Submitted:
24 January 2025
Posted:
24 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
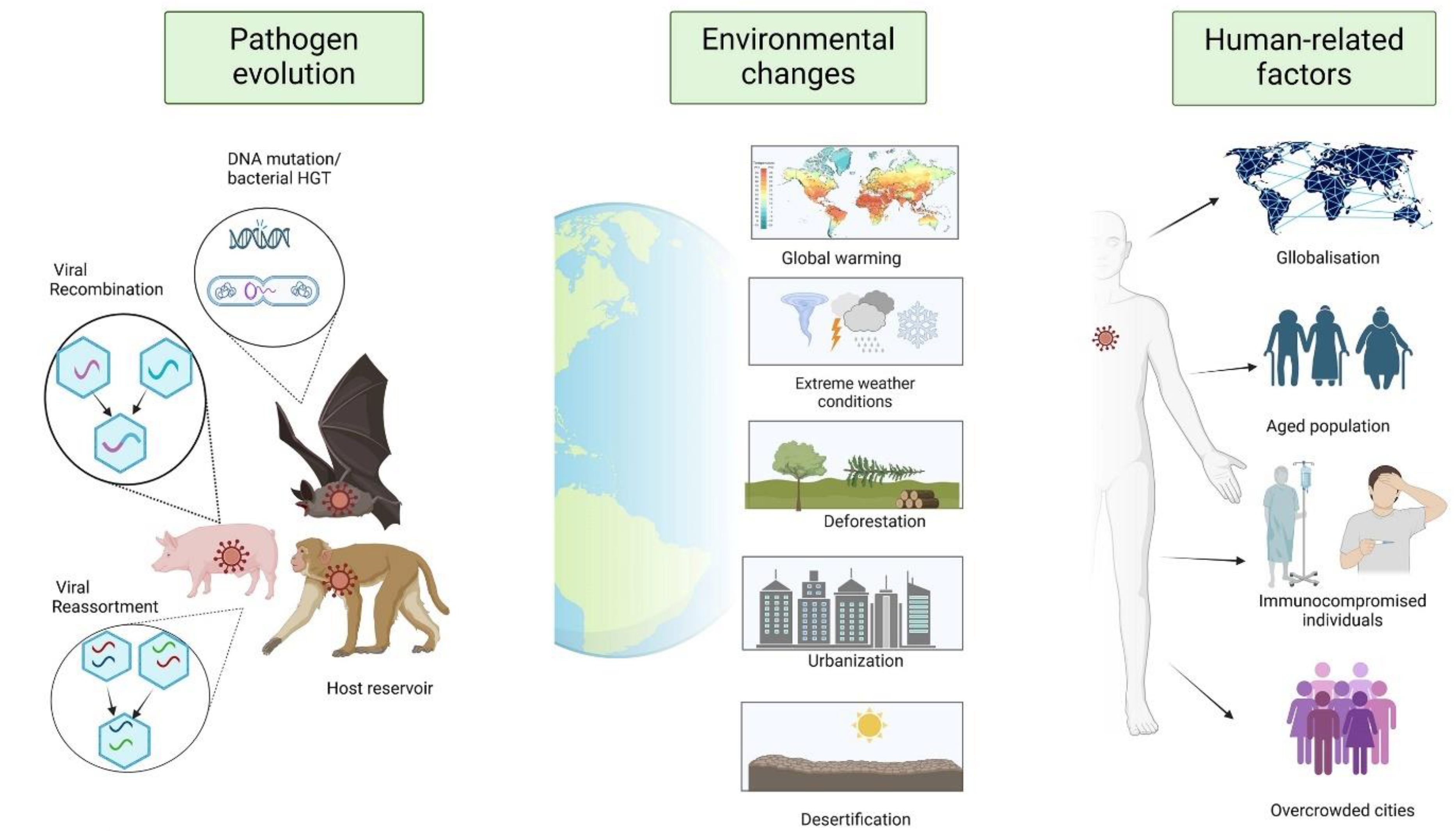
2. Emerging and Reemeerging Infectious Diseases: How and Why They Arise
3. Pathogens with Epidemic and Pandemic Potential
3.1. Zika Virus
3.2. SARS-CoV-2
3.3. Middle East Respiratory Disease (MERS) and Severe Acute Respiratory Syndrome (SARS)
3.4. Ebola Virus Disease and Marburg Virus Disease
3.5. Crimean-Congo Haemorrhagic Fever
3.6. RIFT VALLEY FEVER (RVF)
3.7. LASSA FEVER
3.8. “DISEASE X”
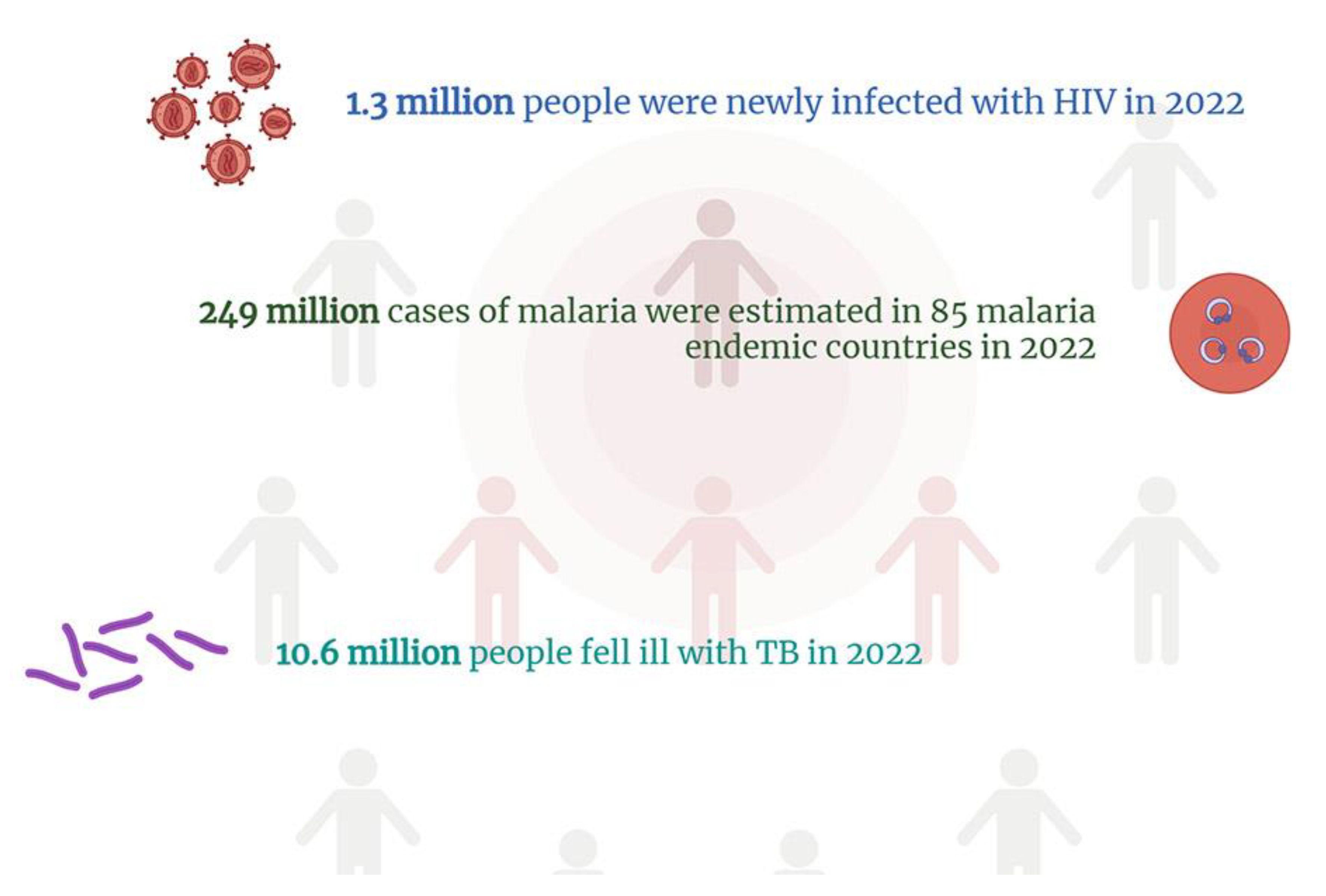
4. Overcoming Challenges: The Neglected Pandemics – HIV/AIDS, Tuberculosis and Malaria
4.1. HIV Infection and AIDS
4.2. Malaria's Impact Worldwide
4.3. Tuberculosis: Old Disease- New Threat
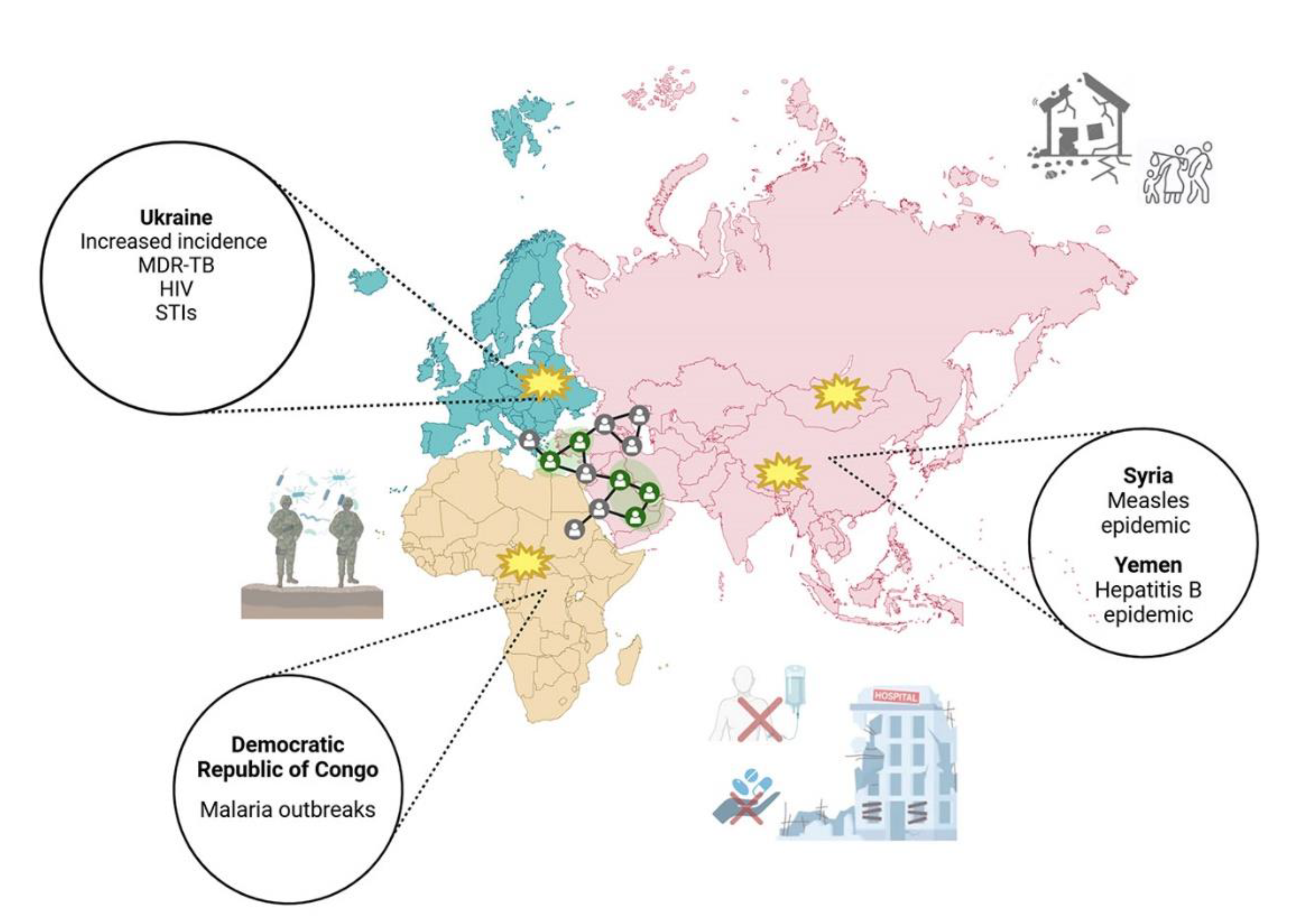
5. Conflict and Infectious Diseases
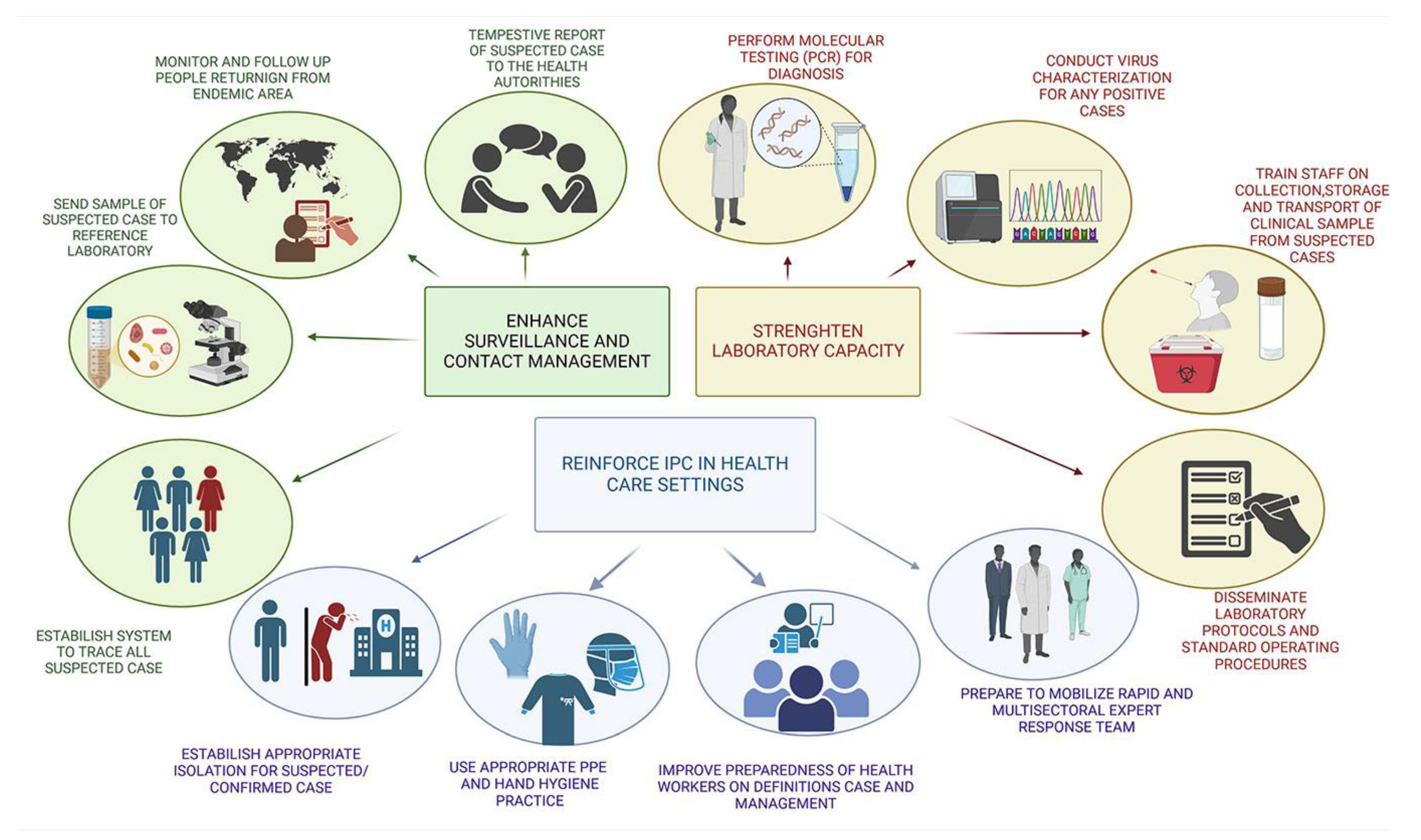
6. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Karlsson, E.K.; Kwiatkowski, D.P.; Sabeti, P.C. Natural selection and infectious disease in human populations. Nature reviews. Genetics 2014, 15, 379–393. [Google Scholar] [CrossRef] [PubMed]
- Seal, S.; Dharmarajan, G.; Khan, I. Evolution of pathogen tolerance and emerging infections: A missing experimental paradigm. eLife 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Li, W.; Wang, Z.; Yang, W.; Li, E.; Xia, X.; Yan, F.; Chiu, S. Emerging and reemerging infectious diseases: global trends and new strategies for their prevention and control. Signal transduction and targeted therapy 2024, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Ristori, M.V.; Guarrasi, V.; Soda, P.; Petrosillo, N.; Gurrieri, F.; Longo, U.G.; Ciccozzi, M.; Riva, E.; Angeletti, S. Emerging Microorganisms and Infectious Diseases: One Health Approach for Health Shared Vision. Genes 2024, 15. [Google Scholar] [CrossRef] [PubMed]
- Hauri, A.M.; Uphoff, H. [Tasks, principles and methods of applied infectious disease epidemiology/field epidemiology]. Bundesgesundheitsblatt, Gesundheitsforschung, Gesundheitsschutz 2005, 48, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Talisuna, A.O.; Okiro, E.A.; Yahaya, A.A.; Stephen, M.; Bonkoungou, B.; Musa, E.O.; Minkoulou, E.M.; Okeibunor, J.; Impouma, B.; Djingarey, H.M.; et al. Spatial and temporal distribution of infectious disease epidemics, disasters and other potential public health emergencies in the World Health Organisation Africa region, 2016-2018. Globalization and health 2020, 16, 9. [Google Scholar] [CrossRef]
- Gollakner, R.; Capua, I. Is COVID-19 the first pandemic that evolves into a panzootic? Veterinaria italiana 2020, 56, 7–8. [Google Scholar] [CrossRef]
- Biondo, C.; Ponzo, E.; Midiri, A.; Ostone, G.B.; Mancuso, G. The Dark Side of Nosocomial Infections in Critically Ill COVID-19 Patients. Life 2023, 13. [Google Scholar] [CrossRef]
- Cunha, C.B.; Opal, S.M. Middle East respiratory syndrome (MERS): a new zoonotic viral pneumonia. Virulence 2014, 5, 650–654. [Google Scholar] [CrossRef]
- Cherry, J.D.; Krogstad, P. SARS: the first pandemic of the 21st century. Pediatric research 2004, 56, 1–5. [Google Scholar] [CrossRef]
