3.1. General Introduction to the Hodgkin-Huxley Model
The electrical behavior of the excitable membrane is described using the HH model [
12], which provides a robust quantitative foundation for understanding the membrane’s dynamic properties. This section details the mathematical representation of the system, emphasizing its nonlinear differential equations, the membrane's ionic dynamics, and their role in generating a periodic driving force for the electromechanical components.
The power contributions in the Hodgkin-Huxley (HH) model can be categorized into distinct components that reflect the energy transformations occurring within the neuronal membrane. These components account for external stimulation, capacitive storage, ionic dissipation, and the influence of electromotive forces. Below is a detailed description of each term.
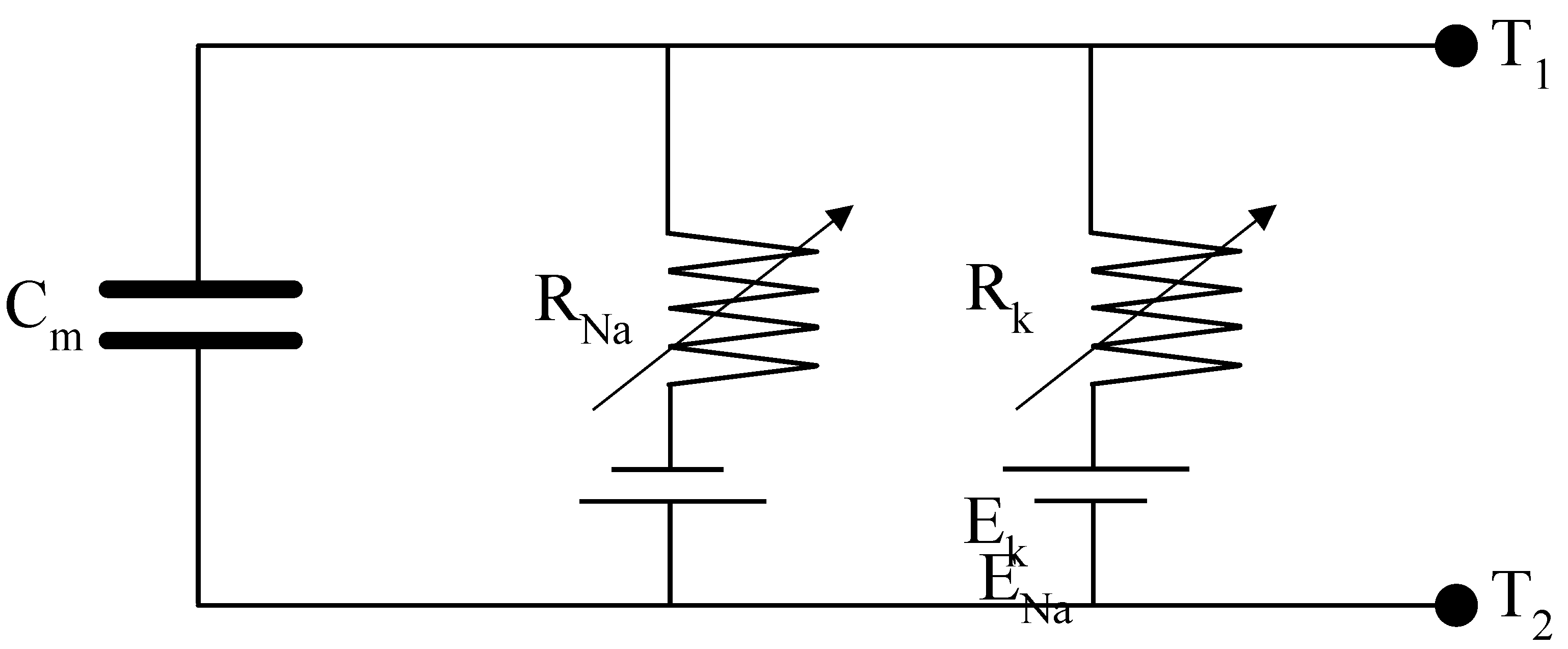
The excitable membrane, represented in
Figure 2, is modeled as a capacitor (
) with a dielectric formed by the lipid bilayer. This bilayer is combined with selective ionic channels that regulate ion flow based on voltage differentials. For this study, the focus is on sodium (
) and potassium (
) ions, which play essential roles in generating and propagating action potentials; in this model the leakage channel is not considered.
The sodium and potassium channels are characterized as variable resistors ( and ), each in series with constant voltage generators to reflect their voltage-dependent behavior. Hodgkin and Huxley demonstrated that the conductances of these channels ( and ) dynamically depend on the transmembrane potential (). During an action potential, the conductances increase significantly, reducing the channel resistances and facilitating ion flow. This decrease in resistance highlights the membrane's changing permeability in response to the electric field and provides clear evidence that the action potential is driven by variations in ionic fluxes across the membrane. The resistances are expressed as and , where the conductances depend on both time and membrane potential.
Unlike the ionic channels, which can be modeled as resistors and respond to a step current input with a corresponding step change in voltage, the neuronal membrane exhibits a continuous voltage response under similar conditions. This distinction arises because the membrane behaves as a capacitor, where altering the potential across it requires modifying the charge stored on its plates. In the neuronal membrane, this process is analogous to redistributing charge across its interfaces through ionic fluxes, which drive depolarization.
The relationship between the voltage across a capacitor and the charge stored on its plates is given by: where represents the charge, and is the capacitance, serving as the proportionality constant between and . The change in charge, and consequently the voltage across the capacitor, is induced by current flow. Current is defined as the rate of charge transfer over time: Substituting this into the capacitor equation yields: From this relationship, it becomes clear that the voltage change across the capacitor in response to a current pulse is directly proportional to the duration of the current. This continuous variation of membrane potential highlights the capacitor-like behavior of the neuronal membrane in contrast to the step responses of purely resistive ionic channels.
In series with these resistors are ionic batteries ( and ), which represent the electromotive forces arising from ionic concentration gradients.
In the HH model, ionic currents are modeled as flow through variable resistances in parallel with a capacitor that represents the membrane. For each ion considered (e.g., sodium and potassium), the current depends on the difference between the membrane potential and the ion's equilibrium potential, which is determined by the Nernst equation. The total current flowing through the membrane () is thus the sum of the capacitive current and the ionic currents.
The capacitive current () is related to the rate of change of the membrane potential () over time. Since the membrane is represented as a capacitor, the capacitive current is given by: where is the membrane capacitance per unit area.
