Submitted:
10 February 2025
Posted:
12 February 2025
You are already at the latest version
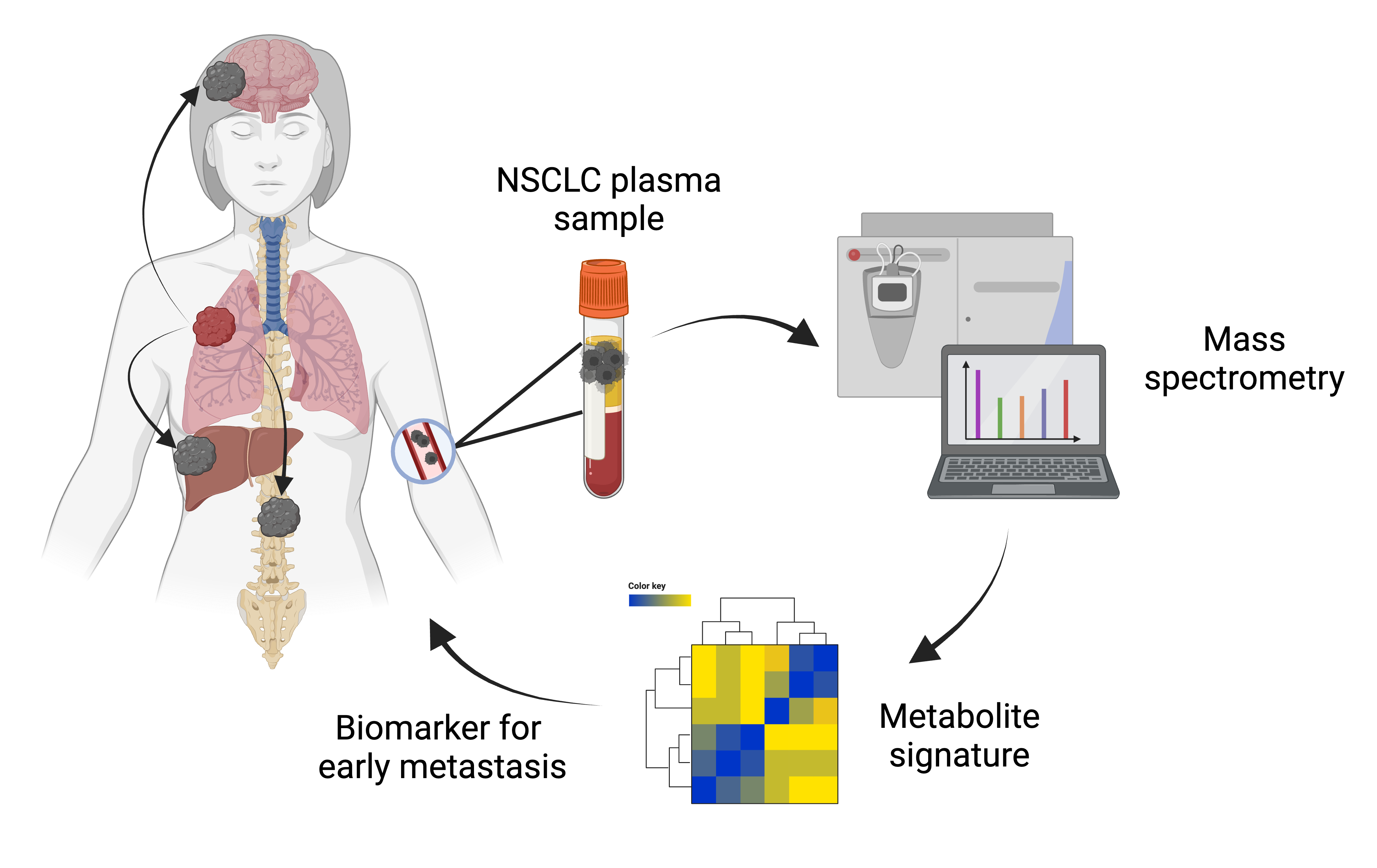
Abstract
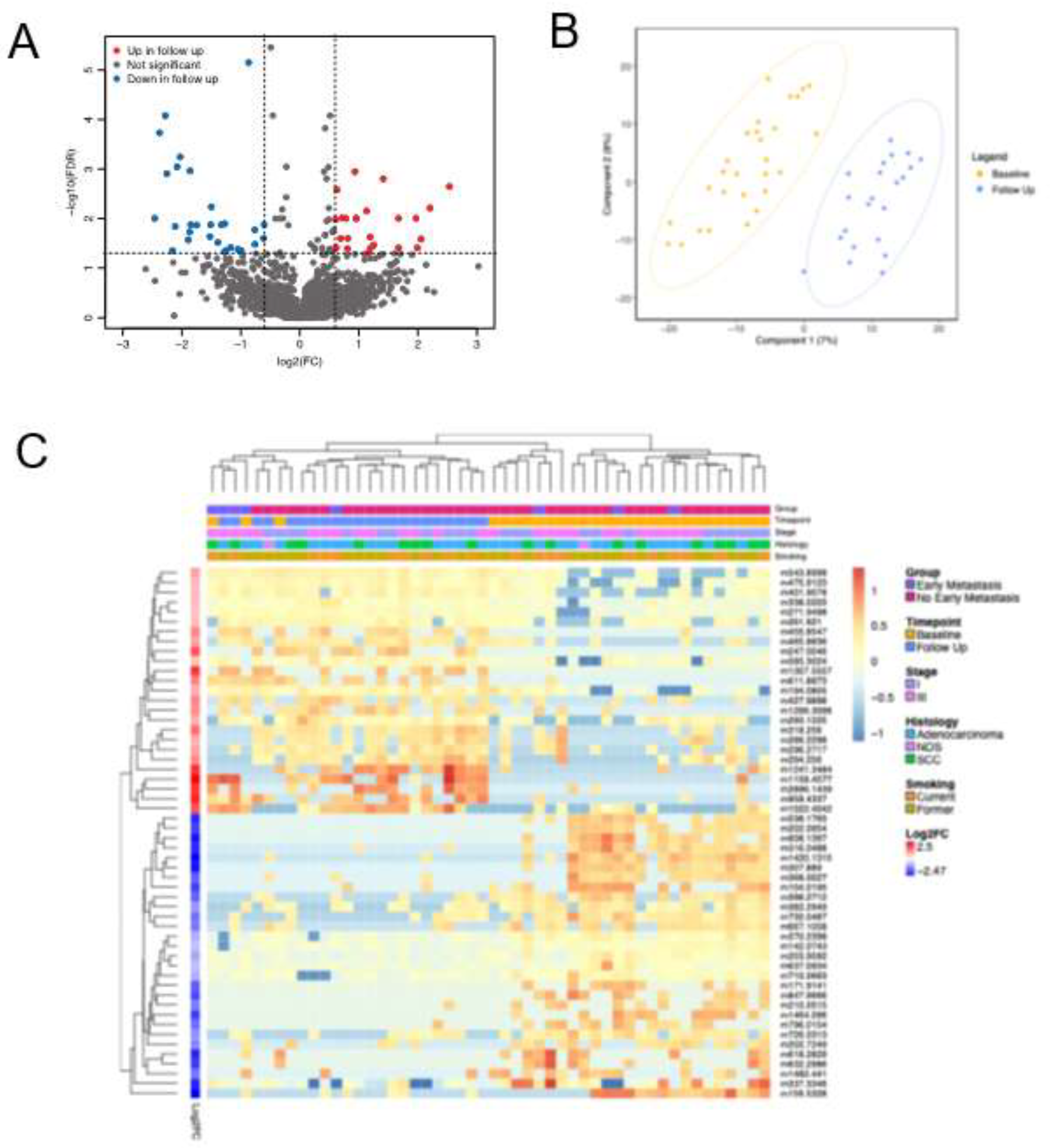
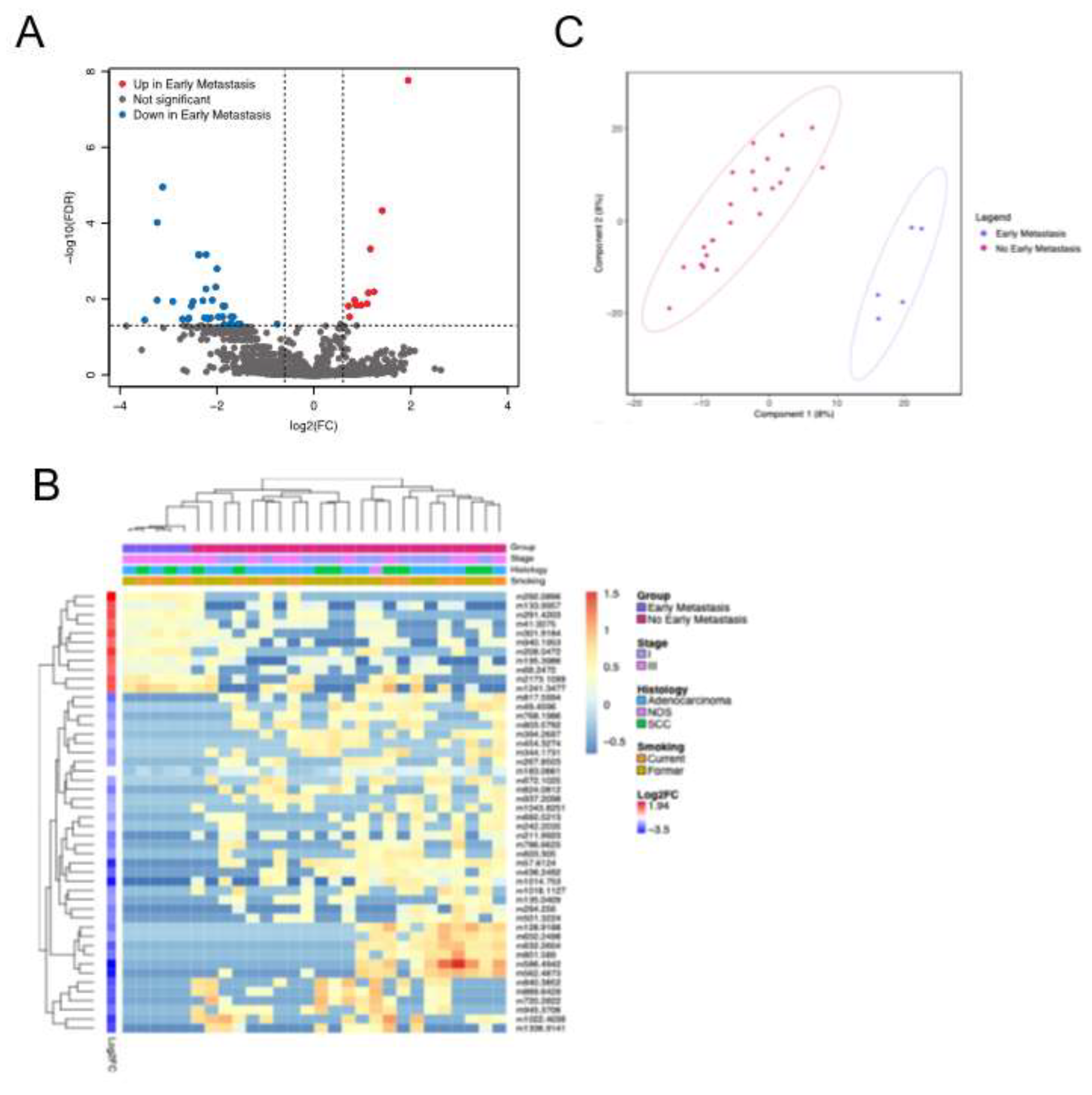
Background/Objectives: The current staging of non-small cell lung cancer (NSCLC) relies on conventional imaging, which lacks sensitivity to detect micrometa-static disease. Functional assessment of NSCLC progression may provide independent information to enhance prediction of metastatic risk.. The objective of this study was to determine if we could identify a metabolomic signature predictive of metastasis in pa-tients with NSCLC treated with definitive radiation. Methods: Plasma samples were collected prospectively from patients enrolled in a clinical trial with non-metastatic NSCLC treated with definitive radiation. Metabolites were extracted and mass spec-trometry-based analysis was performed using a flow injection electrospray (FIE) Fourier transform ion cyclotron resonance (FTICR) mass spectrometry (MS) method. Early metastasis was defined as metastasis within 1 year of radiation treatment. Results: The study cohort included 28 patients. FIE-FITCR produced highly reproducible profiles in technical replicates A total of 48 metabolic features were identified to be different in patients with early metastasis compared to patients without early metastasis (all ad-justed p values < 0.05, Welch’s t-test), including glycerophospholipids, sphingolipids, and fatty acyls. In follow up samples collected after initiation of chemotherapy and radiation treatment, a total of 154 metabolic features were significantly altered in patients who developed early metastasis compared to those who did not. Conclusions: We identified several distinct changes in the metabolic profiles of patients with NSCLC who developed metastatic disease within 1 year of definitive radiation. These findings highlight the potential of metabolomic profiling as a predictive tool for assessing met-astatic risk in NSCLC.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Sample Preparation