- Mena, I.; Nelson, M.I.; Quezada-Monroy, F.; Dutta, J.; Cortes-Fernandez, R.; Lara-Puente, J.H.; Castro-Peralta, F.; Cunha, L.F.; Trovao, N.S.; Lozano-Dubernard, B.; et al. Origins of the 2009 H1N1 influenza pandemic in swine in Mexico. eLife 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Sayres, L.; Hughes, B.L. Contemporary Understanding of Ebola and Zika Virus in Pregnancy. Clinics in perinatology 2020, 47, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Murphy, H.L.; Ly, H. Pathogenicity and virulence mechanisms of Lassa virus and its animal modeling, diagnostic, prophylactic, and therapeutic developments. Virulence 2021, 12, 2989–3014. [Google Scholar] [CrossRef] [PubMed]
- Cuomo-Dannenburg, G.; McCain, K.; McCabe, R.; Unwin, H.J.T.; Doohan, P.; Nash, R.K.; Hicks, J.T.; Charniga, K.; Geismar, C.; Lambert, B.; et al. Marburg virus disease outbreaks, mathematical models, and disease parameters: a systematic review. The Lancet. Infectious diseases 2024, 24, e307–e317. [Google Scholar] [CrossRef] [PubMed]
- Alhilfi, R.A.; Khaleel, H.A.; Raheem, B.M.; Mahdi, S.G.; Tabche, C.; Rawaf, S. Large outbreak of Crimean-Congo haemorrhagic fever in Iraq, 2022. IJID regions 2023, 6, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Youssouf, H.; Subiros, M.; Dennetiere, G.; Collet, L.; Dommergues, L.; Pauvert, A.; Rabarison, P.; Vauloup-Fellous, C.; Le Godais, G.; Jaffar-Bandjee, M.C.; et al. Rift Valley Fever Outbreak, Mayotte, France, 2018-2019. Emerging infectious diseases 2020, 26, 769–772. [Google Scholar] [CrossRef] [PubMed]
- Global Burden of Disease Health Financing Collaborator, N. Health sector spending and spending on HIV/AIDS, tuberculosis, and malaria, and development assistance for health: progress towards Sustainable Development Goal 3. Lancet 2020, 396, 693–724. [Google Scholar] [CrossRef]
- Bray, F.; Laversanne, M.; Cao, B.; Varghese, C.; Mikkelsen, B.; Weiderpass, E.; Soerjomataram, I. Comparing cancer and cardiovascular disease trends in 20 middle- or high-income countries 2000-19: A pointer to national trajectories towards achieving Sustainable Development goal target 3.4. Cancer treatment reviews 2021, 100, 102290. [Google Scholar] [CrossRef]
- Boutayeb, A. The double burden of communicable and non-communicable diseases in developing countries. Transactions of the Royal Society of Tropical Medicine and Hygiene 2006, 100, 191–199. [Google Scholar] [CrossRef]
- Boutayeb, A.; Boutayeb, S. The burden of non communicable diseases in developing countries. International journal for equity in health 2005, 4, 2. [Google Scholar] [CrossRef]
- Church, D.L. Major factors affecting the emergence and re-emergence of infectious diseases. Clinics in laboratory medicine 2004, 24, 559–586. [Google Scholar] [CrossRef] [PubMed]
- Morse, S.S. Factors in the emergence of infectious diseases. Emerging infectious diseases 1995, 1, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.M.; Martin-Granel, E. 2,500-year evolution of the term epidemic. Emerging infectious diseases 2006, 12, 976–980. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.T.; Liang, L.T.; Rosen, J.M. COVID-19: a comparison to the 1918 influenza and how we can defeat it. Postgraduate medical journal 2021, 97, 273–274. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.A.; Sankaran, N. Emergence of epidemic diseases: zoonoses and other origins. Faculty reviews 2022, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Ellwanger, J.H.; Veiga, A.B.G.; Kaminski, V.L.; Valverde-Villegas, J.M.; Freitas, A.W.Q.; Chies, J.A.B. Control and prevention of infectious diseases from a One Health perspective. Genetics and molecular biology 2021, 44, e20200256. [Google Scholar] [CrossRef] [PubMed]
