Submitted:
29 January 2025
Posted:
31 January 2025
You are already at the latest version
Abstract
Keywords:
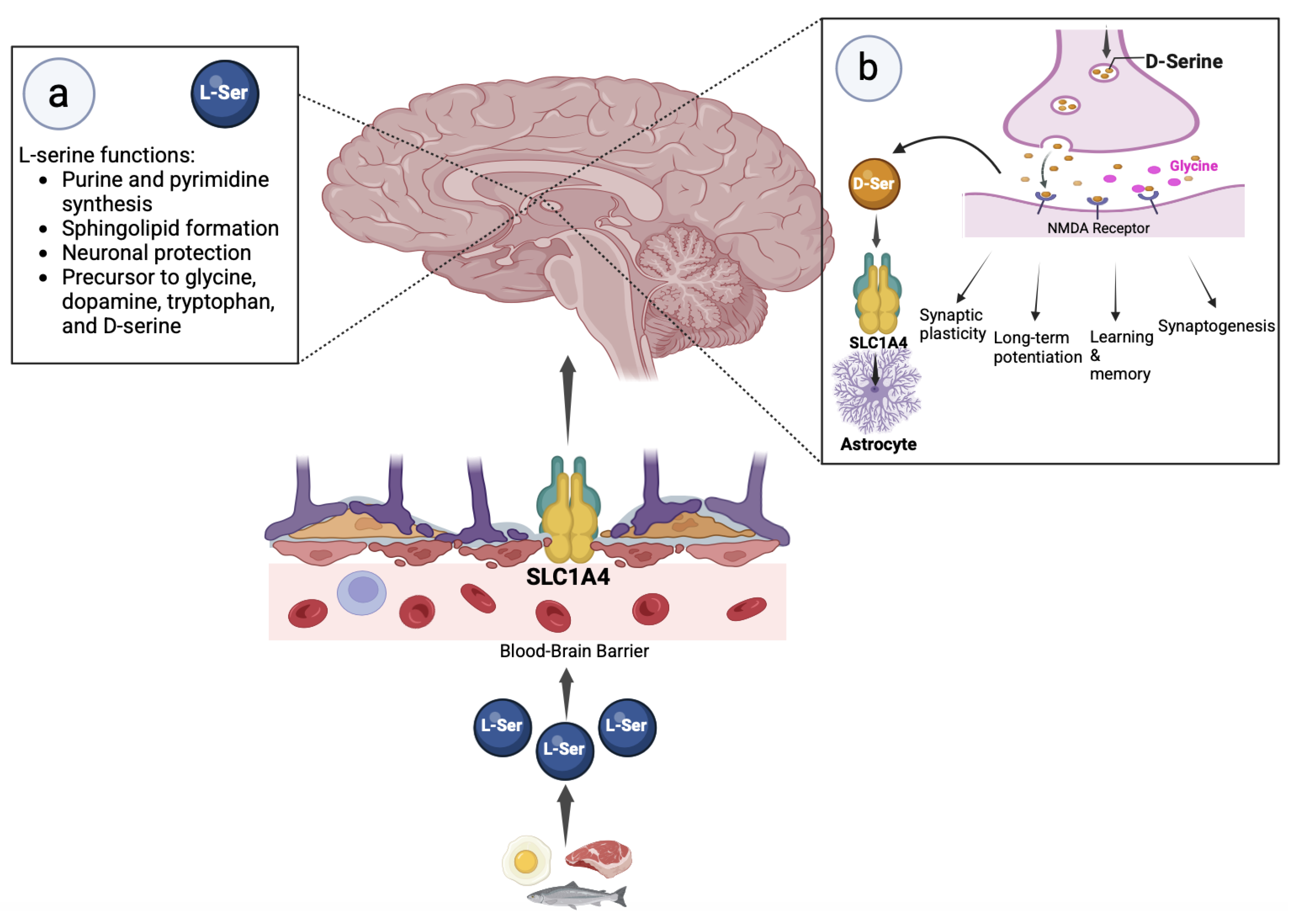
1. Introduction
2. SLC1A4 and Serine Metabolism
3. SLC1A4 in Neurodevelopment
3.1. SPATCCM
3.2. Schizophrenia
4. SLC1A4 and Serine Metabolism in Aging and Neurodegeneration
4.1. Normal Aging
4.2. Alzheimer’s Disease
4.3. Parkinson’s Disease
5. Serine Supplementation
6. Conclusions
References
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| ASCT1 | Alanine-serine-cysteine transporter 1 |
| BBB | Blood-Brain Barrier |
| D-ser | Dextro (D)-serine |
| FAD | Familial Alzheimer’s Disease |
| GluN2 | Glutamate ionotrophic receptor NMDA type subunit 2 |
| GluN3 | Glutamate ionotrophic receptor NMDA type subunit 3 |
| KOMP | Knockout Mouse Project |
| L-ser | Levo (L)-serine |
| LTP | Long-term potentiation |
| NMDA | N-methyl-D-aspartate |
| PD | Parkinson’s disease |
| PCP | Phencyclidine |
| PHGDH | Phosphoglycerate dehydrogenase |
| SLC1A4 | Solute carrier family 1 member 4 gene |
| SLC1A4 | Solute carrier family 1 member 4 protein |
| SNP | Single nucleotide polymorphism |
| SPATCCM | Spastic tetraplegia, thin corpus callosum and progressive microcephaly |
References
- Hofmann, K.; Düker, M.; Fink, T.; Lichter, P.; Stoffel, W. Human Neutral Amino Acid Transporter ASCT1: Structure of the Gene (SLC1A4) and Localization to Chromosome 2p13-p15. Genomics 1994, 24, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Zerangue, N.; Kavanaugh, M.P. ASCT-1 Is a Neutral Amino Acid Exchanger with Chloride Channel Activity. J. Biol. Chem. 1996, 271, 27991–27994. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, E.; Zubedat, S.; Radzishevsky, I.; Valenta, A.C.; Rechnitz, O.; Sason, H.; Sajrawi, C.; Bodner, O.; Konno, K.; Esaki, K.; et al. ASCT1 (Slc1a4) transporter is a physiologic regulator of brain d -serine and neurodevelopment. Proc. Natl. Acad. Sci. 2018, 115, 9628–9633. [Google Scholar] [CrossRef]
- Weiss, M.D.; Derazi, S.; Kilberg, M.S.; Anderson, K.J. Ontogeny and localization of the neutral amino acid transporter ASCT1 in rat brain. Dev. Brain Res. 2001, 130, 183–190. [Google Scholar] [CrossRef]
