Submitted:
20 February 2025
Posted:
20 February 2025
You are already at the latest version
Abstract
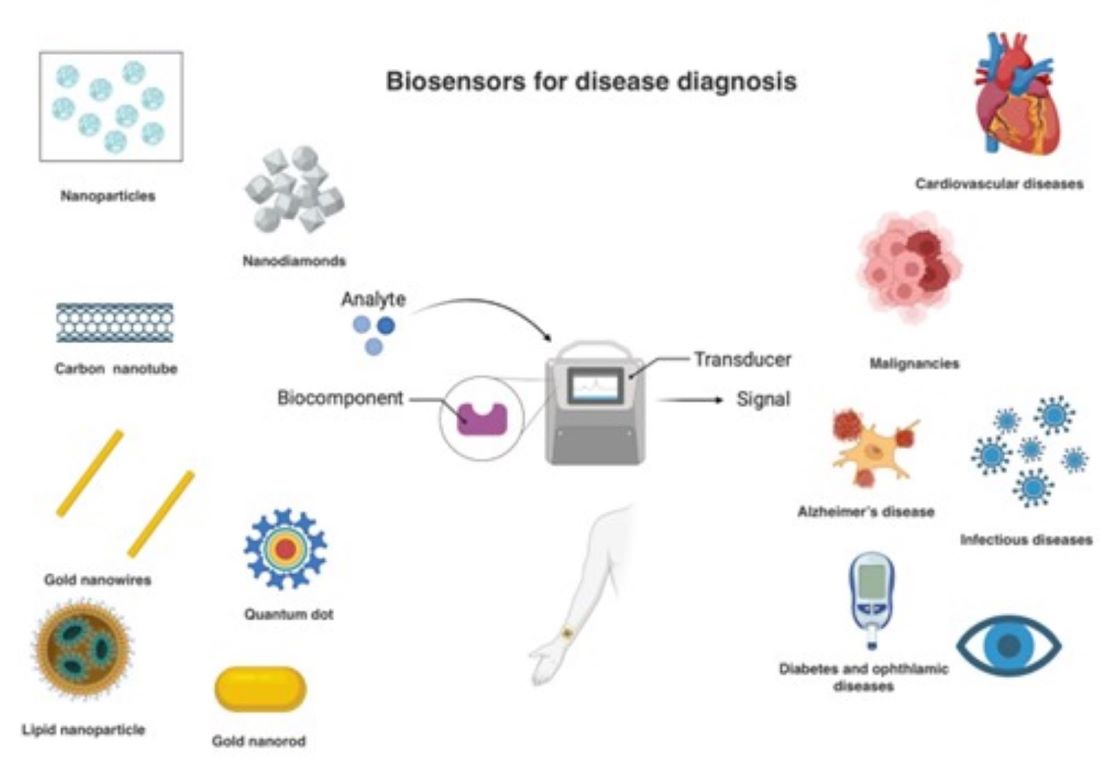
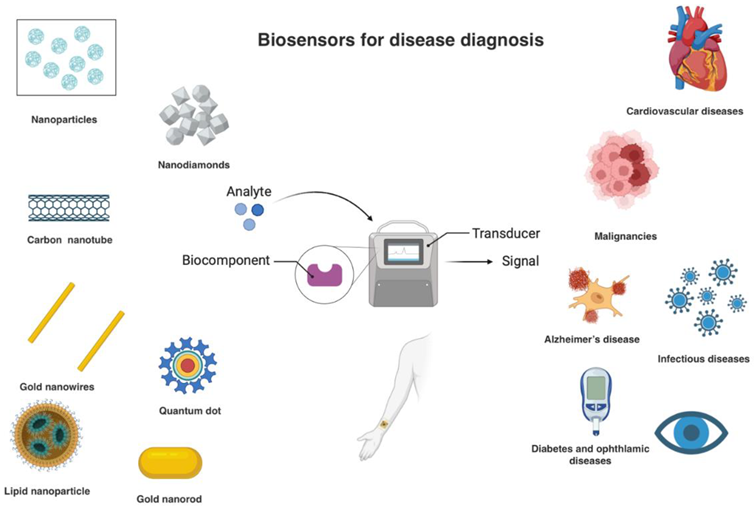
Keywords:
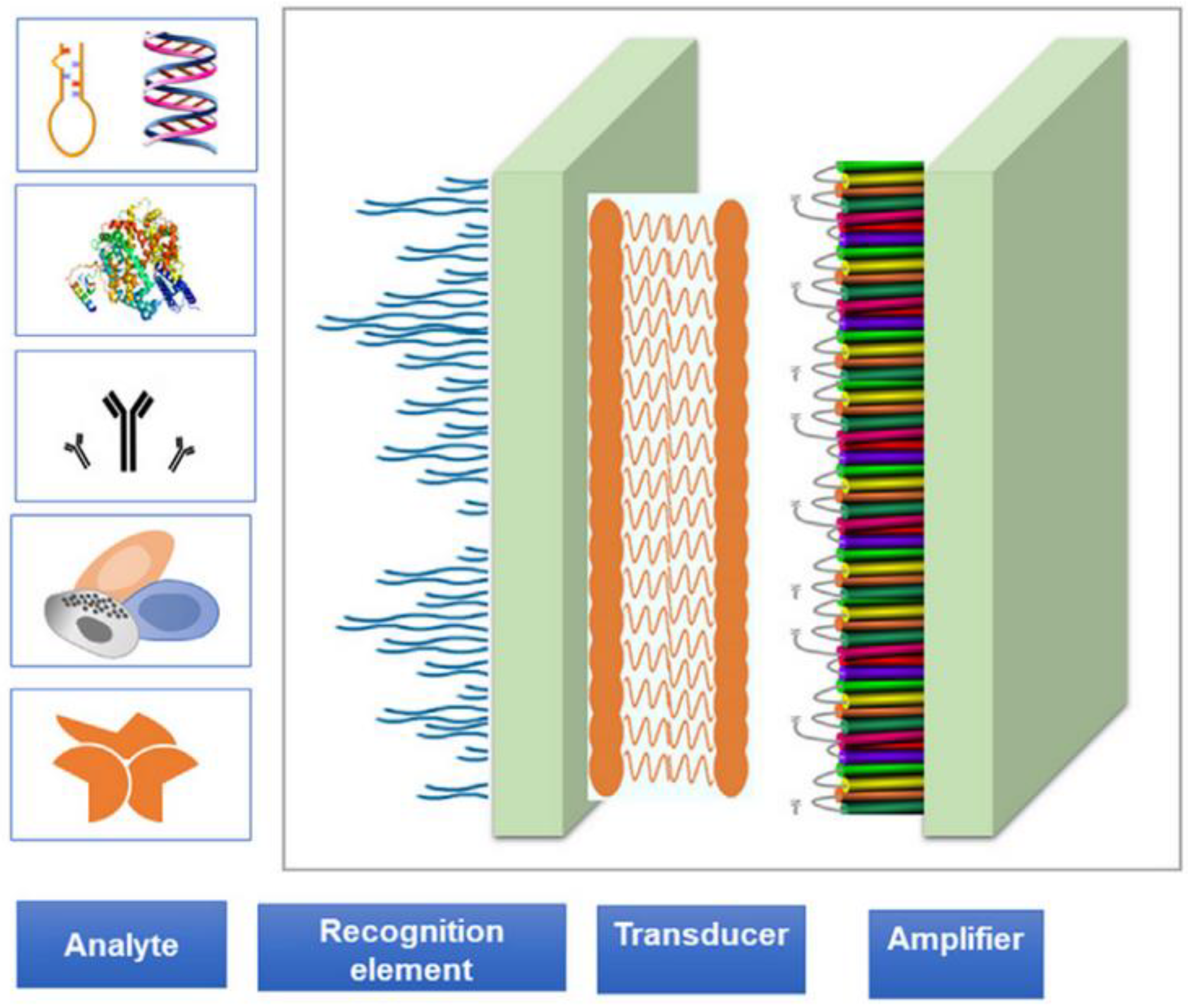
1. Introduction