2.3. FIE-FTICR Experiment
2.4. MS Data Processing
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
| Characteristics | N (%) |
|---|---|
| Sex | |
| Male | 17 (61) |
| Female | 11 (39) |
| Median age (years) | 69 (52 – 84) |
| Stage | |
| I | 13 (46) |
| III | 15 (54) |
| Histology | |
| Squamous Cell Carcinoma | 10 (36) |
| Adenocarcinoma | 17 (61) |
| Not Otherwise Specified | 1 (3) |
| Metastasis | |
| Early Metastasis | 5 (18) |
| No Early Metastasis | 23 (82) |
| Smoking Status | |
| Current | 11 (39) |
| Former | 16 (61) |
| Non-smoker | 0 (0) |
| Treatment | |
| Radiation Only | 12 (43) |
| Radiation + Chemo | 6 (21) |
| Radiation + Chemo/ICI | 8 (29) |
| Radiation + ICI | 2 (7) |
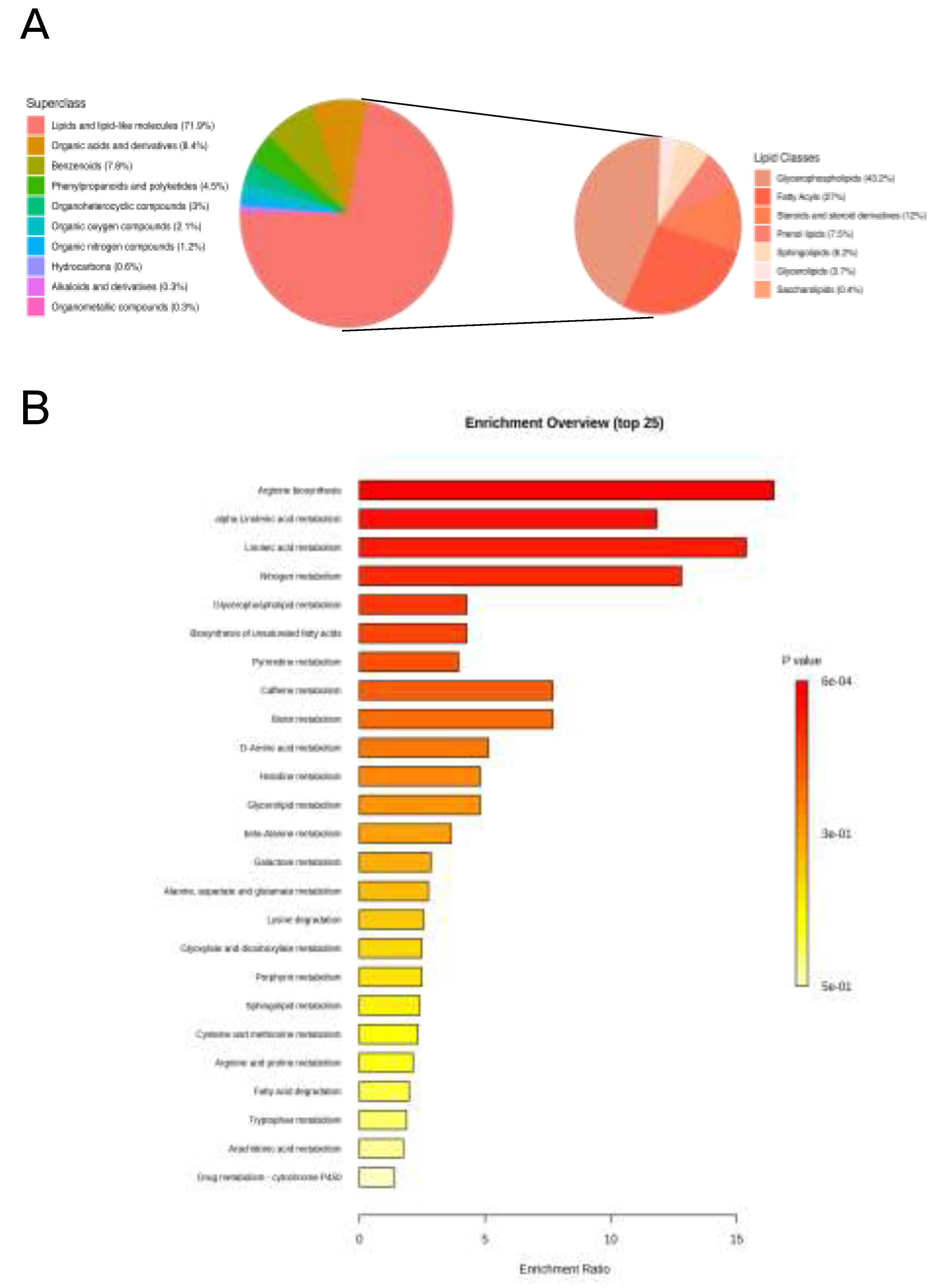
3.2. High-Throughput and Reproducible FIE-FTICR MS-Based Platform
3.3. Serial Analysis of Metabolites Reveal Treatment Effects
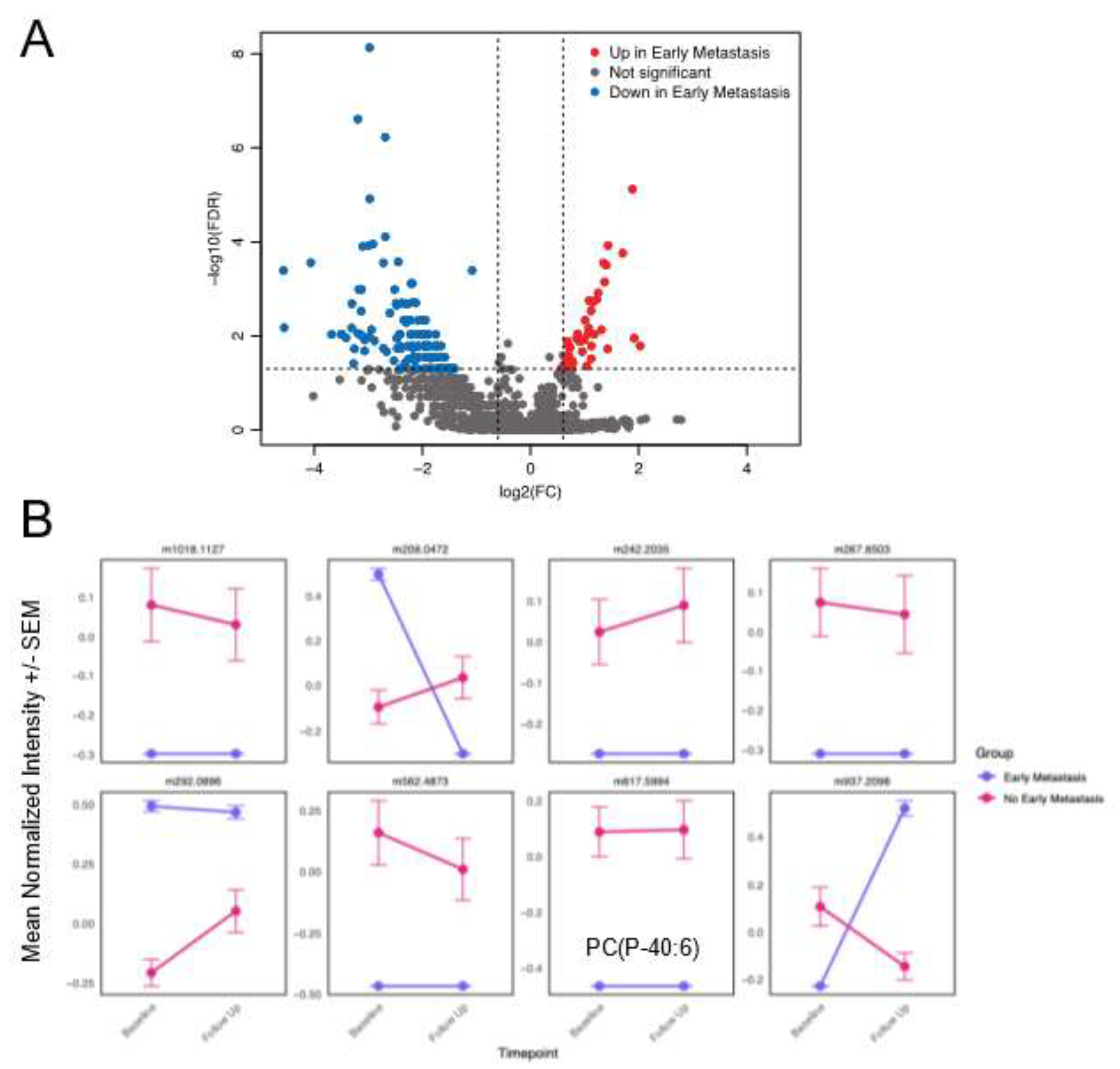
3.4. Distinct Metabolites Characterize Presence of Early Metastases
| Mass | Name | Molecular Formula | Class | Subclass | Parent Level 1 | Log2FC | Adj p |
|---|---|---|---|---|---|---|---|
| 294.256 | FA(19:2) | C19H34O2 | Fatty Acyls | Fatty acids and derivatives | Long-chain fatty acids | -2.174 | 0.034 |
| 344.1731 | 4-ETHOXYCARBONYL-6-ETHYL-5-METHYL-2-(PARA-TOLYL)PERHYDROPYRROLO(3,4-C)PYRROLE-1,3-DIONE (3A,4-CIS-6,6A-TRANS) | C19H24N2O4 | Carboxylic acids and derivatives | Amino acids, peptides, and analogues | Amino acids and derivatives | -1.834 | 0.03 |
| 720.2822 | As-PL(16:0) | C29H58AsO13P | Diarylheptanoids | Linear diarylheptanoids | Linear diarylheptanoids | -2.593 | 0.035 |
