Submitted:
27 January 2025
Posted:
28 January 2025
You are already at the latest version
Abstract
Keywords:
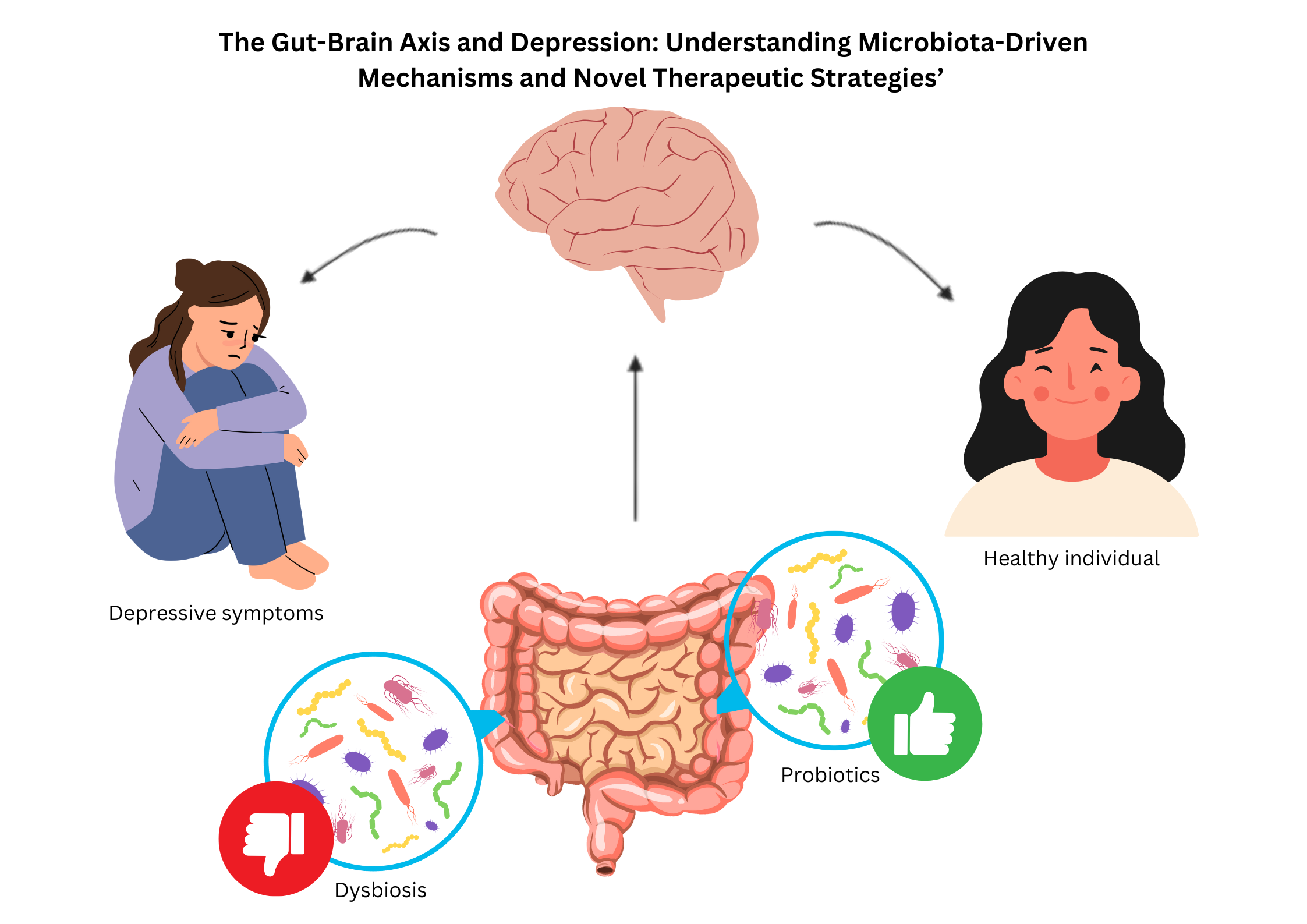
1. Introduction
2. Gut Microbiota System
| Bacteria Genera | Human Studies | Animal Studies | Remarks |
|---|---|---|---|
| Firmicutes | ↓ Decreased in most studies | ↓ Decreased in rodent models of depression | Key SCFA producers (e.g., Faecalibacterium, Coprococcus) |
| ↓ Associated with gut dysbiosis and reduced SCFAs | ↓ Correlated with depressive-like behaviours in rodents | ||
| Bifidobacterium | ↓ Decreased in depression | ↓ Decreased in stress-induced models | Serotonin modulation, probiotic supplementation improves mood |
| Actinobacteria | ↓ Decreased in some studies which usually decreased in Bifidobacterium too | ↑ Increased in certain conditions like high-fat diets and stress | Contains beneficial bacteria like Bifidobacterium |
| Bacteroides | ↓ Decreased in some cases, however it also ↑ increased in others | ↓ Decreased in high-stress or high-fat diet models | Variable depending on species and inflammation |
| Clostridium | ↓ Decreased in beneficial species like Clostridium butyricum in SCFAs production leading to increased inflammation and gut-brain axis disruption | ↑ Increased in pathogenic species like Clostridium perfringens in dysbiotic conditions of stress-induced models. | Loss of SCFA producers affects gut health which may worsen inflammation |
| Faecalibacterium | ↓ Decreased in depression | ↓ Reduced in stress models | Major SCFA producer (anti-inflammatory) |
| Coprococcus | ↓ Depleted in participants with depression | ↓ Reduced in stress-induced animal models | Butyrate producer with anti-inflammatory properties |
| Roseburia | ↓ Decreased in depression | ↓ Decreased in animal models with induced stress | SCFA producer important for gut integrity |
| Dialister | ↓ Decreased in patients with major depressive disorder | ↓ Reduced in animal models with gut dysbiosis | Associated with mood regulation |
| Lactobacillus | ↓ Decreased in depression | ↓ Reduced in rodent models of stress | Produces neurotransmitters like Gamma-aminobutyric acid (GABA) |
| Ruminococcus | ↓ Decreased in depressive patients | ↓ Reduced in animal models | SCFA production and gut health |
| Alistipes | ↓ Decreased in individuals with depression | ↓ Reduced in stress-induced animal models. | Associated with mental signs of depression |
| Prevotella | ↓ Decreased in most studies | ↓ Reduced in rodent models | Associated with carbohydrate metabolism |
3. Current Evidence Linking the Gut Microbiota to Depression
