Submitted:
28 January 2025
Posted:
28 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Stable Transfectants
2.2. Antibodies
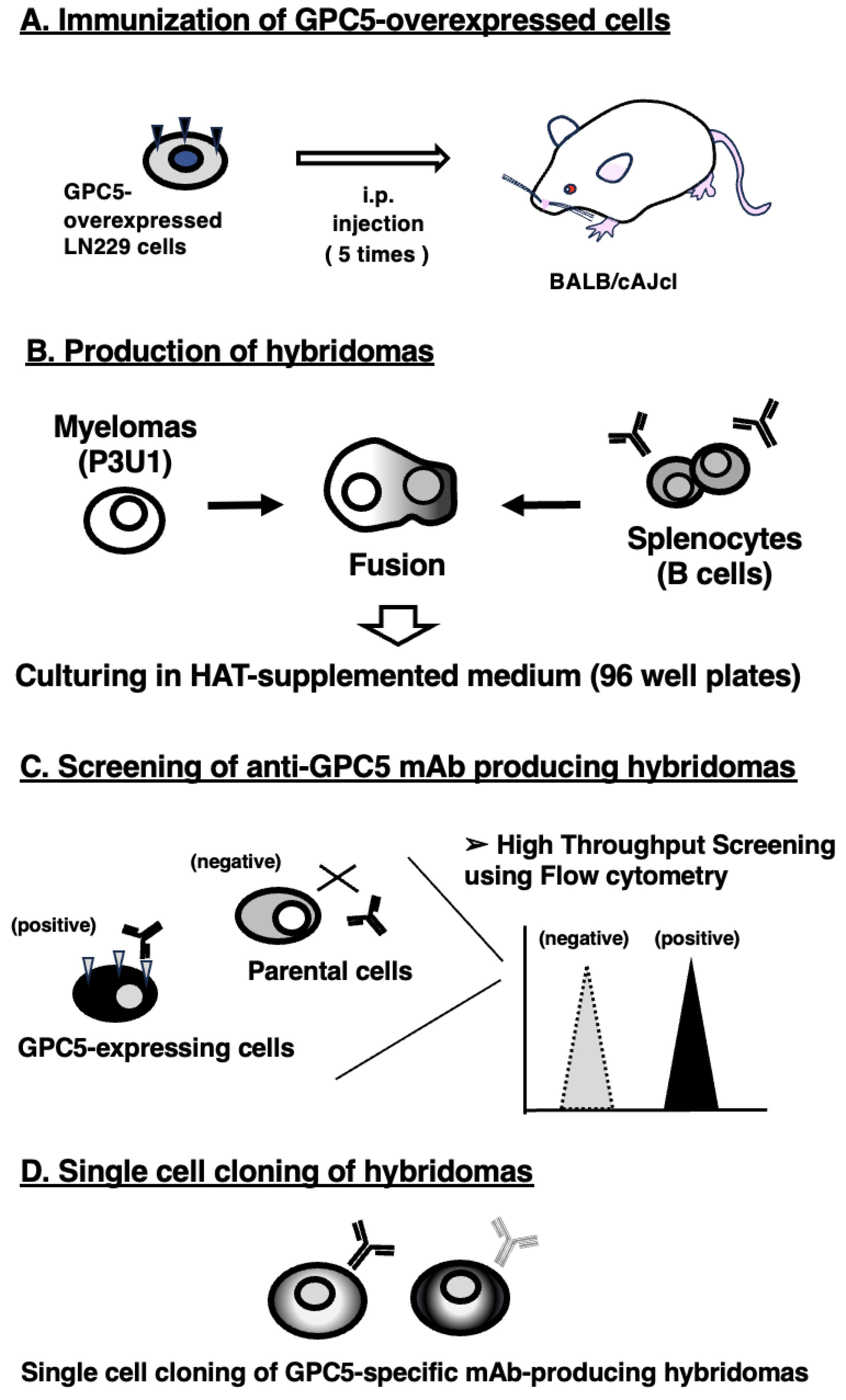
2.3. Development of Hybridomas
2.4. Flow Cytometric Analysis
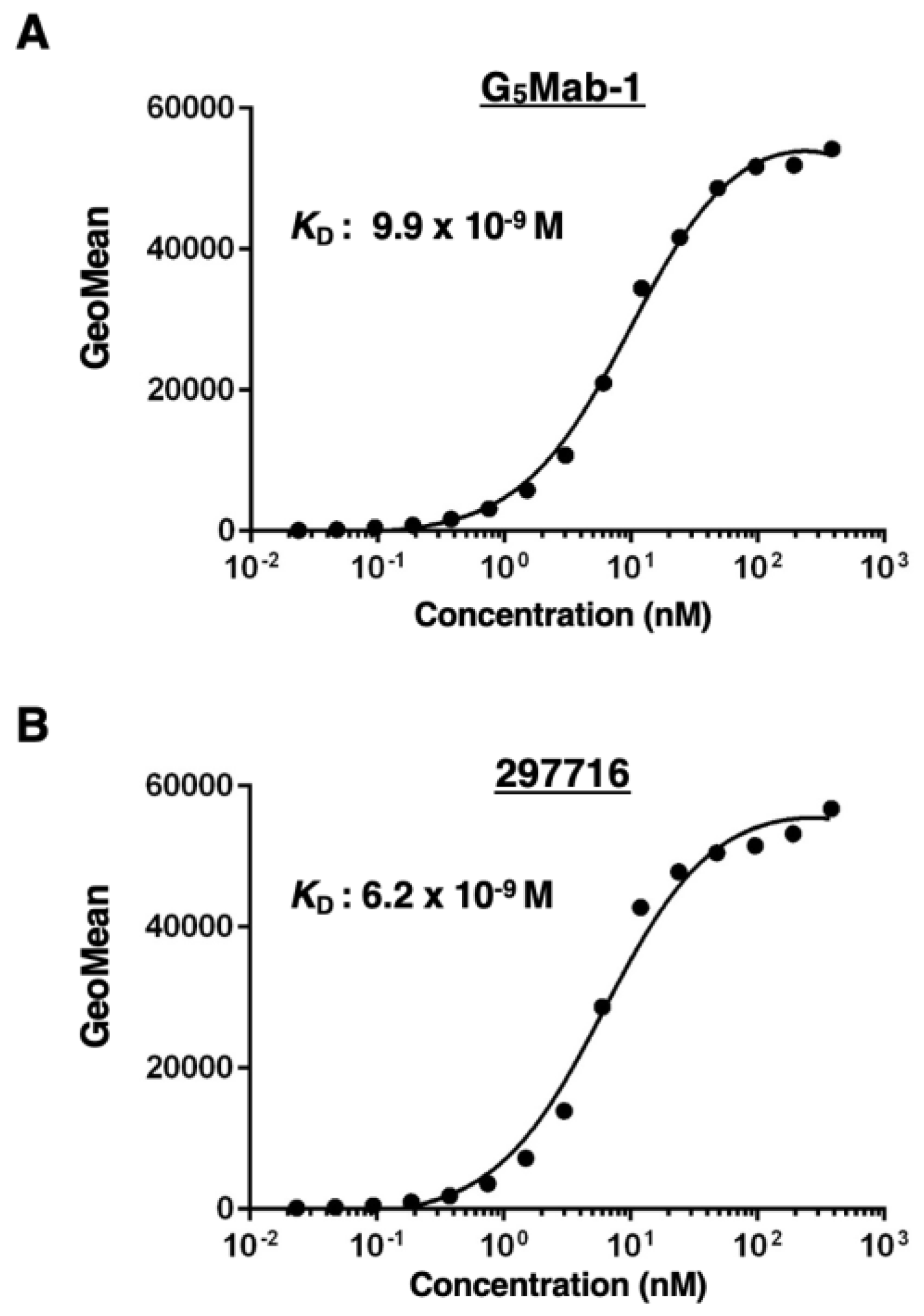
2.5. Determination of Dissociation Constant (KD) by Flow Cytometry
2.6. Western Blot Analysis
2.7. Immunohistochemical Analysis
3. Results
3.1. Development of Anti-GPC5 mAbs Using the CBIS Method
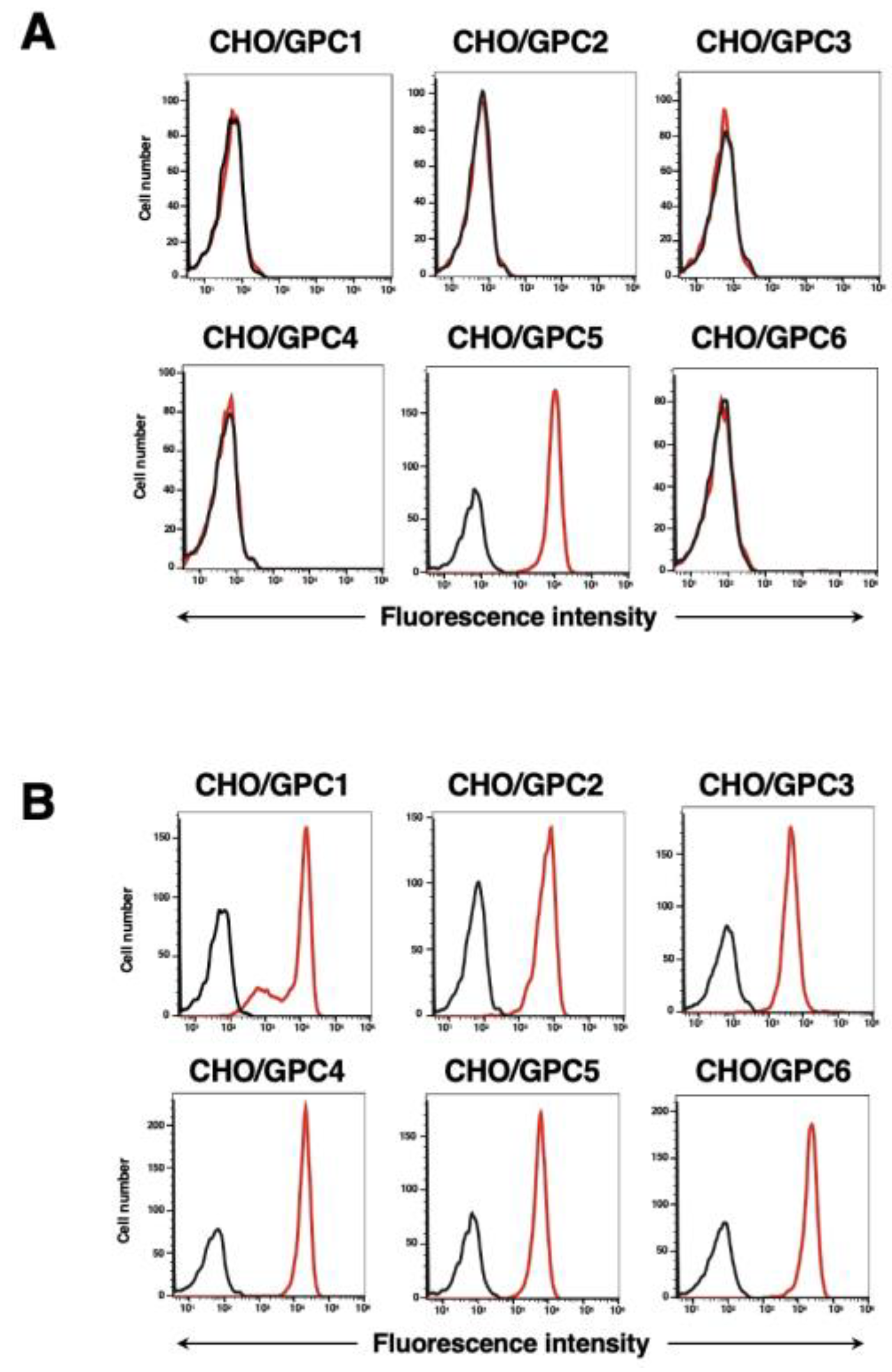
3.2. Evaluation of Antibody Reactivity and Specificity Using Flow Cytometry
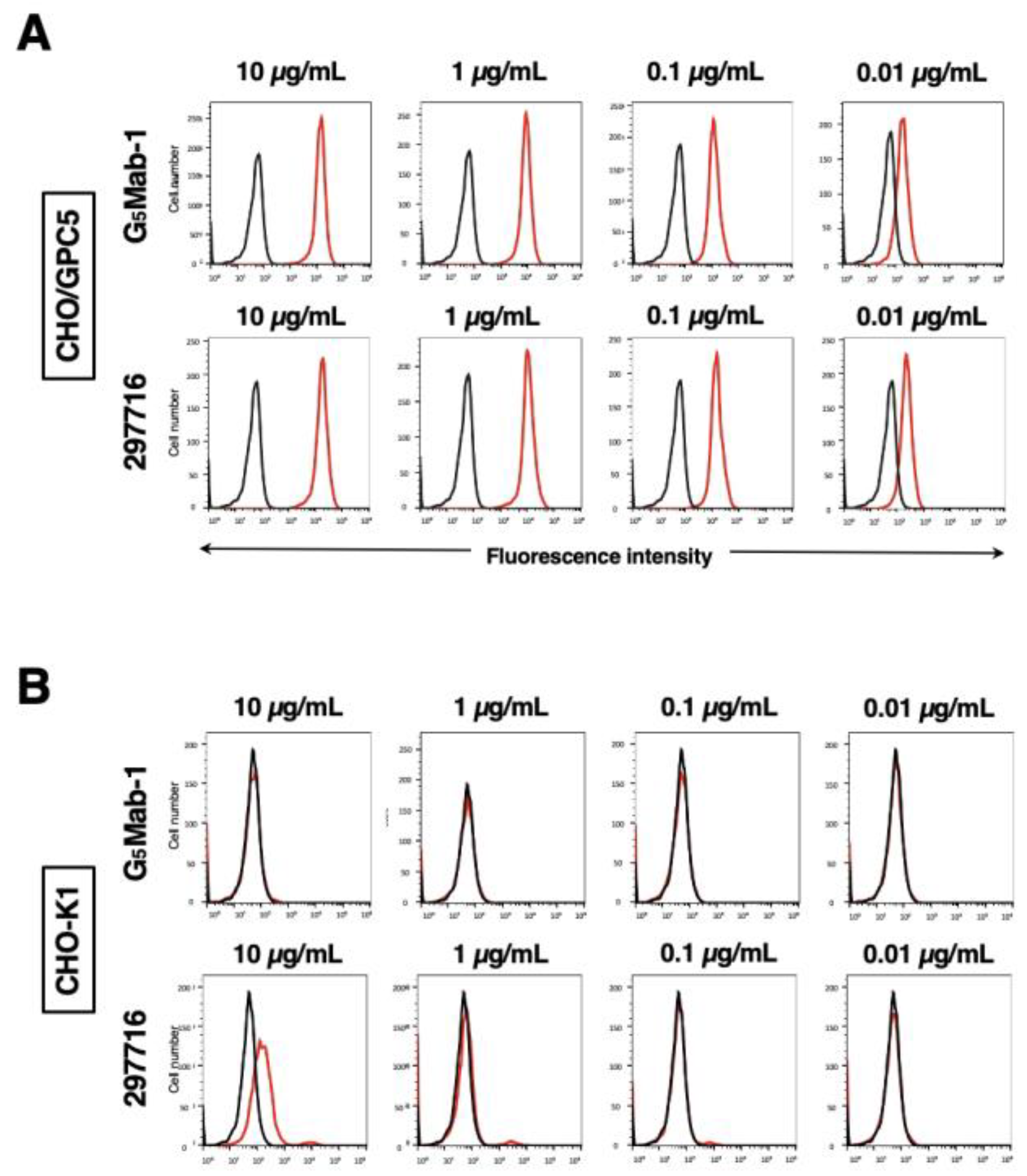
3.3. Specificity of G5Mab-1 to Glypican-Overexpressed CHO-K1 Cells
3.4. Calculation of the Apparent Binding Affinity of Anti-GPC5 mAbs Using Flow Cytometry
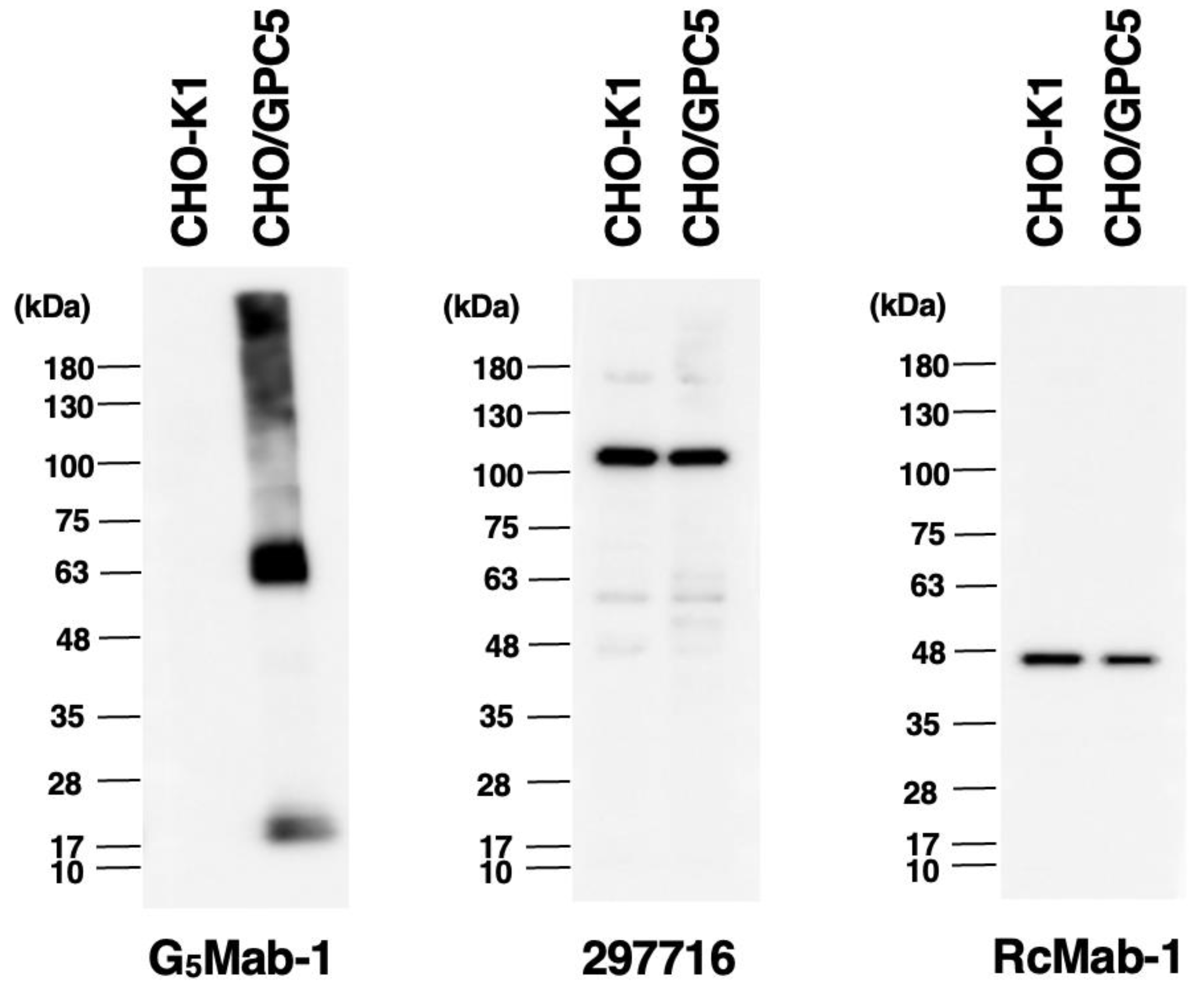
3.5. Western Blot Analyses Using Anti-GPC5 mAbs
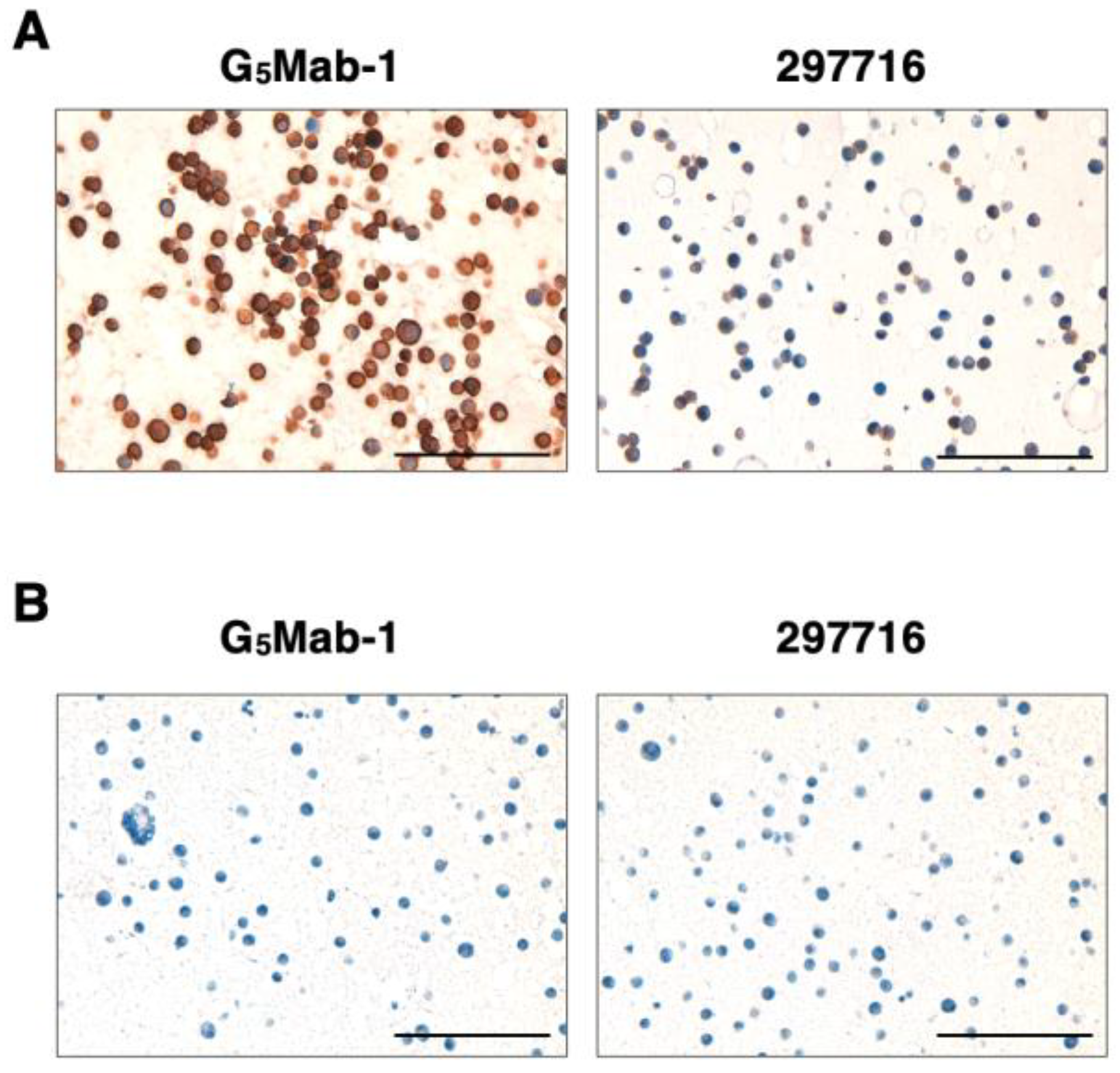
3.6. Immunohistochemistry Using Anti-GPC5 mAbs
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Song, H.H.; Filmus, J. The role of glypicans in mammalian development. Biochim Biophys Acta 2002, 1573, 241–246. [Google Scholar] [CrossRef]
- De Cat, B.; David, G. Developmental roles of the glypicans. Semin Cell Dev Biol 2001, 12, 117–125. [Google Scholar] [CrossRef]
- Bernfield, M.; Götte, M.; Park, P.W.; et al. Functions of cell surface heparan sulfate proteoglycans. Annu Rev Biochem 1999, 68, 729–777. [Google Scholar] [CrossRef] [PubMed]