| 801.589 | PC 36:02 HETE | C44H84NO9P | Glycerophospholipids | Glycerophosphoserines | 1-alkyl,2-acyl-glycerol-3-phosphoserines | -2.19 | 0.032 |
| 817.5994 | PC(P-40:6) | C48H84NO7P | Glycerophospholipids | Glycerophosphocholines | 1-(1Z-alkenyl),2-acyl-glycerophosphocholines | -2.372 | 0.001 |
| 183.0661 | Phosphocholine | C5H14NO4P | Organonitrogen compounds | Quaternary ammonium salts | Cholines | -0.759 | 0.001 |
| 632.2604 | PI(20:5) | C29H45O13P | Glycerophospholipids | Glycerophosphoinositols | PI (Phosphatidylinositols) | -2.528 | 0.016 |
| 786.6625 | SM(40:1) | C45H91N2O6P | Sphingolipids | Phosphosphingolipids | SM (Sphingomyelins) | -2.11 | 0.032 |
| 660.5213 | SM(d31:1) | C36H73N2O6P | Sphingolipids | Phosphosphingolipids | SM (Sphingomyelins) | -1.872 | 0.015 |
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FIE FTICR MS | flow injection electrospray-Fourier transform ion cyclotron resonance mass spectrometry |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| Log2FC | Log2 fold change |
| PLS-DA | Partial Least-Squares Discriminant Analysis |
References
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA Cancer J Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Goldstraw, P.; Chansky, K.; Crowley, J.; Rami-Porta, R.; Asamura, H.; Eberhardt, W.E.E.; Nicholson, A.G.; Groome, P.; Mitchell, A.; Bolejack, V.; et al. The IASLC Lung Cancer Staging Project: Proposals for Revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM Classification for Lung Cancer. J. Thorac. Oncol. 2016, 11, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Zappa, C.; Mousa, S.A. Non-Small Cell Lung Cancer: Current Treatment and Future Advances. Transl. Lung Cancer Res. 2016, 5, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Goldstraw, P.; Chansky, K.; Crowley, J.; Rami-Porta, R.; Asamura, H.; Eberhardt, W.E.E.; Nicholson, A.G.; Groome, P.; Mitchell, A.; Bolejack, V.; et al. The IASLC Lung Cancer Staging Project: Proposals for Revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM Classification for Lung Cancer. J. Thorac. Oncol. 2016, 11, 39–51. [Google Scholar] [CrossRef]