- Foster, A.C.; Farnsworth, J.; Lind, G.E.; Li, Y.-X.; Yang, J.-Y.; Dang, V.; Penjwini, M.; Viswanath, V.; Staubli, U.; Kavanaugh, M.P. D-Serine Is a Substrate for Neutral Amino Acid Transporters ASCT1/SLC1A4 and ASCT2/SLC1A5, and Is Transported by Both Subtypes in Rat Hippocampal Astrocyte Cultures. PLOS ONE 2016, 11, e0156551. [Google Scholar] [CrossRef]
- Krishnan, K.S.; Billups, B. ASC Transporters Mediate D-Serine Transport into Astrocytes Adjacent to Synapses in the Mouse Brain. Biomolecules 2023, 13, 819. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Sadamoto, Y.; Konno, A.; Kon, Y.; Iwanaga, T. Distribution of neutral amino acid transporter ASCT 1 in the non-neuronal tissues of mice. . 2004, 52, 113–24. [Google Scholar]
- Odeh, M.; Sajrawi, C.; Majcher, A.; Zubedat, S.; Shaulov, L.; Radzishevsky, A.; Mizrahi, L.; Chung, W.K.; Avital, A.; Hornemann, T.; et al. A new type of blood–brain barrier aminoacidopathy underlies metabolic microcephaly associated with SLC1A4 mutations. Brain 2024, 147, 3874–3889. [Google Scholar] [CrossRef]
- Damseh, N.; Simonin, A.; Jalas, C.; A Picoraro, J.; Shaag, A.; Cho, M.T.; Yaacov, B.; Neidich, J.; Al-Ashhab, M.; Juusola, J.; et al. Mutations inSLC1A4, encoding the brain serine transporter, are associated with developmental delay, microcephaly and hypomyelination. J. Med Genet. 2015, 52, 541–547. [Google Scholar] [CrossRef]
- Ratz-Mitchem, M.L.; Leary, G.; Grindeland, A.; Silvius, D.; Guter, J.; Kavanaugh, M.P.; Gunn, T.M. Generation and characterization of a knock-in mouse model for spastic tetraplegia, thin corpus callosum, and progressive microcephaly (SPATCCM). Mamm. Genome 2023, 34, 572–585. [Google Scholar] [CrossRef]
- Furuya, S. An essential role for de novo biosynthesis of L-serine in CNS development. . 2008, 312–5. [Google Scholar]
