Submitted:
28 February 2025
Posted:
28 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
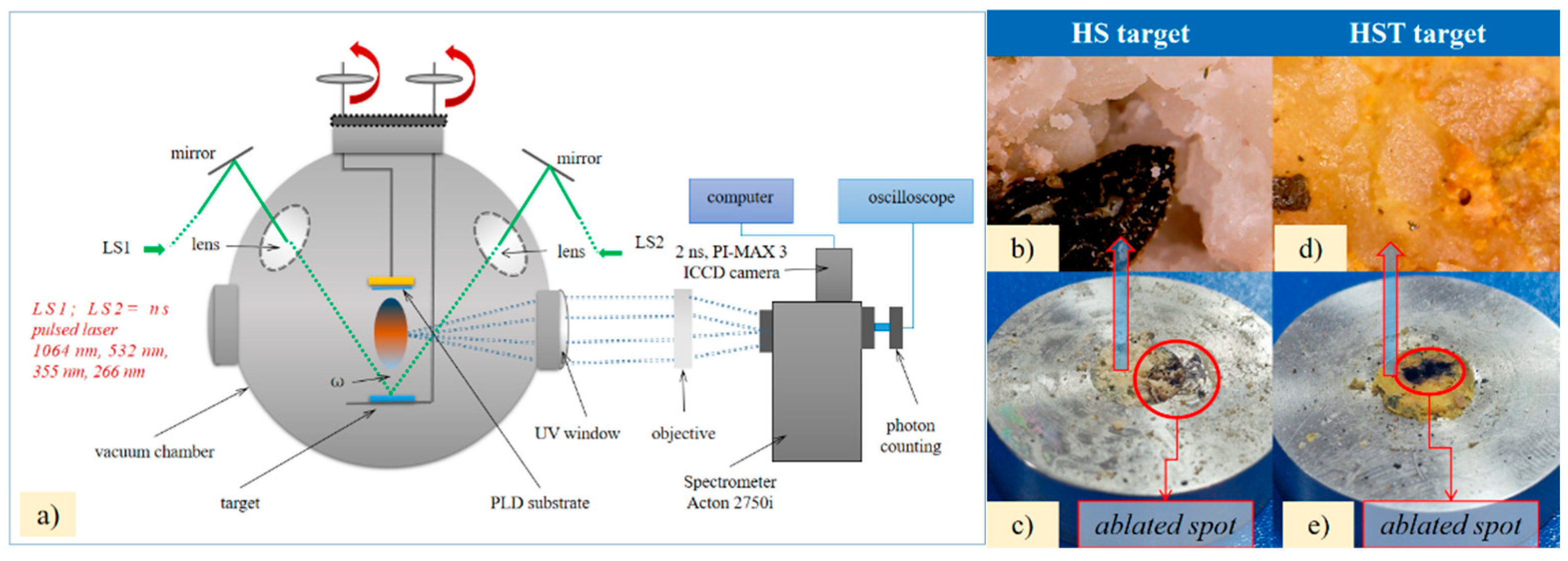
2. Methods and Materials
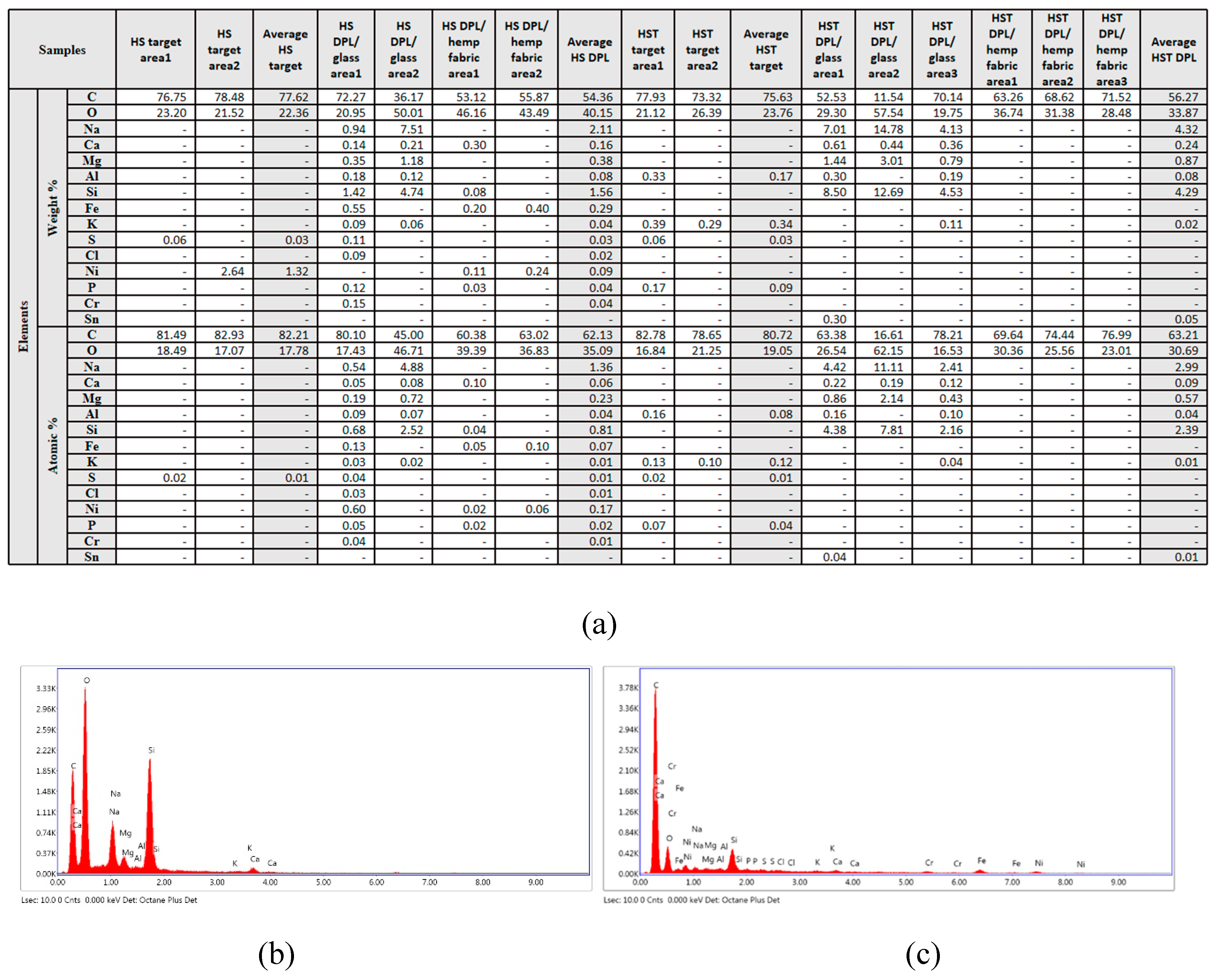
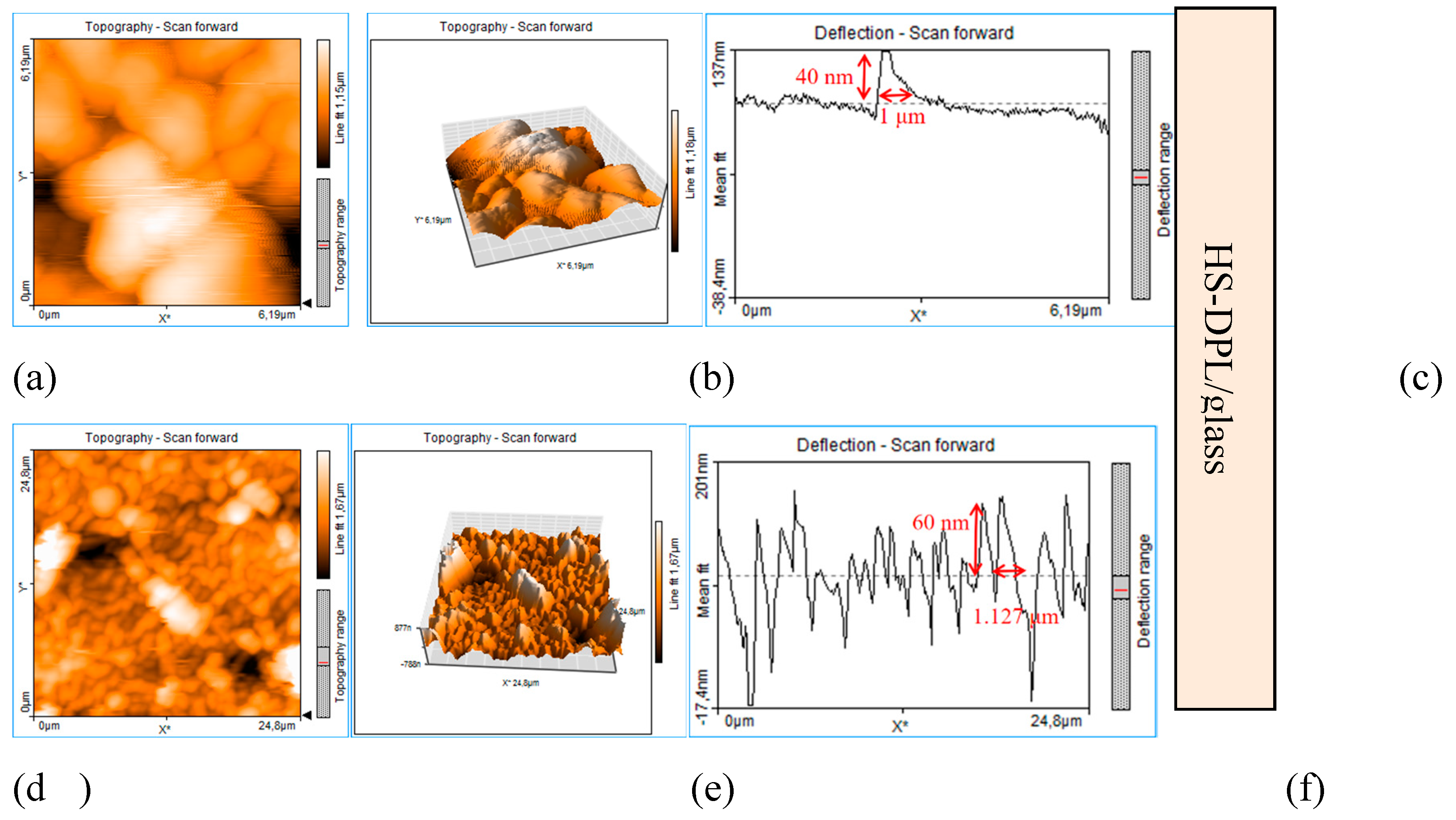
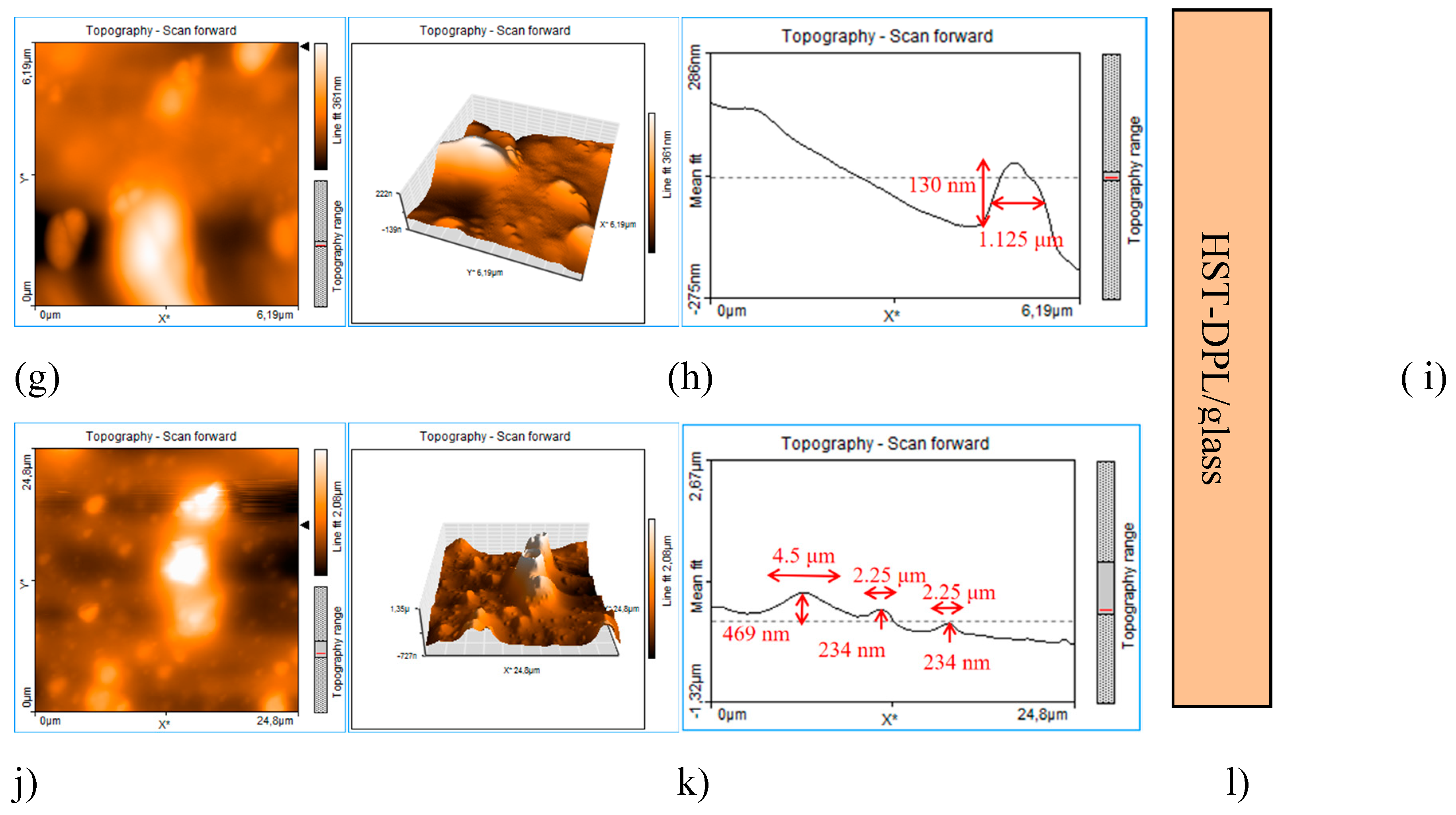
3. Results and Discussions
Transfer Test
4. Conclusions
Aknowledgement
References
- Cocean, I.; Cocean, A.; Postolachi, C.; Pohoata, V.; Cimpoesu, N.; Bulai, G.; Iacomi, F. ; S. Gurlui, S.; Alpha keratin amino acids behvior under high fluence laser interaction. Medical applications, Applied Surface Science 488 (2019) 418–426. [CrossRef]
- Cocean, G.; Cocean, A.; Postolachi, C.; Garofalide, S.; Bulai, G.; Munteanu, B.S.; Cimpoesu, N.; Cocean, I.; Gurlui, S. High-Power Laser Deposition of Chitosan. Polymers: Medical and Environmental Applications. Polymers (Basel). 2022 Apr 10;14(8):1537. [CrossRef]
- Cocean, G.; Cocean, A.; Garofalide, S.; Pelin, V.; Munteanu, B.S.; Pricop, D.A.; Motrescu, I.; Dimitriu, D.G.; Cocean, I.; Gurlui, S. Dual-Pulsed Laser Ablation of Oyster Shell Producing Novel Thin Layers Deposed to Saccharomyces cerevisiae. Polymers 2023, 15, 3953. [Google Scholar] [CrossRef] [PubMed]
- Cocean, A.; Cocean, I.; Cimpoesu, N.; Cocean, G.; Cimpoesu, R.; Postolachi, C.; Popescu, V.; Gurlui, S. Laser Induced Method to Produce Curcuminoid-Silanol Thin Films for Transdermal Patches Using Irradiation of Turmeric Target. Appl. Sci. 2021, 11, 4030. [Google Scholar] [CrossRef]
- Cocean, A.; Cocean, G.; Diaconu, M.; Garofalide, S.; Husanu, F.; Munteanu, B.S.; Cimpoesu, N.; Motrescu, I.; Puiu, I.; Postolachi, C.; Cocean, I.; Gurlui, S. Nano-Biocomposite Materials Obtained from Laser Ablation of Hemp Stalks for Medical Applications and Potential Component in New Solar Cells. Int. J. Mol. Sci. 2023, 24, 3892. [Google Scholar] [CrossRef] [PubMed]