- Boyd, J.A.; Hubbs, J.L.; Kim, D.W.; Hollis, D.; Marks, L.B.; Kelsey, C.R. Timing of Local and Distant Failure in Resected Lung Cancer: Implications for Reported Rates of Local Failure. J. Thorac. Oncol. 2010, 5, 211–214. [Google Scholar] [CrossRef]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Kurata, T.; Chiappori, A.; Lee, K.H.; De Wit, M.; et al. Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC. N. Engl. J. Med. 2018, 379, 2342–2350. [Google Scholar] [CrossRef]
- Garassino, M.C.; Gadgeel, S.; Speranza, G.; Felip, E.; Esteban, E.; Dómine, M.; Hochmair, M.J.; Powell, S.F.; Bischoff, H.G.; Peled, N.; et al. Pembrolizumab Plus Pemetrexed and Platinum in Nonsquamous Non–Small-Cell Lung Cancer: 5-Year Outcomes From the Phase 3 KEYNOTE-189 Study. J. Clin. Oncol. 2023, 41, 1992–1998. [Google Scholar] [CrossRef]
- Kratzer, T.B.; Bandi, P.; Freedman, N.D.; Smith, R.A.; Travis, W.D.; Jemal, A.; Siegel, R.L. Lung Cancer Statistics, 2023. Cancer 2024, 130, 1330–1348. [Google Scholar] [CrossRef]
- Jiao, X.; Krasna, M.J. Clinical Significance of Micrometastasis in Lung and Esophageal Cancer: A New Paradigm in Thoracic Oncology. Ann. Thorac. Surgery 2002, 74, 278–284. [Google Scholar] [CrossRef]
- Elia, I.; Doglioni, G.; Fendt, S.-M. Metabolic Hallmarks of Metastasis Formation. Trends Cell Biol. 2018, 28, 673–684. [Google Scholar] [CrossRef]
- Wei, Q.; Qian, Y.; Yu, J.; Wong, C.C. Metabolic Rewiring in the Promotion of Cancer Metastasis: Mechanisms and Therapeutic Implications. Oncogene 2020, 39, 6139–6156. [Google Scholar] [CrossRef] [PubMed]
