Submitted:
23 January 2025
Posted:
23 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Patients and Methods
- 1.
- Aims and objectives
- 2.
- Study design
- 3.
- Data collection
- 4.
- Definitions
- (1)
- Obesity (Body mass index ≥ 30 kg/m 2).
- (2)
- Acute or chronic kidney disease (presence at diagnosis of plasma creatinine ≥ 2 mg/dL due to acute kidney injure, or as the baseline creatinine in patients with chronic kidney disease).
- (3)
- Chronic obstructive pulmonary disease.
- (4)
- Bronchial asthma.
- (5)
- Hypertension (HTN).
- (6)
- Type 2 diabetes mellitus (T2D).
- (7)
- Cardiovascular events or cardiovascular risk factors: arrhythmia, infarction, cardiomyopathy, heart failure or coronary artery disease or the combination of HTN, T2D and dyslipidemia as cardiovascular risk factors.
- (8)
- Altered liver enzymes (elevation of liver enzyme (aminotransferase) above the upper limit of normal).
- (1)
- A positive real-time polymerase chain reaction (RT-PCR) test for SARS-CoV-2.
- (2)
- A positive antigen detection test [21].
- (3)
- Patients with compatible symptoms and radiological findings suggestive of COVID-19, during the first weeks of the study, prior to the establishment of diagnostic tests [22].
- (4)
- (1)
- Required hospitalization.
- (2)
- Required admission to the Intensive Care Unit (ICU).
- (3)
- Associated respiratory failure: baseline oxygen saturation ≤ 93%, partial blood pressure of oxygen (PaO2) / oxygen concentration (FiO2) ≤ 30 mmHg [25]. Need for non-invasive ventilatory support.
- (4)
- Required invasive mechanical ventilation (IMV).
- (5)
- Died from COVID-19 disease.
3. Study Time
- -
- First period: From March 1st, 2020, to June 21st, 2020, the date on which the state of alarm in Spain ended once the first epidemic wave of COVID-19 ended.
- -
- Second period: From June 22nd to December 6th, 2020.
- -
- Third period: From December 7th, 2020, to March 14th, 2021.
- -
- Fourth period: From March 15th, 2021, to June 19th, 2021.
- -
- Fifth period: From June 20th, 2021, to October 13rd, 2021.
- -
- Sixth period: From October 14th, 2021, to March 27 th, 2022.
- -
- Seventh period: From March 28th, 2022, to December 31st, 2023.
4. Inclusion Criteria
5. Exclusion Criteria
6. Statistital Analysis
- 1.
- Characteristics of the study population
- 2.
- Symptoms
- 3.
- Immunosupression
- 4.
- Diagnosis of COVID-19 disease
- 5.
- Treatment for COVID-19 disease
- 6.
- Vaccination
- 7.
- Severe COVID-19 disease
| Table 4. Respiratory failure in LTR diagnosed with COVID-19 | Logistic Regression Analysis | |||||
| Respiratory failure | ||||||
| Total (N = 307) | No (N = 273) | Yes (N = 34) | p-value | OR (p-value) | 95%CI | |
| Age (years), median (IQR) | 61 (54 – 68) | 61 (53 – 68) | 68 (59 – 73) | 0.002 | 1.05 (0.002) | 1.02 - 1.09 |
| IMC (kg/m2), median (IQR) | 26.3 (23 – 29) | 26.05 (22.7 – 29) | 28 (25.5 – 29.7) | 0.026 | OR 1.09 (0.017) | 1.017 - 1.18 |
| Male sex, N (%) | 209 (68.08%) | 180 (65.93%) | 29 (85.29%) | 0.022 | ||
| Female sex, N (%) | 98 (31.92%) | 93 (34.07%) | 5 (14.71%) | 0.33 (0.028) | 0.12 - 0.89 | |
| Number of vaccines at diagnosis | 3 (0 – 3) | 3 (0 – 3) | 0 (0 – 2) | < 0.001 | 0.52 (< 0.001) | 0.39 - 0. 69 |
| Vaccination N (%) | 211 (68.95%) | 199 (73.16%) | 12 (35.29%) | < 0.001 | 0.2 (< 0.001) | 0.094 - 0.42 |
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 5.87 (3.23 – 9.03) | 6.46 (3.31-9.11) | 2.89 (0.62-4.36) | 0.003 | 0.7 (0.011) | 0.54 - 0.92 |
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 94.46 (41.44-185.21) | 94.46 (41.44-184.49) | 95.08 (51.28-188.75) | 0.79 | ||
| Kidney failure(Creatinine≥ 2 mg/dL) | 23 (7.52%) | 16 (5.88%) | 7 (20.59%) | 0.002 | 4.12 (0.004) | 1.57 - 10.97 |
| COPD, N (%) | 13 (4.25%) | 10 (3.68%) | 3 (8.82%) | 0.16 | ||
| Asthma, N (%) | 6 (1.96%) | 6 (2.21%) | 0 | 0.38 | ||
| HTN, N (%) | 117 (38.24%) | 94 (34.56%) | 23 (67.65%) | < 0.001 | 3.96 (< 0.001) | 1.85 - 8.47 |
| Cardiovascular events and / or risk factors | 181 (59.34%) | 152 (56.09%) | 29 (85.29%) | 0.001 | 4.54 (0.002) | 1.7 - 12 |
| T2D, N (%) | 125 (40.85%) | 102 (37.50%) | 23 (67.65%) | < 0.001 | 3.48 (< 0.001) | 1.63 - 7.45 |
| Altered liver enzymes, N (%) | 20 (6.54%) | 16 (6.81%) | 4 (5.63%) | 0.73 | ||
| Smokers, N (%) | 50 (16.72%) | 44 (16.54%) | 6 (18.18%) | 0.53 | ||
| Asthenia, N (%) | 98 (32.45%) | 81 (30.22%) | 17 (50.00%) | 0.02 | 2.31 (0.023) | 1.12 - 4.75 |
| Dyspnea, N (%) | 45 (14.9 %) | 25 (9.33%) | 20 (58.82%) | < 0.001 | 13.88 (< 0.001) | 6.25 - 30.82 |
| Fever, N (%) | 77 (25.5 %) | 59 (22.01%) | 18 (52.94%) | < 0.001 | 3.98 (< 0.001) | 1.92 - 8.29 |