- Andrew, J. Hill, Claire M. Williams, Benjamin J. Whalley, Gary J. Stephens, Phytocannabinoids as novel therapeutic agents in CNS disorders, Pharmacology & Therapeutics, Volume 133, Issue 1, 2012, Pages 79-97, ISSN 0163-7258. [CrossRef]
- Sandru, I.D.; Paraschivoiu, R.; Gauca, C. Cultura canepii (Hemp cultivation), Helicon Editure, 1996, ISBN 973-574-249-7.
- El-Sohaimy, S.A.; Androsova, N.V.; Toshev, A.D.; El Enshasy, H.A. Nutritional Quality, Chemical, and Functional Characteristics of Hemp (Cannabis sativa ssp. sativa) Protein Isolate. Plants 2022, 11, 2825. [Google Scholar] [CrossRef] [PubMed]
- Francesco Siano, Stefania Moccia, Gianluca Picariello, Gian Luigi Russo, Giuseppe Sorrentino, Michele Di Stasio, Francesco La Cara, Maria Grazia Volpe, Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L. ), Molecules 2019, 24, 83; [CrossRef]
- Andronie, L.; Pop, I.D.; Sobolu, R.; Diaconeasa, Z.; Truță, A.; Hegeduș, C.; Rotaru, A. Characterization of Flax and Hemp Using Spectrometric Methods. Appl. Sci. 2021, 11, 8341. [Google Scholar] [CrossRef]
- Mouslech Z, Valla V. Endocannabinoid system: An overview of its potential in current medical practice. Neuro Endocrinol Lett. 2009;30(2):153-79. [PubMed]
- Pacher P, Bátkai S, Kunos G. The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev. 2006 Sep;58(3):389-462. [CrossRef] [PubMed] [PubMed Central]
- Rodríguez de Fonseca F, Del Arco I, Bermudez-Silva FJ, Bilbao A, Cippitelli A, Navarro M. The endocannabinoid system: physiology and pharmacology. Alcohol Alcohol. 2005 Jan-Feb;40(1):2-14. [CrossRef] [PubMed]