3.1. Microbiota Composition and Diversity
3.2. Probiotics and Prebiotics
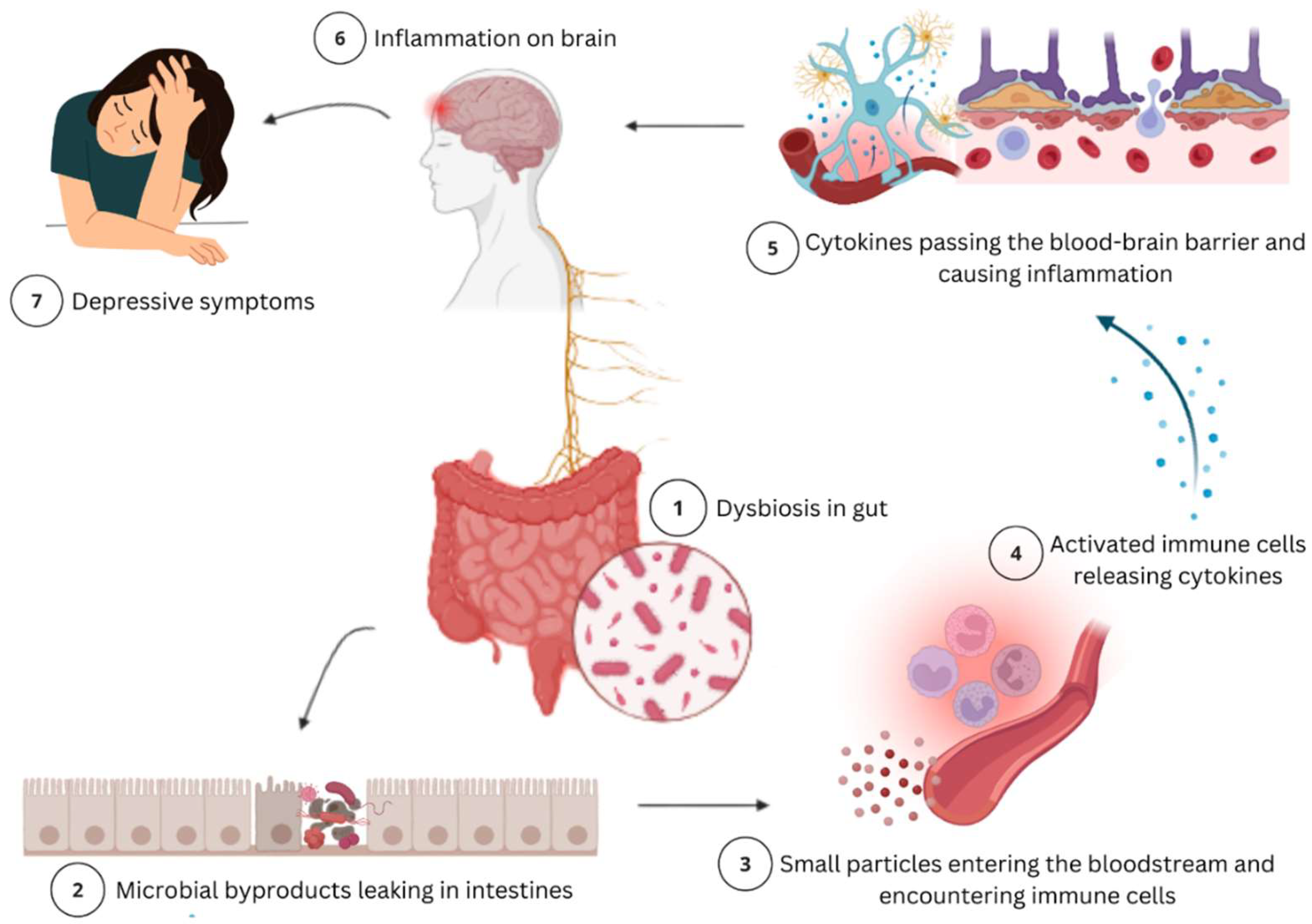
4. Mechanism of Action
4.1. Neuroimmune Modulation
4.2. Neurotransmitter production
4.3. Vagus Nerve Pathways
5. Demographic Differences in Gut Microbiota and Depression
6. Limitation of Current Studies on the Gut-Brain Axis
7. Future Directions and Clinical Translation
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MDD | Major depressive disorder |
| GBA | Gut-brain axis |
| CNS | Central nervous system |
| HPA | Hypothalamus-pituitary-adrenal |
| SCFA | Short-chain fatty acid |
| ENS | Enteric nervous system |
| BDNF | Brain-derived neurotrophic factor |
| ROS | Reactive oxygen species |
| BBB | Blood-brain barrier |
| GABA | Gamma-aminobutyric acid |
| IL-6 | Interleukin-6 |
| IDO1 | Indoleamine 2,3-dioxygenase 1 |
| 5-HT | 5-hydroxytryptamine |
| NTS | Nucleus tractus solitarii |
| DRG | Dorsal root ganglia |
| VNS | Vagus nerve stimulation |
| PET | Positron emission tomography |
| vmPFC | Ventromedial prefrontal cortex |
| TRD | Treatment-resistant depression |
| BMI | Body mass index |
References
- Alli, S. R. , Gorbovskaya, I., Liu, J. C. W., Kolla, N. J., Brown, L., & Müller, D. J. The gut microbiome in Depression and Potential benefit of prebiotics, probiotics and synbiotics: A Systematic review of clinical trials and observational studies. International Journal of Molecular Sciences 2022, 23, 4494. [Google Scholar] [CrossRef]
- Bakshi, I. , Dey, S., Raut, A. J., Katta, S., & Sharma, P. Exploring the Gut-Brain Axis: a comprehensive review of interactions between the gut microbiota and the central nervous system. International Journal for Multidisciplinary Research. [CrossRef]
- Belmaker, R. , & Agam, G. Major depressive disorder. New England Journal of Medicine 2008, 358, 55–68. [Google Scholar] [CrossRef]
- Berthoud, H. , Albaugh, V. L., & Neuhuber, W. L. Gut-brain communication and obesity: understanding functions of the vagus nerve. Journal of Clinical Investigation. [CrossRef]
- Bevilacqua, A. , Campaniello, D., Speranza, B., Racioppo, A., Sinigaglia, M., & Corbo, M. R. An update on prebiotics and on their health effects. Foods 2024, 13, 446. [Google Scholar] [CrossRef]
- Bindels, L. B. , Delzenne, N. M., Cani, P. D., & Walter, J. Towards a more comprehensive concept for prebiotics. Nature Reviews Gastroenterology & Hepatology 2015, 12, 303–310. [Google Scholar] [CrossRef]
- Bonaz, B. , Bazin, T., & Pellissier, S. The vagus nerve at the interface of the Microbiota-Gut-Brain axis. Frontiers in Neuroscience. [CrossRef]
