Submitted:
22 January 2025
Posted:
23 January 2025
You are already at the latest version
Abstract
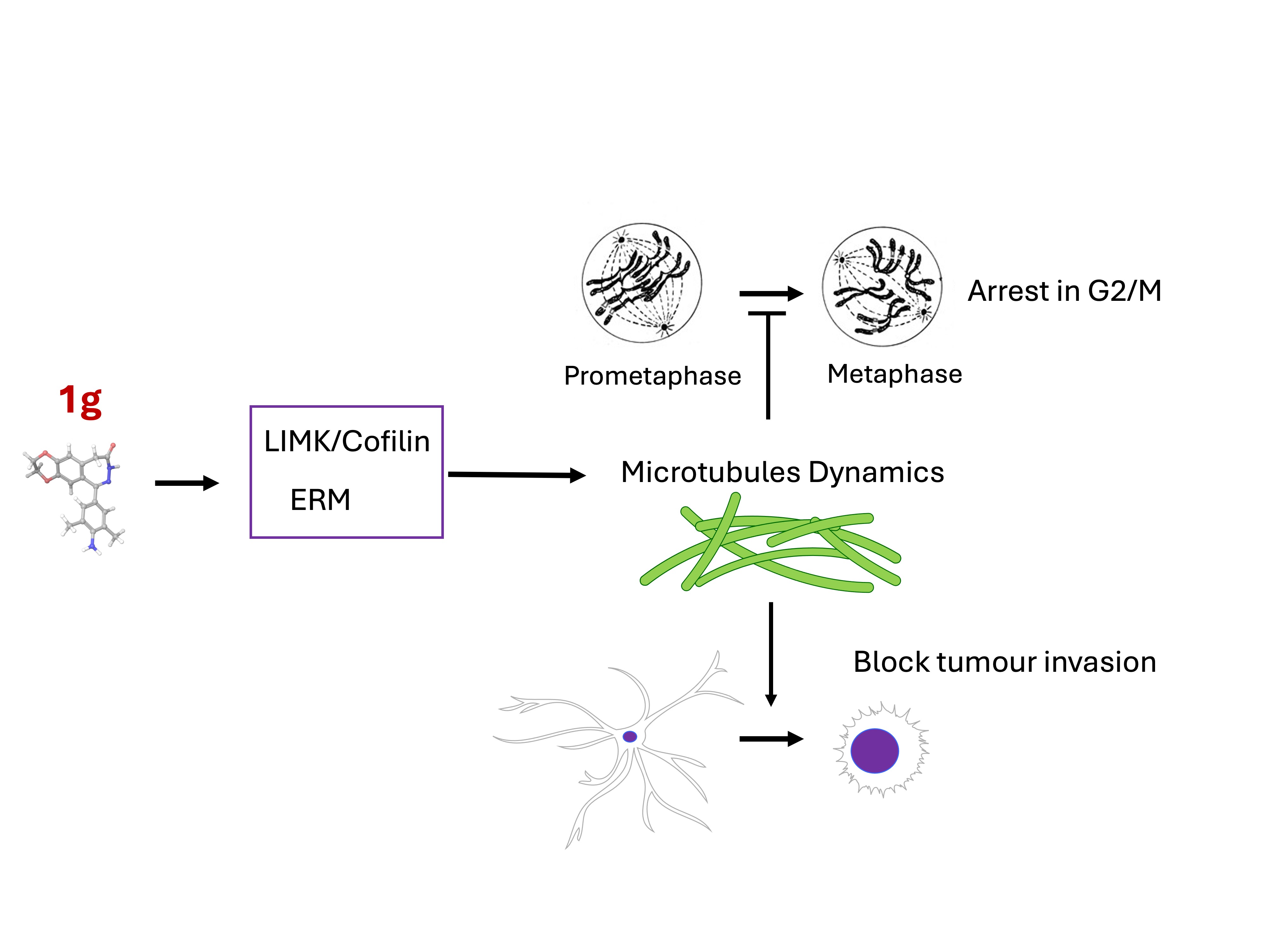
Keywords:
1. Introduction
2. Results
2.1.
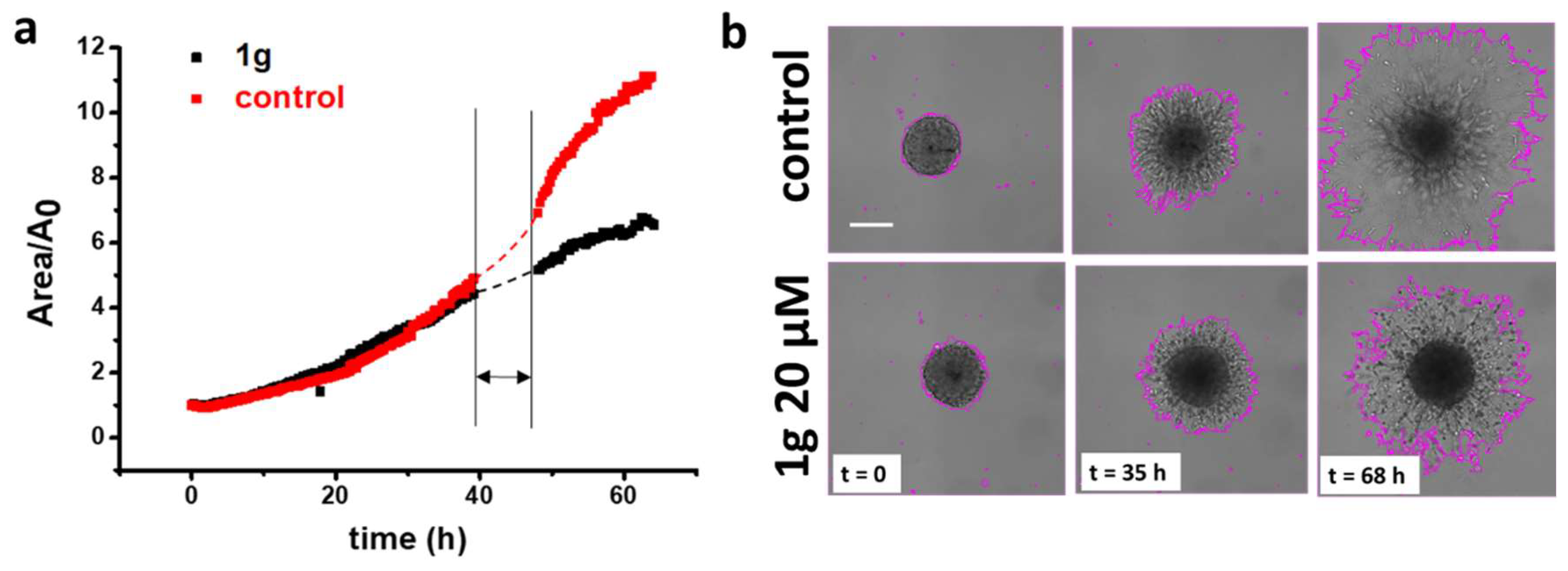
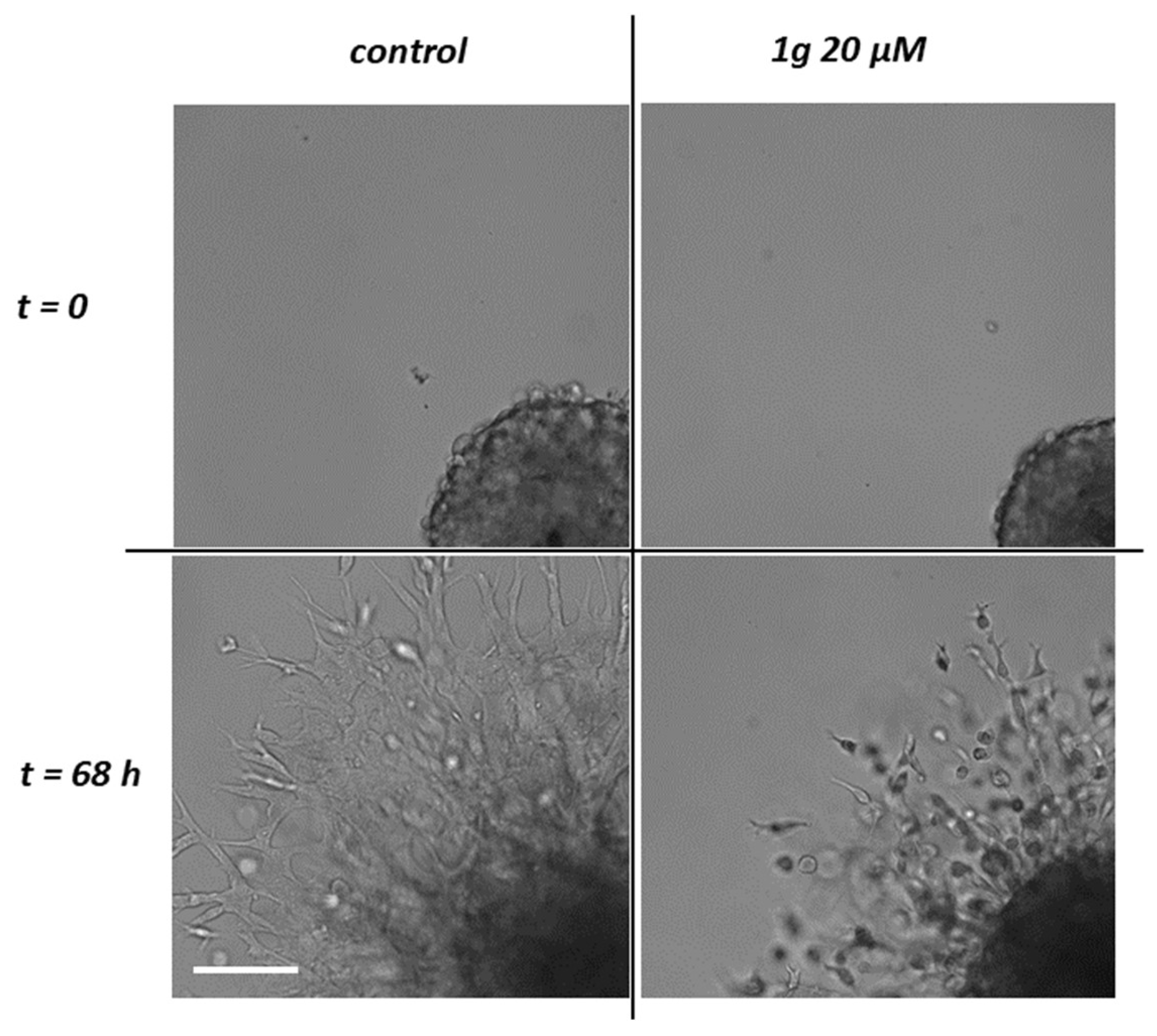
2.1.1. g Reduces U87MG Spheroid Matrix Invasion
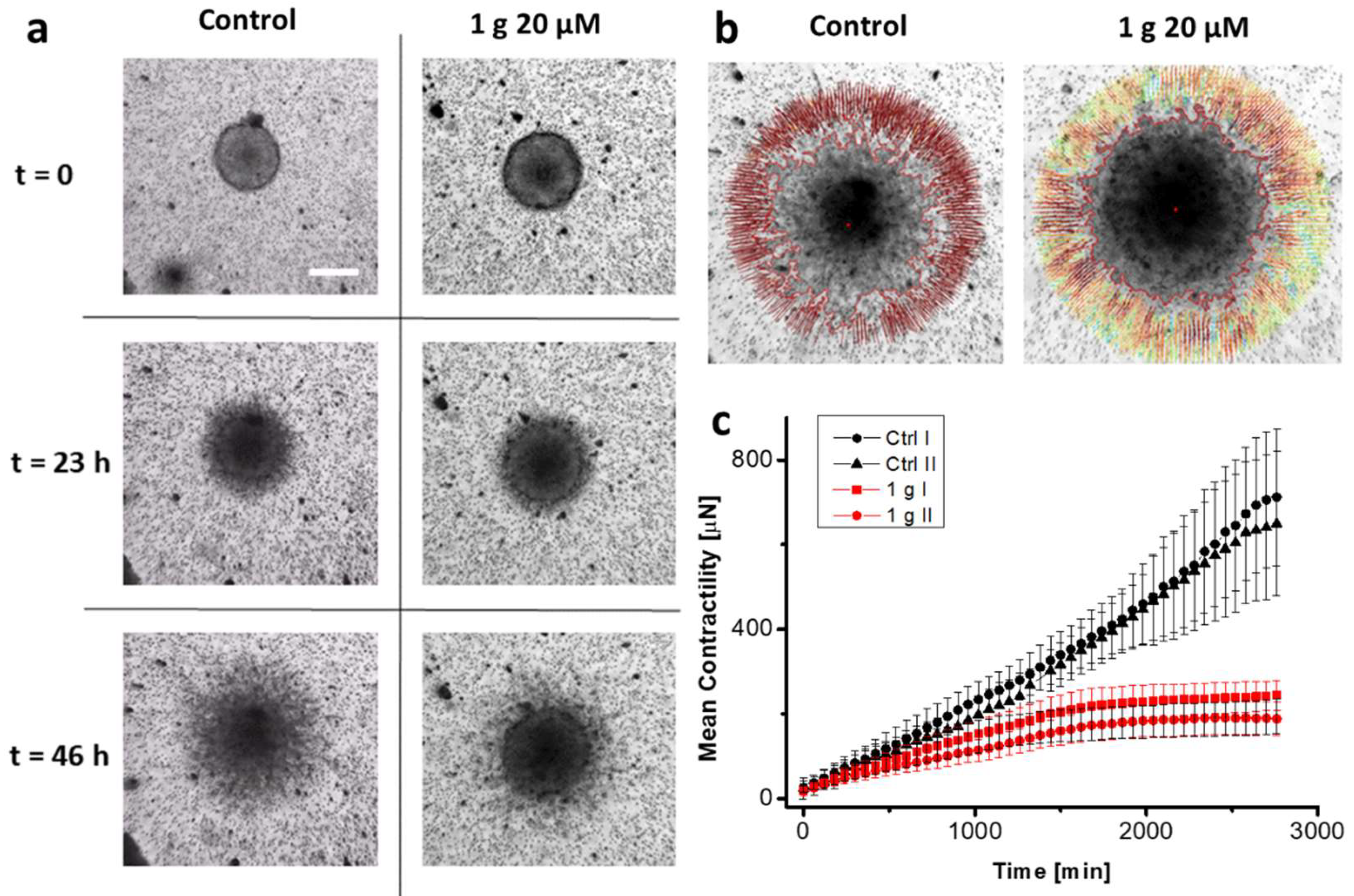
2.1.2. g Reduces Contractility of U87MG Spheroids Embedded in Matrigel
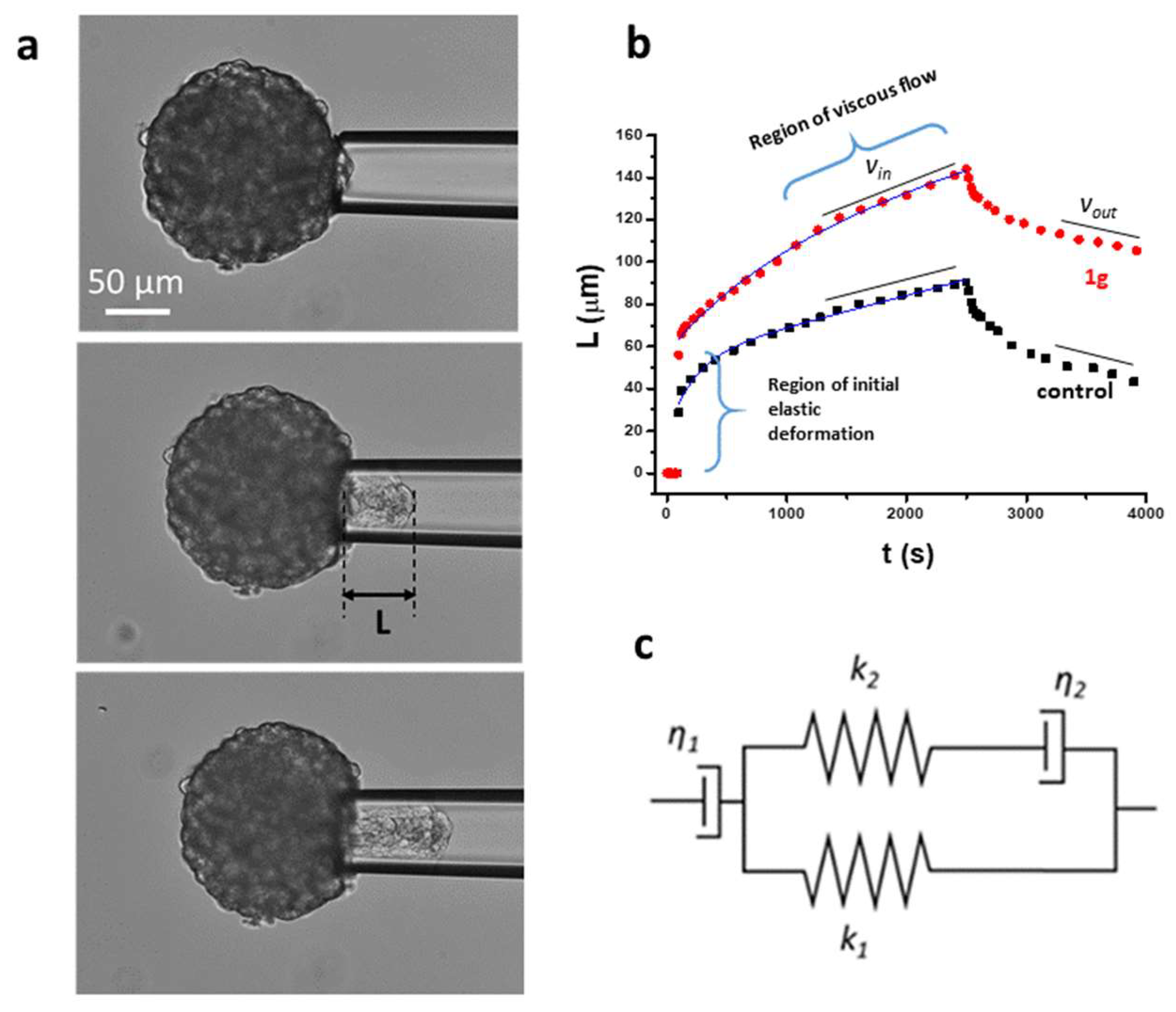
2.1.3. Micropipette Aspiration Technique (MAT) Shows that 1g Decreases Surface Tension of U87MG Spheroids
2.2.
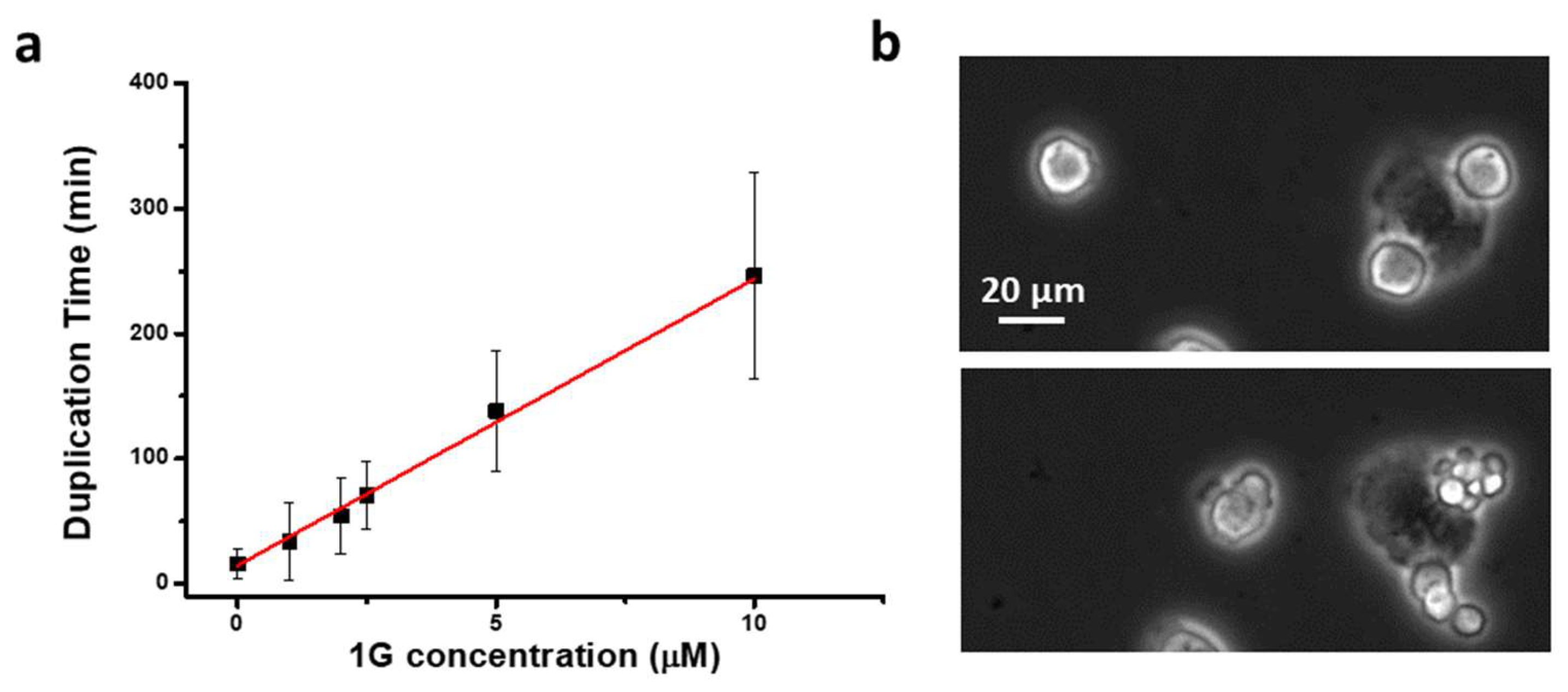
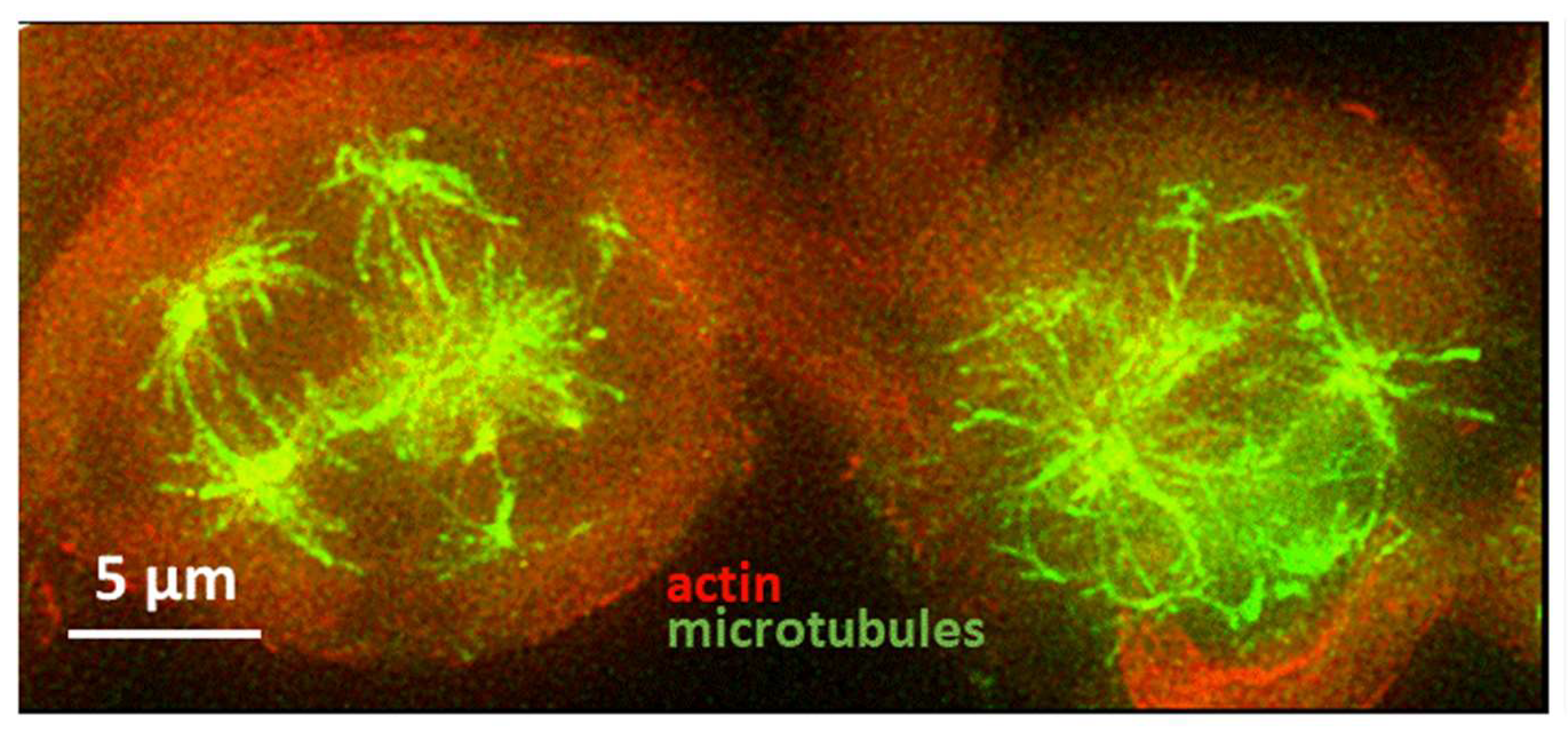
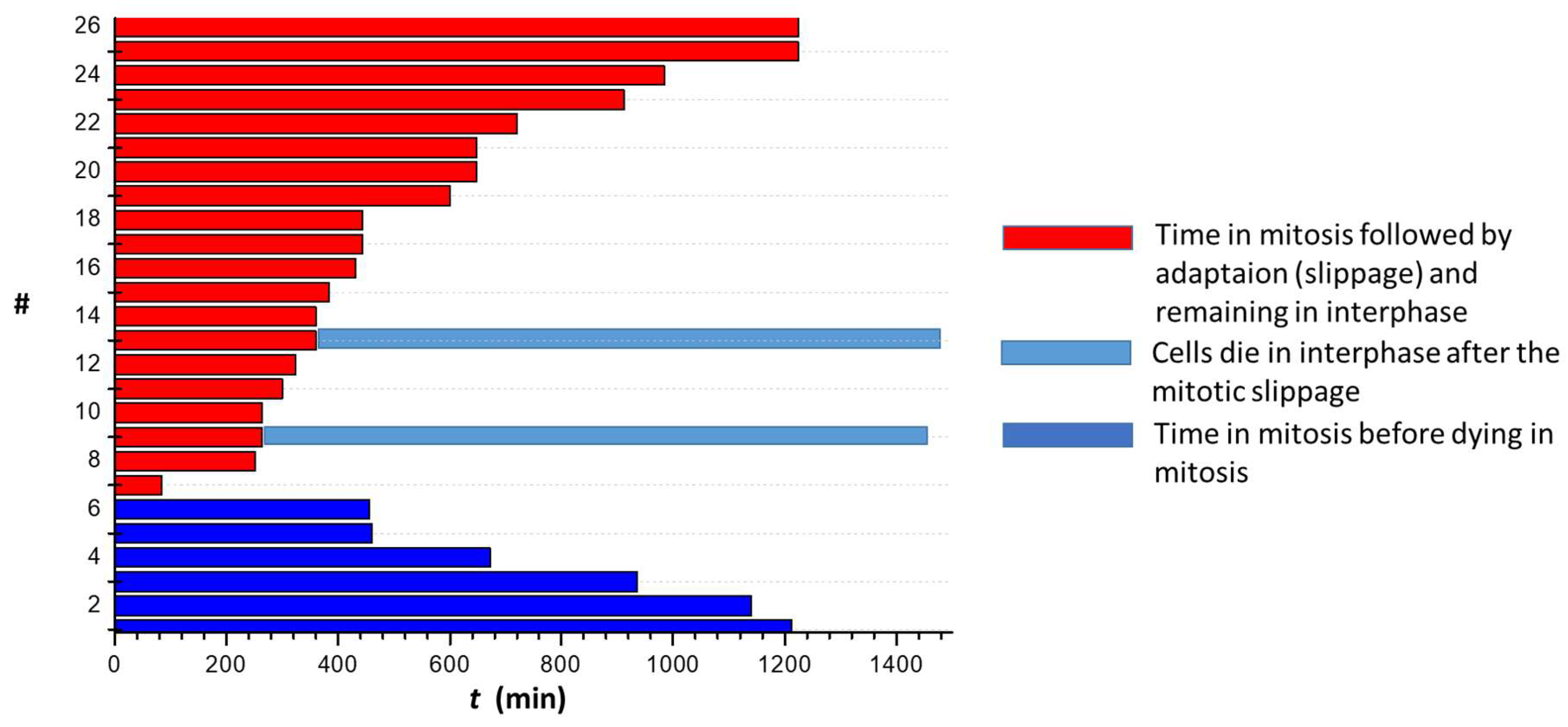
2.2.1. g Reduces Cell Division in 2D Cell Cultures
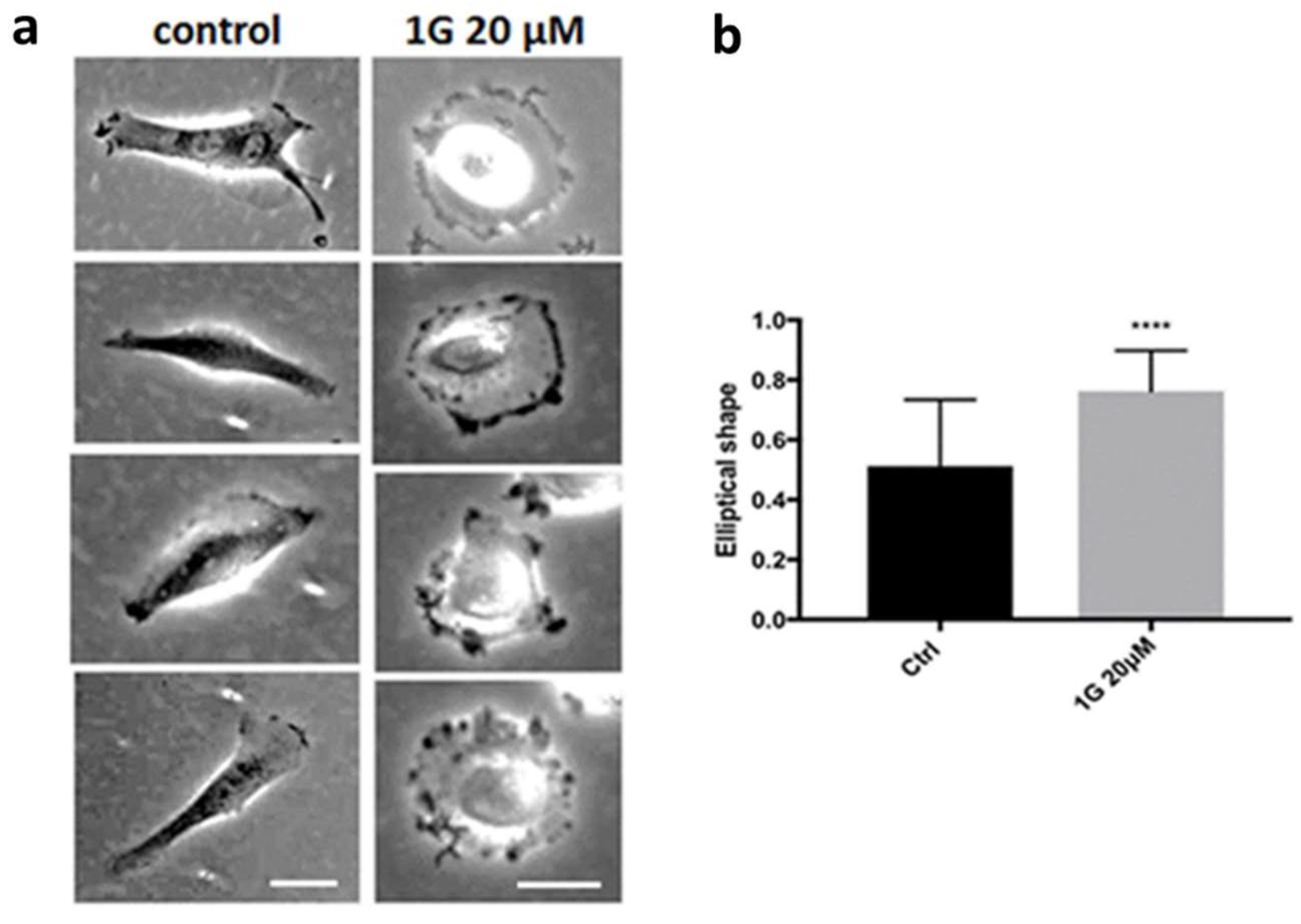
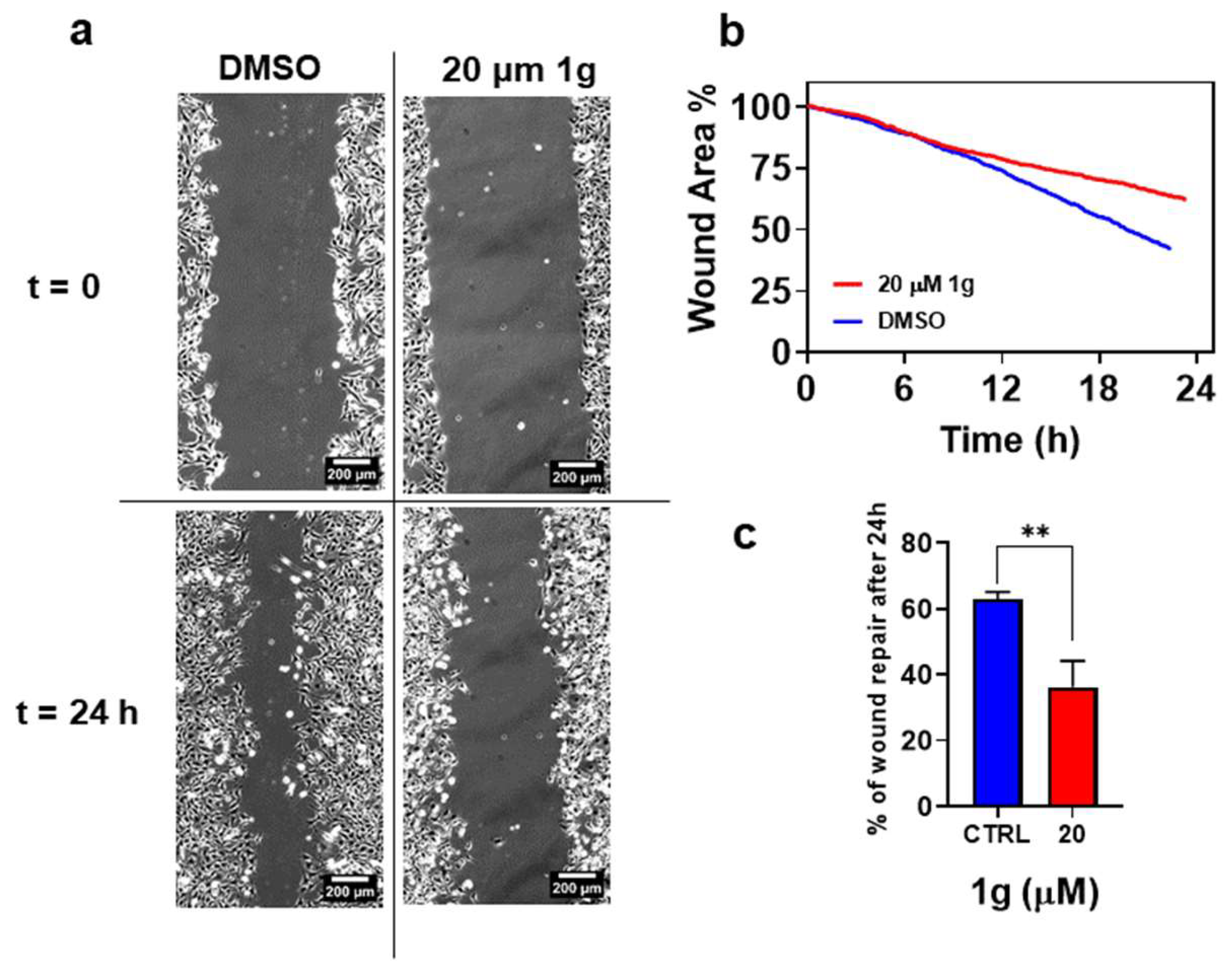
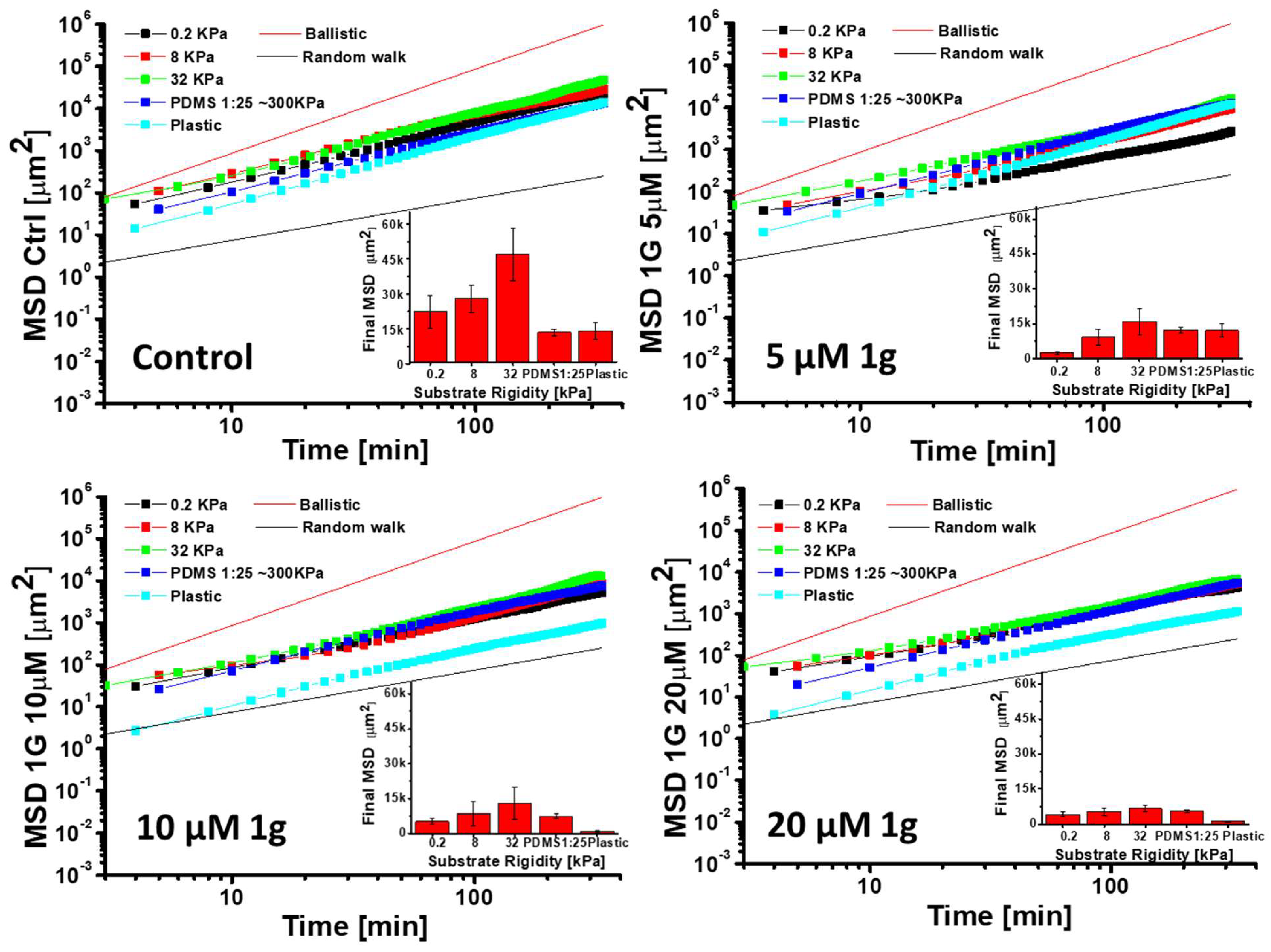
2.2.2. g Inhibits Migration of GBM in 2D Cultures
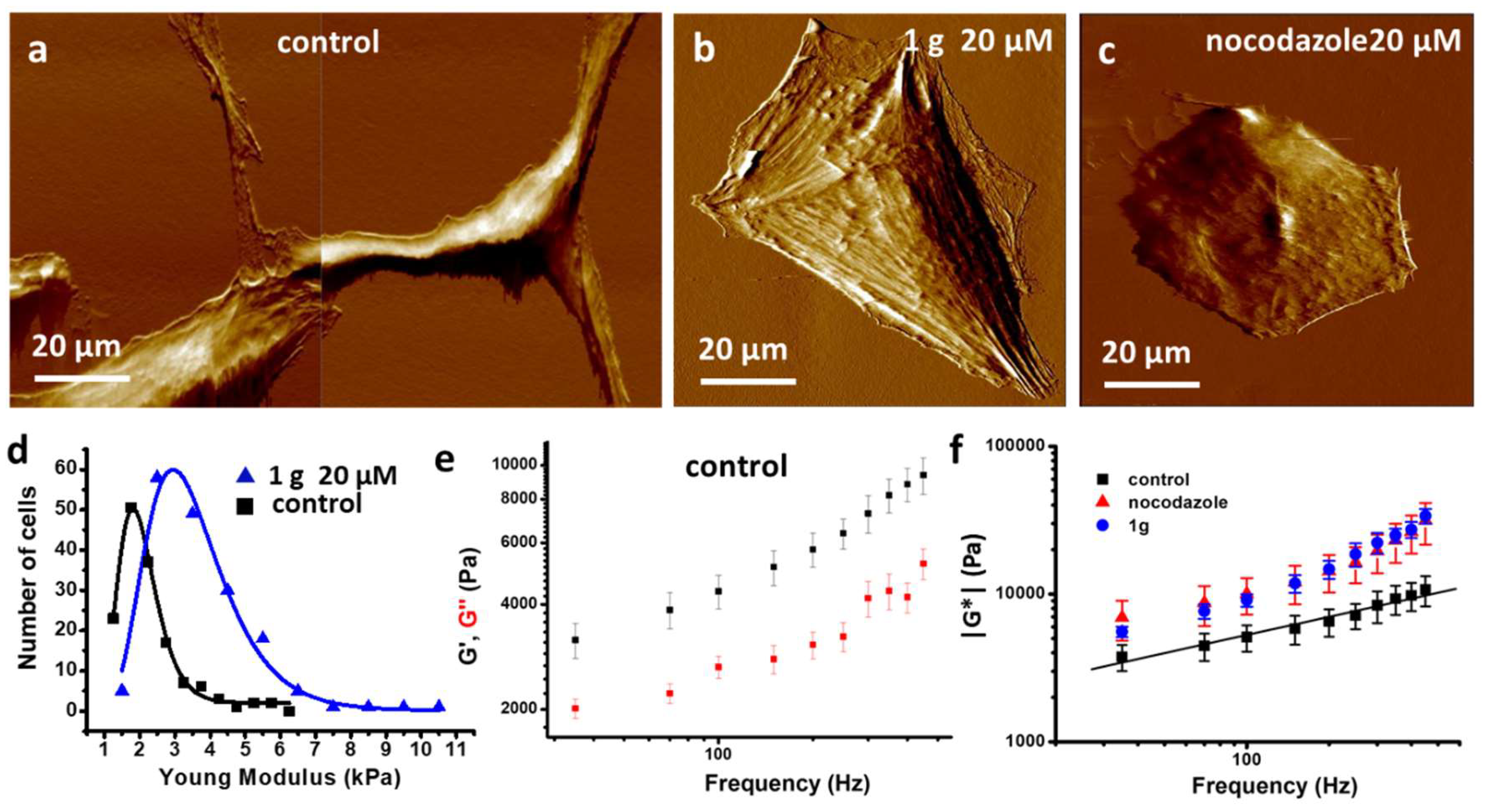
2.2.3. g Increases Cell Stiffness of GBM Cells as Measured by Atomic Force Microscopy
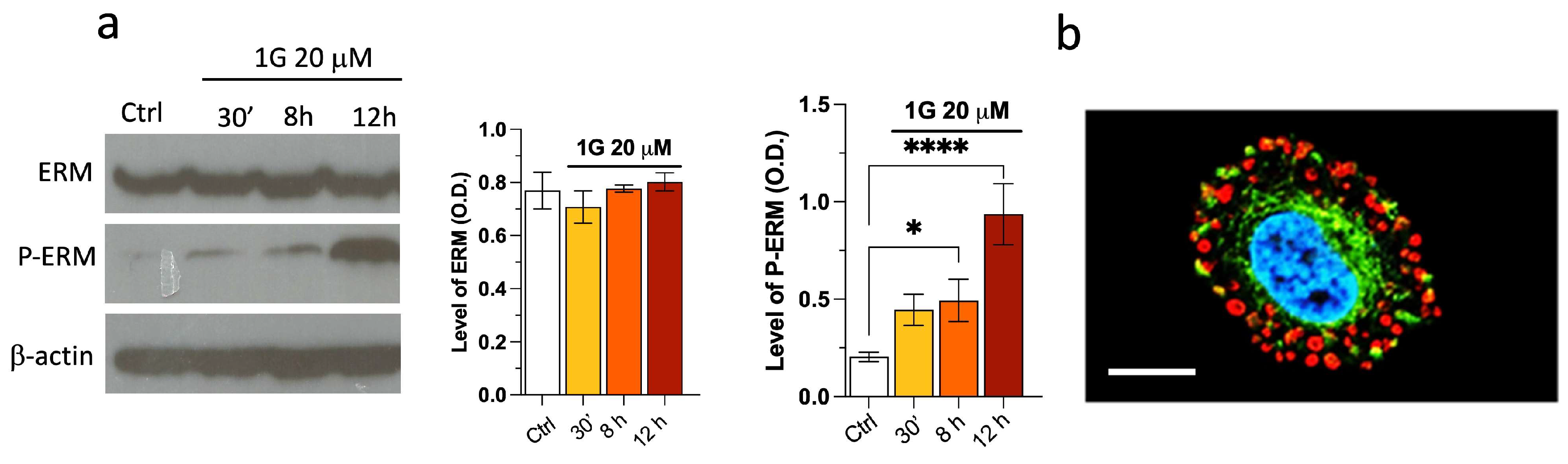
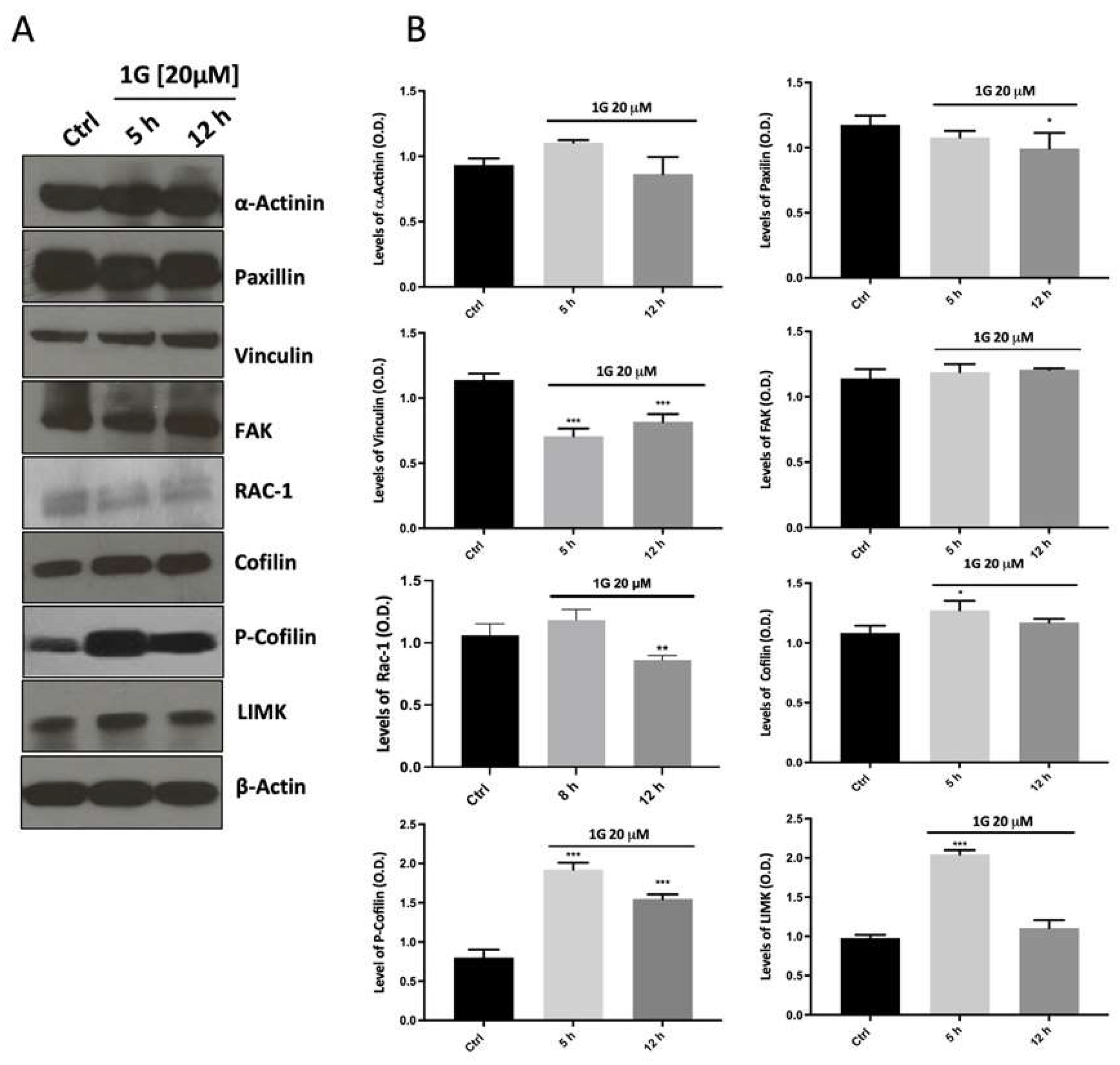
2.3. Modulation of the Principal Pathways Involved in Cell Adhesion and Migration
3. Discussion
4. Materials and Methods
Cell Line and Treatment
2D Cell Migration
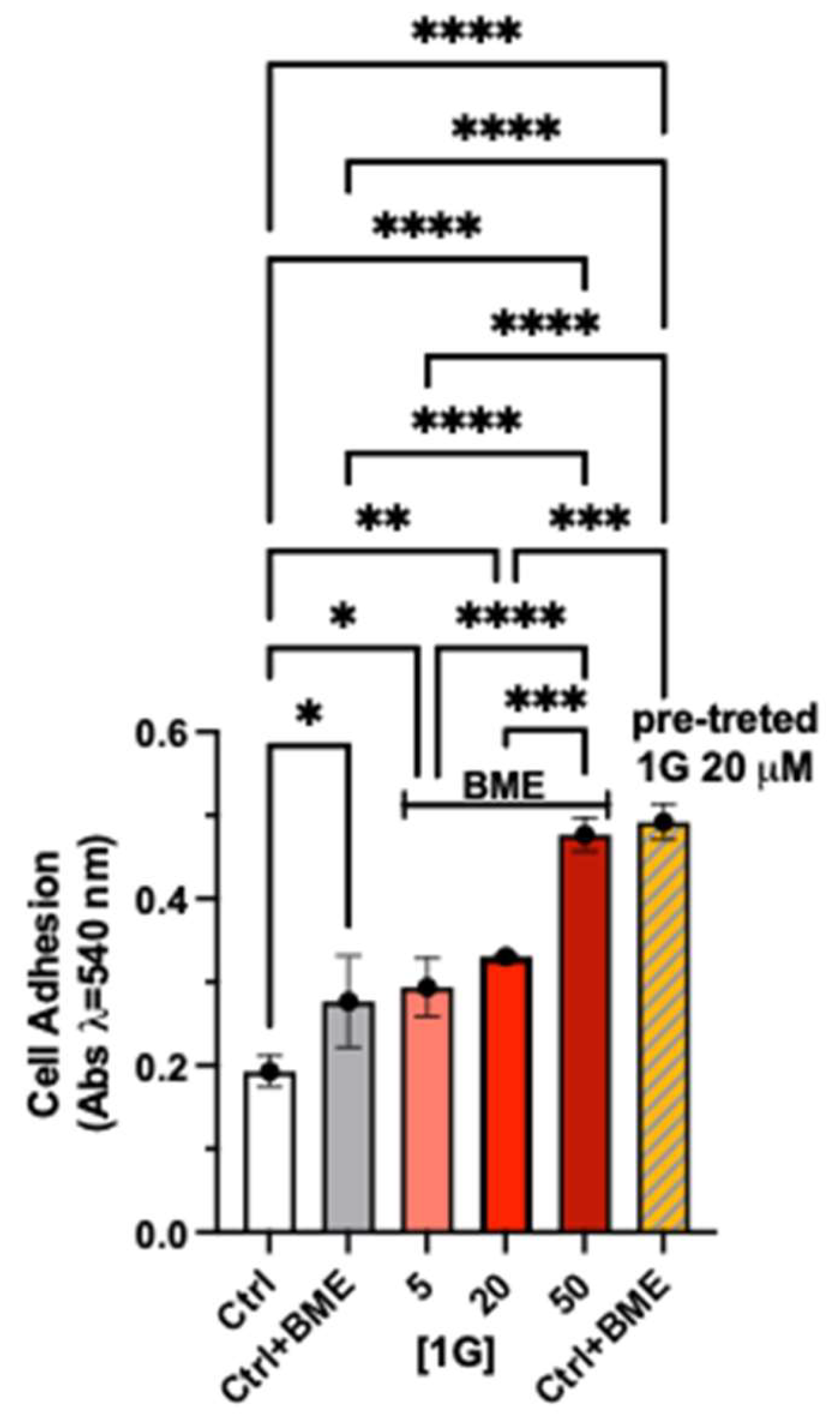
Adhesion Assay
Western Blot
Spheroid Preparation
3D Traction Force Microscopy of Spheroids
Micropipette Aspiration Technique
Dynamic Mechanical Analysis by AFM
Ting Model Analysis of Single Force Curves
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| The following abbreviations are used in this manuscript | . |
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
Appendix A
Appendix A.1
| Title 1 | Title 2 | Title 3 |
| entry 1 | data | data |
| entry 2 | data | data 1 |
Appendix B
References
- Ostrom, Q. T.; Gittleman, H.; Liao, P.; Rouse, C.; Chen, Y.; Dowling, J.; Wolinsky, Y.; Kruchko, C.; Barn-holtz-Sloan, J. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2007-2011. Neuro-Oncology 2014, 16 (suppl 4), iv1–iv63. [CrossRef]
- Grochans, S.; Cybulska, A. M.; Simińska, D.; Korbecki, J.; Kojder, K.; Chlubek, D.; Baranowska-Bosiacka, I. Epidemiology of Glioblastoma Multiforme–Literature Review. Cancers 2022, 14 (10), 2412. [CrossRef]
- Pollard, T. D.; Borisy, G. G. Cellular Motility Driven by Assembly and Disassembly of Actin Filaments. Cell 2003, 113 (4), 549. [CrossRef]
- Nürnberg, A.; Kitzing, T.; Grosse, R. Nucleating Actin for Invasion. Nat Rev Cancer 2011, 11 (3), 177–187. [CrossRef]
- Yamaguchi, H.; Condeelis, J. Regulation of the Actin Cytoskeleton in Cancer Cell Migration and Invasion. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 2007, 1773 (5), 642–652. [CrossRef]
- Nguyen, R. Y.; Xiao, H.; Gong, X.; Arroyo, A.; Cabral, A. T.; Fischer, T. T.; Flores, K. M.; Zhang, X.; Robert, M. E.; Ehrlich, B. E.; Mak, M. Cytoskeletal Dynamics Regulates Stromal Invasion Behavior of Distinct Liver Cancer Subtypes. Commun Biol 2022, 5 (1), 202. [CrossRef]
- Ridley, A. J. Rho GTPases and Cell Migration. Journal of Cell Science 2001, 114 (15), 2713–2722. [CrossRef]
- Giese, A.; Loo, M. A.; Tran, N.; Haskett, D.; Coons, S. W.; Berens, M. E. Dichotomy of Astrocytoma Migration and Proliferation. Int. J. Cancer 1996, 67 (2), 275–282. [CrossRef]
- Xie, Q.; Mittal, S.; Berens, M. E. Targeting Adaptive Glioblastoma: An Overview of Proliferation and Inva-sion. Neuro-Oncology 2014, 16 (12), 1575–1584. [CrossRef]