| Cough, N (%) | 120 (39.74%) | 103 (38.43%) | 17 (50 %) | 0.19 | ||
| Chest pain, N (%) | 12 (3.97%) | 9 (3.36%) | 3 (8.82%) | 0.12 | ||
| Diarrhoea, N (%) | 32 (10.6 %) | 27 (10.07%) | 5 (14.71%) | 0.41 | ||
| Anorexia, N (%) | 14 (4.64%) | 10 (3.73%) | 4 (11.76%) | 0.036 | 3.44 (0.047) | 1.02 - 11.65 |
| Chest X-ray findings,N (%) | <0.001 | |||||
| Not performed | 191 (69.45%) | 189 (77.78%) | 2 (6.25%) | |||
| Normal | 28 (10.18%) | 27 (11.11%) | 1 (3.13%) | |||
| Bilateral pneumonia | 46 (16.73%) | 21 (8.64%) | 25 (78.13%) | 112.5 (< 0.001) | 24.87 – 508.84 | |
| Lobar pneumonia | 10 (3.64%) | 6 (2.47%) | 4 (12.50%) | 63 (< 0.001) | 9.59 – 413.67 | |
| Azathioprine, N (%) | 3 (0.98%) | 2 (0.74%) | 1 (2.94%) | 0.22 | ||
| Corticosteroids, N (%) | 17 (5.56%) | 17 (6.25%) | 0 | 0.13 | ||
| mTORi, N (%) | 40 (13.07%) | 35 (12.87%) | 5 (14.71%) | 0.76 | ||
| MMF, N (%) | 184 (60.13%) | 154 (56.62%) | 30 (88.24%) | < 0.001 | 5.74 (< 0.001) | 1.97 - 16.76 |
| CNIs, N (%) | 189 (61.76%) | 177 (65.07%) | 12 (35.29%) | < 0.001 | 0.29 (< 0.001) | 0.14 - 0.62 |
| Table 5. IMV in LTR diagnosed with COVID-19 | Logistic Regression Analysis | |||||
| Mechanical ventilation | ||||||
| Total (N = 306, 1 lost case) | No (N = 301) | Yes (N = 5) | p-value | OR (p-value) | 95%CI | |
| Age (years), median (IQR) | 61 (54 – 68) | 61 (53 – 68) | 68 (62 – 74) | 0.1 | ||
| IMC (kg/m2), median (IQR) | 26.3 (23 – 29) | 26.3 (22.7 – 29) | 28 (25.5 – 29.7) | 0.21 | ||
| Male sex, N (%) | 208 (67.97%) | 203 (67.44%) | 5 (100 %) | 0.12 | ||
| Female sex, N (%) | 98 (32.03%) | 98 (32.56%) | 0 | |||
| Number of vaccines at diagnosis | 3 (0 – 3) | 3 (0 – 3) | 0 (0 – 2) | 0.2 | ||
| Vaccination N (%) | 211 (69.18%) | 209 (69.67%) | 2 (40.00%) | 0.15 | ||
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 5.87 (3.23 - 9.03) | 5.87 (3.25 - 9.02) | 4.61 (0.1 – 9.11) | 0.56 | ||
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 94.72 (43.31-185.21) | 94.46 (43.31-184.49) | 134.75 (78.23-191.93) | 0.48 | ||
| Kidney failure (Creatinine≥ 2 mg/dL) | 23 (7.54%) | 23 (7.67%) | 0 | 0.52 | ||
| COPD, N (%) | 13 (4.26%) | 13 (4.33%) | 0 | 0.63 | ||
| Asthma, N (%) | 6 (1.97%) | 6 (2 %) | 0 | 0.75 | ||
| HTN, N (%) | 117 (38.36%) | 114 (38.00%) | 3 (60 %) | 0.32 | ||
| Cardiovascular events and / or risk factors | 180 (59.21%) | 176 (58.86%) | 4 (80 %) | 0.34 | ||
| T2D, N (%) | 124 (40.66%) | 119 (39.67%) | 5 (100 %) | 0.006 | ||
| Altered liver enzymes, N (%) | 20 (6.56%) | 20 (6.67%) | 0 | 0.55 | ||
| Smokers, N (%) | 50 (16.78%) | 49 (16.72%) | 1 (20 %) | 0.94 | ||
| Asthenia, N (%) | 98 (32.56%) | 96 (32.43%) | 2 (40 %) | 0.72 | ||
| Dyspnea, N (%) | 45 (14.95%) | 41 (13.85%) | 4 (80 %) | < 0.001 | 24.87 (0.004) | 2.71 – 228.14 |
| Fever, N (%) | 77 (25.58%) | 74 (25.00%) | 3 (60 %) | 0.075 | ||
| Cough, N (%) | 120 (39.87%) | 119 (40.20%) | 1 (20 %) | 0.36 | ||
| Chest pain, N (%) | 12 (3.99%) | 12 (4.05%) | 0 | 0.65 | ||
| Diarrhoea, N (%) | 32 (10.63%) | 31 (10.47%) | 1 (20 %) | 0.49 | ||
| Anorexia, N (%) | 14 (4.65%) | 14 (4.73%) | 0 | 0.62 | ||
| Chest X-ray findings,N (%) | 0.008 | |||||
| Not performed | 191 (69.71%) | 190 (70.63%) | 1 (20%) | |||
| Normal | 28 (10.22%) | 28 (10.41%) | 0 | |||
| Bilateral pneumonia | 45 (16.42%) | 42 (15.61%) | 3 (60%) | 13.57 (0.025) | 1.37 – 133.71 | |
| Lobar pneumonia | 10 (3.65%) | 9 (3.35%) | 1 (20%) | 21.11 (0.036) | 1.22 – 365.44 | |
| Azathioprine, N (%) | 3 (0.98%) | 2 (0.67%) | 1 (20%) | < 0.001 | 37.25 (0.006) | 2.78 – 499.16 |
| Corticosteroids, N (%) | 17 (5.57%) | 17 (5.67%) | 0 ( | 0.58 | ||
| mTORi, N (%) | 40 (13.11%) | 40 (13.33%) | 0 | 0.38 | ||
| MMF, N (%) | 183 (60%) | 179 (59.67%) | 4 (80%) | 0.36 | ||
| CNIs, N (%) | 188 (61.64%) | 186 (62%) | 2 (40%) | 0.32 | ||
| Table 6. Hospital admission in LTR diagnosed with COVID-19 | Logistic Regression Analysis | |||||
| Hospital admission | ||||||
| Total (N = 307) | No (N = 235) | Yes (N = 72) | p-value | OR (p-value) | 95%CI | |
| Age (years), median (IQR) | 61 (54 – 68) | 60 (52 – 67) | 64.5 (56 – 71.5) | 0.002 | 1.04 (p = 0.001) | 1.02 – 1-07 |
| IMC (kg/m2), median (IQR) | 26.3 (23 – 29) | 26.1 (22.5 – 29) | 27.32 (24.2 – 29.7) | 0.009 | 1.08 (p = 0.011) | 1.02 – 1.15 |
| Male sex, N (%) | 209 (68.08%) | 156 (66.38%) | 53 (73.61%) | 0.25 | ||
| Female sex, N (%) | 98 (31.92%) | 79 (33.62%) | 19 (26.39%) | |||
| Number of vaccines at diagnosis | 3 (0 – 3) | 3 (1 – 4) | 0 (0 – 3) | < 0.001 | 0.63 (p < 0.001) | 0.53 – 0.75 |
