Submitted:
21 January 2025
Posted:
22 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
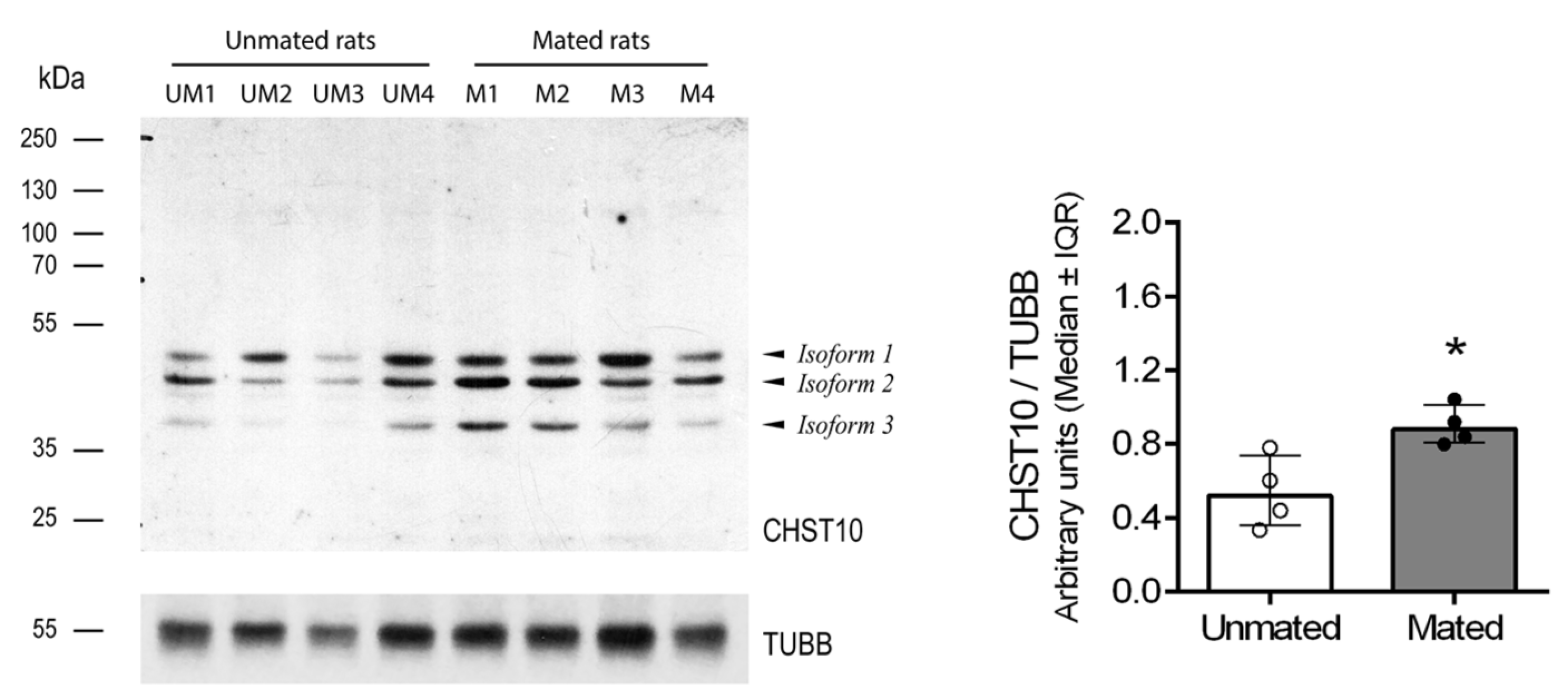
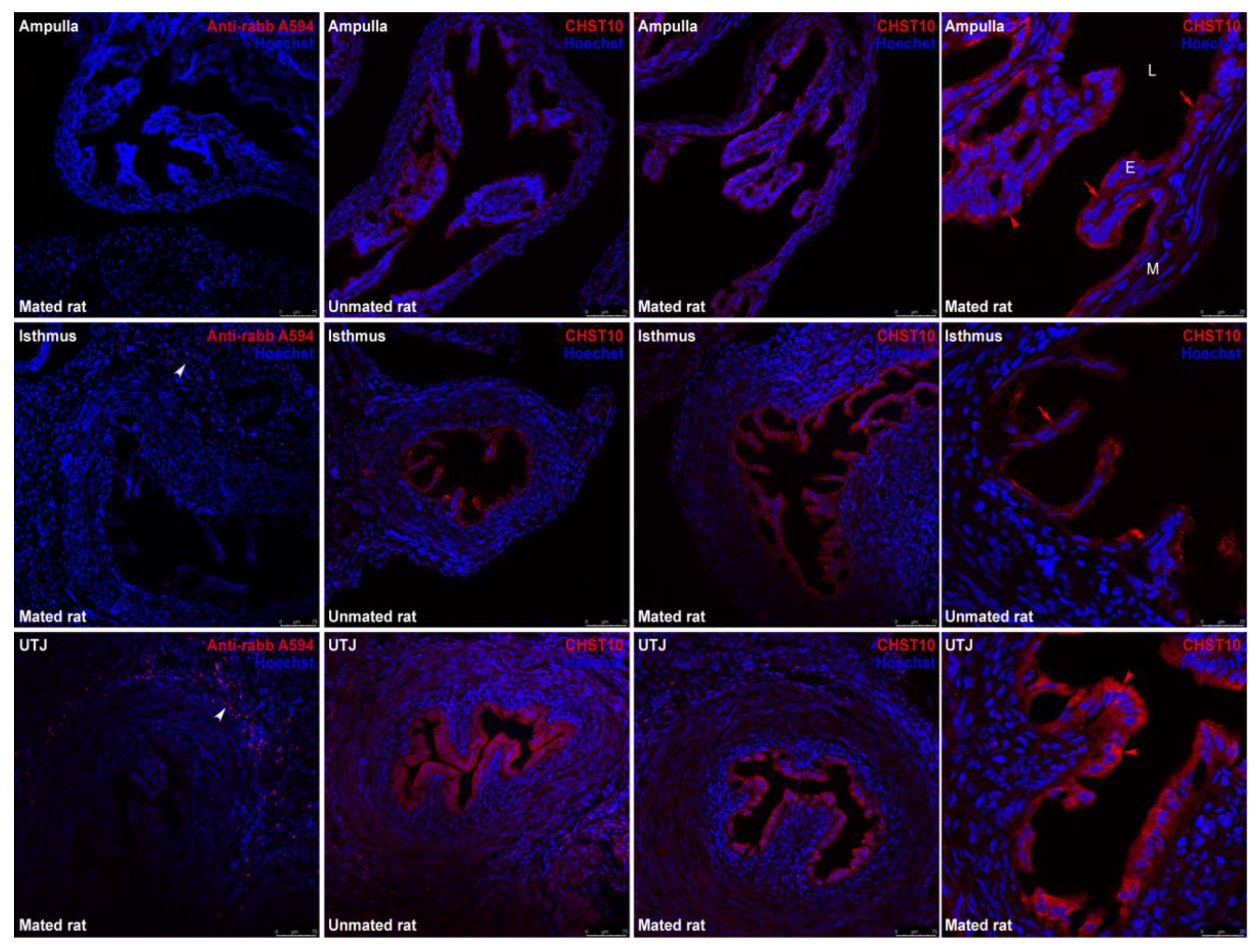
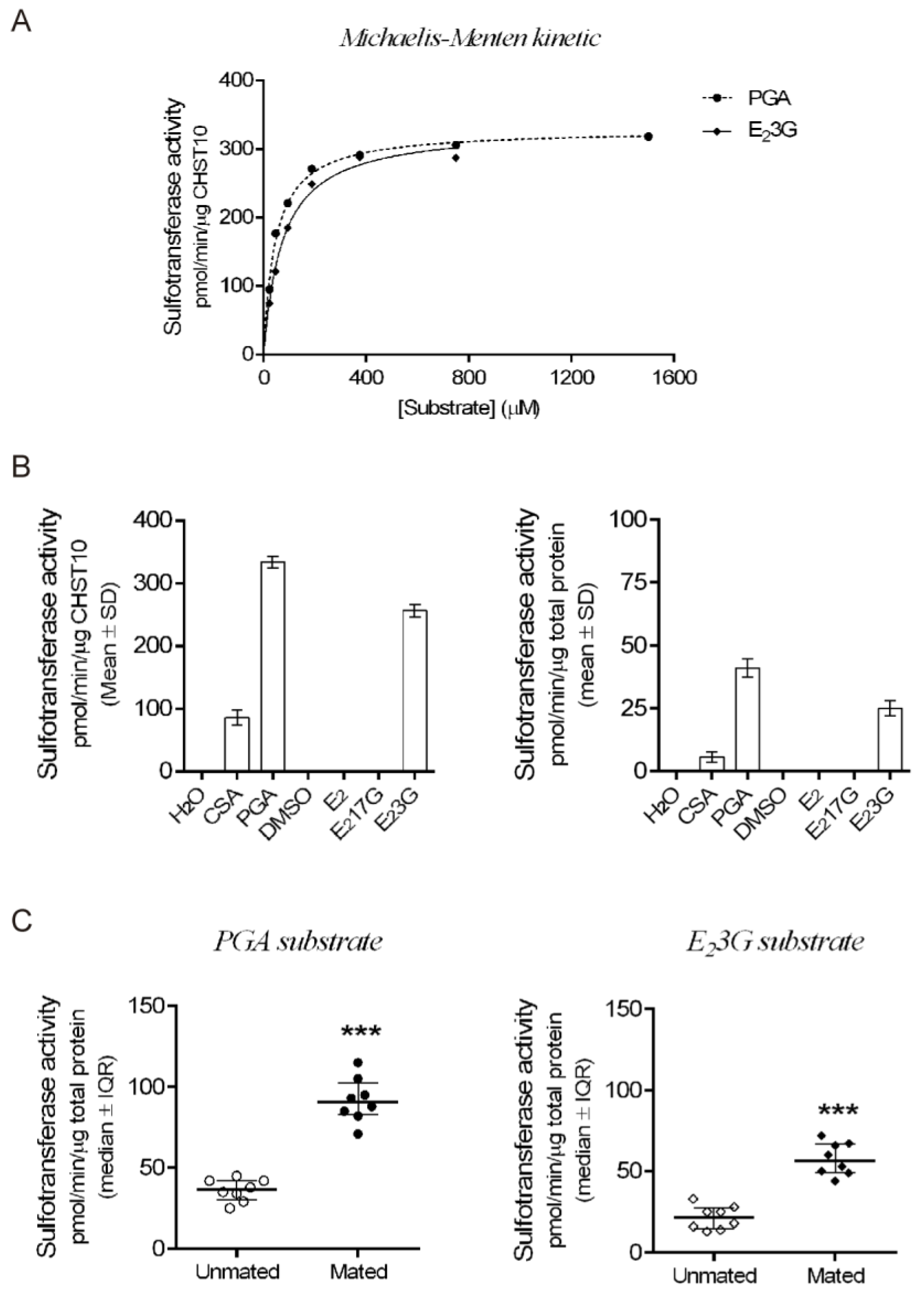
2.1. Mating Increases CHST10 Protein Level and Sulfotransferase Activity in the Oviductal Mucosa
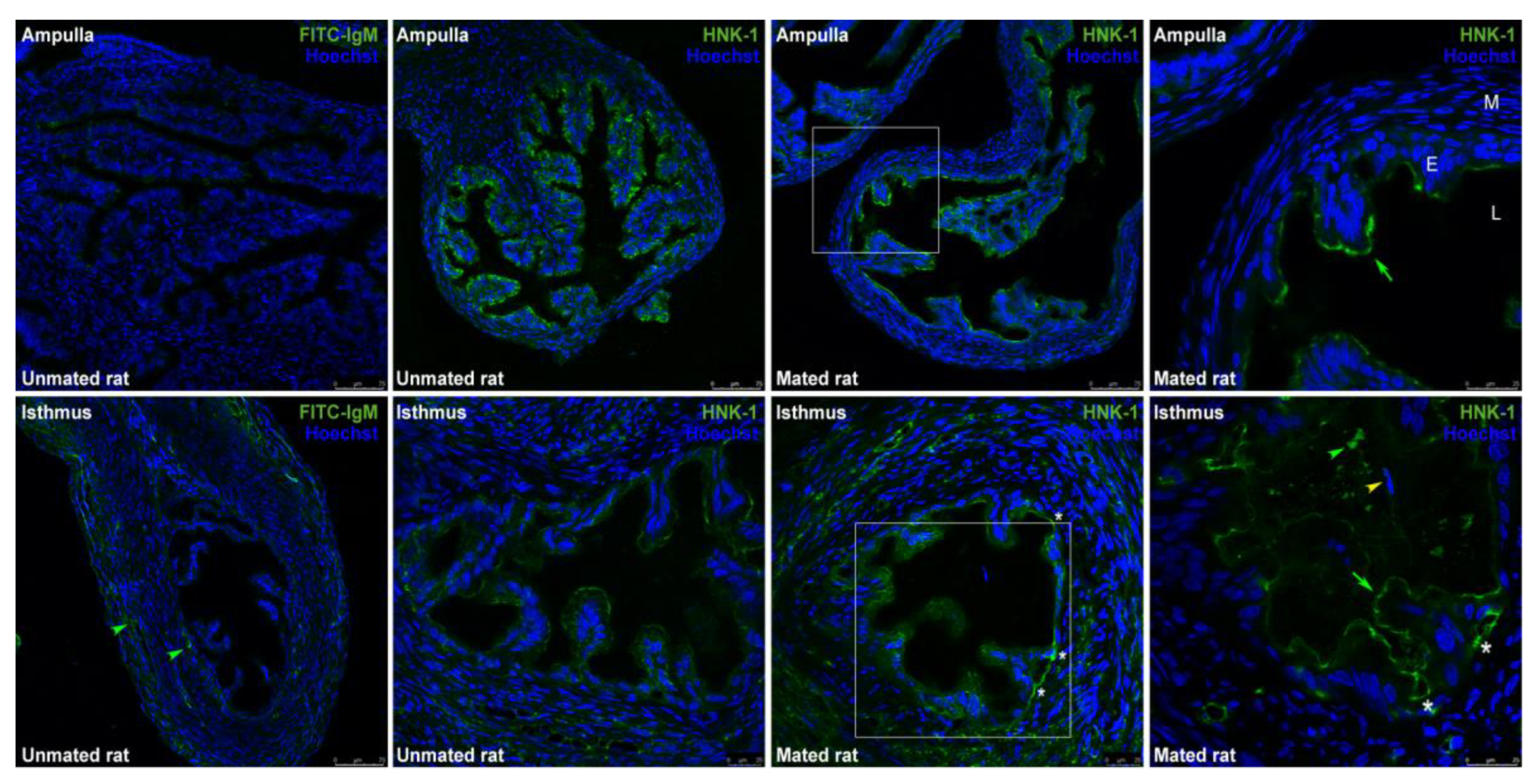
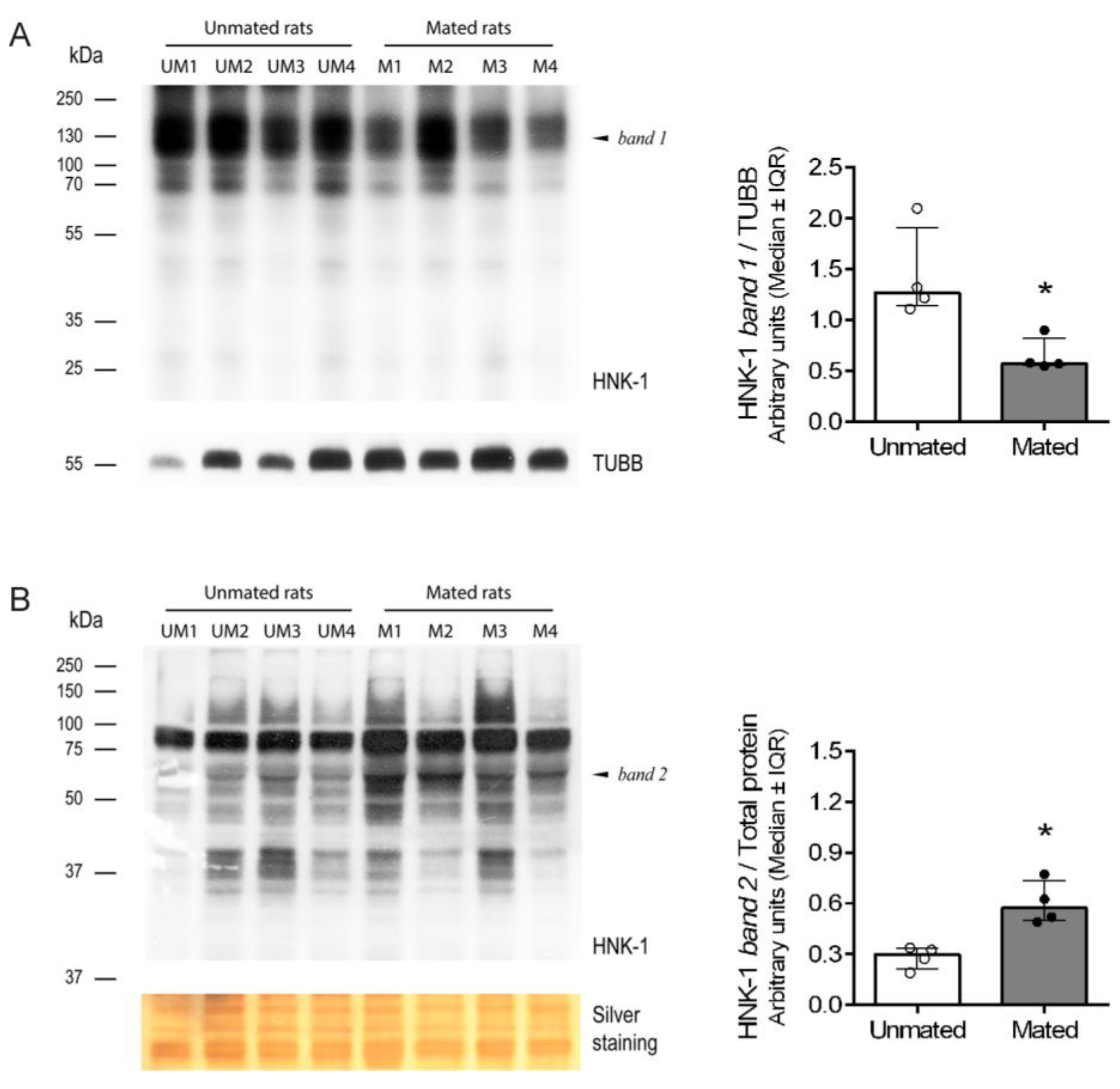
2.2. Mating Changes the Levels of HNK-1 Glycoproteins in the Oviductal Mucosa
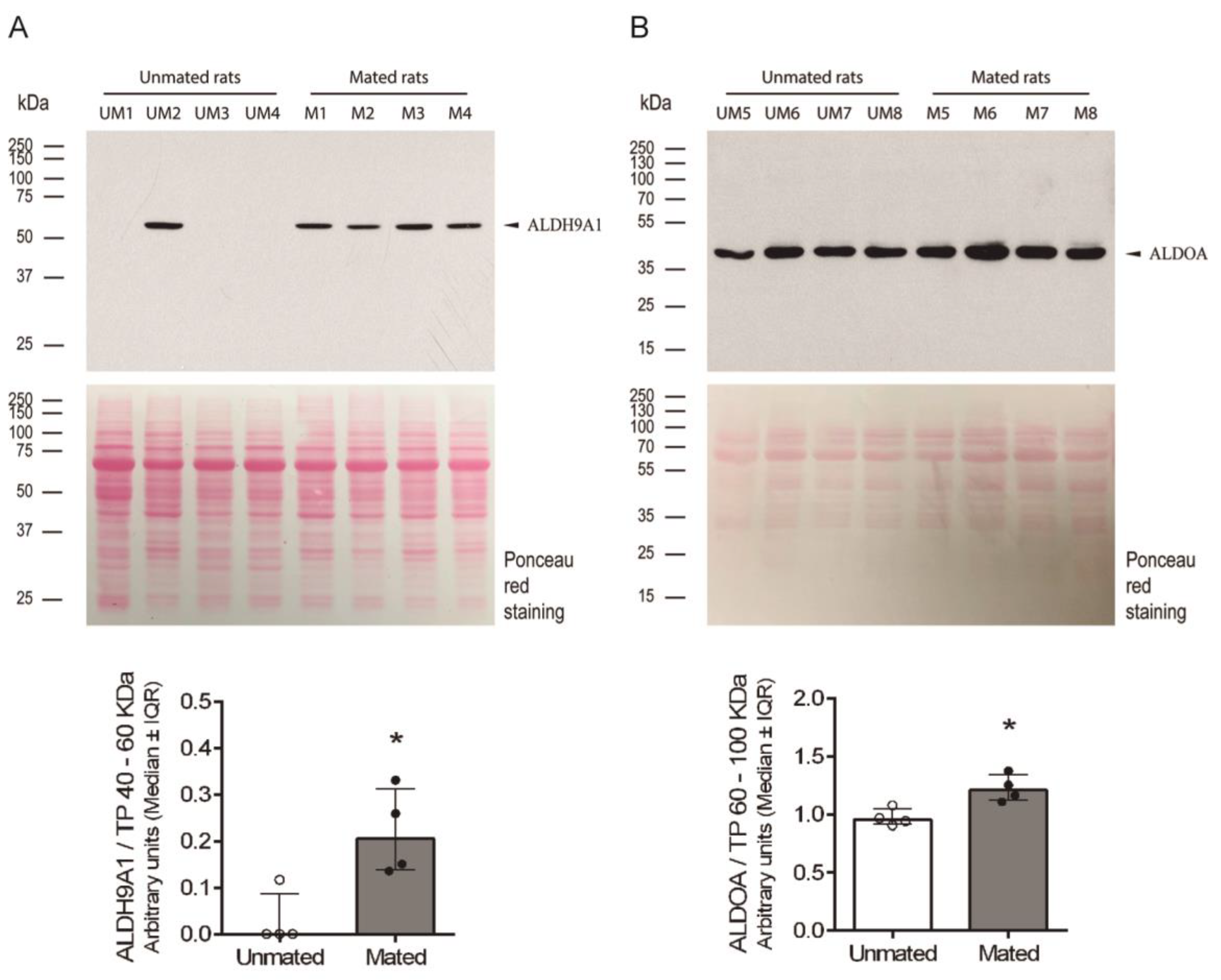
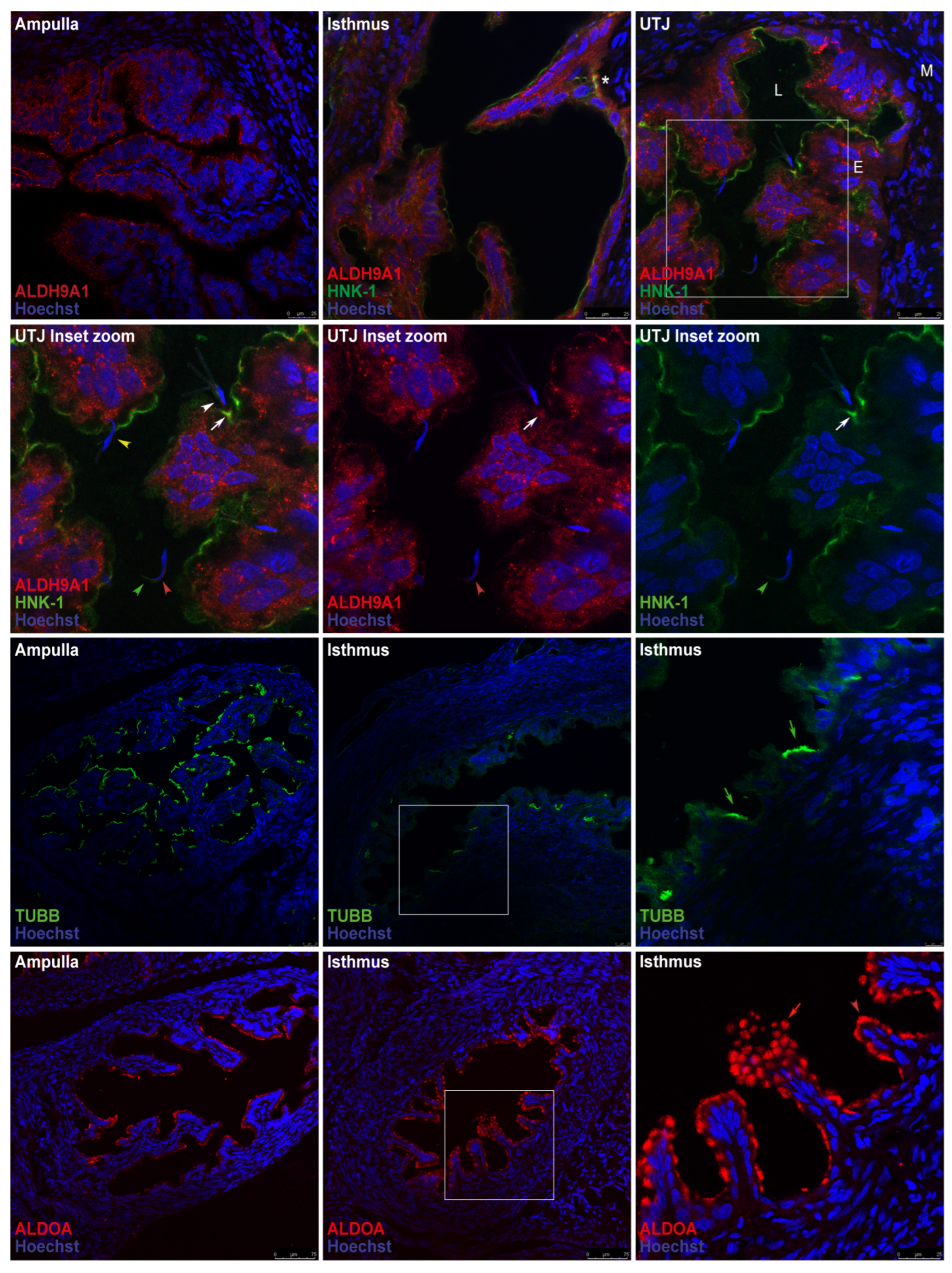
2.3. Mating Induces the Secretion of ALDH9A1 and Increases the Secretion of ALDOA from the Epithelial Cells of Oviductal Mucosa.
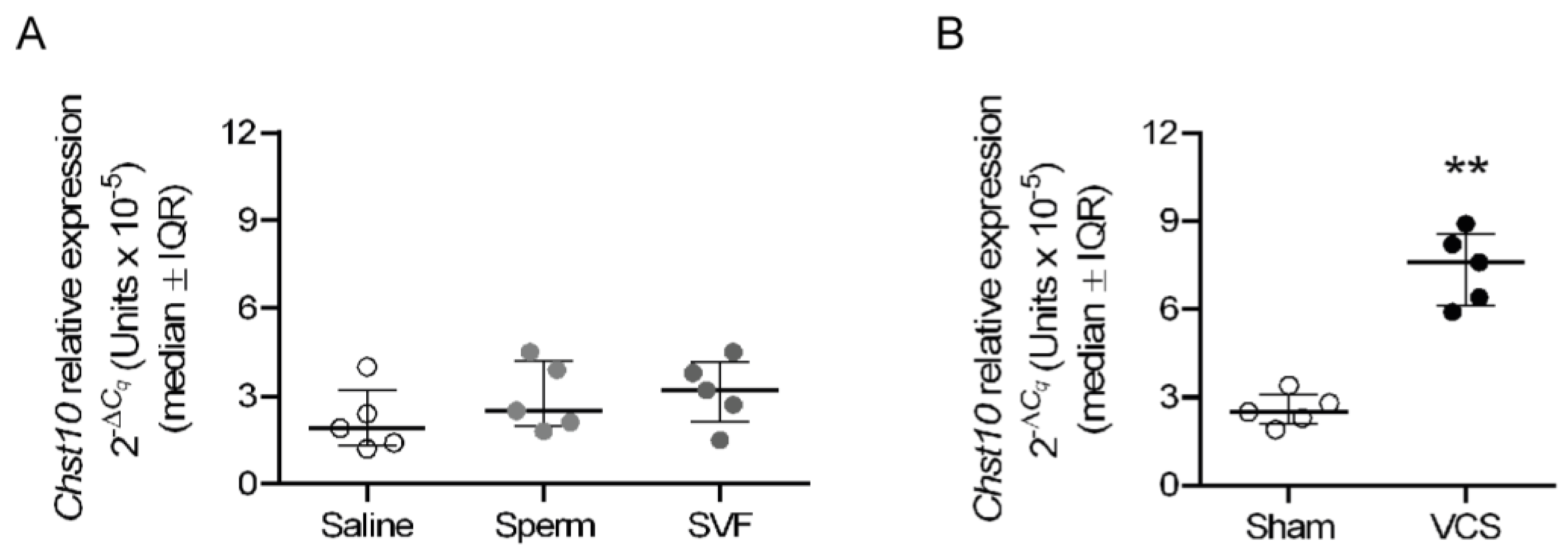
2.4. Vaginocervical Stimulation (VCS) Increases the Expression of Chst10 in the Oviductal Mucosa
3. Discussion
4. Materials and Methods
4.1. Animals
4.1.1. Mating and Sample Collection
4.1.2. Sperm and Seminal Vesicle Fluid (SVF) Collection
4.1.3. Intrauterine Injection of Sperm or SVF
4.1.4. Vaginocervical Stimulation (VCS)
4.2. Sulfotransferase Activity
4.3. Immunofluorescence
4.4. Western Blotting
4.4.1. Sample Preparation:
4.4.2.1. D Electrophoresis and Western Blotting:
4.4.3.2. D Electrophoresis and Western Blotting:
4.5. Protein Identification
4.5.1. Destaining and Drying the Gel Pieces:
4.5.2. Trypsin Digestion:
4.5.3. Liquid Chromatography and Mass Spectrometry Analysis:
4.5.4. Criteria for Protein Identification:
4.6. Relative Gene Expression Levels
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zuniga, L.M.; Andrade, J.C.; Fabrega-Gueren, F.; Orihuela, P.A.; Velasquez, E.V.; Vidal, E.A.; Gutierrez, R.A.; Morales, P.; Gomez-Silva, B.; Croxatto, H.B. Mating induces early transcriptional response in the rat endosalpinx: the role of TNF and RA. Reproduction 2021, 161, 43–59. [Google Scholar] [CrossRef] [PubMed]
- Kared, H.; Martelli, S.; Ng, T.P.; Pender, S.L.; Larbi, A. CD57 in human natural killer cells and T-lymphocytes. Cancer Immunol Immunother 2016, 65, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Morita, I.; Kizuka, Y.; Kakuda, S.; Oka, S. Expression and function of the HNK-1 carbohydrate. J Biochem 2008, 143, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Schachner, M.; Martini, R. Glycans and the modulation of neural-recognition molecule function. Trends Neurosci 1995, 18, 183–191. [Google Scholar] [CrossRef]
- Hashiguchi, T.; Mizumoto, S.; Nishimura, Y.; Tamura, J.; Yamada, S.; Sugahara, K. Involvement of human natural killer-1 (HNK-1) sulfotransferase in the biosynthesis of the GlcUA(3-O-sulfate)-Gal-Gal-Xyl tetrasaccharide found in alpha-thrombomodulin from human urine. J Biol Chem 2011, 286, 33003–33011. [Google Scholar] [CrossRef]
