Submitted:
03 March 2025
Posted:
05 March 2025
Read the latest preprint version here
Abstract
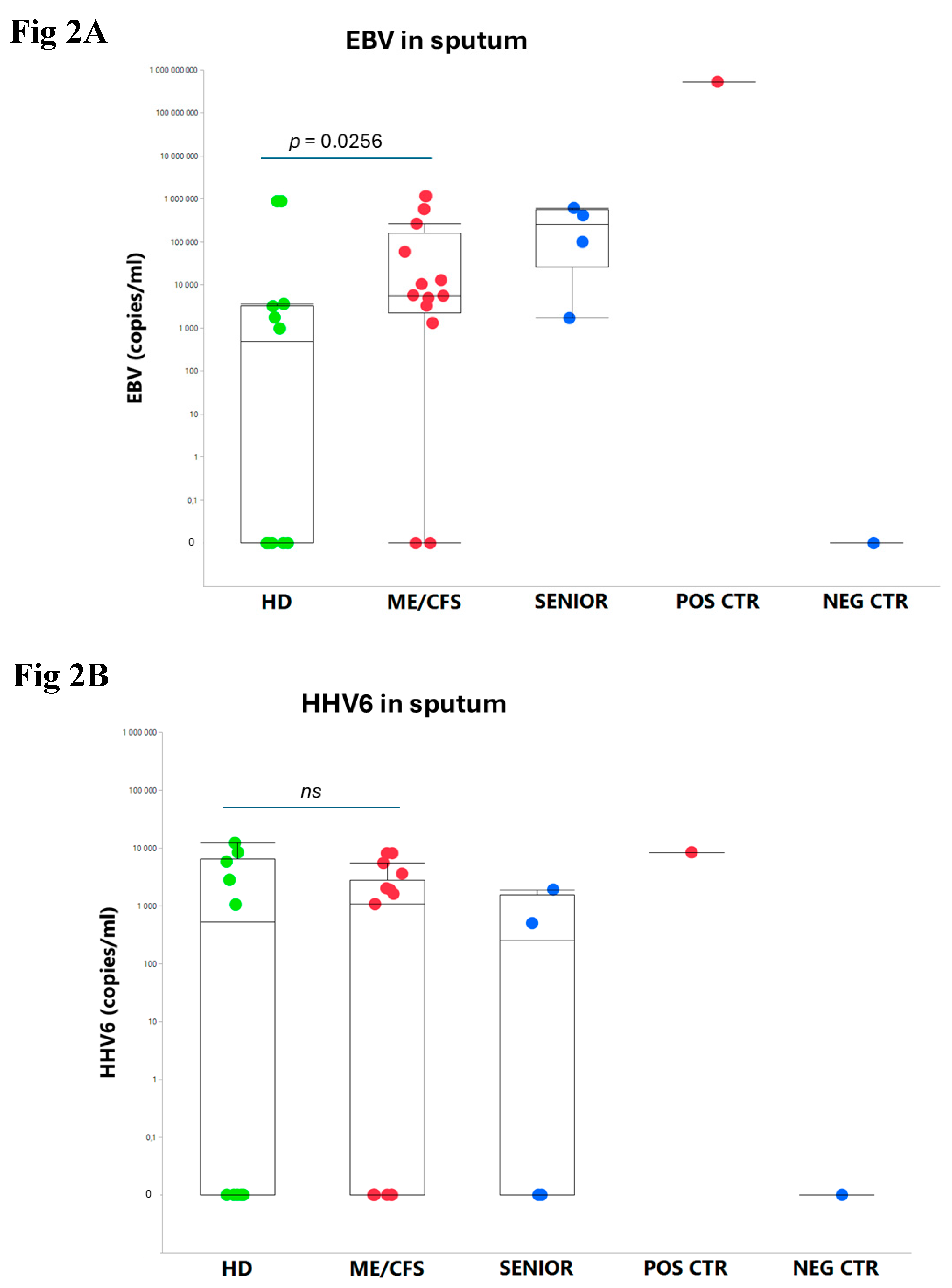
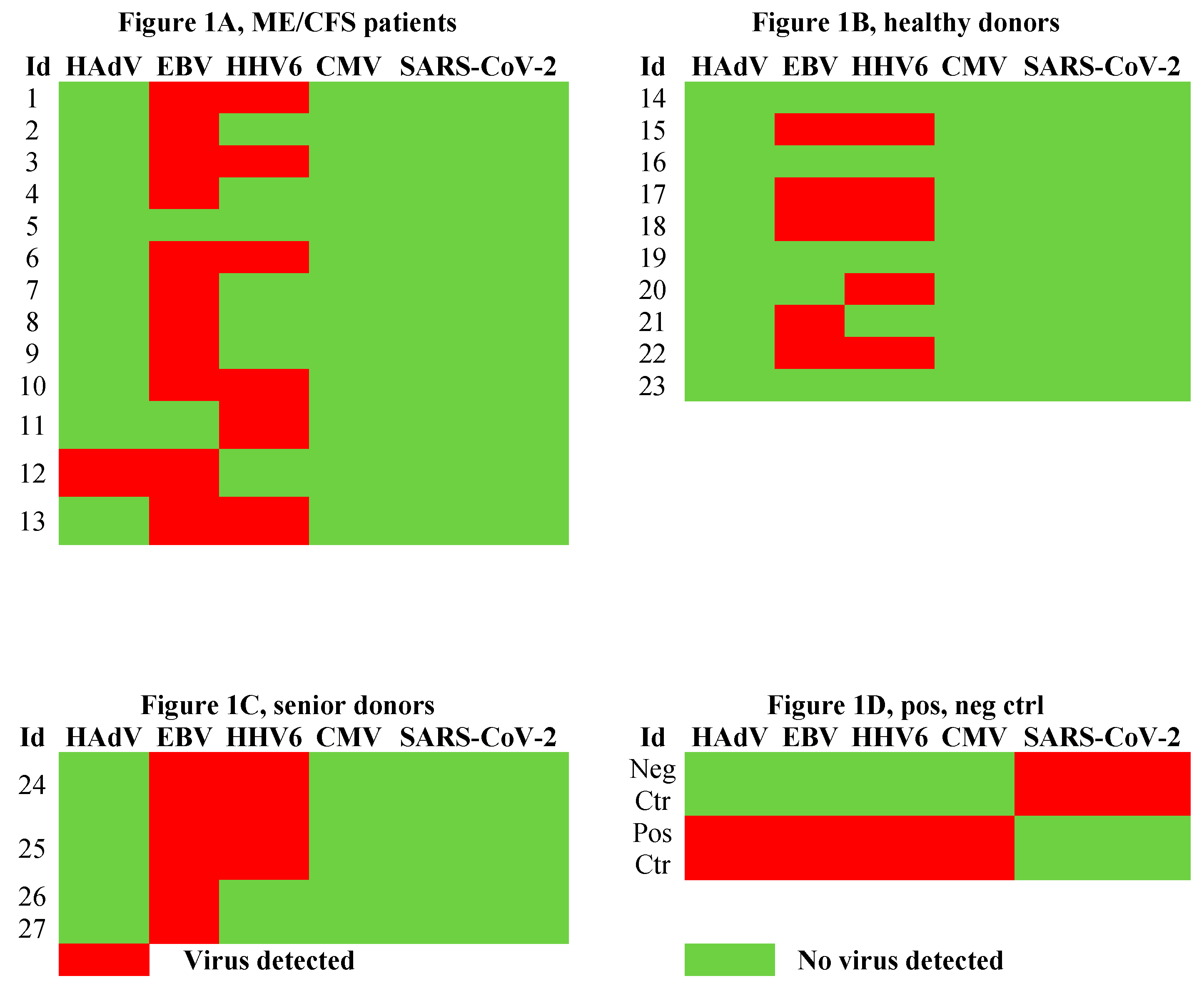
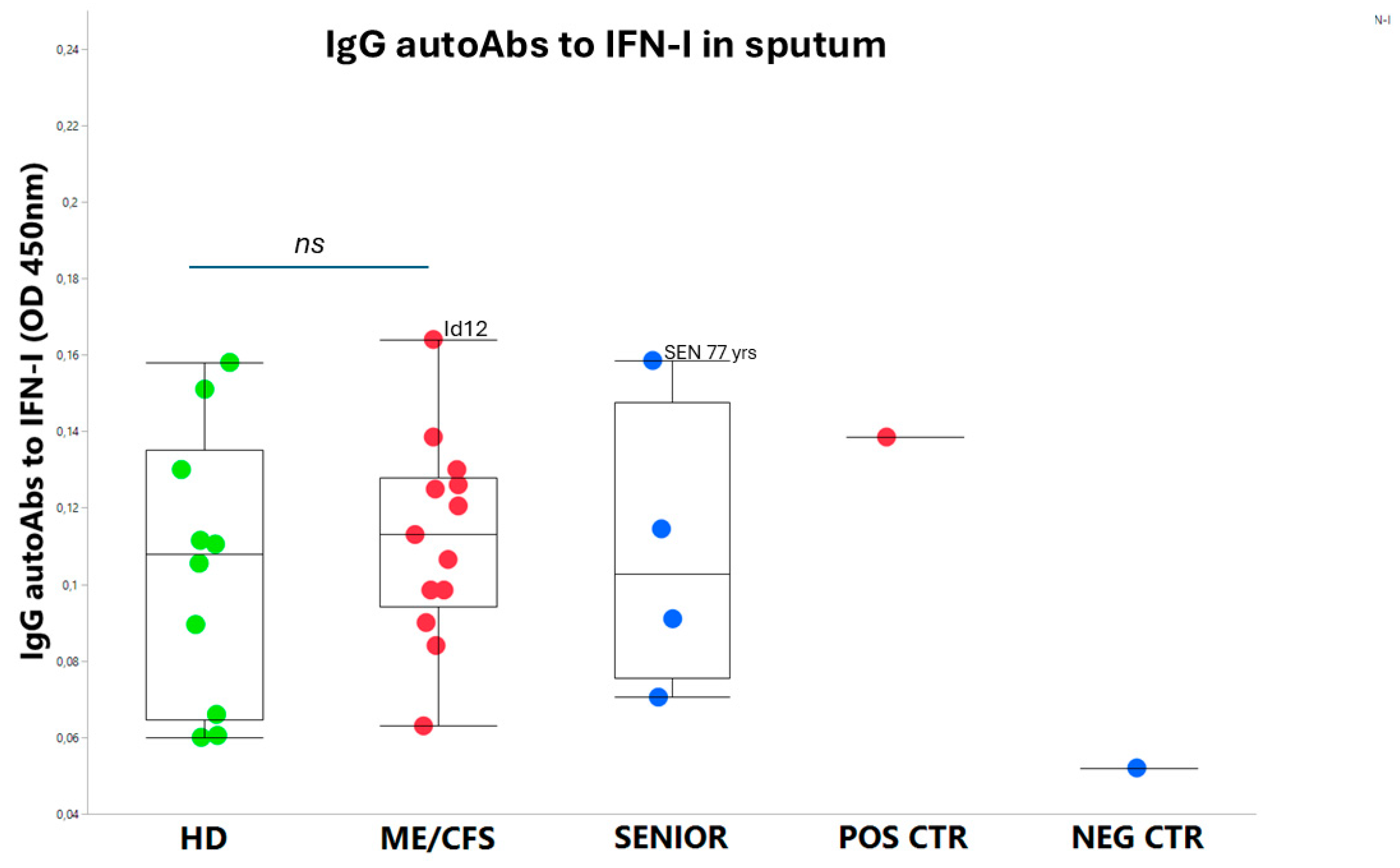
An exhausted antiviral immune response is observed in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and post-SARS-CoV-2 syndrome also termed long COVID. In this study, potential mechanisms behind this exhaustion were investigated. First, the viral load of Epstein-Barr virus (EBV), human adenovirus (HAdV), human cytomegalovirus (HCMV), human herpesvirus 6 (HHV6), and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was determined in sputum samples (n=29) derived from ME/CFS patients (n=13), healthy controls (n=10), elderly healthy controls (n=4), and immunosuppressed controls (n=2). Secondly, autoantibodies (autoAbs) to type I interferon (IFN-I) in sputum were analyzed to possibly explain impaired viral immunity. We found that ME/CFS patients released EBV at a significantly higher level compared to controls (p=0.0256). HHV6 was present in ~50% of all participants at the same level. HAdV was detected in two cases with immunosuppression and severe ME/CFS, respectively. HCMV and SARS-CoV-2 were found only in immunosuppressed controls. Notably, anti-IFN-I autoAbs in ME/CFS and controls did not differ, except in a severe ME/CFS case showing an increased level. We conclude that ME/CFS patients, compared to controls, have a significantly higher load of EBV. IFN-I autoAbs cannot explain IFN-I dysfunction, with the possible exception of severe cases, also reported in severe SARS-CoV-2. We forward that additional mechanisms, such as viral evasion of IFN-I effect via degradation of IFN-receptors; may be present in ME/CFS, which demands further studies.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Ethical Permit
2.3. Sputum Collection
2.4. Analysis of EBV, HCMV, HHV6, HAdV, and SARS-CoV-2 in Sputum
2.5. ELISA Analysis of IgG autoAbs to Type-I IFN
2.6. Statistical Analyses
3. Results
3.1. Viral Load of EBV, HCMV, HHV6, HAdV, and SARS-CoV-2 in Sputum.