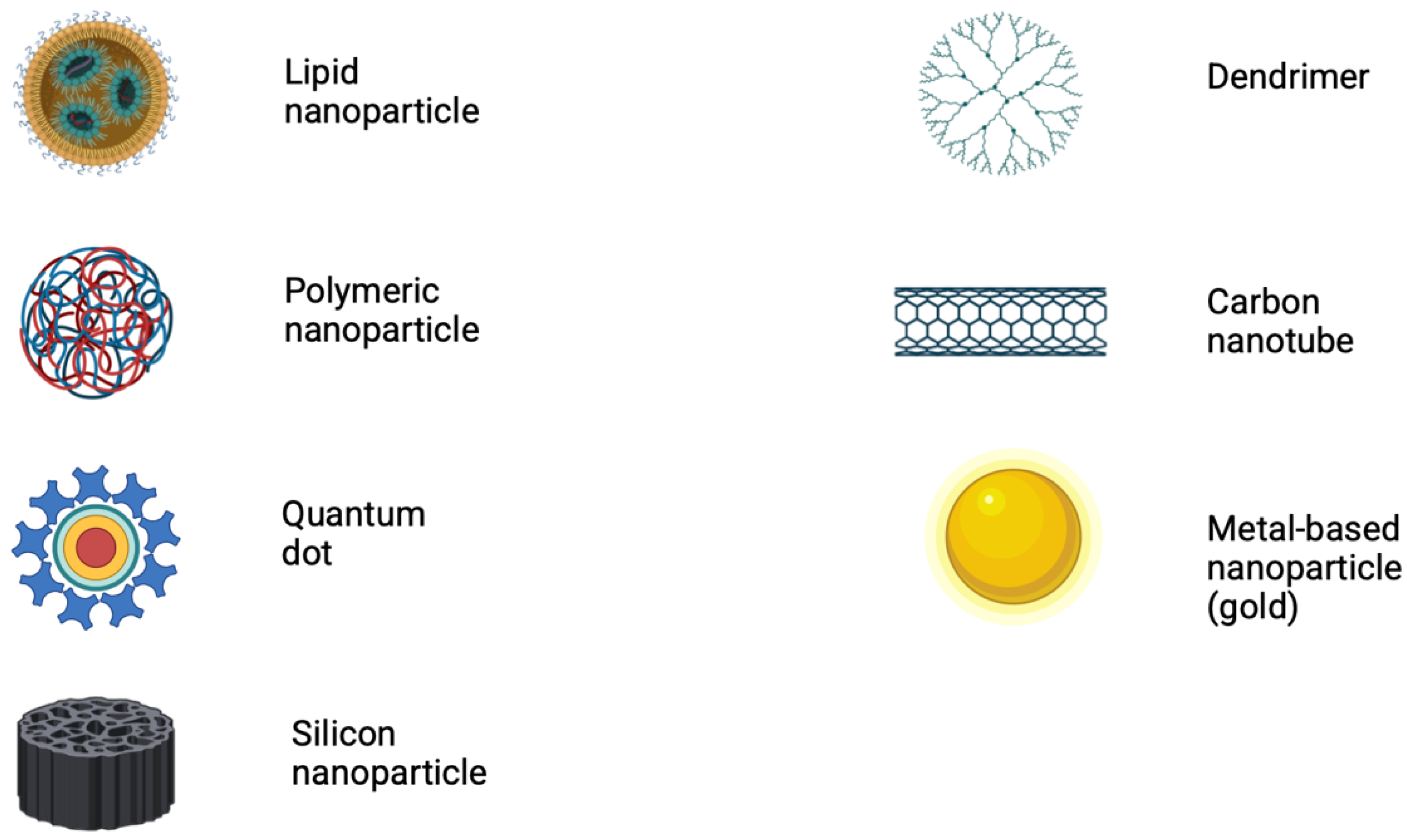
2. Nanomaterials Employed in Nano-Biosensors
- Lipid nanoparticles
- ii.
- Polymeric nanoparticles
- iii.
- Quantum dots
- iv.
- Silicon nanoparticles
- v.
- Dendrimers
- vi.
- Carbon nanoparticles
- vii.
- Metal-based nanoparticles
3. Types of Nanotechnology-Based Biosensors for Disease Diagnosis
- i.
- Electrochemical Nanobiosensors
- ii.
- Optical Nanobiosensors
- iii.
- Piezoelectric Nanobiosensors
- iv.
- Magnetic Nanobiosensors
- v.
