Submitted:
24 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
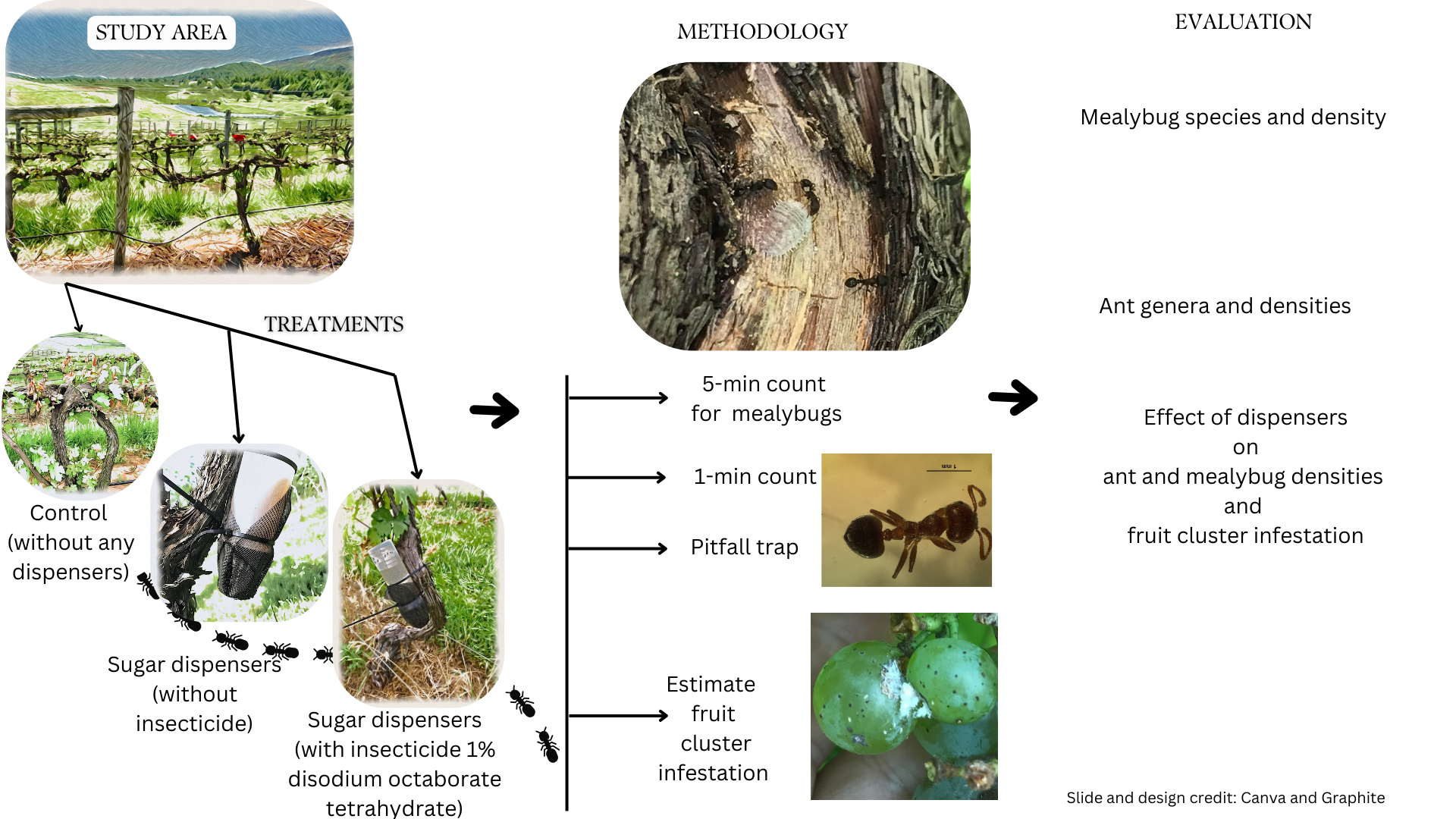
2. Materials and Methods
2.1. Field Sites and Experimental Design
2.2. Sugar Dispensers
2.3. Mealybug Species
2.4. Ant Activity
2.5. Fruit Cluster Infestation