- Ellwanger, J.H.; Chies, J.A.B. Zoonotic spillover: Understanding basic aspects for better prevention. Genetics and molecular biology 2021, 44, e20200355. [Google Scholar] [CrossRef]
- Rodriguez-Lazaro, D.; Cook, N.; Ruggeri, F.M.; Sellwood, J.; Nasser, A.; Nascimento, M.S.; D'Agostino, M.; Santos, R.; Saiz, J.C.; Rzezutka, A.; et al. Virus hazards from food, water and other contaminated environments. FEMS microbiology reviews 2012, 36, 786–814. [Google Scholar] [CrossRef]
- Endale, H.; Mathewos, M.; Abdeta, D. Potential Causes of Spread of Antimicrobial Resistance and Preventive Measures in One Health Perspective-A Review. Infection and drug resistance 2023, 16, 7515–7545. [Google Scholar] [CrossRef]
- Bonneaud, C.; Longdon, B. Emerging pathogen evolution: Using evolutionary theory to understand the fate of novel infectious pathogens. EMBO reports 2020, 21, e51374. [Google Scholar] [CrossRef]
- Holden, M.T.; Hauser, H.; Sanders, M.; Ngo, T.H.; Cherevach, I.; Cronin, A.; Goodhead, I.; Mungall, K.; Quail, M.A.; Price, C.; et al. Rapid evolution of virulence and drug resistance in the emerging zoonotic pathogen Streptococcus suis. PloS one 2009, 4, e6072. [Google Scholar] [CrossRef] [PubMed]
- LaTourrette, K.; Garcia-Ruiz, H. Determinants of Virus Variation, Evolution, and Host Adaptation. Pathogens 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Antunes, W.; Mota, S.; Madureira-Carvalho, A.; Dinis-Oliveira, R.J.; Dias da Silva, D. An Overview of the Recent Advances in Antimicrobial Resistance. Microorganisms 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, G.; Trinchera, M.; Midiri, A.; Zummo, S.; Vitale, G.; Biondo, C. Novel Antimicrobial Approaches to Combat Bacterial Biofilms Associated with Urinary Tract Infections. Antibiotics 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Howard, C.R.; Fletcher, N.F. Emerging virus diseases: can we ever expect the unexpected? Emerging microbes & infections 2012, 1, e46. [Google Scholar] [CrossRef]
- Enserink, M. Emerging infectious diseases. Nipah virus (or a cousin) strikes again. Science 2004, 303, 1121. [Google Scholar] [CrossRef]
- LeDuc, J.W. WHO program on emerging virus diseases. Archives of virology. Supplementum 1996, 11, 13–20. [Google Scholar] [CrossRef]
- McNicholl, J.M.; Downer, M.V.; Udhayakumar, V.; Alper, C.A.; Swerdlow, D.L. Host-pathogen interactions in emerging and re-emerging infectious diseases: a genomic perspective of tuberculosis, malaria, human immunodeficiency virus infection, hepatitis B, and cholera. Annual review of public health 2000, 21, 15–46. [Google Scholar] [CrossRef]
- Baker, R.E.; Mahmud, A.S.; Miller, I.F.; Rajeev, M.; Rasambainarivo, F.; Rice, B.L.; Takahashi, S.; Tatem, A.J.; Wagner, C.E.; Wang, L.F.; et al. Infectious disease in an era of global change. Nature reviews. Microbiology 2022, 20, 193–205. [Google Scholar] [CrossRef]
- Heffernan, C. Climate change and multiple emerging infectious diseases. Veterinary journal 2018, 234, 43–47. [Google Scholar] [CrossRef]