The ionic currents () are driven by the conductances of the sodium and potassium channels ( and ), which depend on both time and the membrane potential, reflecting the voltage-dependent nature of the channels. For sodium and potassium ions, the currents are expressed as: where and are the equilibrium potentials, respectively. These potentials represent the voltages at which there is no net ionic flow for the corresponding ion species.
The total membrane current is given by:
which, for the case of two ion species, can be expanded as: equation:
This formulation captures the dynamic interplay between the capacitive and ionic components of the current, as well as their dependence on the time-varying membrane potential. It provides the foundation for analyzing how the membrane potential evolves during processes like the action potential, where rapid changes in ion channel conductances lead to characteristic voltage fluctuations.
Hodgkin and Huxley developed equations to describe the time-dependent conductances of ion channels, ensuring they remained sufficiently simple for computing action potentials and refractory periods. A significant challenge in modeling these conductances lay in their distinct behaviors during depolarization and repolarization. Specifically, sodium () and potassium () conductances increase with a delay during depolarization and decrease rapidly during repolarization.
Using experimental data obtained via the patch-clamp technique, Hodgkin and Huxley demonstrated that sodium conductance is proportional to the third power of an activation variable, , governed by a first-order differential equation, with an additional term accounting for the progressive inactivation of sodium channels, . Similarly, potassium conductance is proportional to the fourth power of its activation variable, , also governed by a first-order differential equation. These relationships are expressed as:
,
here,
and
represent the maximum attainable values of sodium and potassium conductances, respectively, while
,
, and
are dimensionless gating variables that range between 0 and 1, that indicate the likelihood of the corresponding ion channel being open, allowing ions to move between the intracellular and extracellular fluids. Their values depend on both membrane voltage and time, reflecting the dynamic nature of channel gating. Their dynamics are governed by the following first-order differential equations:
The rate constants
and
are voltage-dependent and describe the transition rates of ion channels between open and closed states. Let
denote a general gating variable, such as
,
, or
. The dynamics of
can be described by:
where is the equilibrium fraction of open channels, and is the relaxation time. These equations highlight the following key points:
Individual ion channel proteins transition stochastically between open and closed states.
The fraction of open channels, , relaxes exponentially toward , the equilibrium value.
The relaxation rate to equilibrium is determined by the time constant .
The voltage-dependence of
and
, as determined by Hodgkin and Huxley's experiments, leads to exponential dependencies of reaction rates on membrane voltage (see
Table 1).
Consequently, the equilibrium fraction and time constant exhibit sigmoidal dependencies on voltage, reflecting the nonlinear reaction kinetics of ion channel gating. This interplay between voltage and gating dynamics forms the foundation for understanding the behavior of excitable membranes and their role in action potential generation.
Indicating with the index
for
(sodium) or
(potassium), the Nernst potentials (
) and maximum conductances (
) are provided in the
Table 2. The term
accounts for different resting potentials commonly adopted in various studies, which are typically either
or
. In this work,
is taken to be
.
At equilibrium, the cell maintains a resting potential, primarily governed by the efflux of ions through weakly active potassium channels. Sodium channels are largely inactive at this state. Disturbances in the membrane potential, if sufficient in magnitude, initiate the activation of sodium channels, leading to a sequence of ionic exchanges that constitutes the action potential.
The dynamics of the membrane are captured by the following system of nonlinear differential equations, derived from the HH model:
Here:
: Membrane capacitance [].
: Transmembrane potential [.
: Maximum conductances of sodium and potassium channels [].
, : Nernst potentials [] for sodium and potassium, respectively.
, represent the activation and inactivation variables for sodium channels, respectively, while n represents the activation variable for potassium channels.
, , : Steady-state activation and inactivation functions (voltage-dependent).
, , : Voltage-dependent time constants.
: External current per unit area applied across the membrane [].
The first equation in system (1) represents the balance of currents across the cell membrane, as illustrated in
Figure 2. This equation accounts for the combined contributions of ionic currents and external current inputs. The remaining three equations describe the dynamics of the ionic channels, specifically the constitutive relationships governing the variable resistances depicted in the diagram in
Figure 2.
3.2. Power Balance in the Hodgkin-Huxley Model
The power contributions in the HH model can be categorized into distinct components that reflect the energy transformations occurring within the neuronal membrane. These components account for external stimulation, capacitive storage, ionic dissipation, and the influence of electromotive forces. Below is a detailed description of each term.
The external power is provided by the applied current and is responsible for driving the system. The instantaneous power supplied by
is given by:
The external power can drive changes in the capacitive charge, sustain ionic currents, or both. In
Figure 5,
is represented in the third subplot from the top.
The power associated with the membrane's capacitive charging or discharging is:
This term reflects the rate at which electrical energy is stored or released from the capacitive component of the membrane. In
Figure 5,
is shown in the second subplot from the top and indicated with
The power dissipated by the ionic currents is given by:
where
and
are the power dissipation rates for sodium and potassium channels, respectively. For each ionic channel:
These terms represent the resistive dissipation of energy due to ion flow through voltage-dependent channels. In
Figure 4,
and
are displayed in the first subplot.
The HH model includes voltage sources representing the Nernst potentials (
and
) for sodium and potassium ions, respectively. These potentials contribute additional power, defined as:
where
and
are the ionic currents through the sodium and potassium channels:
These terms quantify the work done by the ion-specific electromotive forces in driving ionic currents. Unlike
, these contributions do not result in heat dissipation but are essential for maintaining ionic gradients and facilitating proper neural function. In
Figure 5,
and
are also represented in the first subplot.
The power balance of the HH model is described by the following relationship:
indicating that the power supplied externally is distributed among three components: energy storage in the membrane capacitance, dissipative losses through ionic channels and energy exchange facilitated by the Nernst potentials.
3.3. Numerical Simulations of the Hodgkin-Huxley Model
Numerical simulations are performed with the values listed in
Table 1 and
Table 2. Initial condition of Eq. 1 are
and
The external current is used as regulation parameter.
The way the HH model processes external current to generate action potentials is fundamental, as this conversion of synaptic input into action potentials forms the cornerstone of neuronal communication and brain functionality. When a low-level external current is applied, ionic fluctuations occur until the system stabilizes at a new steady state characterized by slightly elevated membrane potentials. With moderate external current input, the membrane potential may briefly exceed the action potential threshold, producing a single spike before returning to a steady state. Biologically, this reflects ion redistribution in response to changes in electrostatic pressure.
