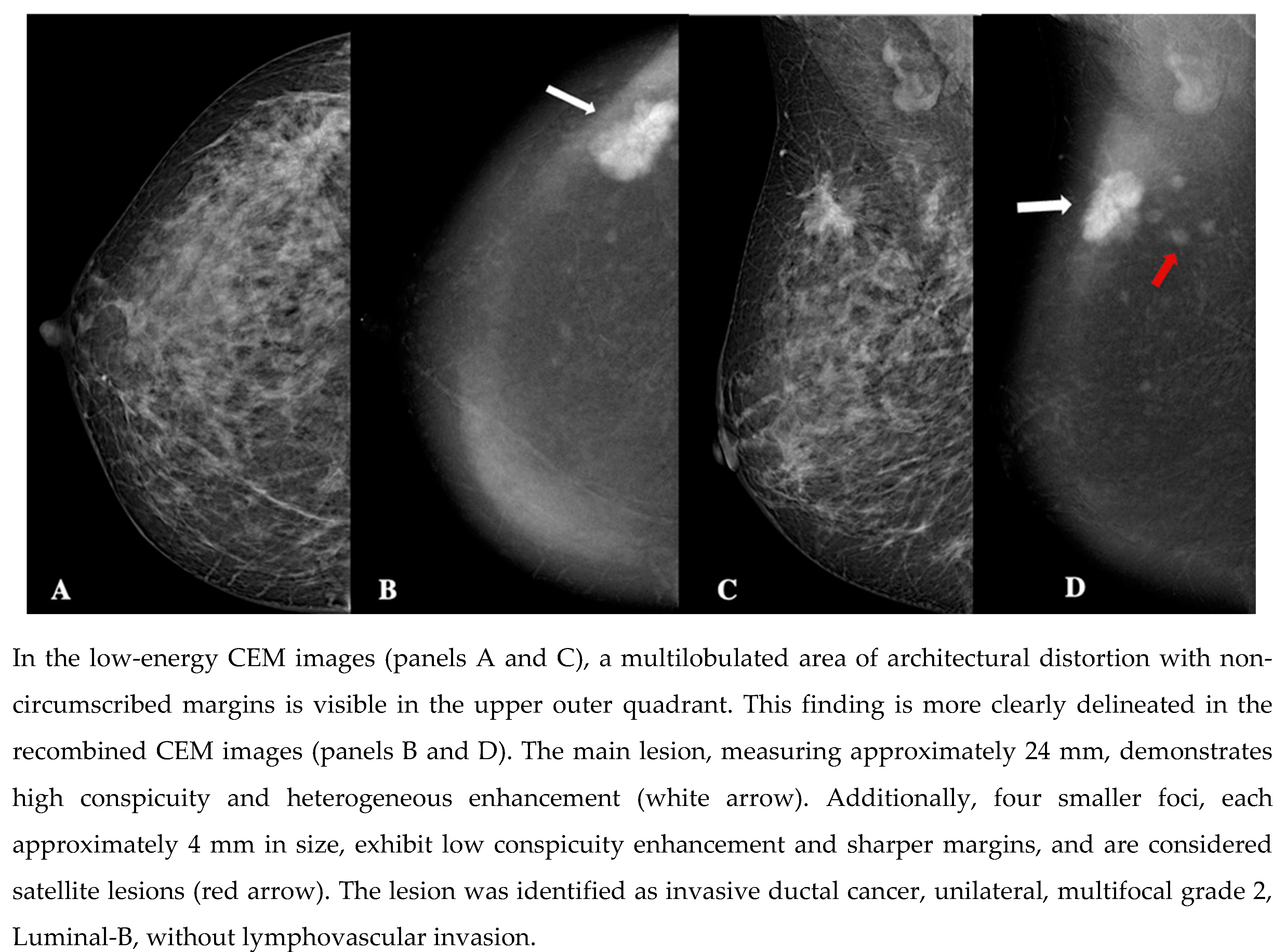
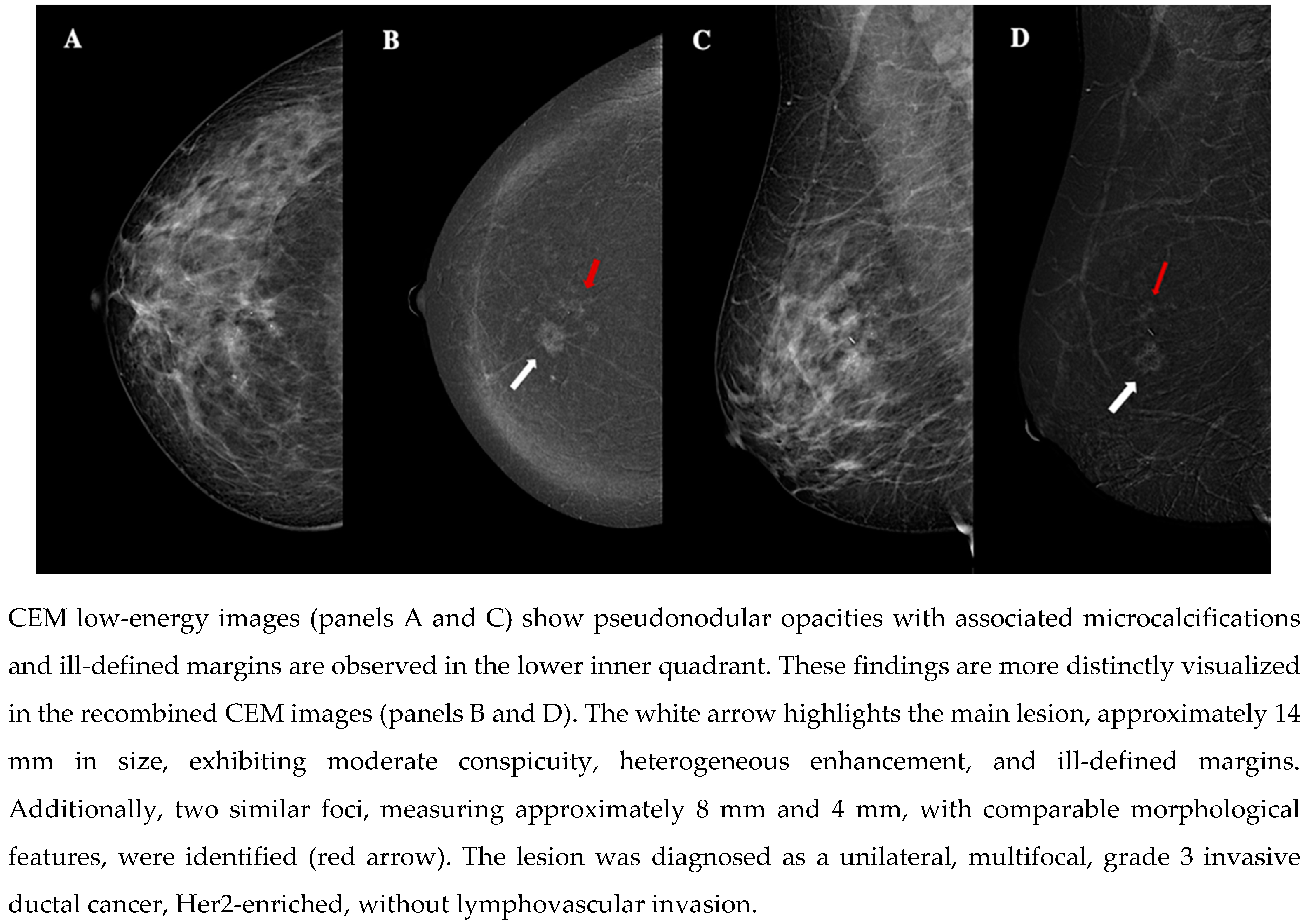
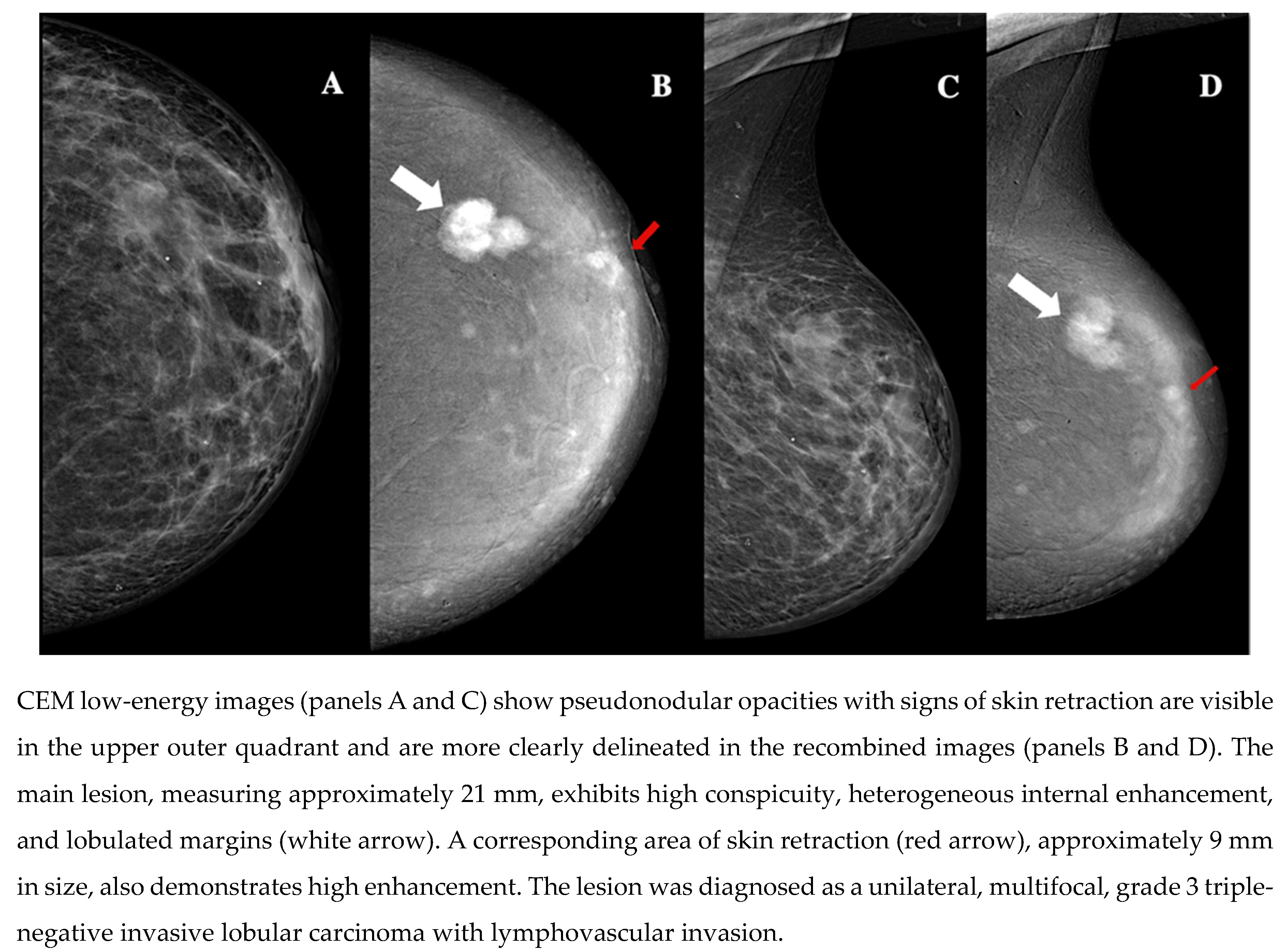
| Demographic characteristics | Age (year, mean ± SD) | 57.2 ± 13.7 |
| Body mass index (kg/m2, mean ± SD) | 23.7 ± 8.1 | |
| Clinical characteristics | Presence of palpable nodule (n, %) | 13 (26%) |
| Presence of nipple secrection (n, %) | 3 (6%) | |
| Presence of retraction (n, %) | 2 (4%) | |
| Presence of cutaneous retraction (n, %) | 3 (6%) | |
| Presence of palpable adenopathy (n, %) | 4 (8%) | |
| Family history of breast cancer (n, %) | 17 (34%) |
| Breast composition | A (almost entirely fatty) (n, %) | 4 (8%) |
| B (scattered fibroglandular areas) (n, %) | 30 (60%) | |
| C (heterogeneously dense) (n, %) | 14 (28%) | |
| D (extremely dense) (n, %) | 2 (4%) | |