- Wikan, N.; Smith, D.R. Zika virus: history of a newly emerging arbovirus. The Lancet. Infectious diseases 2016, 16, e119–e126. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, E.B.; Kramer, L.D. Zika Virus Mosquito Vectors: Competence, Biology, and Vector Control. The Journal of infectious diseases 2017, 216, S976–S990. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.R.; Chen, T.H.; Hancock, W.T.; Powers, A.M.; Kool, J.L.; Lanciotti, R.S.; Pretrick, M.; Marfel, M.; Holzbauer, S.; Dubray, C.; et al. Zika virus outbreak on Yap Island, Federated States of Micronesia. The New England journal of medicine 2009, 360, 2536–2543. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Ortiz, K.; Ansari, A.; Gershwin, M.E. The Zika outbreak of the 21st century. Journal of autoimmunity 2016, 68, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.; Choi, G.K.; Yip, C.C.; Cheng, V.C.; Yuen, K.Y. Zika fever and congenital Zika syndrome: An unexpected emerging arboviral disease. The Journal of infection 2016, 72, 507–524. [Google Scholar] [CrossRef] [PubMed]
- Boyer, S.; Calvez, E.; Chouin-Carneiro, T.; Diallo, D.; Failloux, A.B. An overview of mosquito vectors of Zika virus. Microbes and infection 2018, 20, 646–660. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, F.M.E.; Pietrobon, A.J.; Oliveira, L.M.; Oliveira, L.; Sato, M.N. Maternal-Fetal Interplay in Zika Virus Infection and Adverse Perinatal Outcomes. Frontiers in immunology 2020, 11, 175. [Google Scholar] [CrossRef] [PubMed]
- Possas, C.; Brasil, P.; Marzochi, M.C.; Tanuri, A.; Martins, R.M.; Marques, E.T.; Bonaldo, M.C.; Ferreira, A.G.; Lourenco-de-Oliveira, R.; Nogueira, R.M.R.; et al. Zika puzzle in Brazil: peculiar conditions of viral introduction and dissemination - A Review. Memorias do Instituto Oswaldo Cruz 2017, 112, 319–327. [Google Scholar] [CrossRef]
- McCloskey, B.; Endericks, T. The rise of Zika infection and microcephaly: what can we learn from a public health emergency? Public health 2017, 150, 87–92. [Google Scholar] [CrossRef]
- Gardner, L.; Chen, N.; Sarkar, S. Vector status of Aedes species determines geographical risk of autochthonous Zika virus establishment. PLoS neglected tropical diseases 2017, 11, e0005487. [Google Scholar] [CrossRef]
- Talavera-Aguilar, L.G.; Murrieta, R.A.; Kiem, S.; Cetina-Trejo, R.C.; Baak-Baak, C.M.; Ebel, G.D.; Blitvich, B.J.; Machain-Williams, C. Infection, dissemination, and transmission efficiencies of Zika virus in Aedes aegypti after serial passage in mosquito or mammalian cell lines or alternating passage in both cell types. Parasites & vectors 2021, 14, 261. [Google Scholar] [CrossRef]
- Biondo, C.; Midiri, A.; Gerace, E.; Zummo, S.; Mancuso, G. SARS-CoV-2 Infection in Patients with Cystic Fibrosis: What We Know So Far. Life 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Worobey, M.; Levy, J.I.; Malpica Serrano, L.; Crits-Christoph, A.; Pekar, J.E.; Goldstein, S.A.; Rasmussen, A.L.; Kraemer, M.U.G.; Newman, C.; Koopmans, M.P.G.; et al. The Huanan Seafood Wholesale Market in Wuhan was the early epicenter of the COVID-19 pandemic. Science 2022, 377, 951–959. [Google Scholar] [CrossRef] [PubMed]
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmann, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. The Journal of hospital infection 2020, 104, 246–251. [Google Scholar] [CrossRef]
- Baj, J.; Karakula-Juchnowicz, H.; Teresinski, G.; Buszewicz, G.; Ciesielka, M.; Sitarz, R.; Forma, A.; Karakula, K.; Flieger, W.; Portincasa, P.; et al. COVID-19: Specific and Non-Specific Clinical Manifestations and Symptoms: The Current State of Knowledge. Journal of clinical medicine 2020, 9. [Google Scholar] [CrossRef]
- Filip, R.; Gheorghita Puscaselu, R.; Anchidin-Norocel, L.; Dimian, M.; Savage, W.K. Global Challenges to Public Health Care Systems during the COVID-19 Pandemic: A Review of Pandemic Measures and Problems. Journal of personalized medicine 2022, 12. [Google Scholar] [CrossRef]