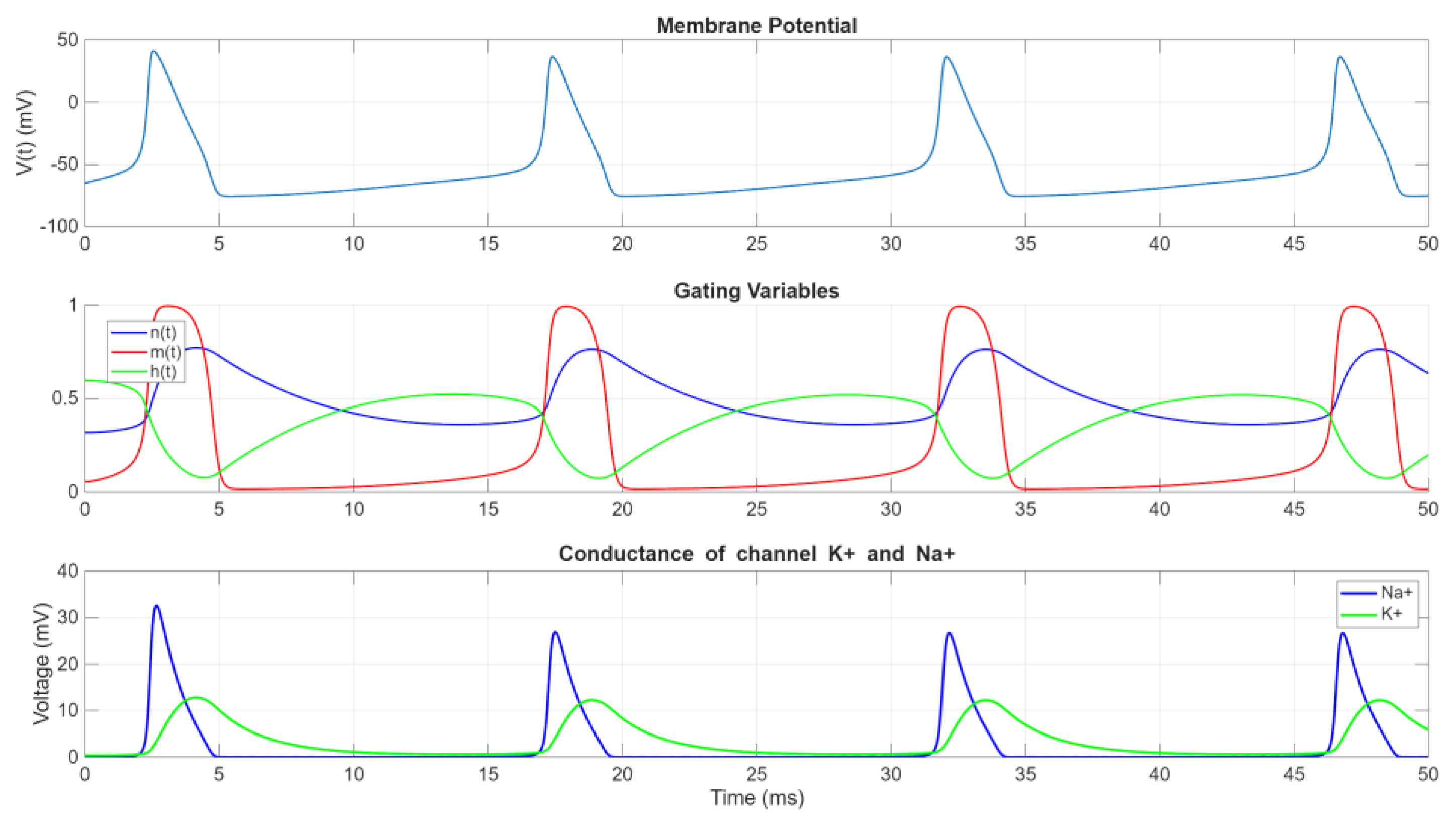
Figure 3 illustrates the neuronal response to
, a steady stream of action potentials is generated. The HH model not only attributes action potential generation to ionic current changes but also faithfully captures the timing and voltage characteristics of these events, ensuring biologically accurate simulations.
Figure 3 further demonstrates how ionic conductance changes correspond to the various phases of an action potential. At the onset of an action potential, the conductance of Na
+ (
) rapidly increases due to the activation of
-gates, enabling a substantial influx of Na
+ ions. This inward current amplifies depolarization through a positive feedback loop, driving the rapid upstroke of the action potential. At the peak, however,
begins to decrease due to the inactivation of Na
+ channels via the
-gates, thereby limiting further Na
+ entry. Sodium ions subsequently leave the cell, driven by electrochemical gradients, as the channels close.
In contrast, potassium conductance () increases more gradually as -gates slowly activate. This delayed efflux of K+ ions counteracts the depolarizing effects of Na+ influx and ultimately dominates during the repolarization phase. The delayed activation of -gates ensures a robust repolarization process, restoring the membrane potential toward its resting state. The persistence of elevated after repolarization results in an after-hyperpolarization, a transient state where the membrane potential becomes more negative than the resting level. This state is critical for resetting the neuronal membrane and ensuring unidirectional action potential propagation.
The interplay between Na+ channel inactivation (-gates closing) and K+ channel activation (-gates opening) creates a refractory period during which further action potential initiation is inhibited. This separation of successive spikes prevents excessive neuronal firing and ensures orderly signal transmission.
The voltage- and time-dependent behavior of the gating variables , , and allows the HH model to accurately capture the biophysical mechanisms underlying action potentials. The distinct time constants and voltage sensitivities of the gating variables contribute to the characteristic waveform of action potentials and their critical role in neural signaling.
