Submitted:
22 January 2025
Posted:
23 January 2025
You are already at the latest version
Abstract
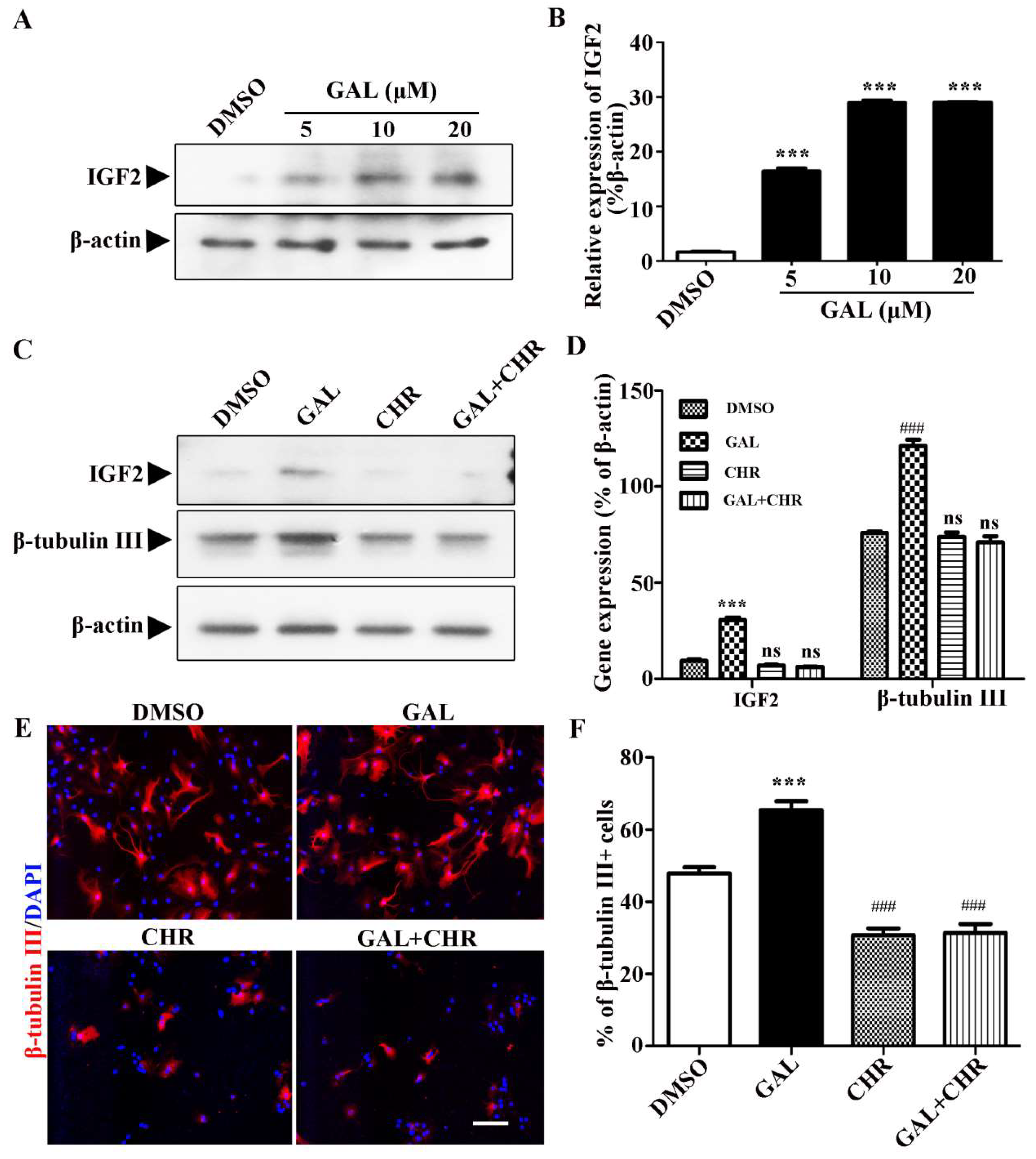
Galanthamine, an alkaloid from the Amaryllidaceae family, serves as an inhibitor of acetylcholinesterase. Beause of its central cholinergic properties, galanthamine is currently being researched as a potential therapeutic agent for the treatment of Alzheimer's disease. Nevertheless, the full extent of its other biological activities is still unclear. Here, we investigated the therapeutic potential of galanthamine in inducing neuronal differentiation and enhancing neurite outgrowth in neural stem and progenitor cells (NSPCs). Our comprehensive analysis revealed significant alterations in neuronal morphology and complexity during neuronal maturation following galanthamine treatment. Specifically, galanthamine significantly increased the percentage of multiple-neurite neurons, suggesting its ability to stimulate neurite formation and promote the development of extensive neuronal networks. Additionally, treatment with galanthamine resulted in a considerable increase in the proportion of mature-appearing neurons, characterized by elongated and intricate dendrites, suggesting its potential in enhancing neural plasticity and repair. Importantly, we further confirmed that galanthamine promotes neuronal differentiation in NSPCs through up-regulation of the IGF2 signaling pathway. Altogether, these findings contribute significantly to our understanding of the effects of galanthamine on Alzheimer's disease and underscore its potential as a therapeutic agent for this neurodegenerative condition.