- Singh, S.; McNab, C.; Olson, R.M.; Bristol, N.; Nolan, C.; Bergstrom, E.; Bartos, M.; Mabuchi, S.; Panjabi, R.; Karan, A.; et al. How an outbreak became a pandemic: a chronological analysis of crucial junctures and international obligations in the early months of the COVID-19 pandemic. Lancet 2021, 398, 2109–2124. [Google Scholar] [CrossRef]
- Jee, Y. WHO International Health Regulations Emergency Committee for the COVID-19 outbreak. Epidemiology and health 2020, 42, e2020013. [Google Scholar] [CrossRef]
- Chong, Z.X.; Liew, W.P.P.; Ong, H.K.; Yong, C.Y.; Shit, C.S.; Ho, W.Y.; Ng, S.Y.L.; Yeap, S.K. Current diagnostic approaches to detect two important betacoronaviruses: Middle East respiratory syndrome coronavirus (MERS-CoV) and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Pathology, research and practice 2021, 225, 153565. [Google Scholar] [CrossRef]
- Omrani, A.S.; Shalhoub, S. Middle East respiratory syndrome coronavirus (MERS-CoV): what lessons can we learn? The Journal of hospital infection 2015, 91, 188–196. [Google Scholar] [CrossRef]
- Rasmussen, S.A.; Gerber, S.I.; Swerdlow, D.L. Middle East respiratory syndrome coronavirus: update for clinicians. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America 2015, 60, 1686–1689. [Google Scholar] [CrossRef] [PubMed]
- Oraby, T.; Tyshenko, M.G.; Balkhy, H.H.; Tasnif, Y.; Quiroz-Gaspar, A.; Mohamed, Z.; Araya, A.; Elsaadany, S.; Al-Mazroa, E.; Alhelail, M.A.; et al. Analysis of the Healthcare MERS-CoV Outbreak in King Abdulaziz Medical Center, Riyadh, Saudi Arabia, June-August 2015 Using a SEIR Ward Transmission Model. International journal of environmental research and public health 2020, 17. [Google Scholar] [CrossRef] [PubMed]
- Pavli, A.; Tsiodras, S.; Maltezou, H.C. Middle East respiratory syndrome coronavirus (MERS-CoV): prevention in travelers. Travel medicine and infectious disease 2014, 12, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Omrani, A.S.; Al-Tawfiq, J.A.; Memish, Z.A. Middle East respiratory syndrome coronavirus (MERS-CoV): animal to human interaction. Pathogens and global health 2015, 109, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Khalafalla, A.I. Zoonotic diseases transmitted from the camels. Frontiers in veterinary science 2023, 10, 1244833. [Google Scholar] [CrossRef]
- Xu, R.H.; He, J.F.; Evans, M.R.; Peng, G.W.; Field, H.E.; Yu, D.W.; Lee, C.K.; Luo, H.M.; Lin, W.S.; Lin, P.; et al. Epidemiologic clues to SARS origin in China. Emerging infectious diseases 2004, 10, 1030–1037. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhao, S.; Teng, T.; Abdalla, A.E.; Zhu, W.; Xie, L.; Wang, Y.; Guo, X. Systematic Comparison of Two Animal-to-Human Transmitted Human Coronaviruses: SARS-CoV-2 and SARS-CoV. Viruses 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Wilder-Smith, A. The severe acute respiratory syndrome: impact on travel and tourism. Travel medicine and infectious disease 2006, 4, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Luu, B.; McCoy-Hass, V.; Kadiu, T.; Ngo, V.; Kadiu, S.; Lien, J. Severe Acute Respiratory Syndrome Associated Infections. Physician assistant clinics 2023, 8, 495–530. [Google Scholar] [CrossRef]
