Submitted:
04 February 2025
Posted:
05 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Epidemiology of SCLC
1.2. Staging of SCLC
1.3. Challenges in Treating SCLC
1.4. Review Objectives
2. Current Status of Limited Stage Small Cell Lung Cancer (LS-SCLC)
2.1. Impact of Lung Cancer Screening
2.2. Current Standard of Care in LS-SCLC
2.3. Multimodal Treatment Approaches
2.3.1. Role of Surgery
2.3.2. Advances in Concomitant Radiation Therapy
2.4. Future of Consolidation Therapies in LS-SCLC
2.5. Smoking Cessation and Supportive Care
3. Current Status of Extensive Stage SCLC (ES-SCLC)
3.1. Current First-Line Systemic Therapy
3.2. Concurrent, Maintenance and Consolidation Immunotherapy for ES-SCLC
3.3. Role of Radiation Therapy in ES-SCLC
3.3.1. The Status of Consolidation Radiation Therapy
3.4. Why and how personalized therapies may be useful
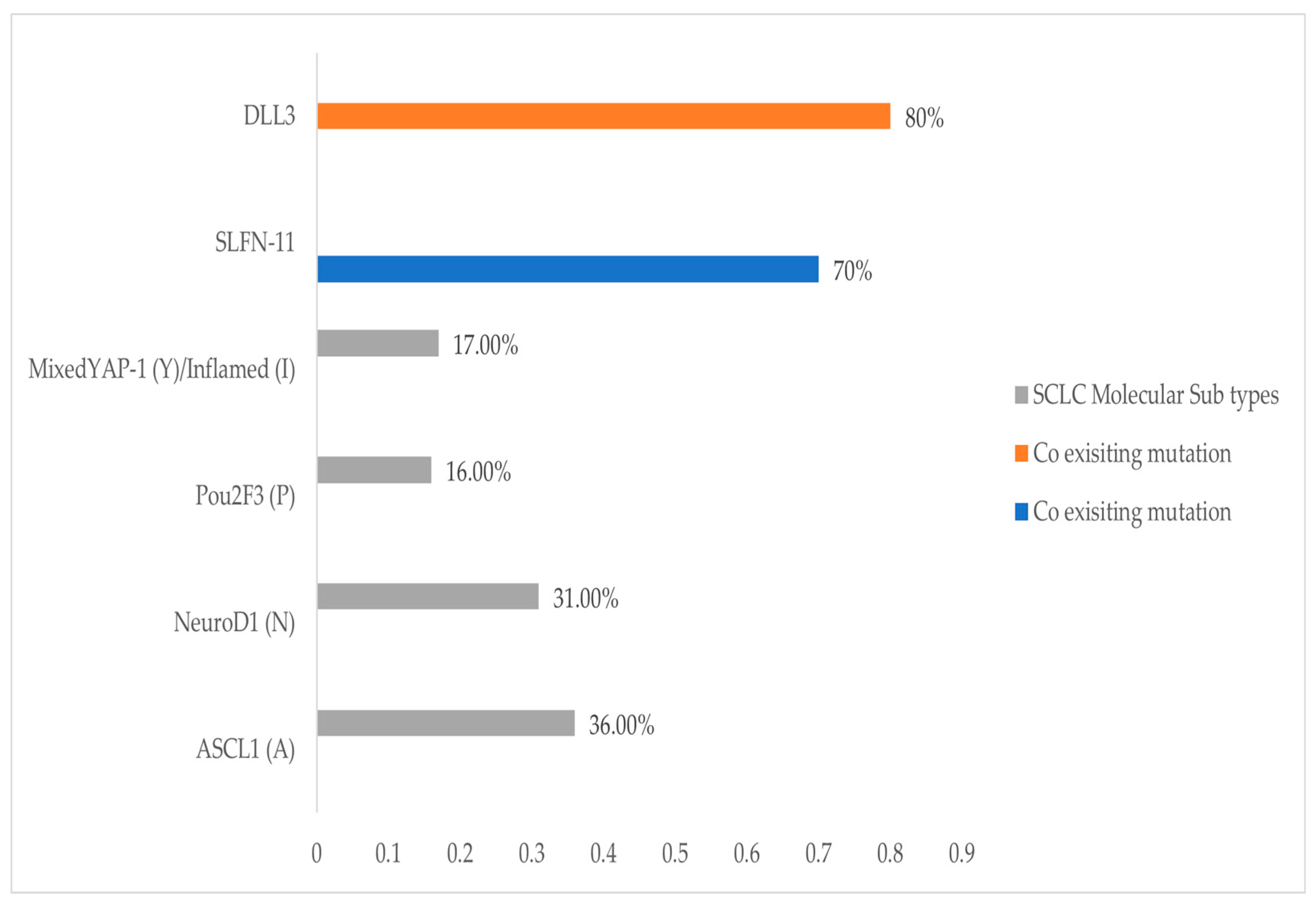
3.4.1. SLFN11
3.4.2. DLL3
3.4.3. Molecular Subtypes and Treatment Implications
3.4.4. Molecular Biomarkers
3.5. The Role of Prophylactic Cranial Radiation
3.6. Second Line Therapeutic Options:
4. Emerging Therapeutic Strategies and Future Directions in SCLC
4.1. Advances in ES-SCLC Therapies: Immunotherapy and Targeted Treatment Advances
4.2. Clinical Trials and Investigational Agents
4.2.1. Evolving therapies in LS-SCLC
4.2.2. Evolving therapies in ES-SCLC
4.3. Novel Therapeutic Approaches
4.3.1. Immunotherapy Beyond First Line
4.3.2. Antibody-Drug Conjugates
4.4. Special Populations
4.4.1. Patients with Brain Metastases
4.4.2. Patients with Borderline Performance Status
4.4.3. Elderly
4.4.4. Patients with SCLC transformation and previous EGFR Mutant Non-Small Cell Lung Cancer Diagnosis
4.4.5. Paraneoplastic Syndromes with Neurologic Manifestations
4.4.6. Paraneoplastic Syndromes without Neurologic Manifestations
4.5. Supportive Care and Quality of Life
5. Future Directions and Research Opportunities
5.1. Biomarker Development
5.2. Overcoming Drug Resistance
5.3. Cost-Effectiveness and Biomarker-Driven Therapies
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Govindan, R.; Page, N.; Morgensztern, D.; Read, W.; Tierney, R.; Vlahiotis, A.; Spitznagel, E.L.; Piccirillo, J. Changing Epidemiology of Small-Cell Lung Cancer in the United States Over the Last 30 Years: Analysis of the Surveillance, Epidemiologic, and End Results Database. J. Clin. Oncol. 2006, 24, 4539–4544. [Google Scholar] [CrossRef] [PubMed]
- Pietanza, M.C.; Byers, L.A.; Minna, J.D.; Rudin, C.M. Small Cell Lung Cancer: Will Recent Progress Lead to Improved Outcomes? Clin. Cancer Res. 2015, 21, 2244–2255. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Gümüş, Z.H.; Colarossi, C.; Memeo, L.; Wang, X.; Kong, C.Y.; Boffetta, P. SCLC: Epidemiology, Risk Factors, Genetic Susceptibility, Molecular Pathology, Screening, and Early Detection. J. Thorac. Oncol. 2022, 18, 31–46. [Google Scholar] [CrossRef] [PubMed]
- https://seer.cancer.gov/statisticsnetwork. Accessed 1.3.2025.
- Zelen, M. Keynote address on biostatistics and data retrieval. Cancer Chemother Rep 3. 1973, 4(2), 31–42. [Google Scholar]
- Micke, P.; Faldum, A.; Metz, T.; Beeh, K.-M.; Bittinger, F.; Hengstler, J.G.; Buhl, R. Staging small cell lung cancer: Veterans Administration Lung Study Group versus International Association for the Study of Lung Cancer—What limits limited disease? Lung Cancer 2002, 37, 271–276. [Google Scholar] [CrossRef]
- Nicholson, A.G.; Chansky, K.; Crowley, J.; Beyruti, R.; Kubota, K.; Turrisi, A.; van Meerbeeck, J.; Rami-Porta, R.; Goldstraw, P.; Asamura, H.; et al. The International Association for the Study of Lung Cancer Lung Cancer Staging Project: Proposals for the Revision of the Clinical and Pathologic Staging of Small Cell Lung Cancer in the Forthcoming Eighth Edition of the TNM Classification for Lung Cancer. J. Thorac. Oncol. 2015, 11, 300–311. [Google Scholar] [CrossRef]
- Li, H.; Zhao, Y.; Ma, T.; Shao, H.; Wang, T.; Jin, S.; Liu, Z. Radiotherapy for extensive-stage small-cell lung cancer in the immunotherapy era. Front. Immunol. 2023, 14, 1132482. [Google Scholar] [CrossRef]
- Kalemkerian, G.P. Staging and imaging of small cell lung cancer. Cancer Imaging 2011, 11, 253–258. [Google Scholar] [CrossRef]
- Arriola, E.; Trigo, J.M.; Sánchez-Gastaldo, A.; Navarro, A.; Perez, C.; Crama, L.; Ponce-Aix, S. Prognostic Value of Clinical Staging According to TNM in Patients With SCLC: A Real-World Surveillance Epidemiology and End-Results Database Analysis. JTO Clin. Res. Rep. 2021, 3, 100266. [Google Scholar] [CrossRef]
- Final Recommendation Statement: Lung Cancer: Screening. 2021; Available from: https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening.