2.6. Data Analysis
3. Results
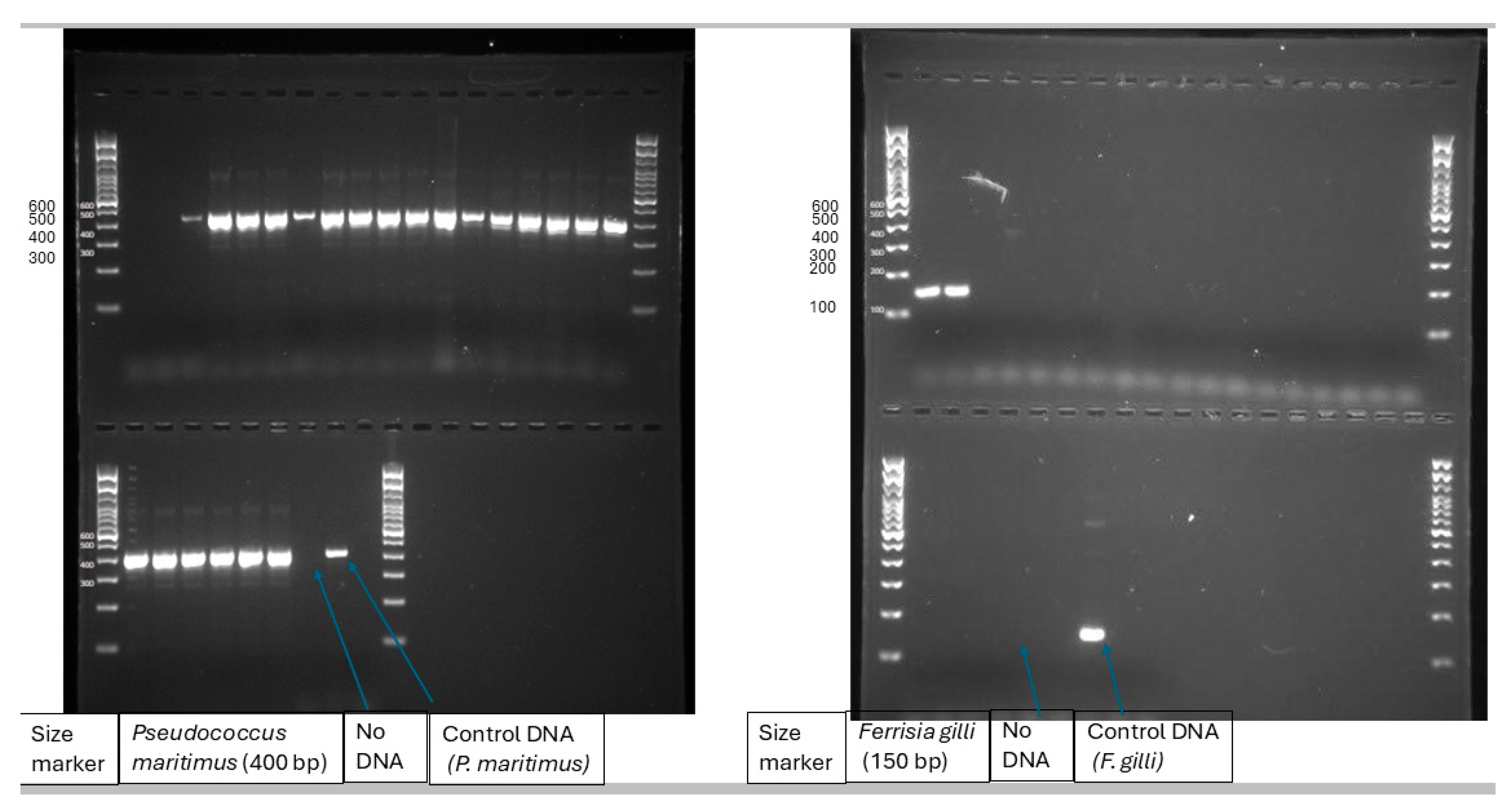
3.1. Mealybug Species in the Vineyard
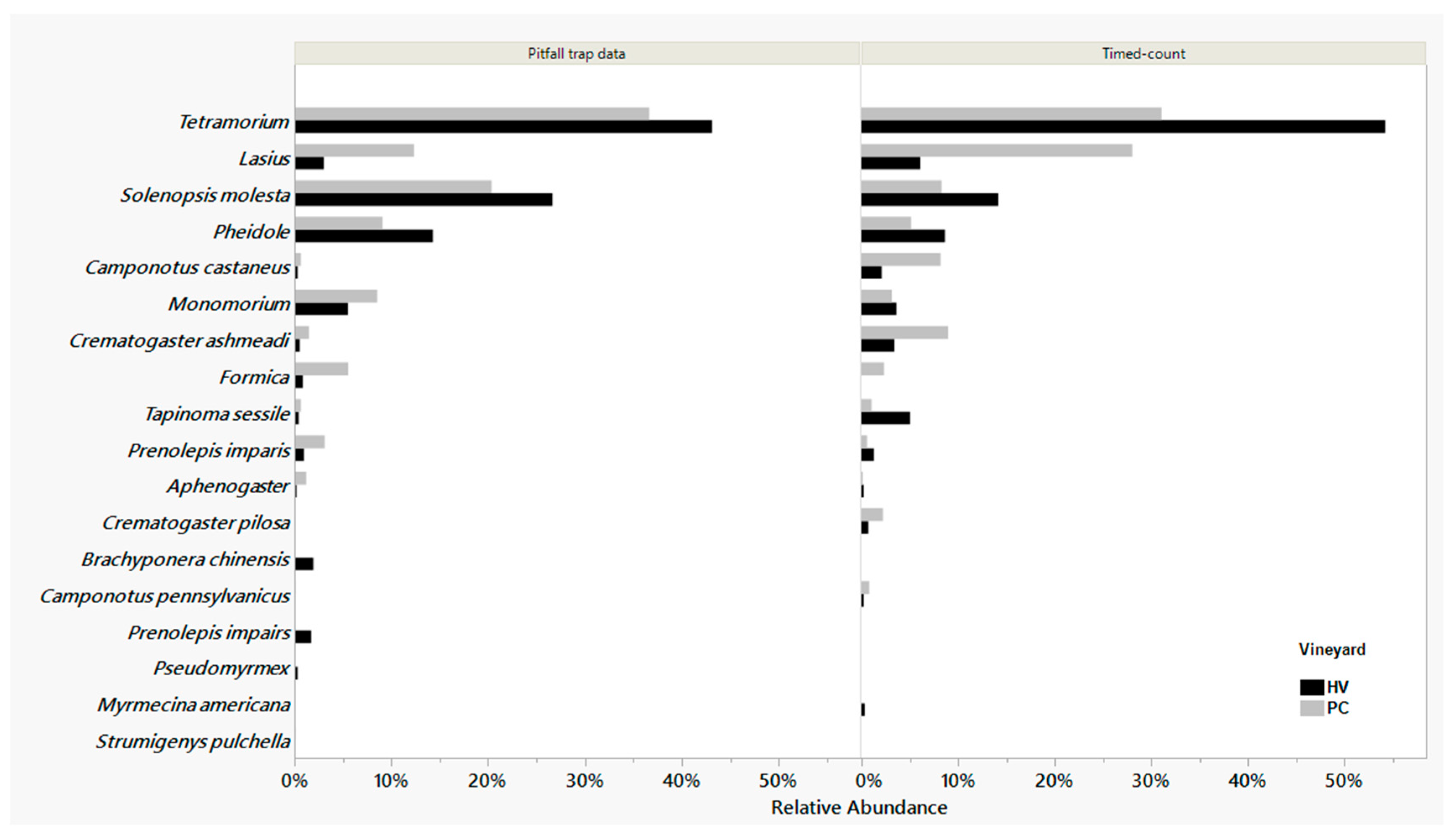
3.2. Ant Species in the Vineyard
3.2.1. Pitfall Trap Data
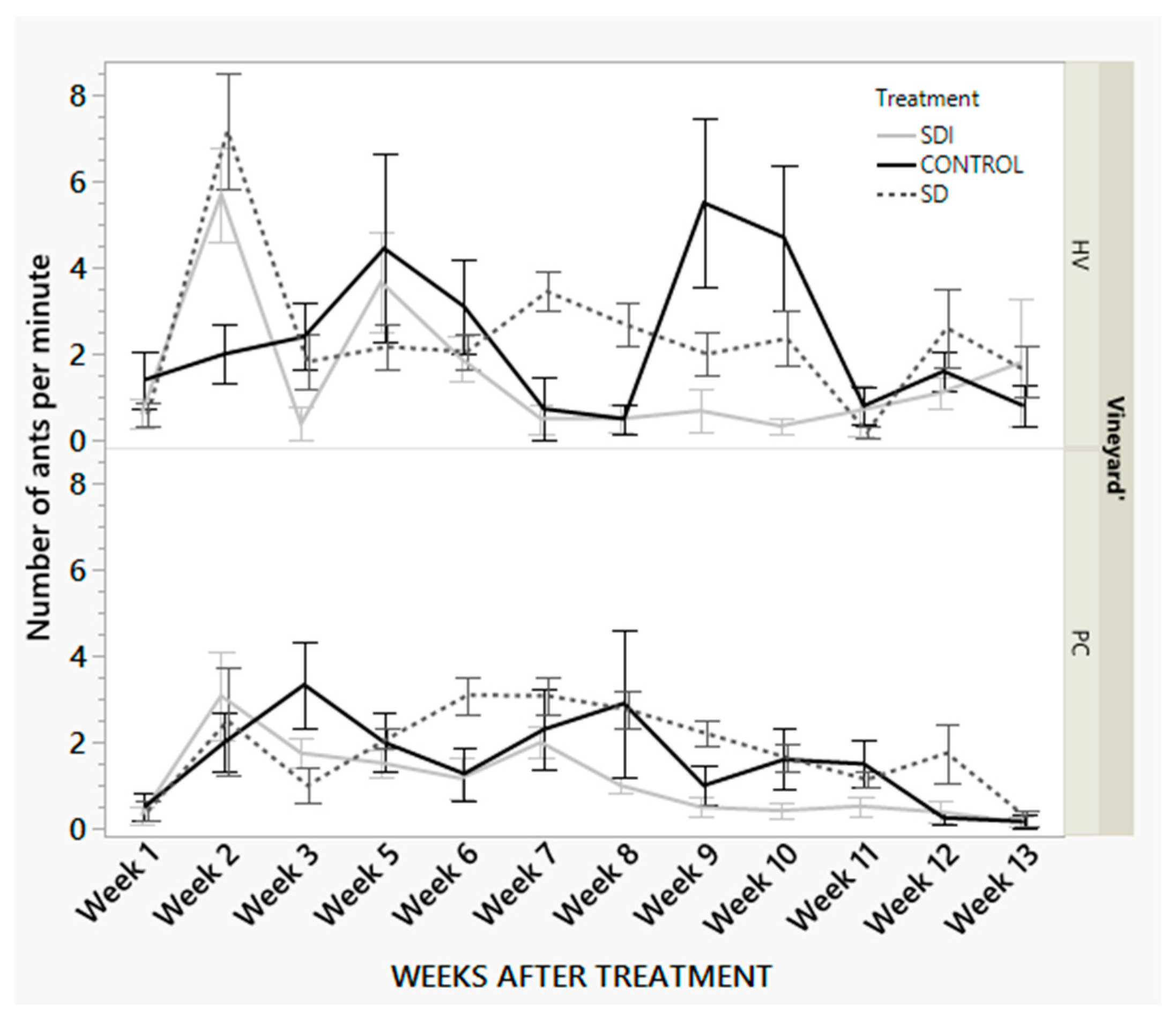
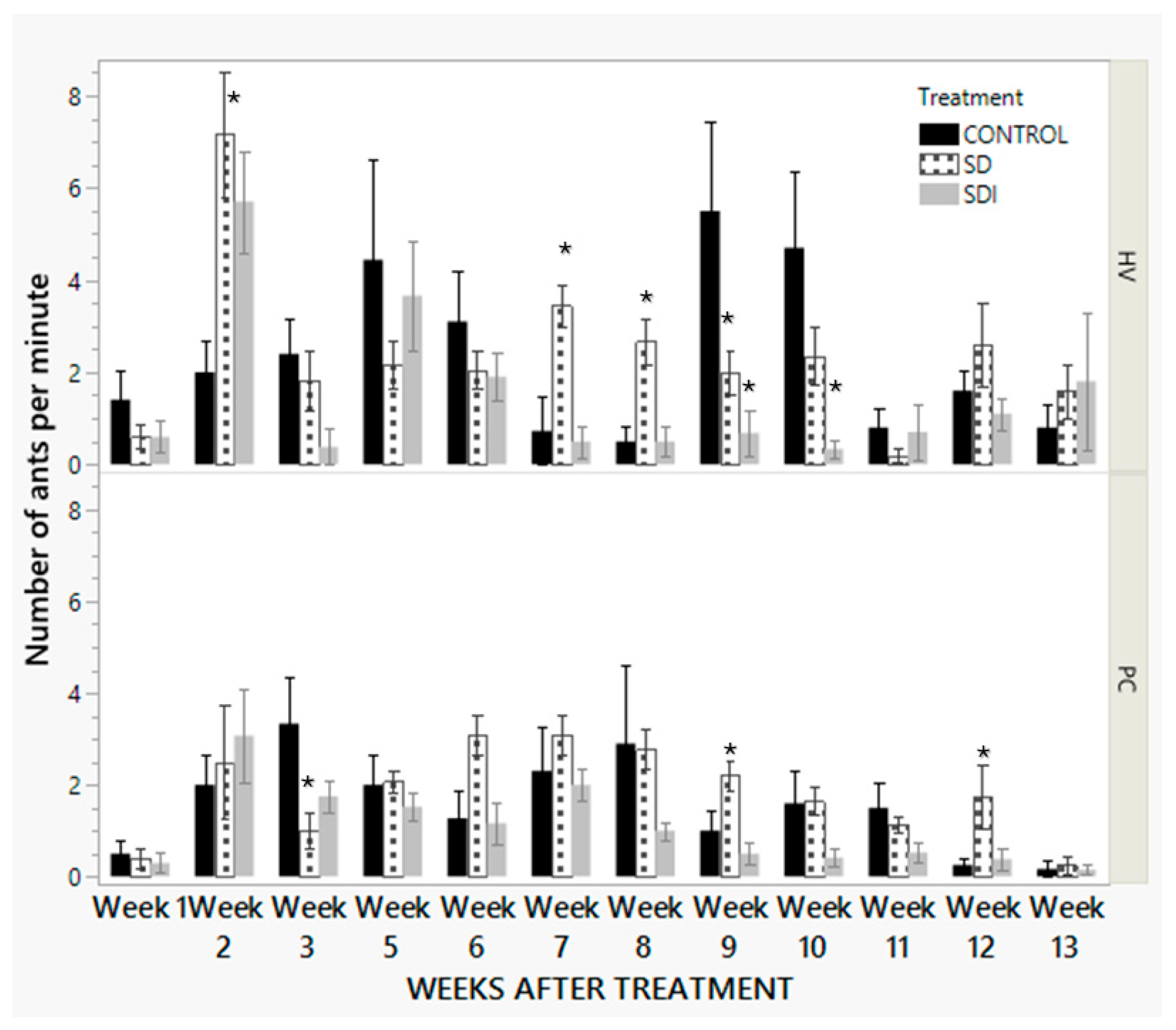
