1. Introduction
Elastic fibers play a critical role in the expansion and contraction of the lung during the respiratory cycle [
1,
2]. They support the architecture of alveolar walls, helping to maintain their shape during expansion and contraction. The degradation of these fibers leads to airflow obstruction, causing hyperinflation of the lung and rupture of alveolar walls. As the elastic fiber network deteriorates, it may also stimulate an inflammatory response mediated by the release of elastin peptides from fragmented elastic fibers [
3].
The proinflammatory effect of the peptides is mediated by their binding to the elastin receptor complex, which plays an important role in multiple biological processes ranging from tissue development and remodeling to cellular signaling pathways that govern cell migration and proliferation [
6]. Structural studies using NMR spectroscopy and other techniques have provided insights into the conformational changes that occur upon binding, shedding light on the dynamics of the peptide-receptor interaction [
7,
8,
9]. Elastin peptides with specific amino acid sequences, such as Val-Gly-Val-Ala-Pro-Gly, exhibit distinct bioactive properties and have been shown to stimulate the chemotaxis of immune cells, particularly neutrophils and macrophages [
7,
10]. This process amplifies the inflammatory response within the lung environment, increasing the production of elastin peptides and resulting in a self-perpetuating cycle of inflammation and tissue destruction.
Based on recent experimental findings, we hypothesize that the advancement of emphysematous changes in the lung involves the dissemination of these peptides through an inflammatory cell population, resulting in the breakdown of elastic fibers, increased mechanical strain on alveolar walls, and airspace enlargement. As in the case of an epidemic, elastin peptides act as a vector for the spread of a self-replicating amino acid sequence responsible for disease propagation. This concept could provide a rationale for developing a treatment that inhibits the spread of these peptides in the lung, thereby slowing the progression of alveolar wall injury and reducing the risk of respiratory failure.
2. Role of Elastin in the Pathogenesis of Pulmonary Emphysema
2.1. Mechanical Properties of Elastic Fibers
The primary component of elastic fibers is the distensible elastin protein, which stores energy during lung expansion [
11,
12]. Elastin lacks the rigid, crystalline structure typical of other structural proteins like collagen, allowing it to stretch significantly without losing its structural integrity. This elasticity is primarily due to a unique amino acid composition rich in glycine, valine, and proline, enabling the protein to adopt flexible conformations. When elastin stretches during inhalation, the decrease in entropy is associated with energy storage. Conversely, returning to a more disordered state during exhalation releases the force needed to expel air from the lungs [
13]. These changes in entropy are facilitated by the hydrophobic regions of elastin, which interact with neighboring water molecules [
14].
2.2. Modeling the Role of Elastic Fiber Injury in Pulmonary Emphysema
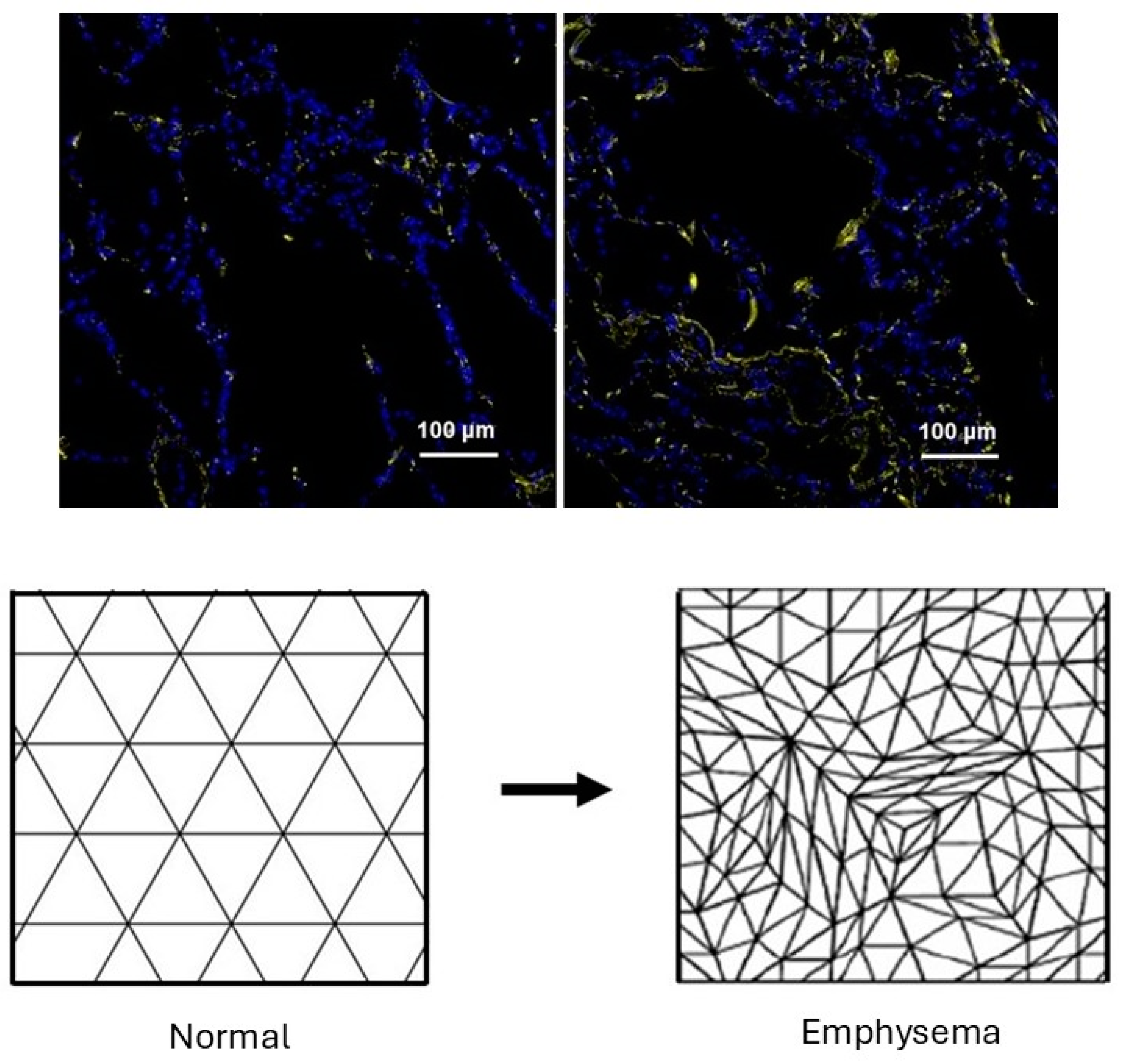
The initial changes associated with the development of pulmonary emphysema involve only localized damage to elastic fibers. As the structural alterations in these fibers progress, they disrupt the normal distribution of mechanical forces, causing the rupture of alveolar walls [
15]. This process may be modeled using percolation theory, which analyzes the movement of fluids through interconnected channels [
16]. A particular percolation model, the random resistor network, may be utilized to investigate how changes in elastic fibers influence lung mechanics [
17]. It involves the indiscriminate disconnection of conducting bonds, resulting in a decreased flow of electrical current. This process is analogous to the impaired transmission of mechanical forces through fragmented elastic fibers. It may be modeled by constructing a network composed of two interconnected units, K1 and K2, representing intact and disintegrating elastic fibers, respectively [
18]. These components are randomly arranged within a three-dimensional lattice, and their relative proportions significantly impact mechanical force transmission throughout the lung.
