Submitted:
06 February 2025
Posted:
07 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
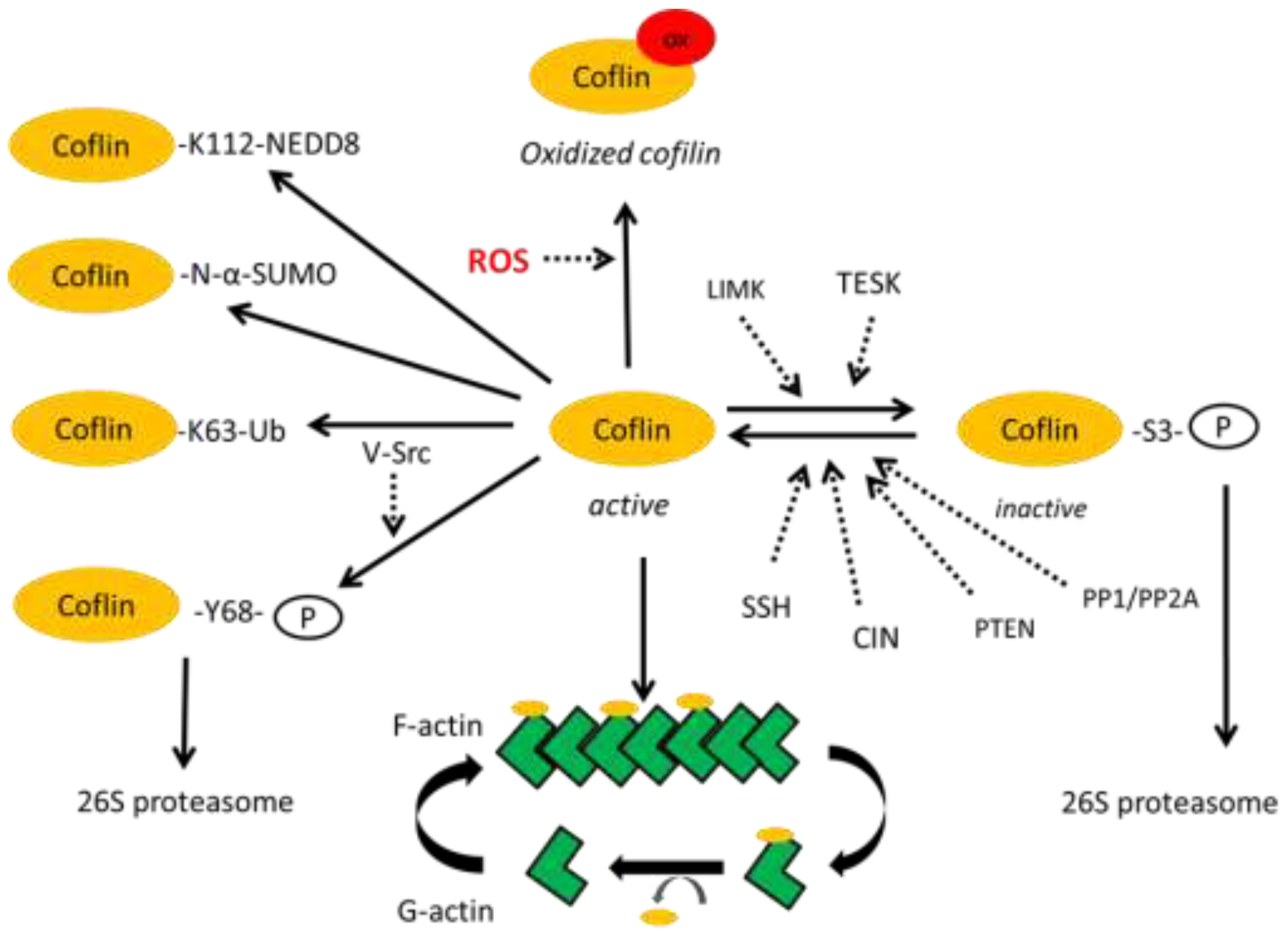
2. Post-Translational Modifications of Cofilin
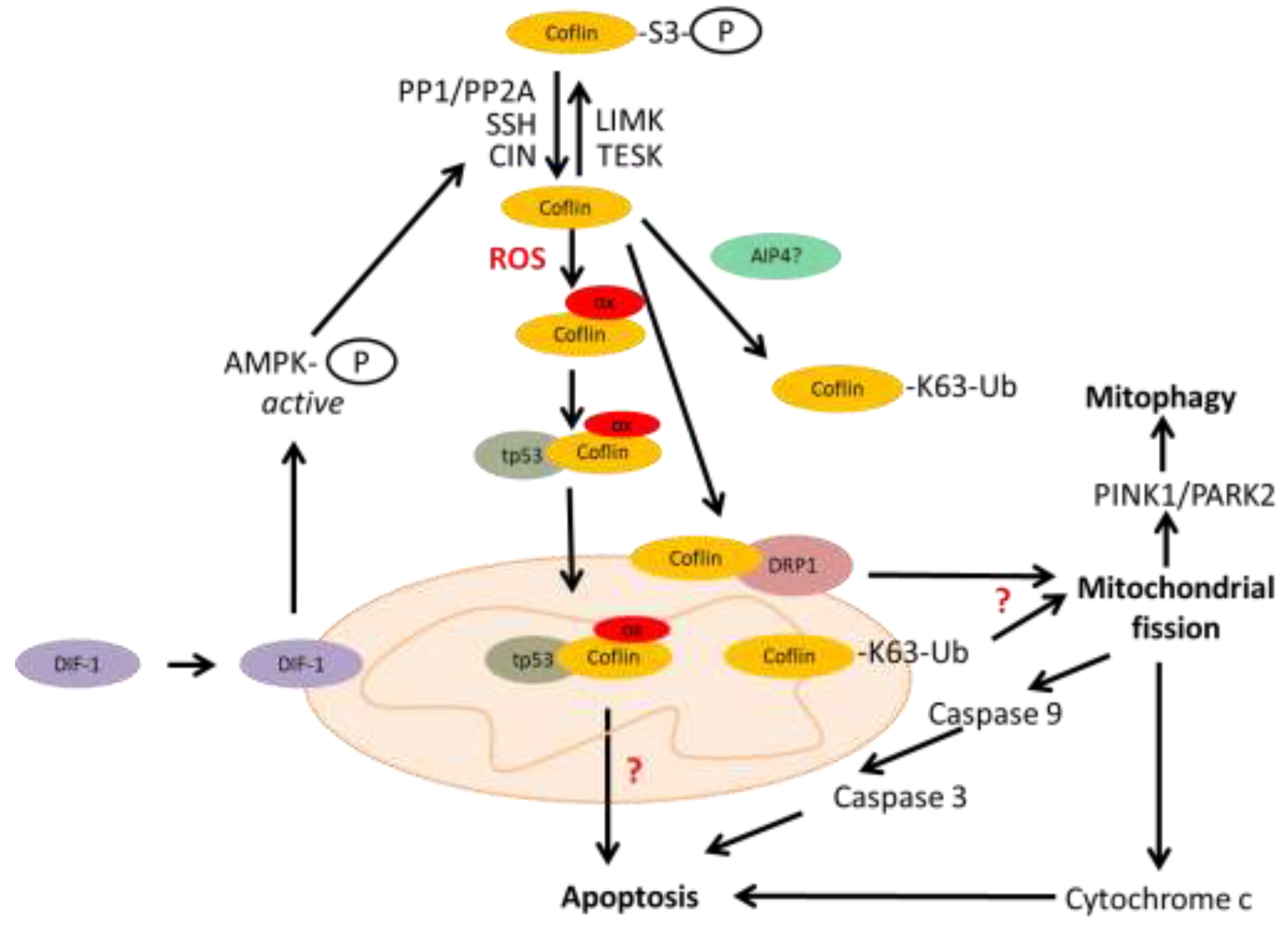
3. The Mitochondrial Localization of Cofilin
4. Cofilin Mediated Mitochondrial Dysfunction During Neurodegeneration
5. Cofilin Mediated Mitochondrial Dysfunction During Tumorigenesis
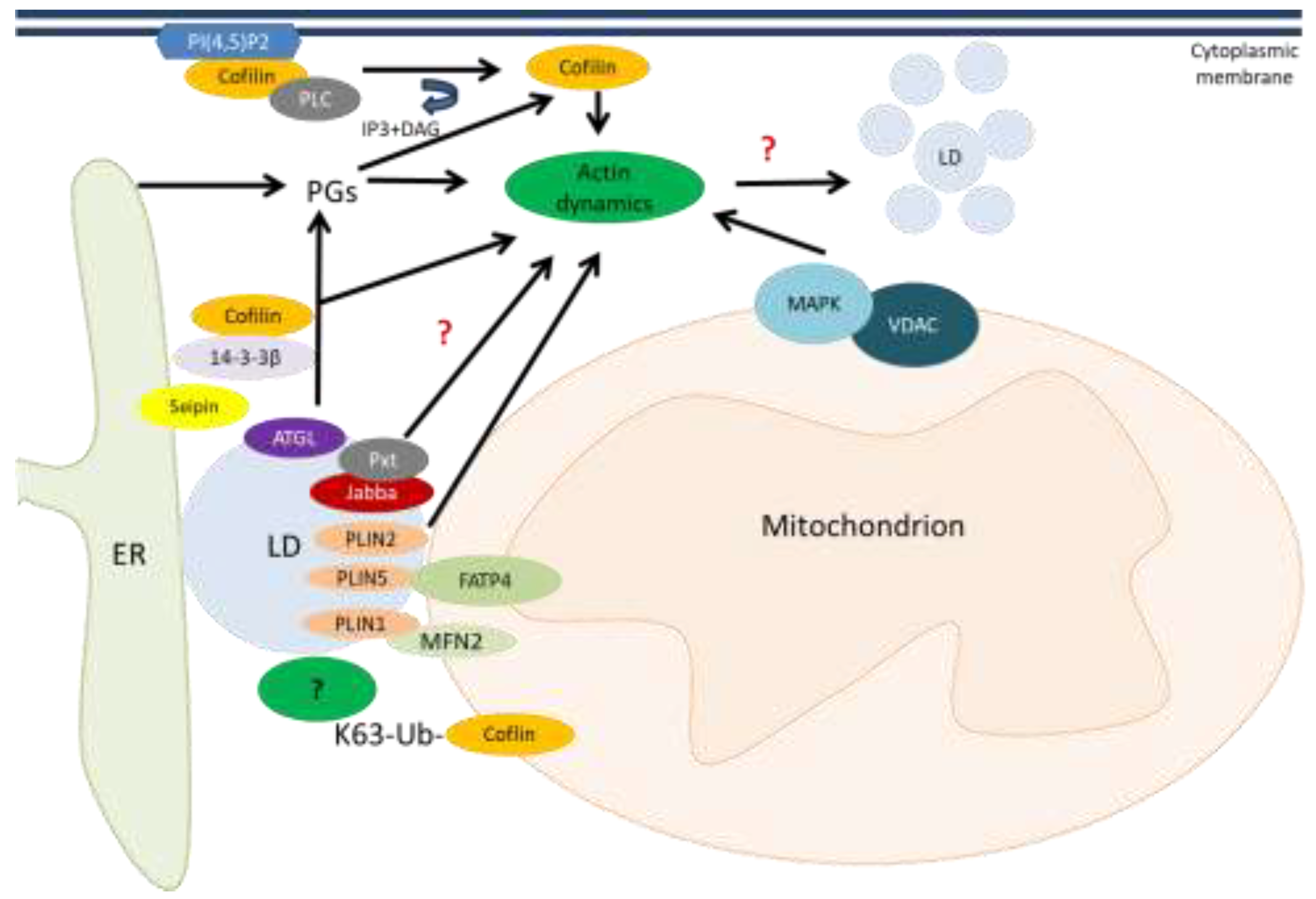
6. Cofilin and Lipid Metabolism
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| ADF | Actin depolymerizing factor |
| AMPK | AMP-activated protein kinase |
| ATGL | Adipose triglyceride lipase |
| CIN | Сhronophin |
| DIF-1 | Differentiation-inducing factor 1 |
| Drp1 | Dynamin-related protein 1 |
| EGF | Epidermal growth factor |
| ER | Endoplasmic reticulum |
| F-actin | Filamentous actin |
| G-actin | Globular actin |
| LD | Lipid droplet |
| LIMK | LIM kinase |
| MFN2 | Mitofusin 2 |
| PD | Parkinson’s disease |
| PG | Prostaglandin |
| PI(4,5)P2 | Phosphatidylinositol 4,5-bisphosphate |
| PLC | Phospholipase C |
| PLIN | Perilipin |
| PP1 | Serine/threonine phosphatase type 1 |
| PP2A | Serine/threonine phosphatase type 2A |
| PTEN | Phosphatase and tensin homolog |
| PTM | Post-translational modification |
| ROS | Reactive oxygen species |
| SSH | Slingshot phosphatase |
| TESK | TES kinase |
| VDAC | Voltage-dependent anion channel |
References
- Nishida, E.; Maekawa, S.; Sakai, H. Cofilin, a Protein in Porcine Brain That Binds to Actin Filaments and Inhibits Their Interactions with Myosin and Tropomyosin. Biochemistry 1984, 23, 5307–5313. [Google Scholar] [CrossRef]
- Alsegiani, A.; Shah, Z. The Role of Cofilin in Age-Related Neuroinflammation. Neural Regen Res 2020, 15, 1451. [Google Scholar] [CrossRef] [PubMed]
- Bamburg, J.; Minamide, L.; Wiggan, O.; Tahtamouni, L.; Kuhn, T. Cofilin and Actin Dynamics: Multiple Modes of Regulation and Their Impacts in Neuronal Development and Degeneration. Cells 2021, 10, 2726. [Google Scholar] [CrossRef] [PubMed]
- Kremneva, E.; Makkonen, M.H.; Skwarek-Maruszewska, A.; Gateva, G.; Michelot, A.; Dominguez, R.; Lappalainen, P. Cofilin-2 Controls Actin Filament Length in Muscle Sarcomeres. Developmental Cell 2014, 31, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Ono, S. Cofilin-Induced Structural Changes in Actin Filaments Stay Local. Proc. Natl. Acad. Sci. U.S.A. 2020, 117, 3349–3351. [Google Scholar] [CrossRef]
- Kanellos, G.; Frame, M.C. Cellular Functions of the ADF/Cofilin Family at a Glance. Journal of Cell Science 2016, jcs.187849. [CrossRef]
- Zuchero, J.B.; Fu, M.; Sloan, S.A.; Ibrahim, A.; Olson, A.; Zaremba, A.; Dugas, J.C.; Wienbar, S.; Caprariello, A.V.; Kantor, C.; et al. CNS Myelin Wrapping Is Driven by Actin Disassembly. Developmental Cell 2015, 34, 152–167. [Google Scholar] [CrossRef]
- Ono, S.; Minami, N.; Abe, H.; Obinata, T. Characterization of a Novel Cofilin Isoform That Is Predominantly Expressed in Mammalian Skeletal Muscle. J Biol Chem 1994, 269, 15280–15286. [Google Scholar] [CrossRef]
- Bamburg, J.R. Proteins of the ADF/Cofilin Family: Essential Regulators of Actin Dynamics. Annu. Rev. Cell Dev. Biol. 1999, 15, 185–230. [Google Scholar] [CrossRef]
- Sollis, E.; Mosaku, A.; Abid, A.; Buniello, A.; Cerezo, M.; Gil, L.; Groza, T.; Güneş, O.; Hall, P.; Hayhurst, J.; et al. The NHGRI-EBI GWAS Catalog: Knowledgebase and Deposition Resource. Nucleic Acids Research 2023, 51, D977–D985. [Google Scholar] [CrossRef]
