Submitted:
18 February 2025
Posted:
18 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Report
2.1. Case Description
2.2. Study Design
3. Results
3.1. Improvements in Body Composition, Bone Mineral Density and Dentition
3.2. Amelioration of Stiffness: Joints, Tympanic Membrane, and Arterial Flexibility
3.3. Short-Term Improvement in Growth and Metabolic Aspects
3.4. Reduction in Inflammatory Cytokines
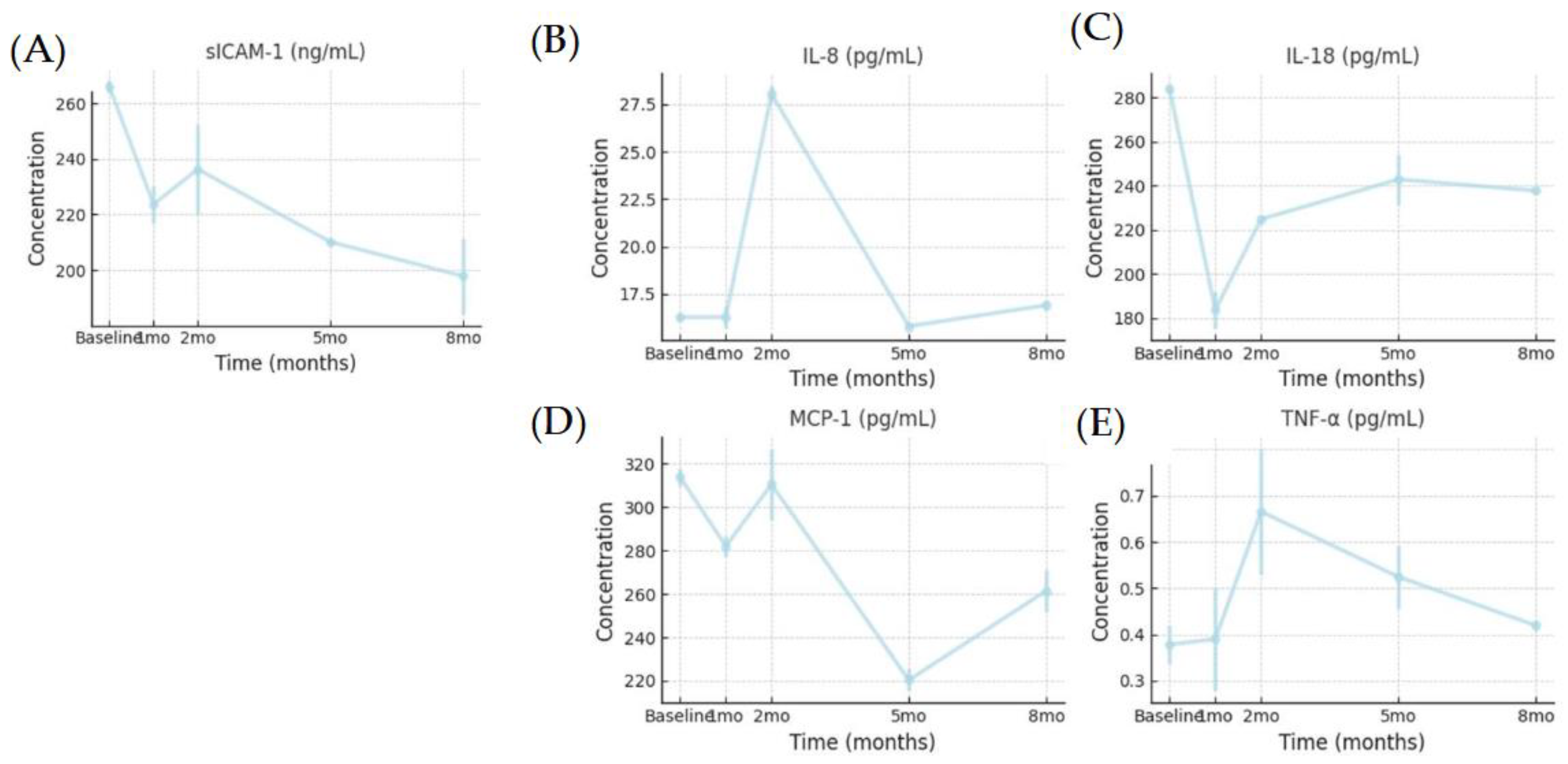
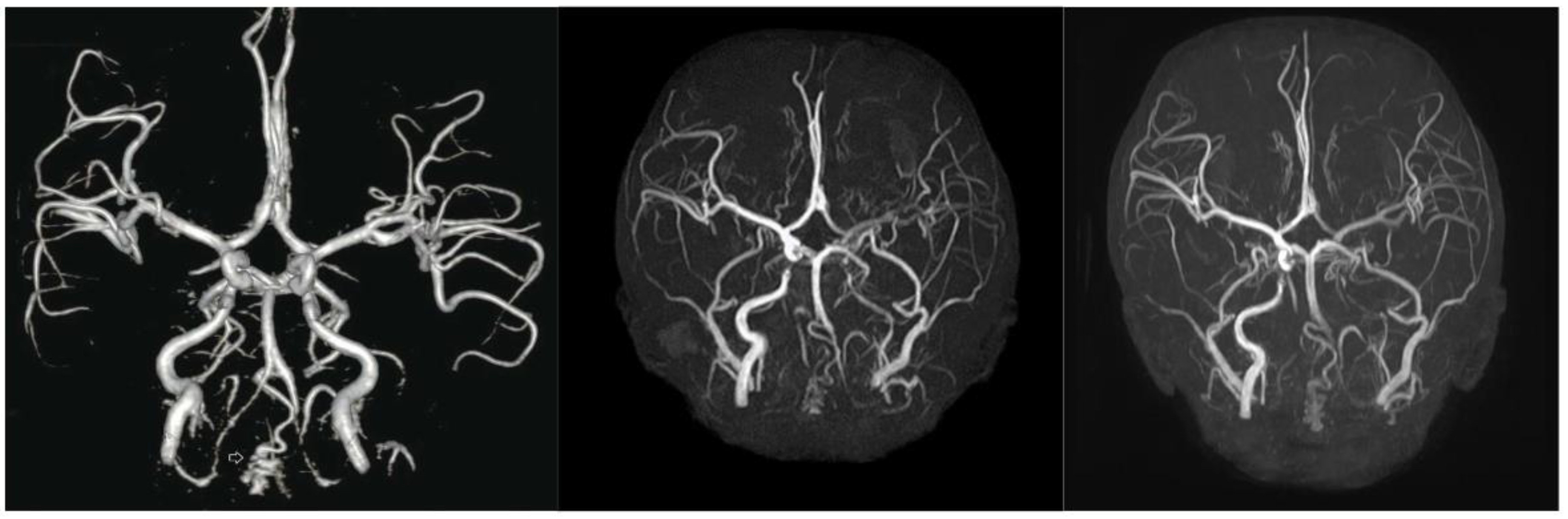
3.5. Lower Efficacy for Atherosclerosis and Cardiovascular Aspects
3.6. Safety of MSC Treatment
- Unfortunately, MSC therapy did not result in an extension of the patient’s lifespan. The study was terminated prematurely at age 8 years and 7 months because of death 10 months after the start of treatment. The cause of death was presumed to be postexercise arrhythmia and was unlikely to be directly related to stem cell treatment. This conclusion is supported by the fact that the initial onset of chest pain occurred approximately one month after the final treatment, and both D-dimer and CK-MB levels remained within normal limits throughout the treatment period. This might be an inevitable consequence of the rapid progression of progeria and its natural course.
- During MSC administration, the only adverse events that occurred within 24 hours of administration were nausea, vomiting and dizziness with the first dose. At the 1st dose, lorazepam was administered concurrently to relieve anxiety. Without lorazepam, the side effects do not appear repeatedly, so it looks less relevant to stem cell therapy. Hand weakness lasting for an hour and a half without headache at 6 days after the first dose might be associated with his basal vascular state, which is less likely with MSC therapy. Frequent epistaxis was thought to be due to the use of clopidogrel. Thirty-six days after the 4th MSC treatment, the patient reported intermittent, stress-induced chest pain lasting 1–2 minutes, which resolved completely. An immediate attempt to conduct additional evaluation was made, but patient refused scheduling delays because various circumstances resulted in unexpected death during daily activities. Other mild adverse reactions associated with infection were noted (Table S1).