When the proportion of K2 units is low, the forces are evenly distributed across the K1 units, causing minimal changes in lung structure. However, as the number of K2 units increases due to elastolysis, the active transmission of mechanical forces in the lung becomes concentrated in the remaining K1 units. The additional strain on the K1 units promotes their transformation to K2 units, resulting in diminished elastic recoil, hyperinflation of the lung, and the rupture of alveolar walls (
Figure 1).
2.3. Role of Elastin Peptides in the Pathogenesis of Pulmonary Emphysema
Early investigations conducted in the 1970s began to elucidate the connection between elastin degradation and the pathophysiology of emphysema. It was shown that proteolytic enzymes released by neutrophils and macrophages play a critical role in alveolar wall injury, laying the groundwork for subsequent research efforts focused on understanding the complex interactions between inflammatory responses and the structural integrity of the lung [
19,
20,
21,
22]. Elastolytic enzymes produced by these cells, including matrix metalloproteinases (MMPs) and serine proteases, are central to the pathogenesis of pulmonary emphysema [
23,
24].
These enzymes are synthesized in response to lung irritants like tobacco smoke and result in the generation of elastin peptide fragments that are critical mediators of inflammation in pulmonary emphysema. These peptides have been shown to interact with an elastin receptor complex that activates immune responses [
25]. The binding of elastin peptides to their receptors activates various intracellular signaling pathways, causing the upregulation of proinflammatory cytokines, further exacerbating lung damage [
26].
Beyond attracting inflammatory cells, elastin peptides play an important role in lung remodeling by facilitating cell proliferation and extracellular matrix remodeling. By promoting the activation of fibroblasts and the secretion of elastases and other enzymes, these peptides can produce changes in matrix composition that adversely affect the mechanical properties of alveolar walls and contribute to the pathogenesis of emphysema [
3,
27]. Studies showed that intratracheal administration of elastin peptides in mice can induce alveolar wall inflammation, significantly increasing alveolar diameter compared to controls [
28]. Elastin peptide-treated mice also demonstrated increased recruitment of neutrophils and macrophages, causing further degradation of elastic fibers [
29].
The proinflammatory activity of these peptides is associated with their binding to the elastin binding protein, a 67 kDa component of the elastin receptor complex [
30]. Before the emergence of pulmonary emphysema, the fragmentation and unraveling of elastic fibers may expose the core elastin protein to enzymatic and oxidative breakdown, facilitating the release of proinflammatory peptides [
31].
To investigate this hypothesis, we developed a hamster model of pulmonary emphysema using elastase and LPS to examine the relationship between lung inflammation and damage to elastic fibers [
10]. The combination of these agents was informed by prior research showing their synergistic interaction in promoting airspace enlargement [
32]. To enhance the impact of LPS, a single low dose of elastase was given to the hamsters with a shorter interval between the enzyme instillation and LPS administration, thereby improving the detection of potential synergistic effects. In contrast to earlier studies that employed multiple elastase treatments over several weeks before administering LPS, this revised model concentrated on a single dose and minimized the timeframe between the two treatments [
33].
This model was used to assess whether pretreatment with elastase altered the structure of elastic fibers, making them more vulnerable to subsequent injury from LPS. The findings revealed that hamsters treated with elastase and LPS exhibited significantly higher levels of cells in bronchoalveolar lavage fluid (BALF) compared to those treated with elastase and saline, saline and LPS, or the control group. Furthermore, the proportion of neutrophils in the BALF was significantly greater in the animals receiving both elastase and LPS, in contrast to those treated with either elastase or LPS alone [
10].