- Erickson-Lawrence, M.F.; Turner, T.T.; Ross, P.; Thomas, T.S.; Oliphant, G. Sulfated oviductal glycoproteins in the rabbit: quantitation by competitive enzyme-linked immunosorbent assay. Biol Reprod 1989, 40, 1299–1310. [Google Scholar] [CrossRef]
- Murray, M.K. Epithelial lining of the sheep ampulla oviduct undergoes pregnancy-associated morphological changes in secretory status and cell height. Biol Reprod 1995, 53, 653–663. [Google Scholar] [CrossRef]
- Suzuki-Anekoji, M.; Suzuki, A.; Wu, S.W.; Angata, K.; Murai, K.K.; Sugihara, K.; Akama, T.O.; Khoo, K.H.; Nakayama, J.; Fukuda, M.N.; et al. In vivo regulation of steroid hormones by the Chst10 sulfotransferase in mouse. J Biol Chem 2013, 288, 5007–5016. [Google Scholar] [CrossRef]
- Forcelledo, M.L.; Vera, R.; Croxatto, H.B. Ovum transport in pregnant, pseudopregnant, and cyclic rats and its relationship to estradiol and progesterone blood levels. Biol Reprod 1981, 24, 760–765. [Google Scholar] [CrossRef]
- Prather, B.; Ethen, C.M.; Machacek, M.; Wu, Z.L. Golgi-resident PAP-specific 3'-phosphatase-coupled sulfotransferase assays. Anal Biochem 2012, 423, 86–92. [Google Scholar] [CrossRef]
- Diekman, A.B. Glycoconjugates in sperm function and gamete interactions: how much sugar does it take to sweet-talk the egg? Cell Mol Life Sci 2003, 60, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Suarez, S.S. Mammalian sperm interactions with the female reproductive tract. Cell Tissue Res 2016, 363, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Topfer-Petersen, E.; Ekhlasi-Hundrieser, M.; Tsolova, M. Glycobiology of fertilization in the pig. Int J Dev Biol 2008, 52, 717–736. [Google Scholar] [CrossRef] [PubMed]