- Klank, R. L.; Decker Grunke, S. A.; Bangasser, B. L.; Forster, C. L.; Price, M. A.; Odde, T. J.; SantaCruz, K. S.; Rosenfeld, S. S.; Canoll, P.; Turley, E. A.; McCarthy, J. B.; Ohlfest, J. R.; Odde, D. J. Biphasic Dependence of Glioma Survival and Cell Migration on CD44 Expression Level. Cell Reports 2017, 18 (1), 23–31. [CrossRef]
- Picariello, H. S.; Kenchappa, R. S.; Rai, V.; Crish, J. F.; Dovas, A.; Pogoda, K.; McMahon, M.; Bell, E. S.; Chandrasekharan, U.; Luu, A.; West, R.; Lammerding, J.; Canoll, P.; Odde, D. J.; Janmey, P. A.; Egelhoff, T.; Rosenfeld, S. S. Myosin IIA Suppresses Glioblastoma Development in a Mechanically Sensitive Manner. Proc. Natl. Acad. Sci. U.S.A. 2019, 116 (31), 15550–15559. [CrossRef]
- Ivkovic, S.; Beadle, C.; Noticewala, S.; Massey, S. C.; Swanson, K. R.; Toro, L. N.; Bresnick, A. R.; Canoll, P.; Rosenfeld, S. S. Direct Inhibition of Myosin II Effectively Blocks Glioma Invasion in the Presence of Multi-ple Motogens. MBoC 2012, 23 (4), 533–542. [CrossRef]
- Hatzikirou, H.; Basanta, D.; Simon, M.; Schaller, K.; Deutsch, A. “Go or Grow”: The Key to the Emergence of Invasion in Tumour Progression? Math. Med. Biol. 2012, 29 (1), 49–65. [CrossRef]
- Giese, A.; Bjerkvig, R.; Berens, M. E.; Westphal, M. Cost of Migration: Invasion of Malignant Gliomas and Implications for Treatment. JCO 2003, 21 (8), 1624–1636. [CrossRef]
- Dhruv, H. D.; McDonough Winslow, W. S.; Armstrong, B.; Tuncali, S.; Eschbacher, J.; Kislin, K.; Loftus, J. C.; Tran, N. L.; Berens, M. E. Reciprocal Activation of Transcription Factors Underlies the Dichotomy between Proliferation and Invasion of Glioma Cells. PLoS ONE 2013, 8 (8), e72134. [CrossRef]
- Ulrich, T. A.; De Juan Pardo, E. M.; Kumar, S. The Mechanical Rigidity of the Extracellular Matrix Regulates the Structure, Motility, and Proliferation of Glioma Cells. Cancer Res. 2009, 69 (10), 4167–4174. [CrossRef]
- Pérez-González, A.; Bévant, K.; Blanpain, C. Cancer Cell Plasticity during Tumor Progression, Metastasis and Response to Therapy. Nat Cancer 2023, 4 (8), 1063–1082. [CrossRef]
- Wu, J.; Jiang, J.; Chen, B.; Wang, K.; Tang, Y.; Liang, X. Plasticity of Cancer Cell Invasion: Patterns and Mechanisms. Transl. Oncol. 2021, 14 (1), 100899. [CrossRef]
- Omuro, A. M. P.; Faivre, S.; Raymond, E. Lessons Learned in the Development of Targeted Therapy for Ma-lignant Gliomas. Mol. Cancer Ther. 2007, 6 (7), 1909–1919. [CrossRef]
- Woehrer, A.; Bauchet, L.; Barnholtz-Sloan, J. S. Glioblastoma Survival: Has It Improved? Evidence from Population-Based Studies. Curr. Opin. Neurol. 2014, 27 (6), 666–674. [CrossRef]
- Nakada, M.; Nakada, S.; Demuth, T.; Tran, N. L.; Hoelzinger, D. B.; Berens, M. E. Molecular Targets of Gli-oma Invasion. Cell. Mol. Life Sci. 2007, 64 (4), 458–478. [CrossRef]
- Beadle, C.; Assanah, M. C.; Monzo, P.; Vallee, R.; Rosenfeld, S. S.; Canoll, P. The Role of Myosin II in Glioma Invasion of the Brain. MBoC 2008, 19 (8), 3357–3368. [CrossRef]
- Manning, T. J.; Parker, J. C.; Sontheimer, H. Role of Lysophosphatidic Acid and Rho in Glioma Cell Motility. Cell Motil. Cytoskeleton 2000, 45 (3), 185–199. [CrossRef]
- Salhia, B.; Rutten, F.; Nakada, M.; Beaudry, C.; Berens, M.; Kwan, A.; Rutka, J. T. Inhibition of Rho-Kinase Affects Astrocytoma Morphology, Motility, and Invasion through Activation of Rac1. Cancer Res. 2005, 65 (19), 8792–8800. [CrossRef]
- Liew, H. Y.; Liew, X. H.; Lin, W. X.; Lee, Y. Z.; Ong, Y. S.; Ogawa, S.; Chong, L. H. Cellular Traction Force Holds the Potential as a Drug Testing Readout for In Vitro Cancer Metastasis. Cel. Mol. Bioeng. 2024, 17 (3), 203–217. [CrossRef]