- Bellenchi, G.C.; Gurniak, C.B.; Perlas, E.; Middei, S.; Ammassari-Teule, M.; Witke, W. N-Cofilin Is Associated with Neuronal Migration Disorders and Cell Cycle Control in the Cerebral Cortex. Genes Dev. 2007, 21, 2347–2357. [Google Scholar] [CrossRef]
- Moon, A.L.; Janmey, P.A.; Louie, K.A.; Drubin, D.G. Cofilin Is an Essential Component of the Yeast Cortical Cytoskeleton. The Journal of cell biology 1993, 120, 421–435. [Google Scholar] [CrossRef]
- Xu, J.; Huang, Y.; Zhao, J.; Wu, L.; Qi, Q.; Liu, Y.; Li, G.; Li, J.; Liu, H.; Wu, H. Cofilin: A Promising Protein Implicated in Cancer Metastasis and Apoptosis. Front. Cell Dev. Biol. 2021, 9, 599065. [Google Scholar] [CrossRef] [PubMed]
- Schratt, G.M.; Tuebing, F.; Nigh, E.A.; Kane, C.G.; Sabatini, M.E.; Kiebler, M.; Greenberg, M.E. A Brain-Specific microRNA Regulates Dendritic Spine Development. Nature 2006, 439, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Zeng, M.; Zhao, Y.; Fang, X. Upregulation of Limk1 Caused by microRNA-138 Loss Aggravates the Metastasis of Ovarian Cancer by Activation of Limk1/Cofilin Signaling. Oncology Reports 2014, 32, 2070–2076. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.-X.; Wang, X.-L.; Zhang, L.-N.; Zhang, J.; Zhao, W. MicroRNA-384 Inhibits the Progression of Esophageal Squamous Cell Carcinoma through Blockade of the LIMK1/Cofilin Signaling Pathway by Binding to LIMK1. Biomedicine & Pharmacotherapy 2019, 109, 751–761. [Google Scholar] [CrossRef]
- Tang, Q.; Ji, Q.; Tang, Y.; Chen, T.; Pan, G.; Hu, S.; Bao, Y.; Peng, W.; Yin, P. Mitochondrial Translocation of Cofilin-1 Promotes Apoptosis of Gastric Cancer BGC-823 Cells Induced by Ursolic Acid. Tumor Biol. 2014, 35, 2451–2459. [Google Scholar] [CrossRef]
- Ockeloen, C.W.; Gilhuis, H.J.; Pfundt, R.; Kamsteeg, E.J.; Agrawal, P.B.; Beggs, A.H.; Dara Hama-Amin, A.; Diekstra, A.; Knoers, N.V.A.M.; Lammens, M.; et al. Congenital Myopathy Caused by a Novel Missense Mutation in the CFL2 Gene. Neuromuscular Disorders 2012, 22, 632–639. [Google Scholar] [CrossRef]
- Tabur, S.; Oztuzcu, S.; Oguz, E.; Demiryürek, S.; Dagli, H.; Alasehirli, B.; Ozkaya, M.; Demiryürek, A.T. Evidence for Elevated (LIMK2 and CFL1) and Suppressed (ICAM1, EZR, MAP2K2, and NOS3) Gene Expressions in Metabolic Syndrome. Endocrine 2016, 53, 465–470. [Google Scholar] [CrossRef]
- Jang, D.-H.; Han, J.-H.; Lee, S.-H.; Lee, Y.-S.; Park, H.; Lee, S.-H.; Kim, H.; Kaang, B.-K. Cofilin Expression Induces Cofilin-Actin Rod Formation and Disrupts Synaptic Structure and Function in Aplysia Synapses. Proc. Natl. Acad. Sci. U.S.A. 2005, 102, 16072–16077. [Google Scholar] [CrossRef]
- Bamburg, J.R.; Bernstein, B.W. Actin Dynamics and Cofilin-actin Rods in Alzheimer Disease. Cytoskeleton 2016, 73, 477–497. [Google Scholar] [CrossRef]
- Lu, L.; Fu, N.; Luo, X.; Li, X.-Y.; Li, X.-P. Overexpression of Cofilin 1 in Prostate Cancer and the Corresponding Clinical Implications. Oncology Letters 2015, 9, 2757–2761. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wu, D.; He, F.; Fu, H.; Xu, J.; Chen, J.; Zhang, S.; Wang, W. Study on the Significance of Cofilin 1 Overexpression in Human Bladder Cancer. Tumori 2017, 103, 537–542. [Google Scholar] [CrossRef]