4. Discussion
5. Literature Review
| Year | Authors | Subjects | Methodology | Key Findings | Mechanism of Action | Clinical Implications | Limitations/Future Directions |
|---|---|---|---|---|---|---|---|
| 2021[28] | Suh YS, et al. | 13-year-old HGPS patient | Cord blood stem cell infusion | Improved skin elasticity, hair growth, weight gain | Cord blood stem cells may provide trophic support and replace damaged cells | Potential noninvasive treatment for HGPS | Single case study; larger trials needed |
| 2020 [27] | Park J et al. | 13-year-old HGPS | adipose SVF containing MSC | Increased height, weight and IGF-1 | anti-inflammatory effects via paracrine signaling | Proposal for the potential treatment of inflammaging-related diseases | Single case study; larger trials needed |
| 2015 [41] | Lo Cicero A, Nissan X | iPSCs | iPSC modeling of HGPS | Improved understanding of disease mechanisms | iPSCs recreate HGPS cellular environment for study | Improved drug screening platform | Translation to in vivo models needed |
| 2011[42] | Zhang J, et al. | iPSC-derived cells | iPSC differentiation and analysis | Vascular smooth muscle and mesenchymal stem cell defects identified | iPSCs reveal specific cellular defects in HGPS | New targets for therapeutic intervention | Validation in patient samples required |
| 2015[44] | Xiong ZM, et al. | HGPS patient-derived fibroblasts | Treatment with methylene blue (MB); confocal microscopy; western blotting; nuclear fractionation | MB improved nuclear morphology and mitochondrial function; increased solubility of progerin | MB upregulates A-type lamin expression and increases progerin solubility; acts as mitochondrial electron carrier | Potential therapeutic agent for cellular abnormalities in HGPS | Not direct stem cell therapy; in vivo studies needed |
| 2008 [45] | Scaffidi P, Misteli T | Human mesenchymal stem cells (hMSCs) | Expression of progerin in hMSCs; analysis of stem cell function | Discovered misregulation leading to premature aging | Progerin interferes with the function of hMSCs | Progerin activates Notch signaling pathway in hMSCs | Limited to in vitro study; in vivo confirmation needed |
| 2012[43] | Lavasani M et al. | Progeroid mice |
Intraperitoneal injection of young wild-type MDSPCs |
Extended lifespan and healthspan of progeroid mice | Secretion of factors by MDSPCs that improve tissue function | Stem cell transplantation as potential therapy for progeria | Limited to animal model; human studies needed |
| 2011[46] | Liu GH et al.; Ho JC et al. | HGPS patient fibroblasts | Generation of iPSCs from HGPS fibroblasts; Differentiation of iPSCs | iPSCs from HGPS patients lack progerin expression but resume upon differentiation | Reprogramming suppresses progerin expression; differentiation resumes aging-associated phenotypes | iPSCs as a model for studying HGPS and drug screening | Limited to in vitro model; in vivo validation needed |
| 2011 [19] | Rosengardten Y et al. |
HGPS mouse model |
Analysis of stem cell populations and wound healing capacity | HGPS mutation causes adult stem cell depletion and impaired wound healing | Progerin accumulation leads to stem cell exhaustion | Stem cell therapies may be beneficial for HGPS patients | Limited to mouse model; human studies needed |