- Zhang, Y.; Shi, X.; Zhao, T.; Huang, C.; Wei, Q.; Tang, X.; Santy, L. C.; Saif, M. T. A.; Zhang, S. A Traction Force Threshold Signifies Metastatic Phenotypic Change in Multicellular Epithelia. Soft Matter 2019, 15 (36), 7203–7210. [CrossRef]
- Vining, K. H.; Mooney, D. J. Mechanical Forces Direct Stem Cell Behaviour in Development and Regenera-tion. Nat. Rev. Mol. Cell Biol 2017, 18 (12), 728–742. [CrossRef]
- Hall, A. Rho GTPases and the Actin Cytoskeleton. Science 1998, 279. [CrossRef]
- Provenzano, P. P.; Keely, P. J. Mechanical Signaling through the Cytoskeleton Regulates Cell Proliferation by Coordinated Focal Adhesion and Rho GTPase Signaling. Journal of Cell Science 2011, 124 (8), 1195–1205. [CrossRef]
- Kumar, S.; Weaver, V. M. Mechanics, Malignancy, and Metastasis: The Force Journey of a Tumor Cell. Can-cer Metastasis Rev 2009, 28 (1–2), 113–127. [CrossRef]
- Li, Y.; Wong, I. Y.; Guo, M. Reciprocity of Cell Mechanics with Extracellular Stimuli: Emerging Opportuni-ties for Translational Medicine. Small 2022, 18 (36), 2107305. [CrossRef]
- Chen, C. S. Mechanotransduction – a Field Pulling Together? J. Cell Sci. 2008, 121 (20), 3285–3292. [CrossRef]
- Vogel, V.; Sheetz, M. Local Force and Geometry Sensing Regulate Cell Functions. Nat Rev Mol Cell Biol 2006, 7 (4), 265–275. [CrossRef]
- Paszek, M. J.; Zahir, N.; Johnson, K. R.; Lakins, J. N.; Rozenberg, G. I.; Gefen, A.; Reinhart-King, C. A.; Margulies, S. S.; Dembo, M.; Boettiger, D.; Hammer, D. A.; Weaver, V. M. Tensional Homeostasis and the Malignant Phenotype. Cancer Cell 2005, 8 (3), 241–254. [CrossRef]
- Hemmat, M.; Castle, B. T.; Odde, D. J. Microtubule Dynamics: Moving toward a Multi-Scale Approach. Curr. Opin. Cell Bio. 2018, 50, 8–13. [CrossRef]
- Akhmanova, A.; Steinmetz, M. O. Control of Microtubule Organization and Dynamics: Two Ends in the Limelight. Nat Rev Mol Cell Biol 2015, 16 (12), 711–726. [CrossRef]
- Pirani, V.; Métivier, M.; Gallaud, E.; Thomas, A.; Ku, S.; Chretien, D.; Ettari, R.; Giet, R.; Corsi, L.; Benaud, C. A Novel Benzodiazepine Derivative That Suppresses Microtubule Dynamics and Impairs Mitotic Progres-sion. J. Cell Sci. 2020, 133 (7), jcs239244. [CrossRef]
- Parenti, S.; Casagrande, G.; Montanari, M.; Espahbodinia, M.; Ettari, R.; Grande, A.; Corsi, L. A Novel 2,3-Benzodiazepine-4-One Derivative AMPA Antagonist Inhibits G2/M Transition and Induces Apoptosis in Human Leukemia Jurkat T Cell Line. Life Sci. 2016, 152, 117–125. [CrossRef]
- Micale, N.; Colleoni, S.; Postorino, G.; Pellicanò, A.; Zappalà, M.; Lazzaro, J.; Diana, V.; Cagnotto, A.; Men-nini, T.; Grasso, S. Structure–Activity Study of 2,3-Benzodiazepin-4-Ones Noncompetitive AMPAR An-tagonists: Identification of the 1-(4-Amino-3-Methylphenyl)-3,5-Dihydro-7,8-Ethylenedioxy-4H-2,3-Benzodiazepin-4-One as Neuropro-tective Agent. Bioorg. & Med. Chem. 2008, 16 (5), 2200–2211. [CrossRef]
- Boot, R. C.; Koenderink, G. H.; Boukany, P. E. Spheroid Mechanics and Implications for Cell Invasion. Adv. Phys.: X 2021, 6 (1), 1978316. [CrossRef]
- Zanoni, M.; Piccinini, F.; Arienti, C.; Zamagni, A.; Santi, S.; Polico, R.; Bevilacqua, A.; Tesei, A. 3D Tumor Spheroid Models for in Vitro Therapeutic Screening: A Systematic Approach to Enhance the Biological Relevance of Data Obtained. Sci Rep 2016, 6 (1), 19103. [CrossRef]
- Han, S. J.; Kwon, S.; Kim, K. S. Challenges of Applying Multicellular Tumor Spheroids in Preclinical Phase. Cancer Cell Int 2021, 21 (1), 152. [CrossRef]