- Petrosillo, N.; Viceconte, G.; Ergonul, O.; Ippolito, G.; Petersen, E. COVID-19, SARS and MERS: are they closely related? Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases 2020, 26, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Breman, J.G.; Heymann, D.L.; Lloyd, G.; McCormick, J.B.; Miatudila, M.; Murphy, F.A.; Muyembe-Tamfun, J.J.; Piot, P.; Ruppol, J.F.; Sureau, P.; et al. Discovery and Description of Ebola Zaire Virus in 1976 and Relevance to the West African Epidemic During 2013-2016. The Journal of infectious diseases 2016, 214, S93–S101. [Google Scholar] [CrossRef] [PubMed]
- Den Boon, S.; Marston, B.J.; Nyenswah, T.G.; Jambai, A.; Barry, M.; Keita, S.; Durski, K.; Senesie, S.S.; Perkins, D.; Shah, A.; et al. Ebola Virus Infection Associated with Transmission from Survivors. Emerging infectious diseases 2019, 25, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Chiappelli, F.; Bakhordarian, A.; Thames, A.D.; Du, A.M.; Jan, A.L.; Nahcivan, M.; Nguyen, M.T.; Sama, N.; Manfrini, E.; Piva, F.; et al. Ebola: translational science considerations. Journal of translational medicine 2015, 13, 11. [Google Scholar] [CrossRef] [PubMed]
- Leroy, E.M.; Kumulungui, B.; Pourrut, X.; Rouquet, P.; Hassanin, A.; Yaba, P.; Delicat, A.; Paweska, J.T.; Gonzalez, J.P.; Swanepoel, R. Fruit bats as reservoirs of Ebola virus. Nature 2005, 438, 575–576. [Google Scholar] [CrossRef] [PubMed]
- El Ayoubi, L.W.; Mahmoud, O.; Zakhour, J.; Kanj, S.S. Recent advances in the treatment of Ebola disease: A brief overview. PLoS pathogens 2024, 20, e1012038. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Sharma, D.; Kumar, S.; Sharma, A.; Rijal, R.; Asija, A.; Adhikari, S.; Rustagi, S.; Sah, S.; Al-Qaim, Z.H.; et al. Emergence of Marburg virus: a global perspective on fatal outbreaks and clinical challenges. Frontiers in microbiology 2023, 14, 1239079. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.P.; Souris, M.; Valdivia-Granda, W. Global Spread of Hemorrhagic Fever Viruses: Predicting Pandemics. Methods in molecular biology 2018, 1604, 3–31. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, R.K.; Sarangi, A.K.; Kandi, V.; Chakraborty, S.; Chandran, D.; Alagawany, M.; Chakraborty, C.; Dhama, K. Recent re-emergence of Marburg virus disease in an African country Ghana after Guinea amid the ongoing COVID-19 pandemic: Another global threat? Current knowledge and strategies to tackle this highly deadly disease having feasible pandemic potential. International journal of surgery 2022, 106, 106863. [Google Scholar] [CrossRef] [PubMed]
- Okesanya, O.J.; Olatunji, G.D.; Kokori, E.; Olaleke, N.O.; Adigun, O.A.; Manirambona, E.; Lucero-Prisno, D.E. , 3rd. Looking Beyond the Lens of Crimean-Congo Hemorrhagic Fever in Africa. Emerging infectious diseases 2024, 30, 1319–1325. [Google Scholar] [CrossRef] [PubMed]
- Papa, A.; Tsergouli, K.; Tsioka, K.; Mirazimi, A. Crimean-Congo Hemorrhagic Fever: Tick-Host-Virus Interactions. Frontiers in cellular and infection microbiology 2017, 7, 213. [Google Scholar] [CrossRef] [PubMed]
- Kodama, C.; Alhilfi, R.A.; Aakef, I.; Khamasi, A.; Mahdi, S.; Hasan, H.M.; Khaleel, R.I.; Naji, M.M.; Esmaeel, N.K.; Haji-Jama, S.; et al. Epidemiological analysis and potential factors affecting the 2022-23 Crimean-Congo hemorrhagic fever outbreak in Iraq. European journal of public health 2025, 35, i6–i13. [Google Scholar] [CrossRef] [PubMed]