- Breit, S. , Kupferberg, A., Rogler, G., & Hasler, G. Vagus nerve as modulator of the Brain–Gut axis in psychiatric and inflammatory disorders. Frontiers in Psychiatry. [CrossRef]
- Carabotti, M. , Scirocco, A., Maselli, M. A., & Severi, C. (2015, ). The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. https://pmc.ncbi.nlm.nih. 1 June 4367. [Google Scholar]
- Carlson, J. L. , Erickson, J. M., Lloyd, B. B., & Slavin, J. L. Health effects and sources of prebiotic dietary fiber. Current Developments in Nutrition 2018, 2, nzy005. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y. , Li, R., & Bai, L. Vagal sensory pathway for the gut-brain communication. Seminars in Cell and Developmental Biology 2023, 156, 228–243. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, P. R. The role of gut microbiota in health and disease: Implications for therapeutic interventions. Universal Research Reports 2024, 11, 16–23. [Google Scholar] [CrossRef]
- Chen, J. , He, S., Fang, L., Wang, B., Bai, S., Xie, J., Zhou, C., Wang, W., & Xie, P. Age-specific differential changes on gut microbiota composition in patients with major depressive disorder. Aging 2020, 12, 2764–2776. [Google Scholar] [CrossRef]
- Chen, J. , Zheng, P., Liu, Y., Zhong, X., Wang, H., Guo, Y., & Xie, P. Sex differences in gut microbiota in patients with major depressive disorder. Neuropsychiatric Disease and Treatment 2018, 14, 647–655. [Google Scholar] [CrossRef]
- Chen, Y. , Xu, J., & Chen, Y. Regulation of neurotransmitters by the gut microbiota and effects on cognition in neurological disorders. Nutrients 2021, 13, 2099. [Google Scholar] [CrossRef]
- Cheng, J. , Hu, H., Ju, Y., Liu, J., Wang, M., Liu, B., & Zhang, Y. Gut microbiota-derived short-chain fatty acids and depression: deep insight into biological mechanisms and potential applications. General Psychiatry 2024, 37, e101374. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J. F. , O’Riordan, K. J., Cowan, C. S. M., Sandhu, K. V., Bastiaanssen, T. F. S., Boehme, M., Codagnone, M. G., Cussotto, S., Fulling, C., Golubeva, A. V., Guzzetta, K. E., Jaggar, M., Long-Smith, C. M., Lyte, J. M., Martin, J. A., Molinero-Perez, A., Moloney, G., Morelli, E., Morillas, E.,... Dinan, T. G. The Microbiota-Gut-Brain axis. Physiological Reviews 2019, 99, 1877–2013. [Google Scholar] [CrossRef] [PubMed]
- Dabboussi, N. , Debs, E., Bouji, M., Rafei, R., & Fares, N. Balancing the Mind: Toward a Complete Picture of the Interplay between Gut Microbiota, Inflammation and Major Depressive Disorder. Brain Research Bulletin 2024, 216, 111056. [Google Scholar] [CrossRef]