- Ye, L.; Sun, Y.; Jiang, Z.; Wang, G. L-Serine, an Endogenous Amino Acid, Is a Potential Neuroprotective Agent for Neurological Disease and Injury. Front. Mol. Neurosci. 2021, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhai, P.-P.; Xu, L.-H.; Yang, J.-J.; Jiang, Z.-L.; Zhao, G.-W.; Sun, L.; Wang, G.-H.; Li, X. Reduction of inflammatory responses by l-serine treatment leads to neuroprotection in mice after traumatic brain injury. Neuropharmacology 2015, 95, 1–11. [Google Scholar] [CrossRef]
- Imarisio, A.; Yahyavi, I.; Avenali, M.; Di Maio, A.; Buongarzone, G.; Galandra, C.; Picascia, M.; Filosa, A.; Gasparri, C.; Monti, M.C.; et al. Blood D-serine levels correlate with aging and dopaminergic treatment in Parkinson's disease. Neurobiol. Dis. 2024, 192, 106413. [Google Scholar] [CrossRef]
- Myint, S.M.M.P.; Sun, L.Y. L-serine: Neurological Implications and Therapeutic Potential. Biomedicines 2023, 11, 2117. [Google Scholar] [CrossRef]
- Basu, A.C.; E Tsai, G.; Ma, C.-L.; Ehmsen, J.T.; Mustafa, A.K.; Han, L.; I Jiang, Z.; A Benneyworth, M.; Froimowitz, M.P.; Lange, N.; et al. Targeted disruption of serine racemase affects glutamatergic neurotransmission and behavior. Mol. Psychiatry 2008, 14, 719–727. [Google Scholar] [CrossRef]
- Miya, K.; Inoue, R.; Takata, Y.; Abe, M.; Natsume, R.; Sakimura, K.; Hongou, K.; Miyawaki, T.; Mori, H. Serine racemase is predominantly localized in neurons in mouse brain. J. Comp. Neurol. 2008, 510, 641–654. [Google Scholar] [CrossRef]
- Benneyworth, M.A.; Li, Y.; Basu, A.C.; Bolshakov, V.Y.; Coyle, J.T. Cell Selective Conditional Null Mutations of Serine Racemase Demonstrate a Predominate Localization in Cortical Glutamatergic Neurons. Cell. Mol. Neurobiol. 2012, 32, 613–624. [Google Scholar] [CrossRef]
- Ehmsen, J.T.; Ma, T.M.; Sason, H.; Rosenberg, D.; Ogo, T.; Furuya, S.; Snyder, S.H.; Wolosker, H. D-Serine in Glia and Neurons Derives from 3-Phosphoglycerate Dehydrogenase. J. Neurosci. 2013, 33, 12464–12469. [Google Scholar] [CrossRef]
- Wolosker, H. Serine racemase and the serine shuttle between neurons and astrocytes. Biochim. et Biophys. Acta (BBA) - Proteins Proteom. 2011, 1814, 1558–1566. [Google Scholar] [CrossRef]
- S. Verleysdonk and B. Hamprecht, “Synthesis and release of L-serine by rat astroglia-rich primary cultures,” Glia, vol. 30, no. 1, pp. 19-26, Mar 2000.