| Vaccination N (%) | 211 (68.95%) | 178 (76.07%) | 33 (45.83%) | < 0.001 | 0.23 (p < 0.001) | 0.15 – 0.46 |
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 5.87 (3.23 – 9.03) | 6.26 (3.28 – 9.18) | 3.61 (2.2 – 6.79) | 0.073 | ||
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 94.46 (41.44-185.21) | 93.05 (40.85-177.64) | 101.98 (43.74-192.44) | 0.26 | ||
| Kidney failure (Creatinine≥ 2 mg/dL) | 23 (7.52%) | 12 (5.13%) | 11 (15.28%) | 0.004 | 3.33 (p = 0.006) | 1.4 – 7.93 |
| COPD, N (%) | 13 (4.25%) | 7 (2.98%) | 6 (8.45%) | 0.0.45 | 3 (p = 0.055) | 0.97 – 9.26 |
| Asthma, N (%) | 6 (1.96%) | 6 (2.55%) | 0 | 0.17 | ||
| HTN, N (%) | 117 (38.24%) | 78 (33.19%) | 39 (54.93%) | < 0.001 | 2.45 (p = 0.001) | 1.43 – 4.21 |
| Cardiovascular events and / or risk factors | 181 (59.34%) | 127 (54.27%) | 54 (76.06%) | 0.001 | 2.67 (p = 0.001) | 1.46 – 4. 89 |
| T2D, N (%) | 125 (40.85%) | 85 (36.17%) | 40 (56.34%) | 0.002 | 2.28 (p = 0.003) | 1.33 – 3.9 |
| Altered liver enzymes, N (%) | 20 (6.54%) | 16 (6.81%) | 4 (5.63%) | 0.73 | ||
| Smokers, N (%) | 50 (16.72%) | 41 (17.75%) | 9 (13.24%) | 0.14 | ||
| Asthenia, N (%) | 98 (32.45%) | 70 (30.43%) | 28 (38.89%) | 0.18 | ||
| Dyspnea, N (%) | 73 (24.17%) | 62 (26.96%) | 11 (15.28%) | 0.043 | 24.56 (p < 0.001) | 10.91 – 55.25 |
| Fever, N (%) | 77 (25.50%) | 41 (17.83%) | 36 (50.00%) | < 0.001 | 4.61 (p < 0.001) | 2.6 – 8.17 |
| Cough, N (%) | 120 (39.74%) | 80 (34.78%) | 40 (55.56%) | 0.002 | 2.34 (p = 0.002) | 1.37 – 4.01 |
| Chest pain, N (%) | 12 (3.97%) | 3 (1.30%) | 9 (12.50%) | < 0.001 | 10.81 (p < 0.001) | 2.84 – 41.12 |
| Diarrhoea, N (%) | 32 (10.60%) | 15 (6.52%) | 17 (23.61%) | < 0.001 | 4.43 (p < 0.001) | 2.08 – 9.42 |
| Anorexia, N (%) | 14 (4.64%) | 8 (3.48%) | 6 (8.33%) | 0.087 | ||
| Chest X-ray findings,N (%) | <0.001 | |||||
| Not performed | 191 (69.45%) | 182 (87.92%) | 9 (13.24%) | |||
| Normal | 28 (10.18%) | 19 (9.18%) | 9 (13.24%) | |||
| Bilateral pneumonia | 46 (16.73%) | 4 (1.93%) | 42 (61.76%) | 212.33 (p < 0.001) | 3.39 – 27.04 | |
| Lobar pneumonia | 10 (3.64%) | 2 (0.97%) | 8 (11.76%) | 80.89 (p < 0.001) | 14.96 – 437.44 | |
| Azathioprine, N (%) | 3 (0.98%) | 1 (0.43%) | 2 (2.78%) | 0.077 | ||
| Corticosteroids, N (%) | 17 (5.56%) | 14 (5.98%) | 3 (4.17%) | 0.56 | ||
| mTORi, N (%) | 40 (13.07%) | 23 (9.83%) | 17 (23.61%) | 0.002 | 2.84 (p = 0.003) | 1.42 – 5.67 |
| MMF, N (%) | 184 (60.13%) | 132 (56.41%) | 52 (72.22%) | 0.017 | 2.01 (p = 0.018) | 1.13 – 3.58 |
| CNIs, N (%) | 189 (61.76%) | 155 (66.24%) | 34 (47.22%) | 0.004 | 0.46 (p = 0.004) | 0.27 – 0.78 |
| Table 7.ICU admission in LTR diagnosed with COVID-19 | Logistic Regression Analysis | |||||
| Total (N = 306, 1 lost case) | Admission to the ICU | |||||
| No (N = 301) | Yes (N = 5) | p-value | OR (p-value) | 95%CI | ||
| Age (years), median (IQR) | 61 (54 – 68) | 61 (53 – 68) | 60 (62 – 74) | 0.1 | ||
| IMC (kg/m2), median (IQR) | 26.3 (23 – 29) | 26.3 (23 – 29) | 29 (25.5 – 34.4) | 0.21 | ||
| Male sex, N (%) | 208 (67.97%) | 203 (67.44%) | 5 (100%) | 0.12 | ||
| Female sex, N (%) | 98 (32.03%) | 98 (32.56%) | 0 | |||
| Number of vaccines at diagnosis | 3 (0 – 3) | 3 (0 – 3) | 0 (0 – 3) | 0.2 | ||
| Vaccination N (%) | 211 (69.18%) | 209 (69.67%) | 2 (40%) | < 0.001 | ||
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 5.87 (3.23 - 9.03) | 5.87 (3.25-9.02) | 4.61 (0.10-9.11) | 0.56 | ||
| Time (months) from vaccination to the onset of symptoms, median (IQR) | 94.46 (41.44-185.21) | 95.28 (45.51-185.31) | 46.26 (24.16-137.70) | 0.12 | ||
| Kidney failure (Creatinine≥ 2 mg/dL) | 23 (7.54%) | 23 (7.67%) | 0 | 0.52 | ||
| COPD, N (%) | 13 (4.26%) | 13 (4.33%) | 0 | 0.63 | ||
| Asthma, N (%) | 6 (1.97%) | 6 (2.00%) | 0 | 0.75 | ||
| HTN, N (%) | 117 (38.36%) | 114 (38%) | 3 (60%) | 0.32 | ||
| Cardiovascular events and / or risk factors | 180 (59.21%) | 176 (58.86%) | 4 (80.00%) | 0.34 | ||
| T2D, N (%) | 124 (40.66%) | 119 (39.67%) | 5 (100%) | 0.006 | ||
| Altered liver enzymes, N (%) | 20 (6.54%) | 19 (6.44%) | 1 (9.09%) | 0.73 | ||
| Smokers, N (%) | 50 (16.78%) | 49 (16.72%) | 1 (20%) | 0.94 | ||
| Asthenia, N (%) | 98 (32.56%) | 96 (32.43%) | 2 (40%) | 0.72 | ||
| Dyspnea, N (%) | 45 (14.95%) | 41 (13.85%) | 4 (80.00%) | < 0.001 | 21.17 (< 0.001) | 5.47 – 81.85 |
| Fever, N (%) | 77 (25.58%) | 74 (25.00%) | 3 (60.00%) | 0.075 | 4.4 (0.014) | 1.35 – 14.3 |
| Cough, N (%) | 120 (39.87%) | 119 (40.2%) | 1 (20%) | 0.36 | ||
| Chest pain, N (%) | 12 (3.99%) | 12 (4.05%) | 0 | 0.65 | ||
| Diarrhoea, N (%) | 32 (10.63%) | 31 (10.47%) | 1 (20%) | 0.49 | ||