- Thomas, A.; Pattanayak, P.; Szabo, E.; Pinsky, P. Characteristics and Outcomes of Small Cell Lung Cancer Detected by CT Screening. Chest 2018, 154, 1284–1290. [Google Scholar] [CrossRef]
- Silva, M.; Galeone, C.; Sverzellati, N.; Marchianò, A.; Calareso, G.; Sestini, S.; La Vecchia, C.; Sozzi, G.; Pelosi, G.; Pastorino, U. Screening with Low-Dose Computed Tomography Does Not Improve Survival of Small Cell Lung Cancer. 11. [CrossRef]
- Haaf, K.T.; van Rosmalen, J.; de Koning, H.J. Lung Cancer Detectability by Test, Histology, Stage, and Gender: Estimates from the NLST and the PLCO Trials. Cancer Epidemiology Biomarkers Prev. 2015, 24, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Fedewa, S.A. Lung Cancer Screening With Low-Dose Computed Tomography in the United States—2010 to 2015. JAMA Oncol. 2017, 3, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Li, Y.; Wang, Q.; Fujiwara, Y.; Kurbanova, T.; Theodoropoulos, N. MA04.02 Lung Cancer Screening Utilization and Its Correlates in Sexual Minorities: An Analysis of the BRFSS 2018. J. Thorac. Oncol. 2021, 16, S144. [Google Scholar] [CrossRef]
- Melzer, A.C.; Wilt, T.J. Expanded Access to Lung Cancer Screening—Implementing Wisely to Optimize Health. JAMA Netw. Open 2021, 4, e210275–e210275. [Google Scholar] [CrossRef]
- Cheng, Y.; Spigel, D.R.; Cho, B.C.; Laktionov, K.K.; Fang, J.; Chen, Y.; Zenke, Y.; Lee, K.H.; Wang, Q.; Navarro, A.; et al. Durvalumab after Chemoradiotherapy in Limited-Stage Small-Cell Lung Cancer. New Engl. J. Med. 2024, 391, 1313–1327. [Google Scholar] [CrossRef]
- https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-limited-stage-small-cell-lung-cancer (accessed on 30th of 24. 20 December.
- Bogart, J.; Wang, X.; Masters, G.; Gao, J.; Komaki, R.; Gaspar, L.E.; Heymach, J.; Bonner, J.; Kuzma, C.; Waqar, S.; et al. High-Dose Once-Daily Thoracic Radiotherapy in Limited-Stage Small-Cell Lung Cancer: CALGB 30610 (Alliance)/RTOG 0538. J. Clin. Oncol. 2023, 41, 2394–2402. [Google Scholar] [CrossRef]
- Faivre-Finn, C.; Snee, M.; Ashcroft, L.; Appel, W.; Barlesi, F.; Bhatnagar, A.; Bezjak, A.; Cardenal, F.; Fournel, P.; Harden, S.; et al. Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol. 2017, 18, 1116–1125. [Google Scholar] [CrossRef]
- van Meerbeeck, J.P.; A Fennell, D.; De Ruysscher, D.K. Small-cell lung cancer. Lancet 2011, 378, 1741–1755. [Google Scholar] [CrossRef]
- Hoda, M.A.; Klikovits, T.; Klepetko, W. Controversies in oncology: surgery for small cell lung cancer? It's time to rethink the case. ESMO Open 2018, 3, e000366. [Google Scholar] [CrossRef]
- Aupérin, A.; Arriagada, R.; Pignon, J.-P.; Le Péchoux, C.; Gregor, A.; Stephens, R.J.; Kristjansen, P.E.; Johnson, B.E.; Ueoka, H.; Wagner, H.; et al. Prophylactic Cranial Irradiation for Patients with Small-Cell Lung Cancer in Complete Remission. New Engl. J. Med. 1999, 341, 476–484. [Google Scholar] [CrossRef]
- Turrisi, A.T. Limited stage small cell lung cancer: treatment and therapy. Curr. Treat. Options Oncol. 2003, 4, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Lad, T.; Piantadosi, S.; Thomas, P.; Payne, D.; Ruckdeschel, J.; Giaccone, G. A Prospective Randomized Trial to Determine the Benefit of Surgical Resection of Residual Disease Following Response of Small Cell Lung Cancer to Combination Chemotherapy. Chest 1994, 106, 320S–323S. [Google Scholar] [CrossRef] [PubMed]
- Fox, W.; Scadding, J. MEDICAL RESEARCH COUNCIL COMPARATIVE TRIAL OF SURGERY AND RADIOTHERAPY FOR PRIMARY TREATMENT OF SMALL-CELLED OR OAT-CELLED CARCINOMA OF BRONCHUS. Lancet 1973, 302, 63–65. [Google Scholar] [CrossRef]
- Liao, M.; Zhao, J.; Zhou, Y. [Multimodality therapy of late stage lung cancer]. 1995, 17, 384–6.
- Liu, T.; Chen, Z.; Dang, J.; Li, G. The role of surgery in stage I to III small cell lung cancer: A systematic review and meta-analysis. PLOS ONE 2018, 13, e0210001. [Google Scholar] [CrossRef] [PubMed]
- Wakeam, E.; Acuna, S.; Leighl, N.; Giuliani, M.; Finlayson, S.; Varghese, T.; Darling, G. Surgery Versus Chemotherapy and Radiotherapy For Early and Locally Advanced Small Cell Lung Cancer: A Propensity-Matched Analysis of Survival. 109, 88. [CrossRef]
- Tsuchiya, R.; Suzuki, K.; Ichinose, Y.; Watanabe, Y.; Yasumitsu, T.; Ishizuka, N.; Kato, H. Phase II trial of postoperative adjuvant cisplatin and etoposide in patients with completely resected stage I-IIIa small cell lung cancer: The Japan Clinical Oncology Lung Cancer Study Group Trial (JCOG9101). J. Thorac. Cardiovasc. Surg. 2005, 129, 977–983. [Google Scholar] [CrossRef]
- Vallières, E.; Shepherd, F.A.; Crowley, J.; Van Houtte, P.; Postmus, P.E.; Carney, D.; Chansky, K.; Shaikh, Z.; Goldstraw, P. The IASLC Lung Cancer Staging Project: Proposals Regarding the Relevance of TNM in the Pathologic Staging of Small Cell Lung Cancer in the Forthcoming (Seventh) Edition of the TNM Classification for Lung Cancer. J. Thorac. Oncol. 2009, 4, 1049–1059. [Google Scholar] [CrossRef]
- Yu, J.B.; Decker, R.H.; Detterbeck, F.C.; Wilson, L.D. Surveillance Epidemiology and End Results Evaluation of the Role of Surgery for Stage I Small Cell Lung Cancer. J. Thorac. Oncol. 2010, 5, 215–219. [Google Scholar] [CrossRef]
- Yang, C.-F.J.; Chan, D.Y.; Speicher, P.J.; Gulack, B.C.; Wang, X.; Hartwig, M.G.; Onaitis, M.W.; Tong, B.C.; D’amico, T.A.; Berry, M.F.; et al. Role of Adjuvant Therapy in a Population-Based Cohort of Patients With Early-Stage Small-Cell Lung Cancer. J. Clin. Oncol. 2016, 34, 1057–1064. [Google Scholar] [CrossRef]
- Badzio, A. A retrospective comparative study of surgery followed by chemotherapy vs. non-surgical management in limited-disease small cell lung cancer. Eur. J. Cardio-Thoracic Surg. [CrossRef]
- Ettinger, D.S.; Wood, D.E.; Aisner, D.L.; Akerley, W.; Bauman, J.R.; Bharat, A.; Bruno, D.S.; Chang, J.Y.; Chirieac, L.R.; D’amico, T.A.; et al. Non–Small Cell Lung Cancer, Version 3.2022, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2022, 20, 497–530. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, D.; Zhou, X.; Bao, W.; Ji, Y.; Sheng, L.; Cheng, L.; Chen, Y.; Du, X.; Qiu, G. Prophylactic cranial irradiation in resected small cell lung cancer: A systematic review with meta-analysis. J. Cancer 2018, 9, 433–439. [Google Scholar] [CrossRef]