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data availability statement
Acknowledgments
Conflicts of interest
References
- De Sandre-Giovannoli, A.; Bernard, R.; Cau, P.; Navarro, C.; Amiel, J.; Boccaccio, I.; Lyonnet, S.; Stewart, C.L.; Munnich, A.; Le Merrer, M.; et al. Lamin a truncation in Hutchinson-Gilford progeria. Science 2003, 300, 2055. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, M.; Brown, W.T.; Gordon, L.B.; Glynn, M.W.; Singer, J.; Scott, L.; Erdos, M.R.; Robbins, C.M.; Moses, T.Y.; Berglund, P. Recurrent de novo point mutations in lamin A cause Hutchinson–Gilford progeria syndrome. Nature 2003, 423, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Jansen, T.; Romiti, R. Progeria infantum (Hutchinson-Gilford syndrome) associated with scleroderma-like lesions and acro-osteolysis: a case report and brief review of the literature. Pediatr Dermatol 2000, 17, 282–285. [Google Scholar] [CrossRef]
- Rork, J.F.; Huang, J.T.; Gordon, L.B.; Kleinman, M.; Kieran, M.W.; Liang, M.G. Initial cutaneous manifestations of Hutchinson-Gilford progeria syndrome. Pediatr Dermatol 2014, 31, 196–202. [Google Scholar] [CrossRef]
- Hennekam, R.C. Hutchinson-Gilford progeria syndrome: review of the phenotype. Am J Med Genet A 2006, 140, 2603–2624. [Google Scholar] [CrossRef]
- Merideth, M.A.; Gordon, L.B.; Clauss, S.; Sachdev, V.; Smith, A.C.; Perry, M.B.; Brewer, C.C.; Zalewski, C.; Kim, H.J.; Solomon, B.; et al. Phenotype and course of Hutchinson-Gilford progeria syndrome. N Engl J Med 2008, 358, 592–604. [Google Scholar] [CrossRef]
- Gordon, L.B.; McCarten, K.M.; Giobbie-Hurder, A.; Machan, J.T.; Campbell, S.E.; Berns, S.D.; Kieran, M.W. Disease progression in Hutchinson-Gilford progeria syndrome: impact on growth and development. Pediatrics 2007, 120, 824–833. [Google Scholar] [CrossRef]
- Nevado, R.M.; Hamczyk, M.R.; Gonzalo, P.; Andres-Manzano, M.J.; Andres, V. Premature Vascular Aging with Features of Plaque Vulnerability in an Atheroprone Mouse Model of Hutchinson-Gilford Progeria Syndrome with Ldlr Deficiency. Cells 2020, 9. [Google Scholar] [CrossRef]
- Xu, Q.; Mojiri, A.; Boulahouache, L.; Morales, E.; Walther, B.K.; Cooke, J.P. Vascular senescence in progeria: role of endothelial dysfunction. Eur Heart J Open 2022, 2, oeac047. [Google Scholar] [CrossRef]
- Gordon, L.B.; Massaro, J.; D'Agostino, R.B., Sr.; Campbell, S.E.; Brazier, J.; Brown, W.T.; Kleinman, M.E.; Kieran, M.W.; Progeria Clinical Trials, C. Impact of farnesylation inhibitors on survival in Hutchinson-Gilford progeria syndrome. Circulation 2014, 130, 27–34. [Google Scholar] [CrossRef]
- Prakash, A.; Gordon, L.B.; Kleinman, M.E.; Gurary, E.B.; Massaro, J.; D'Agostino, R., Sr.; Kieran, M.W.; Gerhard-Herman, M.; Smoot, L. Cardiac Abnormalities in Patients With Hutchinson-Gilford Progeria Syndrome. JAMA Cardiol 2018, 3, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Gordon, L.B.; Shappell, H.; Massaro, J.; D'Agostino, R.B., Sr.; Brazier, J.; Campbell, S.E.; Kleinman, M.E.; Kieran, M.W. Association of Lonafarnib Treatment vs No Treatment With Mortality Rate in Patients With Hutchinson-Gilford Progeria Syndrome. JAMA 2018, 319, 1687–1695. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Jeng, L.J.B.; Chefo, S.; Wang, Y.; Price, D.; Li, X.; Wang, J.; Li, R.J.; Ma, L.; Yang, Y.; et al. FDA approval summary for lonafarnib (Zokinvy) for the treatment of Hutchinson-Gilford progeria syndrome and processing-deficient progeroid laminopathies. Genet Med 2023, 25, 100335. [Google Scholar] [CrossRef] [PubMed]