- Sugishita, H.; Kuwabara, Y.; Toku, K.; Doi, L.; Yang, L.; Mitoma, J.; Furuya, S.; Hirabayashi, Y.; Maeda, N.; Sakanaka, M.; et al. L-Serine regulates the activities of microglial cells that express very low level of 3-phosphoglycerate dehydrogenase, an enzyme for L-serine biosynthesis. J. Neurosci. Res. 2001, 64, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Furuya, S.; Watanabe, M. Novel neuroglial and glioglial relationships mediated by L-serine metabolism. Arch. Histol. Cytol. 2003, 66, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Shimizu, H.; Koike, T.; Furuya, S.; Watanabe, M. Neutral Amino Acid Transporter ASCT1 Is Preferentially Expressed in l-Ser-Synthetic/Storing Glial Cells in the Mouse Brain with Transient Expression in Developing Capillaries. J. Neurosci. 2003, 23, 550–560. [Google Scholar] [CrossRef]
- Yamamoto, T.; Nishizaki, I.; Furuya, S.; Hirabayashi, Y.; Takahashi, K.; Okuyama, S.; Yamamoto, H. Characterization of rapid and high-affinity uptake of L-serine in neurons and astrocytes in primary culture. FEBS Lett. 2003, 548, 69–73. [Google Scholar] [CrossRef]
- Le Bail, M.; Martineau, M.; Sacchi, S.; Yatsenko, N.; Radzishevsky, I.; Conrod, S.; Ouares, K.A.; Wolosker, H.; Pollegioni, L.; Billard, J.-M.; et al. Identity of the NMDA receptor coagonist is synapse specific and developmentally regulated in the hippocampus. Proc. Natl. Acad. Sci. 2014, 112, 201416668–E213. [Google Scholar] [CrossRef]
- Mothet, J.; Le Bail, M.; Billard, J. Time and space profiling of NMDA receptor co-agonist functions. J. Neurochem. 2015, 135, 210–225. [Google Scholar] [CrossRef]
- Papouin, T.; Ladépêche, L.; Ruel, J.; Sacchi, S.; Labasque, M.; Hanini, M.; Groc, L.; Pollegioni, L.; Mothet, J.-P.; Oliet, S.H. Synaptic and Extrasynaptic NMDA Receptors Are Gated by Different Endogenous Coagonists. Cell 2012, 150, 633–646. [Google Scholar] [CrossRef]
- Henneberger, C.; Bard, L.; King, C.; Jennings, A.; Rusakov, D.A. NMDA Receptor Activation: Two Targets for Two Co-Agonists. Neurochem. Res. 2013, 38, 1156–1162. [Google Scholar] [CrossRef]
- Henneberger, C.; Papouin, T.; Oliet, S.H.R.; Rusakov, D.A. Long-term potentiation depends on release of d-serine from astrocytes. Nature 2010, 463, 232–236. [Google Scholar] [CrossRef]
- Mothet, J.-P.; Parent, A.T.; Wolosker, H.; Brady, R.O., Jr.; Linden, D.J.; Ferris, C.D.; Rogawski, M.A.; Snyder, S.H. D-Serine is an endogenous ligand for the glycine site of the N-methyl-D-aspartate receptor. Proc. Natl. Acad. Sci. USA 2000, 97, 4926–4931. [Google Scholar] [CrossRef]
- Radzishevsky, I.; Sason, H.; Wolosker, H. D-Serine. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Abdelrahman, H.A.; Al-Shamsi, A.; John, A.; Ali, B.R.; Al-Gazali, L. A Novel SLC1A4 Mutation (p.Y191*) Causes Spastic Tetraplegia, Thin Corpus Callosum, and Progressive Microcephaly (SPATCCM) With Seizure Disorder. Child Neurol. Open 2019, 6. [Google Scholar] [CrossRef] [PubMed]
- Conroy, J.; Allen, N.M.; Gorman, K.; O'Halloran, E.; Shahwan, A.; Lynch, B.; A Lynch, S.; Ennis, S.; King, M.D. Novel European SLC1A4 variant: infantile spasms and population ancestry analysis. J. Hum. Genet. 2016, 61, 761–764. [Google Scholar] [CrossRef]
- Heimer, G.; Marek-Yagel, D.; Eyal, E.; Barel, O.; Levi, D.O.; Hoffmann, C.; Ruzzo, E.; Ganelin-Cohen, E.; Lancet, D.; Pras, E.; et al. SLC1A4 mutations cause a novel disorder of intellectual disability, progressive microcephaly, spasticity and thin corpus callosum. Clin. Genet. 2015, 88, 327–335. [Google Scholar] [CrossRef]