- Patel, T.; Jain, N. Multicellular Tumor Spheroids: A Convenient in Vitro Model for Translational Cancer Research. Life Sci. 2024, 358, 123184. [CrossRef]
- Tevis, K. M.; Colson, Y. L.; Grinstaff, M. W. Embedded Spheroids as Models of the Cancer Microenviron-ment. Adv. Biosys. 2017, 1 (10), 1700083. [CrossRef]
- Mark, C.; Grundy, T. J.; Strissel, P. L.; Böhringer, D.; Grummel, N.; Gerum, R.; Steinwachs, J.; Hack, C. C.; Beckmann, M. W.; Eckstein, M.; Strick, R.; O’Neill, G. M.; Fabry, B. Collective Forces of Tumor Spheroids in Three-Dimensional Biopolymer Networks. eLife 2020, 9, e51912. [CrossRef]
- Koch, T. M.; Münster, S.; Bonakdar, N.; Butler, J. P.; Fabry, B. 3D Traction Forces in Cancer Cell Invasion. PLoS ONE 2012, 7 (3), e33476. [CrossRef]
- Kopanska, K. S.; Alcheikh, Y.; Staneva, R.; Vignjevic, D.; Betz, T. Tensile Forces Originating from Cancer Spheroids Facilitate Tumor Invasion. PLoS ONE 2016, 11 (6), e0156442. [CrossRef]
- Steinwachs, J.; Metzner, C.; Skodzek, K.; Lang, N.; Thievessen, I.; Mark, C.; Münster, S.; Aifantis, K. E.; Fabry, B. Three-Dimensional Force Microscopy of Cells in Biopolymer Networks. Nat Methods 2016, 13 (2), 171–176. [CrossRef]
- Buchmann, B.; Fernández, P.; Bausch, A. R. The Role of Nonlinear Mechanical Properties of Biomimetic Hydrogels for Organoid Growth. Biophys. Rev. 2021, 2 (2), 021401. [CrossRef]
- Gonzalez-Rodriguez, D.; Guevorkian, K.; Douezan, S.; Brochard-Wyart, F. Soft Matter Models of Develop-ing Tissues and Tumors. Science 2012, 338 (6109), 910–917. [CrossRef]
- Ehrig, S.; Schamberger, B.; Bidan, C. M.; West, A.; Jacobi, C.; Lam, K.; Kollmannsberger, P.; Petersen, A.; To-mancak, P.; Kommareddy, K.; Fischer, F. D.; Fratzl, P.; Dunlop, J. W. C. Surface Tension Determines Tissue Shape and Growth Kinetics. Sci. Adv. 2019. [CrossRef]
- Nagle, I.; Richert, A.; Quinteros, M.; Janel, S.; Buysschaert, E.; Luciani, N.; Debost, H.; Thevenet, V.; Wilhelm, C.; Prunier, C.; Lafont, F.; Padilla-Benavides, T.; Boissan, M.; Reffay, M. Surface Tension of Model Tissues during Malignant Transformation and Epithelial–Mesenchymal Transition. Front. Cell Dev. Biol. 2022, 10, 926322. [CrossRef]
- Guevorkian, K.; Colbert, M.-J.; Durth, M.; Dufour, S.; Brochard-Wyart, F. Aspiration of Biological Viscoelas-tic Drops. Phys. Rev. Lett. 2010, 104 (21), 218101. [CrossRef]
- Guevorkian, K.; Maître, J.-L. Micropipette Aspiration. In Methods in Cell Biology; Elsevier, 2017; Vol. 139, pp 187–201. [CrossRef]
- Boot, R. C.; Roscani, A.; Van Buren, L.; Maity, S.; Koenderink, G. H.; Boukany, P. E. High-Throughput Mechanophenotyping of Multicellular Spheroids Using a Microfluidic Micropipette Aspiration Chip. Lab Chip 2023, 23 (7), 1768–1778. [CrossRef]
- Guevorkian, K.; Gonzalez-Rodriguez, D.; Carlier, C.; Dufour, S.; Brochard-Wyart, F. Mechanosensitive Shivering of Model Tissues under Controlled Aspiration. Proc. Natl. Acad. Sci. U.S.A. 2011, 108 (33), 13387–13392. [CrossRef]
- Ferraro, G.; Mozzicafreddo, M.; Ettari, R.; Corsi, L.; Monti, M. C. A Proteomic Platform Unveils the Brain Glycogen Phosphorylase as a Potential Therapeutic Target for Glioblastoma Multiforme. IJMS 2022, 23 (15), 8200. [CrossRef]
- Venere, M.; Horbinski, C.; Crish, J. F.; Jin, X.; Vasanji, A.; Major, J.; Burrows, A. C.; Chang, C.; Prokop, J.; Wu, Q.; Sims, P. A.; Canoll, P.; Summers, M. K.; Rosenfeld, S. S.; Rich, J. N. The Mitotic Kinesin KIF11 Is a Driver of Invasion, Proliferation, and Self-Renewal in Glioblastoma. Sci. Transl. Med. 2015, 7 (304). [CrossRef]