- Voshol, H.; van Zuylen, C.W.; Orberger, G.; Vliegenthart, J.F.; Schachner, M. Structure of the HNK-1 carbohydrate epitope on bovine peripheral myelin glycoprotein P0. J Biol Chem 1996, 271, 22957–22960. [Google Scholar] [CrossRef] [PubMed]
- Bakker, H.; Friedmann, I.; Oka, S.; Kawasaki, T.; Nifant'ev, N.; Schachner, M.; Mantei, N. Expression cloning of a cDNA encoding a sulfotransferase involved in the biosynthesis of the HNK-1 carbohydrate epitope. J Biol Chem 1997, 272, 29942–29946. [Google Scholar] [CrossRef]
- Nagase, T.; Shimoda, Y.; Sanai, Y.; Nakamura, S.; Harii, K.; Osumi, N. Differential expression of two glucuronyltransferases synthesizing HNK-1 carbohydrate epitope in the sublineages of the rat myogenic progenitors. Mech Dev 2000, 98, 145–149. [Google Scholar] [CrossRef]
- Nakagawa, N.; Manya, H.; Toda, T.; Endo, T.; Oka, S. Human natural killer-1 sulfotransferase (HNK-1ST)-induced sulfate transfer regulates laminin-binding glycans on alpha-dystroglycan. J Biol Chem 2012, 287, 30823–30832. [Google Scholar] [CrossRef]
- Pasqualini, J.R. Estrogen sulfotransferases in breast and endometrial cancers. Ann N Y Acad Sci 2009, 1155, 88–98. [Google Scholar] [CrossRef]