- Barnes, H.; See, K.; Barnett, S.; Manser, R. Surgery for limited-stage small-cell lung cancer. Cochrane Database Syst. Rev. 2017, 2017, CD011917. [Google Scholar] [CrossRef]
- Martucci, N.; Morabito, A.; La Rocca, A.; De Luca, G.; De Cecio, R.; Botti, G.; Totaro, G.; Muto, P.; Picone, C.; Esposito, G.; et al. Surgery in Small-Cell Lung Cancer. Cancers 2021, 13, 390. [Google Scholar] [CrossRef] [PubMed]
- Turrisi, A.T.; Kim, K.; Blum, R.; Sause, W.T.; Livingston, R.B.; Komaki, R.; Wagner, H.; Aisner, S.; Johnson, D.H. Twice-Daily Compared with Once-Daily Thoracic Radiotherapy in Limited Small-Cell Lung Cancer Treated Concurrently with Cisplatin and Etoposide. New Engl. J. Med. 1999, 340, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, D.; Wong, A.T.; Schwartz, D.; Rineer, J. Utilization of Hyperfractionated Radiation in Small-Cell Lung Cancer and Its Impact on Survival. J. Thorac. Oncol. 2015, 10, 1770–1775. [Google Scholar] [CrossRef]
- Grønberg, B.H.; Killingberg, K.T.; Fløtten. ; Brustugun, O.T.; Hornslien, K.; Madebo, T.; Langer, S.W.; Schytte, T.; Nyman, J.; Risum, S.; et al. High-dose versus standard-dose twice-daily thoracic radiotherapy for patients with limited stage small-cell lung cancer: an open-label, randomised, phase 2 trial. Lancet Oncol. 2021, 22, 321–331. [Google Scholar] [CrossRef]
- Shirvani, S.M.; Juloori, A.; Allen, P.K.; Komaki, R.; Liao, Z.; Gomez, D.; O'Reilly, M.; Welsh, J.; Papadimitrakopoulou, V.; Cox, J.D.; et al. Comparison of 2 Common Radiation Therapy Techniques for Definitive Treatment of Small Cell Lung Cancer. Int. J. Radiat. Oncol. 2013, 87, 139–147. [Google Scholar] [CrossRef]
- Martinussen, H.M.; Reymen, B.; Wanders, R.; Troost, E.G.; Dingemans, A.-M.C.; Öllers, M.; Houben, R.; De Ruysscher, D.; Lambin, P.; van Baardwijk, A. Is selective nodal irradiation in non-small cell lung cancer still safe when using IMRT? Results of a prospective cohort study. Radiother. Oncol. 2016, 121, 322–327. [Google Scholar] [CrossRef]
- Noronha, V.; Sekhar, A.; Patil, V.M.; Menon, N.; Joshi, A.; Kapoor, A.; Prabhash, K. Systemic therapy for limited stage small cell lung carcinoma. J. Thorac. Dis. 2020, 12, 6275–6290. [Google Scholar] [CrossRef]
- Christodoulou, M.; Blackhall, F.; Mistry, H.; Leylek, A.; Knegjens, J.; Remouchamps, V.; Martel-Lafay, I.; Farré, N.; Zwitter, M.; Lerouge, D.; et al. Compliance and Outcome of Elderly Patients Treated in the Concurrent Once-Daily Versus Twice-Daily Radiotherapy (CONVERT) Trial. J. Thorac. Oncol. 2018, 14, 63–71. [Google Scholar] [CrossRef]
- Salem, A.; Mistry, H.; Hatton, M.; Locke, I.; Monnet, I.; Blackhall, F.; Faivre-Finn, C. Association of Chemoradiotherapy With Outcomes Among Patients With Stage I to II vs Stage III Small Cell Lung Cancer. JAMA Oncol. 2019, 5, e185335–e185335. [Google Scholar] [CrossRef]
- Shepherd, F.A.; Crowley, J.; Van Houtte, P.; Postmus, P.E.; Carney, D.; Chansky, K.; Shaikh, Z.; Goldstraw, P. The International Association for the Study of Lung Cancer Lung Cancer Staging Project: Proposals Regarding the Clinical Staging of Small Cell Lung Cancer in the Forthcoming (Seventh) Edition of the Tumor, Node, Metastasis Classification for Lung Cancer. J. Thorac. Oncol. 2007, 2, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Manoharan, P.; Salem, A.; Mistry, H.; Gornall, M.; Harden, S.; Julyan, P.; Locke, I.; McAleese, J.; McMenemin, R.; Mohammed, N.; et al. 18F-Fludeoxyglucose PET/CT in SCLC: Analysis of the CONVERT Randomized Controlled Trial. J. Thorac. Oncol. 2019, 14, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Walls, G.M.; Mistry, H.; Barlesi, F.; Bezjak, A.; Le Pechoux, C.; O'Brien, M.; Van Meerbeeck, J.P.; Blackhall, F.; Faivre-Finn, C. Long-Term Outcomes After Concurrent Once- or Twice-Daily Chemoradiation in Limited-Stage Small Cell Lung Cancer: A Brief Report From the CONVERT Trial. Int. J. Radiat. Oncol. 2024, 119, 1386–1390. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. NCCN Guidelines, /: Available from:https, 1462.
- Higgins, K.; Hu, C.; Ross, H.; Jabbour, S.; Kozono, D.; Owonikoko, T.; Dib, E.; Brownstein, J.; Kuzma, C.; Kotecha, R.; et al. Concurrent Chemoradiation ± Atezolizumab (atezo) in Limited-Stage Small Cell Lung Cancer (LS-SCLC): Results of NRG Oncology/Alliance LU005. Int. J. Radiat. Oncol. 2024, 120, S2. [Google Scholar] [CrossRef]
- Kenfield, S.A.; Stampfer, M.J.; Rosner, B.A.; Colditz, G.A. Smoking and Smoking Cessation in Relation to Mortality in Women. JAMA 2008, 299, 2037–2047. [Google Scholar] [CrossRef]
- Feigin, V.L.; Stark, B.A.; Johnson, C.O.; Roth, G.A.; Bisignano, C.; Abady, G.G.; Abbasifard, M.; Abbasi-Kangevari, M.; Abd-Allah, F.; Abedi, V.; et al. Global, regional, and national burden of stroke and its risk factors, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021, 20, 795–820. [Google Scholar] [CrossRef]
- Deeks, J.J.; Higgins, J.P.; Altman, D.G. ; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2019; pp. 241–284, https://doi.org/10.1002/9781119536604.ch10.meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2019; Sons, Ltd.: Hoboken, NJ, USA, 2019; pp. 241–284. [Google Scholar] [CrossRef]
- Hayden, J.A.; Van Der Windt, D.A.; Cartwright, J.L.; Côté, P.; Bombardier, C. Assessing Bias in Studies of Prognostic Factors. Ann. Intern. Med. 2013, 158, 280–286. [Google Scholar] [CrossRef]
- LC survival rates. 2021; Available from: https://www.cancer.org/cancer/lung-cancer/detection-diagnosis-staging/survival-rates.html.
- Chen, J.; Jiang, R.; Garces, Y.I.; Jatoi, A.; Stoddard, S.M.; Sun, Z.; Marks, R.S.; Liu, Y.; Yang, P. Prognostic factors for limited-stage small cell lung cancer: A study of 284 patients. Lung Cancer 2009, 67, 221–226. [Google Scholar] [CrossRef]
- Lung Cancer Fact Sheet. 2021; Available from: https://gco.iarc.fr/today/data/factsheets/cancers/15-Lung-fact-sheet.