- Paine-Saunders, S.; Viviano, B.L.; Saunders, S. GPC6, a novel member of the glypican gene family, encodes a product structurally related to GPC4 and is colocalized with GPC5 on human chromosome 13. Genomics 1999, 57, 455–458. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, S.; Lamanna, W.C.; Esko, J.D. Heparan sulfate proteoglycans. Cold Spring Harb Perspect Biol 2011, 3. [Google Scholar] [CrossRef]
- Li, F.; Shi, W.; Capurro, M.; Filmus, J. Glypican-5 stimulates rhabdomyosarcoma cell proliferation by activating Hedgehog signaling. J Cell Biol 2011, 192, 691–704. [Google Scholar] [CrossRef]
- Filmus, J.; Capurro, M. The role of glypicans in Hedgehog signaling. Matrix Biol 2014, 35, 248–252. [Google Scholar] [CrossRef]
- de Moraes, G.F.A.; Listik, E.; Justo, G.Z.; Vicente, C.M.; Toma, L. The Glypican proteoglycans show intrinsic interactions with Wnt-3a in human prostate cancer cells that are not always associated with cascade activation. BMC Mol Cell Biol 2021, 22, 26. [Google Scholar] [CrossRef] [PubMed]
- Saunders, S.; Paine-Saunders, S.; Lander, A.D. Expression of the cell surface proteoglycan glypican-5 is developmentally regulated in kidney, limb, and brain. Dev Biol 1997, 190, 78–93. [Google Scholar] [CrossRef] [PubMed]
- Luxardi, G.; Galli, A.; Forlani, S.; et al. Glypicans are differentially expressed during patterning and neurogenesis of early mouse brain. Biochem Biophys Res Commun 2007, 352, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Handel, A.E.; Ramagopalan, S.V. GPC5 and lung cancer in multiple sclerosis. Lancet Oncol 2010, 11, 714. [Google Scholar] [CrossRef] [PubMed]