- Bergers, G.; Fendt, S.-M. The Metabolism of Cancer Cells during Metastasis. Nat. Rev. Cancer 2021, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, K.; Morii, E. Metabolic Reprogramming of Cancer Cells during Tumor Progression and Metastasis. Metabolites 2021, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Duan, Z.; Li, Z.; Ge, F.; Wei, R.; Kong, L. The Significance of Glycolysis in Tumor Progression and Its Relationship with the Tumor Microenvironment. Front. Pharmacol. 2022, 13, 1091779. [Google Scholar] [CrossRef]
- Bamji-Stocke, S.; Berkel, V. van; Miller, D.M.; Frieboes, H.B. A Review of Metabolism-Associated Biomarkers in Lung Cancer Diagnosis and Treatment. Metabolomics 2018, 14, 81. [Google Scholar] [CrossRef]
- Fahrmann, J.F.; Kim, K.; DeFelice, B.C.; Taylor, S.L.; Gandara, D.R.; Yoneda, K.Y.; Cooke, D.T.; Fiehn, O.; Kelly, K.; Miyamoto, S. Investigation of Metabolomic Blood Biomarkers for Detection of Adenocarcinoma Lung Cancer. Cancer Epidemiol. Biomarkers Prev. 2015, 24, 1716–1723. [Google Scholar] [CrossRef]
- Lim, S.L.; Jia, Z.; Lu, Y.; Zhang, H.; Ng, C.T.; Bay, B.H.; Shen, H.M.; Ong, C.N. Metabolic Signatures of Four Major Histological Types of Lung Cancer Cells. Metabolomics 2018, 14, 118. [Google Scholar] [CrossRef]
- Hori, S.; Nishiumi, S.; Kobayashi, K.; Shinohara, M.; Hatakeyama, Y.; Kotani, Y.; Hatano, N.; Maniwa, Y.; Nishio, W.; Bamba, T.; et al. A Metabolomic Approach to Lung Cancer. Lung Cancer 2011, 74, 284–292. [Google Scholar] [CrossRef]
- Zheng, L.; Hu, F.; Huang, L.; Lu, J.; Yang, X.; Xu, J.; Wang, S.; Shen, Y.; Zhong, R.; Chu, T.; et al. Association of Metabolomics with PD-1 Inhibitor plus Chemotherapy Outcomes in Patients with Advanced Non-Small-Cell Lung Cancer. J. Immunother. Cancer 2024, 12, e008190. [Google Scholar] [CrossRef]
- Mei, L.; Zhang, Z.; Li, X.; Yang, Y.; Qi, R. Metabolomics Profiling in Prediction of Chemo-Immunotherapy Efficiency in Advanced Non-Small Cell Lung Cancer. Front. Oncol. 2023, 12. [Google Scholar] [CrossRef]