- Feigin, V.L.; Abate, M.D.; Abate, Y.H.; ElHafeez, S.A.; Abd-Allah, F.; Abdelalim, A.; Abdelkader, A.; Abdelmasseh, M.; Abd-Elsalam, S.; Abdi, P.; et al. Global, regional, and national burden of stroke and its risk factors, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Neurol. 2024, 23, 973–1003. [Google Scholar] [CrossRef]
- Mascaux, C.; Paesmans, M.; Berghmans, T.; Branle, F.; Lafitte, J.; Lemaı̂tre, F.; Meert, A.; Vermylen, P.; Sculier, J. A systematic review of the role of etoposide and cisplatin in the chemotherapy of small cell lung cancer with methodology assessment and meta-analysis. 30, 36. [CrossRef]
- Rossi, A.; Di Maio, M.; Chiodini, P.; Rudd, R.M.; Okamoto, H.; Skarlos, D.V.; Früh, M.; Qian, W.; Tamura, T.; Samantas, E.; et al. Carboplatin- or Cisplatin-Based Chemotherapy in First-Line Treatment of Small-Cell Lung Cancer: The COCIS Meta-Analysis of Individual Patient Data. J. Clin. Oncol. 2012, 30, 1692–1698. [Google Scholar] [CrossRef]
- Horn, L.; Mansfield, A.S.; Szczęsna, A.; Havel, L.; Krzakowski, M.; Hochmair, M.J.; Huemer, F.; Losonczy, G.; Johnson, M.L.; Nishio, M.; et al. First-Line Atezolizumab plus Chemotherapy in Extensive-Stage Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2220–2229. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Dziadziuszko, R.; Sugawara, S.; Kao, S.; Hochmair, M.; Huemer, F.; Castro, G.; Havel, L.; Caro, R.; Losonczy, G.; et al. OA01.04 Five-Year Survival in Patients with ES-SCLC Treated with Atezolizumab in IMpower133: Imbrella a Extension Study Results. J. Thorac. Oncol. 2023, 18, S44–S45. [Google Scholar] [CrossRef]
- Farago, A.F.; Keane, F.K. Current standards for clinical management of small cell lung cancer. Transl. Lung Cancer Res. 2018, 7, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Roth, B.J.; Johnson, D.H.; Einhorn, L.H.; Schacter, L.P.; Cherng, N.C.; Cohen, H.J.; Crawford, J.; A Randolph, J.; Goodlow, J.L.; O Broun, G. Randomized study of cyclophosphamide, doxorubicin, and vincristine versus etoposide and cisplatin versus alternation of these two regimens in extensive small-cell lung cancer: a phase III trial of the Southeastern Cancer Study Group. J. Clin. Oncol. 1992, 10, 282–291. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Dvorkin, M.; Chen, Y.; Reinmuth, N.; Hotta, K.; Trukhin, D.; Statsenko, G.; Hochmair, M.J.; Özgüroğlu, M.; Ji, J.H.; et al. Durvalumab plus platinum–etoposide versus platinum–etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, phase 3 trial. Lancet 2019, 394, 1929–1939. [Google Scholar] [CrossRef]
- Goldman, J.W.; Dvorkin, M.; Chen, Y.; Reinmuth, N.; Hotta, K.; Trukhin, D.; Statsenko, G.; Hochmair, M.J.; Ji, J.H.; Garassino, M.C.; et al. Durvalumab, with or without tremelimumab, plus platinum–etoposide versus platinum–etoposide alone in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): updated results from a randomised, controlled, open-label, phase 3 trial. 22, 65. [CrossRef]
- Spigel, D.R.; Cheng, Y.; Cho, B.C.; Laktionov, K.K.; Fang, J.; Chen, Y.; Zenke, Y.; Lee, K.H.; Wang, Q.; Navarro, A.; et al. ADRIATIC: Durvalumab (D) as consolidation treatment (tx) for patients (pts) with limited-stage small-cell lung cancer (LS-SCLC). J. Clin. Oncol. 2024, 42, LBA5–LBA5. [Google Scholar] [CrossRef]
- Xie, M.; Vuko, M.; Rodriguez-Canales, J.; Zimmermann, J.; Schick, M.; O’brien, C.; Paz-Ares, L.; Goldman, J.W.; Garassino, M.C.; Gay, C.M.; et al. Molecular classification and biomarkers of outcome with immunotherapy in extensive-stage small-cell lung cancer: analyses of the CASPIAN phase 3 study. Mol. Cancer 2024, 23, 1–17. [Google Scholar] [CrossRef]
- Concannon, K.; Heeke, S.; Sahu, M.; Tang, X.; Sasaki, K.; Patel, S.; Raso, M.G.; Tran, H.; Gay, C.; Byers, L. Abstract 1398: Clinical and transcriptomic analysis demonstrates improved survival and unique gene expression signatures among SCLC arising in patients with minimal tobacco use. Cancer Res. 2023, 83, 1398–1398. [Google Scholar] [CrossRef]
- Genentech Provides Update on Phase III SKYSCRAPER-02 Study in Extensive-Stage Small Cell Lung Cancer. 2022.
- Jazz Pharmaceuticals Announces Statistically Significant Overall Survival and Progression-Free Survival Results for Zepzelca® (lurbinectedin) and Atezolizumab Combination in First-Line Maintenance Therapy for Extensive-Stage Small Cell Lung Cancer. 2024; Available from: https://investor.jazzpharma.com/news-releases/news-release-details/jazz-pharmaceuticals-announces-statistically-significant-overall. National Library of Medicine (U.S.), Testing the Addition of Radiation Therapy to the Usual Immune Therapy Treatment (Atezolizumab) for Extensive Stage Small Cell Lung Cancer, The RAPTOR Trial. Identifier NCT04402788. [Google Scholar]
- Jeremic, B.; Shibamoto, Y.; Nikolic, N.; Milicic, B.; Milisavljevic, S.; Dagovic, A.; Aleksandrovic, J.; Radosavljevic-Asic, G. Role of Radiation Therapy in the Combined-Modality Treatment of Patients With Extensive Disease Small-Cell Lung Cancer: A Randomized Study. J. Clin. Oncol. 1999, 17, 2092–2092. [Google Scholar] [CrossRef]
- Slotman, B.J.; van Tinteren, H.; O Praag, J.; Knegjens, J.L.; El Sharouni, S.Y.; Hatton, M.; Keijser, A.; Faivre-Finn, C.; Senan, S. Use of thoracic radiotherapy for extensive stage small-cell lung cancer: a phase 3 randomised controlled trial. Lancet 2015, 385, 36–42. [Google Scholar] [CrossRef]
- Gore, E.M.; Hu, C.; Sun, A.Y.; Grimm, D.F.; Ramalingam, S.S.; Dunlap, N.E.; Higgins, K.A.; Werner-Wasik, M.; Allen, A.M.; Iyengar, P.; et al. Randomized Phase II Study Comparing Prophylactic Cranial Irradiation Alone to Prophylactic Cranial Irradiation and Consolidative Extracranial Irradiation for Extensive-Disease Small Cell Lung Cancer (ED SCLC): NRG Oncology RTOG 0937. J. Thorac. Oncol. 2017, 12, 1561–1570. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Ramkumar, K.; Cardnell, R.J.; Gay, C.M.; Stewart, C.A.; Wang, W.-L.; Fujimoto, J.; Wistuba, I.I.; Byers, L.A. A wake-up call for cancer DNA damage: the role of Schlafen 11 (SLFN11) across multiple cancers. Br. J. Cancer 2021, 125, 1333–1340. [Google Scholar] [CrossRef]
- Sivakumar, S.; Moore, J.A.; Montesion, M.; Sharaf, R.; Lin, D.I.; Colón, C.I.; Fleischmann, Z.; Ebot, E.M.; Newberg, J.Y.; Mills, J.M.; et al. Integrative Analysis of a Large Real-World Cohort of Small Cell Lung Cancer Identifies Distinct Genetic Subtypes and Insights into Histologic Transformation. Cancer Discov. 2023, 13, 1572–1591. [Google Scholar] [CrossRef] [PubMed]