- Mohamed, F.E.; Ghattas, M.A.; Almansoori, T.M.; Tabouni, M.; Baydoun, I.; Kizhakkedath, P.; John, A.; Alblooshi, H.; Shaukat, Q.; Al-Jasmi, F. Novel compound heterozygous variants (c.971delA/c.542C > T) in SLC1A4 causes spastic tetraplegia, thin corpus callosum, and progressive microcephaly: a case report and mutational analysis. Front. Pediatr. 2023, 11, 1183574. [Google Scholar] [CrossRef]
- Pironti, E.; Salpietro, V.; Cucinotta, F.; Granata, F.; Mormina, E.; Efthymiou, S.; Scuderi, C.; Gagliano, A.; Houlden, H.; Di Rosa, G. A novel SLC1A4 homozygous mutation causing congenital microcephaly, epileptic encephalopathy and spastic tetraparesis: a video-EEG and tractography – case study. J. Neurogenetics 2018, 32, 316–321. [Google Scholar] [CrossRef]
- Sarigecili, E.; Bulut, F.D.; Anlas, O. A rare cause of microcephaly, thin corpus callosum and refractory epilepsy due to a novel SLC1A4 gene mutation. Clin. Neurol. Neurosurg. 2022, 218, 107283. [Google Scholar] [CrossRef]
- Sedláčková, L.; Laššuthová, P.; Štěrbová, K.; Vlčková, M.; Kudr, M.; Buksakowska, I.; Staněk, D.; Seeman, P. Severe neurodevelopmental disorder with intractable seizures due to a novel SLC1A4 homozygous variant. Eur. J. Med Genet. 2021, 64, 104263. [Google Scholar] [CrossRef]
- Srour, M.; Hamdan, F.F.; Gan-Or, Z.; Labuda, D.; Nassif, C.; Oskoui, M.; Gana-Weisz, M.; Orr-Urtreger, A.; Rouleau, G.; Michaud, J. A homozygous mutation in SLC1A4 in siblings with severe intellectual disability and microcephaly. Clin. Genet. 2015, 88, E1–E4. [Google Scholar] [CrossRef]
- Pujol-Giménez, J.; Mirzaa, G.; Blue, E.E.; Albano, G.; Miller, D.E.; Allworth, A.; Bennett, J.T.; Byers, P.H.; Chanprasert, S.; Chen, J.; et al. Dominant-negative variant in SLC1A4 causes an autosomal dominant epilepsy syndrome. Ann. Clin. Transl. Neurol. 2023, 10, 1046–1053. [Google Scholar] [CrossRef]
- Skarnes, W.C.; Rosen, B.; West, A.P.; Koutsourakis, M.; Bushell, W.; Iyer, V.; Mujica, A.O.; Thomas, M.; Harrow, J.; Cox, T.; et al. A conditional knockout resource for the genome-wide study of mouse gene function. Nature 2011, 474, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Arizanovska, D.; Emodogo, J.A.; Lally, A.P.; Palavicino-Maggio, C.B.; Liebl, D.J.; Folorunso, O.O. Cross species review of the physiological role of d-serine in translationally relevant behaviors. Amino Acids 2023, 55, 1501–1517. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Sagata, N.; Takeuchi, N.; Tanaka, M.; Ninomiya, H.; Iwata, N.; Ozaki, N.; Shibata, H.; Fukumaki, Y. Association study of polymorphisms in the neutral amino acid transporter genes SLC1A4, SLC1A5 and the glycine transporter genes SLC6A5, SLC6A9with schizophrenia. BMC Psychiatry 2008, 8, 58–58. [Google Scholar] [CrossRef]
- Okubo, R.; Okada, M.; Motomura, E. Dysfunction of the NMDA Receptor in the Pathophysiology of Schizophrenia and/or the Pathomechanisms of Treatment-Resistant Schizophrenia. Biomolecules 2024, 14, 1128. [Google Scholar] [CrossRef]
- S. I. Deutsch, J. S. I. Deutsch, J. Mastropaolo, B. L. Schwartz, R. B. Rosse, and J. M. Morihisa, “A “glutamatergic hypothesis” of schizophrenia. Rationale for pharmacotherapy with glycine,” Clin Neuropharmacol, vol. 12, no. 1, pp. 1-13, Feb 1989.