- De Leeuw, M. , Verhoeve, S. I., Van Der Wee, N. J., Van Hemert, A. M., Vreugdenhil, E., & Coomans, C. P. The role of the circadian system in the etiology of depression. Neuroscience & Biobehavioral Reviews 2023, 153, 105383. [Google Scholar] [CrossRef]
- Evrensel, A. , Ünsalver, B. Ö., & Ceylan, M. E. Neuroinflammation, Gut-Brain axis and depression. Psychiatry Investigation 2019, 17, 2–8. [Google Scholar] [CrossRef]
- Foster, J. A. , Baker, G. B., & Dursun, S. M. (2021b). The relationship between the gut Microbiome-Immune System-Brain axis and major depressive disorder. Frontiers in Neurology, 12. [CrossRef]
- Gao, M. , Wang, J., Liu, P., Tu, H., Zhang, R., Zhang, Y., Sun, N., & Zhang, K. Gut microbiota composition in depressive disorder: a systematic review, meta-analysis, and meta-regression. Translational Psychiatry. [CrossRef]
- Hao, W. , Ma, Q., Wang, L., Yuan, N., Gan, H., He, L., Li, X., Huang, J., & Chen, J. Gut dysbiosis induces the development of depression-like behavior through abnormal synapse pruning in microglia-mediated by complement C3. Microbiome. [CrossRef]
- Hu, X. , Li, Y., Wu, J., Zhang, H., Huang, Y., Tan, X., Wen, L., Zhou, X., Xie, P., Olasunkanmi, O. I., Zhou, J., Sun, Z., Liu, M., Zhang, G., Yang, J., Zheng, P., & Xie, P. Changes of gut microbiota reflect the severity of major depressive disorder: a cross sectional study. Translational Psychiatry. [CrossRef]
- Johnson, K. V. Gut microbiome composition and diversity are related to human personality traits. Human Microbiome Journal 2019, 15, 100069. [Google Scholar] [CrossRef]
- Kano, M. , & Fukudo, S. (2023). Gut–brain interactions. In Elsevier eBooks (pp. 312–333). [CrossRef]
- Kerff, F. , Pasco, J. A., Williams, L. J., Jacka, F. N., Loughman, A., & Dawson, S. L. (2024). Associations between oral microbiota pathogens and elevated depressive and anxiety symptoms in men. bioRxiv (Cold Spring Harbor Laboratory). [CrossRef]
- Kölliker-Frers, R. , Udovin, L., Otero-Losada, M., Kobiec, T., Herrera, M. I., Palacios, J., Razzitte, G., & Capani, F. Neuroinflammation: An integrating overview of Reactive-Neuroimmune cell interactions in health and disease. Mediators of Inflammation 2021, 2021, 1–20. [Google Scholar] [CrossRef]
- Kumar, A. , Pramanik, J., Goyal, N., Chauhan, D., Sivamaruthi, B. S., Prajapati, B. G., & Chaiyasut, C. Gut microbiota in Anxiety and Depression: Unveiling the relationships and management options. Pharmaceuticals 2023, 16, 565. [Google Scholar] [CrossRef]