- Inno, A.; Stagno, A.; Gori, S. Schlafen-11 (SLFN11): a step forward towards personalized medicine in small-cell lung cancer? Transl. Lung Cancer Res. 2018, 7, S341–S345. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C.M.; Poirier, J.T.; Byers, L.A.; Dive, C.; Dowlati, A.; George, J.; Heymach, J.V.; Johnson, J.E.; Lehman, J.M.; MacPherson, D.; et al. Molecular subtypes of small cell lung cancer: a synthesis of human and mouse model data. Nat. Rev. Cancer 2019, 19, 289–297. [Google Scholar] [CrossRef]
- Karim, N.A.; Miao, J.; Reckamp, K.L.; Gay, C.M.; Byers, L.A.; Zhao, Y.-Q.; Redman, M.W.; Carrizosa, D.R.; Wang, W.-L.; Petty, W.J.; et al. Phase II randomized study of maintenance atezolizumab (A) versus atezolizumab + talazoparib (AT) in patients with SLFN11 positive extensive stage small cell lung cancer. S1929. S1929. J. Thorac. Oncol. 2024. [CrossRef]
- Yang, W.; Wang, W.; Li, Z.; Wu, J.; Huang, X.; Li, J.; Zhang, X.; Ye, X. Delta-like ligand 3 in small cell lung cancer: Potential mechanism and treatment progress. Crit. Rev. Oncol. 2023, 191, 104136. [Google Scholar] [CrossRef]
- Bellis, R.Y.; Adusumilli, P.S.; Amador-Molina, A. DLL3-targeted CAR T-cell therapy in pre-clinical models for small cell lung cancer: safety, efficacy, and challenges. Transl. Lung Cancer Res. 2024, 13, 694–698. [Google Scholar] [CrossRef]
- Puri, S.; Naqash, A.R.; Elliott, A.; Kerrigan, K.C.; Patel, S.B.; Seeber, A.; Kocher, F.; Uprety, D.; Mamdani, H.; Kulkarni, A.; et al. Real-world multiomic characterization of small cell lung cancer subtypes to reveal differential expression of clinically relevant biomarkers. J. Clin. Oncol. 2021, 39, 8508–8508. [Google Scholar] [CrossRef]
- Ireland, A.S.; Micinski, A.M.; Kastner, D.W.; Guo, B.; Wait, S.J.; Spainhower, K.B.; Conley, C.C.; Chen, O.S.; Guthrie, M.R.; Soltero, D.; et al. MYC Drives Temporal Evolution of Small Cell Lung Cancer Subtypes by Reprogramming Neuroendocrine Fate. Cancer Cell 2020, 38, 60–78e12. [Google Scholar] [CrossRef]
- Owonikoko, T.K.; Dwivedi, B.; Chen, Z.; Zhang, C.; Barwick, B.; Ernani, V.; Zhang, G.; Gilbert-Ross, M.; Carlisle, J.; Khuri, F.R.; et al. YAP1 Expression in SCLC Defines a Distinct Subtype With T-cell–Inflamed Phenotype. J. Thorac. Oncol. 2020, 16, 464–476. [Google Scholar] [CrossRef]
- Gay, C.M.; Stewart, C.A.; Park, E.M.; Diao, L.; Groves, S.M.; Heeke, S.; Nabet, B.Y.; Fujimoto, J.; Solis, L.M.; Lu, W.; et al. Patterns of transcription factor programs and immune pathway activation define four major subtypes of SCLC with distinct therapeutic vulnerabilities. 2021, 39, 346–360. e7. [CrossRef]
- Hu, H.; Qiu, Y.; Guo, M.; Huang, Y.; Fang, L.; Peng, Z.; Ji, W.; Xu, Y.; Shen, S.; Yan, Y.; et al. Targeted Hsp70 expression combined with CIK-activated immune reconstruction synergistically exerts antitumor efficacy in patient-derived hepatocellular carcinoma xenograft mouse models. Oncotarget 2014, 6, 1079–1089. [Google Scholar] [CrossRef] [PubMed]
- Lantuejoul, S.; Fernandez-Cuesta, L.; Damiola, F.; Girard, N.; McLeer, A. New molecular classification of large cell neuroendocrine carcinoma and small cell lung carcinoma with potential therapeutic impacts. Transl. Lung Cancer Res. 2020, 9, 2233–2244. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Randulfe, I.; Leporati, R.; Gupta, B.; Liu, S.; Califano, R. Recent advances and future strategies in first-line treatment of ES-SCLC. Eur. J. Cancer 2024, 200, 113581. [Google Scholar] [CrossRef]
- Yu, L.; Lai, Q.; Gou, L.; Feng, J.; Yang, J. Opportunities and obstacles of targeted therapy and immunotherapy in small cell lung cancer. J. Drug Target. 2020, 29, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Padda, S.K.; Weiss, J.; Owonikoko, T.K. Advances in Treatment of Recurrent Small Cell Lung Cancer (SCLC): Insights for Optimizing Patient Outcomes from an Expert Roundtable Discussion. Adv. Ther. 2021, 38, 5431–5451. [Google Scholar] [CrossRef]
- Reck, M.; Liu, S.; Mansfield, A.; Mok, T.; Scherpereel, A.; Reinmuth, N.; Garassino, M.; De Carpeno, J.; Califano, R.; Nishio, M.; et al. IMpower133: Updated overall survival (OS) analysis of first-line (1L) atezolizumab (atezo) + carboplatin + etoposide in extensive-stage SCLC (ES-SCLC). Ann. Oncol. 2019, 30, v710–v711. [Google Scholar] [CrossRef]
- Zhu, L.; Qin, J. Predictive biomarkers for immunotherapy response in extensive-stage SCLC. J. Cancer Res. Clin. Oncol. 2024, 150, 1–11. [Google Scholar] [CrossRef]
- Herbreteau, G.; Langlais, A.; Greillier, L.; Audigier-Valette, C.; Uwer, L.; Hureaux, J.; Moro-Sibilot, D.; Guisier, F.; Carmier, D.; Madelaine, J.; et al. Circulating Tumor DNA as a Prognostic Determinant in Small Cell Lung Cancer Patients Receiving Atezolizumab. J. Clin. Med. 2020, 9, 3861. [Google Scholar] [CrossRef]
- Nong, J.; Gong, Y.; Guan, Y.; Yi, X.; Yi, Y.; Chang, L.; Yang, L.; Lv, J.; Guo, Z.; Jia, H.; et al. Circulating tumor DNA analysis depicts subclonal architecture and genomic evolution of small cell lung cancer. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef]
- Sivapalan, L.; Iams, W.T.; Belcaid, Z.; Scott, S.C.; Niknafs, N.; Balan, A.; White, J.R.; Kopparapu, P.; Cann, C.; Landon, B.V.; et al. Dynamics of Sequence and Structural Cell-Free DNA Landscapes in Small-Cell Lung Cancer. Clin. Cancer Res. 2023, 29, 2310–2323. [Google Scholar] [CrossRef] [PubMed]
- Nie, W.; Wang, Z.-J.; Zhang, K.; Li, B.; Cai, Y.-R.; Wen, F.-C.; Zhang, D.; Bai, Y.-Z.; Zhang, X.-Y.; Wang, S.-Y.; et al. ctDNA-adjusted bTMB as a predictive biomarker for patients with NSCLC treated with PD-(L)1 inhibitors. BMC Med. 2022, 20, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Heitzer, E.; Haque, I.S.; Roberts, C.E.S.; Speicher, M.R. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat. Rev. Genet. 2019, 20, 71–88. [Google Scholar] [CrossRef]
- Batool, S.M.; Yekula, A.; Khanna, P.; Hsia, T.; Gamblin, A.S.; Ekanayake, E.; Escobedo, A.K.; Gil You, D.; Castro, C.M.; Im, H.; et al. The Liquid Biopsy Consortium: Challenges and opportunities for early cancer detection and monitoring. Cell Rep. Med. 2023, 4, 101198. [Google Scholar] [CrossRef]