- Harwalkar, K.; Ford, M.J.; Teng, K.; Yamanaka, N.; Yang, B.; Burtscher, I.; Lickert, H.; Yamanaka, Y. Anatomical and cellular heterogeneity in the mouse oviduct-its potential roles in reproduction and preimplantation developmentdagger. Biol Reprod 2021, 104, 1249–1261. [Google Scholar] [CrossRef]
- La Spina, F.A.; Puga Molina, L.C.; Romarowski, A.; Vitale, A.M.; Falzone, T.L.; Krapf, D.; Hirohashi, N.; Buffone, M.G. Mouse sperm begin to undergo acrosomal exocytosis in the upper isthmus of the oviduct. Dev Biol 2016, 411, 172–182. [Google Scholar] [CrossRef]
- Muro, Y.; Hasuwa, H.; Isotani, A.; Miyata, H.; Yamagata, K.; Ikawa, M.; Yanagimachi, R.; Okabe, M. Behavior of Mouse Spermatozoa in the Female Reproductive Tract from Soon after Mating to the Beginning of Fertilization. Biol Reprod 2016, 94, 80. [Google Scholar] [CrossRef] [PubMed]
- Suarez, S.S. Sperm transport and motility in the mouse oviduct: observations in situ. Biol Reprod 1987, 36, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Cortes, P.P.; Orihuela, P.A.; Zuniga, L.M.; Velasquez, L.A.; Croxatto, H.B. Sperm binding to oviductal epithelial cells in the rat: role of sialic acid residues on the epithelial surface and sialic acid-binding sites on the sperm surface. Biol Reprod 2004, 71, 1262–1269. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.T.; Yanagimachi, R. The viability of hamster spermatozoa stored in the isthmus of the oviduct: the importance of sperm-epithelium contact for sperm survival. Biol Reprod 1990, 42, 450–457. [Google Scholar] [CrossRef]