- Wang, F.; Wu, D.; Fu, H.; He, F.; Xu, C.; Zhou, J.; Li, D.; Li, G.; Xu, J.; Wu, Q.; et al. Cofilin 1 Promotes Bladder Cancer and Is Regulated by TCF7L2. Oncotarget 2017, 8, 92043–92054. [Google Scholar] [CrossRef] [PubMed]
- Sousa-Squiavinato, A.C.M.; Rocha, M.R.; Barcellos-de-Souza, P.; De Souza, W.F.; Morgado-Diaz, J.A. Cofilin-1 Signaling Mediates Epithelial-Mesenchymal Transition by Promoting Actin Cytoskeleton Reorganization and Cell-Cell Adhesion Regulation in Colorectal Cancer Cells. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 2019, 1866, 418–429. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liao, R.; Li, H.; Liu, L.; Chen, X.; Chen, H. Expression of Cofilin-1 and Transgelin in Esophageal Squamous Cell Carcinoma. Med Sci Monit 2015, 21, 2659–2665. [Google Scholar] [CrossRef]
- Tsai, C.; Chang, C.; Lin, B.; Wu, Y.; Wu, M.; Lin, L.; Huang, W.; Holz, J.D.; Sheu, T.; Lee, J.; et al. Up-regulation of Cofilin-1 in Cell Senescence Associates with Morphological Change and P27 kip1 -mediated Growth Delay. Aging Cell 2021, 20, e13288. [Google Scholar] [CrossRef]
- Andrianantoandro, E.; Pollard, T.D. Mechanism of Actin Filament Turnover by Severing and Nucleation at Different Concentrations of ADF/Cofilin. Molecular Cell 2006, 24, 13–23. [Google Scholar] [CrossRef]
- Namme, J.N.; Bepari, A.K.; Takebayashi, H. Cofilin Signaling in the CNS Physiology and Neurodegeneration. IJMS 2021, 22, 10727. [Google Scholar] [CrossRef]
- Yap, C.T.; Simpson, T.I.; Pratt, T.; Price, D.J.; Maciver, S.K. The Motility of Glioblastoma Tumour Cells Is Modulated by Intracellular Cofilin Expression in a Concentration-Dependent Manner. Cell Motil. Cytoskeleton 2005, 60, 153–165. [Google Scholar] [CrossRef]
- Tsai, C.-H.; Chiu, S.-J.; Liu, C.-C.; Sheu, T.-J.; Hsieh, C.-H.; Keng, P.C.; Lee, Y.-J. Regulated Expression of Cofilin and the Consequent Regulation of P27kip1 Are Essential for G1 Phase Progression. Cell Cycle 2009, 8, 2365–2374. [Google Scholar] [CrossRef]
- Zhang, H.; Lechuga, T.J.; Tith, T.; Wang, W.; Wing, D.A.; Chen, D. S-Nitrosylation of Cofilin-1 Mediates Estradiol-17β-Stimulated Endothelial Cytoskeleton Remodeling. Molecular Endocrinology 2015, 29, 434–444. [Google Scholar] [CrossRef]
- Vallée, B.; Doudeau, M.; Godin, F.; Bénédetti, H. Characterization at the Molecular Level Using Robust Biochemical Approaches of a New Kinase Protein. JoVE 2019, 59820. [Google Scholar] [CrossRef]
- Hunter, T. The Age of Crosstalk: Phosphorylation, Ubiquitination, and Beyond. Molecular Cell 2007, 28, 730–738. [Google Scholar] [CrossRef] [PubMed]
- Akimov, V.; Rigbolt, K.T.G.; Nielsen, M.M.; Blagoev, B. Characterization of Ubiquitination Dependent Dynamics in Growth Factor Receptor Signaling by Quantitative Proteomics. Mol. BioSyst. 2011, 7, 3223. [Google Scholar] [CrossRef]
- Beltrao, P.; Albanèse, V.; Kenner, L.R.; Swaney, D.L.; Burlingame, A.; Villén, J.; Lim, W.A.; Fraser, J.S.; Frydman, J.; Krogan, N.J. Systematic Functional Prioritization of Protein Posttranslational Modifications. Cell 2012, 150, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.Y.; DerMardirossian, C.; Bokoch, G.M. Cofilin Phosphatases and Regulation of Actin Dynamics. Current Opinion in Cell Biology 2006, 18, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Toshima, J.; Toshima, J.Y.; Amano, T.; Yang, N.; Narumiya, S.; Mizuno, K. Cofilin Phosphorylation by Protein Kinase Testicular Protein Kinase 1 and Its Role in Integrin-Mediated Actin Reorganization and Focal Adhesion Formation. MBoC 2001, 12, 1131–1145. [Google Scholar] [CrossRef]