- Khan, P.; Siddiqui, J.A.; Kshirsagar, P.G.; Venkata, R.C.; Maurya, S.K.; Mirzapoiazova, T.; Perumal, N.; Chaudhary, S.; Kanchan, R.K.; Fatima, M.; et al. MicroRNA-1 attenuates the growth and metastasis of small cell lung cancer through CXCR4/FOXM1/RRM2 axis. Mol. Cancer 2023, 22, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhou, N.; Deng, H.; Chen, X.; Chen, Q.; Wang, Q.; Sun, L.; Wen, Y.; Cao, X.; Luo, Z.; et al. Prognostic value of circulating tumor DNA using target next-generation sequencing in extensive-stage small-cell lung cancer. Lung Cancer 2023, 178, 11–19. [Google Scholar] [CrossRef]
- Sadeghi, M.S.; Lotfi, M.; Soltani, N.; Farmani, E.; Fernandez, J.H.O.; Akhlaghitehrani, S.; Mohammed, S.H.; Yasamineh, S.; Kalajahi, H.G.; Gholizadeh, O. Recent advances on high-efficiency of microRNAs in different types of lung cancer: a comprehensive review. Cancer Cell Int. 2023, 23, 1–30. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, J.; Guo, C.; Wang, M.; Wang, C.; Yan, Y.; Sun, L.; Wang, D.; Zhang, L.; Yu, H.; et al. Proteogenomic characterization of small cell lung cancer identifies biological insights and subtype-specific therapeutic strategies. Cell 2024, 187, 184–203.e28. [Google Scholar] [CrossRef]
- Sen, T.; Dotsu, Y.; Corbett, V.; Puri, S.; Sen, U.; A Boyle, T.; Mack, P.; Hirsch, F.; Aljumaily, R.; Naqash, A.R.; et al. Pulmonary neuroendocrine neoplasms: the molecular landscape, therapeutic challenges, and diagnosis and management strategies. . 2025, 26, e13–e33. [Google Scholar] [CrossRef]
- Zhu, Y.; Cui, Y.; Zheng, X.; Zhao, Y.; Sun, G. Small-cell lung cancer brain metastasis: From molecular mechanisms to diagnosis and treatment. Biochim. et Biophys. Acta (BBA) - Mol. Basis Dis. 2022, 1868, 166557. [Google Scholar] [CrossRef]
- Li, N.; Chu, Y.; Song, Q. Brain Metastasis in Patients with Small Cell Lung Cancer. Int. J. Gen. Med. 2021, ume 14, 10131–10139. [Google Scholar] [CrossRef]
- Slotman, B.; Faivre-Finn, C.; Kramer, G.; Rankin, E.; Snee, M.; Hatton, M.; Postmus, P.; Collette, L.; Musat, E.; Senan, S. Prophylactic Cranial Irradiation in Extensive Small-Cell Lung Cancer. New Engl. J. Med. 2007, 357, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Yamanaka, T.; Seto, T.; Harada, H.; Nokihara, H.; Saka, H.; Nishio, M.; Kaneda, H.; Takayama, K.; Ishimoto, O.; et al. Prophylactic cranial irradiation versus observation in patients with extensive-disease small-cell lung cancer: a multicentre, randomised, open-label, phase 3 trial. 18. [CrossRef]
- Gaebe, K.; Erickson, A.W.; Li, A.Y.; Youssef, A.N.; Sharma, B.; Chan, K.K.; Lok, B.H.; Das, S. Re-examining prophylactic cranial irradiation in small cell lung cancer: a systematic review and meta-analysis. eClinicalMedicine 2024, 67, 102396. [Google Scholar] [CrossRef] [PubMed]
- de Dios, N.R.; Couñago, F.; Murcia-Mejía, M.; Rico-Oses, M.; Calvo-Crespo, P.; Samper, P.; Vallejo, C.; Luna, J.; Trueba, I.; Sotoca, A.; et al. Randomized Phase III Trial of Prophylactic Cranial Irradiation With or Without Hippocampal Avoidance for Small-Cell Lung Cancer (PREMER): A GICOR-GOECP-SEOR Study. J. Clin. Oncol. 2021, 39, 3118–3127. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.-J.; Cho, B.C.; Felip, E.; Korantzis, I.; Ohashi, K.; Majem, M.; Juan-Vidal, O.; Handzhiev, S.; Izumi, H.; Lee, J.-S.; et al. Tarlatamab for Patients with Previously Treated Small-Cell Lung Cancer. New Engl. J. Med. 2023, 389, 2063–2075. [Google Scholar] [CrossRef]
- Dingemans, A.-M.C.; Ahn, M.-J.; Blackhall, F.H.; Reck, M.; Hummel, H.-D.; Ramalingam, S.S.; Johnson, M.L.; Akamatsu, H.; Wolf, J.; Sands, J.; et al. DeLLphi-301: Tarlatamab phase 2 trial in small cell lung cancer (SCLC)—Efficacy and safety analyzed by presence of brain metastasis. J. Clin. Oncol. 2024, 42, 8015–8015. [Google Scholar] [CrossRef]
- Dhillon, S. Tarlatamab: First Approval. Drugs 2024, 84, 995–1003. [Google Scholar] [CrossRef]
- Desai, A.; Smith, C.J.; Ashara, Y.; Orme, J.J.; Zanwar, S.; Potter, A.; Hocum, C.; Moffett, J.N.; Schwecke, A.J.; Manochakian, R.; et al. Real-World Outcomes With Lurbinectedin in Second-Line Setting and Beyond for Extensive Stage Small Cell Lung Cancer. Clin. Lung Cancer 2023, 24, 689–695.e1. [Google Scholar] [CrossRef]
- Dowlati, A.; Chiang, A.C.; Cervantes, A.; Babu, S.; Hamilton, E.; Wong, S.F.; Tazbirkova, A.; Sullivan, I.G.; van Marcke, C.; Italiano, A.; et al. Phase 2 Open-Label Study of Sacituzumab Govitecan as Second-Line Therapy in Patients With Extensive-Stage Small Cell Lung Cancer: Results From TROPiCS-03. . 2025. [CrossRef]
- U.S. FDA grants breakthrough therapy designation to Trodelvy® (sacituzumab govitecan-hziy) for second-line treatment of extensive-stage small cell lung cancer. Gilead Sciences, Inc. , 2024. https://tinyurl.com/3af9n47n. Accessed 1.3.2025. 17 December.
- Gray, J.E.; Heist, R.S.; Starodub, A.N.; Camidge, D.R.; Kio, E.A.; Masters, G.A.; Purcell, W.T.; Guarino, M.J.; Misleh, J.; Schneider, C.J.; et al. Therapy of Small Cell Lung Cancer (SCLC) with a Topoisomerase-I–inhibiting Antibody–Drug Conjugate (ADC) Targeting Trop-2, Sacituzumab Govitecan. Clin. Cancer Res. 2017, 23, 5711–5719. [Google Scholar] [CrossRef]
- Pons-Tostivint, E.; Ezzedine, R.; Goronflot, T.; Crequit, P.; Chatellier, T.; Raimbourg, J.; Bennouna, J.; Leprieur, E.G.; Porte, M. Second-line treatment outcomes after first-line chemotherapy plus immunotherapy in Extensive-Stage small cell lung cancer (ES-SCLC) patients: A large French multicenter study. Lung Cancer 2024, 194. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.-Q.; Lin, X.; Li, Y.; Lu, S. Irinotecan as a Second-line Chemotherapy for Small Cell Lung Cancer: a Systemic Analysis. Asian Pac. J. Cancer Prev. 2015, 16, 1993–1995. [Google Scholar] [CrossRef] [PubMed]
- Karim, N.A.; Bhatt, A.; Chiec, L.; Curry, R. Systemic Chemotherapy for Progression of Brain Metastases in Extensive-Stage Small Cell Lung Cancer. Case Rep. Oncol. Med. 2015, 2015, 1–3. [Google Scholar] [CrossRef]
- Masuda, N.; Fukuoka, M.; Kusunoki, Y.; Matsui, K.; Takifuji, N.; Kudoh, S.; Negoro, S.; Nishioka, M.; Nakagawa, K.; Takada, M. CPT-11: a new derivative of camptothecin for the treatment of refractory or relapsed small-cell lung cancer. J. Clin. Oncol. 1992, 10, 1225–1229. [Google Scholar] [CrossRef]
- Sevinc, A.; Kalender, M.E.; Altinbas, M.; Ozkan, M.; Dikilitas, M.; Camci, C.; (Asmo), A.S.O.M.O. Irinotecan as a second-line monotherapy for small cell lung cancer. . 2011, 12, 1055–9. [Google Scholar]
- Denham CA, et al. A phase II study of irinotecan inpatients with previously treated small-cell lung cancer (SCLC), 4: Am Soc Clin Oncol. 1998. 17(abstract 1736), 1998.