- Brown, S.C.; Kruppa, G.; Dasseux, J.-L. Metabolomics Applications of FT-ICR Mass Spectrometry. Mass Spectrom. Rev. 2005, 24, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Wancewicz, B.; Zhu, Y.; Fenske, R.J.; Weeks, A.M.; Wenger, K.; Pabich, S.; Daniels, M.; Punt, M.; Nall, R.; Peter, D.C.; et al. Metformin Monotherapy Alters the Human Plasma Lipidome Independent of Clinical Markers of Glycemic Control and Cardiovascular Disease Risk in a Type 2 Diabetes Clinical Cohort. J. Pharmacol. Exp. Ther. 2023, 386, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Truchan, N.A.; Fenske, R.J.; Sandhu, H.K.; Weeks, A.M.; Patibandla, C.; Wancewicz, B.; Pabich, S.; Reuter, A.; Harrington, J.M.; Brill, A.L.; et al. Human Islet Expression Levels of Prostaglandin E2 Synthetic Enzymes, But Not Prostaglandin EP3 Receptor, Are Positively Correlated with Markers of β-Cell Function and Mass in Nondiabetic Obesity. ACS Pharmacol. Transl. Sci. 2021, 4, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Schaid, M.D.; Harrington, J.M.; Kelly, G.M.; Sdao, S.M.; Merrins, M.J.; Kimple, M.E. EP3 Signaling Is Decoupled from the Regulation of Glucose-Stimulated Insulin Secretion in β-Cells Compensating for Obesity and Insulin Resistance. Islets 2023, 15, 2223327. [Google Scholar] [CrossRef]
- Zhu, Y.; Wancewicz, B.; Schaid, M.; Tiambeng, T.N.; Wenger, K.; Jin, Y.; Heyman, H.; Thompson, C.J.; Barsch, A.; Cox, E.D.; et al. Ultrahigh-Resolution Mass Spectrometry-Based Platform for Plasma Metabolomics Applied to Type 2 Diabetes Research. J. Proteome Res. 2021, 20, 463–473. [Google Scholar] [CrossRef]
- Baschnagel, A.M.; Flakus, M.J.; Wallat, E.M.; Wuschner, A.E.; Chappell, R.J.; Bayliss, R.A.; Kimple, R.J.; Christensen, G.E.; Reinhardt, J.M.; Bassetti, M.F.; et al. A Phase 2 Randomized Clinical Trial Evaluating 4-Dimensional Computed Tomography Ventilation-Based Functional Lung Avoidance Radiation Therapy for Non-Small Cell Lung Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2024, 119, 1393–1402. [Google Scholar] [CrossRef]