- Gordon, L.B.; Kleinman, M.E.; Miller, D.T.; Neuberg, D.S.; Giobbie-Hurder, A.; Gerhard-Herman, M.; Smoot, L.B.; Gordon, C.M.; Cleveland, R.; Snyder, B.D.; et al. Clinical trial of a farnesyltransferase inhibitor in children with Hutchinson-Gilford progeria syndrome. Proc Natl Acad Sci U S A 2012, 109, 16666–16671. [Google Scholar] [CrossRef]
- Gordon, L.B.; Kleinman, M.E.; Massaro, J.; D'Agostino, R.B., Sr.; Shappell, H.; Gerhard-Herman, M.; Smoot, L.B.; Gordon, C.M.; Cleveland, R.H.; Nazarian, A.; et al. Clinical Trial of the Protein Farnesylation Inhibitors Lonafarnib, Pravastatin, and Zoledronic Acid in Children With Hutchinson-Gilford Progeria Syndrome. Circulation 2016, 134, 114–125. [Google Scholar] [CrossRef]
- Abutaleb, N.O.; Atchison, L.; Choi, L.; Bedapudi, A.; Shores, K.; Gete, Y.; Cao, K.; Truskey, G.A. Lonafarnib and everolimus reduce pathology in iPSC-derived tissue engineered blood vessel model of Hutchinson-Gilford Progeria Syndrome. Sci Rep 2023, 13, 5032. [Google Scholar] [CrossRef]
- Halaschek-Wiener, J.; Brooks-Wilson, A. Progeria of stem cells: stem cell exhaustion in Hutchinson-Gilford progeria syndrome. J Gerontol A Biol Sci Med Sci 2007, 62, 3–8. [Google Scholar] [CrossRef]
- Bridger, J.M.; Kill, I.R. Aging of Hutchinson-Gilford progeria syndrome fibroblasts is characterised by hyperproliferation and increased apoptosis. Exp Gerontol 2004, 39, 717–724. [Google Scholar] [CrossRef]
- Rosengardten, Y.; McKenna, T.; Grochova, D.; Eriksson, M. Stem cell depletion in Hutchinson-Gilford progeria syndrome. Aging Cell 2011, 10, 1011–1020. [Google Scholar] [CrossRef]
- Prolla, T.A. Multiple roads to the aging phenotype: insights from the molecular dissection of progerias through DNA microarray analysis. Mech Ageing Dev 2005, 126, 461–465. [Google Scholar] [CrossRef]
- Dabrowska, S.; Andrzejewska, A.; Janowski, M.; Lukomska, B. Immunomodulatory and Regenerative Effects of Mesenchymal Stem Cells and Extracellular Vesicles: Therapeutic Outlook for Inflammatory and Degenerative Diseases. Front Immunol 2020, 11, 591065. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Ge, J.; Huang, C.; Liu, H.; Jiang, H. Application of mesenchymal stem cell therapy for aging frailty: from mechanisms to therapeutics. Theranostics 2021, 11, 5675–5685. [Google Scholar] [CrossRef] [PubMed]
- Ogay, V.; Sekenova, A.; Li, Y.; Issabekova, A.; Saparov, A. The Therapeutic Potential of Mesenchymal Stem Cells in the Treatment of Atherosclerosis. Curr Stem Cell Res Ther 2021, 16, 897–913. [Google Scholar] [CrossRef]
- Kirwin, T.; Gomes, A.; Amin, R.; Sufi, A.; Goswami, S.; Wang, B. Mechanisms underlying the therapeutic potential of mesenchymal stem cells in atherosclerosis. Regen Med 2021, 16, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Dong, N.; Hong, H.; Qi, J.; Zhang, S.; Wang, J. Mesenchymal Stem Cells: Therapeutic Mechanisms for Stroke. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Drole Torkar, A.; Plesnik, E.; Groselj, U.; Battelino, T.; Kotnik, P. Carotid Intima-Media Thickness in Healthy Children and Adolescents: Normative Data and Systematic Literature Review. Front Cardiovasc Med 2020, 7, 597768. [Google Scholar] [CrossRef]