- Liu, P. , Liu, Z., Wang, J., Wang, J., Gao, M., Zhang, Y., Yang, C., Zhang, A., Li, G., Li, X., Liu, S., Liu, L., Sun, N., & Zhang, K. Immunoregulatory role of the gut microbiota in inflammatory depression. Immunoregulatory role of the gut microbiota in inflammatory depression. Nature Communications 2024, 15. [Google Scholar] [CrossRef]
- Liu, R. T. , Rowan-Nash, A. D., Sheehan, A. E., Walsh, R. F., Sanzari, C. M., Korry, B. J., & Belenky, P. Reductions in anti-inflammatory gut bacteria are associated with depression in a sample of young adults. Brain Behavior and Immunity 2020, 88, 308–324. [Google Scholar] [CrossRef]
- Mansuy-Aubert, V. , & Ravussin, Y. Short chain fatty acids: the messengers from down below. Frontiers in Neuroscience. [CrossRef]
- Marques, S. I. , Sá, S. I., Carmo, H., Carvalho, F., & Silva, J. P. Pharmaceutical-mediated neuroimmune modulation in psychiatric/psychological adverse events. Progress in Neuro-Psychopharmacology and Biological Psychiatry 2024, 135, 111114. [Google Scholar] [CrossRef]
- Mhanna, A. , Martini, N., Hmaydoosh, G., Hamwi, G., Jarjanazi, M., Zaifah, G., Kazzazo, R., Mohamad, A. H., & Alshehabi, Z. The correlation between gut microbiota and both neurotransmitters and mental disorders: A narrative review. Medicine 2024, 103, e37114. [Google Scholar] [CrossRef] [PubMed]
- Miyaho, K. , Sanada, K., Kurokawa, S., Tanaka, A., Tachibana, T., Ishii, C., Noda, Y., Nakajima, S., Fukuda, S., Mimura, M., Kishimoto, T., & Iwanami, A. The Potential Impact of Age on Gut Microbiota in Patients with Major Depressive Disorder: A Secondary Analysis of the Prospective Observational Study. Journal of Personalized Medicine 2022, 12, 1827. [Google Scholar] [CrossRef]
- Morys, J. , Małecki, A., & Nowacka-Chmielewska, M. Stress and the gut-brain axis: an inflammatory perspective. Frontiers in Molecular Neuroscience. [CrossRef]
- Nahas, Z. , Teneback, C., Chae, J., Mu, Q., Molnar, C., Kozel, F. A., Walker, J., Anderson, B., Koola, J., Kose, S., Lomarev, M., Bohning, D. E., & George, M. S. Serial Vagus Nerve Stimulation Functional MRI in Treatment-Resistant Depression. Neuropsychopharmacology 2007, 32, 1649–1660. [Google Scholar] [CrossRef]
- Nakhal, M. M. , Yassin, L. K., Alyaqoubi, R., Saeed, S., Alderei, A., Alhammadi, A., Alshehhi, M., Almehairbi, A., Houqani, S. A., BaniYas, S., Qanadilo, H., Ali, B. R., Shehab, S., Statsenko, Y., Meribout, S., Sadek, B., Akour, A., & Hamad, M. I. K. The Microbiota–Gut–Brain Axis and Neurological Disorders: A Comprehensive review. Life 2024, 14, 1234. [Google Scholar] [CrossRef]
- Niemela, L. , Lamoury, G., Carroll, S., Morgia, M., Yeung, A., & Oh, B. Exploring gender differences in the relationship between gut microbiome and depression - a scoping review. Frontiers in Psychiatry 2024, 15. [Google Scholar] [CrossRef]