3.2. IgG autoAbs Against IFN-I in ME/CFS Patients
4. Discussion
4.1. ME/CFS Immune and Antiviral Dysregulation -Unknown Mechanisms
4.2. Overload of Epstein-Barr Virus in ME/CFS
4.3. Human Adenovirus and Herpesvirus Reactivation in Airways
4.4. Latent Virus – Host Immune Balance
4.5. Limitation of the Study
4.6. Future Perspectives and Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Vahratian, A.; Lin, J.S.; Bertolli, J.; Unger, E.R. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome in Adults: United States, 2021-2022. NCHS Data Brief. 2023, 1–8. [Google Scholar] [CrossRef]
- Valdez, A.R.; Hancock, E.E.; Adebayo, S.; Kiernicki, D.J.; Proskauer, D.; Attewell, J.R.; Bateman, L.; DeMaria, A., Jr.; Lapp, C.W.; Rowe, P.C.; et al. Estimating Prevalence, Demographics, and Costs of ME/CFS Using Large Scale Medical Claims Data and Machine Learning. Front Pediatr. 2018, 6, 412. [Google Scholar] [CrossRef]
- Bateman, L.; Bested, A.C.; Bonilla, H.F.; Chheda, B.V.; Chu, L.; Curtin, J.M.; Dempsey, T.T.; Dimmock, M.E.; Dowell, T.G.; Felsenstein, D.; et al. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Essentials of Diagnosis and Management. Mayo Clin Proc. 2021, 96, 2861–2878. [Google Scholar] [CrossRef]
- Hanson, M.R. The viral origin of myalgic encephalomyelitis/chronic fatigue syndrome. PLoS Pathog. 2023, 19, e1011523. [Google Scholar] [CrossRef]
- Hickie, I.; Davenport, T.; Wakefield, D.; Vollmer-Conna, U.; Cameron, B.; Vernon, S.D.; Reeves, W.C.; Lloyd, A.; Dubbo Infection Outcomes Study, G. Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: Prospective cohort study. BMJ. 2006, 333, 575. [Google Scholar] [CrossRef]
- Naess, H.; Sundal, E.; Myhr, K.M.; Nyland, H.I. Postinfectious and chronic fatigue syndromes: Clinical experience from a tertiary-referral centre in Norway. In Vivo. 2010, 24, 185–188. [Google Scholar]
- Blomberg, J.; Gottfries, C.G.; Elfaitouri, A.; Rizwan, M.; Rosén, A. Infection Elicited Autoimmunity and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: An Explanatory Model. Front Immunol. 2018, 9, 229. [Google Scholar] [CrossRef]