| Anorexia, N (%) | 14 (4.65%) | 14 (4.73%) | 0 | 0.62 | ||
| Chest X-ray findings,N (%) | 0.008 | |||||
| Not performed | 191 (69.71%) | 190 (70.63%) | 1 (20%) | |||
| Normal | 28 (10.22%) | 28 (10.41%) | 0 | |||
| Bilateral pneumonia | 45 (16.42%) | 42 (15.61%) | 3 (60%) | 28.5 (0.002) | 3.34 – 243.27 | |
| Lobar pneumonia | 10 (3.65%) | 9 (3.35%) | 1 (20%) | 21.11 (0.036) | 1.22 – 365.44) | |
| Azathioprine, N (%) | 3 (0.98%) | 2 (0.67%) | 1 (20%) | < 0.001 | 13.27 (0.041) | 1.12 – 157.67 |
| Corticosteroids, N (%) | 17 (5.57%) | 17 (5.67%) | 0 | 0.58 | ||
| mTORi, N (%) | 40 (13.11%) | 40 (13.33%) | 0 | 0.38 | ||
| MMF, N (%) | 183 (60%) | 179 (59.67%) | 4 (80%) | 0.36 | ||
| CNIs, N (%) | 188 (61.64%) | 186 (62%) | 2 (40%) | 0.32 | ||
| Table 8. Multivariate analysis | ||||
| Respiratory failure | Hospital admission | |||
| Area under ROC curve = 0.851 | Area under ROC curve = 0.896 | |||
| OR (p-value) | 95%CI | OR (p-value) | 95%CI | |
| Age | 1.056 (0.045) | 1 – 1.1 | ||
| Vaccination | 0.16 (< 0.001) | 0.072 – 0.37 | 0.2 (< 0.001) | 0.09 – 0.44 |
| Kidney injury | 5.33 (0.006) | 1.62 – 17.52 | 4.29 (0.013) | 1.35 – 13.58 |
| HTN | 3.69 (0.002) | 1.61 – 8.45 | 3.25 (0.002) | 1.54 – 6.89 |
| Dyspnea at diagnosis | 18.83 (< 0.001) | 7.61 – 46.56 | ||
| Fever at diagnosis | 4.08 (< 0.001) | 1.9 – 8.74 | ||
| mTORi | 2.8 (0.036) | 1.07 – 7.32 | ||
| MMF | 2.73 (0.008) | 1.49 – 14.71 | ||
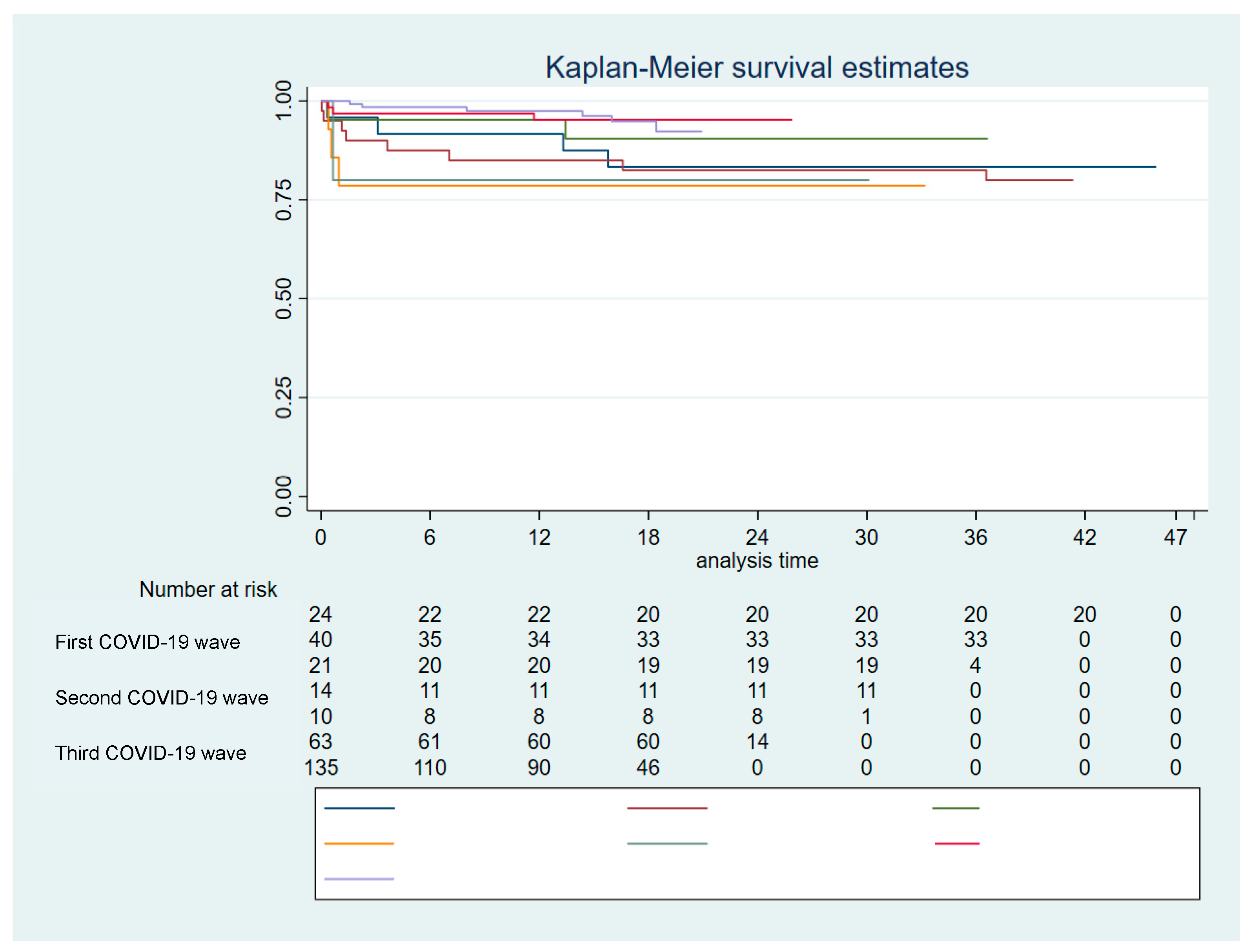
| Table 9. Death from COVID-19 | ||||||
| Death due to COVID-19 | Univariant survival analysis | |||||
| Total (N=307) | No (N = 295) | Yes (N = 12) | p-value | Hazard ratio (p-value) | 95% CI | |
| Age (years), median (IQR) | 61 (54.00-68.00) | 61(53-68) | 68 (61.5-71.5) | 0.034 | ||
| Age (years), mean (SD) | 59.03 (14.03) | 58.72 (14.16) | 66.50 (7.24) | 0.06 | ||
| IMC (kg/m2), median (IQR) | 26.30 (23 – 29) | 26.10(22.9 - 29) | 29.05 (26.8 - 35.25) | 0.008 | ||
| IMC (kg/m2), mean (SD) | 26.35 (4.68) | 26.17 (4.56) | 30.54 (5.59) | 0.001 | ||
| Obesity, N (%) | 62 (20.67%) | 56 (19.44%) | 6 (50.00%) | 0.01 | 3.94 (0.002) | 1.27 - 12.22 |
| Male sex, N (%) | 209 (68.08%) | 199 (67.46%) | 10 (83.33%) | 0.25 | ||
| Doses of vaccination at diagnosis | 3 (0 – 3) | 3 (0 – 3) | 0 (0 – 1.5) | 0.002 | 0.56 (0.006) | 0.32 - 0.83 |
| Vaccine (doses) | 0.062 | |||||
| 0 | 93 (30.39%) | 85 (28.91%) | 8 (66.67%) | |||
| 1 | 16 (5.23%) | 15 (5.1%) | 1 (8.33%) | |||
| 2 | 38 (12.42%) | 36 (12.24%) | 2 (16.67%) | |||
| 3 | 92 (30.07%) | 91 (30.95%) | 1 (8.33%) | |||
| 4 | 54 (17.65%) | 54 (18.37%) | 0 | |||
| 5 | 13 (4.25%) | 13 (4.42%) | 0 | |||
| Kidney injury (creatinine ≥ 2 mg/dL) | 23 (7.52%) | 19 (6.46%) | 4 (33.33%) | <0.001 | 6.9 (0.002) | 2.08 - 23.02 |
| COPD, N (%) | 13 (4.25%) | 11 (3.74%) | 2 (16.67%) | 0.030 | 4.64 (0.047) | 1.02 - 21.22 |
| Asthma, N (%) | 6 (1.96%) | 6 (2.04%) | 0 (0.00%) | 0.62 | ||
| HTN, N (%) | 117 (38.24%) | 109 (37.07%) | 8 (66.67%) | 0.039 | ||
| Cardiovascular events and / or risk factors | 181 (59.34%) | 170 (58.02%) | 11 (91.67%) | 0.020 | ||
| T2D, N (%) | 125 (40.85%) | 115 (39.12%) | 10 (83.33%) | 0.002 | 7.39 (0.01) | 1.62 - 33.76 |
| Altered liver enzymes, N (%) | 20 (6.54%) | 19 (6.46%) | 1 (8.33%) | 0.80 | ||
| Smokers | 50 (16.72%) | 48 (16.72%) | 2 (16.67%) | 0.14 | ||
| Azathioprine, N (%) | 3 (0.98%) | 3 (1.02%) | 0 | 0.73 | ||
| Corticosteroids, N (%) | 17 (5.56%) | 16 (5.44%) | 1 (8.33%) | 0.67 | ||
| mTORi, N (%) | 40 (13.07%) | 39 (13.27%) | 1 (8.33%) | 0.62 | ||
| MMF, N (%) | 184 (60.13%) | 172 (58.5%) | 12 (100%) | 0.004 | ||
| CNIs, N (%) | 189 (61.76%) | 187 (63.61%) | 2 (16.67%) | 0.001 | 0.12 (0.006) | 0.026 - 0.55 |
| Period | <0.001 | |||||
| 1th wave | 24 (7.82%) | 23 (7.80%) | 1 (8.33%) | |||
| 2nd wave | 40 (13.03%) | 36 (12.20%) | 4 (33.33%) | |||
| 3rd wave | 21 (6.84%) | 20 (6.78%) | 1 (8.33%) | |||
| 4th wave | 14 (4.56%) | 11 (3.73%) | 3 (25.00%) | |||
| 5th wave | 10 (3.26%) | 8 (2.71%) | 2 (16.67%) | |||
| 6th wave | 63 (20.52%) | 62 (21.02%) | 1 (8.33%) | |||
| 7th wave | 135 (43.97%) | 135 (45.76%) | 0 (0.00%) | |||
7.1. Respiratory failure
7.2. Invasive mechanical ventilation
7.3. Hospital admission
7.4. Admission to the ICU
7.5. Multivariate analysis of respiratory failure and hospital admission
8. COVID-19 mortality
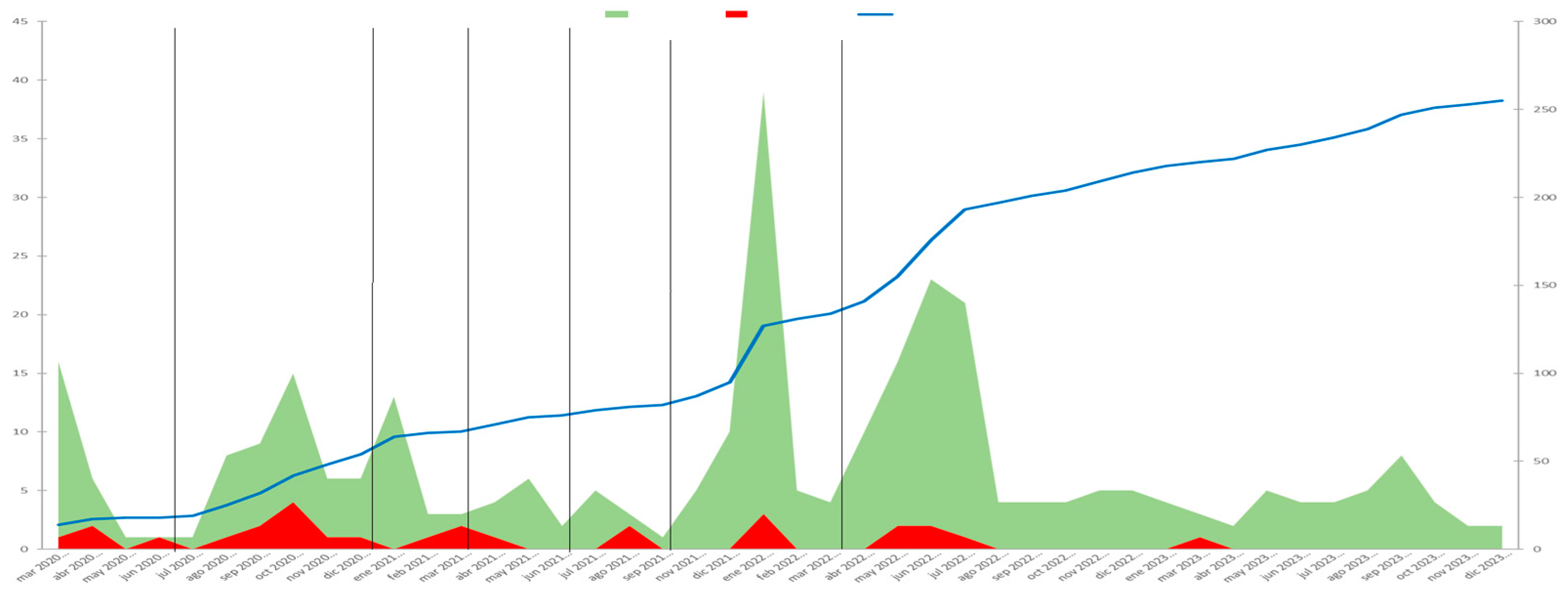
9. Evolution of severe disease throughout the COVID-19 waves
7. Discussion
Author Contributions
Conflicts of Interest
References
- World Health Organization. WHO COVID-19 Dashboard [Internet]. Geneva: WHO. Available at: https://data.who.int/dashboards/covid19/cases.