3.2.2. One-Minute Visual Count Data
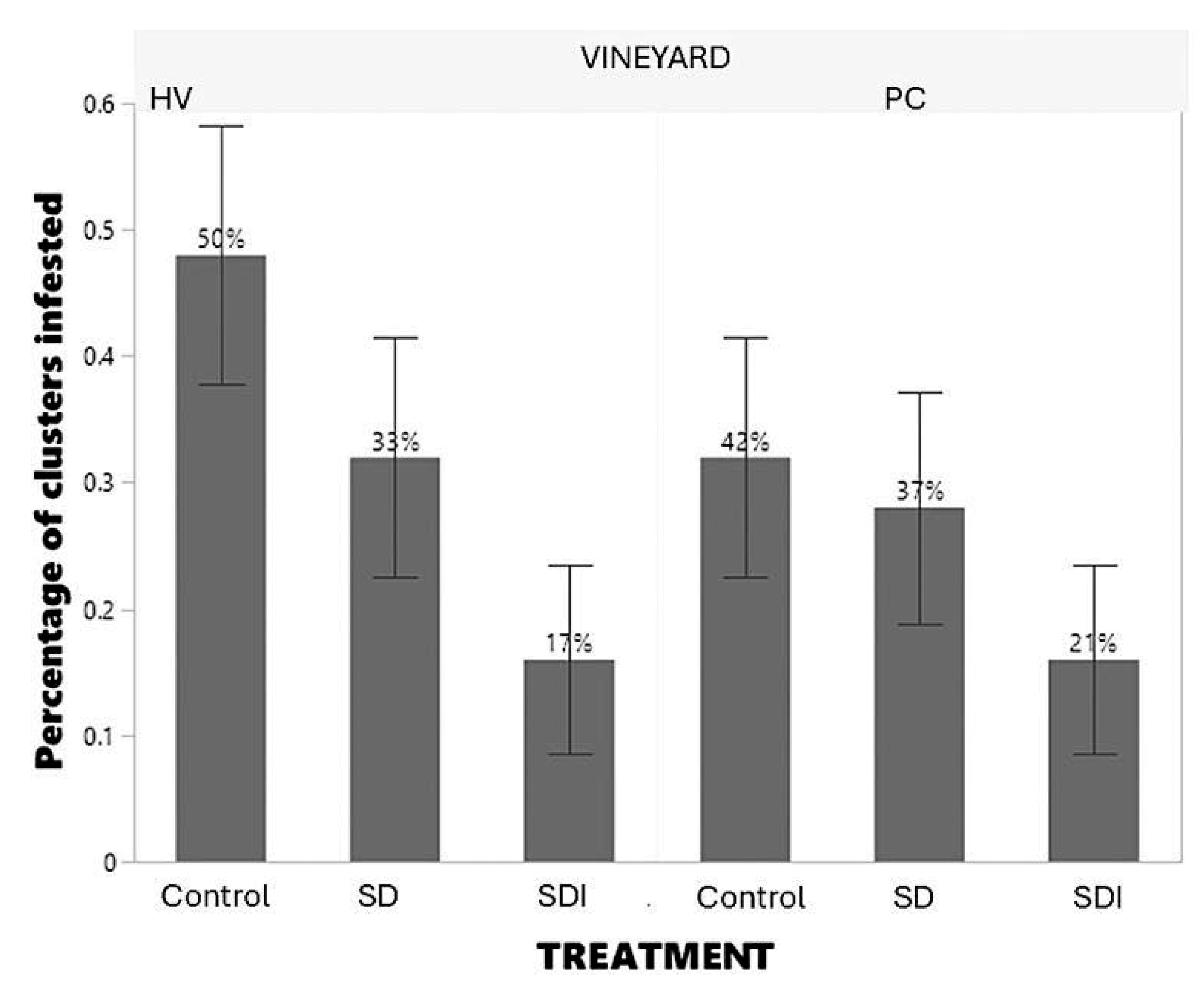
3.2.4. Fruit Cluster Injury Due to the Presence of Mealybugs
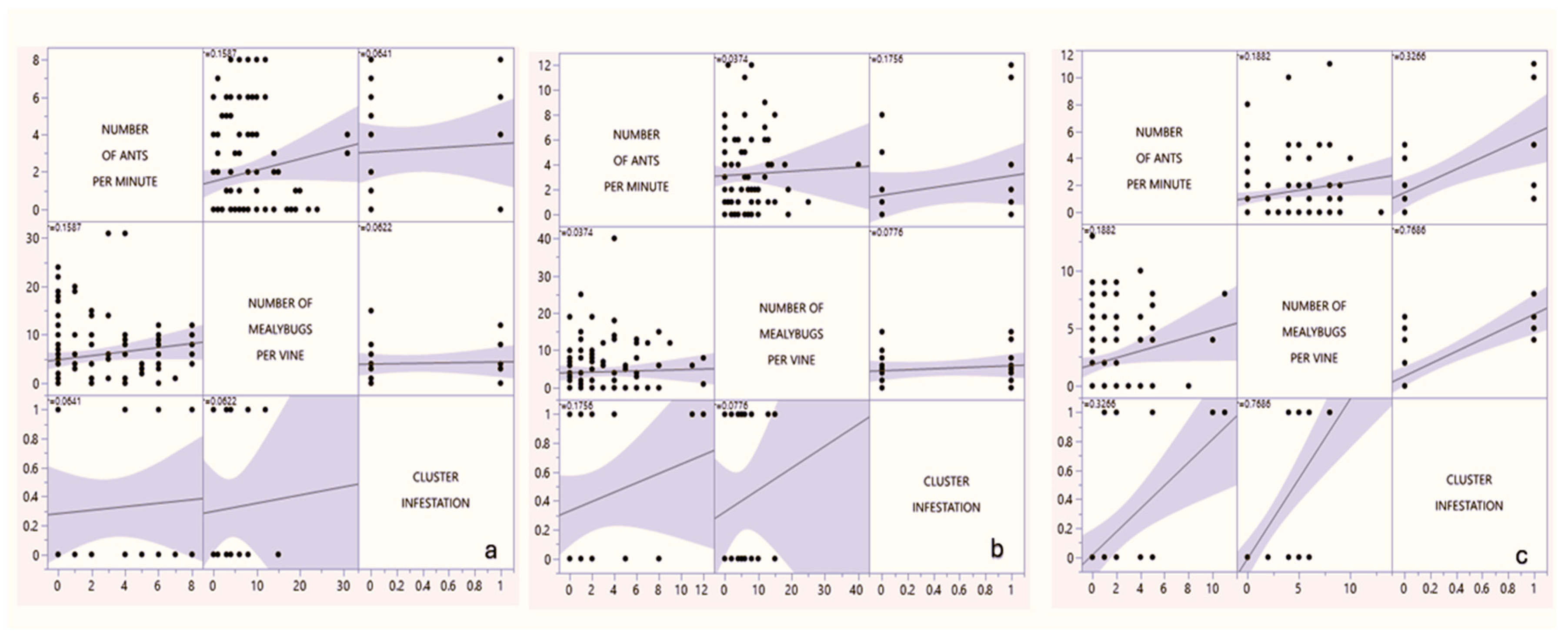
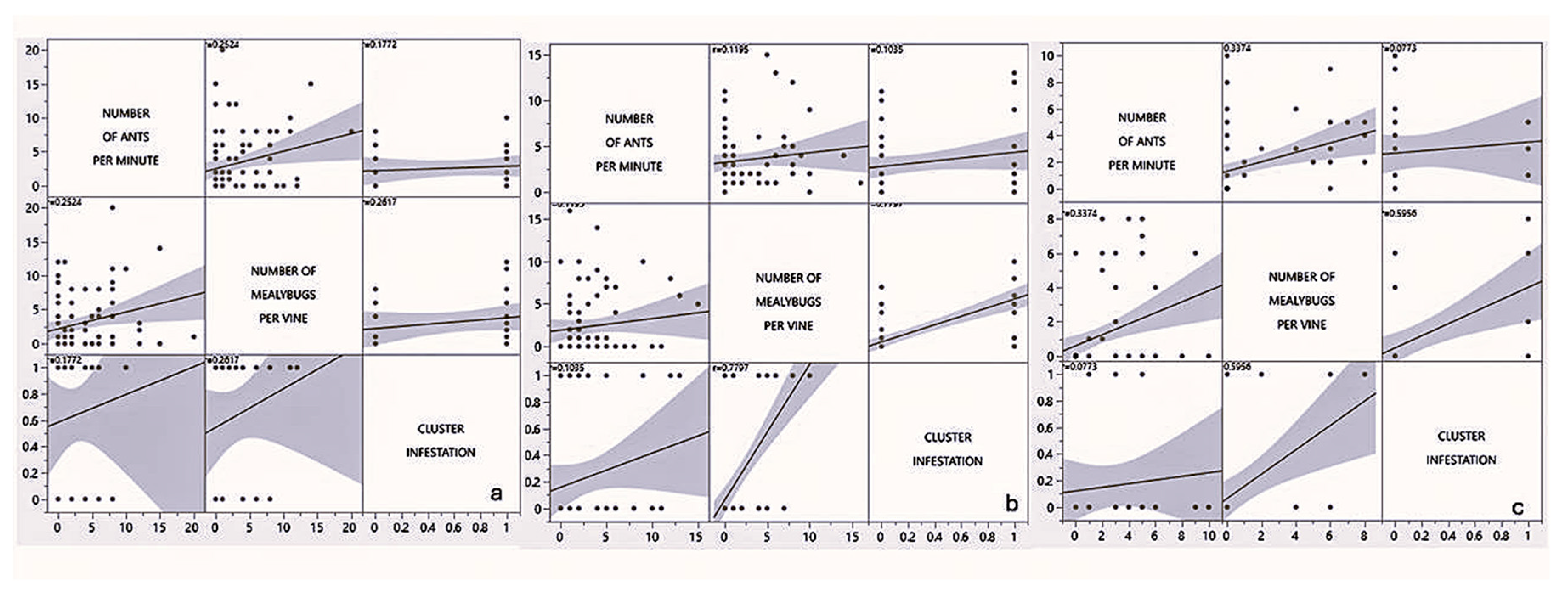
3.2.5. Relation Between Ants, Mealybugs and Cluster Infestation
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mani, M.; Amala, U. Fruit crops: grapevine. In Mealybugs and their Management in Agricultural and Horticultural Crops, Mani, M., Shivaraju, C., Eds.; Springer India: New Delhi, 2016; pp. 329-351.