- Komaroff, A.L.; Lipkin, W.I. Insights from myalgic encephalomyelitis/chronic fatigue syndrome may help unravel the pathogenesis of postacute COVID-19 syndrome. Trends Mol Med. 2021, 27, 895–906. [Google Scholar] [CrossRef]
- Apostolou, E.; Rizwan, M.; Moustardas, P.; Sjögren, P.; Bertilson, B.C.; Bragée, B.; Polo, O.; Rosén, A. Saliva antibody-fingerprint of reactivated latent viruses after mild/asymptomatic COVID-19 is unique in patients with myalgic-encephalomyelitis/chronic fatigue syndrome. Front Immunol. 2022, 13, 949787. [Google Scholar] [CrossRef]
- Hannestad, U.; Apostolou, E.; Sjögren, P.; Bragée, B.; Polo, O.; Bertilson, B.C.; Rosén, A. Post-COVID sequelae effect in chronic fatigue syndrome: SARS-CoV-2 triggers latent adenovirus in the oral mucosa. Front Med (Lausanne). 2023, 10, 1208181. [Google Scholar] [CrossRef]
- Schindele, A.; Holm, A.; Kraft, S.; Nylander, K.; Allard, A.; Olofsson, K. Cross-evaluating Epstein-Barr virus, human papilloma virus, human cytomegalovirus and human adenovirus in nasal polyps and turbinate mucosa. Acta Otolaryngol. 2025, 145, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.T.; Lidsky, P.V.; Xiao, Y.; Cheng, R.; Lee, I.T.; Nakayama, T.; Jiang, S.; He, W.; Demeter, J.; Knight, M.G.; et al. SARS-CoV-2 replication in airway epithelia requires motile cilia and microvillar reprogramming. Cell. 2023, 186, 112–130.e120. [Google Scholar] [CrossRef] [PubMed]
- Vojdani, A.; Vojdani, E.; Saidara, E.; Maes, M. Persistent SARS-CoV-2 Infection, EBV, HHV-6 and Other Factors May Contribute to Inflammation and Autoimmunity in Long COVID. Viruses. 2023, 15. [Google Scholar] [CrossRef]
- Albanese, M.; Tagawa, T.; Hammerschmidt, W. Strategies of Epstein-Barr virus to evade innate antiviral immunity of its human host. Front Microbiol. 2022, 13, 955603. [Google Scholar] [CrossRef]
- Hale, B.G. Autoantibodies targeting type I interferons: Prevalence, mechanisms of induction, and association with viral disease susceptibility. Eur J Immunol. 2023, 53, e2250164. [Google Scholar]