- Thermal or Calorimetric Nanobiosensors
4. Nanobiosensors for Disease Diagnosis
4.1. Cardiovascular Disease
4.2. Cancer
| Biomarker | Cancer | Nanomaterial | Method | Reference |
|---|---|---|---|---|
| HER 2 | Breast | Metal NPs, QDs, CNTs, graphene | EC, optical, piezoelectric | [30] |
| CA 15.3 | Breast | Metal NPs | EC | [29] |
| SCC-Ag | Ovarian | CNTs | EC | [31] |
| CA 125 | Ovarian | Metal nanocomposite (NC), QDs, Magnetic NPs | Optical, EC | [32] |
| HE4 | Ovarian | Metal NC | EC | [33] |
| PSA | Prostate | Graphene, metal NPs, QDs, conducting polymers (CPs) | EC, magnetic, fluorescence | [34,35,36,37] |
| CEA | Colorectal, pancreatic, lung | Graphene, CPs, QDss | EC | [38] |
| AFP | Hepatocellular | Graphene, metal NPs, CPs | EC | [39] |
| NSE | Lung | Metal NWs, QDs, Graphene | EC | [40] |
| CYFRA-21-1 | Oral, Lung | Metal NPs, QDs | [41] | |
| FLT3-ITD Mutations | Acute myeloid leukemia | Metal NRs | EC | [42] |
| EGFR III | Glioma | QDs | EC | [24] |
| Anti-apoptotic protein | Colorectal | Graphene | Optical | [43] |
| PDGF-B, thrombin | Gastric | Metal-NPs | Optical-SERS,SPR | [25] |
4.3. Infectious Diseases
| Virus/Bacterial | Nanoparticles | Detection Methods | Reference |
|---|---|---|---|
| Hepatitis—C virus (HCV) | AuNPs | Colorimetric | [46] |
| SARS-CoV-2 | AuNPs | Colorimetric | [47] |
| MERS-CoV | AgNPs | Colorimetric | [48] |
| AuNPs | Electrochemiluminescence | [49] | |
| Genetic material (Bacteria or Virus) | Magnetic NPs | PCR | [50] |
| Infuenza Virus | AgNPs | PCR | [51] |
| AuNPs | Thermometric | [52] | |
| E. coli | AuNPs | Colorimetric | [63] |
| HBV | AuNPs | Colorimetric | [53] |
| QD | Fluorimetric | [54] | |
| EBOV | MNPs | EC | [55] |
| AgNPs | Colorimetric | [56] | |
| HIV | MNPs | Fluorimetric | [57] |
| AuNPs | SERS | [58] | |
| silica nanoparticles, AuNPs | Electrochemiluminometry | [59] | |
| HPV | graphene, Au nanorod | EC | [60] |
| AgNPs | Colorimetric | [61] | |
| MTB (Mycobacterium tuberculosis) | AgNPs | Colorimetric | [64] |
| Salmonella | Gold-coated magnetic nanoparticle | Colorimetric | [62] |
4.4. Neurological Diseases
4.5. Other Diseases
6. Conclusion and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanjay Kisan Metkar and Koyeli Girigoswami,” Diagnostic Biosensors in Medicine- a Review”, Biocatalysis and Agricultural Biotechnology, vol.17, p. 271-283, January 2019. [CrossRef]
- Song, F. X., Xu, X., Ding, H., Yu, L., Huang, H., Hao, J., Wu, C., Liang, R., & Zhang, S. (2023). Recent Progress in Nanomaterial-Based Biosensors and Theranostic Nanomedicine for Bladder Cancer. In Biosensors (Vol. 13, Issue 1). MDPI. [CrossRef]
- Wang, S., Zhao, Z., Haque, F., & Guo, P. (2018). Engineering of protein nanopores for sequencing, chemical or protein sensing and disease diagnosis. In Current Opinion in Biotechnology (Vol. 51, pp. 80–89). Elsevier Ltd. [CrossRef]
- X. Ma and S. J. Sim, “Single plasmonic nanostructures for biomedical diagnosis,” Journal of Materials Chemistry B, vol. 8, no. 29. Royal Society of Chemistry, pp. 6197–6216, Aug. 07, 2020. [CrossRef]
- S. Sreejith, J. Ajayan, J. M. Radhika, N. v. Uma Reddy, and M. Manikandan, “Recent advances in nano biosensors: An overview,” Measurement: Journal of the International Measurement Confederation, vol. 236. Elsevier B.V., Aug. 15, 2024. [CrossRef]
- R. C. Popescu, M. Oana, M. Fufă, and A. M. Grumezescu, “Metal-based nanosystems for diagnosis,” Rom J Morphol Embryol, vol. 56, no. 2, pp. 635–649, 2015, [Online]. Available: http://www.rjme.ro/.