- Gülck, T.; Møller, B. L. Phytocannabinoids: Origins and Biosynthesis, Trends in Plant Science, 20, Vol. 25, No. 20 October. [CrossRef]
- Ashton, C.H. Pharmacology and effects of cannabis: a brief review, British Journal of Psychiatry (2001), 178, 101-106. [CrossRef]
- Malcolm Begg, Pál Pacher, Sándor Bátkai, Douglas Osei-Hyiaman, László Offertáler, Fong Ming Mo, Jie Liu, George Kunos, Evidence for novel cannabinoid receptors, Pharmacology & Therapeutics, Volume 106, Issue 2, 2005, Pages 133-145, ISSN 0163-7258. [CrossRef]
- Natalia Battista, Monia Di Tommaso, Monica Bari, Mauro Maccarrone, The endocannabinoid system: an overview, REVIEW article Front. Behav. Neurosci., Sec. 14 March 2012. [CrossRef]
- Sakakibara, I.; Ikeya, Y.; Hayashi, K.; Mitsuhashi, H. Three phenyldihydronaphthalene lignanamides from fruits of Cannabis Sativa, Phytochemistry, Vol.31, No.9, pp. 3219. [Google Scholar]
- Yuefang Zhou, Shanshan Wang, Hongxiang Lou, Peihong Fan, Chemical constituents of hemp (Cannabis sativa L.) seed with potential antineuroinflammatory activity, Phytochemistry Letters 23 (2018) 57–61. [CrossRef]
- Oier Aizpurua-Olaizola, Umut Soydaner, Ekin Öztü rk, Daniele Schibano,Yilmaz Simsir, Patricia Navarro, Nestor Etxebarria, and Aresatz Usobiaga, Evolution of the Cannabinoid and Terpene Content during the Growth of Cannabis sativa Plants from Different Chemotypes, J. Nat. Prod. 2016, 79, 324−331. [CrossRef]
- Alex, J. Straiker, Greg Maguire, Ken Mackie, James Lindsey; Localization of Cannabinoid CB1 Receptors in the Human Anterior Eye and Retina. Invest. Ophthalmol. Vis. Sci. 1999;40(10):2442-2448.