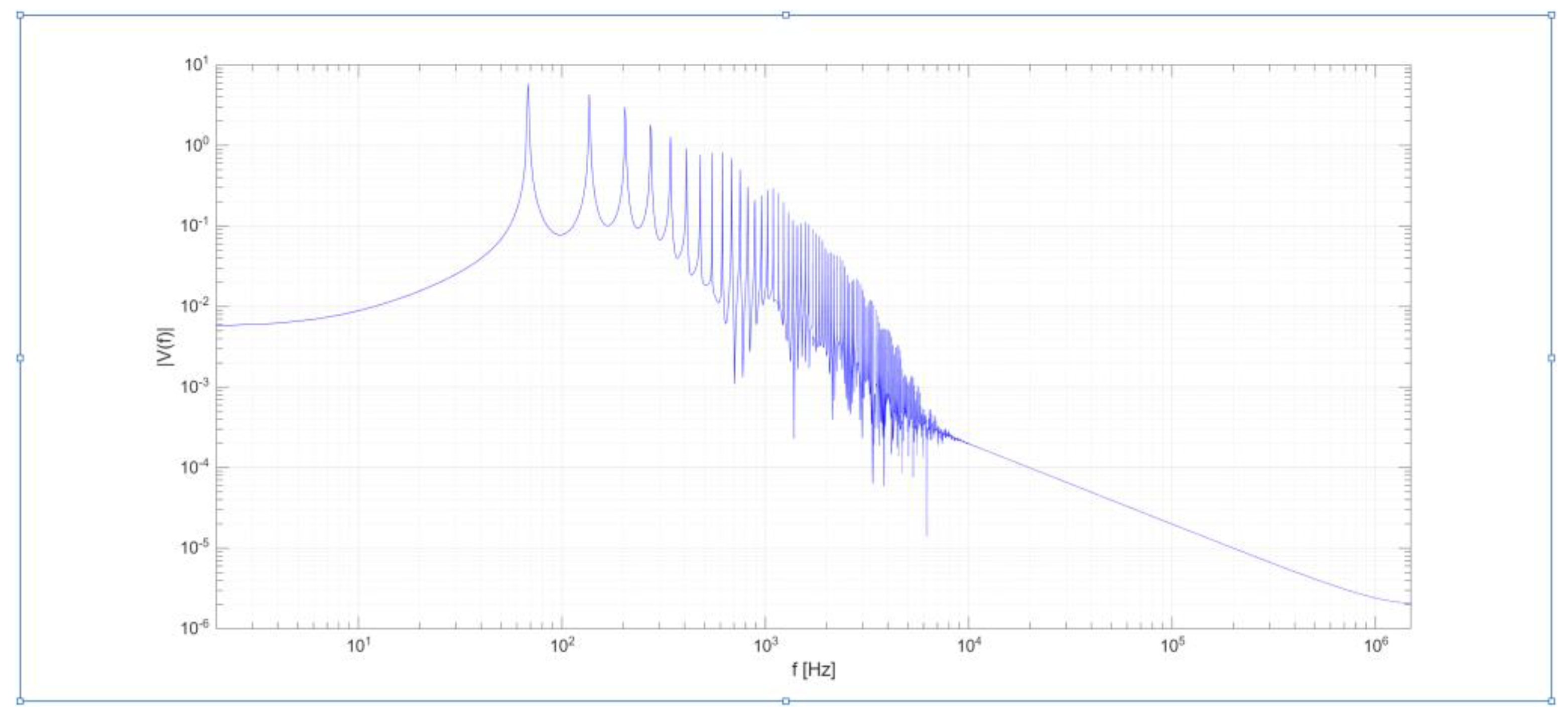
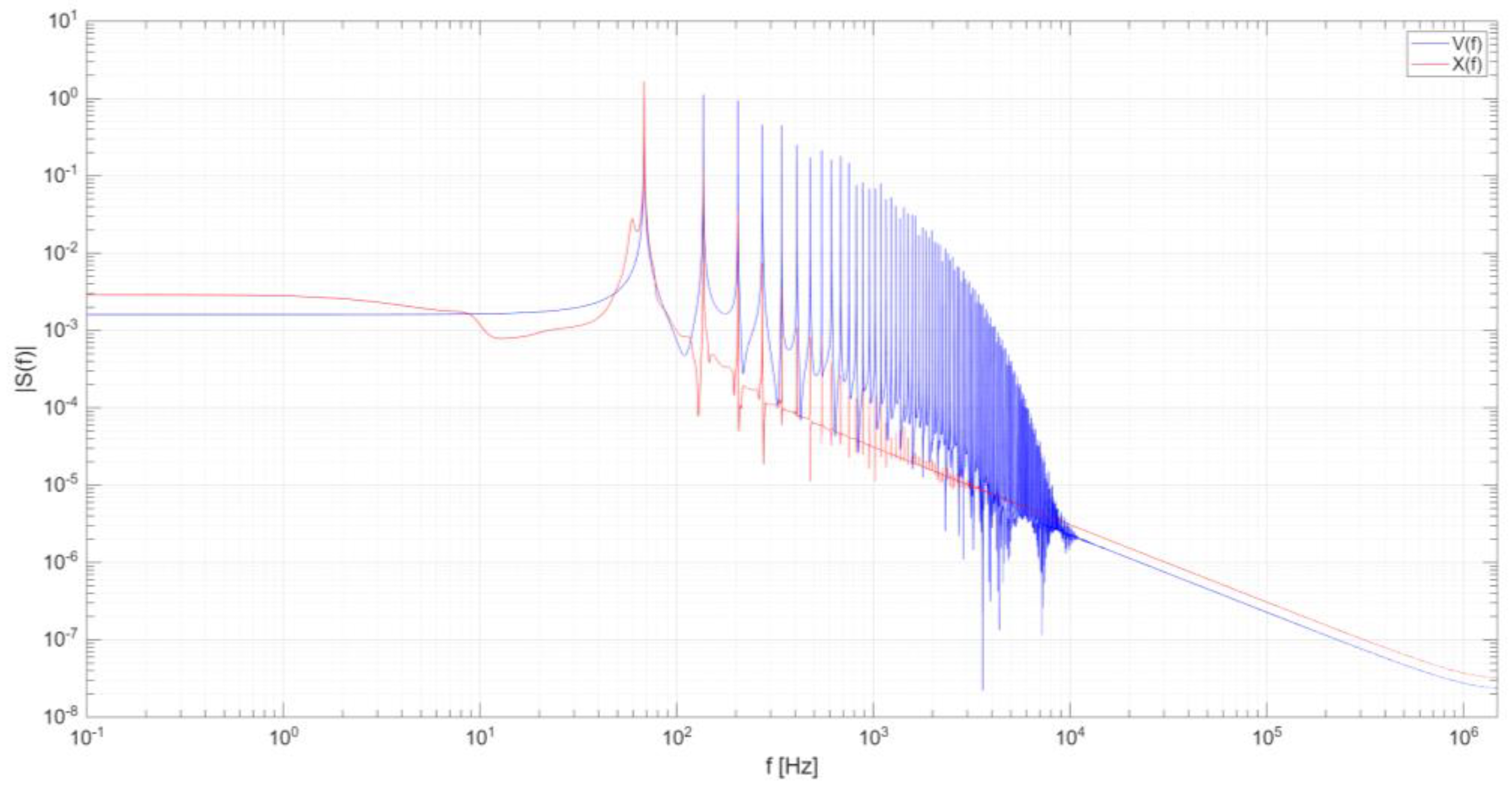
Figure 4 illustrates the Fourier transformation of the signal, highlighting a dominant frequency slightly below 70 Hz. This result is consistent with the temporal periodicity observed in
Figure 3, where the signal exhibits a period of approximately 0.015 seconds.