- Carruthers, B.M. Definitions and aetiology of myalgic encephalomyelitis: How the Canadian consensus clinical definition of myalgic encephalomyelitis works. J Clin Pathol. 2007, 60, 117–119. [Google Scholar] [CrossRef]
- Carruthers, B.M.; van de Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J Intern Med. 2011, 270, 327–338. [Google Scholar] [CrossRef]
- Hernroth, B.E.; Conden-Hansson, A.C.; Rehnstam-Holm, A.S.; Girones, R.; Allard, A.K. Environmental factors influencing human viral pathogens and their potential indicator organisms in the blue mussel, Mytilus edulis: The first Scandinavian report. Appl Environ Microbiol. 2002, 68, 4523–4533. [Google Scholar] [CrossRef]
- Schindele, A.; Holm, A.; Nylander, K.; Allard, A.; Olofsson, K. Mapping human papillomavirus, Epstein-Barr virus, cytomegalovirus, adenovirus, and p16 in laryngeal cancer. Discov Oncol. 2022, 13, 18. [Google Scholar] [CrossRef]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brunink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020, 25. [Google Scholar] [CrossRef]
- Gervais, A.; Rovida, F.; Avanzini, M.A.; Croce, S.; Marchal, A.; Lin, S.C.; Ferrari, A.; Thorball, C.W.; Constant, O.; Le Voyer, T.; et al. Autoantibodies neutralizing type I IFNs underlie West Nile virus encephalitis in approximately 40% of patients. J Exp Med. 2023, 220. [Google Scholar] [CrossRef]
- Eaton-Fitch, N.; Rudd, P.; Er, T.; Hool, L.; Herrero, L.; Marshall-Gradisnik, S. Immune exhaustion in ME/CFS and long COVID. JCI Insight. 2024, 9. [Google Scholar] [CrossRef]
- Blomberg, J.; Rizwan, M.; Bohlin-Wiener, A.; Elfaitouri, A.; Julin, P.; Zachrisson, O.; Rosén, A.; Gottfries, C.G. Antibodies to Human Herpesviruses in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients. Front Immunol. 2019, 10, 1946. [Google Scholar] [CrossRef]
- Apostolou, E.; Rosén, A. Epigenetic reprograming in myalgic encephalomyelitis/chronic fatigue syndrome: A narrative of latent viruses. J Intern Med. 2024, 296, 93–115. [Google Scholar] [CrossRef]
- Choutka, J.; Jansari, V.; Hornig, M.; Iwasaki, A. Unexplained post-acute infection syndromes. Nat Med. 2022, 28, 911–923. [Google Scholar] [CrossRef]