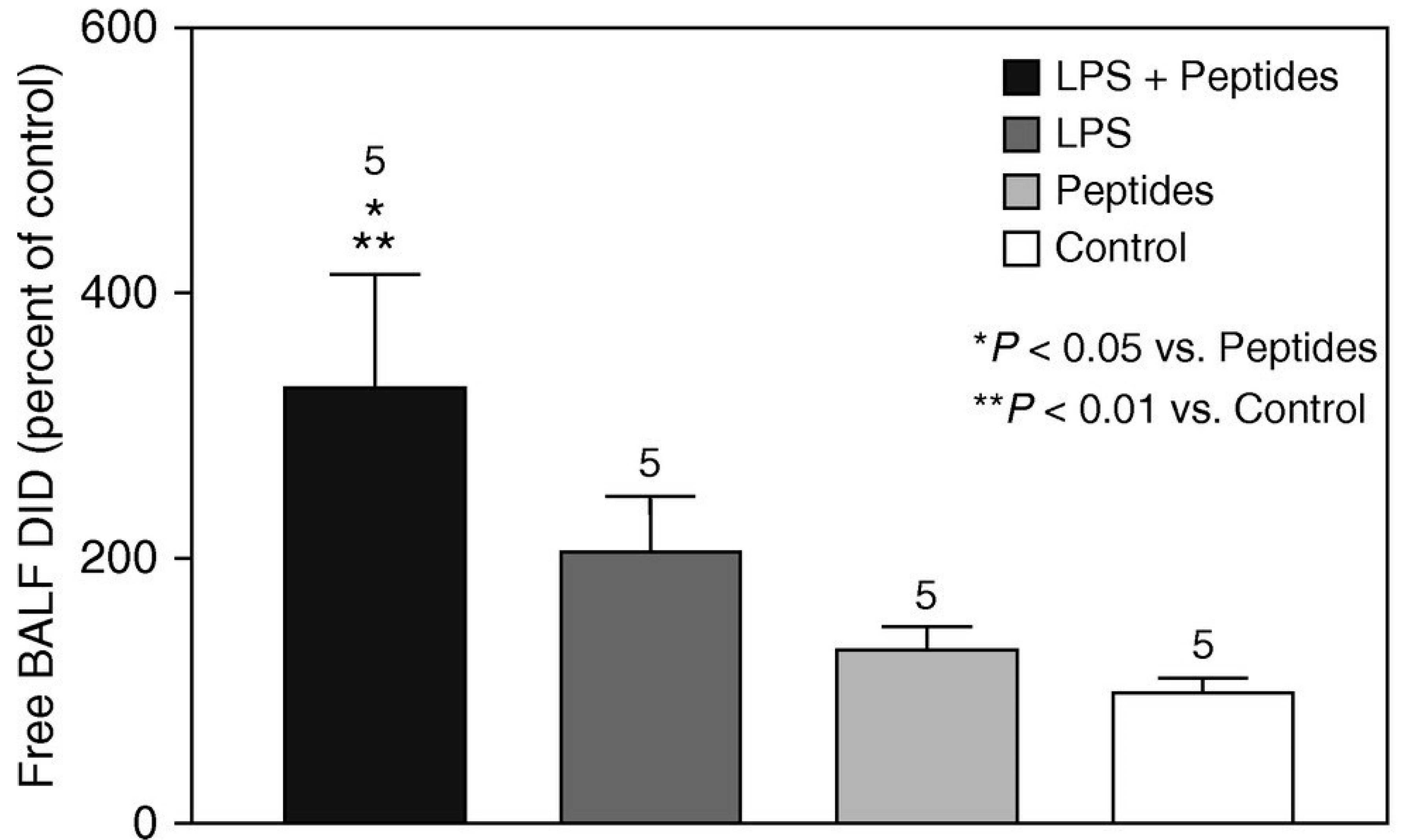
The proinflammatory effects of elastin peptides released from damaged elastic fibers were investigated in a lung injury model induced by LPS. When elastin peptides and LPS were administered together via intratracheal instillation, there was a marked increase in the levels of neutrophils and markers of inflammation in bronchoalveolar lavage fluid (BALF) compared to the administration of either agent on its own [
10]. The proinflammatory effect of the peptides was also reflected by an increase in elastolysis, as measured by BALF desmosine levels (
Figure 2).
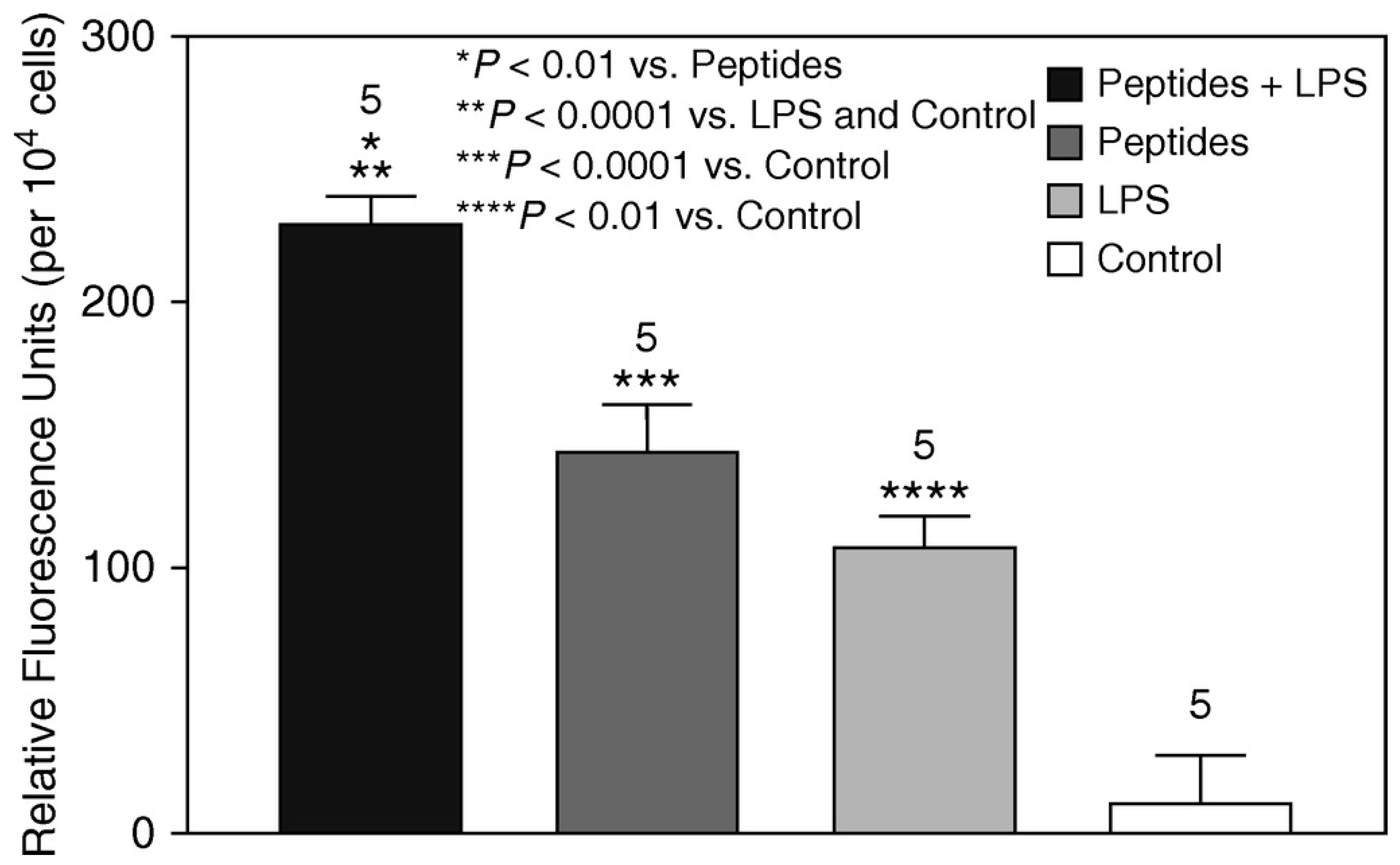
In vitro experiments were conducted using BALF macrophages from untreated animals to determine the chemotactic properties of elastin peptides [
10]. While exposure to elastin peptides or LPS alone significantly increased chemotaxis compared to the control group, combining the two agents produced an even more significant enhancement in chemotactic response (
Figure 3).