- Coon, H.; Myles-Worsley, M.; Tiobech, J.; Hoff, M.; Rosenthal, J.; Bennett, P.; Reimherr, F.; Wender, P.; Dale, P.; Polloi, A.; et al. Evidence for a chromosome 2p13–14 schizophrenia susceptibility locus in families from Palau, Micronesia. Mol. Psychiatry 1998, 3, 521–527. [Google Scholar] [CrossRef]
- E Straub, R.; MacLean, C.J.; Ma, Y.; Webb, B.T.; Myakishev, M.V.; Harris-Kerr, C.; Wormley, B.; Sadek, H.; Kadambi, B.; A O'Neill, F.; et al. Genome-wide scans of three independent sets of 90 Irish multiplex schizophrenia families and follow-up of selected regions in all families provides evidence for multiple susceptibility genes. Mol. Psychiatry 2002, 7, 542–559. [Google Scholar] [CrossRef]
- Halberstadt, A.L. The Phencyclidine-Glutamate Model of Schizophrenia. Clin. Neuropharmacol. 1995, 18, 237–249. [Google Scholar] [CrossRef]
- Krystal, J.H.; Karper, L.P.; Seibyl, J.P.; Freeman, G.K.; Delaney, R.; Bremner, J.D.; Heninger, G.R.; Bowers, M.B., Jr.; Charney, D.S. Subanesthetic effects of the noncompetitive NMDA antagonist, ketamine, in humans: Psychotomimetic, perceptual, cognitive, and neuroendocrine responses Arch. Gen. Psychiatry 1994, 51, 199–214. [CrossRef]
- Skowronek, M.H.; Georgi, A.; Jamra, R.A.; Schumacher, J.; Becker, T.; Schmael, C.; Paul, T.; Deschner, M.; Höfels, S.; Wulff, M.; et al. No association between genetic variants at the ASCT1 gene and schizophrenia or bipolar disorder in a German sample. Psychiatr. Genet. 2006, 16, 233–234. [Google Scholar] [CrossRef]
- Cho, S.-E.; Na, K.-S.; Cho, S.-J.; Kang, S.G. Low d-serine levels in schizophrenia: A systematic review and meta-analysis. 634, 51. [CrossRef]
- Guan, J.; Cai, J.J.; Ji, G.; Sham, P.C. Commonality in dysregulated expression of gene sets in cortical brains of individuals with autism, schizophrenia, and bipolar disorder. Transl. Psychiatry 2019, 9, 1–15. [Google Scholar] [CrossRef]
- Hardingham, G.E.; Bading, H. Synaptic versus extrasynaptic NMDA receptor signalling: implications for neurodegenerative disorders. Nat. Rev. Neurosci. 2010, 11, 682–696. [Google Scholar] [CrossRef] [PubMed]
- Orzylowski, M.; Fujiwara, E.; Mousseau, D.D.; Baker, G.B. An Overview of the Involvement of D-Serine in Cognitive Impairment in Normal Aging and Dementia. Front. Psychiatry 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.G.; Zukin, R.S. NMDA receptor trafficking in synaptic plasticity and neuropsychiatric disorders. Nat. Rev. Neurosci. 2007, 8, 413–426. [Google Scholar] [CrossRef]
- Nava-Gómez, L.; Calero-Vargas, I.; Higinio-Rodríguez, F.; Vázquez-Prieto, B.; Olivares-Moreno, R.; Ortiz-Retana, J.; Aranda, P.; Hernández-Chan, N.; Rojas-Piloni, G.; Alcauter, S.; et al. Aging-Associated Cognitive Decline is Reversed by D-Serine Supplementation. eneuro 2022, 9. [Google Scholar] [CrossRef]