- Su, Y.; Yuan, D.; Chen, D.G.; Ng, R.H.; Wang, K.; Choi, J.; Li, S.; Hong, S.; Zhang, R.; Xie, J.; et al. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell. 2022, 185, 881–895.e820. [Google Scholar] [CrossRef]
- Le Voyer, T.; Parent, A.V.; Liu, X.; Cederholm, A.; Gervais, A.; Rosain, J.; Nguyen, T.; Perez Lorenzo, M.; Rackaityte, E.; Rinchai, D.; et al. Autoantibodies against type I IFNs in humans with alternative NF-kappaB pathway deficiency. Nature. 2023, 623, 803–813. [Google Scholar] [CrossRef]
- Bastard, P.; Vazquez, S.E.; Liu, J.; Laurie, M.T.; Wang, C.Y.; Gervais, A.; Le Voyer, T.; Bizien, L.; Zamecnik, C.; Philippot, Q.; et al. Vaccine breakthrough hypoxemic COVID-19 pneumonia in patients with auto-Abs neutralizing type I IFNs. Sci Immunol. 2023, 8, eabp8966. [Google Scholar] [CrossRef]
- Philippot, Q.; Fekkar, A.; Gervais, A.; Le Voyer, T.; Boers, L.S.; Conil, C.; Bizien, L.; de Brabander, J.; Duitman, J.W.; Romano, A.; et al. Autoantibodies Neutralizing Type I IFNs in the Bronchoalveolar Lavage of at Least 10% of Patients During Life-Threatening COVID-19 Pneumonia. J Clin Immunol. 2023, 43, 1093–1103. [Google Scholar] [CrossRef]
- Gervais, A.; Marchal, A.; Fortova, A.; Berankova, M.; Krbkova, L.; Pychova, M.; Salat, J.; Zhao, S.; Kerrouche, N.; Le Voyer, T.; et al. Autoantibodies neutralizing type I IFNs underlie severe tick-borne encephalitis in approximately 10% of patients. J Exp Med. 2024, 221. [Google Scholar] [CrossRef]
- Feng, A.; Yang, E.Y.; Moore, A.R.; Dhingra, S.; Chang, S.E.; Yin, X.; Pi, R.; Mack, E.K.; Volkel, S.; Gessner, R.; et al. Autoantibodies are highly prevalent in non-SARS-CoV-2 respiratory infections and critical illness. JCI Insight. 2023, 8. [Google Scholar] [CrossRef]
- Walter, J.E.; Rosen, L.B.; Csomos, K.; Rosenberg, J.M.; Mathew, D.; Keszei, M.; Ujhazi, B.; Chen, K.; Lee, Y.N.; Tirosh, I.; et al. Broad-spectrum antibodies against self-antigens and cytokines in RAG deficiency. J Clin Invest. 2015, 125, 4135–4148. [Google Scholar] [CrossRef] [PubMed]