- Le Chevalier T, I.N. , Chomy P, et al. A phase II study of irinotecan (CPT-11) in patients with small cell lung cancer (SCLC) progressing after initial response to first line chemotherapy (CT), 4: Am Soc Clin Oncol. 1997. 16(abstract 1), 1997. [Google Scholar]
- O'Brien, M.E.; Ciuleanu, T.-E.; Tsekov, H.; Shparyk, Y.; Čučeviá, B.; Juhasz, G.; Thatcher, N.; Ross, G.A.; Dane, G.C.; Crofts, T. Phase III Trial Comparing Supportive Care Alone With Supportive Care With Oral Topotecan in Patients With Relapsed Small-Cell Lung Cancer. J. Clin. Oncol. 2006, 24, 5441–5447. [Google Scholar] [CrossRef]
- von Pawel, J.; Schiller, J.H.; Shepherd, F.A.; Fields, S.Z.; Kleisbauer, J.; Chrysson, N.G.; Stewart, D.J.; Clark, P.I.; Palmer, M.C.; Depierre, A.; et al. Topotecan Versus Cyclophosphamide, Doxorubicin, and Vincristine for the Treatment of Recurrent Small-Cell Lung Cancer. J. Clin. Oncol. 1999, 17, 658–658. [Google Scholar] [CrossRef]
- Zheng, J.; Deng, Y.; Huang, B.; Chen, X. Efficacy and safety of immune checkpoint inhibitors combined with chemotherapy as first-line treatment for extensive-stage small cell lung cancer: a meta-analysis based on mixed-effect models. Front. Med. 2023, 10, 1198950. [Google Scholar] [CrossRef]
- Perez, J.M.T.; Leary, A.; Besse, B.; Castellano, D.E.; Aix, S.P.; Arrondeau, J.; Moreno, V.; Doger, B.; Lopez, R.; Awada, A.; et al. Efficacy and safety of lurbinectedin (PM1183) in small cell lung cancer (SCLC): Results from a phase 2 study. J. Clin. Oncol. 2018, 36, 8570–8570. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, Q.; Li, K.; Shi, J.; Wu, L.; Han, B.; Chen, G.; He, J.; Wang, J.; Qin, H.; et al. OA13.03 Anlotinib as Third-Line or Further-Line Treatment in Relapsed SCLC: A Multicentre, Randomized, Double-Blind Phase 2 Trial. J. Thorac. Oncol. 2018, 13, S351–S352. [Google Scholar] [CrossRef]
- Pietanza, M.C.; Waqar, S.N.; Krug, L.M.; Dowlati, A.; Hann, C.L.; Chiappori, A.; Owonikoko, T.K.; Woo, K.M.; Cardnell, R.J.; Fujimoto, J.; et al. Randomized, Double-Blind, Phase II Study of Temozolomide in Combination With Either Veliparib or Placebo in Patients With Relapsed-Sensitive or Refractory Small-Cell Lung Cancer. J. Clin. Oncol. 2018, 36, 2386–2394. [Google Scholar] [CrossRef] [PubMed]
- Aliaga, P.T.; Del Signore, E.; Fuorivia, V.; Spitaleri, G.; Asnaghi, R.; Attili, I.; Corvaja, C.; Schianca, A.C.; Passaro, A.; de Marinis, F. The Evolving Scenario of ES-SCLC Management: From Biology to New Cancer Therapeutics. Genes 2024, 15, 701. [Google Scholar] [CrossRef] [PubMed]
- Lallo, A.; Frese, K.K.; Morrow, C.J.; Sloane, R.; Gulati, S.; Schenk, M.W.; Trapani, F.; Simms, N.; Galvin, M.; Brown, S.; et al. The Combination of the PARP Inhibitor Olaparib and the WEE1 Inhibitor AZD1775 as a New Therapeutic Option for Small Cell Lung Cancer. Clin. Cancer Res. 2018, 24, 5153–5164. [Google Scholar] [CrossRef] [PubMed]
- Nemala, A.; Ullah, A.; Yacur, M.; Karim, N.A. Do Current Lung Cancer Clinical Trials Represent All Patient Populations Including Minorities? Clin. Lung Cancer 2023, 24, 573–580. [Google Scholar] [CrossRef]
- Grenda, A.; Krawczyk, P.; Obara, A.; Gajek. ; Łomża-Łaba, A.; Milanowski, J. Transitioning to a Personalized Approach in Molecularly Subtyped Small-Cell Lung Cancer (SCLC). Int. J. Mol. Sci. 2024, 25, 4208. [Google Scholar] [CrossRef]
- Clinicaltrials.gov website last accessed 1/3/2025.
- Trigo, J.; Subbiah, V.; Besse, B.; Moreno, V.; López, R.; Sala, M.A.; Peters, S.; Ponce, S.; Fernández, C.; Alfaro, V.; et al. Lurbinectedin as second-line treatment for patients with small-cell lung cancer: a single-arm, open-label, phase 2 basket trial. Lancet Oncol. 2020, 21, 645–654. [Google Scholar] [CrossRef]
- Rossi, A.; Sacco, P.C.; Sgambato, A.; Casaluce, F.; Santabarbara, G.; Palazzolo, G.; Maione, P.; Gridelli, C. Optimal drugs for second-line treatment of patients with small-cell lung cancer. Expert Opin. Pharmacother. 2016, 17, 969–976. [Google Scholar] [CrossRef]
- Torasawa, M.; Horinouchi, H.; Nomura, S.; Igawa, S.; Asai, M.; Ishii, H.; Wakui, H.; Ushio, R.; Asao, T.; Namba, Y.; et al. Reconsidering the Cutoff Value for Sensitive and Refractory Relapses in Extensive-Stage SCLC in the Era of Immunotherapy. J. Thorac. Oncol. 2023, 19, 325–336. [Google Scholar] [CrossRef]
- Calles, A.; Navarro, A.; Doger, B.; de Miguel, M.J.; Alvarez, R.; Moreno, V.; Ramón, J.; del Mar Galera López, M.; Hernandez-Guerrero, T.; Corral de la Fuente, E.; et al. A phase 1/2 trial of lurbinectedin (L) in combination with pembrolizumab (P) in relapsed small cell lung cancer (SCLC): The LUPER study. Journal of Clinical Oncology. 40(16_suppl): p. 8581-8581.
- Desai, A.; Abdayem, P.; Adjei, A.A.; Planchard, D. Antibody-drug conjugates: A promising novel therapeutic approach in lung cancer. Lung Cancer 2021, 163, 96–106. [Google Scholar] [CrossRef]
- Cani, M.; Napoli, V.M.; Garbo, E.; Ferrari, G.; Del Rio, B.; Novello, S.; Passiglia, F. Targeted Therapies in Small Cell Lung Cancer: From Old Failures to Novel Therapeutic Strategies. Int. J. Mol. Sci. 2023, 24, 8883. [Google Scholar] [CrossRef]
- Deneka, A.Y.; Boumber, Y.; Beck, T.; Golemis, E.A. Tumor-Targeted Drug Conjugates as an Emerging Novel Therapeutic Approach in Small Cell Lung Cancer (SCLC). Cancers 2019, 11, 1297. [Google Scholar] [CrossRef] [PubMed]
- Erickson, H.K.; Phillips, G.D.L.; Leipold, D.D.; Provenzano, C.A.; Mai, E.; Johnson, H.A.; Gunter, B.; Audette, C.A.; Gupta, M.; Pinkas, J.; et al. The Effect of Different Linkers on Target Cell Catabolism and Pharmacokinetics/Pharmacodynamics of Trastuzumab Maytansinoid Conjugates. Mol. Cancer Ther. 2012, 11, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Drago, J.Z.; Modi, S.; Chandarlapaty, S. Unlocking the potential of antibody–drug conjugates for cancer therapy. Nat. Rev. Clin. Oncol. 2021, 18, 327–344. [Google Scholar] [CrossRef] [PubMed]
- Owen, D.H.; Giffin, M.J.; Bailis, J.M.; Smit, M.-A.D.; Carbone, D.P.; He, K. DLL3: an emerging target in small cell lung cancer. J. Hematol. Oncol. 2019, 12, 1–8. [Google Scholar] [CrossRef]
- Starodub, A.; Camidge, D.R.; Scheff, R.J.; Thomas, S.S.; Guarino, M.J.; Masters, G.A.; Kalinsky, K.; Gandhi, L.; Bardia, A.; Messersmith, W.A.; et al. Trop-2 as a therapeutic target for the antibody-drug conjugate (ADC), sacituzumab govitecan (IMMU-132), in patients (pts) with previously treated metastatic small-cell lung cancer (mSCLC). J. Clin. Oncol. 2016, 34, 8559–8559. [Google Scholar] [CrossRef]
- Seute, T.; Leffers, P.; Velde, G.P.T.; Twijnstra, A. Detection of brain metastases from small cell lung cancer. Cancer 2008, 112, 1827–1834. [Google Scholar] [CrossRef]
- Hirsch, F.R.; Paulson, O.B.; Hansen, H.H.; Larsen, S.O. Intracranial metastases in small cell carcinoma of the lung prognostic aspects. Cancer 1983, 51, 529–533. [Google Scholar] [CrossRef]
- Kristjansen, P.E.; Kristensen, C.A. The role of prophylactic cranial irradiation in the management of small cell lung cancer. Cancer Treat. Rev. 1993, 19, 3–16. [Google Scholar] [CrossRef]
- Rusthoven, C.G.; Yamamoto, M.; Bernhardt, D.; Smith, D.E.; Gao, D.; Serizawa, T.; Yomo, S.; Aiyama, H.; Higuchi, Y.; Shuto, T.; et al. Evaluation of First-line Radiosurgery vs Whole-Brain Radiotherapy for Small Cell Lung Cancer Brain Metastases. JAMA Oncol. 2020, 6, 1028–1037. [Google Scholar] [CrossRef]
- Pietanza, M.C.; Kadota, K.; Huberman, K.; Sima, C.S.; Fiore, J.J.; Sumner, D.K.; Travis, W.D.; Heguy, A.; Ginsberg, M.S.; Holodny, A.I.; et al. Phase II Trial of Temozolomide in Patients with Relapsed Sensitive or Refractory Small Cell Lung Cancer, with Assessment of Methylguanine-DNA Methyltransferase as a Potential Biomarker. Clin. Cancer Res. 2012, 18, 1138–1145. [Google Scholar] [CrossRef]
- A Phase 3, Randomized, Double-blind, Placebo-controlled, Multicenter Study of Tarlatamab Therapy in Subjects With Limited-Stage Small-Cell Lung Cancer (LS-SCLC) Who Have Not Progressed Following Concurrent Chemoradiation Therapy. 2023.