- Mowry, E.M.; Carey, R.F.; Blasco, M.R.; et al. Multiple sclerosis susceptibility genes: associations with relapse severity and recovery. PLoS One 2013, 8, e75416. [Google Scholar] [CrossRef]
- Li, N.; Spetz, M.R.; Ho, M. The Role of Glypicans in Cancer Progression and Therapy. J Histochem Cytochem 2020, 68, 841–862. [Google Scholar] [CrossRef]
- Veugelers, M.; Vermeesch, J.; Reekmans, G.; et al. Characterization of glypican-5 and chromosomal localization of human GPC5, a new member of the glypican gene family. Genomics 1997, 40, 24–30. [Google Scholar] [CrossRef]
- Ota, A.; Tagawa, H.; Karnan, S.; et al. Identification and characterization of a novel gene, C13orf25, as a target for 13q31-q32 amplification in malignant lymphoma. Cancer Res 2004, 64, 3087–3095. [Google Scholar] [CrossRef]
- Li, Y.; Miao, L.; Cai, H.; et al. The overexpression of glypican-5 promotes cancer cell migration and is associated with shorter overall survival in non-small cell lung cancer. Oncol Lett 2013, 6, 1565–1572. [Google Scholar] [CrossRef]
- Ojopi, E.P.; Rogatto, S.R.; Caldeira, J.R.; Barbiéri-Neto, J.; Squire, J.A. Comparative genomic hybridization detects novel amplifications in fibroadenomas of the breast. Genes Chromosomes Cancer 2001, 30, 25–31. [Google Scholar] [CrossRef]
- Williamson, D.; Selfe, J.; Gordon, T.; et al. Role for amplification and expression of glypican-5 in rhabdomyosarcoma. Cancer Res 2007, 67, 57–65. [Google Scholar] [CrossRef]
- Li, Y.; Yang, P. GPC5 gene and its related pathways in lung cancer. J Thorac Oncol 2011, 6, 2–5. [Google Scholar] [CrossRef]