- Ariza, M.E. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: The Human Herpesviruses Are Back! Biomolecules. 2021, 11. [Google Scholar] [CrossRef]
- Shikova, E.; Reshkova, V.; Kumanova capital, A.C.; Raleva, S.; Alexandrova, D.; Capo, N.; Murovska, M.; On Behalf Of The European Network On Me/Cfs, E. Cytomegalovirus, Epstein-Barr virus, and human herpesvirus-6 infections in patients with myalgic small ie, Cyrillicncephalomyelitis/chronic fatigue syndrome. J Med Virol. 2020, 92, 3682–3688. [Google Scholar]
- Jeong, J.H.; Kim, K.H.; Jeong, S.H.; Park, J.W.; Lee, S.M.; Seo, Y.H. Comparison of sputum and nasopharyngeal swabs for detection of respiratory viruses. J Med Virol. 2014, 86, 2122–2127. [Google Scholar] [CrossRef]
- Lion, T. Adenovirus persistence, reactivation, and clinical management. FEBS Lett. 2019, 593, 3571–3582. [Google Scholar] [CrossRef]
- Garnett, C.T.; Erdman, D.; Xu, W.; Gooding, L.R. Prevalence and quantitation of species C adenovirus DNA in human mucosal lymphocytes. J Virol. 2002, 76, 10608–10616. [Google Scholar] [CrossRef]
- Lion, T. Adenovirus infections in immunocompetent and immunocompromised patients. Clin Microbiol Rev. 2014, 27, 441–462. [Google Scholar] [CrossRef]
- Kosulin, K.; Haberler, C.; Hainfellner, J.A.; Amann, G.; Lang, S.; Lion, T. Investigation of adenovirus occurrence in pediatric tumor entities. J Virol. 2007, 81, 7629–7635. [Google Scholar] [CrossRef]
- Virgin, H.W.; Wherry, E.J.; Ahmed, R. Redefining chronic viral infection. Cell. 2009, 138, 30–50. [Google Scholar]
- Levin, D.; Schneider, W.M.; Hoffmann, H.H.; Yarden, G.; Busetto, A.G.; Manor, O.; Sharma, N.; Rice, C.M.; Schreiber, G. Multifaceted activities of type I interferon are revealed by a receptor antagonist. Sci Signal. 2014, 7, ra50. [Google Scholar] [CrossRef] [PubMed]
- Crow, Y.J.; Stetson, D.B. The type I interferonopathies: 10 years on. Nat Rev Immunol. 2022, 22, 471–483. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Q.; Zheng, C.; Wang, L. MAVS: The next STING in cancers and other diseases. Crit Rev Oncol Hematol. 2024, 207, 104610. [Google Scholar] [CrossRef]