- Suarez, S.S. Regulation of sperm storage and movement in the mammalian oviduct. Int J Dev Biol 2008, 52, 455–462. [Google Scholar] [CrossRef]
- Holt, W.V.; Fazeli, A. The oviduct as a complex mediator of mammalian sperm function and selection. Mol Reprod Dev 2010, 77, 934–943. [Google Scholar] [CrossRef]
- Muzio, G.; Maggiora, M.; Paiuzzi, E.; Oraldi, M.; Canuto, R.A. Aldehyde dehydrogenases and cell proliferation. Free Radic Biol Med 2012, 52, 735–746. [Google Scholar] [CrossRef]
- Kurys, G.; Ambroziak, W.; Pietruszko, R. Human aldehyde dehydrogenase. Purification and characterization of a third isozyme with low Km for gamma-aminobutyraldehyde. J Biol Chem 1989, 264, 4715–4721. [Google Scholar] [CrossRef]
- Jin, J.Y.; Chen, W.Y.; Zhou, C.X.; Chen, Z.H.; Yu-Ying, Y.; Ni, Y.; Chan, H.C.; Shi, Q.X. Activation of GABAA receptor/Cl- channel and capacitation in rat spermatozoa: HCO3- and Cl- are essential. Syst Biol Reprod Med 2009, 55, 97–108. [Google Scholar] [CrossRef]
- Kurata, S.; Hiradate, Y.; Umezu, K.; Hara, K.; Tanemura, K. Capacitation of mouse sperm is modulated by gamma-aminobutyric acid (GABA) concentration. J Reprod Dev 2019, 65, 327–334. [Google Scholar] [CrossRef]