- Navarrete, C.; García-Martín, A.; Rolland, A.; DeMesa, J.; Muñoz, E. Cannabidiol and Other Cannabinoids in Demyelinating Diseases. Int. J. Mol. Sci. 2021, 22, 2992. [Google Scholar] [CrossRef]
- Abyadeh M, Gupta V, Paulo JA, Gupta V, Chitranshi N, Godinez A, Saks D, Hasan M, Amirkhani A, McKay M, Salekdeh GH, Haynes PA, Graham SL, Mirzaei M. A Proteomic View of Cellular and Molecular Effects of Cannabis. Biomolecules. 2021 Sep 27;11(10):1411. [CrossRef] [PubMed] [PubMed Central]
- Soheila Rezapour-Firouzi, Seyed Rafie Arefhosseini, Farhoudi Mehdi, Ebrahimi-Mamaghani Mehrangiz, Behzad Baradarand, Elyar Sadeghihokmabad, Somaiyeh Mostafaei, Seyed Mohammad Bagher Fazljou, Mohammad-ali Torbati, Sarvin Sanaie, Fatemeh Zamani,Immunomodulatory and therapeutic effects of Hot-nature diet and co-supplemented hemp seed, evening primrose oils intervention in multiple sclerosis patients, Complementary Therapies in Medicine (2013) 21, 473—480. [CrossRef]
- Khan, H. , Ghori, F.K., Ghani, U. et al. Cannabinoid and endocannabinoid system: a promising therapeutic intervention for multiple sclerosis. Mol Biol Rep 49, 5117–5131 (2022). [CrossRef]
- Ahmed Nafis, Ayoub Kasrati, Chaima Alaoui Jamali, Noureddine Mezrioui, William Setzer, Abdelaziz Abbad, Lahcen Hassani, Antioxidant activity and evidence for synergism of Cannabis sativa (L.) essential oil with antimicrobial standards, Industrial Crops & Products 137 (2019) 396–400. [CrossRef]
- Jose Ignacio Alonso-Esteban, Jose Pinela, Ana Ciric, Ricardo C. Calhelha, Marina Sokovic, Isabel C.F.R. Ferreira, Lillian Barros, Esperanza Torija-Isasa, María de Cortes Sanchez-Mata, Chemical composition and biological activities of whole and dehulled hemp (Cannabis sativa L.) seeds, Food Chemistry 374 (2022) 131754. [CrossRef]
- Moqejwa, T.; Marimuthu, T.; Kondiah, P.P.D.; Choonara, Y.E. Development of Stable Nano-Sized Transfersomes as a Rectal Colloid for Enhanced Delivery of Cannabidiol. Pharmaceutics 2022, 14, 703. [Google Scholar] [CrossRef] [PubMed]
- Cocean, A.; Cocean, G.; Postolachi, C.; Garofalide, S.; Pricop, D.A.; Munteanu, B.S.; Bulai, G.; Cimpoesu, N.; Motrescu, I.; Pelin, V.; Ababei, R. V; Dimitriu, D.-G. *; Cocean, I; Silviu Gurlui, S.*, High Energy Pulsed Laser Beam to Produce a Thin Layer of Crystalline Silver without Heating the Deposition Substrate and Its Catalytic Effects. Quantum Beam Sci. [CrossRef]
- Pretsch, E.; Buhlmann, P.; Badertscher, M. Structure Determination of Organic Compounds. Tables of Spectral Data, 4th ed.; Revised and Enlarged Edition; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-3-540-93810-1. [Google Scholar]
- Bruker Optics. "Differentiating Cannabis with FTIR". News-Medical. https://www.news-medical.net/whitepaper/20190614/Differentiating-Cannabis-with-FTIR.aspx. (accessed August 28, 2024).