- Domínguez-Gil B, Coll E, Fernández-Ruiz M, Corral E, Del Río F, Zaragoza R, et al. COVID-19 in Spain: Transplantation in the midst of the pandemic. Am J Transplant. 2020 Sep;20(9):2593-2598. doi: 10.1111/ajt.15983. Epub 2020 May 27. PMID: 32359194; PMCID: PMC7267131. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7267131/.
- De Carlis R, Vella I, Incarbone N, Centonze L, Buscemi V, Lauterio A, et al. Impact of the COVID-19 pandemic on liver donation and transplantation: A review of the literature. World J Gastroenterol. 2021 Mar 14;27(10):928-938. doi: 10.3748/wjg.v27.i10.928. PMID: 33776364. Available at: https://www.wjgnet.com/1007-9327/full/v27/i10/928.htm. [CrossRef]
- Theocharidou E, Adebayo D. Challenges in liver transplantation in the context of a major pandemic. World J Transplant. 2022 Nov;12(11):347-358. doi: 10.5500/wjt.v12.i11.347. PMID: 36437846. Available at: https://www.wjgnet.com/2220-3230/full/v12/i11/347.htm. [CrossRef]
- Aubert O, Yoo D, Zielinski D, Cozzi E, Cardillo M, Dürr M, et al. COVID-19 pandemic and worldwide organ transplantation: a population-based study. Lancet Public Health.2021;6:e709–e719.
- Michaels MG, La Hoz RM, Danziger-Isakov L, Blumberg EA, Kumar D, Green M, et al. Coronavirus disease 2019: Implications of emerging infections for transplantation. Am J Transplant. 2020 Jul;20(7):1768-1772. doi: 10.1111/ajt.15832. Epub 2020 Mar 16. PMID: 32090448; PMCID: PMC9800450. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9800450/. [CrossRef]
- Guarino M, Cossiga V, Loperto I, Esposito I, Ortolani R, Fiorentino A, et al. COVID-19 in liver transplant recipients: incidence, hospitalization and outcome in an Italian prospective double-centre study. Sci Rep. 2022 Mar 22;12(1):4831. doi: 10.1038/s41598-022-08947-x. PMID: 35318432; PMCID: PMC8940902. Available at:https://pubmed.ncbi.nlm.nih.gov/35318432/. [CrossRef]
- Colmenero J, Rodríguez-Perálvarez M, Salcedo M, Arias-Milla A, Muñoz-Serrano A, Graus J, et al. Epidemiological pattern, incidence, and outcomes of COVID-19 in liver transplant patients. J Hepatol. 2021 Jan;74(1):148-155. doi: 10.1016/j.jhep.2020.07.040. Epub 2020 Aug 1. PMID: 32750442; PMCID: PMC7395653. Available at:https://pubmed.ncbi.nlm.nih.gov/32750442/. [CrossRef]
- Kulkarni AV, Tevethia HV, Premkumar M, Arab JP, Candia R, Kumar K, et al. Impact of COVID-19 on liver transplant recipients-A systematic review and meta-analysis.EClinicalMedicine. 2021 Aug;38:101025. doi: 10.1016/j.eclinm.2021.101025. Epub 2021 Jul 13. PMID: 34278287; PMCID: PMC8276632. [CrossRef]
- Inayat F, Patel P, Ali H, Afzal A, Tahir H, Chaudhry A, et al. Impact of COVID-19 on liver transplant recipients: A nationwide cohort study evaluating hospitalization, transplant rejection, and inpatient mortality. World J Transplant. 2024 Jun 18;14(2):90866. doi: 10.5500/wjt.v14.i2.90866. PMID: 38947960; PMCID: PMC11212588. [CrossRef]
- John BV, Deng Y, Khakoo NS, Taddei TH, Kaplan DE, Dahman B. Coronavirus disease 2019 vaccination is associated with reduced severe acute respiratory syndrome coronavirus 2 infection and death in liver transplant recipients. Gastroenterology. 2022;162:645-647.e2.
- Hamm SR, Rezahosseini O, Møller DL, Loft JA, Poulsen JR, Knudsen JD, et al. Incidence and severity of SARS-CoV-2 infections in liver and kidney transplant recipients in the post-vaccination era: real-life data from Denmark. Am J Transplant. 2022;22:2637-2650.
- Moon AM, Webb GJ, García-Juárez I, Kulkarni AV, Adali G, Wong DK, et al. SARS-CoV-2 infections among patients with liver disease and liver transplantation who received COVID-19 vaccination. Hepatol Commun. 2022 Apr;6(4):889-897. doi: 10.1002/hep4.1853. Epub 2021 Nov 9. PMID: 34708575; PMCID: PMC8652790. [CrossRef]
- Organización Nacional de Trasplantes (ONT). Centros autorizados de obtención y trasplante de órganos, tejidos y células [Internet]. Madrid: ONT. Available at:https://www.ont.es/red-espanola-de-donacion-y-trasplante/centros-autorizados-de-obtencion-trasplante-de-organos-tejidos-y-celulas-2-2-2/.
- Organización Nacional de Trasplantes (ONT). Balance 2023 [Internet]. Madrid: ONT; 2024 Jan 17. Available at: https://www.ont.es/wp-content/uploads/2024/01/BALANCE_2023_ONT_17012024.pdf.
- Loinaz C, Marcacuzco A, Fernández-Ruiz M, Caso O, Cambra F, San Juan R, et al. Varied clinical presentation and outcome of SARS-CoV-2 infection in liver transplant recipients: Initial experience at a single center in Madrid, Spain. Transpl Infect Dis. 2020 Oct;22(5):e13372. doi: 10.1111/tid.13372. Epub 2020 Jul 1. PMID: 32562561; PMCID: PMC7323090. [CrossRef]
- Coll E, Fernández-Ruiz M, Sánchez-Álvarez JE, Martínez-Fernández JR, Crespo M, Gayoso J, et al. COVID-19 in transplant recipients: The Spanish experience. Am J Transplant. 2021 May;21(5):1825-1837. doi: 10.1111/ajt.16369. Epub 2020 Nov 10. PMID: 33098200; PMCID: PMC9906239. [CrossRef]
- Coll E, Fernández-Ruiz M, Padilla M, Moreso F, Hernández-Vicente A, Yañez I, et al. COVID-19 in Solid Organ Transplant Recipients in Spain Throughout 2020: Catching the Wave? Transplantation. 2021 Oct 1;105(10):2146-2155. doi: 10.1097/TP.0000000000003873. PMID: 34241988; PMCID: PMC8487705. [CrossRef]
- Belli LS, Duvoux C, Cortesi PA, Facchetti R, Iacob S, Perricone G, et al. COVID-19 in liver transplant candidates: pretransplant and post-transplant outcomes - an ELITA/ELTR multicentre cohort study. Gut. 2021 Oct;70(10):1914-1924. doi: 10.1136/gutjnl-2021-324879. Epub 2021 Jul 19. PMID: 34281984. [CrossRef]
- Mansoor E, Perez A, Abou-Saleh M, et al. Clinical characteristics, hospitalization, and mortality rates of coronavirus disease 2019 among liver transplant patients in the United States: a multicenter research network study. Gastroenterology. 2021;160(1):459-462.e1.