- Miyagi, S.; Watanabe, T.; Hara, Y.; Arata, M.; Uddin, M.K.; Mantoku, K.; Sago, K.; Yanagi, Y.; Suzuki, T.; Masud, H.; et al. A STING inhibitor suppresses EBV-induced B cell transformation and lymphomagenesis. Cancer Sci. 2021, 112, 5088–5099. [Google Scholar] [CrossRef]
- Pasquesi, G.I.M.; Allen, H.; Ivancevic, A.; Barbachano-Guerrero, A.; Joyner, O.; Guo, K.; Simpson, D.M.; Gapin, K.; Horton, I.; Nguyen, L.L.; et al. Regulation of human interferon signaling by transposon exonization. Cell. 2024, 187, 7621–7636.e7619. [Google Scholar] [CrossRef]
- Peluso, M.J.; Deeks, S.G. Mechanisms of long COVID and the path toward therapeutics. Cell. 2024, 187, 5500–5529. [Google Scholar] [CrossRef]
- Kosulin, K.; Geiger, E.; Vecsei, A.; Huber, W.D.; Rauch, M.; Brenner, E.; Wrba, F.; Hammer, K.; Innerhofer, A.; Potschger, U.; et al. Persistence and reactivation of human adenoviruses in the gastrointestinal tract. Clin Microbiol Infect. 2016, 22, 381 e381‐381 e388. [Google Scholar] [CrossRef]
- Alvarez-Cardona, J.J.; Whited, L.K.; Chemaly, R.F. Brincidofovir: Understanding its unique profile and potential role against adenovirus and other viral infections. Future Microbiol. 2020, 15, 389–400. [Google Scholar] [CrossRef]
- El-Haddad, D.; El Chaer, F.; Vanichanan, J.; Shah, D.P.; Ariza-Heredia, E.J.; Mulanovich, V.E.; Gulbis, A.M.; Shpall, E.J.; Chemaly, R.F. Brincidofovir (CMX-001) for refractory and resistant CMV and HSV infections in immunocompromised cancer patients: A single-center experience. Antiviral Res. 2016, 134, 58–62. [Google Scholar] [CrossRef]
- Camargo, J.F.; Morris, M.I.; Abbo, L.M.; Simkins, J.; Saneeymehri, S.; Alencar, M.C.; Lekakis, L.J.; Komanduri, K.V. The use of brincidofovir for the treatment of mixed dsDNA viral infection. J Clin Virol. 2016, 83, 1–4. [Google Scholar] [CrossRef]
- Hill, J.A.; Nichols, W.G.; Marty, F.M.; Papanicolaou, G.A.; Brundage, T.M.; Lanier, R.; Zerr, D.M.; Boeckh, M.J. Oral brincidofovir decreases the incidence of HHV-6B viremia after allogeneic HCT. Blood. 2020, 135, 1447–1451. [Google Scholar] [CrossRef] [PubMed]