- Tufani, A. Qureshi, and J. H. Niazi, “Iron oxide nanoparticles based magnetic luminescent quantum dots (MQDs) synthesis and biomedical/biological applications: A review,” Materials Science and Engineering C, vol. 118. Elsevier Ltd, Jan. 01, 2021. [CrossRef]
- Aykaç et al., “An Overview on Recent Progress of Metal Oxide/Graphene/CNTs-Based Nanobiosensors,” Nanoscale Research Letters, vol. 16, no. 1. Springer, 2021. [CrossRef]
- M. Ramesh, R. Janani, C. Deepa, and L. Rajeshkumar, “Nanotechnology-Enabled Biosensors: A Review of Fundamentals, Design Principles, Materials, and Applications,” Biosensors, vol. 13, no. 1. MDPI, Jan. 01, 2023. [CrossRef]
- M. A. Darwish, W. Abd-Elaziem, A. Elsheikh, and A. A. Zayed, “Advancements in nanomaterials for nanosensors: a comprehensive review,” Nanoscale Advances, vol. 6, no. 16. Royal Society of Chemistry, pp. 4015–4046, May 24, 2024. [CrossRef]
- S. A. Qureshi, W. W. W. Hsiao, L. Hussain, H. Aman, T. N. Le, and M. Rafique, “Recent Development of Fluorescent Nanodiamonds for Optical Biosensing and Disease Diagnosis,” Biosensors, vol. 12, no. 12. MDPI, Dec. 01, 2022. [CrossRef]
- Fereshteh Rahdan et al., “MicroRNA electrochemical biosensors for pancreatic cancer”,Clinica Chimica Acta, vol.548, 1 August 2023. [CrossRef]
- Jules L. Hammond et al., “Electrochemical biosensors and nanobiosensors”, Essays in Biochemistry, vol. 60 (1), 30 June 2016, p. 69-80. [CrossRef]
- Lino et al., “Biosensors as diagnostic tools in clinical applications”, Biochimica et Biophysica Acta (BBA) – Reviews on Cancer, vol. 1877 (3), May 2022. [CrossRef]
- Anchal Pradhan et al., “Biosensors as Nano-Analytical Tools for COVID-19 Detection”, Sensors, vol. 21(23), 7823, 2021. [CrossRef]
- Kai Wu et al., “Magnetic-Nanosensor-Based Virus and Pathogen Detection Strategies before and during COVID-19”, ACS Applied Nano Materials, vol.3,p. 9560-9580, 2020. [CrossRef]
- T. A. Tabish, H. Hayat, A. Abbas, and R. J. Narayan, “Graphene Quantum Dots-Based Electrochemical Biosensing Platform for Early Detection of Acute Myocardial Infarction.,” Biosensors, vol. 12, no. 2, Jan. 2022. [CrossRef]
- M. Gachpazan et al., “A review of biosensors for the detection of B-type natriuretic peptide as an important cardiovascular biomarker.,” Anal. Bioanal. Chem., vol. 413, no. 24, pp. 5949–5967, Oct. 2021. [CrossRef]
- J. Li, F. Centurion, R. Chen, and Z. Gu, “Intravascular Imaging of Atherosclerosis by Using Engineered Nanoparticles.,” Biosensors, vol. 13, no. 3, Feb. 2023. [CrossRef]
- Q. Hu, Z. Fang, J. Ge, and H. Li, “Nanotechnology for cardiovascular diseases.,” Innov. (Cambridge, vol. 3, no. 2, p. 100214, Mar. 2022. [CrossRef]
- M. Yang, Z. Ye, Y. Ren, M. Farhat, and P.-Y. Chen, “Recent Advances in Nanomaterials Used for Wearable Electronics,” Micromachines, vol. 14, no. 3. 2023.
- W. A. D. M. Jayathilaka et al., “Significance of Nanomaterials in Wearables: A Review on Wearable Actuators and Sensors.,” Adv. Mater., vol. 31, no. 7, p. e1805921, Feb. 2019. [CrossRef]
- L. Tang, J. Yang, Y. Wang, and R. Deng, “Recent Advances in Cardiovascular Disease Biosensors and Monitoring Technologies.,” ACS sensors, vol. 8, no. 3, pp. 956–973, Mar. 2023. [CrossRef]
- R. Shandilya, A. Bhargava, N. Bunkar, R. Tiwari, I. Y. Goryacheva, and P. K. Mishra, “Nanobiosensors: Point-of-care approaches for cancer diagnostics,” Biosens. Bioelectron., vol. 130, pp. 147–165, 2019. [CrossRef]
- cP. Shao et al., “Aptamer-Based Functionalized SERS Biosensor for Rapid and Ultrasensitive Detection of Gastric Cancer-Related Biomarkers.,” Int. J. Nanomedicine, vol. 18, pp. 7523–7532, 2023. [CrossRef]