With the aid of
Figure 5 and
Figure 6 it is possible to understand the power fluxes within the membrane. Specifically, to facilitate the visualization of power exchanges, the first subplot of
Figure 6 displays the combined power contributions from the individual ionic channels (
and
) along with the capacitive power (
). These components are related to the external power (
), represented in the second subplot.
Figure 4.
FFT of the signal .
Figure 4.
FFT of the signal .
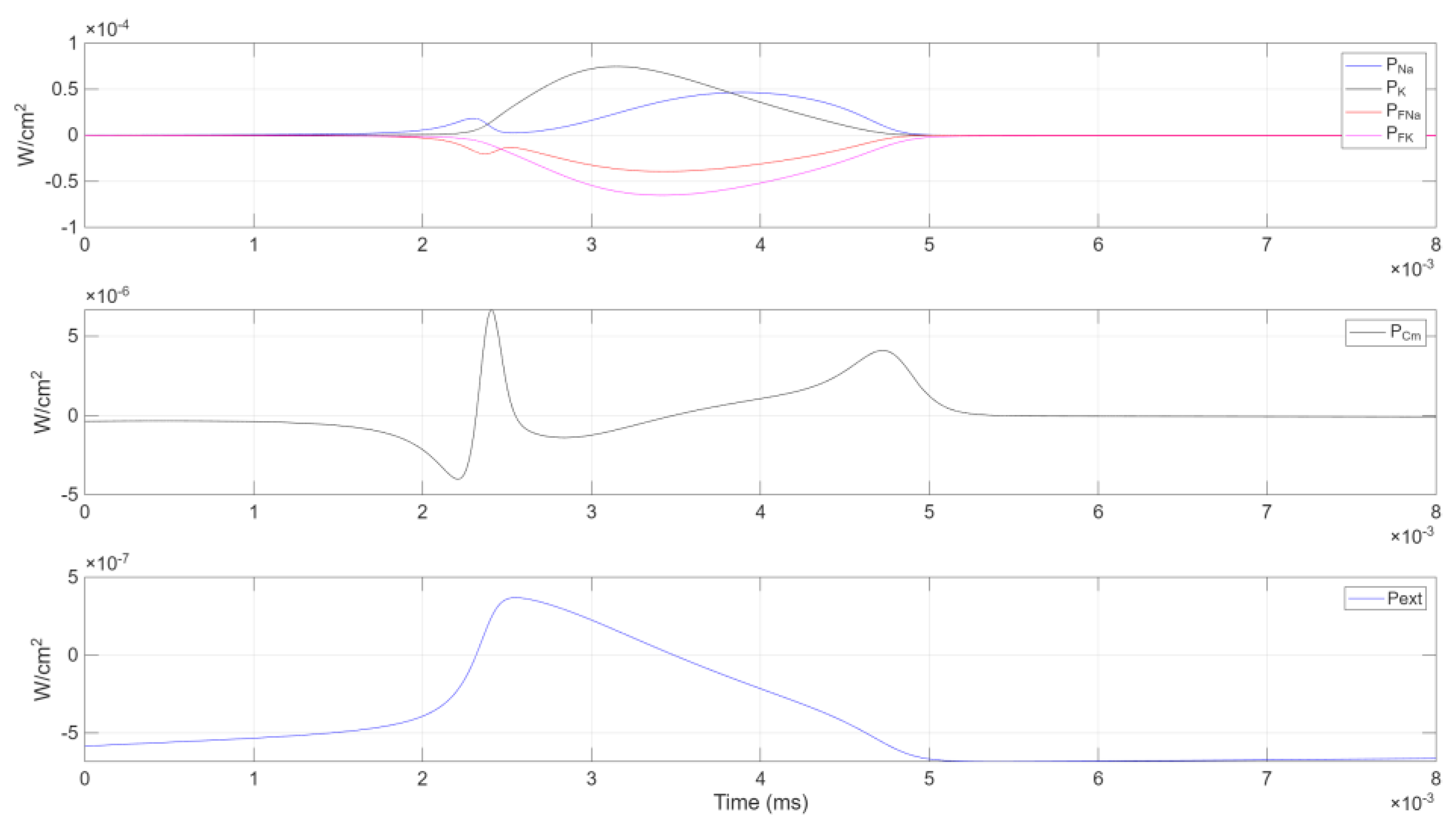
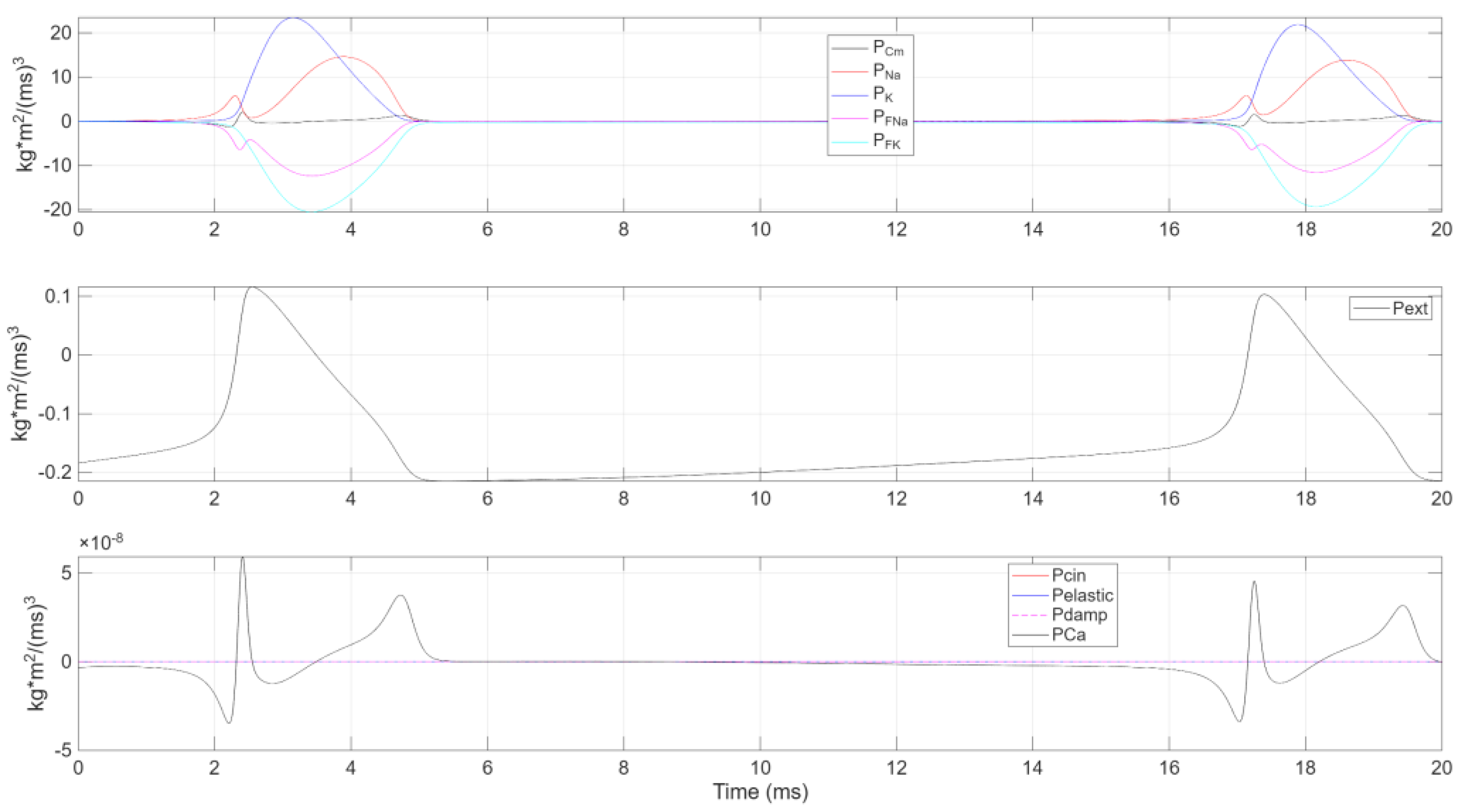
Figure 5.