- Wang, F.; Lin, S. L. Knockdown of Kinesin KIF11 Abrogates Directed Migration in Response to Epidermal Growth Factor-Mediated Chemotaxis. Biochem. Biophys. Res. Commun. 2014, 452 (3), 642–648. [CrossRef]
- Zasadil, L. M.; Andersen, K. A.; Yeum, D.; Rocque, G. B.; Wilke, L. G.; Tevaarwerk, A. J.; Raines, R. T.; Bur-kard, M. E.; Weaver, B. A. Cytotoxicity of Paclitaxel in Breast Cancer Is Due to Chromosome Missegrega-tion on Multipolar Spindles. Sci. Transl. Med. 2014, 6 (229). [CrossRef]
- Nishimura, K.; Johmura, Y.; Deguchi, K.; Jiang, Z.; Uchida, K. S. K.; Suzuki, N.; Shimada, M.; Chiba, Y.; Hi-rota, T.; Yoshimura, S. H.; Kono, K.; Nakanishi, M. Cdk1-Mediated DIAPH1 Phosphorylation Maintains Metaphase Cortical Tension and Inactivates the Spindle Assembly Checkpoint at Anaphase. Nat Commun 2019, 10 (1), 981. [CrossRef]
- Field, J. J.; Kanakkanthara, A.; Miller, J. H. Microtubule-Targeting Agents Are Clinically Successful Due to Both Mitotic and Interphase Impairment of Microtubule Function. Bioorg. & Med. Chem. 2014, 22 (18), 5050–5059. [CrossRef]
- Rieder, C. L.; Maiato, H. Stuck in Division or Passing Through. Dev. Cell 2004, 7 (5), 637–651. [CrossRef]
- Weaver, B. A. A.; Cleveland, D. W. Decoding the Links between Mitosis, Cancer, and Chemotherapy: The Mitotic Checkpoint, Adaptation, and Cell Death. Cancer Cell 2005, 8 (1), 7–12. [CrossRef]
- Miroshnikova, Y. A.; Mouw, J. K.; Barnes, J. M.; Pickup, M. W.; Lakins, J. N.; Kim, Y.; Lobo, K.; Persson, A. I.; Reis, G. F.; McKnight, T. R.; Holland, E. C.; Phillips, J. J.; Weaver, V. M. Tissue Mechanics Promote IDH1-Dependent HIF1α–Tenascin C Feedback to Regulate Glioblastoma Aggression. Nat Cell Biol 2016, 18 (12), 1336–1345. [CrossRef]
- Grundy, T. J.; De Leon, E.; Griffin, K. R.; Stringer, B. W.; Day, B. W.; Fabry, B.; Cooper-White, J.; O’Neill, G. M. Differential Response of Patient-Derived Primary Glioblastoma Cells to Environmental Stiffness. Sci Rep 2016, 6 (1), 23353. [CrossRef]
- Zhang, C.; Tan, Y.; Feng, J.; Huang, C.; Liu, B.; Fan, Z.; Xu, B.; Lu, T. Exploration of the Effects of Substrate Stiffness on Biological Responses of Neural Cells and Their Mechanisms. ACS Omega 2020, 5 (48), 31115–31125. [CrossRef]
- Feng, J.; Tang, Y.; Xu, Y.; Sun, Q.; Liao, F.; Han, D. Substrate Stiffness Influences the Outcome of Antitumor Drug Screening in Vitro. Clin. Hemorheol. Microcirc. 2013, 55 (1), 121–131. [CrossRef]
- Bangasser, B. L.; Shamsan, G. A.; Chan, C. E.; Opoku, K. N.; Tüzel, E.; Schlichtmann, B. W.; Kasim, J. A.; Fuller, B. J.; McCullough, B. R.; Rosenfeld, S. S.; Odde, D. J. Shifting the Optimal Stiffness for Cell Migration. Nat Commun 2017, 8 (1), 15313. [CrossRef]
- Seetharaman, S.; Vianay, B.; Roca, V.; Farrugia, A. J.; De Pascalis, C.; Boëda, B.; Dingli, F.; Loew, D.; Vass-ilopoulos, S.; Bershadsky, A.; Théry, M.; Etienne-Manneville, S. Microtubules Tune Mechanosensitive Cell Responses. Nat. Mater. 2022, 21 (3), 366–377. [CrossRef]
- Suresh, S.; Spatz, J.; Mills, J. P.; Micoulet, A.; Dao, M.; Lim, C. T.; Beil, M.; Seufferlein, T. Connections be-tween Single-Cell Biomechanics and Human Disease States: Gastrointestinal Cancer and Malaria. Acta Bi-omaterialia 2005, 1 (1), 15–30. [CrossRef]
- The Physical Sciences - Oncology Centers Network; Agus, D. B.; Alexander, J. F.; Arap, W.; Ashili, S.; Aslan, J. E.; Austin, R. H.; Backman, V.; Bethel, K. J.; Bonneau, R.; Chen, W.-C. et al. A Physical Sciences Network Characterization of Non-Tumorigenic and Metastatic Cells. Sci Rep 2013, 3 (1), 1449. [CrossRef]