- Puente, M.A.; Tartaglione, C.M.; Ritta, M.N. Bull sperm acrosome reaction induced by gamma-aminobutyric acid (GABA) is mediated by GABAergic receptors type A. Anim Reprod Sci 2011, 127, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.X.; Yuan, Y.Y.; Roldan, E.R. gamma-Aminobutyric acid (GABA) induces the acrosome reaction in human spermatozoa. Mol Hum Reprod 1997, 3, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Martin del Rio, R. Gamma-aminobutyric acid system in rat oviduct. J Biol Chem 1981, 256, 9816–9819. [Google Scholar] [CrossRef] [PubMed]
- Orensanz, L.M.; Fernandez, I.; Martin del Rio, R.; Storm-Mathisen, J. Gamma-aminobutyric acid in the rat oviduct. Adv Biochem Psychopharmacol 1986, 42, 265–274. [Google Scholar]
- Koncitikova, R.; Vigouroux, A.; Kopecna, M.; Sebela, M.; Morera, S.; Kopecny, D. Kinetic and structural analysis of human ALDH9A1. Biosci Rep 2019, 39. [Google Scholar] [CrossRef]
- Fernandez, C.; Sharrard, R.M.; Talbot, M.; Reed, B.D.; Monks, N. Evaluation of the significance of polyamines and their oxidases in the aetiology of human cervical carcinoma. Br J Cancer 1995, 72, 1194–1199. [Google Scholar] [CrossRef]
- Mann, T. Secretory function of the prostate, seminal vesicle and other male accessory organs of reproduction. J Reprod Fertil 1974, 37, 179–188. [Google Scholar] [CrossRef]
- Breitbart, H.; Rubinstein, S.; Lax, Y. Regulatory mechanisms in acrosomal exocytosis. Rev Reprod 1997, 2, 165–174. [Google Scholar] [CrossRef]