3. Elastin Peptides as a Vector in the Transmission of Alveolar Wall Injury
3.1. Emergent Behavior: A Fundamental Feature of Epidemics and Pulmonary Emphysema
While the relationship between excess protease activity and alveolar wall injury plays a central role in pulmonary emphysema, other mechanisms may be more directly responsible for the morphologic abnormalities associated with the disease. Changes in the distribution of mechanical forces may be necessary for converting proteolytic injury into airspace enlargement [
34,
35]. This hypothesis is supported by in-silico studies showing that local variations in alveolar wall elasticity evolve into global morphological alterations that resemble those seen in pulmonary emphysema [
36].
This finding is consistent with the principle of emergence in which complex interactions at different levels of scale produce spontaneous reorganization of chemical and physical systems [
37,
38]. An example of an emergent phenomenon is an epidemic, in which the transmission of the disease vector depends on the unpredictable interaction of various factors, such as population density and mobility [
39,
40]. The incidence of infection may remain uncertain until it involves a relatively large population.
The progression of pulmonary emphysema may incorporate analogous mechanisms, where indeterminate events regulate the transition from normal to enlarged airspaces. The lung remodeling cannot be predicted by analyzing individual components such as elastase activity or antiprotease levels. Instead, it may require the identification of specific patterns of molecular and macroscopic behavior that are part of a self-organizing process at multiple scale levels [
41].
3.2. Experimental Evidence for an Epidemic-Like Mechanism in Pulmonary Emphysema
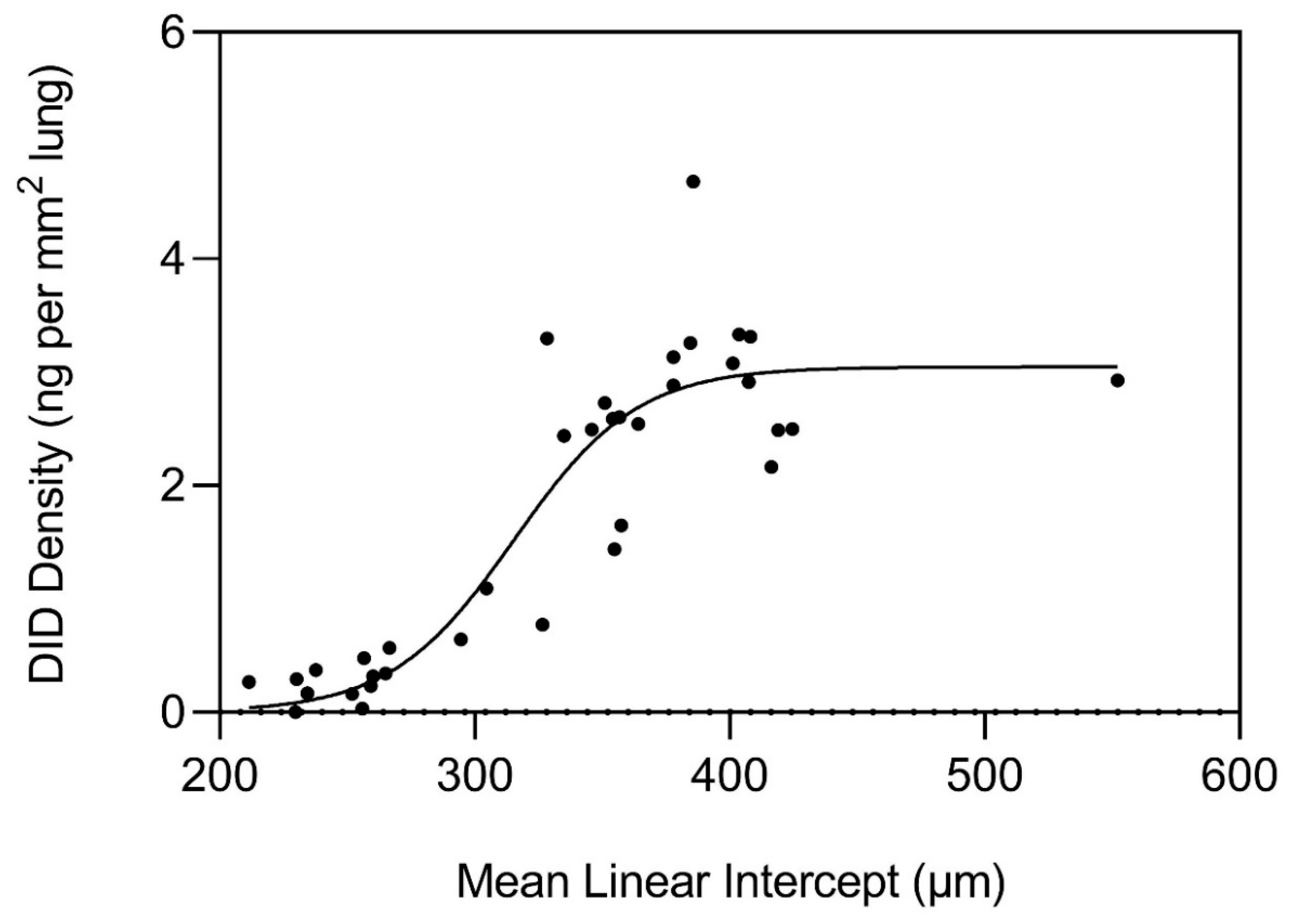
Measurements of elastin crosslinks in postmortem lungs from COPD patients provide evidence that an epidemic-like mechanism is responsible for elastic fiber injury and repair in pulmonary emphysema [
42]. The main parameter used to evaluate “infection” is structural alterations in elastic fibers that result in the dysfunctional transmission of mechanical forces that induce airspace distention. As shown in
Figure 4, desmosine crosslink density in lung tissue sections exponentially increases when the alveolar diameter exceeds 300 µm and plateaus beyond 400 µm. This finding is consistent with a repair process that initially counteracts alveolar wall injury and subsequently undergoes a decompensatory phase involving more widespread transmission of uneven mechanical forces that induce alveolar wall rupture. The increase in crosslinking is also accompanied by abnormal elastin deposition that further impairs energy storage by elastic fibers (
Figure 5).