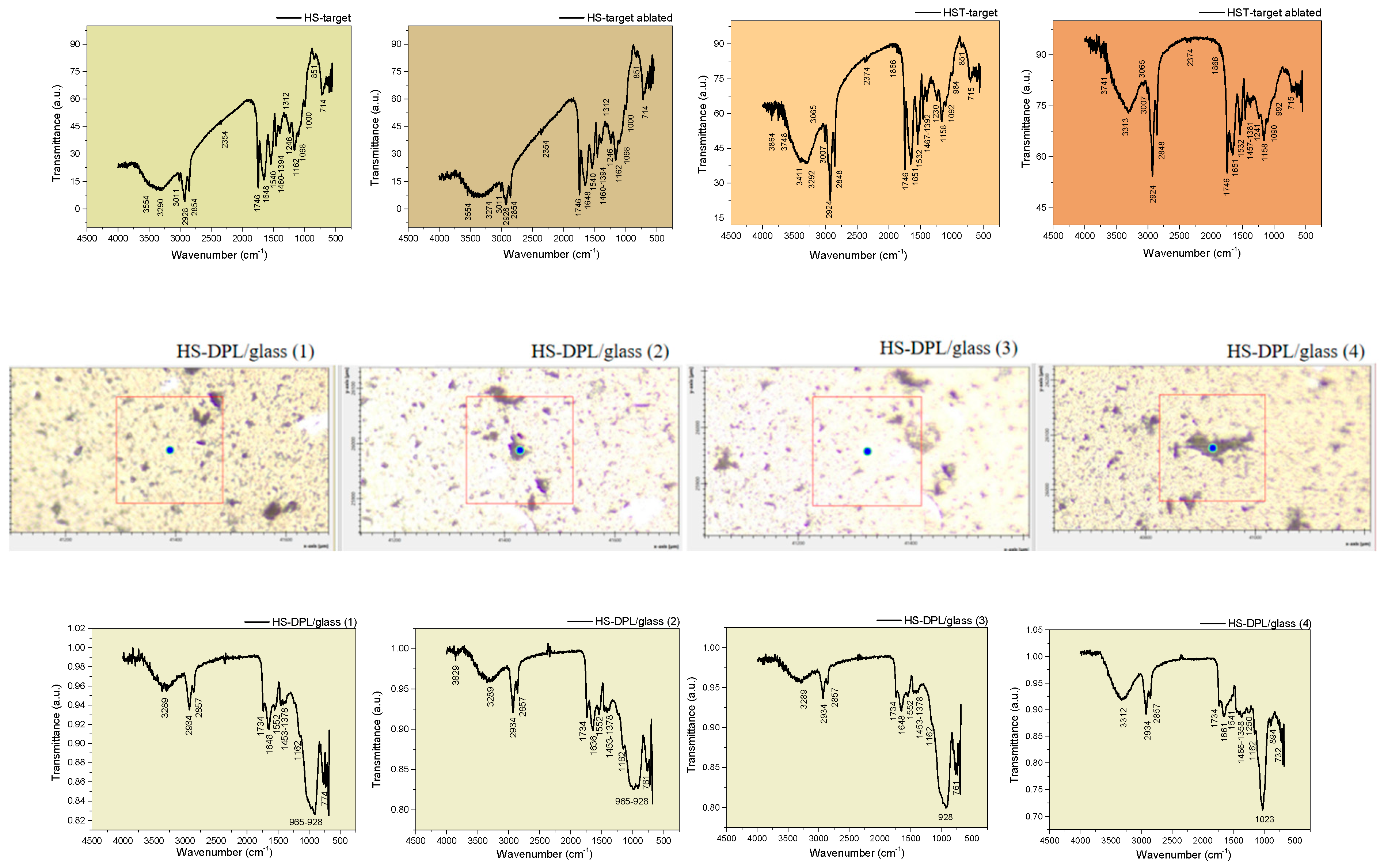
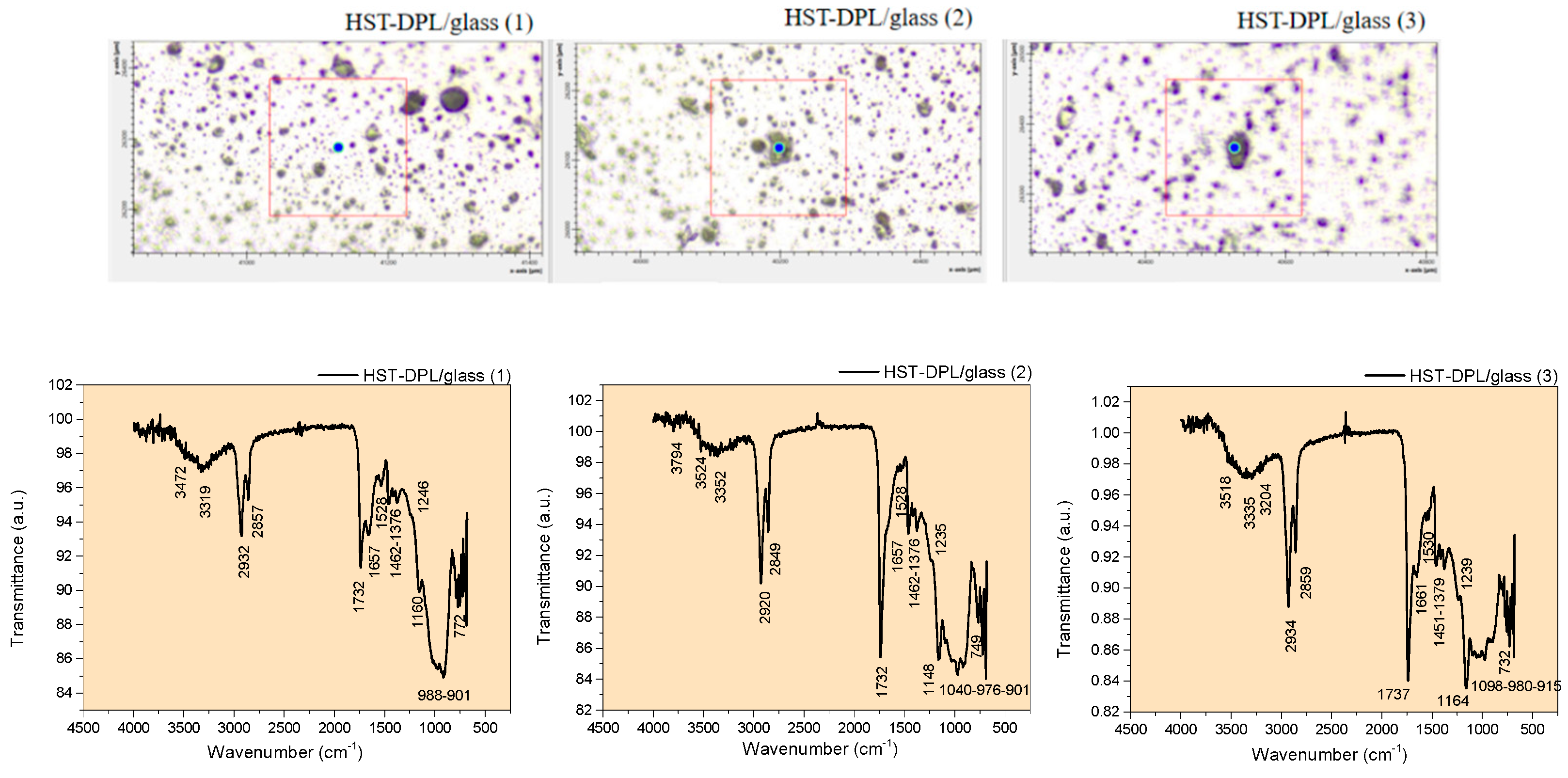



| Vibration bands [cm-1] |
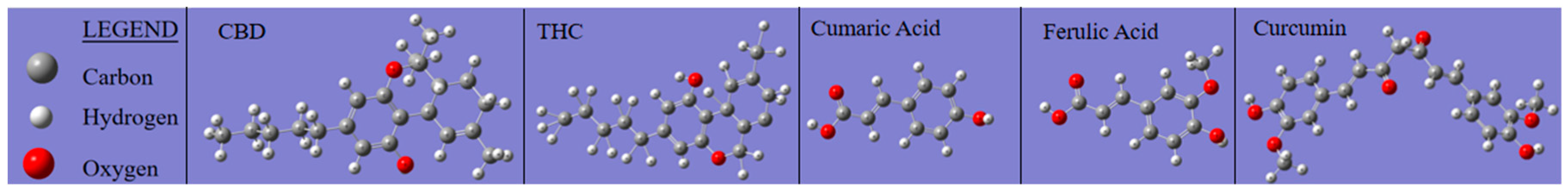
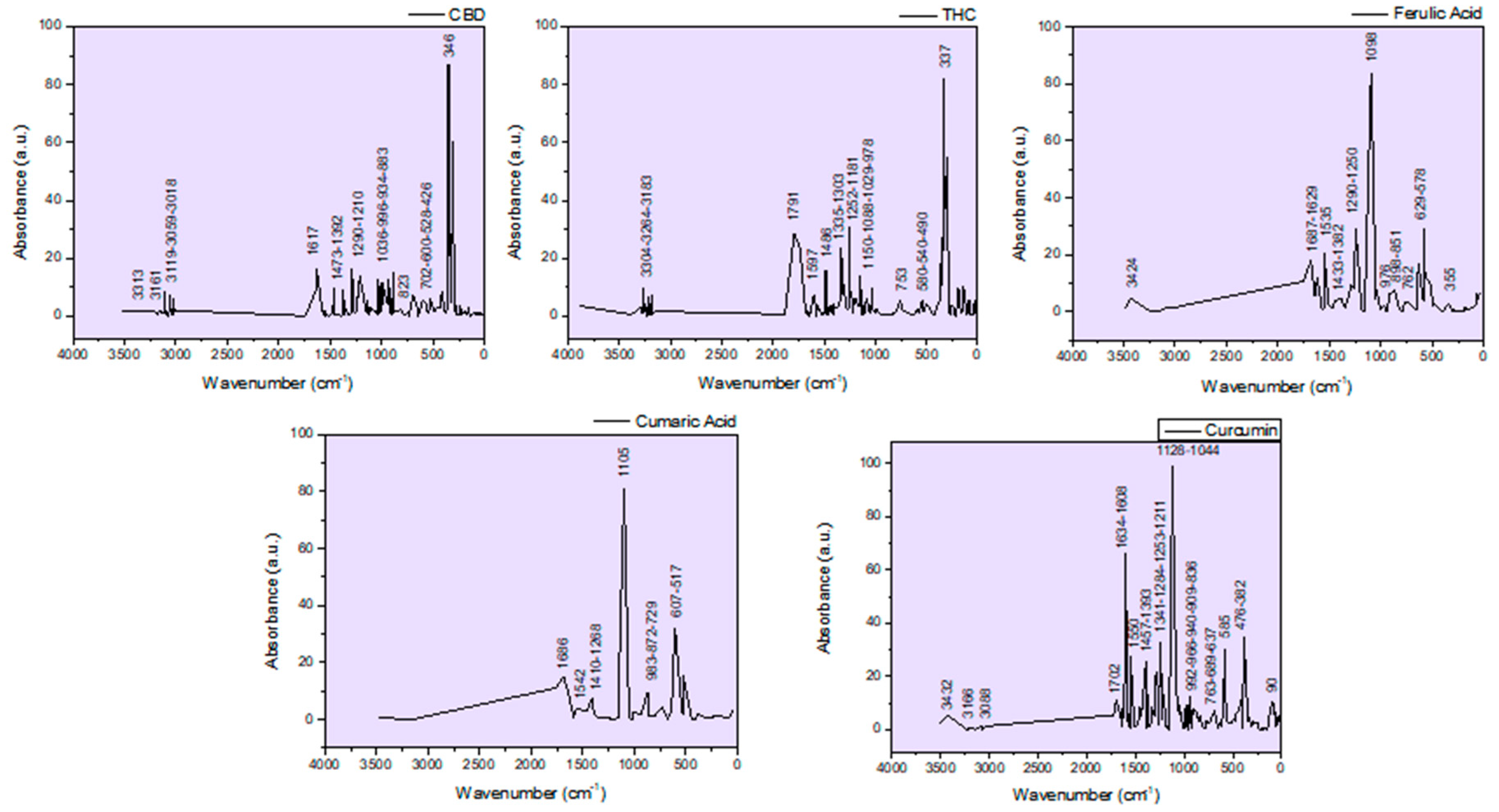
Functional groups identified based on Pretsch et al., 2009 [30] and on the Gaussian 6 IR spectra simulation performed in this study (Figure 3) |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| HS-target | HS-target ablated | HS-DPL/ glass (1) |
HS-DPL/ glass (2) |
HS-DPL/ glass (3) |
HS-DPL/ glass (4) |
HS-T-target | HST-target ablated | HST- DPL/ glass (1) |
HST- DPL/ glass (2) |
HST- DPL/ glass (3) |
|
| - | OH stretching, free | - | 3829 | - | - | 3855 3748 |
- 3741 |
- |
- 3749 |
- | OH stretching, free |
| 3554 3290 |
3554 3274 |
3495 3375 3289 |
3460 3343 3289 |
3558 3375 3289 |
3410 3312 3258 |
3411 3292 |
- 3313 |
3472 3319 |
3524 3352 |
3518 3335 |
OH and NH stretching, free and H-bonded; C-H aromatic stretching (3200-3312 cm-1 bands are assigned to aromatic CH and phenolic OH). Lignanamides (3364 cm-1) Gaussian simulation: CBD (3313 cm-1; 3161 cm-1); THC (3304 cm-1; 3264 cm-1); Ferulic Acid (3424 cm-1); Coumaric Acid (3500 cm-1); Curcumin (3432 cm-1) |
| 3011 | 3011 | - | - | - | - | 3006 | 3007 | - | - | - | CH aromatic stretching Gaussian simulation: CBD (3119 cm-1; 3059 cm-1; 3018 cm-1); Curcumin (3166 cm-1; 3088 cm-1) |
| 2928 | 2928 | 2934 | 2934 | 2934 | 2934 | 2924 | 2924 | 2932 | 2920 | 2934 | CH aliphatic asymmetric stretching |
| 2854 | 2854 | 2857 | 2857 | 2857 | 2857 | 2848 | 2848 | 2857 | 2849 | 2859 | CH aliphatic symmetric stretching |
| 2354 | 2354 | - | - | - | - | 2374 | 2374 | - | - | - | CO2 |
| - | - | - | - | - | - | 1866 | 1866 | - | - | - | C=O stretching (assigned to hemp oil) Esthers; aldehyde ether |
| 1746 | 1746 | 1734 | 1734 | 1734 | 1734 | 1746 | 1746 | 1732 | 1732 | 1737 | C=O stretching (assigned to THC; hemp oil; curcumin) Esthers; cyclopentanone 1745 cm-1); aldehyde ether Gaussian simulation: THC (1791cm-1); Curcumin (1702 cm-1) |