- Yüce M, Filiztekin E, Özkaya KG. COVID-19 diagnosis - A review of current methods. Biosens Bioelectron. 2021 Jan 15;172:112752. doi: 10.1016/j.bios.2020.112752. Epub 2020 Oct 24. PMID: 33126180; PMCID: PMC7584564. [CrossRef]
- Vieceli T, Oliveira Filho CM, Berger M, Saadi MP, Salvador PA, Anizelli LB, et al. A predictive score for COVID-19 diagnosis using clinical, laboratory and chest image data. Braz J Infect Dis. 2020 Jul-Aug;24(4):343-348. doi: 10.1016/j.bjid.2020.06.009. Epub 2020 Jul 25. PMID: 32721387; PMCID: PMC7381934. [CrossRef]
- Vengesai A, Midzi H, Kasambala M, Mutandadzi H, Mduluza-Jokonya TL, Rusakaniko S, et al. A systematic and meta-analysis review on the diagnostic accuracy of antibodies in the serological diagnosis of COVID-19. Syst Rev. 2021 May 26;10(1):155. doi: 10.1186/s13643-021-01689-3. PMID: 34039423; PMCID: PMC8152206. [CrossRef]
- Sharma A, Balda S, Apreja M, Kataria K, Capalash N, Sharma P. COVID-19 Diagnosis: Current and Future Techniques. Int J Biol Macromol. 2021 Dec 15;193(Pt B):1835-1844. doi: 10.1016/j.ijbiomac.2021.11.016. Epub 2021 Nov 12. PMID: 34774862; PMCID: PMC8585557. [CrossRef]
- Diagnosis and treatment protocol for novel coronavirus pneumonia (Trial Version 7). Chin Med J (Engl). 2020;133(9):1087-1095.
- Informe n° 182. Situación de COVID-19 en España. Informe COVID-19. [Internet]. 2021 . Available at: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/Paginas/InformesCOVID-19.aspx.
- Kute VB, Tullius SG, Rane H, Chauhan S, Mishra V, Meshram HS. Global Impact of the COVID-19 Pandemic on Solid Organ Transplant. Transplant Proc. 2022 Jul-Aug;54(6):1412-1416. doi: 10.1016/j.transproceed.2022.02.009. Epub 2022 Feb 10. PMID: 35337665; PMCID: PMC8828418. [CrossRef]
- Inayat F, Patel P, Ali H, Afzal A, Tahir H, Chaudhry A, Ishtiaq R, Rehman AU, Darji K, Afzal MS, Nawaz G, Giammarino A, Satapathy SK. Impact of COVID-19 on liver transplant recipients: A nationwide cohort study evaluating hospitalization, transplant rejection, and inpatient mortality. World J Transplant. 2024 Jun 18;14(2):90866. doi: 10.5500/wjt.v14.i2.90866. PMID: 38947960; PMCID: PMC11212588. [CrossRef] [PubMed]
- Montiel Villalonga P, Martínez-Alpuente I, Fernández-Ruiz M, Len Ó, Bodro M, Los-Arcos I, et al. Transplantation of organs from SARS-CoV-2-positive donors: Preliminary experience from Spain. Transpl Infect Dis. 2023 Feb;25(1):e14008. doi: 10.1111/tid.14008. Epub 2023 Jan 19. PMID: 36659870. [CrossRef]
- Romagnoli R, Gruttadauria S, Tisone G, Ettorre GM, De Carlis L, Martini S, et al. Liver transplantation from active COVID-19 donors: A lifesaving opportunity worth grasping? Am J Transplant. 2021 Dec;21(12):3919-3925. doi: 10.1111/ajt.16823. Epub 2021 Sep 13. PMID: 34467627; PMCID: PMC8653300. [CrossRef]
- Webb GJ, Marjot T, Cook JA, Aloman C, Armstrong MJ, Brenner EJ, et al. Outcomes following SARS-CoV-2 infection in liver transplant recipients: an international registry study.Lancet Gastroenterol Hepatol. 2020 Nov;5(11):1008-1016. doi: 10.1016/S2468-1253(20)30271-5. Epub 2020 Aug 28. PMID: 32866433; PMCID: PMC7455160. [CrossRef]
- Becchetti C, Zambelli MF, Pasulo L, et al; COVID-LT group. COVID-19 in an international European liver transplant recipient cohort. Gut. 2020;69:1832–1840.
- Rabiee A, Sadowski B, Adeniji N, Perumalswami PV, Nguyen V, Moghe A, et al; COLD Consortium. Liver Injury in Liver Transplant Recipients With Coronavirus Disease 2019 (COVID-19): U.S. Multicenter Experience. Hepatology. 2020 Dec;72(6):1900-1911. doi: 10.1002/hep.31574. Epub 2020 Dec 9. PMID: 32964510; PMCID: PMC7537191. [CrossRef]
- Eren-Kutsoylu OO, Egeli T, Agalar C, Ozbilgin M, Ozgen-Alpaydin A, Unek T, et al. COVID-19 in Liver Transplant Patients: A University Hospital Experience. Transplant Proc. 2023 Jun;55(5):1223-1225. doi: 10.1016/j.transproceed.2023.01.032. Epub 2023 Mar 21. PMID: 37087296; PMCID: PMC10027957. [CrossRef]
- Shafiq M, Gibson C. Clinical outcomes of coronavirus disease 2019 in liver transplant recipients. World J Hepatol. 2022 Jun 27;14(6):1142-1149. doi: 10.4254/wjh.v14.i6.1142. PMID: 35978674; PMCID: PMC9258250. [CrossRef]
- Fu S, Chen D, Zhang Y, Wu M. Clinical characteristics of liver transplant recipients with COVID-19 and analysis of risk factors for the severe disease. J Infect Dev Ctries. 2024 Apr 30;18(4):542-549. doi: 10.3855/jidc.18764. PMID: 38728648. [CrossRef]
- Lee BT, Perumalswami PV, Im GY, et al; COBE Study Group. COVID-19 in liver transplant recipients: An initial experience from the US epicenter. Gastroenterology. 2020;159:1176–1178.e2.
- Forns X, Navasa M. Inmunosupresión en el trasplante hepático en la era COVID-19. Rev Esp Enferm Dig. 2020;112(10):758-764.
- Parente A, Manzia TM, Angelico R, Tirotta F, Muiesan P, Tisone G, et al. COVID-19, liver transplant, and immunosuppression: Allies or foes? Liver Transpl. 2020;26(12):1562-1564. doi: 10.1002/lt.26028. [CrossRef]
- Karruli A, Spiezia S, Boccia F, Gagliardi M, Patauner F, Salemme A, et al. Effect of immunosuppression maintenance in solid organ transplant recipients with COVID-19: Systematic review and meta-analysis. Transpl Infect Dis. 2021 Aug;23(4):e13595. doi: 10.1111/tid.13595. Epub 2021 Mar 18. PMID: 33641202; PMCID: PMC7995235. [CrossRef]
- Fix OK, Hameed B, Fontana RJ, Kwok RM, McGuire BM, Mulligan DC, et al. Clinical Best Practice Advice for Hepatology and Liver Transplant Providers During the COVID-19 Pandemic: AASLD Expert Panel Consensus Statement. Hepatology. 2020;72:287-304. doi: 10.1002/hep.31281. PMID: 32298473. [CrossRef]
- Marjot T, Eberhardt CS, Boettler T, Belli LS, Berenguer M, Buti M, et al. Impact of COVID-19 on the liver and on the care of patients with chronic liver disease, hepatobiliary cancer, and liver transplantation: An updated EASL position paper. J Hepatol. 2022;77:1161-1197. doi: 10.1016/j.jhep.2022.07.008. PMID: 35868584. [CrossRef]
- Caballero-Marcos A, Citores MJ, Alonso-Fernández R, Rodríguez-Perálvarez M, Valerio M, Graus Morales J, et al. Decreased Long-Term Severe Acute Respiratory Syndrome Coronavirus 2-Specific Humoral Immunity in Liver Transplantation Recipients 12 Months After Coronavirus Disease 2019. Liver Transpl. 2022 Jun;28(6):1039-1050. doi: 10.1002/lt.26389. Epub 2022 Jan 17. PMID: 34919762. [CrossRef]
- Liu FC, Xie M, Rao W. Clinical application of COVID-19 vaccine in liver transplant recipients. Hepatobiliary Pancreat Dis Int. 2024 Aug;23(4):339-343. doi: 10.1016/j.hbpd.2023.08.010. Epub 2023 Aug 12. PMID: 37620225. [CrossRef]
- Rabinowich L, Grupper A, Baruch R, Ben-Yehoyada M, Halperin T, Turner D, et al. Low immunogenicity to SARS-CoV-2 vaccination among liver transplant recipients. J Hepatol. 2021 Aug;75(2):435-438. doi: 10.1016/j.jhep.2021.04.020. Epub 2021 Apr 21. PMID: 33892006; PMCID: PMC8058047. [CrossRef]
- World Health Organization. Coronavirus disease (COVID-19) vaccines. [Internet]. [cited 2024 Jan 7]. Available at: https://www.who.int/es/emergencies/diseases/novel-coronavirus-2019/question-and-answers-hub/q-a-detail/coronavirus-disease-(covid-19)-vaccines.