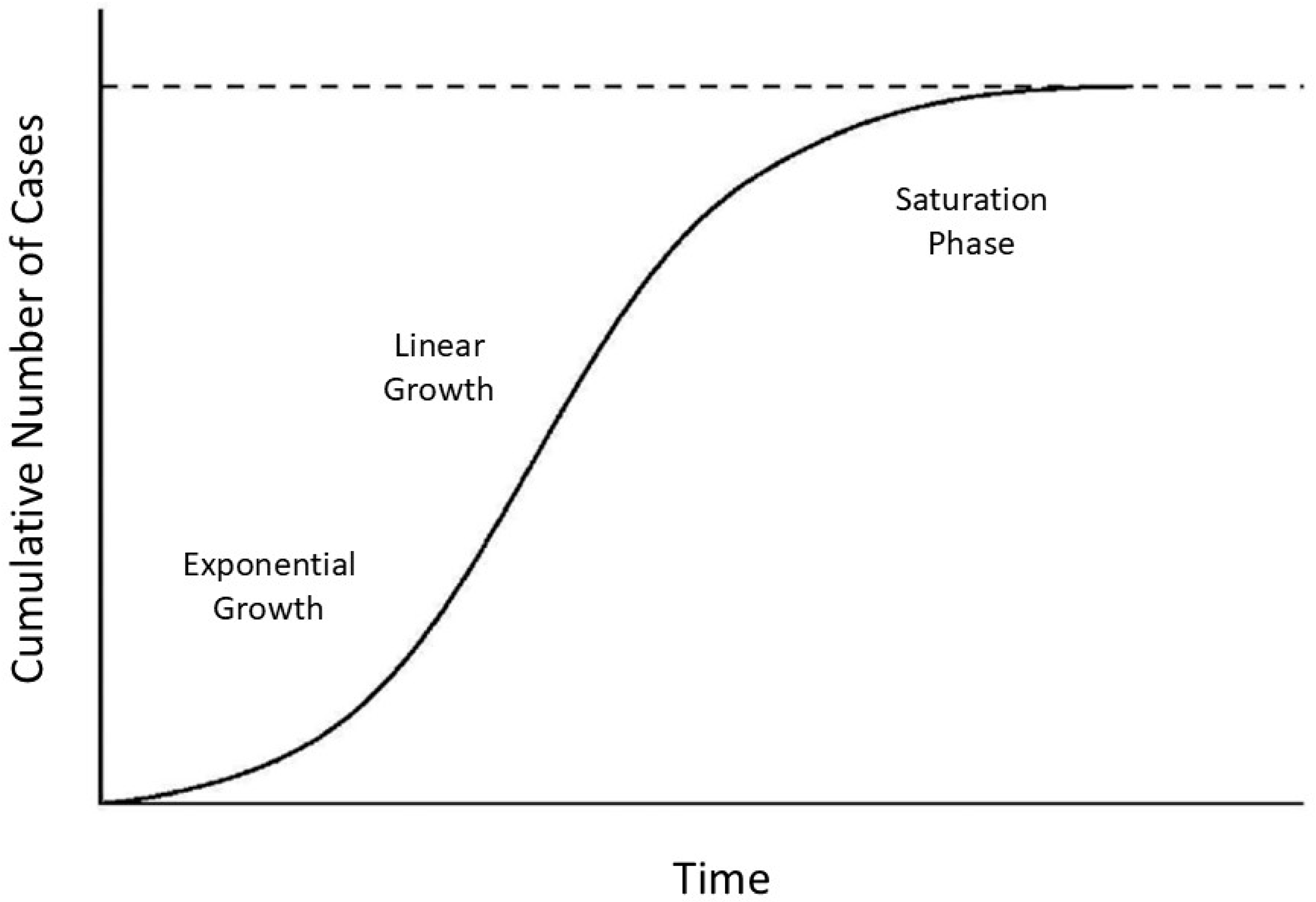
The shape of the crosslink density curve is similar to that of a communicable infection, where the cumulative number of cases is characterized by exponential, linear, and saturation phases (
Figure 6) [
43]. As in the case of an epidemic, the increase in dysfunctional elastic fibers results in a phase transition to an organized disease state reflected at increasing levels of scale by molecular changes in the extracellular matrix, microscopic airspace enlargement, and a loss of physiological function.
We hypothesize that the spread of airspace enlargement through the lung requires a morphological landscape composed of a diffuse population of inflammatory cells subject to activation by the spread of elastin peptides that act as a vector for the progression of alveolar wall injury. This mechanism involves increasing elastic fiber injury that accelerates the release of elastin peptides, resulting in a self-perpetuating disease mechanism that contains the features of an epidemic.
An important feature of this process is its potential synergistic interaction with the uneven mechanical forces responsible for airspace distention and rupture [
34]. The combination of elastin peptide-induced elastic fiber injury and loss of elastic recoil results in the progressive mechanical failure of alveolar walls and the further development of airspace enlargement. The continued spread of emphysematous changes eventually undergoes a phase transition to an active disease state involving a reorganization of lung architecture less amenable to therapeutic intervention.
4. Therapeutic Considerations
4.1. Leveraging the Relationship Between Pulmonary Emphysema and Epidemics
Similar to physical systems, epidemics can undergo phase transitions where small changes in parameters, such as contact rates, can lead to abrupt changes in the outbreak dynamics, from endemic to epidemic or extinction [
39]. Spatial dynamics also influence epidemic spread. The movement of people in geographic spaces leads to random spread and the formation of "hot spots" where the incidence of the disease is particularly high. Models incorporating spatial dynamics often exhibit complex emergent patterns arising from simple local rules about movement and transmission [
44,
45].