- Pfeiffer, D.G. Major insect and mite pests of grape in eastern North America. In Wine Grape Production Guide for Eastern North America, Wolf, T.K., Ed.; Natural Resource, Agriculture, and Engineering Service (NRAES): Ithaca, New York, 2008; pp. 241-261.
- Jones, T.J. Grapevine viruses and associated vectors in Virginia: Survey, vector management, and development of efficient grapevine virus testing methods. Virginia Polytechnic Institute and State University, Blacksburg, Virginia, 2016.
- Geiger, C.A.; Daane, K.M. Seasonal movement and distribution of the grape mealybug (Homoptera: Pseudococcidae): developing a sampling program for San Joaquin valley vineyards. Journal of Economic Entomology 2001, 94, 291-301. [CrossRef]
- Grasswitz, T.R.; James, D.G. Movement of grape mealybug, Pseudococcus maritimus, on and between host plants. Entomologia Experimentalis et Applicata 2008, 129, 268-275. [CrossRef]
- Franco, J.C.; Zada, A.; Mendel, Z. Novel approaches for the management of mealybug pests. In Biorational Control of Arthropod Pests: Application and Resistance Management, Ishaaya, I., Horowitz, A.R., Eds.; Springer Netherlands: Dordrecht, 2009; pp. 233-278.
- Daane, K.; Almeida, b.; Bell, V.; Walker, b.; Botton, M.; Fallahzadeh, M.; Mani, M.; Daane, K.; Almeida, R.; Bell, V.; et al. Biology and management of mealybugs in vineyards. In Arthropod Management in Vineyards: Pests, Approaches, and Future Directions, Bostanian, N.J., Vincent, C., Issacs, R., Eds.; Springer Dordrecht, Heidelberg, New York, London, 2012; pp. 271-307.
- Mani, M.; Krishnamoorthy, A.; Shivaraju, C. Biological suppression of major mealybug species on horticultural crops in India. Journal of Horticultural Sciences 2011, 6, 85-100. [CrossRef]
- Mansour, R.; Belzunces, L.P.; Suma, P.; Zappalà, L.; Mazzeo, G.; Grissa-Lebdi, K.; Russo, A.; Biondi, A. Vine and citrus mealybug pest control based on synthetic chemicals. A review. Agronomy for Sustainable Development 2018, 38, 1-20. [CrossRef]
- Styrsky, J.D.; Eubanks, M.D. Ecological consequences of interactions between ants and honeydew-producing insects. Proceedings of Biological sciences / The Royal Society 2006, 274, 151-164. [CrossRef]
- Xu, C.; Su, J.; Qu, X.; Zhou, A. Ant-mealybug mutualism modulates the performance of co-occurring herbivores. Scientific Reports 2019, 9, 13004. [CrossRef]
- Wilder, S.M.; Holway, D.A.; Suarez, A.V.; LeBrun, E.G.; Eubanks, M.D. Intercontinental differences in resource use reveal the importance of mutualisms in fire ant invasions. Proc Natl Acad Sci U S A 2011, 108, 20639-20644. [CrossRef]
- Fernández de Bobadilla, M.; Ramírez, N.M.; Calvo-Agudo, M.; Dicke, M.; Tena, A. Honeydew management to promote biological control. Current Opinion in Insect Science 2024, 61, 101151. [CrossRef]
- Beltrà, A.; Navarro-Campos, C.; Calabuig, A.; Estopà, L.; Wäckers, F.L.; Pekas, A.; Soto, A. Association between ants (Hymenoptera: Formicidae) and the vine mealybug (Hemiptera: Pseudococcidae) in table-grape vineyards in Eastern Spain. Pest Manag Sci 2017, 73, 2473-2480. [CrossRef]
- Daane, K.M.; Sime, K.R.; Hogg, B.N.; Bianchi, M.L.; Cooper, M.L.; Rust, M.K.; Klotz, J.H. Effects of liquid insecticide baits on Argentine ants in California's coastal vineyards. Crop Protection 2006, 25, 592-603. [CrossRef]
- Parrilli, M.; Profeta, M.; Casoli, L.; Gambirasio, F.; Masetti, A.; Burgio, G. Use of sugar dispensers to disrupt ant attendance and improve biological control of mealybugs in vineyard. Insects 2021, 12, 330.
- Perez-Rodriguez, J.; Pekas, A.; Tena, A.; Wäckers, F. Sugar provisioning for ants enhances biological control of mealybugs in citrus. Biological Control 2021, 157, 104573. [CrossRef]
- Nondillo, A.; Andzeiewski, S.; Bello Fialho, F.; Bueno, O.C.; Botton, M. Control of Linepithema micans (Hymenoptera: Formicidae) and Eurhizococcus brasiliensis (Hemiptera: Margarodidae) in vineyards using toxic baits. Journal of Economic Entomology 2016, 109, 1660-1666. [CrossRef]
- Klimes, P.; Borovanska, M.; Plowman, N.S.; Leponce, M. How common is trophobiosis with hoppers (Hemiptera: Auchenorrhyncha) inside ant nests (Hymenoptera: Formicidae)? Novel interactions from New Guinea and a worldwide overview. Myrmecological News 2018, 26, 31-45.
- Rust, M.K.; Reierson, D.A.; Klotz, J.H. Delayed toxicity as a critical factor in the efficacy of aqueous baits for controlling Argentine ants (Hymenoptera: Formicidae). J Econ Entomol 2004, 97, 1017-1024. [CrossRef]
- Wcislo, W.T. Trophallaxis in weakly social bees (Apoidea). Ecological Entomology 2015, 41, 37-39. [CrossRef]
- Daane, K.M.; Cooper, M.L.; Sime, K.R.; Nelson, E.H.; Battany, M.C.; Rust, M.K. Testing baits to control Argentine ants (Hymenoptera: Formicidae) in vineyards. J Econ Entomol 2008, 101, 699-709. [CrossRef]
- Daane, K.M.; Middleton, M.C.; Sforza, R.; Cooper, M.L.; Walton, V.M.; Walsh, D.B.; Zaviezo, T.; Almeida, R.P. Development of a multiplex PCR for identification of vineyard mealybugs. Environ Entomol 2011, 40, 1595-1603. [CrossRef]
- Calixto, A.; Marvin, K.; Dean, A. Sampling ants with pitfall traps using either propylene glycol or water as a preservative. Southwestern Entomologist 2007, 32, 87-91. [CrossRef]
- Ward, D.F.; New, T.R.; Yen, A.L. Effects of pitfall trap spacing on the abundance, richness and composition of invertebrate catches. Journal of Insect Conservation 2001, 5, 47-53. [CrossRef]
- Fisher, B.L.; Cover, S.P.; Kirsch, G.; Kane, J.; Nobile, A. Ants of North America: A guide to the genera, 1 ed.; University of California Press: 2007.