- World Health Organization. Coronavirus disease (COVID-19): vaccines. Available at: https://extranet.who.int/prequal/vaccines/covid-19-vaccines-who-emergency-use-listing.
- John BV, Deng Y, Khakoo NS, Taddei TH, Kaplan DE, Dahman B. Coronavirus disease 2019 vaccination is associated with reduced severe acute respiratory syndrome coronavirus 2 infection and death in liver transplant recipients. Gastroenterology. 2022;162:645–647 e2.
- Hamm SR, Rezahosseini O, Møller DL, Loft JA, Poulsen JR, Knudsen JD, et al. Incidence and severity of SARS-CoV-2 infections in liver and kidney transplant recipients in the post-vaccination era: real-life data from Denmark. Am J Transplant. 2022;22:2637–2650.
- Cornberg M, Buti M, Eberhardt CS, Grossi PA, Shouval D. EASL position paper on the use of COVID-19 vaccines in patients with chronic liver diseases, hepatobiliary cancer and liver transplant recipients. J Hepatol. 2021;74:944-951.
- Fix OK, Blumberg EA, Chang KM, Chu J, Chung RT, Goacher EK, Hameed B, Kaul DR, Kulik LM, Kwok RM, McGuire BM, Mulligan DC, Price JC, Reau NS, Reddy KR, Reynolds A, Rosen HR, Russo MW, Schilsky ML, Verna EC, Ward JW, Fontana RJ; AASLD COVID-19 Vaccine Working Group. American Association for the Study of Liver Diseases Expert Panel Consensus Statement: Vaccines to Prevent Coronavirus Disease 2019 Infection in Patients With Liver Disease. Hepatology. 2021 Aug;74(2):1049-1064. doi: 10.1002/hep.31751. PMID: 33577086; PMCID: PMC8014184. Available at: https://www.aasld.org/sites/default/files/2022-10/AASLD%20COVID-19%20Guidance%20Document%2010.06.2022F.pdf. [CrossRef]
- Gkoufa A, Saridaki M, Georgakopoulou VE, Spandidos DA, Cholongitas E. COVID-19 vaccination in liver transplant recipients (Review). Exp Ther Med. 2023 May 3;25(6):291. doi: 10.3892/etm.2023.11990. PMID: 37206557; PMCID: PMC10189597. [CrossRef]
- Nimmo A, Gardiner D, Ushiro-Lumb I, Ravanan R, Forsythe JLR. The Global Impact of COVID-19 on Solid Organ Transplantation: Two Years Into a Pandemic. Transplantation. 2022 Jul 1;106(7):1312-1329. doi: 10.1097/TP.0000000000004151. Epub 2022 Apr 11. PMID: 35404911; PMCID: PMC9213067. [CrossRef]
- Chen X, Luo D, Mei B, et al. Immunogenicity of COVID-19 vaccines in solid organ transplant recipients: a systematic review and metaanalysis. Clin Microbiol Infect. 2023;29:441–456.
- Herrera S, Colmenero J, Pascal M, Escobedo M, Castel MA, Sole-González E, Palou E, Egri N, Ruiz P, Mosquera M, Moreno A, Juan M, Vilella A, Soriano A, Farrero M, Bodro M. Cellular and humoral immune response after mRNA-1273 SARS-CoV-2 vaccine in liver and heart transplant recipients. Am J Transplant. 2021 Dec;21(12):3971-3979. doi: 10.1111/ajt.16768. Epub 2021 Aug 4. PMID: 34291552; PMCID: PMC9800111. [CrossRef]
- Kabaçam G, Dayangaç M, Üçbilek E, Erçin CN, Günsar F, Akyıldız M, Akarsu M, Demir M, Kaymakoğlu S, Karasu Z, İdilman R. The COVID-19 pandemic: Clinical practice advice for gastroenterologists, hepatologists, and liver transplant specialists. Turk J Gastroenterol. 2020; 31(11): 841-852.
- Buehrle DJ, Sutton RR, McCann EL, Lucas AE. A review of treatment and prevention of coronavirus disease 2019 among solid organ transplant recipients. Viruses. 2021;13(9):1706.
- Navidi Z, Pakzad Moghadam SH, Iravani MM, Orandi A, Orandi A, Ghazi SF, Fallah E, Malekabad ES, Khorramnia S. Remdesivir in solid organ transplant recipients with COVID-19: a systematic review and meta-analysis. Clin Transplant Res. 2024 Sep 30;38(3):212-221. doi: 10.4285/ctr.24.0031. PMID: 39344698; PMCID: PMC11464156. [CrossRef]
- World Health Organization. WHO recommends highly successful COVID-19 therapy and calls for wide geographical distribution and transparency from originator. WHO; 2022 Apr 22. Available from: https://www.who.int/news/item/22-04-2022-who-recommends-highly-successful-covid-19-therapy-and-calls-for-wide-geographical-distribution-and-transparency-from-originator.
- Korula P, Alexander H, John JS, Kirubakaran R, Singh B, Tharyan P, Rupali P. Favipiravir for treating COVID-19. Cochrane Database Syst Rev. 2024 Feb 5;2(2):CD015219. doi: 10.1002/14651858.CD015219.pub2. PMID: 38314855; PMCID: PMC10840071. [CrossRef]
- Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for Coronavirus disease 2019 (COVID-19). A review. JAMA. Apr 13; doi: 10.1001/jama.2020.6019. [CrossRef]
- Asselah T, Durantel D, Pasmant E, Lau G, Schinazi RF. COVID-19: Discovery, diagnostics and drug development. J Hepatol. 2020;73(1):1-12.
- Fishbane S, Hirsch JS, Nair V. Special Considerations for Paxlovid Treatment Among Transplant Recipients With SARS-CoV-2 Infection. Am J Kidney Dis. 2022 Apr;79(4):480-482. doi: 10.1053/j.ajkd.2022.01.001. Epub 2022 Jan 12. PMID: 35032591; PMCID: PMC8754454. [CrossRef]
| Table 1. A. Symptoms at diagnosis | p-value | ||||||||
| Period | 1st wave | 2nd wave | 3rd wave | 4th wave | 5th wave | 6th wave | 7th wave | ||
| Number of cases | N = 302(5 lost cases) | 24 | 40 | 21 | 14 | 10 | 63 | 135 | |
| Odynophagia | 30 (9.93%) | 1 (4.17%) | 0 | 1 (4.76%) | 1 (7.14%) | 1 (10%) | 5 (8.06%) | 21 (16.03%) | 0.068 |
| Rhinorrhoea | 46 (15.23%) | 0 | 1 (2.5%) | 2 (9.52%) | 0 | 1 (10%) | 14 (22.58%) | 28 (21.37%) | 0.004 |
| Nausea or vomiting | 13 (4.30%) | 1 (4.17%) | 4 (10%) | 2 (9.52%) | 0 | 0 | 2 (3.23%) | 4 (3.05%) | 0.39 |
| Asthenia | 98 (32.45%) | 5 (20.83%) | 8 (20%) | 5 (23.81%) | 4 (28.57%) | 4 (40%) | 14 (22.58%) | 58 (44.27%) | 0.011 |