Both elastin peptide-induced lung injury and infectious epidemics are characterized by similar phases: initiation, exponential growth, stabilization, and resolution [
43]. Initial exposure leads to localized responses that can escalate into widespread damage. Initially protective, an overactive inflammatory response results in tissue damage in both lung injuries and uncontrolled infections. Understanding these similarities could lead to preventive strategies, promoting therapeutic intervention before a phase transition involving irreversible lung injury.
4.2. Aerosolized Hyaluronan: A Potential Treatment for the Emergent Properties of Pulmonary Emphysema
The potential significance of elastin-derived peptides indicates that decreasing their levels in the lungs could mitigate the progression of the disease. While most current therapeutic strategies for treating pulmonary emphysema have primarily concentrated on elastase inhibitors, our laboratory has explored using an aerosolized formulation containing low molecular weight hyaluronan (HA), a long-chain polysaccharide. Previous research demonstrated that pretreatment with hyaluronidase exacerbates airspace enlargement in an emphysema model induced by intratracheal elastase, whereas animals pretreated with HA exhibited significantly reduced airspace enlargement in emphysema models induced by either elastase or cigarette smoke [
46,
47]. This protective effect arises from HA's ability to bind to elastic fibers, acting as a physical barrier against various agents that degrade elastin, although it does not function as an elastase inhibitor. The therapeutic potential of enhancing the extracellular matrix with exogenously administered HA is strengthened by a study showing significantly lower lung HA levels in patients with pulmonary emphysema resulting from alpha-1 antiprotease deficiency.
The clinical efficacy of HA was assessed in a 28-day trial using levels of peptide-free desmosine in urine as a biomarker of elastin degradation in patients with pulmonary emphysema due to alpha-1 antiprotease deficiency. Inhalation of this agent twice daily significantly reduced urine desmosine levels throughout the trial. This finding suggests that aerosolized HA can reduce the release of elastin peptides and slow the progression of alveolar wall injury. Furthermore, HA's hydrophilic properties may also increase energy storage in elastin, thereby mitigating the mechanical strain that contributes to the fragmentation of elastic fibers. This concept is supported by a study indicating that HA and other proteoglycans reduce the uneven distribution of forces in the extracellular matrix [
49].
The potential role of HA in multiple mechanisms of lung injury provides a rationale for developing drugs that inhibit the broader process of disease emergence rather than individual components of the inflammatory reaction associated with airspace enlargement. Aside from alpha-1 antiprotease augmentation in a small subgroup with genetically induced pulmonary emphysema, elastase inhibitors and other anti-inflammatory agents have shown limited success in treating the disease [
50,
51,
52,
53]. This finding may be due to the complexity of emergent phenomena, where the reorganization of a system depends on numerous interactions at different levels of scale rather than the elementary properties of the constituents. Consequently, the loss of specific molecular components of lung injury could be circumvented by the higher-level effects of structural alterations in alveolar walls.
4.3. Targeting Convergence Points in the Pathogenesis of Pulmonary Emphysema
Since the release of elastin peptides reflects a number of pathogenetic mechanisms, it may represent a point in the emergence of pulmonary emphysema where a group of agents interact concurrently to form a converging whole. Blocking the effects of these peptides by using antibodies to prevent their attachment to elastin receptor complexes or chemically modifying these receptors or their elastin binding protein component could provide alternative approaches to therapeutic intervention.
Pharmacological agents that selectively inhibit signaling through the ERC or modulate its interaction with elastin are currently under investigation. Potential drug candidates include small molecules that target ERC-associated pathways, monoclonal antibodies that disrupt ERC-elastin interactions, and gene therapy approaches aimed at downregulating ERC expression or activity [
30].
While preclinical data highlight the potential of targeting the ERC for treating pulmonary emphysema, challenges remain in translating these findings into clinical practice. Safety profiles, pharmacokinetics, and patient variability need careful evaluation through well-designed clinical trials. Identifying reliable biomarkers to predict response to ERC-targeting therapies will be crucial for patient stratification and optimizing treatment outcomes. Future studies should focus on developing biomarker panels associated with ERC signaling pathways.
Considering the complex nature of pulmonary emphysema, a multi-modal approach combining ERC inhibitors with other proposed treatments, such as aerosolized HA, may produce synergistic interactions that greatly enhance therapeutic efficacy. This strategy may be particularly suited to the complexity of emergent processes involved in the pathogenesis of this disease.
5. Conclusions
The studies discussed in the current paper support the role of elastin-derived peptides in the pathogenesis of pulmonary emphysema. These peptides interact with specific receptors, initiating signaling events promoting inflammation and elastic fiber damage. The resulting airspace enlargement correlates with an increase in elastin crosslink density, and the graph of that relationship corresponds to that associated with the transmission of a communicable disease.
While this finding suggests that elastin peptides act as a vector in propagating alveolar wall injury, further studies are warranted to validate this concept, including clinical trials that determine the therapeutic effects of inhibiting elastin peptide activity. This treatment approach could slow the progression of pulmonary emphysema and significantly lower the risk of respiratory failure.
References
- Mecham RP. Elastin in lung development and disease pathogenesis. Matrix Biol. (2018) 73:6–20. [CrossRef]
- Hogg JC, Timens W. The pathology of chronic obstructive pulmonary disease.
- Heinz A. Elastases and elastokines: elastin degradation and its significance in health and disease. Crit Rev Biochem Mol Biol. 2020 Jun;55(3):252-273. Epub 2020 Jun 12. [CrossRef] [PubMed]
- Fulop T, Khalil A, Larbi A. The role of elastin peptides in modulating the immune response in aging and age-related diseases. Pathol Biol (Paris). 2012 Feb;60(1):28-33. Epub 2011 Nov 17. [CrossRef] [PubMed]
- Lahire S, Fichel C, Rubaszewski O, Lerévérend C, Audonnet S, Visneux V, Perotin JM, Deslée G, Le Jan S, Potteaux S, Le Naour R, Pommier A. Elastin-derived peptides favor type 2 innate lymphoid cells in chronic obstructive pulmonary disease. Am J Physiol Lung Cell Mol Physiol. 2024 Jun 1;326(6):L812-L820. Epub 2024 May 7. [CrossRef] [PubMed]
- Houghton, A. M. (2015). The role of elastin-derived peptides in pulmonary inflammation and emphysema. *Nature Reviews Pulmonology*, 11(3), 149-158.