- Takeuchi, M.; Takeuchi, K.; Takai, T.; et al. Subcellular localization of glypican-5 is associated with dynamic motility of the human mesenchymal stem cell line U3DT. PLoS One 2021, 16, e0226538. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, S.; Zhang, D.; et al. A lung cancer gene GPC5 could also be crucial in breast cancer. Mol Genet Metab 2011, 103, 104–105. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, Z.; Wang, L.; et al. Prognostic significance of GPC5 expression in patients with prostate cancer. Tumour Biol 2016, 37, 6413–6418. [Google Scholar] [CrossRef]
- Hong, X.; Zhang, Z.; Pan, L.; et al. MicroRNA-301b promotes the proliferation and invasion of glioma cells through enhancing activation of Wnt/β-catenin signaling via targeting Glypican-5. Eur J Pharmacol 2019, 854, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhao, J.; Yin, X.; et al. miR-297 acts as an oncogene by targeting GPC5 in lung adenocarcinoma. Cell Prolif 2016, 49, 636–643. [Google Scholar] [CrossRef]
- Sun, Y.; Xu, K.; He, M.; Fan, G.; Lu, H. Overexpression of Glypican 5 (GPC5) Inhibits Prostate Cancer Cell Proliferation and Invasion via Suppressing Sp1-Mediated EMT and Activation of Wnt/β-Catenin Signaling. Oncol Res 2018, 26, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Chen, Y.; Zhou, Y.; et al. GPC5 suppresses lung cancer progression and metastasis via intracellular CTDSP1/AhR/ARNT signaling axis and extracellular exosome secretion. Oncogene 2021, 40, 4307–4323. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, Z.; Qiu, M.; et al. Glypican-5 is a novel metastasis suppressor gene in non-small cell lung cancer. Cancer Lett 2013, 341, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Sheu, C.C.; Ye, Y.; et al. Genetic variants and risk of lung cancer in never smokers: a genome-wide association study. Lancet Oncol 2010, 11, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Yu, Z.; Liu, Q.; et al. GPC5, a novel epigenetically silenced tumor suppressor, inhibits tumor growth by suppressing Wnt/β-catenin signaling in lung adenocarcinoma. Oncogene 2016, 35, 6120–6131. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Han, C.; Liu, J.; et al. GPC5, a tumor suppressor, is regulated by miR-620 in lung adenocarcinoma. Mol Med Rep 2014, 9, 2540–2546. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, X.; Sha, K.; et al. miR-709 up-regulated in hepatocellular carcinoma, promotes proliferation and invasion by targeting GPC5. Cell Prolif 2015, 48, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Itai, S.; Nakamura, T.; et al. Monoclonal Antibody L(1)Mab-13 Detected Human PD-L1 in Lung Cancers. Monoclon Antib Immunodiagn Immunother 2018, 37, 110–115. [Google Scholar] [CrossRef]
- Suzuki, H.; Tanaka, T.; Kudo, Y.; et al. A Rat Anti-Mouse CD39 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2023, 42, 203–208. [Google Scholar] [CrossRef]
- Li, G.; Suzuki, H.; Asano, T.; et al. Development of a Novel Anti-EpCAM Monoclonal Antibody for Various Applications. Antibodies (Basel) 2022, 11. [Google Scholar] [CrossRef]