- Schulze, M.; Hutterer, M.; Sabo, A.; Hoja, S.; Lorenz, J.; Rothhammer-Hampl, T.; Herold-Mende, C.; Floßbach, L.; Monoranu, C.; Riemenschneider, M.J. Chronophin Regulates Active Vitamin B6 Levels and Transcriptomic Features of Glioblastoma Cell Lines Cultured under Non-Adherent, Serum-Free Conditions. BMC Cancer 2018, 18, 524. [Google Scholar] [CrossRef]
- Oleinik, N.V.; Krupenko, N.I.; Krupenko, S.A. ALDH1L1 Inhibits Cell Motility via Dephosphorylation of Cofilin by PP1 and PP2A. Oncogene 2010, 29, 6233–6244. [Google Scholar] [CrossRef]
- Vitolo, M.I.; Boggs, A.E.; Whipple, R.A.; Yoon, J.R.; Thompson, K.; Matrone, M.A.; Cho, E.H.; Balzer, E.M.; Martin, S.S. Loss of PTEN Induces Microtentacles through PI3K-Independent Activation of Cofilin. Oncogene 2013, 32, 2200–2210. [Google Scholar] [CrossRef]
- Prudent, R.; Demoncheaux, N.; Diemer, H.; Collin-Faure, V.; Kapur, R.; Paublant, F.; Lafanechère, L.; Cianférani, S.; Rabilloud, T. A Quantitative Proteomic Analysis of Cofilin Phosphorylation in Myeloid Cells and Its Modulation Using the LIM Kinase Inhibitor Pyr1. PLoS ONE 2018, 13, e0208979. [Google Scholar] [CrossRef] [PubMed]
- Yoo, Y.; Ho, H.J.; Wang, C.; Guan, J.-L. Tyrosine Phosphorylation of Cofilin at Y68 by V-Src Leads to Its Degradation through Ubiquitin–Proteasome Pathway. Oncogene 2010, 29, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Pfannstiel, J.; Cyrklaff, M.; Habermann, A.; Stoeva, S.; Griffiths, G.; Shoeman, R.; Faulstich, H. Human Cofilin Forms Oligomers Exhibiting Actin Bundling Activity. Journal of Biological Chemistry 2001, 276, 49476–49484. [Google Scholar] [CrossRef]
- Weng, W.; Gu, X.; Yang, Y.; Zhang, Q.; Deng, Q.; Zhou, J.; Cheng, J.; Zhu, M.X.; Feng, J.; Huang, O.; et al. N-Terminal α-Amino SUMOylation of Cofilin-1 Is Critical for Its Regulation of Actin Depolymerization. Nat Commun 2023, 14, 5688. [Google Scholar] [CrossRef]
- Vogl, A.M.; Phu, L.; Becerra, R.; Giusti, S.A.; Verschueren, E.; Hinkle, T.B.; Bordenave, M.D.; Adrian, M.; Heidersbach, A.; Yankilevich, P.; et al. Global Site-Specific Neddylation Profiling Reveals That NEDDylated Cofilin Regulates Actin Dynamics. Nat Struct Mol Biol 2020, 27, 210–220. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, R. Towards Computational Models of Identifying Protein Ubiquitination Sites. CDT 2019, 20, 565–578. [Google Scholar] [CrossRef] [PubMed]
- Gainullin, M.R.; Zhukov, I.Y.; Zhou, X.; Mo, Y.; Astakhova, L.; Ernberg, I.; Matskova, L. Degradation of Cofilin Is Regulated by Cbl, AIP4 and Syk Resulting in Increased Migration of LMP2A Positive Nasopharyngeal Carcinoma Cells. Sci Rep 2017, 7, 9012. [Google Scholar] [CrossRef]
- Buneeva, O.; Medvedev, A. Atypical Ubiquitination and Parkinson’s Disease. IJMS 2022, 23, 3705. [Google Scholar] [CrossRef]
- Cao, L.; Liu, X.; Zheng, B.; Xing, C.; Liu, J. Role of K63-Linked Ubiquitination in Cancer. Cell Death Discov. 2022, 8, 410. [Google Scholar] [CrossRef]
- Madiraju, C.; Novack, J.P.; Reed, J.C.; Matsuzawa, S. K63 Ubiquitination in Immune Signaling. Trends in Immunology 2022, 43, 148–162. [Google Scholar] [CrossRef]