- Frank, M.G.; Weaver, G.; Raabe, V.; State of the Clinical Science Working Group of the National Emerging Pathogens, T. ; Education Center's Special Pathogens Research, N.; State of the Clinical Science Working Group of the National Emerging Pathogens Training Education Center's Special Pathogens Research, N. Crimean-Congo Hemorrhagic Fever Virus for Clinicians-Epidemiology, Clinical Manifestations, and Prevention. Emerging infectious diseases 2024, 30, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Alkan, C.; Jurado-Cobena, E.; Ikegami, T. Advancements in Rift Valley fever vaccines: a historical overview and prospects for next generation candidates. NPJ vaccines 2023, 8, 171. [Google Scholar] [CrossRef] [PubMed]
- Hartman, A. Rift Valley Fever. Clinics in laboratory medicine 2017, 37, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Kenawy, M.A.; Abdel-Hamid, Y.M.; Beier, J.C. Rift Valley Fever in Egypt and other African countries: Historical review, recent outbreaks and possibility of disease occurrence in Egypt. Acta tropica 2018, 181, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Reyna, R.A.; Littlefield, K.E.; Shehu, N.; Makishima, T.; Maruyama, J.; Paessler, S. The Importance of Lassa Fever and Its Disease Management in West Africa. Viruses 2024, 16. [Google Scholar] [CrossRef]
- Shieh, W.J.; Demby, A.; Jones, T.; Goldsmith, C.S.; Rollin, P.E.; Ksiazek, T.G.; Peters, C.J.; Zaki, S.R. Pathology and Pathogenesis of Lassa Fever: Novel Immunohistochemical Findings in Fatal Cases and Clinico-pathologic Correlation. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America 2022, 74, 1821–1830. [Google Scholar] [CrossRef] [PubMed]
- Marien, J.; Borremans, B.; Kourouma, F.; Baforday, J.; Rieger, T.; Gunther, S.; Magassouba, N.; Leirs, H.; Fichet-Calvet, E. Evaluation of rodent control to fight Lassa fever based on field data and mathematical modelling. Emerging microbes & infections 2019, 8, 640–649. [Google Scholar] [CrossRef]
- Lawrence, P.; Heung, M.; Nave, J.; Henkel, C.; Escudero-Perez, B. The natural virome and pandemic potential: Disease X. Current opinion in virology 2023, 63, 101377. [Google Scholar] [CrossRef] [PubMed]
- Bell, D.; Schultz Hansen, K. Relative Burdens of the COVID-19, Malaria, Tuberculosis, and HIV/AIDS Epidemics in Sub-Saharan Africa. The American journal of tropical medicine and hygiene 2021, 105, 1510–1515. [Google Scholar] [CrossRef]
- Davis, S.L. Human rights and the Global Fund to Fight AIDS, Tuberculosis, and Malaria. Health and human rights 2014, 16, 134–147. [Google Scholar] [PubMed]
- Strong, K.; Noor, A.; Aponte, J.; Banerjee, A.; Cibulskis, R.; Diaz, T.; Ghys, P.; Glaziou, P.; Hereward, M.; Hug, L.; et al. Monitoring the status of selected health related sustainable development goals: methods and projections to 2030. Global health action 2020, 13, 1846903. [Google Scholar] [CrossRef] [PubMed]
- Payagala, S.; Pozniak, A. The global burden of HIV. Clinics in dermatology 2024, 42, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Kharsany, A.B.; Karim, Q.A. HIV Infection and AIDS in Sub-Saharan Africa: Current Status, Challenges and Opportunities. The open AIDS journal 2016, 10, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.R.; Rao, A.; Rucinski, K.B.; Lyons, C.; Viswasam, N.; Comins, C.A.; Olawore, O.; Baral, S. HIV-Related Implementation Research for Key Populations: Designing for Individuals, Evaluating Across Populations, and Integrating Context. Journal of acquired immune deficiency syndromes 2019, 82 Suppl 3, S206–S216. [Google Scholar] [CrossRef]