- Tanaka, T.; Ohishi, T.; Asano, T.; et al. An anti-TROP2 monoclonal antibody TrMab-6 exerts antitumor activity in breast cancer mouse xenograft models. Oncol Rep 2021, 46. [Google Scholar] [CrossRef] [PubMed]
- Fujii, Y.; Kaneko, M.; Neyazaki, M.; et al. PA tag: a versatile protein tagging system using a super high affinity antibody against a dodecapeptide derived from human podoplanin. Protein Expr Purif 2014, 95, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Ikota, H.; Nobusawa, S.; Arai, H.; et al. Evaluation of IDH1 status in diffusely infiltrating gliomas by immunohistochemistry using anti-mutant and wild type IDH1 antibodies. Brain Tumor Pathol 2015, 32, 237–244. [Google Scholar] [CrossRef]
- Chui, M.H.; Have, C.; Hoang, L.N.; et al. Genomic profiling identifies GPC5 amplification in association with sarcomatous transformation in a subset of uterine carcinosarcomas. J Pathol Clin Res 2018, 4, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Suzuki, H.; Ohishi, T.; et al. Antitumor activities of a defucosylated anti-EpCAM monoclonal antibody in colorectal carcinoma xenograft models. Int J Mol Med 2023, 51. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Suzuki, H.; Ohishi, T.; Kaneko, M.K.; Kato, Y. Antitumor activities against breast cancers by an afucosylated anti-HER2 monoclonal antibody H(2) Mab-77-mG(2a) -f. Cancer Sci 2024, 115, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. A Cancer-Specific Monoclonal Antibody against HER2 Exerts Antitumor Activities in Human Breast Cancer Xenograft Models. Int J Mol Sci, 2024; 25. [Google Scholar]