- Noh, H. , Jang, H., Kim, G., Zouiouich, S., Cho, S., Kim, H., Kim, J., Choe, J., Gunter, M. J., Ferrari, P., Scalbert, A., & Freisling, H. Taxonomic composition and diversity of the gut microbiota in relation to habitual dietary intake in Korean adults. Nutrients 2021, 13, 366. [Google Scholar] [CrossRef]
- Otte, C. , Gold, S. M., Penninx, B. W., Pariante, C. M., Etkin, A., Fava, M., Mohr, D. C., & Schatzberg, A. F. Major depressive disorder. F. Major depressive disorder. Nature Reviews Disease Primers 2016, 2. [Google Scholar] [CrossRef]
- Rahmannia, M. , Poudineh, M., Mirzaei, R., Aalipour, M. A., Bonjar, A. H. S., Goudarzi, M., Kheradmand, A., Aslani, H. R., Sadeghian, M., Nasiri, M. J., & Sechi, L. A. Strain-specific effects of probiotics on depression and anxiety: a meta-analysis. Gut Pathogens 2024, 16. [Google Scholar] [CrossRef]
- Ren, Y. , Wu, J., Wang, Y., Zhang, L., Ren, J., Zhang, Z., Chen, B., Zhang, K., Zhu, B., Liu, W., Li, S., & Li, X. Lifestyle patterns influence the composition of the gut microbiome in a healthy Chinese population. Lifestyle patterns influence the composition of the gut microbiome in a healthy Chinese population. Scientific Reports 2023, 13. [Google Scholar] [CrossRef]
- Reyes-Martínez, S. , Segura-Real, L., Gómez-García, A. P., Tesoro-Cruz, E., Constantino-Jonapa, L. A., Amedei, A., & Aguirre-García, M. M. Neuroinflammation, Microbiota-Gut-Brain axis, and Depression: the vicious circle. Journal of Integrative Neuroscience 2023, 22, 65. [Google Scholar] [CrossRef]
- Sackeim, H. Vagus Nerve Stimulation (VNSTM) for Treatment-Resistant Depression Efficacy, Side effects, and Predictors of outcome. Neuropsychopharmacology 2001, 25, 713–728. [Google Scholar] [CrossRef] [PubMed]
- Sardooi, A. R. , Reisi, P., & Yazdi, A. Protective effect of honey on learning and memory impairment, depression and neurodegeneration induced by chronic unpredictable mild stress. Physiology and Pharmacology 2020, 25, 21–35. [Google Scholar] [CrossRef]
- Silva, Y. P. , Bernardi, A., & Frozza, R. L. The role of Short-Chain fatty acids from gut microbiota in Gut-Brain communication. Frontiers in Endocrinology. [CrossRef]
- Singh, J. , Vanlallawmzuali, N., Singh, A., Biswal, S., Zomuansangi, R., Lalbiaktluangi, C., Singh, B. P., Singh, P. K., Vellingiri, B., Iyer, M., Ram, H., Udey, B., & Yadav, M. K. Microbiota-Brain Axis: Exploring the role of gut microbiota in psychiatric disorders - A Comprehensive review. Asian Journal of Psychiatry 2024, 97, 104068. [Google Scholar] [CrossRef] [PubMed]