Keywords:
1. Introduction
2. Results
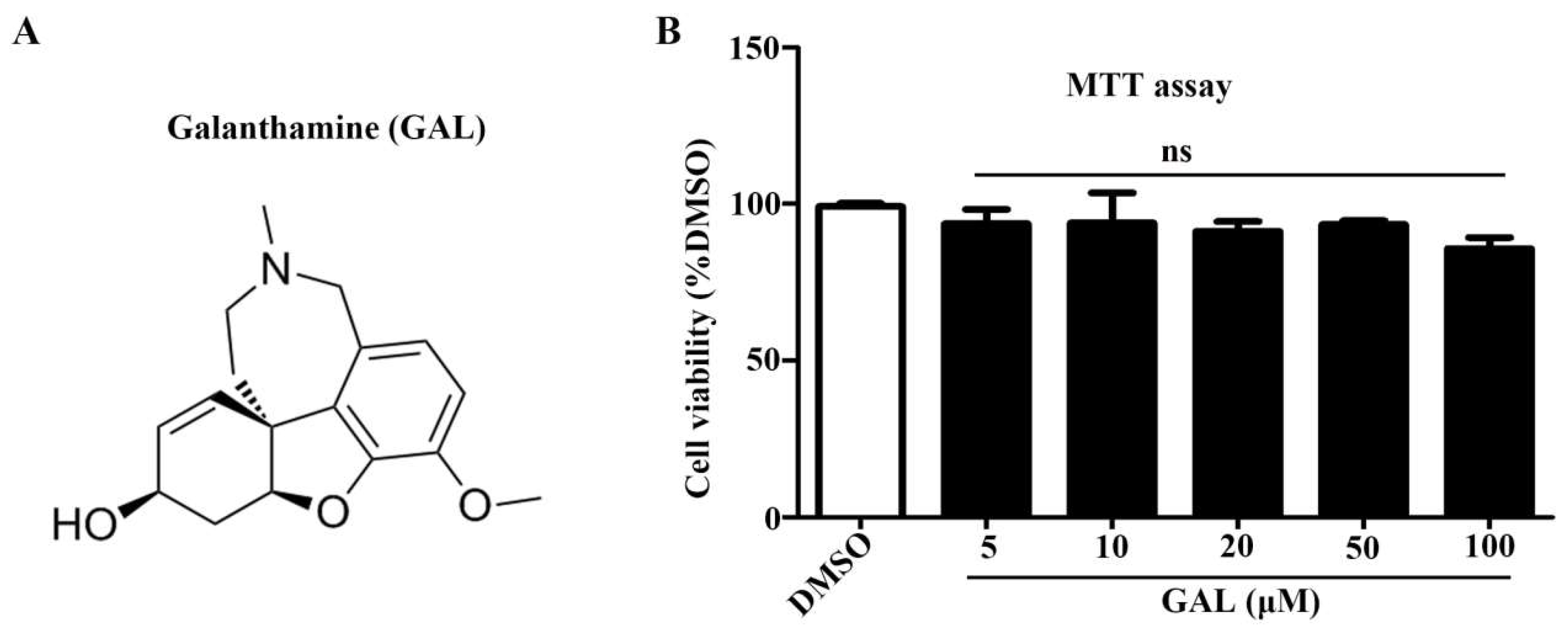
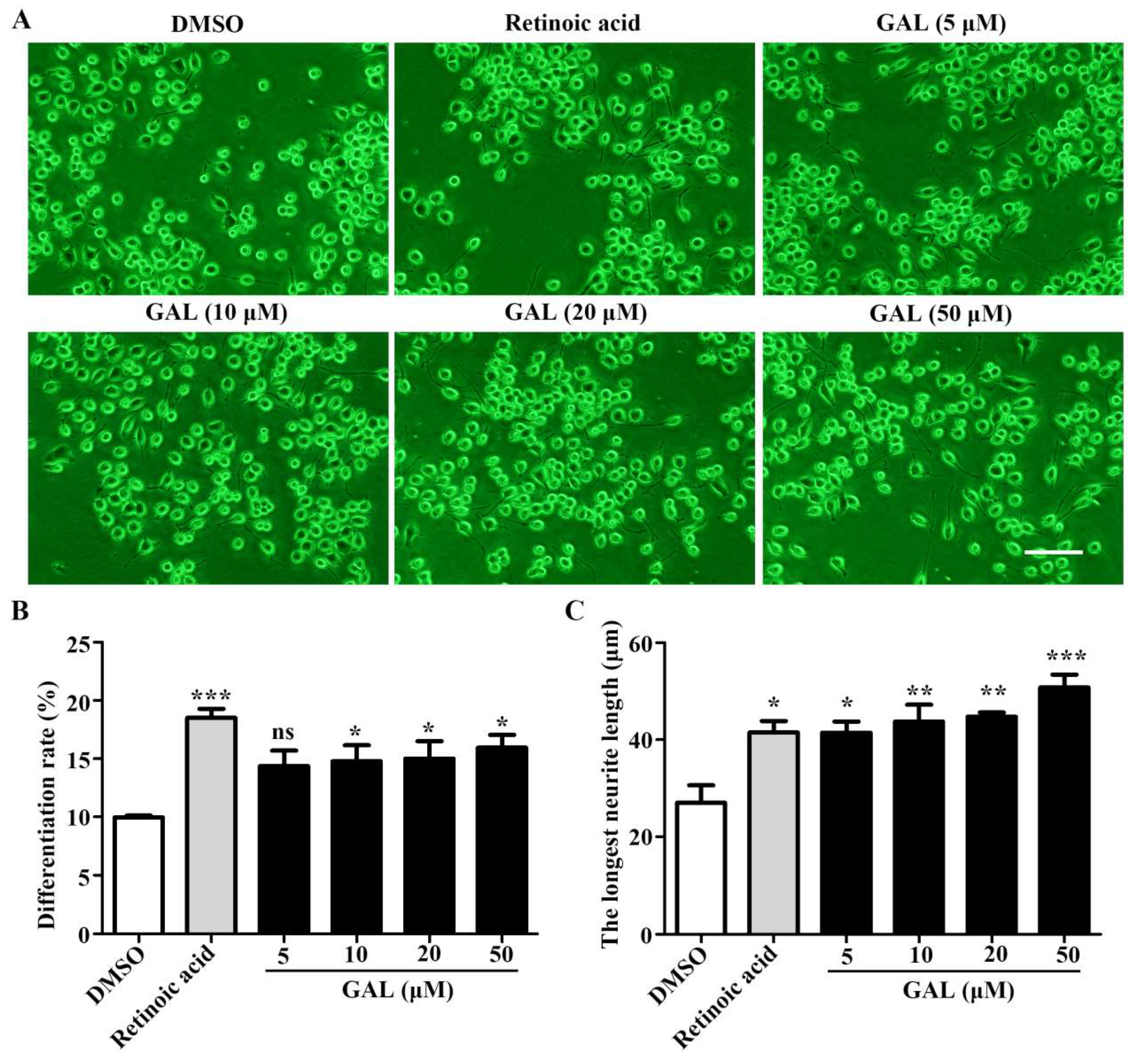
2.1. Galanthamine Induces the Differentiation and Promotes Neurite Outgrowth of Neuro-2a Cells
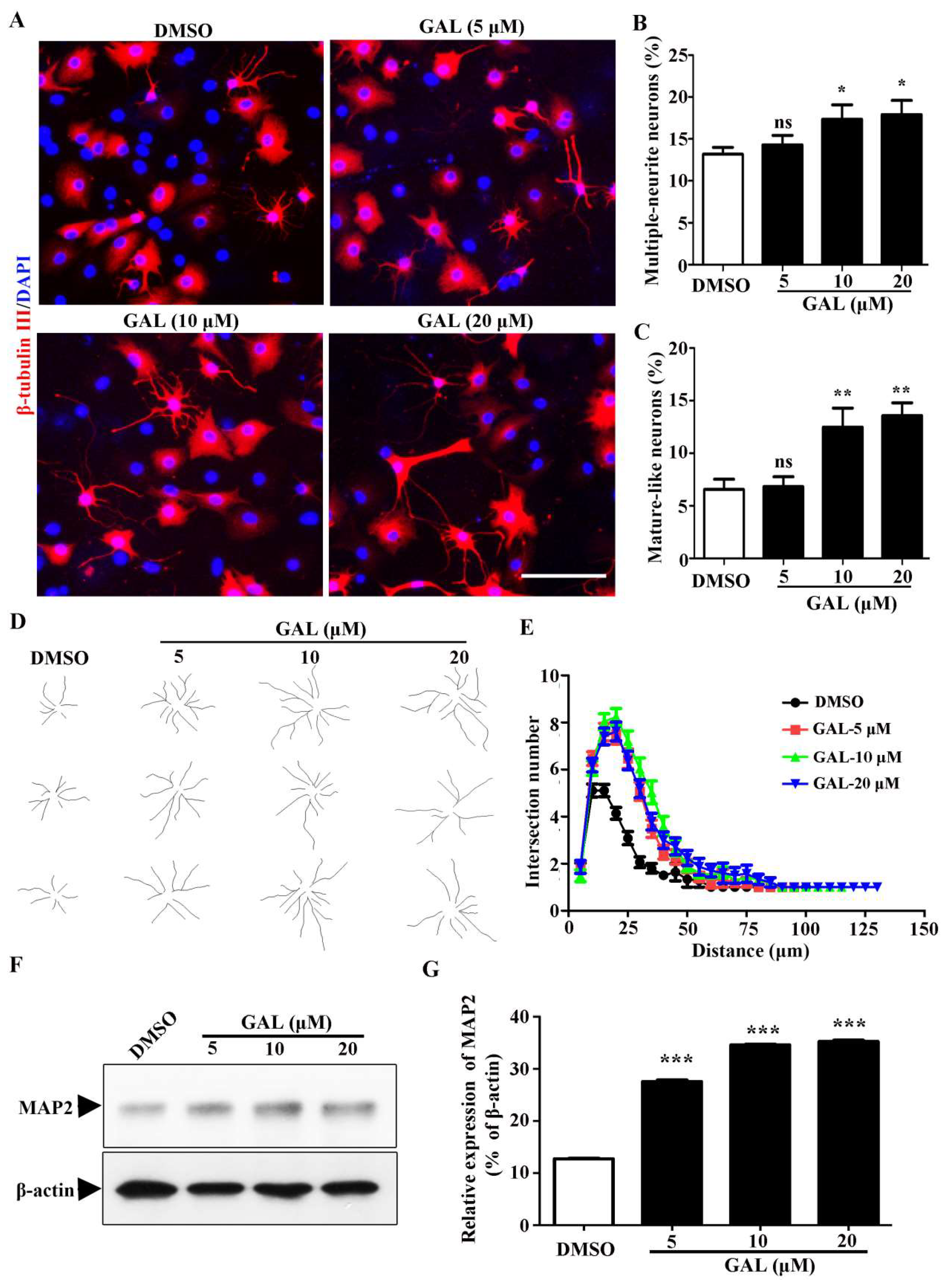
2.2. Galanthamine Promotes the Differentiation of NSPCs
2.3. Galanthamine Stimulates the Maturation Process of Newborn Neurons Derived from NSPCs
2.4. Galanthamine Promotes Neuronal Differentiation of NSPCs by Up-Regulating IGF2
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Culture
4.3. MTT Analysis
4.4. Western Bloting
4.5. Immunostaining
4.6. Statistical Analysis
Funding
References
- Stiles, J.; Jernigan, T. L., The Basics of Brain Development. Neuropsychology Review 2010, 20, (4), 327-348. [CrossRef]
- Markesbery, W. R.; Lovell, M. A., Neuropathologic Alterations in Mild Cognitive Impairment: A Review. Journal of Alzheimer’s Disease 2010, 19, (1), 221-228.
- Temple, S., The development of neural stem cells. Nature 2001, 414, (6859), 112-117. [CrossRef]
- Abraham, R.; Verfaillie, C. M., Neural differentiation and support of neuroregeneration of non-neural adult stem cells. Progress in Brain Research 2012, 201, 17.