- Zhu, A.X.; Gold, P.J.; El-Khoueiry, A.B.; et al. First-in-man phase I study of GC33, a novel recombinant humanized antibody against glypican-3, in patients with advanced hepatocellular carcinoma. Clin Cancer Res 2013, 19, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Ohkawa, S.; Okusaka, T.; et al. Japanese phase I study of GC33, a humanized antibody against glypican-3 for advanced hepatocellular carcinoma. Cancer Sci 2014, 105, 455–462. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Puig, O.; Daniele, B.; et al. Randomized phase II placebo controlled study of codrituzumab in previously treated patients with advanced hepatocellular carcinoma. J Hepatol 2016, 65, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Shi, Y.; Kaseb, A.O.; et al. Chimeric Antigen Receptor-Glypican-3 T-Cell Therapy for Advanced Hepatocellular Carcinoma: Results of Phase I Trials. Clin Cancer Res 2020, 26, 3979–3989. [Google Scholar] [CrossRef]
- Veugelers, M.; De Cat, B.; Ceulemans, H.; et al. Glypican-6, a new member of the glypican family of cell surface heparan sulfate proteoglycans. J Biol Chem 1999, 274, 26968–26977. [Google Scholar] [CrossRef]
- Capurro, M.I.; Xiang, Y.Y.; Lobe, C.; Filmus, J. Glypican-3 promotes the growth of hepatocellular carcinoma by stimulating canonical Wnt signaling. Cancer Res 2005, 65, 6245–6254. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Tang, Z.; Zhang, Y.F.; et al. Immunotoxin targeting glypican-3 regresses liver cancer via dual inhibition of Wnt signalling and protein synthesis. Nat Commun 2015, 6, 6536. [Google Scholar] [CrossRef]