- Foroozandeh, M. Abdouss, H. SalarAmoli, M. Pourmadadi, and F. Yazdian, “An electrochemical aptasensor based on g-C3N4/Fe3O4/PANI nanocomposite applying cancer antigen_125 biomarkers detection,” Process Biochem., vol. 127, pp. 82–91, 2023. [CrossRef]
- M. Li et al., “CePO4/CeO2 heterostructure and enzymatic action of D-Fe2O3 co-amplify luminol-based electrochemiluminescence immunosensor for NSE detection,” Biosens. Bioelectron., vol. 214, p. 114516, 2022. [CrossRef]
- Y. Cao et al., “Application of functional peptides in the electrochemical and optical biosensing of cancer biomarkers,” Chem. Commun., vol. 59, no. 23, pp. 3383–3398, 2023. [CrossRef]
- P. Ranjan et al., “Biosensor-based diagnostic approaches for various cellular biomarkers of breast cancer: A comprehensive review.,” Anal. Biochem., p. 113996, 2020. [CrossRef]
- S. Chupradit et al., “Recent advances in biosensor devices for HER-2 cancer biomarker detection,” Anal. Methods, vol. 14, no. 13, pp. 1301–1310, 2022. [CrossRef]
- T. Liang, Q. Qu, Y. Chang, S. C. B. Gopinath, and X. T. Liu, “Diagnosing ovarian cancer by identifying SCC-antigen on a multiwalled carbon nanotube-modified dielectrode sensor,” Biotechnol. Appl. Biochem., vol. 66, no. 6, pp. 939–944, Nov. 2019. [CrossRef]
- Elhami et al., “Sensitive and Cost-Effective Tools in the Detection of Ovarian Cancer Biomarkers.,” Anal. Sci. Adv., vol. 5, no. 9–10, p. e202400029, Oct. 2024. [CrossRef]
- R. Hu, Z. Mu, F. Gong, M. Qing, Y. Yuan, and L. Bai, “A signal-on electrochemical aptasensor for sensitive detection of human epididymis protein 4 based on functionalized metal–organic framework/ketjen black nanocomposite,” J. Mater. Sci., vol. 58, pp. 1–13, Jun. 2023. [CrossRef]
- Pothipor et al., “Highly sensitive biosensor based on graphene–poly (3-aminobenzoic acid) modified electrodes and porous-hollowed-silver-gold nanoparticle labelling for prostate cancer detection,” Sensors Actuators B Chem., vol. 296, Jun. 2019. [CrossRef]
- P. Jia et al., “DNA precisely regulated Au nanorods/Ag2S quantum dots satellite structure for ultrasensitive detection of prostate cancer biomarker,” Sensors Actuators B Chem., vol. 347, p. 130585, Aug. 2021. [CrossRef]
- S. Mohammadi and Z. Mohammadi, “Functionalized NiFe2O4/mesopore silica anchored to guanidine nanocomposite as a catalyst for synthesis of 4H-chromenes under ultrasonic irradiation,” J. Compos. Compd., vol. 3, no. 7, pp. 84–90, 2021. [CrossRef]
- B. Kavosi, A. Navaee, and A. Salimi, “Amplified fluorescence resonance energy transfer sensing of prostate specific antigen based on aggregation of CdTe QDs/antibody and aptamer decorated of AuNPs-PAMAM dendrimer,” J. Lumin., vol. 204, pp. 368–374, 2018. [CrossRef]
- F. Sharifianjazi et al., “Biosensors and nanotechnology for cancer diagnosis (lung and bronchus, breast, prostate, and colon): a systematic review.,” Biomed. Mater., vol. 17, no. 1, Dec. 2021. [CrossRef]
- J.-H. Zhang, M. Liu, F. Zhou, H.-L. Yan, and Y.-G. Zhou, “Homogeneous Electrochemical Immunoassay Using an Aggregation-Collision Strategy for Alpha-Fetoprotein Detection.,” Anal. Chem., vol. 95, no. 5, pp. 3045–3053, Feb. 2023. [CrossRef]
- T. Thenrajan and J. Wilson, “Biosensors for cancer theranostics,” Biosens. Bioelectron. X, vol. 12, p. 100232, 2022. [CrossRef]
- S. Kumar et al., “Effect of Brownian motion on reduced agglomeration of nanostructured metal oxide towards development of efficient cancer biosensor,” Biosens. Bioelectron., vol. 102, pp. 247–255, 2018. [CrossRef]
- R. Thevendran et al., “Reverse Electrochemical Sensing of FLT3-ITD Mutations in Acute Myeloid Leukemia Using Gold Sputtered ZnO-Nanorod Configured DNA Biosensors,” Biosensors, vol. 12, no. 3. 2022. [CrossRef]