The figure displays the power components of a single charge-discharge cycle organized into three subplots due to differing -axis scales: (top) ionic power contributions (, , , ); (middle) capacitive power (); (bottom) external power ().
Figure 5.
The figure displays the power components of a single charge-discharge cycle organized into three subplots due to differing -axis scales: (top) ionic power contributions (, , , ); (middle) capacitive power (); (bottom) external power ().
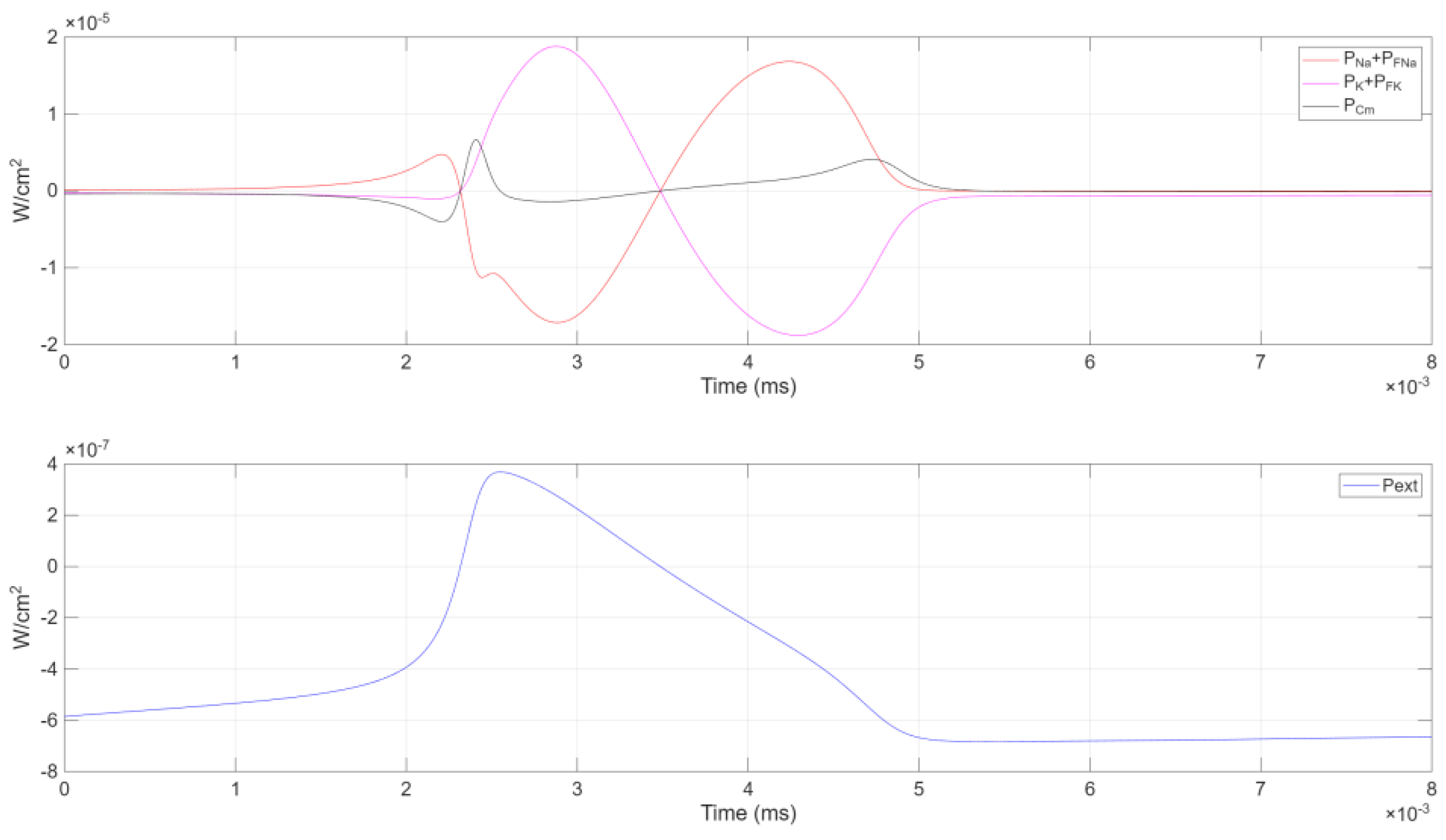
Figure 6.
The first subplot shows the combined power contributions from the ionic channels ( and ) and the capacitive power (). The second subplot illustrates the external power ().
Figure 6.
The first subplot shows the combined power contributions from the ionic channels ( and ) and the capacitive power (). The second subplot illustrates the external power ().
Figure 6 reveals the interdependent temporal evolution of
,
, and the combined ionic channel powers
and
:
Initial Phase. transitions from negative values to zero, reflecting a diminishing energy supply from the external current. It then peaks positively around . Meanwhile, —the power in the membrane capacitance —shows a complementary response: dropping to a negative extremum, then surging to a positive peak shortly thereafter.