| 1648 | 1648 | 1648 | 1636 | 1648 | 1661 | 1651 | 1651 | 1657 | 1657 | 1661 | C=O stretching in amides Lignanamides (1656 cm-1) C=C bending in alkenes (specific to cannabinoids side chain) Gaussian simulation: CBD (1617 cm-1); Ferulic Acid (1687 cm-1; 1629 cm-1); coumaric Acid (1686 cm-1);Curcumin (1634 cm-1; 1608 cm-1) |
| 1540 | 1540 | 1552 | 1552 | 1552 | 1541 | 1532 | 1532 | 1528 | 1528 | 1530 | NH bending (deformation) in amides Lignanamides (1514 cm-1) C=C bending in alkenes (specific to CBD side chain) Gaussian simulation: THC (1597 cm-1); Ferulic Acid (1535 cm-1); coumaric Acid (1542 cm-1); Curcumin (1550 cm-1) |
| 1460 | 1460 | 1453 | 1453 | 1453 | 1466 | 1467 | 1457 | 1462 | 1462 | 1451 | OH bending in alcohols and COOH (carboxylic acids) CH bending of methyl and methylen group Gaussian simulation: CBD (1473 cm-1); THC (1486 cm-1); Ferulic Acid (1433 cm-1); coumaric Acid (1410 cm-1); Curcumin (1457 cm-1) |
| 1394 1312 |
1394 1312 |
1378 | 1378 | 1378 | 1358 | 1392 | 1381 | 1376 | 1376 | 1379 | OH bending in phenols CH3 bending Gaussian simulation: CBD (1392 cm-1); THC (1335 cm-1; 1303 cm-1); Ferulic Acid (1382 cm-1); Curcumin (1393 cm-1) |
| 1246 | 1246 | 1237sh | 1237sh | - | 1250 | 1230 | 1241sh | 1246sh | 1235sh | 1239sh | Ar-C-OH bending CN stretching in aromatic amine CO ring skeletal stretching in epoxides Gaussian simulation: CBD (1290 cm-1; 1210 cm-1); THC (1252 cm-1); Ferulic Acid (1290 cm-1; 1250 cm-1); coumaric Acid (1268 cm-1) |
| 1162 | 1162 | 1162sh | 1162sh | 1162sh | 1162sh | 1158 | 1158 | 1160 | 1148 | 1164 | Ar-C-OH bending C=C bending in alkenes CO ring skeletal vibrations in epoxides Gaussian simulation: THC (1181 cm-1; 1150 cm-1); Curcumin (1128 cm-1) |
| 1098sh | 1098sh | - | - | - | 1023 | 1092sh | 1090sh | - | 1040 | 1098 | C=C bending in alkenes Gaussian simulation: CBD (1036 cm-1); THC (1088 cm-1; 1029 cm-1); Ferulic Acid (1098 cm-1); coumaric Acid (1105 cm-1); Curcumin (1044 cm-1) |
| 1000sh 908 |
1000sh | 965 928 |
962 928 |
928 | - | 984sh | 992sh | 988- 901 |
976- 901 |
980- 915 |
C=C bending in alkenes Gaussian simulation: CBD (966 cm-1; 934 cm-1); THC (978 cm-1); Ferulic Acid (976 cm-1); coumaric acid (983 cm-1); Curcumin (992 cm-1; 966 cm-1; 940 cm-1; 909 cm-1) |