- Ba Konko Cire, E.H.; Roh, M.E.; Diallo, A.; Gadiaga, T.; Seck, A.; Thiam, S.; Gaye, S.; Diallo, I.; Lo, A.C.; Diouf, E.; et al. Mass drug administration to reduce malaria incidence in a low-to-moderate endemic setting: short-term impact results from a cluster randomised controlled trial in Senegal. medRxiv : the preprint server for health sciences. [CrossRef]
- Oladipo, H.J.; Tajudeen, Y.A.; Oladunjoye, I.O.; Yusuff, S.I.; Yusuf, R.O.; Oluwaseyi, E.M.; AbdulBasit, M.O.; Adebisi, Y.A.; El-Sherbini, M.S. Increasing challenges of malaria control in sub-Saharan Africa: Priorities for public health research and policymakers. Annals of medicine and surgery 2022, 81, 104366. [Google Scholar] [CrossRef]
- Segala, F.V.; Di Gennaro, F.; Ictho, J.; L'Episcopia, M.; Onapa, E.; Marotta, C.; De Vita, E.; Amone, J.; Iacobelli, V.; Ogwang, J.; et al. Impact of antimalarial resistance and COVID-19 pandemic on malaria care among pregnant women in Northern Uganda (ERASE): protocol of a prospective observational study. BMC infectious diseases 2022, 22, 668. [Google Scholar] [CrossRef]
- Falzon, D.; Zignol, M.; Bastard, M.; Floyd, K.; Kasaeva, T. The impact of the COVID-19 pandemic on the global tuberculosis epidemic. Frontiers in immunology 2023, 14, 1234785. [Google Scholar] [CrossRef]
- A spotlight on the tuberculosis epidemic in South Africa. Nature communications 2024, 15, 1290. [CrossRef]
- Collaborators, G.B.D.T. Global, regional, and national age-specific progress towards the 2020 milestones of the WHO End TB Strategy: a systematic analysis for the Global Burden of Disease Study 2021. The Lancet. Infectious diseases 2024, 24, 698–725. [Google Scholar] [CrossRef]
- Yenet, A.; Nibret, G.; Tegegne, B.A. Challenges to the Availability and Affordability of Essential Medicines in African Countries: A Scoping Review. ClinicoEconomics and outcomes research : CEOR 2023, 15, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Sundermann, A.J.; Kumar, P.; Griffith, M.P.; Waggle, K.D.; Srinivasa, V.R.; Raabe, N.; Mills, E.G.; Coyle, H.; Ereifej, D.; Creager, H.M.; et al. Genomic Surveillance for Enhanced Healthcare Outbreak Detection and Control. medRxiv : the preprint server for health sciences 2024. [CrossRef]
- Nerlander, L.; Champezou, L.; Gomes Dias, J.; Aspelund, G.; Berlot, L.; Constantinou, E.; Diaz, A.; Epstein, J.; Fogarassy, E.; Hernando, V.; et al. Sharp increase in gonorrhoea notifications among young people, EU/EEA, July 2022 to June 2023. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin 2024, 29. [Google Scholar] [CrossRef] [PubMed]
- Marou, V.; Vardavas, C.I.; Aslanoglou, K.; Nikitara, K.; Plyta, Z.; Leonardi-Bee, J.; Atkins, K.; Condell, O.; Lamb, F.; Suk, J.E. The impact of conflict on infectious disease: a systematic literature review. Conflict and health 2024, 18, 27. [Google Scholar] [CrossRef]
- Mehtar, S.; AlMhawish, N.; Shobak, K.; Reingold, A.; Guha-Sapir, D.; Haar, R.J. Measles in conflict-affected northern Syria: results from an ongoing outbreak surveillance program. Conflict and health 2021, 15, 95. [Google Scholar] [CrossRef] [PubMed]
- Gebreyohannes, E.A.; Wolde, H.F.; Akalu, T.Y.; Clements, A.C.A.; Alene, K.A. Impacts of armed conflicts on tuberculosis burden and treatment outcomes: a systematic review. BMJ open 2024, 14, e080978. [Google Scholar] [CrossRef] [PubMed]
- Kvasnevska, Y.; Faustova, M.; Voronova, K.; Basarab, Y.; Lopatina, Y. Impact of war-associated factors on spread of sexually transmitted infections: a systemic review. Frontiers in public health 2024, 12, 1366600. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