- Capurro, M.I.; Xu, P.; Shi, W.; et al. Glypican-3 inhibits Hedgehog signaling during development by competing with patched for Hedgehog binding. Dev Cell 2008, 14, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Grisaru, S.; Cano-Gauci, D.; Tee, J.; Filmus, J.; Rosenblum, N.D. Glypican-3 modulates BMP- and FGF-mediated effects during renal branching morphogenesis. Dev Biol 2001, 231, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Awad, W.; Adamczyk, B.; Örnros, J.; et al. Structural Aspects of N-Glycosylations and the C-terminal Region in Human Glypican-1. J Biol Chem 2015, 290, 22991–23008. [Google Scholar] [CrossRef] [PubMed]
- De Cat, B.; Muyldermans, S.Y.; Coomans, C.; et al. Processing by proprotein convertases is required for glypican-3 modulation of cell survival, Wnt signaling, and gastrulation movements. J Cell Biol 2003, 163, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Steigmeyer, A.D.; Lowery, S.C.; Rangel-Angarita, V.; Malaker, S.A. Decoding Extracellular Protein Glycosylation in Human Health and Disease. Annu Rev Anal Chem (Palo Alto Calif) 2025. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Kaneko, M.K. A Cancer-specific Monoclonal Antibody Recognizes the Aberrantly Glycosylated Podoplanin. Scientific Reports 2014, 4, 5924. [Google Scholar] [CrossRef] [PubMed]
- Arimori, T.; Mihara, E.; Suzuki, H.; et al. Locally misfolded HER2 expressed on cancer cells is a promising target for development of cancer-specific antibodies. Structure 2024, 32, 536–549.e535. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