- K. Ratajczak, B. E. Krazinski, A. E. Kowalczyk, B. Dworakowska, S. Jakiela, and M. Stobiecka, “Optical Biosensing System for the Detection of Survivin mRNA in Colorectal Cancer Cells Using a Graphene Oxide Carrier-Bound Oligonucleotide Molecular Beacon.,” Nanomater. (Basel, Switzerland), vol. 8, no. 7, Jul. 2018. [CrossRef]
- Jinqi Deng, Shuai Zhao,Yuan Liu, Chao Liu, and Jiashu Sun ‘’Nanosensors for Diagnosis of Infectious Diseases’’ ,2019. [CrossRef]
- K . Yadav , Rahul Deo Yadav , Heena Tabassum ,” Recent Developments in Nanotechnology-Based Biosensors for the Diagnosis of Coronavirus’’ ,Sarita Malti Arya3 2023. [CrossRef]
- Shawky SM, Bald D, Azzazy HM” Direct detection of unamplifed hepatitis C virus RNA using unmodifed gold nanoparticles’’. Clin Biochem 43(13–14):1163–1168, 2010. [CrossRef]
- Pramanik A, Gao Y, Patibandla S, Mitra D, McCandless MG, Fassero LA et al “The rapid diagnosis and efective inhibition of coronavirus using spike antibody attached gold nanoparticles”. Nanoscale Advances 3(6):1588–15,2021. [CrossRef]
- Ahmed SR, Nagy É, Neethirajan S” Self-assembled star-shaped chiroplasmonic gold nanoparticles for an ultrasensitive chiro-immunosensor for viruses.” RSC Adv 7(65):40849–40857, 2017 . [CrossRef]
- Teengam P, Siangproh W, Tuantranont A, Vilaivan T, Chailapakul O, Henry CS “Multiplex paper-based colorimetric DNA sensor using pyrrolidinyl peptide nucleic acid-induced AgNPs aggregation for detecting MERS-CoV, MTB, and HPV oligonucleotides’’. Anal Chem 89(10):5428–543,2017. [CrossRef]
- Bromberg L, Raduyk S, Hatton TA “Functional magnetic nanoparticles for biodefense and biological threat monitoring and surveillance”. Anal Chem 81(14):5637–5645,2009 . [CrossRef]
- Shanmukh S, Jones L, Driskell J, Zhao Y, Dluhy R, Tripp RA “Rapid and sensitive detection of respiratory virus molecular signatures using a silver nanorod array SERS substrate”. Nano Lett 6(11):2630–2636,2006. [CrossRef]
- Bui, M.-P. N.; Ahmed, S.; Abbas, A. “Single-Digit Pathogen and Attomolar Detection with the Naked Eye Using Liposome-Amplified Plasmonic Immunoassay”. Nano Lett.6239−6246.6239−6246,2015. [CrossRef]
- Peng, M.-P.; Ma, W.; Long, Y.-T. “Alcohol Dehydrogenase-Catalyzed Gold Nanoparticle Seed-Mediated Growth Allows Reliable Detection of Disease Biomarkers with the Naked Eye’’. Anal. Chem.,2015. [CrossRef]
- Kim, J.; Biondi, M. J.; Feld, J. J.; Chan, W. C. W. “Clinical Validation of Quantum Dot Barcode Diagnostic Technology”. ACS Nano , 10, 4742−4753,2016. [CrossRef]
- Ciftci, S.; Canovas, R.; Neumann, F.; Paulraj, T.; Nilsson, M.; Crespo, G. A.; Madaboosi, N. “The sweet detection of rolling circle amplification: Glucose-based electrochemical genosensor for the detection of viral nucleic acid’’. Biosens. Bioelectron. 151, 112002,2020. [CrossRef]
- Kurdekar, A. D.; Avinash Chunduri, L. A.; Manohar, C. S.; Haleyurgirisetty, M. K.; Hewlett, I. K.; Venkataramaniah, K. “Streptavidin-conjugated gold nanoclusters as ultrasensitive fluorescent sensors for early diagnosis of HIV infection”. Sci. Adv.4, eaar6280,2018. [CrossRef]
- Fu, X.; Cheng, Z.; Yu, J.; Choo, P.; Chen, L.; Choo, J. “A SERS-based lateral flow assay biosensor for highly sensitive detection of HIV-1 DNA”. Biosens. Bioelectron.78, 530−537, 2016. [CrossRef]
- Zhou, L.; Huang, J.; Yu, B.; Liu, Y.; You, T. ANovel ‘’Electrochemiluminescence Immunosensor for the Analysis of HIV-1 p24 Antigen Based on P-RGO@Au@Ru-SiO2 Composite”. ACS Appl. Mater. Interfaces 7, 24438−24445 ,2015. [CrossRef]
- Huang, H.; Bai, W.; Dong, C.; Guo, R.; Liu, Z.” An ultrasensitive electrochemical DNA biosensor based on graphene/Au nanorod/polythionine for human papillomavirus DNA detection”. Biosens.Bioelectron. 68, 442−446,2015. [CrossRef]