- Grimley, M.S.; Maron, G. editors. Preliminary Results of a Phase 2a Clinical Trial to Evaluate Safety, Tolerability and Antiviral Activity of Intravenous Brincidofovir (BCV IV) in Immunocompromised Patients with Adenovirus Infection. 65th ASH Annual Meeting; 2023; San Diego, CA: Blood.
- Kosulin, K. Intestinal HAdV Infection: Tissue Specificity, Persistence, and Implications for Antiviral Therapy. Viruses. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Radke, J.R.; Cook, J.L. Human adenovirus infections: Update and consideration of mechanisms of viral persistence. Curr Opin Infect Dis. 2018, 31, 251–256. [Google Scholar] [CrossRef]
- Roy, S.; Calcedo, R.; Medina-Jaszek, A.; Keough, M.; Peng, H.; Wilson, J.M. Adenoviruses in lymphocytes of the human gastro-intestinal tract. PLoS ONE. 2011, 6, e24859. [Google Scholar] [CrossRef]
| ME/CFS patients | Healthy controls | Senior controls | |||||||||||||
| Sample | Duration of | Disease | Sex | Age | Sample | Duration of | Sex | Age | Sample | Duration of | Sex | Age | |||
| Id | ME/CFS (yrs) | Severity | yrs | Id | ME/CFS (yrs) | yrs | Id | ME/CFS (yrs) | yrs | ||||||
| 1 | 8 | 1 | F | 61 | 14 | NA | F | 56 | 24 | NA | F | 65 | |||
| 2 | 13 | 2 | F | 60 | 15 | NA | F | 33 | 25 | NA | M | 77 | |||
| 3 | 10 | 2 | F | 58 | 16 | NA | F | 61 | 26 | NA | F | 72 | |||
| 4 | 12 | 1 | F | 56 | 17 | NA | F | 61 | 27 | NA | M | 75 | |||
| 5 | 28 | 1 | F | 54 | 18 | NA | F | 37 | Median | 73.5 | |||||
| 6 | 12 | 1 | F | 54 | 19 | NA | F | 49 | Range | 65 - 77 | |||||
| 7 | 10 | 1 | F | 53 | 20 | NA | F | 66 | |||||||
| 8 | 14 | 1 | F | 49 | 21 | NA | M | 48 | |||||||
| 9 | 14 | 2 | F | 48 | 22 | NA | F | 33 | Positive, negative control | ||||||
| 10 | 8 | 1 | F | 37 | 23 | NA | F | 66 | Sample | Duration of | Sex | Age | |||
| 11 | 17 | 2 | F | 37 | Median | 54 | Id | ME/CFS (yrs) | yrs | ||||||
| 12 | 12 | 3 | F | 22 | Range | 33 - 66 | NEG CTR | NA | F | 46 | |||||
| 13 | 27 | 1 | F | 59 | POS CTR | NA | F | 54 | |||||||
| Median | 12 | 52.5 | |||||||||||||
| Range | 8- 28 | 22 - 61 | |||||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