- Ruiying, C.; Zeyun, L.; Yongliang, Y.; Zijia, Z.; Ji, Z.; Xin, T.; Xiaojian, Z. A Comprehensive Analysis of Metabolomics and Transcriptomics in Non-Small Cell Lung Cancer. PLoS One 2020, 15, e0232272. [Google Scholar] [CrossRef]
- Xu, Y.; Ding, K.; Peng, Z.; Ding, L.; Li, H.; Fan, Y. Evaluating for Correlations between Specific Metabolites in Patients Receiving First-Line or Second-Line Immunotherapy for Metastatic or Recurrent NSCLC: An Exploratory Study Based on Two Cohorts. Mole. Cancer Ther. 2024, 23, 733–742. [Google Scholar] [CrossRef]
- Rocha, C.M.; Barros, A.S.; Goodfellow, B.J.; Carreira, I.M.; Gomes, A.; Sousa, V.; Bernardo, J.; Carvalho, L.; Gil, A.M.; Duarte, I.F. NMR Metabolomics of Human Lung Tumours Reveals Distinct Metabolic Signatures for Adenocarcinoma and Squamous Cell Carcinoma. Carcinogenesis 2015, 36, 68–75. [Google Scholar] [CrossRef]
- Merino Salvador, M.; Gómez de Cedrón, M.; Moreno Rubio, J.; Falagán Martínez, S.; Sánchez Martínez, R.; Casado, E.; Ramírez de Molina, A.; Sereno, M. Lipid Metabolism and Lung Cancer. Crit. Rev. Oncol. Hematol. 2017, 112, 31–40. [Google Scholar] [CrossRef]
- Martin-Perez, M.; Urdiroz-Urricelqui, U.; Bigas, C.; Benitah, S.A. The Role of Lipids in Cancer Progression and Metastasis. Cell Metabolism 2022, 34, 1675–1699. [Google Scholar] [CrossRef] [PubMed]
- Breslow, D.K.; Weissman, J.S. Membranes in Balance: Mechanisms of Sphingolipid Homeostasis. Mol. Cell 2010, 40, 267. [Google Scholar] [CrossRef]
- Hao, D.; Sengupta, A.; Ding, K.; Ubeydullah, E.; Krishnaiah, S.; Leighl, N.B.; Shepherd, F.A.; Seymour, L.; Weljie, A. Metabolites as Prognostic Markers for Metastatic Non-Small Cell Lung Cancer (NSCLC) Patients Treated with First-Line Platinum-Doublet Chemotherapy. Cancers (Basel) 2020, 12, 1926. [Google Scholar] [CrossRef] [PubMed]