| Myalgias | 73 (24.17%) | 2 (8.33%) | 12 (30%) | 4 (19.05%) | 2 (14.29%) | 2 (20%) | 11 (17.74%) | 40 (30.53%) | 0.15 |
| Dyspnea | 45 (14.90%) | 7 (29.17%) | 10 (25%) | 2 (9.52%) | 3 (21.43%) | 1 (10%) | 11 (17.74%) | 11 (8.40%) | 0.043 |
| Ageusia | 11 (3.64%) | 0 | 2 (5%) | 0 | 0 | 3 (30%) | 3 (4.84%) | 3 (2.29%) | <0.001 |
| Anosmia | 15 (4.97%) | 0 | 4 (10%) | 1 (4.76%) | 0 | 4 (40%) | 2 (3.23%) | 4 (3.05%) | <0.001 |
| Chest pain | 12 (3.97%) | 2 (8.33%) | 1 (2.5%) | 1 (4.76%) | 1 (7.14%) | 0 | 4 (6.45%) | 3 (2.29%) | 0.65 |
| Diarrhoea | 32 (10.60%) | 7 (29.17%) | 7 (17.5%) | 2 (9.52%) | 1 (7.14%) | 1 (10%) | 4 (6.45%) | 10 (7.63%) | 0.039 |
| Febricle | 58 (19.21%) | 2 (8.33%) | 0 | 2 (9.52%) | 1 (7.14%) | 3 (30%) | 13 (20.97%) | 37 (28.24%) | 0.001 |
| Fever | 77 (25.50%) | 6 (25.00%) | 16 (40%) | 7 (33.33%) | 1 (7.14%) | 2 (20%) | 15 (24.19%) | 30 (22.9%) | 0.22 |
| Anorexia | 14 (4.64%) | 0 | 3 (7.5%) | 2 (9.52%) | 0 | 0 | 3 (4.84%) | 6 (4.58%) | 0.65 |
| Headache | 33 (10.93%) | 1 (4.17%) | 1 (2.5%) | 2 (9.52%) | 1 (7.14%) | 0 | 10 (16.13%) | 18 (13.74%) | 0.22 |
| Conjunctivitis | 1 (0.33%) | 0 | 1 (2.5%) | 0 | 0 | 0 | 0 | 0 | 0.36 |
| Expectoration | 26 (8.61%) | 1 (4.17%) | 1 (2.5%) | 1 (4.76%) | 2 (14.29%) | 0 | 6 (9.68%) | 15 (11.45%) | 0.44 |
| Cough | 120 (39.74%) | 14 (58.33%) | 16 (40%) | 4 (19.05%) | 2 (14.29%) | 3 (30%) | 18 (29.03%) | 63 (48.09%) | 0.006 |
| Asymptomatic | 60 (19.87%) | 4 (16.67%) | 7 (17.50%) | 7 (33.33%) | 4 (28.57%) | 3 (30%) | 16 (25.81%) | 19 (14.50%) | 0.26 |
| B. Chest X-ray findings | p-value | ||||||||
| Period | 1st wave | 2nd wave | 3rd wave | 4th wave | 5th wave | 6th wave | 7th wave | < 0.001 | |
| Number of cases | N = 275 (32 lost cases) | 24 | 40 | 21 | 14 | 10 | 63 | 135 | |
| Not performed | 191 (69.45%) | 7 (30.43%) | 24 (61.54%) | 11 (57.89%) | 4 (30.77%) | 4 (57.14%) | 42 (77.78%) | 99 (82.5%) | |
| Normal | 28 (10.18%) | 5 (21.74%) | 0 | 1 (5.26%) | 2 (15.38%) | 0 | 4 (7.41%) | 16 (13.33%) | |
| Bilateral pneumonia | 46 (16.73%) | 9 (39.13%) | 13 (33.33%) | 7 (36.84%) | 6 (46.15%) | 3 (42.86%) | 6 (11.11%) | 2 (1.67%) | |
| Lobar pneumonia | 10 (3.64%) | 2 (8.70%) | 2 (5.13%) | 0 | 1 (7.69%) | 0 | 2 (3.70%) | 3 (2.50%) | |
| Table 2. Treatment received in each period | ||||||||
| Period | 1st wave | 2nd wave | 3rd wave | 4th wave | 5th wave | 6th wave | 7th wave | |
| Number of cases | N = 307 | 24 | 40 | 21 | 14 | 10 | 63 | 135 |
| Antibiotics | 50 (16.29%) | 11 (45.83%) | 13 (32.50%) | 2 (9.52%) | 5 (35.71%) | 2 (20.00%) | 7 (11.11%) | 10 (7.41%) |
| Anticoagulation | 38 (12.38%) | 5 (20.83%) | 12 (30.00%) | 4 (19.05%) | 6 (42.86%) | 5 (50.00%) | 5 (7.94%) | 1 (0.74%) |
| Antimalarials | 13 (4.23%) | 12 (50.00%) | 1 (2.50%) | 0 | 0 | 0 | 0 | 0 |
| Corticosteroids | 45 (14.66%) | 3 (12.50%) | 13 (32.50%) | 4 (19.05%) | 5 (35.71%) | 5 (50.00%) | 10 (15.87%) | 5 (3.70%) |
| Interferon | 2 (0.65%) | 2 (8.33%) | 0 | 0 | 0 | 0 | 0 | 0 |
| Kaletra | 3 (0.98%) | 3 (12.50%) | 0 | 0 | 0 | 0 | 0 | 0 |
| Tocilizumab | 8 (2.61%) | 2 (8.33%) | 0 | 1 (4.76%) | 3 (21.43%) | 1 (10.00%) | 1 (1.59%) | 0 |
| Remdesivir | 27 (8.79%) | 0 | 3 (7.50%) | 0 | 1 (7.14%) | 1 (10.00%) | 4 (6.35%) | 18 (13.33%) |
| Paxlovid | 3 (1.02%) | 0 | 0 | 0 | 0 | 0 | 0 | 3 (2.46%) |
| Table 3. Vaccination | |||||||||
| Period | 1st wave | 2nd wave | 3rd wave | 4th wave | 5th wave | 6th wave | 7th wave | ||
| Number of cases | 24 | 40 | 21 | 14 | 10 | 63 | 135 | ||
| Number of vaccines at diagnosis | |||||||||
| 0 | 93 (30.39%) | 24 (100%) | 40 (100%) | 21 (100%) | 8 (57.14%) | 0 | 0 | 0 | |
| 1 | 16 (5.23%) | 0 | 0 | 0 | 5 (35.71%) | 1 (10%) | 5 (8.06%) | 5 (3.70%) | |
| 2 | 38 (12.42%) | 0 | 0 | 0 | 1 (7.14%) | 9 (90%) | 17 (27.42%) | 11 (8.15%) | |
| 3 | 92 (30.07%) | 0 | 0 | 0 | 0 | 0 | 38 (61.29%) | 54 (40%) | |
| 4 | 54 (17.65%) | 0 | 0 | 0 | 0 | 0 | 2 (3.23%) | 52 (38.52%) | |
| 5 | 13 (4.25%) | 0 | 0 | 0 | 0 | 0 | 0 | 13 (9.63%) | |
| Number of vaccines at diagnosis: median (IQR) | 3 (0 – 3) | 0 | 0 | 0 | 0 (0 – 1) | 2 (2 – 2) | 3 (2 – 3) | 3 (3 – 4) | |
| Time from vaccine to diagnosis (months) | 5.87 (3.23-9.03) | 0.49 (0.3 - 0.52) | 2.89 (1.77-3.28) | 3.61 (3.11-4.98) | 7.34 (4.23-10.62) | ||||
| Table 10. Evolution of severe disease throughout the COVID-19 waves | p | ||||||||
| Period | 1st wave | 2nd wave | 3rd wave | 4th wave | 5th wave | 6th wave | 7th wave | ||
| Number of cases | 24/307 (7.82%) | 40/307 (13.03%) | 21/307 (6.84%) | 14/307(4.56%) | 10/307 (3.25%) | 63/307 (20.52%) | 135/307 (43.97%) | ||
| Respiratory failure | 34/307 (11.07%) | 3 (12.50%) | 12 (30%) | 3 (14.29%) | 6 (42.86%) | 4 (40%) | 4 (6.35%) | 2 (1.48%) | <0.001 |
| IMV | 5/307 (1.63%) | 0 | 2 (5%) | 0 | 1 (7.69%) | 0 | 1 (1.59%) | 1 (0.74%) | 0.29 |
| Admission to the ICU | 12/307 (3.91%) | 1 (4.17%) | 2 (5%) | 0 | 2 (14.29%) | 0 | 5 (7.94%) | 2 (1.48%) | 0.11 |
| Hospitalization | 72/307 (23.45%) | 11 (45.83%) | 16 (40%) | 7 (33.33%) | 9 (64.29%) | 5 (50%) | 11 (17.46%) | 13 (9.63%) | <0.001 |
| Death due to COVID-19 | 12/307 (3.91%) | 1 (4.17%) | 4 (10%) | 1 (4.76%) | 3 (21.43%) | 2 (20%) | 1 (1.59%) | 0 (0.00%) | <0.001 |
| Severity | <0.001 | ||||||||
| Not severe COVID-19 disease | 243/307 (79.15%) | 15 (62.50%) | 24 (60%) | 15 (71.43%) | 6 (42.86%) | 6 (60) | 52 (82.54%) | 125 (92.59%) | |
| Severe COVID-19 disease | 64/307 (20.85%) | 9 (37.50%) | 16 (40%) | 6 (28.57%) | 8 (57.14%) | 4 (40%) | 11 (17.46%) | 10 (7.41%) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