- Xu, W.; Mezencev, R.; Kim, B.; Wang, L.; McDonald, J.; Sulchek, T. Cell Stiffness Is a Biomarker of the Meta-static Potential of Ovarian Cancer Cells. PLoS ONE 2012, 7 (10), e46609. [CrossRef]
- Luo, Q.; Kuang, D.; Zhang, B.; Song, G. Cell Stiffness Determined by Atomic Force Microscopy and Its Cor-relation with Cell Motility. Biochimica et Biophysica Acta (BBA) - General Subjects 2016, 1860 (9), 1953–1960. [CrossRef]
- Urbanska, M.; Guck, J. Single-Cell Mechanics: Structural Determinants and Functional Relevance. Annu. Rev. Biophys. 2024, 53 (1), 367–395. [CrossRef]
- Zhang, G.; Long, M.; Wu, Z.-Z.; Yu, W.-Q. Mechanical Properties of Hepatocellular Carcinoma Cells. WJG 2002, 8 (2), 243. [CrossRef]
- Mizuno, D.; Tardin, C.; Schmidt, C. F.; MacKintosh, F. C. Nonequilibrium Mechanics of Active Cytoskeletal Networks. Science 2007, 315 (5810), 370–373. [CrossRef]
- Alcaraz, J.; Buscemi, L.; Grabulosa, M.; Trepat, X.; Fabry, B.; Farré, R.; Navajas, D. Microrheology of Human Lung Epithelial Cells Measured by Atomic Force Microscopy. Biophys. J. 2003, 84 (3), 2071–2079. [CrossRef]
- Mahaffy, R. E.; Shih, C. K.; MacKintosh, F. C.; Käs, J. Scanning Probe-Based Frequency-Dependent Micro-rheology of Polymer Gels and Biological Cells. Phys. Rev. Lett. 2000, 85 (4), 880–883. [CrossRef]
- Bershadsky, A.; Chausovsky, A.; Becker, E.; Lyubimova, A.; Geiger, B. Involvement of Microtubules in the Control of Adhesion-Dependent Signal Transduction. Curr. Biol. 1996, 6 (10), 1279–1289. [CrossRef]
- Liu, B. P.; Chrzanowska-Wodnicka, M.; Burridge, K. Microtubule Depolymerization Induces Stress Fibers, Focal Adhesions, and DNA Synthesis via the GTP-Binding Protein Rho. Cell Adhesion and Communication 1998, 5 (4), 249–255. [CrossRef]
- Takahashi, R.; Okajima, T. Mapping Power-Law Rheology of Living Cells Using Multi-Frequency Force Modulation Atomic Force Microscopy. Appl. Phys. Lett. 2015, 107 (17), 173702. [CrossRef]
- Efremov, Y. M.; Wang, W.-H.; Hardy, S. D.; Geahlen, R. L.; Raman, A. Measuring Nanoscale Viscoelastic Parameters of Cells Directly from AFM Force-Displacement Curves. Sci Rep 2017, 7 (1), 1541. [CrossRef]
- Areti, A.; Komirishetty, P.; Zochodne, D. W. Collaborative Roles for RAC1, ERM Proteins and PTEN During Adult Sensory Axon Regeneration. Mol Neurobiol 2024. [CrossRef]
- Schneider, I. C.; Hays, C. K.; Waterman, C. M. Epidermal Growth Factor–Induced Contraction Regulates Paxillin Phosphorylation to Temporally Separate Traction Generation from De-Adhesion. MBoC 2009, 20 (13), 3155–3167. [CrossRef]
- Panopoulos, A.; Howell, M.; Fotedar, R.; Margolis, R. L. Glioblastoma Motility Occurs in the Absence of Ac-tin Polymer. MBoC 2011, 22 (13), 2212–2220. [CrossRef]
- Rosenblatt, J.; Cramer, L. P.; Baum, B.; McGee, K. M. Myosin II-Dependent Cortical Movement Is Required for Centrosome Separation and Positioning during Mitotic Spindle Assembly. Cell 2004, 117 (3), 361–372. [CrossRef]
- Chang, Y.-C.; Nalbant, P.; Birkenfeld, J.; Chang, Z.-F.; Bokoch, G. M. GEF-H1 Couples Nocodazole-Induced Microtubule Disassembly to Cell Contractility via RhoA. MBoC 2008, 19 (5), 2147–2153. [CrossRef]
- Lv, S.; Chen, Z.; Mi, H.; Yu, X. Cofilin Acts as a Booster for Progression of Malignant Tumors Represented by Glioma. CMAR 2022, Volume 14, 3245–3269. [CrossRef]
- Chignola, R.; Schenetti, A.; Andrighetto, G.; Chiesa, E.; Foroni, R.; Sartoris, S.; Tridente, G.; Liberati, D. Forecasting the Growth of Multicell Tumour Spheroids: Implications for the Dynamic Growth of Solid Tumours. Cell Prolif. 2000, 33 (4), 219–229. [CrossRef]