Sodium Channel Power. The combined sodium channel power closely mirrors but with opposite polarity. It peaks positively at —when is at its minimum—and then decreases to a local minimum at . Shortly after the external power peak, it reaches an absolute minimum at .
Potassium Channel Power. The total potassium channel power lags behind the sodium channel, exhibiting an opposite trend. When rises, drops, and vice versa. Its positive peak emerges slightly delayed, in line with potassium’s role in repolarization.
Convergence and Next Cycle. Notably, at , all internal power terms converge to zero, while remains positive and continues to decrease. Soon after, becomes positive and becomes negative, with both reaching extreme values at . A subsequent relative maximum in appears at . Finally, dips into negative territory, completing one cycle of power exchange and preparing the system for the next.
This analysis underscores the cyclical energy flow within the HH framework. Depolarization involves strong coupling between capacitive charging and sodium ion flux, while repolarization is facilitated by potassium’s delayed conductance. After hyperpolarization, the system transitions to a state where the external current again becomes the primary energy source, allowing the cycle to repeat.
These power exchanges emphasize both the electrical storage and dissipation mechanisms inherent in neuronal activity. The external energy supply sustains the rapid influx and efflux of ions through voltage-gated channels, restoring the membrane potential after each depolarization event. Through this lens, the Hodgkin–Huxley model not only captures the electrophysiological behavior of excitable cells but also provides a framework for analyzing the energetics and metabolic costs of neuronal signaling.
3.4. Integration of the Hodgkin-Huxley Model with a Elettro-Mechanical Oscillator
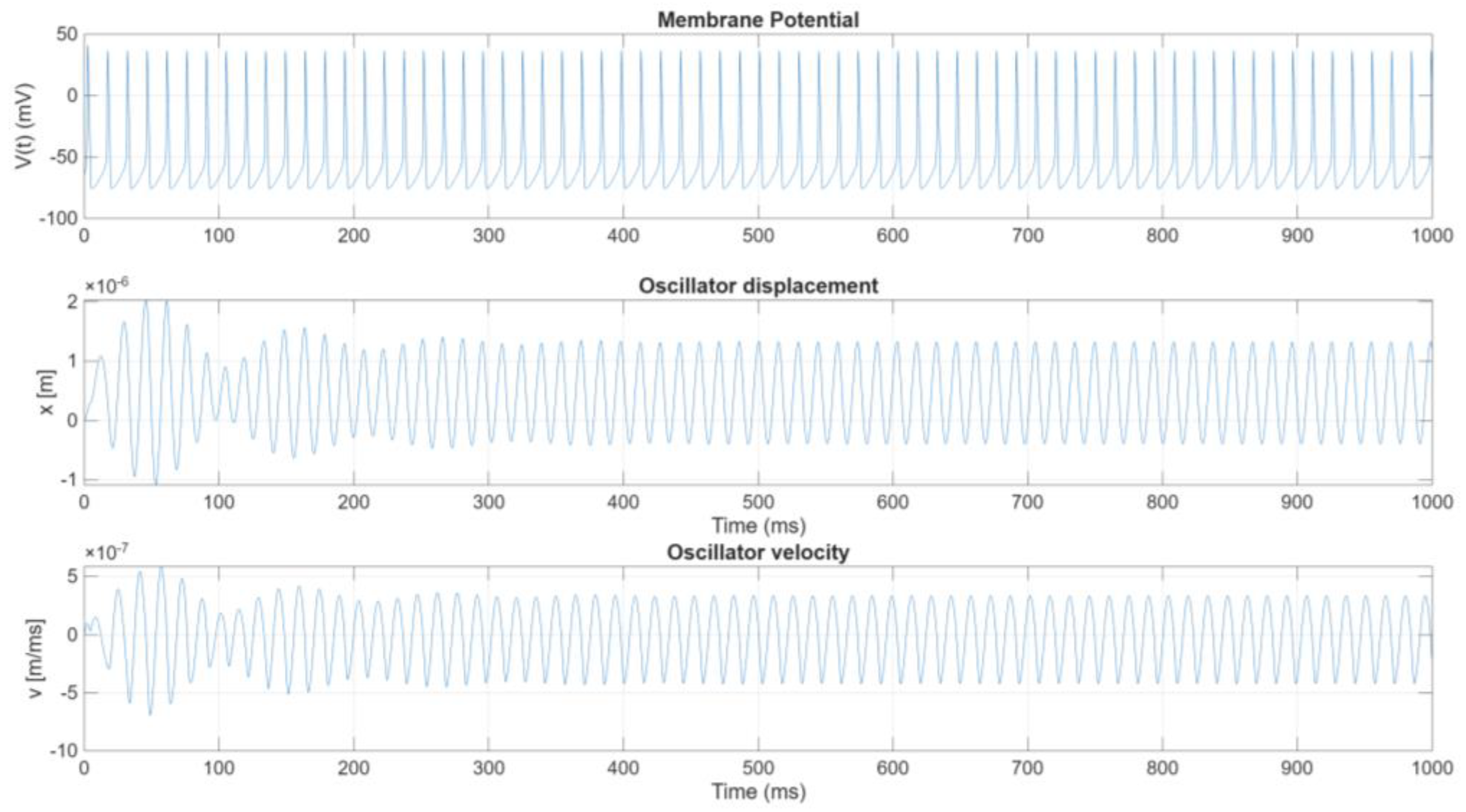
The coupling of the HH neuronal model with a electro-mechanical oscillator introduces a novel bioelectromechanical system, enabling the direct conversion of membrane potentials into mechanical motion. This integration leverages the dynamic properties of the HH model and incorporates the capacitive behavior of the membrane into a resonating mechanical structure. The system’s equations are presented below in coherence with the notation used in this study.