- Liu, H.; Weng, W.; Guo, R.; Zhou, J.; Xue, J.; Zhong, S.; Cheng, J.; Zhu, M.X.; Pan, S.-J.; Li, Y. Olig2 SUMOylation Protects against Genotoxic Damage Response by Antagonizing P53 Gene Targeting. Cell Death Differ 2020, 27, 3146–3161. [Google Scholar] [CrossRef]
- Unwin, R.D.; Craven, R.A.; Harnden, P.; Hanrahan, S.; Totty, N.; Knowles, M.; Eardley, I.; Selby, P.J.; Banks, R.E. Proteomic Changes in Renal Cancer and Co-ordinate Demonstration of Both the Glycolytic and Mitochondrial Aspects of the Warburg Effect. Proteomics 2003, 3, 1620–1632. [Google Scholar] [CrossRef] [PubMed]
- Chua, B.T.; Volbracht, C.; Tan, K.O.; Li, R.; Yu, V.C.; Li, P. Mitochondrial Translocation of Cofilin Is an Early Step in Apoptosis Induction. Nat Cell Biol 2003, 5, 1083–1089. [Google Scholar] [CrossRef]
- Rath, S.; Sharma, R.; Gupta, R.; Ast, T.; Chan, C.; Durham, T.J.; Goodman, R.P.; Grabarek, Z.; Haas, M.E.; Hung, W.H.W.; et al. MitoCarta3.0: An Updated Mitochondrial Proteome Now with Sub-Organelle Localization and Pathway Annotations. Nucleic Acids Res 2021, 49, D1541–D1547. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, L.; Waclawczyk, M.S.; Tang, S.; Hanschmann, E.-M.; Gellert, M.; Rust, M.B.; Culmsee, C. Cofilin1 Oxidation Links Oxidative Distress to Mitochondrial Demise and Neuronal Cell Death. Cell Death Dis 2021, 12, 953. [Google Scholar] [CrossRef] [PubMed]
- Kovaleva, T.F.; Maksimova, N.S.; Pchelin, P.V.; Pershin, V.I.; Tkachenko, N.M.; Gainullin, M.R.; Mukhina, I.V. A New Cofilin-Dependent Mechanism for the Regulation of Brain Mitochondria Biogenesis and Degradation. Sovrem Tehnol Med 2020, 12, 6. [Google Scholar] [CrossRef]
- Lapeña-Luzón, T.; Rodríguez, L.R.; Beltran-Beltran, V.; Benetó, N.; Pallardó, F.V.; Gonzalez-Cabo, P. Cofilin and Neurodegeneration: New Functions for an Old but Gold Protein. Brain Sciences 2021, 11, 954. [Google Scholar] [CrossRef]
- Hoffmann, L.; Rust, M.B.; Culmsee, C. Actin(g) on Mitochondria – a Role for Cofilin1 in Neuronal Cell Death Pathways. Biological Chemistry 2019, 400, 1089–1097. [Google Scholar] [CrossRef]
- Li, G.-B.; Zhang, H.-W.; Fu, R.-Q.; Hu, X.-Y.; Liu, L.; Li, Y.-N.; Liu, Y.-X.; Liu, X.; Hu, J.-J.; Deng, Q.; et al. Mitochondrial Fission and Mitophagy Depend on Cofilin-Mediated Actin Depolymerization Activity at the Mitochondrial Fission Site. Oncogene 2018, 37, 1485–1502. [Google Scholar] [CrossRef]
- Roh, S.; Woo, J.A.; Lakshmana, M.K.; Uhlar, C.; Ankala, V.; Boggess, T.; Liu, T.; Hong, Y.; Mook-Jung, I.; Kim, S.J.; et al. Mitochondrial Dysfunction and Calcium Deregulation by the RanBP9-cofilin Pathway. FASEB j. 2013, 27, 4776–4789. [Google Scholar] [CrossRef]
- Li, G.; Zhou, J.; Budhraja, A.; Hu, X.; Chen, Y.; Cheng, Q.; Liu, L.; Zhou, T.; Li, P.; Liu, E.; et al. Mitochondrial Translocation and Interaction of Cofilin and Drp1 Are Required for Erucin-Induced Mitochondrial Fission and Apoptosis. Oncotarget 2015, 6, 1834–1849. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Miura, K.; Han, R.; Seto-Tetsuo, F.; Arioka, M.; Igawa, K.; Tomooka, K.; Sasaguri, T. Differentiation-Inducing Factor 1 Activates Cofilin through Pyridoxal Phosphatase and AMP-Activated Protein Kinase, Resulting in Mitochondrial Fission. Journal of Pharmacological Sciences 2023, 152, 39–49. [Google Scholar] [CrossRef]
- Klamt, F.; Zdanov, S.; Levine, R.L.; Pariser, A.; Zhang, Y.; Zhang, B.; Yu, L.-R.; Veenstra, T.D.; Shacter, E. Oxidant-Induced Apoptosis Is Mediated by Oxidation of the Actin-Regulatory Protein Cofilin. Nat Cell Biol 2009, 11, 1241–1246. [Google Scholar] [CrossRef]