- Marušić, M.; Bajzer, Ž.; Freyer, J. P.; Vuk-Pavlović, S. Analysis of Growth of Multicellular Tumour Sphe-roids by Mathematical Models. Cell Prolif. 1994, 27 (2), 73–94. [CrossRef]
- Bangasser, B. L.; Rosenfeld, S. S.; Odde, D. J. Determinants of Maximal Force Transmission in a Mo-tor-Clutch Model of Cell Traction in a Compliant Microenvironment. Biophys. J. 2013, 105 (3), 581–592. [CrossRef]
- Stroka, K. M.; Aranda-Espinoza, H. Neutrophils Display Biphasic Relationship between Migration and Substrate Stiffness. Cell Motil. Cytoskeleton 2009, 66 (6), 328–341. [CrossRef]
- Peyton, S. R.; Putnam, A. J. Extracellular Matrix Rigidity Governs Smooth Muscle Cell Motility in a Bipha-sic Fashion. J.Cell.Physiol. 2005, 204 (1), 198–209. [CrossRef]
- (95) DiMilla, P. A.; Barbee, K.; Lauffenburger, D. A. Mathematical Model for the Effects of Adhesion and Mechanics on Cell Migration Speed. Biophys. J. 1991, 60 (1), 15–37. [CrossRef]
- DiMilla, P.; Stone, J.; Quinn, J.; Albelda, S.; Lauffenburger, D. Maximal Migration of Human Smooth Muscle Cells on Fibronectin and Type IV Collagen Occurs at an Intermediate Attachment Strength. JCB 1993, 122 (3), 729–737. [CrossRef]
- Prahl, L. S.; Odde, D. J. Modeling Cell Migration Mechanics. In Biomechanics in Oncology; Dong, C., Zahir, N., Konstantopoulos, K., Eds.; Advances in Experimental Medicine and Biology; Springer International Pub-lishing: Cham, 2018; Vol. 1092, pp 159–187. [CrossRef]
- Kamath, K.; Smiyun, G.; Wilson, L.; Jordan, M. A. Mechanisms of Inhibition of Endothelial Cell Migration by Taxanes. Cytoskeleton 2014, 71 (1), 46–60. [CrossRef]
- Honoré, S.; Pagano, A.; Gauthier, G.; Bourgarel-Rey, V.; Verdier-Pinard, P.; Civiletti, K.; Kruczynski, A.; Braguer, D. Antiangiogenic Vinflunine Affects EB1 Localization and Microtubule Targeting to Adhesion Sites. Mol. Cancer Ther. 2008, 7 (7), 2080–2089. [CrossRef]
- Rafiq, N. B. M.; Nishimura, Y.; Plotnikov, S. V.; Thiagarajan, V.; Zhang, Z.; Shi, S.; Natarajan, M.; Viasnoff, V.; Kanchanawong, P.; Jones, G. E.; Bershadsky, A. D. A Mechano-Signalling Network Linking Microtubules, Myosin IIA Filaments and Integrin-Based Adhesions. Nat. Mater. 2019, 18 (6), 638–649. [CrossRef]
- Prahl, L. S.; Bangasser, P. F.; Stopfer, L. E.; Hemmat, M.; White, F. M.; Rosenfeld, S. S.; Odde, D. J. Microtu-bule-Based Control of Motor-Clutch System Mechanics in Glioma Cell Migration. Cell Reports 2018, 25 (9), 2591-2604.e8. [CrossRef]
- Krylyshkina, O.; Anderson, K. I.; Kaverina, I.; Upmann, I.; Manstein, D. J.; Small, J. V.; Toomre, D. K. Na-nometer Targeting of Microtubules to Focal Adhesions. JCB 2003, 161 (5), 853–859. [CrossRef]
- Bouchet, B. P.; Gough, R. E.; Ammon, Y.-C.; Van De Willige, D.; Post, H.; Jacquemet, G.; Altelaar, A. M.; Heck, A. J.; Goult, B. T.; Akhmanova, A. Talin-KANK1 Interaction Controls the Recruitment of Cortical Microtubule Stabilizing Complexes to Focal Adhesions. eLife 2016, 5, e18124. [CrossRef]
- Castle, B. T.; McCubbin, S.; Prahl, L. S.; Bernens, J. N.; Sept, D.; Odde, D. J. Mechanisms of Kinetic Stabiliza-tion by the Drugs Paclitaxel and Vinblastine. MBoC 2017, 28 (9), 1238–1257. [CrossRef]
- Kwon, M.; Godinho, S. A.; Chandhok, N. S.; Ganem, N. J.; Azioune, A.; Thery, M.; Pellman, D. Mechanisms to Suppress Multipolar Divisions in Cancer Cells with Extra Centrosomes. Genes Dev. 2008, 22 (16), 2189–2203. [CrossRef]
- Bates, D.; Eastman, A. Microtubule Destabilising Agents: Far More than Just Antimitotic Anticancer Drugs. Brit J Clinical Pharma 2017, 83 (2), 255–268. [CrossRef]