- Castiglione Morelli MA, Bisaccia F, Spisani S, De Biasi M, Traniello S, Tamburro AM. Structure-activity relationships for some elastin-derived peptide chemoattractants. J Pept Res. 1997 Jun;49(6):492-9. [CrossRef] [PubMed]
- Hernández B, Crowet JM, Thiery J, Kruglik SG, Belloy N, Baud S, Dauchez M, Debelle L. Structural Analysis of Nonapeptides Derived from Elastin. Biophys J. 2020 Jun 2;118(11):2755-2768. Epub 2020 Apr 25. [CrossRef] [PubMed] [PubMed Central]
- López Barreiro D, Minten IJ, Thies JC, Sagt CMJ. Structure-Property Relationships of Elastin-like Polypeptides: A Review of Experimental and Computational Studies. ACS Biomater Sci Eng. 2023 Jul 10;9(7):3796-3809. Epub 2021 Jul 12. [CrossRef] [PubMed]
- Mehraban S, Gu G, Ma S, Liu X, Turino G, Cantor J. The Proinflammatory Activity of Structurally Altered Elastic Fibers. Am J Respir Cell Mol Biol. 2020 Nov;63(5):699-706. [CrossRef] [PubMed]
- Depenveiller C, Baud S, Belloy N, Bochicchio B, Dandurand J, Dauchez M, Pepe A, Pomès R, Samouillan V, Debelle L. Structural and physical basis for the elasticity of elastin. Q Rev Biophys. 2024 Mar 19;57:e3. [CrossRef] [PubMed]
- Trębacz H, Barzycka A. Mechanical Properties and Functions of Elastin: An Overview. Biomolecules. 2023 Mar 22;13(3):574. [CrossRef] [PubMed] [PubMed Central]
- Trębacz H, Barzycka A. Mechanical properties and functions of elastin: an overview. Biomol Ther. 2023;13:574.
- Li B, Daggett V. Molecular basis for the extensibility of elastin. J Muscle Res Cell Motil. 2002;23:561–73.
- Bates JH, Davis GS, Majumdar A, Butnor KJ, Suki B. Linking parenchymal disease progression to changes in lung mechanical function by percolation. Am J Respir Crit Care Med. 2007;176:617–23.
- Bunde A, Kantelhart JW. Diffusion and conduction in percolation systems In: P Heitjans and J Karger, editors. Diffusion in condensed matter. Berlin: Springer (2005). 895–914.
- Redner S. Fractal and multifractal scaling of electrical conduction in random resistor networks In: PA Meyers, editor. Encyclopedia of complexity and systems science. Berlin: Springer (2009). 3737–54.
- Murphy KD, Hunt GW, Almond DP. Evidence of emergent scaling in mechanical systems. Philos Mag. 2006;86:3325–38.
- Rabe KF, Watz H. Chronic obstructive pulmonary disease. Lancet. 2017;389(10082):1931-1940.
- Churg A, Wright JL. Proteases and emphysema. Curr Mol Med. 2006;6(3):289-308.
- Upadhyay P, Wu CW, Pham A, Zeki AA, Royer CM, Kodavanti UP, Takeuchi M, Bayram H, Pinkerton KE. Animal models and mechanisms of tobacco smoke-induced chronic obstructive pulmonary disease (COPD). J Toxicol Environ Health B Crit Rev. 2023 Jul 4;26(5):275-305. Epub 2023 May 14. [CrossRef] [PubMed] [PubMed Central]
- Churg A, Cosio M, Wright JL. Mechanisms of cigarette smoke-induced COPD: insights from animal models. Am J Physiol Lung Cell Mol Physiol. 2008 Apr;294(4):L612-31. Epub 2008 Jan 25. [CrossRef] [PubMed]
- Christopoulou ME, Papakonstantinou E, Stolz D. Matrix Metalloproteinases in Chronic Obstructive Pulmonary Disease. Int J Mol Sci. 2023;24(4):3786.
- Houghton, A. M. (2015). "The role of neutrophils in the development of emphysema: Lessons from animal models." *Amer J of Resp Cell Mol Biol, 53(5), 707-711. [CrossRef]
- Meghraoui-Kheddar A, Pierre A, Sellami M, Audonnet S, Lemaire F, Le Naour R. Elastin receptor (S-gal) occupancy by elastin peptides modulates T-cell response during murine emphysema. Am J Physiol Lung Cell Mol Physiol. 2017 Sep 1;313(3):L534-L547. Epub 2017 Jun 1. [CrossRef] [PubMed]
- Le Page A, Khalil A, Vermette P, Frost EH, Larbi A, Witkowski JM, Fulop T. The role of elastin-derived peptides in human physiology and diseases. Matrix Biol. 2019 Nov;84:81-96. Epub 2019 Jul 8. [CrossRef] [PubMed]
- de Castro Brás LE, Frangogiannis NG. Extracellular matrix-derived peptides in tissue remodeling and fibrosis. Matrix Biology: Journal of the International Society for Matrix Biology. 2020 Sep;91-92:176-187. [CrossRef] [PubMed] [PubMed Central]
- Sellami M, Meghraoui-Kheddar A, Terryn C, Fichel C, Bouland N, Diebold MD, Guenounou M, Héry-Huynh S, Le Naour R. Induction and regulation of murine emphysema by elastin peptides. Am J Physiol Lung Cell Mol Physiol. 2016 Jan 1;310(1):L8-23. Epub 2015 Oct 30. [CrossRef] [PubMed]
- Houghton AM, Quintero PA, Perkins DL, Kobayashi DK, Kelley DG, Marconcini LA, Mecham RP, Senior RM, Shapiro SD. Elastin fragments drive disease progression in a murine model of emphysema. J Clin Invest. 2006 Mar;116(3):753-9. Epub 2006 Feb 9. [CrossRef] [PubMed] [PubMed Central]
- Tembely D, Henry A, Vanalderwiert L, Toussaint K, Bennasroune A, Blaise S, Sartelet H, Jaisson S, Galés C, Martiny L, Duca L, Romier-Crouzet B, Maurice P. The Elastin Receptor Complex: An Emerging Therapeutic Target Against Age-Related Vascular Diseases. Front Endocrinol (Lausanne). 2022 Feb 11;13:815356. [CrossRef] [PubMed] [PubMed Central]
- Szychowski KA, Skóra B. The elastin-derived peptide (VGVAPG) activates autophagy in neuroblastoma (SH-SY5Y) cells via peroxisome proliferator-activated receptor gamma (PPARγ). Mol Cell Neurosci. 2023 Dec;127:103902. Epub 2023 Nov 2. [CrossRef] [PubMed]
- de Oliveira MV, Rocha NN, Santos RS, Rocco MRM, de Magalhães RF, Silva JD, Souza SAL, Capelozzi VL, Pelosi P, Silva PL, Rocco PRM. Endotoxin-induced emphysema exacerbation: A novel model of chronic obstructive pulmonary disease exacerbations causing cardiopulmonary impairment and diaphragm dysfunction. Front Physiol. 2019;10:664.