- Kotiadis, V.N.; Leadsham, J.E.; Bastow, E.L.; Gheeraert, A.; Whybrew, J.M.; Bard, M.; Lappalainen, P.; Gourlay, C.W. Identification of New Surfaces of Cofilin That Link Mitochondrial Function to the Control of Multi-Drug Resistance. Journal of Cell Science 2012, jcs.099390. [CrossRef]
- Cichon, J.; Sun, C.; Chen, B.; Jiang, M.; Chen, X.A.; Sun, Y.; Wang, Y.; Chen, G. Cofilin Aggregation Blocks Intracellular Trafficking and Induces Synaptic Loss in Hippocampal Neurons. Journal of Biological Chemistry 2012, 287, 3919–3929. [Google Scholar] [CrossRef] [PubMed]
- Kovaleva, T.F.; Maksimova, N.S.; Zhukov, I.Yu.; Pershin, V.I.; Mukhina, I.V.; Gainullin, M.R. Cofilin: Molecular and Cellular Functions and Its Role in the Functioning of the Nervous System. Neurochem. J. 2019, 13, 11–19. [Google Scholar] [CrossRef]
- Yan, M.; Meng, L.; Dai, L.; Zhang, X.; Chen, G.; Zheng, Y.; Zha, Y.; Zeng, Y.; Zhang, Z. Cofilin 1 Promotes the Aggregation and Cell-to-Cell Transmission of α-Synuclein in Parkinson’s Disease. Biochemical and Biophysical Research Communications 2020, 529, 1053–1060. [Google Scholar] [CrossRef]
- Ordonez, D.G.; Lee, M.K.; Feany, M.B. α-Synuclein Induces Mitochondrial Dysfunction through Spectrin and the Actin Cytoskeleton. Neuron 2018, 97, 108–124.e6. [Google Scholar] [CrossRef]
- Kang, D.E.; Woo, J.A. Cofilin, a Master Node Regulating Cytoskeletal Pathogenesis in Alzheimer’s Disease. JAD 2019, 72, S131–S144. [Google Scholar] [CrossRef] [PubMed]
- Minamide, L.S.; Maiti, S.; Boyle, J.A.; Davis, R.C.; Coppinger, J.A.; Bao, Y.; Huang, T.Y.; Yates, J.; Bokoch, G.M.; Bamburg, J.R. Isolation and Characterization of Cytoplasmic Cofilin-Actin Rods. Journal of Biological Chemistry 2010, 285, 5450–5460. [Google Scholar] [CrossRef]
- Munsie, L.N.; Desmond, C.R.; Truant, R. Cofilin Nuclear-Cytoplasmic Shuttling Affects Cofilin-Actin Rod Formation During Stress. Journal of Cell Science 2012, jcs.097667. [CrossRef]
- Bernstein, B.W.; Chen, H.; Boyle, J.A.; Bamburg, J.R. Formation of Actin-ADF/Cofilin Rods Transiently Retards Decline of Mitochondrial Potential and ATP in Stressed Neurons. American Journal of Physiology-Cell Physiology 2006, 291, C828–C839. [Google Scholar] [CrossRef]
- Rehklau, K.; Gurniak, C.B.; Conrad, M.; Friauf, E.; Ott, M.; Rust, M.B. ADF/Cofilin Proteins Translocate to Mitochondria during Apoptosis but Are Not Generally Required for Cell Death Signaling. Cell Death Differ 2012, 19, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, H.; Li, J.; Jiang, X.; Zhang, Y.; Wu, Q.; Shen, L.; Shi, J.; Gao, N. ROCK1 Activation-Mediated Mitochondrial Translocation of Drp1 and Cofilin Are Required for Arnidiol-Induced Mitochondrial Fission and Apoptosis. J Exp Clin Cancer Res 2020, 39, 37. [Google Scholar] [CrossRef] [PubMed]
- Rehklau, K.; Hoffmann, L.; Gurniak, C.B.; Ott, M.; Witke, W.; Scorrano, L.; Culmsee, C.; Rust, M.B. Cofilin1-Dependent Actin Dynamics Control DRP1-Mediated Mitochondrial Fission. Cell Death Dis 2017, 8, e3063–e3063. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Miao, R.; Wei, J.; Wu, H.; Tian, J. Advances in Multi-Omics Study of Biomarkers of Glycolipid Metabolism Disorder. Computational and Structural Biotechnology Journal 2022, 20, 5935–5951. [Google Scholar] [CrossRef]