- Beatty, W. Decision support using nonparametric statistics; 2018.
- JMP® 18 JMP Statistical Discovery LLC, Cary, NC, USA, 2024.
- AntPro, K. AntPro Insect Control System ™. Available online: (accessed on.
- Varela, L.G. Grape mealybug (Pseudococcus maritimus) life cycle in the North Coast. University of California Cooperative Extension 2005.
- Greenberg, L.; Klotz, J.H. Argentine Ant (Hymenoptera: Formicidae) trail pheromone enhances consumption of liquid sucrose solution. Journal of Economic Entomology 2000, 93, 119-122. [CrossRef]
- Saarinen, E.V. Acrobat ant Crematogaster ashmeadi Emery (Insecta: Hymenoptera: Formicidae: Myrmicinae); IFAS/University of Florida: Gainsville, Florida, 2021.
- Tschinkel, W. The natural history of the arboreal ant, Crematogaster ashmeadi. Journal of Insect Science 2002, 2, 1-12. [CrossRef]
- MacGown, J.A. Ants (Formicidae) of the southeastern United States. Available online: (accessed on.
- Stock, T.; Gouge, D. Integrated pest management for ants in schools; Pacific Northwest Extension: 2022; p. 10.
- Wilson, E.O. Pheidole in the New World: A dominant, hyperdiverse ant genus; Harvard University Press: 2003.
- Delabie, J.H.C.; Fowler, H.G. Soil and litter cryptic ant assemblages of Bahian cocoa plantations. Pedobiologia 1995, 39, 423-433. [CrossRef]
- Sheikh, A.; Ganaie, G.; Thomas, M.; Bhandari, R.; Rather, Y.A. Ant pitfall trap sampling: An overview. Journal of Entomological Research 2018, 42, 421-436. [CrossRef]