- Wang, Y.; Qin, Z.; Boulware, D. R.; Pritt, B. S.; Sloan, L. M.; Gonzalez, I. J.; Bell, D.; Rees-Channer, R. R.; Chiodini, P.; Chan, W. ́C. W.; Bischof, J. C. “Thermal Contrast Amplification Reader Yielding 8-Fold Analytical Improvement for Disease Detection with Lateral Flow Assays”. Anal. Chem.88, 11774−11782, 2016. [CrossRef]
- Srivastava, M.; Srivastava, N.; Mishra, P.; Malhotra, B.D.” Prospects of nanomaterials-enabled biosensors for COVID-19 detection”.Sci. Total Environ., 754, 142363,2020. [CrossRef]
- Ishikawa, F.N.; Curreli, M.; Olson, C.A.; Liao, H.-I.; Sun, R.; Roberts, R.W.; Cote, R.J.; Thompson, M.E.; Zhou, C.“ Importance ofControlling Nanotube Density for Highly Sensitive and Reliable Biosensors Functional in Physiological Conditions”. ACS Nano, 4,6914–6922,2010. [CrossRef]
- Naumih M. Noah and Peter M. Ndangili . “Current Trends of Nanobiosensors for Point-of-Care Diagnostics”,2019. [CrossRef]
- Baptista P PE, Eaton P, Doria G, Miranda A, Gomes I, Quaresma P, Franco R “Gold nanoparticles for the development of clinical diagnosis methods”. Anal Bioanal Chem 391:943–50,2008. [CrossRef]
- Kaushik, R. D. Jayant, S. Tiwari, A. Vashist, and M. Nair, “Nano-biosensors to detect beta-amyloid for Alzheimer’s disease management,” Biosensors and Bioelectronics, vol. 80, pp. 273–287, Jun. 2016. [CrossRef]
- G. Pilvenyte et al., “Molecularly imprinted polymers for the recognition of biomarkers of certain neurodegenerative diseases,” J. Pharm. Biomed. Anal., vol. 228, p. 115343, 2023.
- B. B. Apaydın, T. Çamoğlu, Z. C. Canbek Özdil, D. Gezen-Ak, D. Ege, and M. Gülsoy, “Chitosan-enhanced sensitivity of mercaptoundecanoic acid (MUA)- capped gold nanorod based localized surface plasmon resonance (LSPR) biosensor for detection of alpha-synuclein oligomer biomarker in parkinson’s disease.,” Biotechnol. Appl. Biochem., vol. 72, no. 1, pp. 150–163, Feb. 2025.
- Z. Aghili, N. Nasirizadeh, A. Divsalar, S. Shoeibi, and P. Yaghmaei, “A highly sensitive miR-195 nanobiosensor for early detection of Parkinson’s disease.,” Artif. cells, nanomedicine, Biotechnol., vol. 46, no. sup1, pp. 32–40, 2018.
- L. N. Rizalputri et al., “Facile and controllable synthesis of monodisperse gold nanoparticle bipyramid for electrochemical dopamine sensor,” Nanotechnology, vol. 34, no. 5, p. 55502, 2023.
- N. Chauhan, S. Pareek, W. Rosario, R. Rawal, and U. Jain, “An insight into the state of nanotechnology-based electrochemical biosensors for PCOS detection,” Analytical Biochemistry, vol. 687, p. 115412, Apr. 2024. [CrossRef]
- S. Neethirajan, X. Weng, A. Tah, J. O. Cordero, and K. V. Ragavan, “Nano-biosensor platforms for detecting food allergens – New trends,” Sensing and Bio-Sensing Research, vol. 18, pp. 13–30, Apr. 2018. [CrossRef]
- Y. Ma et al., “Nanomaterials in the diagnosis and treatment of ophthalmic diseases,” Nano Today, vol. 54, p. 102117, Feb. 2024. [CrossRef]
- H. Khosravi Ardakani, M. Gerami, M. Chashmpoosh, N. Omidifar, and A. Gholami, “Recent Progress in Nanobiosensors for Precise Detection of Blood Glucose Level,” Biochemistry Research International, vol. 2022, p. 2964705, Jan. 2022. [CrossRef]
- Nan Fu Chiu, Ying Hao Wang, and Chen Yu Chen, “Clinical Application for Screening Down’s Syndrome by Using Carboxylated Graphene Oxide-Based Surface Plasmon Resonance Aptasensors,” International journal of nanomedicine, vol. Volume 15, pp. 8131–8149, Oct. 2020. [CrossRef]
- Yi Zhao, Mingju Pu, Yanan Wang, Liangmin Yu, Xinyu Song, and Zhiyu He, “Application of nanotechnology in acute kidney injury: From diagnosis to therapeutic implications,” Journal of Controlled Release, vol. 336, pp. 233–251, Aug. 2021. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