- Chen, Y.; Chou, W.-C.; Ding, Y.-M.; Wu, Y.-C. Caffeine Inhibits Migration in Glioma Cells through the ROCK-FAK Pathway. Cell Physiol Biochem 2014, 33 (6), 1888–1898. [CrossRef]
- Pardi, R. Signal Transduction by Adhesion Receptors. Nature Education 3(9):38.
- Kim, Y.-B.; Choi, S.; Choi, M.-C.; Oh, M.-A.; Lee, S.-A.; Cho, M.; Mizuno, K.; Kim, S.-H.; Lee, J. W. Cell Adhe-sion-Dependent Cofilin Serine 3 Phosphorylation by the Integrin-Linked Kinase·c-Src Complex. JBC 2008, 283 (15), 10089–10096. [CrossRef]
- Beri, P.; Popravko, A.; Yeoman, B.; Kumar, A.; Chen, K.; Hodzic, E.; Chiang, A.; Banisadr, A.; Placone, J. K.; Carter, H.; Fraley, S. I.; Katira, P.; Engler, A. J. Cell Adhesiveness Serves as a Biophysical Marker for Meta-static Potential. Cancer Res. 2020, 80 (4), 901–911. [CrossRef]
- Leguay, K.; Decelle, B.; Elkholi, I. E.; Bouvier, M.; Côté, J.-F.; Carréno, S. Interphase Microtubule Disassembly Is a Signaling Cue That Drives Cell Rounding at Mitotic Entry. JCB 2022, 221 (6), e202109065. [CrossRef]
- Solinet, S.; Mahmud, K.; Stewman, S. F.; Ben El Kadhi, K.; Decelle, B.; Talje, L.; Ma, A.; Kwok, B. H.; Carreno, S. The Actin-Binding ERM Protein Moesin Binds to and Stabilizes Microtubules at the Cell Cortex. JCB 2013, 202 (2), 251–260. [CrossRef]
- Fievet, B. T.; Gautreau, A.; Roy, C.; Del Maestro, L.; Mangeat, P.; Louvard, D.; Arpin, M. Phosphoinositide Binding and Phosphorylation Act Sequentially in the Activation Mechanism of Ezrin. JCB 2004, 164 (5), 653–659. [CrossRef]
- Tachibana, K.; Haghparast, S. M. A.; Miyake, J. Inhibition of Cell Adhesion by Phosphorylated Ezrin/Radixin/Moesin. Cell Adhes.Migr. 2015, 9 (6), 502–512. [CrossRef]
- Fukata, M.; Watanabe, T.; Noritake, J.; Nakagawa, M.; Yamaga, M.; Kuroda, S.; Matsuura, Y.; Iwamatsu, A.; Perez, F.; Kaibuchi, K. Rac1 and Cdc42 Capture Microtubules through IQGAP1 and CLIP-170. Cell 2002, 109 (7), 873–885. [CrossRef]
- Michaelson, D.; Abidi, W.; Guardavaccaro, D.; Zhou, M.; Ahearn, I.; Pagano, M.; Philips, M. R. Rac1 Accu-mulates in the Nucleus during the G2 Phase of the Cell Cycle and Promotes Cell Division. JCB 2008, 181 (3), 485–496. [CrossRef]
- Stehbens, S. J.; Paszek, M.; Pemble, H.; Ettinger, A.; Gierke, S.; Wittmann, T. CLASPs Link Fo-cal-Adhesion-Associated Microtubule Capture to Localized Exocytosis and Adhesion Site Turnover. Nat Cell Biol 2014, 16 (6), 558–570. [CrossRef]
- Grill, S. W.; Howard, J.; Schäffer, E.; Stelzer, E. H. K.; Hyman, A. A. The Distribution of Active Force Gener-ators Controls Mitotic Spindle Position. Science 2003, 301 (5632), 518–521. [CrossRef]
- Ragazzini, G.; Mescola, A.; Corsi, L.; Alessandrini, A. Fabrication of a Low-Cost on-Stage Cell Incubator with Full Automation. J. Biol. Educ. 2018 53(2), 165–173.
- Di Cerbo, A.; Rubino, V.; Morelli, F.; Ruggiero, G.; Landi, R.; Guidetti, G.; Canello, S.; Terrazzano, G.; Ales-sandrini, A. Mechanical Phenotyping of K562 Cells by the Micropipette Aspiration Technique Allows Iden-tifying Mechanical Changes Induced by Drugs. Sci Rep 2018, 8 (1), 1219. [CrossRef]
- Hans-Jurgen, B.; Jaschke, Manfred. I Calculation of Thermal Noise in Atomic Force Microscopy. Nanotech-nology 1995, 6, 1. [CrossRef]
- Alcaraz, J.; Buscemi, L.; Puig-de-Morales, M.; Colchero, J.; Baró, A.; Navajas, D. Correction of Microrheo-logical Measurements of Soft Samples with Atomic Force Microscopy for the Hydrodynamic Drag on the Cantilever. Langmuir 2002, 18 (3), 716–721. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