- Daane, K.M.; Sime, K.R.; Fallon, J.; Cooper, M.L. Impacts of Argentine ants on mealybugs and their natural enemies in California’s coastal vineyards. Ecological Entomology 2007, 32, 583-596. [CrossRef]

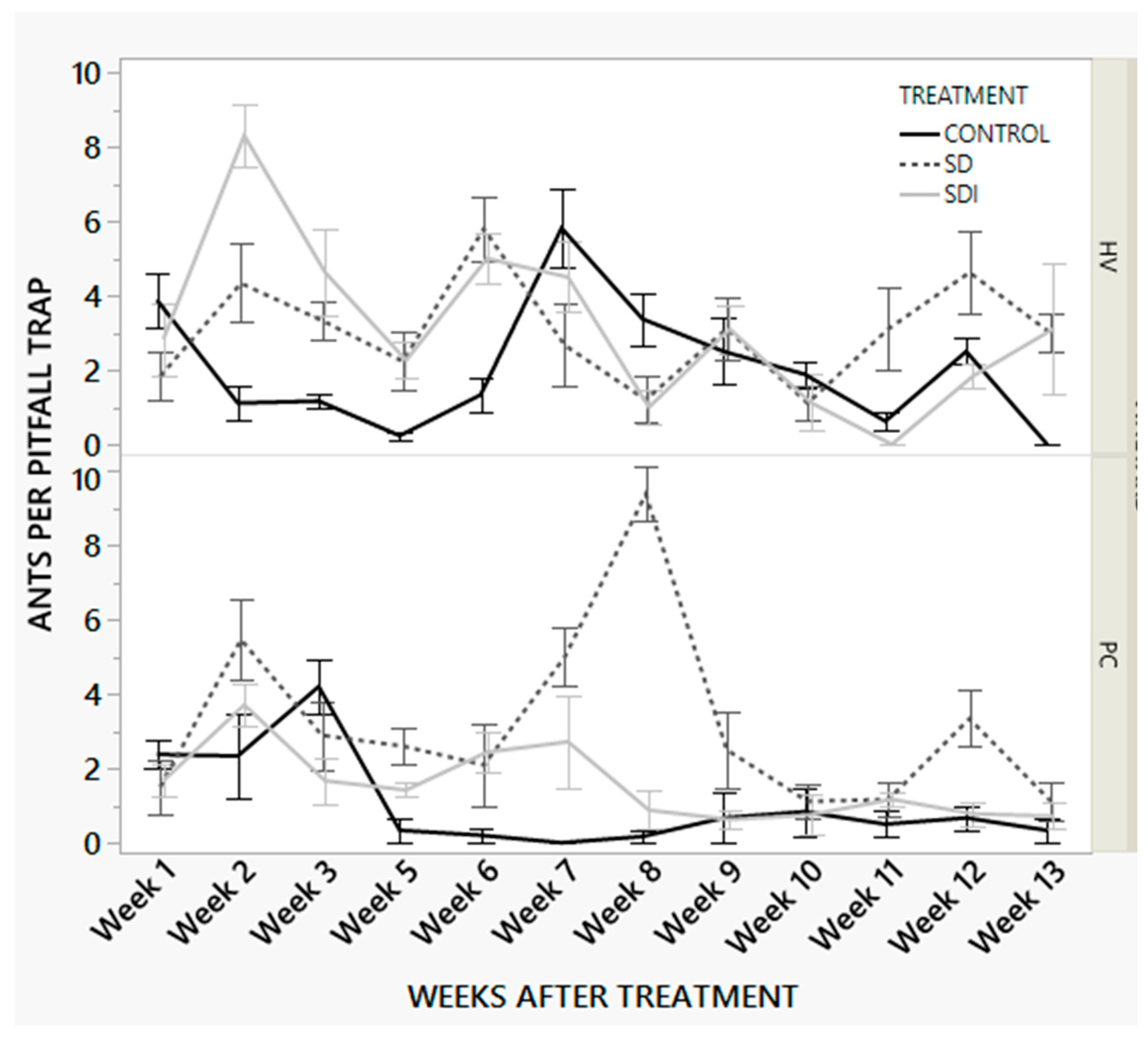
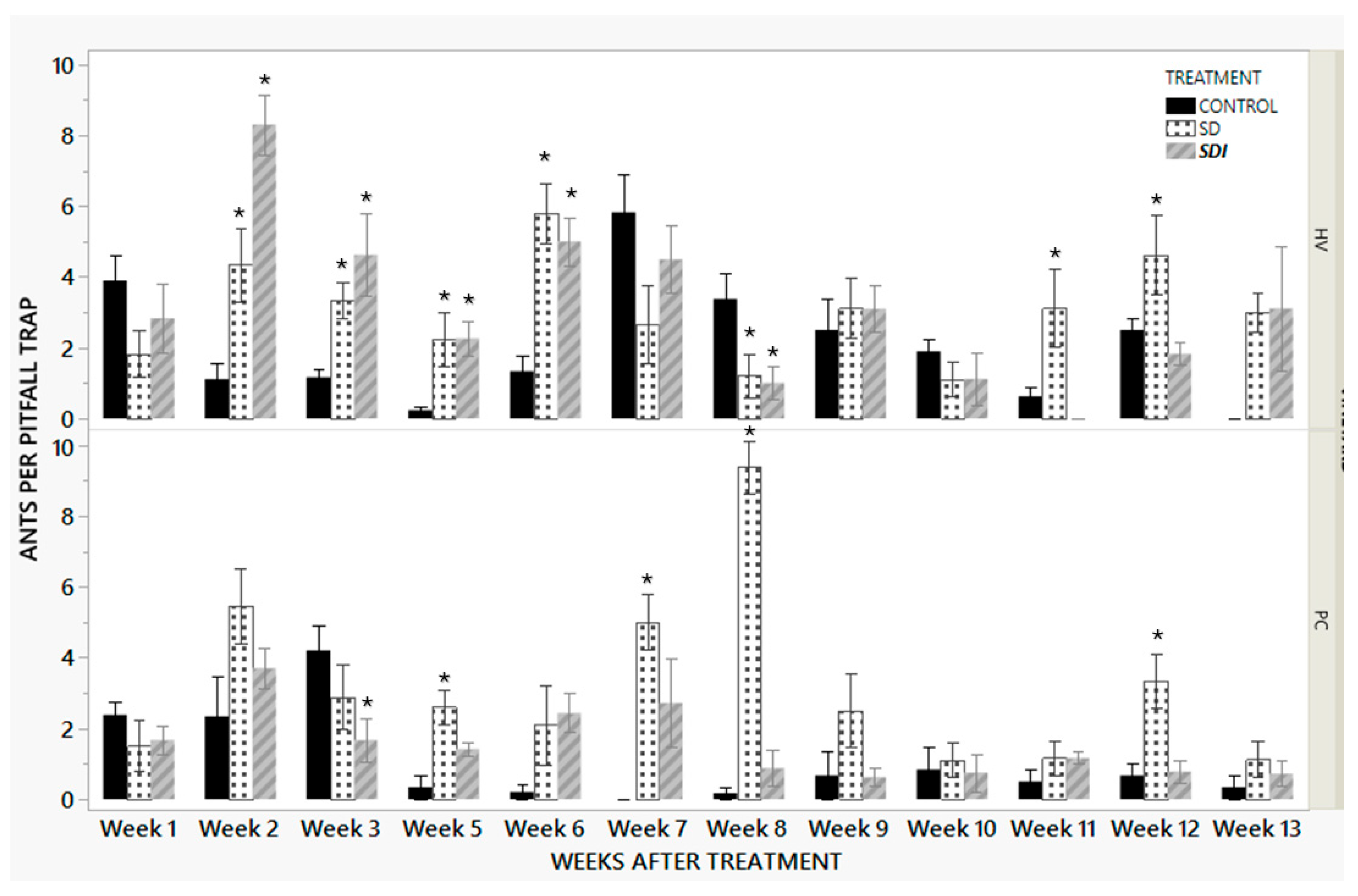
| Factor | Nparm | DF | F | p |
|---|---|---|---|---|
| Treatment | 2 | 2 | 20.52 | <0.0001 |
| Weeks after Treatment | 11 | 11 | 9.28 | <0.0001 |
| Vineyard | 1 | 1 | 16.19 | <0.0001 |
| Vineyard*Weeks after treatment | 11 | 11 | 2.94 | 0.0008 |
| Treatment*Weeks after treatment | 22 | 22 | 3.34 | <0.0001 |
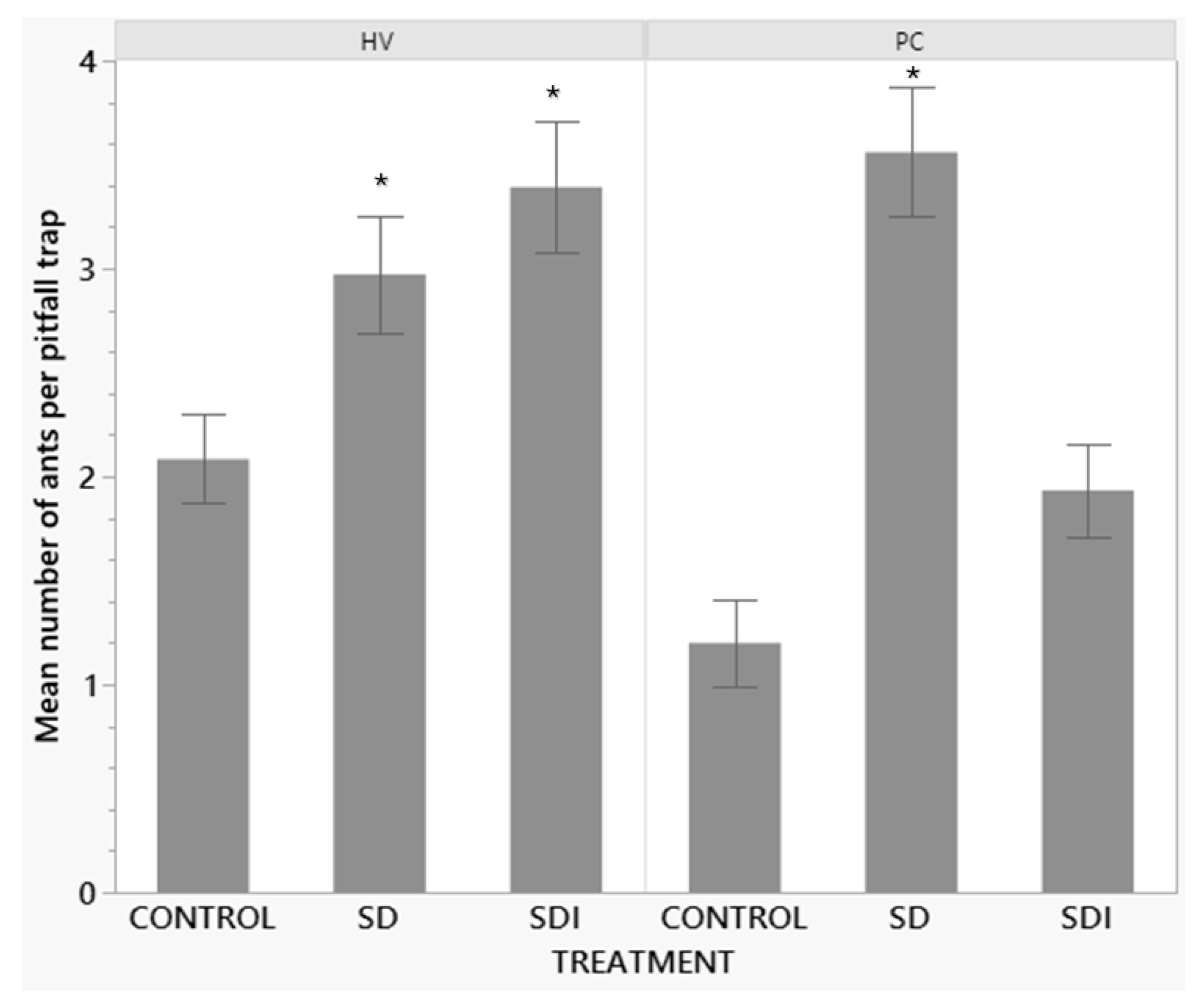
| Treatment | mean±SE | Treatment Comparison | t-Ratio | P |
|---|---|---|---|---|
| Control | 1.68±0.176 | Control-SD | -6.38 | <0.0001 |
| Sugar dispenser without insecticide (SD) | 3.2±0.167 | Control-SDI | -2.89 | 0.0112 |
| Sugar dispenser with insecticide (SDI) | 2.56±0.168 | SD-SDI | 3.58 | 0.0011 |
| Vineyard | Treatment | mean±SE | Treatment Comparison | t-Ratio | P |
|---|---|---|---|---|---|
| HV | Control | 1.68±0.176 | Control-SD | -6.38 | <0.0001 |
| Sugar dispenser without insecticide (SD) | 3.2±0.167 | Control-SDI | -2.89 | 0.0112 | |
| Sugar dispenser with insecticide (SDI) | 2.56±0.168 | SD-SDI | 3.58 | 0.0011 | |
| PC | Control | 1.21±0.241 | Control-SD | -6.72 | <0.0001 |