- Izdebska, M.; Zielińska, W.; Hałas-Wiśniewska, M.; Grzanka, A. Involvement of Actin and Actin-Binding Proteins in Carcinogenesis. Cells 2020, 9, 2245. [Google Scholar] [CrossRef]
- Lv, S.; Chen, Z.; Mi, H.; Yu, X. Cofilin Acts as a Booster for Progression of Malignant Tumors Represented by Glioma. Cancer Manag Res 2022, 14, 3245–3269. [Google Scholar] [CrossRef]
- Wu, J.; Wang, H.; Guo, X.; Chen, J. Cofilin-Mediated Actin Dynamics Promotes Actin Bundle Formation during Drosophila Bristle Development. MBoC 2016, 27, 2554–2564. [Google Scholar] [CrossRef]
- Satoh, M.; Takano, S.; Sogawa, K.; Noda, K.; Yoshitomi, H.; Ishibashi, M.; Mogushi, K.; Takizawa, H.; Otsuka, M.; Shimizu, H.; et al. Immune-complex Level of Cofilin-1 in Sera Is Associated with Cancer Progression and Poor Prognosis in Pancreatic Cancer. Cancer Science 2017, 108, 795–803. [Google Scholar] [CrossRef]
- Yan, J.; Wang, A.; Cao, J.; Chen, L. Apelin/APJ System: An Emerging Therapeutic Target for Respiratory Diseases. Cell. Mol. Life Sci. 2020, 77, 2919–2930. [Google Scholar] [CrossRef]
- Li, J.; Yang, R.; Yang, H.; Chen, S.; Wang, L.; Li, M.; Yang, S.; Feng, Z.; Bi, J. NCAM Regulates the Proliferation, Apoptosis, Autophagy, EMT, and Migration of Human Melanoma Cells via the Src/Akt/mTOR/Cofilin Signaling Pathway. J of Cellular Biochemistry 2020, 121, 1192–1204. [Google Scholar] [CrossRef]
- Song, H.; Pu, J.; Wang, L.; Wu, L.; Xiao, J.; Liu, Q.; Chen, J.; Zhang, M.; Liu, Y.; Ni, M.; et al. ATG16L1 Phosphorylation Is Oppositely Regulated by CSNK2/Casein Kinase 2 and PPP1/Protein Phosphatase 1 Which Determines the Fate of Cardiomyocytes during Hypoxia/Reoxygenation. Autophagy 2015, 11, 1308–1325. [Google Scholar] [CrossRef] [PubMed]
- Celeste Morley, S.; Sun, G.; Bierer, B.E. Inhibition of Actin Polymerization Enhances Commitment to and Execution of Apoptosis Induced by Withdrawal of Trophic Support. J of Cellular Biochemistry 2003, 88, 1066–1076. [Google Scholar] [CrossRef]
- Wang, C.; Youle, R.J. The Role of Mitochondria in Apoptosis. Annu. Rev. Genet. 2009, 43, 95–118. [Google Scholar] [CrossRef]
- Springer, W.; Kahle, P.J. Regulation of PINK1-Parkin-Mediated Mitophagy. Autophagy 2011, 7, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Fedorowicz, M.A.; De Vries-Schneider, R.L.A.; Rüb, C.; Becker, D.; Huang, Y.; Zhou, C.; Alessi Wolken, D.M.; Voos, W.; Liu, Y.; Przedborski, S. Cytosolic Cleaved PINK 1 Represses P Arkin Translocation to Mitochondria and Mitophagy. EMBO Reports 2014, 15, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Tan, Y. Lipid Droplet–Mitochondria Contacts in Health and Disease. IJMS 2024, 25, 6878. [Google Scholar] [CrossRef]
- Davis, J.; Meyer, T.; Smethurst, D.; Neuhaus, L.; Heyden, J.; Broeskamp, F.; Edrich, E.; Knittelfelder, O.; Smolnig, M.; Kolb, D.; et al. Loss of Actin Dynamics Leads to VDAC Dependent MAPK Signalling and Altered Lipid Homeostasis That Promotes Cell Death in Yeast Cells 2022.
- Cui, L.; Liu, P. Two Types of Contact Between Lipid Droplets and Mitochondria. Front. Cell Dev. Biol. 2020, 8, 618322. [Google Scholar] [CrossRef]
- Miner, G.E.; So, C.M.; Edwards, W.; Herring, L.E.; Coleman, R.A.; Klett, E.L.; Cohen, S. Perilipin 5 Interacts with Fatp4 at Membrane Contact Sites to Promote Lipid Droplet-to-Mitochondria Fatty Acid Transport; Cell Biology, 2022.