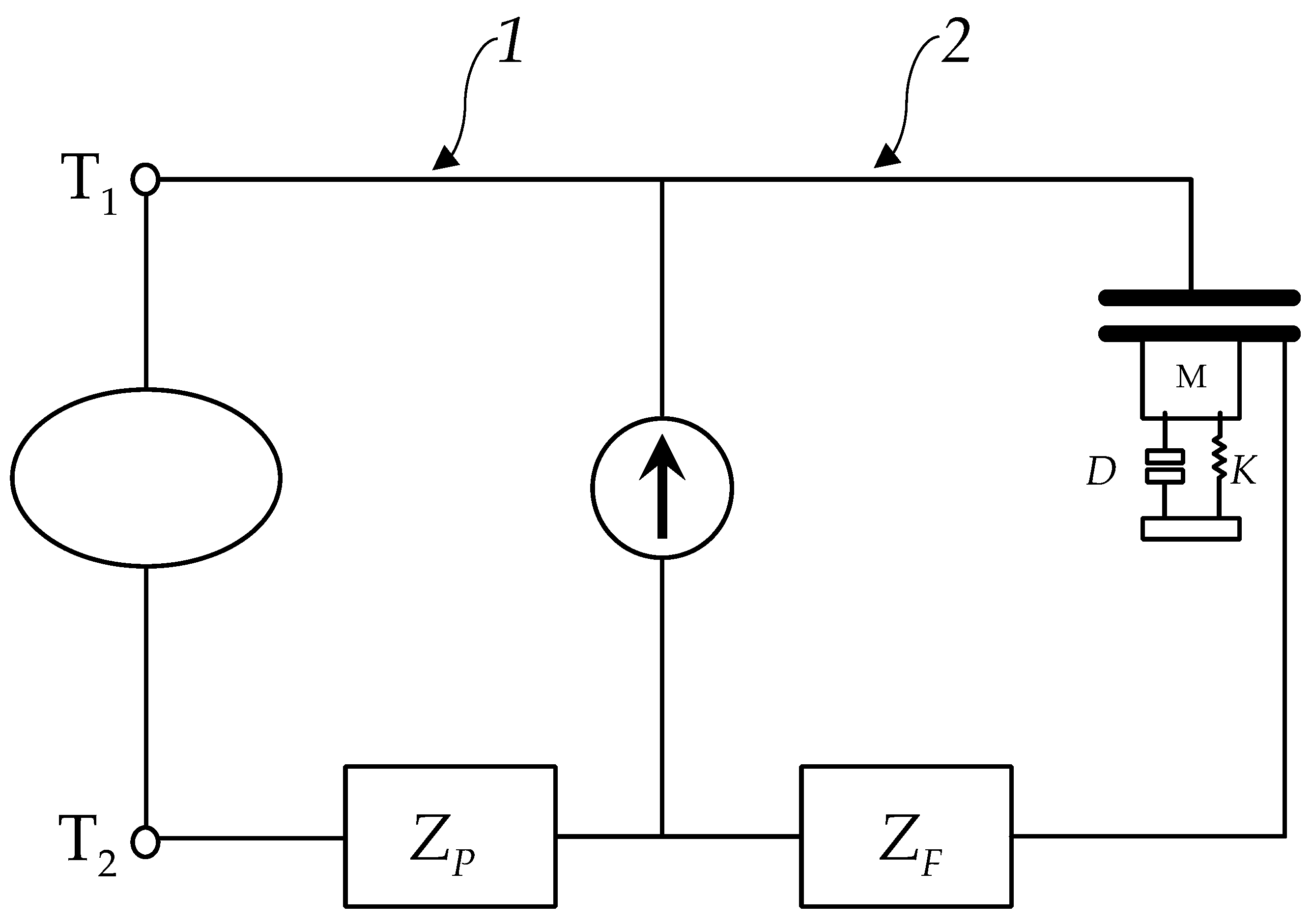
Focusing on the analysis of the driving circuit (1) with the electromechanical converting circuit (2), as illustrated in
Figure 1, the complete system integrates the neuronal membrane described by the HH model with a mechanical resonator. This resonator comprises a mass (M), a spring with stiffness (K), and a damping element characterized by the coefficient (D). Additionally, the mechanical oscillator is equipped with a capacitor, whose capacitance varies with the displacement of the oscillator’s movable plate.
To ensure the compatibility of the two systems, the HH model equations (1) —formulated in terms of specific quantities such as current per unit area—are scaled by the total membrane area
, in this case, a cell radius of 5000
was assumed. The membrane dynamics are governed by the following set of equations:
Here is the current in capacitor coupled to the elastic mechanical oscillator.
The mechanical oscillator is described by the following second-order differential equation:
where:
is the displacement of the movable plate of the capacitor,
is the surface area of the capacitor plates,
is the displacement-dependent capacitance of the capacitor, is the permittivity of free space,
represents the rest distance between the fixed and movable plates of the capacitor incorporated into the resonator, it ensures the capacitor has a finite capacitance in its rest state.
The term on the right-hand side of the equation represents the electrostatic force exerted by the capacitor, which depends on the square of the transmembrane potential and the displacement-dependent capacitance.
Note that to integrate this equation with the HH model, which utilizes time in milliseconds, we redefine the time variable
as
(ms):
This substitution affects the time derivatives as follows:
Substituting these into the mechanical oscillator equation yields:
where:
is time in milliseconds (ms),
remains in meters (m),
,
, and
retain their SI units (kg, N·s/m, and N/m respectively). This normalization ensures that the time scales of the mechanical oscillator are directly compatible with those of the HH neuronal model, facilitating coherent integration of the bioelectrical and mechanical dynamics.
When the HH model is integrated with the mechanical oscillator, the membrane potential influences the resonator’s motion through the variable capacitor, and the displacement x in turn modifies the capacitance seen by the membrane. This two-way feedback links the bioelectrical and mechanical components and can produce complex dynamical behavior. Rewriting eq.s (2) and (3) in a normal form:
the last equation can also be written as where: and Energy Considerations
The powers associated with the electro-mechanical resonator in the coupled HH and resonator system are derived from the mechanical energy components and their interactions with the capacitor.
The variable capacitor depends on the resonator’s displacement x(t), the instantaneous energy stored in this capacitor is:
. Consequently, the instantaneous power associated with
Ca arises from the time derivative of this electrostatic energy:
. After substitution with the actual value of
, the formula produces:
The first term quantifies the power contribution due to the mechanical motion of the capacitor plate, while the second term, corresponds to the conventional power exchange from changes in the voltage across the capacitor at a given capacitance.
Power due to the resonator's kinetic energy, to the spring force and dissipated by the resonator's damping are listed below:
The total power balance for the resonator alone is:
When the resonator is coupled with the HH model, the complete power balance reads:
recalling that the previously calculated powers for the HH model, in section 3.2, were specific powers, they must be multiplied by the cell surface area
to obtain the total power contributions.
The energy exchanges in the coupled system can be categorized into: i) Electrochemical Energy, represented by the ionic currents and the capacitive charging/discharging of the membrane; ii) Mechanical Energy: comprising kinetic energy, potential energy, and dissipation; iii) Electrostatic Energy, stored in the capacitor of the mechanical oscillator.