| Microcalcifications | Present (n, %) / Absent (n, %) | 11 (22%) / 39 (78%) |
| Architectural distorsion | Present (n, %) / Absent (n, %) | 34 (68%) / 16 (32%) |
| Mean lesion dimension | Size (mm—mean ± SD) | 23.4 ± 10.9 |
| Lesion side | Right (n,%) / Left (n, %) | 26 (62%) / 24 (58%) |
| Lesion location | UOQ (n,%) / UIQ (n,%) | 25 (50%) / 7 (14%) |
| LOQ (n,%) / LIQ (n,%) | 12 (24%) / 6 (12%) | |
| Lesion shape | Oval (n, %) / Round (n, %) / Irregular (n,%) | 31 (62%) / 9 (18%) / 10 (20%) |
| Lesion margins | NC (n, %) / C (n, %) | 7 (14%) / 43 (86%) |
| Lesion internal enhancement | Homogeneous (n, %) | 8 (16%) |
| Heterogeneous (n, %) | 37 (74%) | |
| Rim enhancement (n, %) | 5 (10%) | |
| Lesion enhancement conspicuity | Low (n, %) | 25 (50%) |
| Moderate (n, %) | 10 (20%) | |
| High (n, %) | 15 (30%) | |
| Associated Features | Nipple retraction (n, %) | 4 (8%) |
| Nipple invasion (n, %) | 2 (4%) | |
| Skin retraction (n, %) | 2 (4%) | |
| Skin thickening (n, %) | 3 (6%) | |
| Skin invasion (n, %) | 9 (18%) | |
| Axillary adenopathy (n, %) | 3 (6%) | |
| Background parenchymal enhancement | Minimal (n, %) | 26 (52%) |
| Mild (n, %) | 15 (30%) | |
| Moderate–Marked (n, %) | 9 (18%) |
| Mean lesion dimension | Size (mm) (mean ± SD) | 23.7 ± 14.3 |
| Histological phenotype | Ductal (n, %) | 36 (72%) |
| Lobular (n, %) | 14 (28%) | |
| Grading (Elston–Ellis) | Grade 1 (n, %) | 7 (14%) |
| Grade 2 (n, %) | 25 (50%) | |
| Grade 3 (n, %) | 18 (36%) | |
| Immunophenotype | Luminal-A (n, %) | 18 (36%) |
| Luminal-B (n, %) | 26 (52%) | |
| HER2-enriched (n, %) | 2 (4%) | |
| Triple negative (n, %) | 4 (8%) | |
| Lymphovascular invasion | Present (n, %) / Absent (n, %) | 16 (32%) / 34 (68%) |
| CEM findings | Histopathological/Clinical Characteristics | Statistical Relationship |
| Tumor dimension | Tumor size | Positive correlation (ρ = 0.788, p < 0.001) |
| Grade 3 | Significant association (p = 0.017) | |
| Not circumscribed margins | Luminal-B | Significant association (p = 0.001) |
| High lesion conspicuity | Luminal-B (n, %) | Significant association (p = 0.001) |
| Triple negative (n, %) | Significant association (p = 0.001) | |
| Tumor size | Positive correlation (ρ = 0.517, p < 0.001) | |
| BPE | Age | Negative correlation (ρ = -0.286, p = 0.049) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