- 2024 Alzheimer's disease facts and figures. Alzheimer's Dement. 2024, 20, 3708–3821. [CrossRef]
- Le Douce, J.; Maugard, M.; Veran, J.; Matos, M.; Jégo, P.; Vigneron, P.-A.; Faivre, E.; Toussay, X.; Vandenberghe, M.; Balbastre, Y.; et al. Impairment of Glycolysis-Derived l-Serine Production in Astrocytes Contributes to Cognitive Deficits in Alzheimer’s Disease. 2020, 31, 503–517. e8. [CrossRef]
- Madeira, C.; Lourenco, M.V.; Vargaslopes, C.; Suemoto, C.K.; Brandao, C.O.; Reis, T.; Leite, R.E.P.; Laks, J.; Jacobfilho, W.; Pasqualucci, C.A.; et al. d-serine levels in Alzheimer’s disease: implications for novel biomarker development. Transl. Psychiatry 2015, 5, e561–e561. [Google Scholar] [CrossRef]
- Ni, X.; Inoue, R.; Wu, Y.; Yoshida, T.; Yaku, K.; Nakagawa, T.; Saito, T.; Saido, T.C.; Takao, K.; Mori, H. Regional contributions of D-serine to Alzheimer’s disease pathology in male AppNL–G–F/NL–G–F mice. Front. Aging Neurosci. 2023, 15, 1211067. [Google Scholar] [CrossRef]
- Piubelli, L.; Pollegioni, L.; Rabattoni, V.; Mauri, M.; Cariddi, L.P.; Versino, M.; Sacchi, S. Serum d-serine levels are altered in early phases of Alzheimer’s disease: towards a precocious biomarker. Transl. Psychiatry 2021, 11, 1–8. [Google Scholar] [CrossRef]
- Puris, E.; Saveleva, L.; Maciel, I.d.S.; Kanninen, K.M.; Auriola, S.; Fricker, G. Protein Expression of Amino Acid Transporters Is Altered in Isolated Cerebral Microvessels of 5xFAD Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2022, 60, 732–748. [Google Scholar] [CrossRef]
- Ito, S.; Yagi, R.; Ogata, S.; Masuda, T.; Saito, T.; Saido, T.; Ohtsuki, S. Proteomic alterations in the brain and blood–brain barrier during brain Aβ accumulation in an APP knock-in mouse model of Alzheimer’s disease. Fluids Barriers CNS 2023, 20, 1–18. [Google Scholar] [CrossRef]
- Clayton, D.A.; Mesches, M.H.; Alvarez, E.; Bickford, P.C.; Browning, M.D. A Hippocampal NR2B Deficit Can Mimic Age-Related Changes in Long-Term Potentiation and Spatial Learning in the Fischer 344 Rat. J. Neurosci. 2002, 22, 3628–3637. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, T.; Punzo, D.; Devoto, P.; Rosini, E.; Paciotti, S.; Sacchi, S.; Li, Q.; Thiolat, M.-L.; Véga, C.; Carella, M.; et al. The levels of the NMDA receptor co-agonist D-serine are reduced in the substantia nigra of MPTP-lesioned macaques and in the cerebrospinal fluid of Parkinson’s disease patients. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Campanelli, F.; Natale, G.; Marino, G.; Ghiglieri, V.; Calabresi, P. Striatal glutamatergic hyperactivity in Parkinson's disease. Neurobiol. Dis. 2022, 168, 105697. [Google Scholar] [CrossRef]
- Cenci, M.A.; Skovgård, K.; Odin, P. Non-dopaminergic approaches to the treatment of motor complications in Parkinson's disease. Neuropharmacology 2022, 210, 109027. [Google Scholar] [CrossRef]