- Pak, J.; Lee, J.H.; Jeon, J.H.; Kim, Y.B.; Jeong, B.C.; Lee, S.H. Potential Benefits of Allogeneic Haploidentical Adipose Tissue-Derived Stromal Vascular Fraction in a Hutchinson-Gilford Progeria Syndrome Patient. Front Bioeng Biotechnol 2020, 8, 574010. [Google Scholar] [CrossRef]
- Suh, M.R.; Lim, I.; Kim, J.; Yang, P.S.; Choung, J.S.; Sim, H.R.; Ha, S.C.; Kim, M. Efficacy of Cord Blood Cell Therapy for Hutchinson-Gilford Progeria Syndrome-A Case Report. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Murtada, S.I.; Mikush, N.; Wang, M.; Ren, P.; Kawamura, Y.; Ramachandra, A.B.; Li, D.S.; Braddock, D.T.; Tellides, G.; Gordon, L.B.; et al. Lonafarnib improves cardiovascular function and survival in a mouse model of Hutchinson-Gilford progeria syndrome. Elife 2023, 12. [Google Scholar] [CrossRef]
- Olsen, F.J.; Gordon, L.B.; Smoot, L.; Kleinman, M.E.; Gerhard-Herman, M.; Hegde, S.M.; Mukundan, S.; Mahoney, T.; Massaro, J.; Ha, S.; et al. Progression of Cardiac Abnormalities in Hutchinson-Gilford Progeria Syndrome: A Prospective Longitudinal Study. Circulation 2023, 147, 1782–1784. [Google Scholar] [CrossRef]
- Kang, S.M.; Seo, S.; Song, E.J.; Kweon, O.; Jo, A.H.; Park, S.; Woo, T.G.; Kim, B.H.; Oh, G.T.; Park, B.J. Progerinin, an Inhibitor of Progerin, Alleviates Cardiac Abnormalities in a Model Mouse of Hutchinson-Gilford Progeria Syndrome. Cells 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Erdos, M.R.; Cabral, W.A.; Tavarez, U.L.; Cao, K.; Gvozdenovic-Jeremic, J.; Narisu, N.; Zerfas, P.M.; Crumley, S.; Boku, Y.; Hanson, G.; et al. A targeted antisense therapeutic approach for Hutchinson-Gilford progeria syndrome. Nat Med 2021, 27, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Sougawa, Y.; Miyai, N.; Utsumi, M.; Miyashita, K.; Takeda, S.; Arita, M. Brachial-ankle pulse wave velocity in healthy Japanese adolescents: reference values for the assessment of arterial stiffness and cardiovascular risk profiles. Hypertens Res 2020, 43, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Kaur, R.; Kumari, P.; Pasricha, C.; Singh, R. ICAM-1 and VCAM-1: Gatekeepers in various inflammatory and cardiovascular disorders. Clin Chim Acta 2023, 548, 117487. [Google Scholar] [CrossRef]
- Bui, T.M.; Wiesolek, H.L.; Sumagin, R. ICAM-1: A master regulator of cellular responses in inflammation, injury resolution, and tumorigenesis. J Leukoc Biol 2020, 108, 787–799. [Google Scholar] [CrossRef]
- Lin, J.; Kakkar, V.; Lu, X. Impact of MCP-1 in atherosclerosis. Curr Pharm Des 2014, 20, 4580–4588. [Google Scholar] [CrossRef]
- Bahrami, A.; Sathyapalan, T.; Sahebkar, A. The Role of Interleukin-18 in the Development and Progression of Atherosclerosis. Curr Med Chem 2021, 28, 1757–1774. [Google Scholar] [CrossRef]
- Kabat, M.; Bobkov, I.; Kumar, S.; Grumet, M. Trends in mesenchymal stem cell clinical trials 2004-2018: Is efficacy optimal in a narrow dose range? Stem Cells Transl Med 2020, 9, 17–27. [Google Scholar] [CrossRef]
- Kurtzberg, J.; Prockop, S.; Teira, P.; Bittencourt, H.; Lewis, V.; Chan, K.W.; Horn, B.; Yu, L.; Talano, J.A.; Nemecek, E.; et al. Allogeneic human mesenchymal stem cell therapy (remestemcel-L, Prochymal) as a rescue agent for severe refractory acute graft-versus-host disease in pediatric patients. Biol Blood Marrow Transplant 2014, 20, 229–235. [Google Scholar] [CrossRef]