- Menendez, J.A.; Lupu, R. Fatty Acid Synthase and the Lipogenic Phenotype in Cancer Pathogenesis. Nat. Rev. Cancer 2007, 7, 763–777. [Google Scholar] [CrossRef] [PubMed]
- Peck, B.; Schulze, A. Lipid Metabolism at the Nexus of Diet and Tumor Microenvironment. Trends Cancer 2019, 5, 693–703. [Google Scholar] [CrossRef]
- Kowalczyk, T.; Kisluk, J.; Pietrowska, K.; Godzien, J.; Kozlowski, M.; Reszeć, J.; Sierko, E.; Naumnik, W.; Mróz, R.; Moniuszko, M.; et al. The Ability of Metabolomics to Discriminate Non-Small-Cell Lung Cancer Subtypes Depends on the Stage of the Disease and the Type of Material Studied. Cancers 2021, 13, 3314. [Google Scholar] [CrossRef]
- Zhang, M.; He, J.; Li, T.; Hu, H.; Li, X.; Xing, H.; Wang, J.; Yang, F.; Ma, Q.; Liu, B.; et al. Accurate Classification of Non-Small Cell Lung Cancer (NSCLC) Pathology and Mapping of EGFR Mutation Spatial Distribution by Ambient Mass Spectrometry Imaging. Front. Oncol. 2019, 9, 804. [Google Scholar] [CrossRef]
- Hao, D.; Sarfaraz, M.O.; Farshidfar, F.; Bebb, D.G.; Lee, C.Y.; Card, C.M.; David, M.; Weljie, A.M. Temporal Characterization of Serum Metabolite Signatures in Lung Cancer Patients Undergoing Treatment. Metabolomics 2016, 12, 58. [Google Scholar] [CrossRef]
- Overy, D.P.; Enot, D.P.; Tailliart, K.; Jenkins, H.; Parker, D.; Beckmann, M.; Draper, J. Explanatory Signal Interpretation and Metabolite Identification Strategies for Nominal Mass FIE-MS Metabolite Fingerprints. Nat. Protoc. 2008, 3, 471–485. [Google Scholar] [CrossRef]
- Yu, Z.; Kastenmüller, G.; He, Y.; Belcredi, P.; Möller, G.; Prehn, C.; Mendes, J.; Wahl, S.; Roemisch-Margl, W.; Ceglarek, U.; et al. Differences between Human Plasma and Serum Metabolite Profiles. PLoS One 2011, 6, e21230. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