- Gardoni, F.; Di Luca, M. Targeting glutamatergic synapses in Parkinson's disease. Curr. Opin. Pharmacol. 2015, 20, 24–28. [Google Scholar] [CrossRef]
- Di Maio, A.; Nuzzo, T.; Gilio, L.; Serra, M.; Buttari, F.; Errico, F.; De Rosa, A.; Bassi, M.S.; Morelli, M.; Sasabe, J.; et al. Homeostasis of serine enantiomers is disrupted in the post-mortem caudate putamen and cerebrospinal fluid of living Parkinson's disease patients. Neurobiol. Dis. 2023, 184, 106203. [Google Scholar] [CrossRef]
- Goh, K.K.; Wu, T.-H.; Chen, C.-H.; Lu, M.-L. Efficacy of N-methyl-D-aspartate receptor modulator augmentation in schizophrenia: A meta-analysis of randomised, placebo-controlled trials. J. Psychopharmacol. 2021, 35, 236–252. [Google Scholar] [CrossRef]
- Sehatpour, P.; Iosifescu, D.V.; De Baun, H.M.; Shope, C.; Mayer, M.R.; Gangwisch, J.; Dias, E.; Sobeih, T.; Choo, T.-H.; Wall, M.M.; et al. Dose-Dependent Augmentation of Neuroplasticity-Based Auditory Learning in Schizophrenia: A Double-Blind, Placebo-Controlled, Randomized, Target Engagement Clinical Trial of the NMDA Glutamate Receptor Agonist d-serine. Biol. Psychiatry 2023, 94, 164–173. [Google Scholar] [CrossRef]
- Wolosker, H.; Balu, D.T. d-Serine as the gatekeeper of NMDA receptor activity: implications for the pharmacologic management of anxiety disorders. Transl. Psychiatry 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Otte, D.-M.; de Arellano, M.L.B.; Bilkei-Gorzo, A.; Albayram, Ö.; Imbeault, S.; Jeung, H.; Alferink, J.; Zimmer, A. Effects of Chronic D-Serine Elevation on Animal Models of Depression and Anxiety-Related Behavior. PLOS ONE 2013, 8, e67131. [Google Scholar] [CrossRef]
- Malkesman, O.; Austin, D.R.; Tragon, T.; Wang, G.; Rompala, G.; Hamidi, A.B.; Cui, Z.; Young, W.S.; Nakazawa, K.; Zarate, C.A.; et al. Acute d-serine treatment produces antidepressant-like effects in rodents. Int. J. Neuropsychopharmacol. 2011, 15, 1135–1148. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Calandrelli, R.; Girardini, J.; Yan, Z.; Tan, Z.; Xu, X.; Hiniker, A.; Zhong, S. PHGDH expression increases with progression of Alzheimer’s disease pathology and symptoms. Cell Metab. 2022, 34, 651–653. [Google Scholar] [CrossRef]
- Gelfin, E.; Kaufman, Y.; Korn-Lubetzki, I.; Bloch, B.; Kremer, I.; Javitt, D.C.; Heresco-Levy, U. D-serine adjuvant treatment alleviates behavioural and motor symptoms in Parkinson's disease. Int. J. Neuropsychopharmacol. 2012, 15, 543–549. [Google Scholar] [CrossRef]
- Lyda, B.R.; Leary, G.P.; Farnsworth, J.; Seaver, B.; Silvius, D.; Kavanaugh, M.P.; Esslinger, C.S.; Natale, N.R. Discovery and Synthesis of Hydroxy-l-Proline Blockers of the Neutral Amino Acid Transporters SLC1A4 (ASCT1) and SLC1A5 (ASCT2). Molecules 2024, 29, 2330. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