- Nie, L.; Yao, D.; Chen, S.; Wang, J.; Pan, C.; Wu, D.; Liu, N.; Tang, Z., Directional induction of neural stem cells, a new therapy for neurodegenerative diseases and ischemic stroke. Cell Death Discovery 2023, 9, (1). [CrossRef]
- Pincus, D. W.; Goodman, R. R.; Fraser, R. A. R.; Maiken, N.; Goldman, S. A., Neural Stem and Progenitor Cells: A Strategy for Gene Therapy and Brain Repair. Neurosurgery 1998, (4), 858-867. [CrossRef]
- More, S. V.; Koppula, S.; Kim, I. S.; Kumar, H.; Kim, B. W.; Choi, D. K., The Role of Bioactive Compounds on the Promotion of Neurite Outgrowth. Molecules 2012, 17, (6). [CrossRef]
- Harvey, A. L., The pharmacology of galanthamine and its analogues. Pharmacology & Therapeutics 1995, 68, (1), 113. [CrossRef]
- Bickel, U.; Thomsen, T.; Weber, W.; Fischer, J. P.; Bachus, R.; Nitz, M.; Kewitz, H., Pharmacokinetics of galanthamine in humans and corresponding cholinesterase inhibition. Clinical Pharmacology & Therapeutics 1991, 50, (4), 420-8. [CrossRef]
- Kong, Y. R.; Tay, K. C.; Su, Y. X.; Wong, C. K.; Khaw, K. Y., Potential of Naturally Derived Alkaloids as Multi-Targeted Therapeutic Agents for Neurodegenerative Diseases. Molecules 2021, 26, (3), 728. [CrossRef]
- Wilkins, H.; Newhouse, P., Acetylcholinesterase and Cognitive Enhancement. 2010.
- Afonso; Caricati-Neto; and; Luiz; Carlos; Abech; D’angelo; and; Haydee; Reuter, Enhancement of purinergic neurotransmission by galantamine and other acetylcholinesterase inhibitors in the rat vas deferens - ScienceDirect. European Journal of Pharmacology 2004, 503, (1-3), 191-201.
- Marco, L.; Carreiras, M. D. C., Galanthamine, a Natural Product for the Treatment of Alzheimer’s Disease. Recent Patents on CNS Drug Discovery 2012, 1, (1), 105-111.
- Rainer, M., Galanthamine in Alzheimer’s Disease. CNS Drugs 1997, 7, 89-97. [CrossRef]
- Gool, W. A. V., Use of galantamine to treat vascular dementia. Lancet 2002, 360, (9344), 1512-1513. [CrossRef]
- Pohanka, M., Acetylcholinesterase inhibitors: a patent review (2008 – present). Expert Opinion on Therapeutic Patents 2012. [CrossRef]
- Mona; Mehta; Abdu; Adem; Marwan; Sabbagh, New acetylcholinesterase inhibitors for Alzheimer’s disease. International journal of Alzheimer’s disease 2012.
- Jin, K.; Xie, L.; Mao, X. O.; Greenberg, D. A., Alzheimer’s disease drugs promote neurogenesis. Brain Research 2006, 1085, (1), 183-188. [CrossRef]
- Kita, Y.; Ago, Y.; Takano, E.; Fukada, A.; Takuma…, K., Galantamine increases hippocampal insulin-like growth factor 2 expression via α7 nicotinic acetylcholine receptors in mice. Psychopharmacology 2013, 225, (3), 543-551. [CrossRef]
- Kita, Y.; Ago, Y.; Higashino, K.; Asada, K.; Takano, E.; Takuma, K.; Matsuda, T., Galantamine promotes adult hippocampal neurogenesis via M1 muscarinic and α7 nicotinic receptors in mice. International Journal of Neuropsychopharmacology 2014, 17, (12), 1957-68. [CrossRef]
- Xiang, X.; Jiang, X.; Lin, H.; Yu, M.; Wu, L.; Zhou, R., Acylhydrazone Derivative A5 Promotes Neurogenesis by Up-Regulating Neurogenesis-Related Genes and Inhibiting Cell-Cycle Progression in Neural Stem/Progenitor Cells. Molecules 2024, 29, (14), 3330. [CrossRef]
- Xiang, X.; Jiang, X.; Lu, X., Regulation of neural stem cell self-renewal, proliferation and differentiation by the RhoA guanine nucleotide exchange factor Arhgef 1. Gene 2023, 863, 147306. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