- Rahnea-Nita, R.-A.; Stoian, A.-R.; Anghel, R.-M.; Rebegea, L.-F.; Ciuhu, A.-N.; Bacinschi, X.-E.; Zgura, A.-F.; Trifanescu, O.-G.; Toma, R.-V.; Constantin, G.B.; et al. The Efficacy of Immunotherapy in Long-Term Survival in Non-Small Cell Lung Cancer (NSCLC) Associated with the Syndrome of Inappropriate Antidiuretic Hormone Secretion (SIADH). Life 2023, 13, 1279. [Google Scholar] [CrossRef] [PubMed]
- Bahij, R.; Jeppesen, S.S.; Olsen, K.E.; Halekoh, U.; Holmskov, K.; Hansen, O. Outcome of treatment in patients with small cell lung cancer in poor performance status. Acta Oncol. 2019, 58, 1612–1617. [Google Scholar] [CrossRef] [PubMed]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.R. Depression in cancer patients: Pathogenesis, implications and treatment (Review). Oncol. Lett. 2015, 9, 1509–1514. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network website. Available online: https://www.nccn.org/professionals/physician_gls/pdf/older_adult.pdf. (accessed on December 30th, 2024).
- Corso, C.D.; Rutter, C.E.; Park, H.S.; Lester-Coll, N.H.; Kim, A.W.; Wilson, L.D.; Husain, Z.A.; Lilenbaum, R.C.; Yu, J.B.; Decker, R.H. Role of Chemoradiotherapy in Elderly Patients With Limited-Stage Small-Cell Lung Cancer. J. Clin. Oncol. 2015, 33, 4240–4246. [Google Scholar] [CrossRef]
- Pallis, A.G.; Shepherd, F.A.; Lacombe, D.; Gridelli, C. Treatment of small-cell lung cancer in elderly patients. Cancer 2010, 116, 1192–1200. [Google Scholar] [CrossRef]
- Gridelli, C.; Casaluce, F.; Sgambato, A.; Monaco, F.; Guida, C. Treatment of limited-stage small cell lung cancer in the elderly, chemotherapy vs. sequential chemoradiotherapy vs. concurrent chemoradiotherapy: that’s the question. Transl. Lung Cancer Res. 2016, 5, 150–154. [Google Scholar] [CrossRef]
- Zhang, B.; Lewis, W.; Stewart, C.A.; Morris, B.B.; Solis, L.M.; Serrano, A.; Xi, Y.; Wang, Q.; Lopez, E.R.; Concannon, K.; et al. Brief Report: Comprehensive Clinicogenomic Profiling of Small Cell Transformation From EGFR-Mutant NSCLC Informs Potential Therapeutic Targets. JTO Clin. Res. Rep. 2023, 5, 100623. [Google Scholar] [CrossRef]
- Mambetsariev, I.; Arvanitis, L.; Fricke, J.; Pharaon, R.; Baroz, A.R.; Afkhami, M.; Koczywas, M.; Massarelli, E.; Salgia, R. Small Cell Lung Cancer Transformation following Treatment in EGFR-Mutated Non-Small Cell Lung Cancer. J. Clin. Med. 2022, 11, 1429. [Google Scholar] [CrossRef]
- Soomro, Z.; Youssef, M.; Yust-Katz, S.; Jalali, A.; Patel, A.J.; Mandel, J. Paraneoplastic syndromes in small cell lung cancer. J. Thorac. Dis. 2020, 12, 6253–6263. [Google Scholar] [CrossRef]
- Sato, Y.; Fujiwara, S.; Hata, A.; Kida, Y.; Masuda, T.; Amimoto, H.; Matsumoto, H.; Miyoshi, K.; Otsuka, K.; Tomii, K. Clinical Impact of Pre-Existing Autoantibodies in Patients With SCLC Treated With Immune Checkpoint Inhibitor: A Multicenter Prospective Observational Study. JTO Clin. Res. Rep. 2023, 4, 100608. [Google Scholar] [CrossRef] [PubMed]
- Gozzard, P.; Woodhall, M.; Chapman, C.; Nibber, A.; Waters, P.; Vincent, A.; Lang, B.; Maddison, P. Paraneoplastic neurologic disorders in small cell lung carcinoma. Neurology 2015, 85, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Keogh, A.; Finn, S.; Radonic, T. Emerging Biomarkers and the Changing Landscape of Small Cell Lung Cancer. Cancers 2022, 14, 3772. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Kuang, P.; Wang, L.; Li, W.; Chen, B.; Liu, Y.; Wang, H.; Zhao, S.; Ye, L.; Yu, F.; et al. Mechanisms of drugs-resistance in small cell lung cancer: DNA-related, RNA-related, apoptosis-related, drug accumulation and metabolism procedure. Transl. Lung Cancer Res. 2020, 9, 768–786. [Google Scholar] [CrossRef]
- Oronsky, B.; Reid, T.R.; Oronsky, A.; Carter, C.A. What's New in SCLC? A Review. Neoplasia 2017, 19, 842–847. [Google Scholar] [CrossRef]
- Li, H.; Zhao, P.; Tian, L.; Lu, Y.; Wang, X.; Shao, W.; Cheng, Y. Advances in biomarkers for immunotherapy in small-cell lung cancer. Front. Immunol. 2024, 15. [Google Scholar] [CrossRef]
- Rudin, C.; Ahn, M.-J.; Johnson, M.; Hann, C.; Girard, N.; Nishio, M.; Cheng, Y.; Hayashi, H.; Kim, Y.J.; Navarro, A.; et al. OA04.03 Ifinatamab Deruxtecan (I-DXd) in Extensive-Stage Small Cell Lung Cancer (ES-SCLC): Interim Analysis of Ideate-lung01. J. Thorac. Oncol. 2024, 19, S15–S16. [Google Scholar] [CrossRef]
- Subbiah, S.; Nam, A.; Garg, N.; Behal, A.; Kulkarni, P.; Salgia, R. Small Cell Lung Cancer from Traditional to Innovative Therapeutics: Building a Comprehensive Network to Optimize Clinical and Translational Research. J. Clin. Med. 2020, 9, 2433. [Google Scholar] [CrossRef]
- Yi, L.; Zhou, Z.; Zeng, X.; Tan, C.; Liu, Q. First-line treatments for extensive-stage small-cell lung cancer with immune checkpoint inhibitors plus chemotherapy: a China-based cost-effectiveness analysis. Front. Immunol. 2024, 15, 1408928. [Google Scholar] [CrossRef]
| Clinical trial | Investigational agent | Country |
|---|---|---|
| NCT05353257 | Serplulimab + carboplatin/cisplatin-etoposide + radiotherapy | USA, Europe and China |
| NCT04155034 | MRI versus PCI (Prophylactic Cranial Irradiation) and MRI | USA |
| NCT04829708 | MRI verses PCI | China |
| NCT05623267 | Sugemalimab | China |
| NCT06117774 | Tarlatamab | USA, south America, Australia, Europe and Asia |
| NCT06095583 | Tifcemalimab and toripalimab | USA, Europe and Asia |
| NCT06430866 | ABP 234 and Pembrolizumab | South America, Europe and Asia |
| NCT02201992 | Crizotinib | USA |
| Clinical trial | Investigational agent | Country |
|---|---|---|
| NCT04402788 | Atezolizumab | USA |
| NCT04155034 | PCI, MRI verses MRI | USA and South Korea |
| NCT05468489 | Serplulimab + chemotherapy (carboplatin-etoposide) | USA |
| NCT06211036 | Tarlatamab and Durvalumab | USA, Europe, Australia, South America |
| NCT05223647 | Chemo-immunotherapy plus thoracic radiotherapy | Sweden, Finland, Norway |
| NCT05844150 | PM8002+ Etoposide+ platinum | China |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