| 851 | 851 | - | - | - | 894 | C=C bending in alkenes Gaussian simulation: CBD (883 cm-1; 823 cm-1); Ferulic Acid (898 cm-1; 851 cm-1); coumaric Acid (872 cm-1); Curcumin (836 cm-1) |
|||||
| 714 | 714 | 774 | 761 | 761 | 732 | 715 | 715 | 772 | 749 | 732 | C=C skeletal vibrations Gaussian simulation: CBD (702 cm-1); THC (753 cm-1); Ferulic Acid (762 cm-1); coumaric Acid (729 cm-1); Curcumin (736 cm-1) |
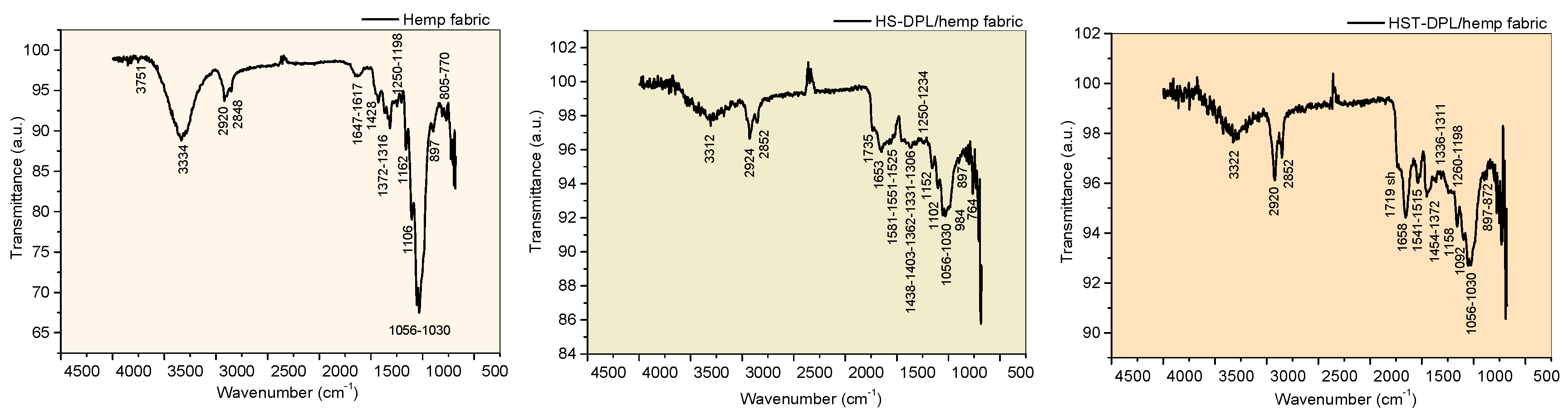
| Vibration bands [cm-1] |
Functional groups identified based on Pretsch et al., 2009 [30] and on the Gaussian 6 IR spectra simulation performed in this study (Figure 3) |
||||
|---|---|---|---|---|---|
| HS-target | HST-target | Hemp fabric | HS-DPL/hemp fabric | HST-DPL/hemp faric | |
| - | 3855 | - | - | - | OH stretching, free |
| 3748 | 3751 | - | - | OH stretching, free | |
| 3554-3290 | 3411-3292 | 3334 | 3312 | 3322 | OH and NH stretching, free and H-bonded CH aromatic stretching (3200-3312 cm-1 bands are assigned to aromatic CH and phenolic OH) Lignanamides (3364 cm-1) Gaussian simulation: CBD (3313 cm-1; 3161 cm-1); THC (3304 cm-1; 3264 cm-1); Ferulic Acid (3424 cm-1); coumaric Acid (3500 cm-1); Curcumin (3432 cm-1) |
| 3011 | 3065sh 3006 |
- | - | - | CH aromatic stretching Gaussian simulation: CBD (3119 cm-1; 3059 cm-1; 3018 cm-1); Curcumin (3166 cm-1; 3088 cm-1) |
| 2928 | 2924 | 2920 | 2924 | 2920 | CH aliphatic asymmetric stretching |
| 2854 | 2848 | 2848 | 2852 | 2852 | CH aliphatic symmetric stretching |
| 2354 | 2374 | - | - | - | CO2 |
| - | 1866 | - | - | - | C=O stretching (assigned to hemp oil [ ]) Esthers; aldehyde ether |
| 1746 | 1746 | - | 1735 | 1719sh | C=O stretching (assigned to THC, hemp oil , curcumin) Esthers; cyclopentanone (1745 cm-1); aldehyde ether Gaussian simmulation: THC (1791 cm-1); Curcumin (1702) |
| 1648 | 1651 | 1647-1617 | 1653 | 1658 | C=O stretching in amides Lignanamides (1656 cm-1) C=C bending in alkene (specific to CBD side chain Gaussian simulation: CBD (1617cm-1); Ferulic Acid (1687 cm-1; 1629 cm-1); coumaric Acid (1686 cm-1); Curcumin (1634 cm-1; 1608 cm-1) |