| Sugar dispenser without insecticide (SD) | 3.4±0.205 | Control-SDI | -1.54 | 0.2729 | |
| Sugar dispenser with insecticide (SDI) | 1.76±0.209 | SD-SDI | 5.59 | <0.0001 |
| Factor | Nparm | DF | F | P |
|---|---|---|---|---|
| Treatment | 2 | 2 | 13.45 | <0.0001 |
| Weeks after Treatment | 11 | 11 | 9.71 | <0.0001 |
| Vineyard | 1 | 1 | 11.23 | 0.0008 |
| Vineyard*Weeks after treatment | 11 | 11 | 3.82 | <0.0001 |
| Treatment*Weeks after treatment | 22 | 22 | 3.10 | <0.0001 |
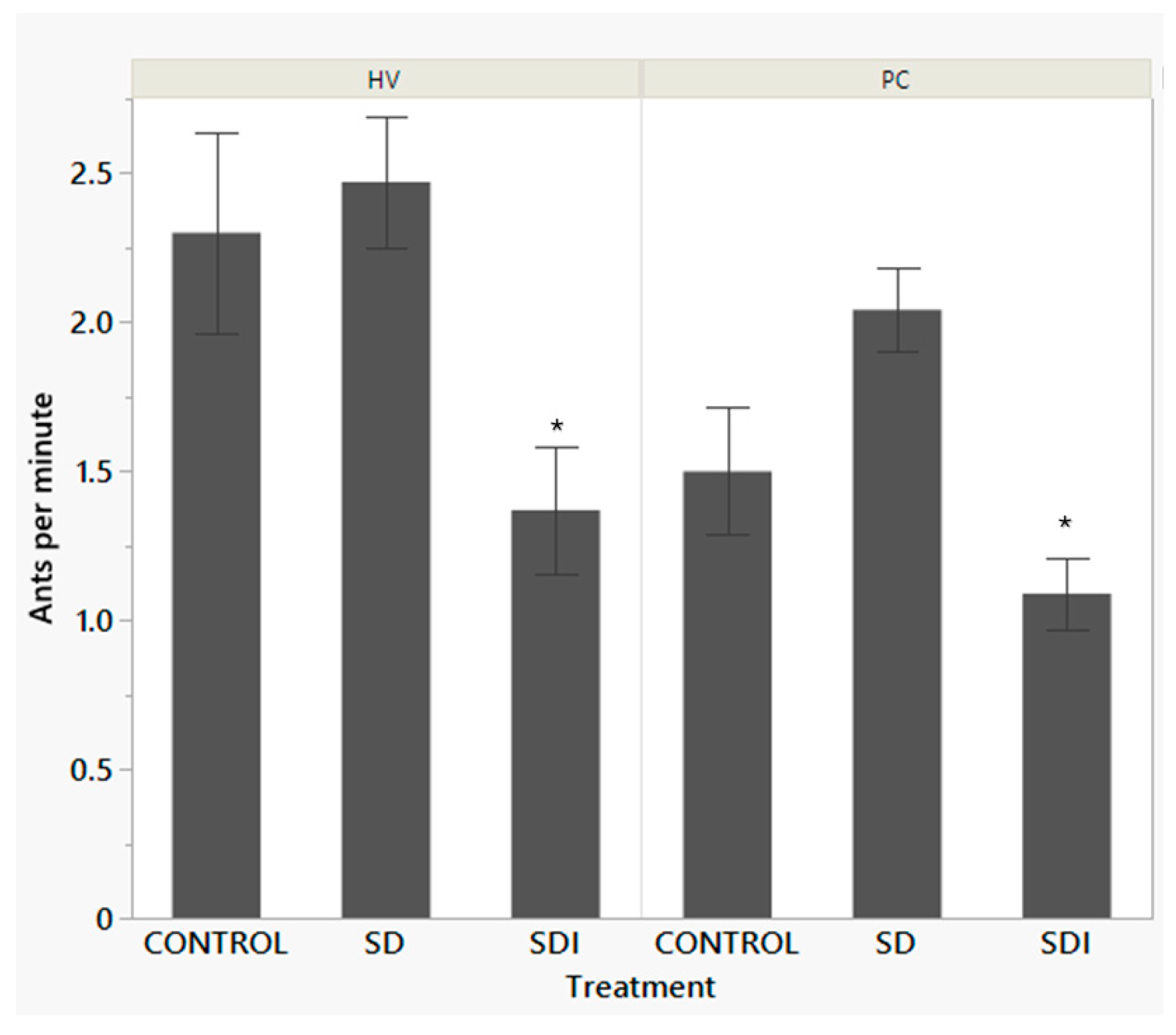
| Treatment | mean±SE | Treatment Comparison | t-Ratio | P |
|---|---|---|---|---|
| Control | 1.93±0.146 | Control-SD | -1.08 | 0.5256 |
| Sugar dispenser without insecticide (SD) | 2.143±0.128 | SDI-Control | -3.57 | 0.0011 |
| Sugar dispenser with insecticide (SDI) | 1.237±0.129 | SDI-SD | -5.00 | <0.0001 |
| wi | Treatment | mean±SE | Treatment Comparison | t-Ratio | P |
|---|---|---|---|---|---|
| HV | Control | 2.33±0.243 | Control-SD | -0.18 | 0.98 |
| Sugar dispenser without insecticide (SD) | 2.39±0.224 | Control-SDI | 2.57 | 0.028 | |
| Sugar dispenser with insecticide (SDI) | 1.49±0.219 | SD-SDI | 2.87 | 0.0119 | |
| PC | Control | 1.57±0.169 | Control-SD | -1.15 | 0.48 |
| Sugar dispenser without insecticide (SD) | 1.82±0.142 | Control-SDI | 2.22 | 0.069 | |
| Sugar dispenser with insecticide (SDI) | 1.06±0.150 | SD-SDI | 3.66 | 0.0008 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