- Ullah, H. , Arbab, S., Tian, Y., Chen, Y., Liu, C., Li, Q., & Li, K. Crosstalk between gut microbiota and host immune system and its response to traumatic injury. Crosstalk between gut microbiota and host immune system and its response to traumatic injury. Frontiers in Immunology 2024, 15. [Google Scholar] [CrossRef]
- Viana, T. R. X. RELATIONSHIP OF GUT MICROBIOTA AND MENTAL HEALTH: THE INFLUENCE OF THE GUT-BRAIN AXIS. International Journal of Health Science 2024, 4, 2–6. [Google Scholar] [CrossRef]
- Wingfield, B. , Lapsley, C., McDowell, A., Miliotis, G., McLafferty, M., O’Neill, S. M., Coleman, S., McGinnity, T. M., Bjourson, A. J., & Murray, E. K. Variations in the oral microbiome are associated with depression in young adults. K. Variations in the oral microbiome are associated with depression in young adults. Scientific Reports 2021, 11. [Google Scholar] [CrossRef]
- Wu, H. , & Wu, E. The role of gut microbiota in immune homeostasis and autoimmunity. Gut Microbes 2012, 3, 4–14. [Google Scholar] [CrossRef]
- Xiong, R. , Li, J., Cheng, J., Zhou, D., Wu, S., Huang, S., Saimaiti, A., Yang, Z., Gan, R., & Li, H. The role of gut microbiota in anxiety, depression, and other mental disorders as well as the protective effects of dietary components. Nutrients 2023, 15, 3258. [Google Scholar] [CrossRef]
- Yu, C. D. , Xu, Q. J., & Chang, R. B. Vagal sensory neurons and gut-brain signaling. Current Opinion in Neurobiology 2020, 62, 133–140. [Google Scholar] [CrossRef]
- Zhang, Q. , Yun, Y., An, H., Zhao, W., Ma, T., Wang, Z., & Yang, F. Gut microbiome composition associated with major depressive disorder and sleep quality. Gut microbiome composition associated with major depressive disorder and sleep quality. Frontiers in Psychiatry 2021, 12. [Google Scholar] [CrossRef]
- Zhang, K. , Liu, P., Liu, Z., Wang, J., Wang, J., Gao, M., Zhang, Y., Yang, C., Zhang, A., Li, G., Li, X., Liu, S., Liu, L., & Sun, N. (2023). Gut microbiota characteristics and its immunoregulatory role in inflammatory depression: joint clinical and animal data. Research Square (Research Square). [CrossRef]
- Zhang, W. , Xiao, D., Mao, Q., & Xia, H. Role of neuroinflammation in neurodegeneration development. Signal Transduction and Targeted Therapy 2023, 8. [Google Scholar] [CrossRef]
- Zheng, P. , Zeng, B., Zhou, C., Liu, M., Fang, Z., Xu, X., Zeng, L., Chen, J., Fan, S., Du, X., Zhang, X., Yang, D., Yang, Y., Meng, H., Li, W., Melgiri, N. D., Licinio, J., Wei, H., & Xie, P. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism. Molecular Psychiatry 2016, 21, 786–796. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