| 1540 | 1532 | - | 1581 1551 1525 |
1541 1515 |
NH bending (deformation) in amides Lignanamides (1514 cm-1) C=C bending in alkene (specific to CBD side chain) Gaussian simulation: THC (1597 cm-1); Ferulic Acid (1535 cm-1); coumaric Acid (1542 cm-1); Curcumin (1550 cm-1) |
| 1460 | 1467 | 1428 | 1438 1403 |
1454 | OH bending in alcohols and COOH (carboxylic acids) CH bending of methyl and methylen group Gaussian simulation: CBD (1473 cm-1); THC (1486 cm-1); Ferulic Acid (1433 cm-1); coumaric Acid (1410 cm-1); Curcumin (1457 cm-1) |
| 1394 1312 |
1392 | 1372 1316 |
1362 1331 1306 |
1372 1336 1311 |
OH bending in phenols CH3 bending Gaussian simulation: CBD (1392 cm-1); THC (1335 cm-1; 1303 cm-1); ferulic Acid (1382 cm-1); Curcumin (1393 cm-1) |
| 1246 | 1230 | 1250 | 1250 1234 |
1260 | Ar-C-OH bending CN stretching in aromatic amine CO ring skeletal vibrations in epoxides Gaussian simulation: CBD (1290 cm-1; 1210 cm-1); THC (1252 cm-1); Ferulic Acid (1290 cm-1; 1250 cm-1); coumaric Acid (1268 cm-1) |
| 1162 | 1158 | 1198;1162 | 1198 1158 |
1152 | Ar-C-OH bending C=C bending in alkenes CO ring skeletal vibrations in epoxides Gaussian simulation: THC (1181 cm-1; 1150 cm-1) |
| 1098sh | 1092sh | 1106 | 1102 1056 1030 |
1056 1030 |
C=C bending in alkenes Gaussian simulation: CBD (1036 cm-1); THC (1088 cm-1; 1029 cm-1); ferulic Acid (1098 cm-1); coumaric Acid (1105 cm-1); Curcumin (1044 v) |
| 1000sh 908 |
984sh | - | 984 | - | C=C bending in alkenes Gaussian simulation: CBD (966 cm-1; 934 v); THC (978 cm-1); Ferulic Acid 976 cm-1); coumaric Acid (983 cm-1); Curcumin (992 cm-1; 966 cm-1; 940 cm-1; 909 cm-1) |
| 851 | - | 897 805 |
897 | 897 872 |
C=C bending in alkenes Gaussian simulation: CBD (883 cm-1; 823 cm-1); Ferulic Acid (898 cm-1; 851 cm-1); coumaric Acid (972 cm-1); Curcumin (836 cm-1) |
| 714 | 715 | 770 | - | 764 | CC skeletal vibrations Gaussian simulation: CBD (702 cm-1); THC (753 cm-1); Ferulic Acid (762 cm-1); coumaric Acid (729 cm-1); Curcumin (736) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