- Shin, H.T.; Lee, S.H.; Yoon, H.S.; Heo, J.H.; Lee, S.B.; Byun, J.W.; Shin, J.; Cho, Y.K.; Chung, E.; Jeon, M.S.; et al. Long-term efficacy and safety of intravenous injection of clonal mesenchymal stem cells derived from bone marrow in five adults with moderate to severe atopic dermatitis. J Dermatol 2021, 48, 1236–1242. [Google Scholar] [CrossRef]
- Lo Cicero, A.; Nissan, X. Pluripotent stem cells to model Hutchinson-Gilford progeria syndrome (HGPS): Current trends and future perspectives for drug discovery. Ageing Res Rev 2015, 24, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lian, Q.; Zhu, G.; Zhou, F.; Sui, L.; Tan, C.; Mutalif, R.A.; Navasankari, R.; Zhang, Y.; Tse, H.F.; et al. A human iPSC model of Hutchinson Gilford Progeria reveals vascular smooth muscle and mesenchymal stem cell defects. Cell Stem Cell 2011, 8, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Lavasani, M.; Robinson, A.R.; Lu, A.; Song, M.; Feduska, J.M.; Ahani, B.; Tilstra, J.S.; Feldman, C.H.; Robbins, P.D.; Niedernhofer, L.J.; et al. Muscle-derived stem/progenitor cell dysfunction limits healthspan and lifespan in a murine progeria model. Nat Commun 2012, 3, 608. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Z.M.; Choi, J.Y.; Wang, K.; Zhang, H.; Tariq, Z.; Wu, D.; Ko, E.; LaDana, C.; Sesaki, H.; Cao, K. Methylene blue alleviates nuclear and mitochondrial abnormalities in progeria. Aging Cell 2016, 15, 279–290. [Google Scholar] [CrossRef]
- Scaffidi, P.; Misteli, T. Lamin A-dependent misregulation of adult stem cells associated with accelerated ageing. Nat Cell Biol 2008, 10, 452–459. [Google Scholar] [CrossRef]
- Liu, G.H.; Barkho, B.Z.; Ruiz, S.; Diep, D.; Qu, J.; Yang, S.L.; Panopoulos, A.D.; Suzuki, K.; Kurian, L.; Walsh, C.; et al. Recapitulation of premature ageing with iPSCs from Hutchinson-Gilford progeria syndrome. Nature 2011, 472, 221–225. [Google Scholar] [CrossRef]

| -2 years | baseline | 2 months | 5 months | 8 months | ||
|---|---|---|---|---|---|---|
| Before treatment | 1 month after 2nd MSC |
3 months after 3rd MSC |
6 months after 3rd MSC |
|||
| Growth | Height (cm) z-score for height |
93.2-4.65 | 101.2-5.41 | 102.1-5.41 | 102.2-5.46 | 103-5.73 |
| Weight (kg) z-score for weight |
11.6-5.56 | 12.1-6.89 | 12.6-6.48 | 12.7-6.3 | 13-6.14 | |
| ALP (U/L) | 178 | 207 | 218 | 166 | 207 | |
| IGF-1 (ng/mL) | 145.5 | 173.1 | 235.6 | 118 | 104.4 | |
| z-score for IGF-1 | -0.15 | 0.03 | 1.32 | -1.24 | -1.49 | |
| IGFBP3 (ng/mL) | 1446 | 1786 | 2664 | 2608 | 2147 | |
| z-score for IGFBP3 | -1.96 | -1.6 | 0.37 | -0.04 | -0.96 | |
| Metabolic | HbA1c (%) | 5.8 | 5.5 | 5.9 | 5.8 | |
| AST/ALT (U/L) | 31/22 | 34/35 | 38/38 | 31/41 | 34/45 | |
| Cholesterol (mg/dL) | 219 | 180 | 165 | 149 | 115 | |
| Cardiac | CK-MB (normal 0-5 ng/ml) | 4.2 | 3.9 | 2.7 | 1.9 | 3.2 |
| D-dimer (normal 0-0.5μg/ml) | 1.67 | 0.40 | 0.37 | 0.28 | ||
| BaPWV (cm/sec) rt/lt | 1113/1228 | 1011/1097 | ||||
| cIMT mean (mm) rt/lt | 0.43/0.36 | 0.47/0.46 | 0.61/0.51 | |||
| max (mm) rt/lt | 0.60/0.52 | 0.60/0.68 | 0.80/0.68 | |||
| TTE- TDI e’(cm/s) (normal>8 cm/s) E/e (normal< 8) |
7 |
8 16.1 |
5.89 12.28 |
|||
| EF (normal 50-70%) | 64.36 | 70 | 56.2 | 56.2 | ||
| FS (normal 25-45%) | 34.38 | 40 | 28.3 | 28.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