- Kobayashi S, Fujinawa R, Ota F, Kobayashi S, Angata T, Ueno M, Maeno T, Kitazume S, Yoshida K, Ishii T, Gao C, Ohtsubo K, Yamaguchi Y, Betsuyaku T, Kida K, Taniguchi N. A single dose of lipopolysaccharide into mice with emphysema mimics human chronic obstructive pulmonary disease exacerbation as assessed by micro-computed tomography. Am J Respir Cell Mol Biol. 2013;49:971-977.
- Suki B, Sato S, Parameswaran H, Szabari MV, Takahashi A, Bartolák-Suki E. Emphysema and mechanical stress-induced lung remodeling. Physiology (Bethesda). 2013 Nov;28(6):404-13. [CrossRef] [PubMed] [PubMed Central]
- Villa B, Erranz B, Cruces P, Retamal J, Hurtado DE. Mechanical and morphological characterization of the emphysematous lung tissue. Acta Biomater. 2024 Jun;181:282-296. Epub 2024 May 3. [CrossRef] [PubMed]
- Suki B, Bates JH. Lung tissue mechanics as an emergent phenomenon. J Appl Physiol. (2011) 110:1111–8. [CrossRef]
- Sun GQ, Jusup M, Jin Z, Wang Y, Wang Z. Pattern transitions in spatial epidemics: mechanisms and emergent properties. Phys Life Rev. (2016) 19:43–73. [CrossRef]
- Winkler T, Suki B. Emergent structure-function relations in emphysema and asthma. Crit Rev Biomed Eng. (2011) 39:263–80. [CrossRef]
- Sander LM, Warren CP, Sokolov IM, Simon C, Koopman J. Percolation on heterogeneous networks as a model for epidemics. Math Biosci. (2002) 180:293–305. [CrossRef]
- Artime O, De Domenico M. From the origin of life to pandemics: emergent phenomena in complex systems. Philos Trans A Math Phys Eng Sci. 2022 Jul 11;380(2227):20200410. Epub 2022 May 23. [CrossRef] [PubMed] [PubMed Central]
- Kalantari, S., Nazemi, E., & Masoumi, B. (2020). Emergence phenomena in self-organizing systems: a systematic literature review of concepts, researches, and future prospects. Journal of Organizational Computing and Electronic Commerce, 30(3), 224–265. [CrossRef]
- Fagiola M, Reznik S, Riaz M, Qyang Y, Lee S, Avella J, Turino G, Cantor J. The relationship between elastin cross linking and alveolar wall rupture in human pulmonary emphysema. Am J Physiol Lung Cell Mol Physiol. 2023;324(6):L747-L755.
- Vasconcelos, G.L., Macêdo, A.M.S., Duarte-Filho, G.C. et al. Power law behaviour in the saturation regime of fatality curves of the COVID-19 pandemic. Sci Rep 11, 4619 (2021). [CrossRef]
- Browne, C. A., Amchin, D. B., Schneider, J., & Datta, S. S. (2021). Infection Percolation: A Dynamic Network Model of Disease Spreading. Frontiers in Physics, 9, Article 645954. [CrossRef]
- Contreras DA, Cencetti G, Barrat A. Infection patterns in simple and complex contagion processes on networks. PLoS Comput Biol. 2024 Jun 10;20(6):e1012206. [CrossRef] [PubMed] [PubMed Central]
- Cantor JO, Cerreta JM, Keller S, Turino GM. Modulation of airspace enlargement in elastase-induced emphysema by intratracheal instillment of hyaluronidase and hyaluronic acid. Exp Lung Res. (1995) 21:423–36. [CrossRef]
- Cantor JO, Cerreta JM, Ochoa M, Ma S, Chow T, Grunig G, Turino GM. Aerosolized hyaluronan limits airspace enlargement in a mouse model of cigarette smoke-induced pulmonary emphysema. Exp Lung Res. 2005 May;31(4):417-30.
- Cantor JO, Ma S, Liu X, Campos MA, Strange C, Stocks JM, et al. A 28-day clinical trial of aerosolized hyaluronan in alpha-1 antiprotease deficiency COPD using desmosine as a surrogate marker for drug efficacy. Respir Med. 2021;182:106402.
- Takahashi A, Majumdar A, Parameswaran H, Bartolák-Suki E, Suki B. Proteoglycans maintain lung stability in an elastase-treated mouse model of emphysema. Am J Respir Cell Mol Biol. (2014) 51:26–33. [CrossRef]
- Strange C. Antiproteases and alpha-1 antitrypsin augmentation therapy. Respir Care. (2018) 63:690–8. [CrossRef]
- Kuna P, Jenkins M, O’Brien CD, Fahy WA. AZD9668, a neutrophil elastase inhibitor, plus ongoing budesonide/formoterol in patients with COPD. Respir Med. (2012) 106:531–9. [CrossRef]
- Lucas SD, Costa E, Guedes RC, Moreira R. Targeting COPD: advances on low- molecular-weight inhibitors of human neutrophil elastase. Med Res Rev. (2013) 33:E73– E101. [CrossRef]
- Groutas WC, Dou D, Alliston KR. Neutrophil elastase inhibitors. Expert Opin Ther Pat. (2011) 21:339–54. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).