- Giedt, M.S.; Thomalla, J.M.; Johnson, M.R.; Lai, Z.W.; Tootle, T.L.; Welte, M.A. Adipose Triglyceride Lipase Promotes Prostaglandin-Dependent Actin Remodeling by Regulating Substrate Release from Lipid Droplets; Cell Biology, 2021.
- Serezani, C.H.; Kane, S.; Medeiros, A.I.; Cornett, A.M.; Kim, S.-H.; Marques, M.M.; Lee, S.-P.; Lewis, C.; Bourdonnay, E.; Ballinger, M.N.; et al. PTEN Directly Activates the Actin Depolymerization Factor Cofilin-1 During PGE 2 -Mediated Inhibition of Phagocytosis of Fungi. Sci. Signal. 2012, 5. [Google Scholar] [CrossRef]
- Yang, W.; Thein, S.; Wang, X.; Bi, X.; Ericksen, R.E.; Xu, F.; Han, W. BSCL2/Seipin Regulates Adipogenesis through Actin Cytoskeleton Remodelling. Human Molecular Genetics 2014, 23, 502–513. [Google Scholar] [CrossRef]
- Samstag, Y.; John, I.; Wabnitz, G.H. Cofilin: A Redox Sensitive Mediator of Actin Dynamics during T-cell Activation and Migration. Immunological Reviews 2013, 256, 30–47. [Google Scholar] [CrossRef]
- Wang, J.; Fang, N.; Xiong, J.; Du, Y.; Cao, Y.; Ji, W.-K. An ESCRT-Dependent Step in Fatty Acid Transfer from Lipid Droplets to Mitochondria through VPS13D−TSG101 Interactions. Nat Commun 2021, 12, 1252. [Google Scholar] [CrossRef]
- Pfisterer, S.G.; Gateva, G.; Horvath, P.; Pirhonen, J.; Salo, V.T.; Karhinen, L.; Varjosalo, M.; Ryhänen, S.J.; Lappalainen, P.; Ikonen, E. Role for Formin-like 1-Dependent Acto-Myosin Assembly in Lipid Droplet Dynamics and Lipid Storage. Nat Commun 2017, 8, 14858. [Google Scholar] [CrossRef]
- Cui, Y.; Man, S.; Tao, J.; Liu, Y.; Ma, L.; Guo, L.; Huang, L.; Liu, C.; Gao, W. The Lipid Droplet in Cancer: From Being a Tumor-supporting Hallmark to Clinical Therapy. Acta Physiologica 2024, e14087. [Google Scholar] [CrossRef] [PubMed]
- Safi, R.; Menéndez, P.; Pol, A. Lipid Droplets Provide Metabolic Flexibility for Cancer Progression. FEBS Letters 2024, 598, 1301–1327. [Google Scholar] [CrossRef] [PubMed]
- Petan, T. Lipid Droplets in Cancer. In Organelles in Disease; Pedersen, S.H.F., Barber, D.L., Eds.; Reviews of Physiology, Biochemistry and Pharmacology; Springer International Publishing: Cham, 2020; Vol. 185, pp. 53–86 ISBN 978-3-031-22594-9.
- Jarc, E.; Petan, T. A Twist of FATe: Lipid Droplets and Inflammatory Lipid Mediators. Biochimie 2020, 169, 69–87. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Dutra, F.S.; Bozza, P.T. Lipid Droplets Diversity and Functions in Inflammation and Immune Response. Expert Review of Proteomics 2021, 18, 809–825. [Google Scholar] [CrossRef]
- Zadoorian, A.; Du, X.; Yang, H. Lipid Droplet Biogenesis and Functions in Health and Disease. Nat Rev Endocrinol 2023, 19, 443–459. [Google Scholar] [CrossRef]
- Farmer, B.C.; Walsh, A.E.; Kluemper, J.C.; Johnson, L.A. Lipid Droplets in Neurodegenerative Disorders. Front. Neurosci. 2020, 14, 742. [Google Scholar] [CrossRef]
- Yin, F. Lipid Metabolism and Alzheimer’s Disease: Clinical Evidence, Mechanistic Link and Therapeutic Promise. The FEBS Journal 2023, 290, 1420–1453. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Song, X.; Wang, Y.; Huang, L.; Luo, W.; Li, F.; Qin, S.; Wang, Y.; Xiao, J.; Wu, Y.; et al. Dysregulation of Cofilin-1 Activity—the Missing Link between Herpes Simplex Virus Type-1 Infection and Alzheimer’s Disease. Critical Reviews in Microbiology 2020, 46, 381–396. [Google Scholar] [CrossRef]
- Smith, L.K.; Babcock, I.W.; Minamide, L.S.; Shaw, A.E.; Bamburg, J.R.; Kuhn, T.B. Direct Interaction of HIV Gp120 with Neuronal CXCR4 and CCR5 Receptors Induces Cofilin-Actin Rod Pathology via a Cellular Prion Protein- and NOX-Dependent Mechanism. PLoS ONE 2021, 16, e0248309. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Ding, Y.; Chen, Y.; Zhang, S.; Huo, C.; Wang, Y.; Yu, J.; Zhang, P.; Na, H.; Zhang, H.; et al. The Proteomics of Lipid Droplets: Structure, Dynamics, and Functions of the Organelle Conserved from Bacteria to Humans. J Lipid Res 2012, 53, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-Y.; Leu, J.-D.; Lee, Y.-J. The Actin Depolymerizing Factor (ADF)/Cofilin Signaling Pathway and DNA Damage Responses in Cancer. IJMS 2015, 16, 4095–4120. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Elzakra, N.; Xu, S.; Xiao, G.G.; Yang, Y.; Hu, S. Investigation of Three Potential Autoantibodies in Sjogren’s Syndrome and Associated MALT Lymphoma. Oncotarget 2017, 8, 30039–30049. [Google Scholar] [CrossRef]
- Hou, M.; Liu, X.; Cao, J.; Chen, B. SEPT7 Overexpression Inhibits Glioma Cell Migration by Targeting the Actin Cytoskeleton Pathway. Oncology Reports 2016, 35, 2003–2010. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