- Rubinstein, S.; Breitbart, H. Role of spermine in mammalian sperm capacitation and acrosome reaction. Biochem J 1991, 278 ( Pt 1) Pt 1, 25–28. [Google Scholar] [CrossRef]
- Shabtay, O.; Breitbart, H. CaMKII prevents spontaneous acrosomal exocytosis in sperm through induction of actin polymerization. Dev Biol 2016, 415, 64–74. [Google Scholar] [CrossRef]
- Cervantes, M.P.; Palomino, J.M.; Adams, G.P. In vivo imaging in the rabbit as a model for the study of ovulation-inducing factors. Lab Anim 2015, 49, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gunnet, J.W.; Freeman, M.E. The mating-induced release of prolactin: a unique neuroendocrine response. Endocr Rev 1983, 4, 44–61. [Google Scholar] [CrossRef] [PubMed]
- Shafik, A.; El Sebai, O.; Shafik, A.A.; Shafik, I. Oviduct contractile response to vaginal distension: identification of vagino-tubal reflex. Arch Gynecol Obstet 2005, 271, 148–151. [Google Scholar] [CrossRef]
- Blandau, R.J.; Money, W.L. Observations on the rate of transport of spermatozoa in the female genital tract of the rat The Anatomical Record 1944, 90, 255-260. [CrossRef]
- Kovarik, A.; Hlubinova, K.; Vrbenska, A.; Prachar, J. An improved colloidal silver staining method of protein blots on nitrocellulose membranes. Folia Biol (Praha) 1987, 33, 253–257. [Google Scholar] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
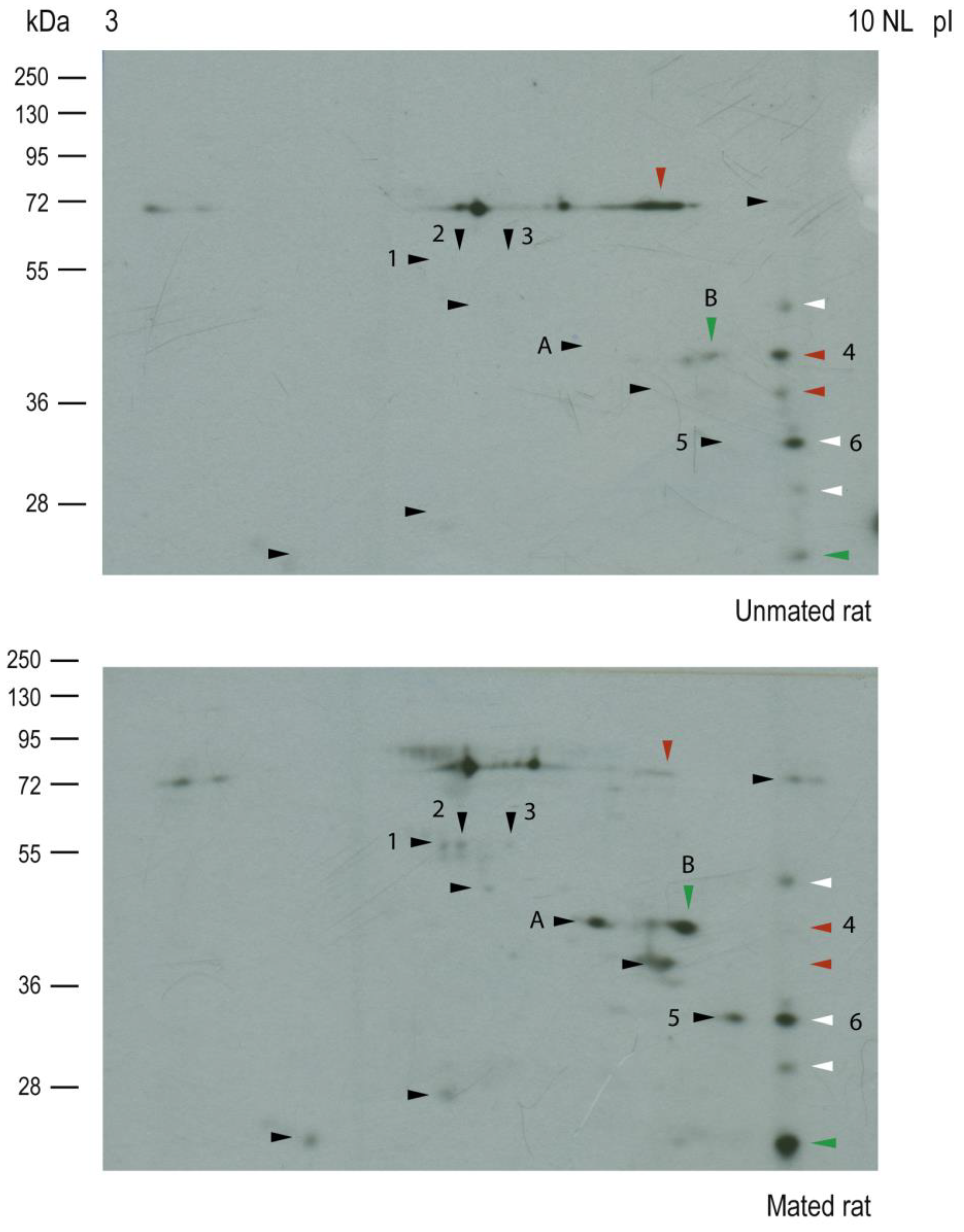
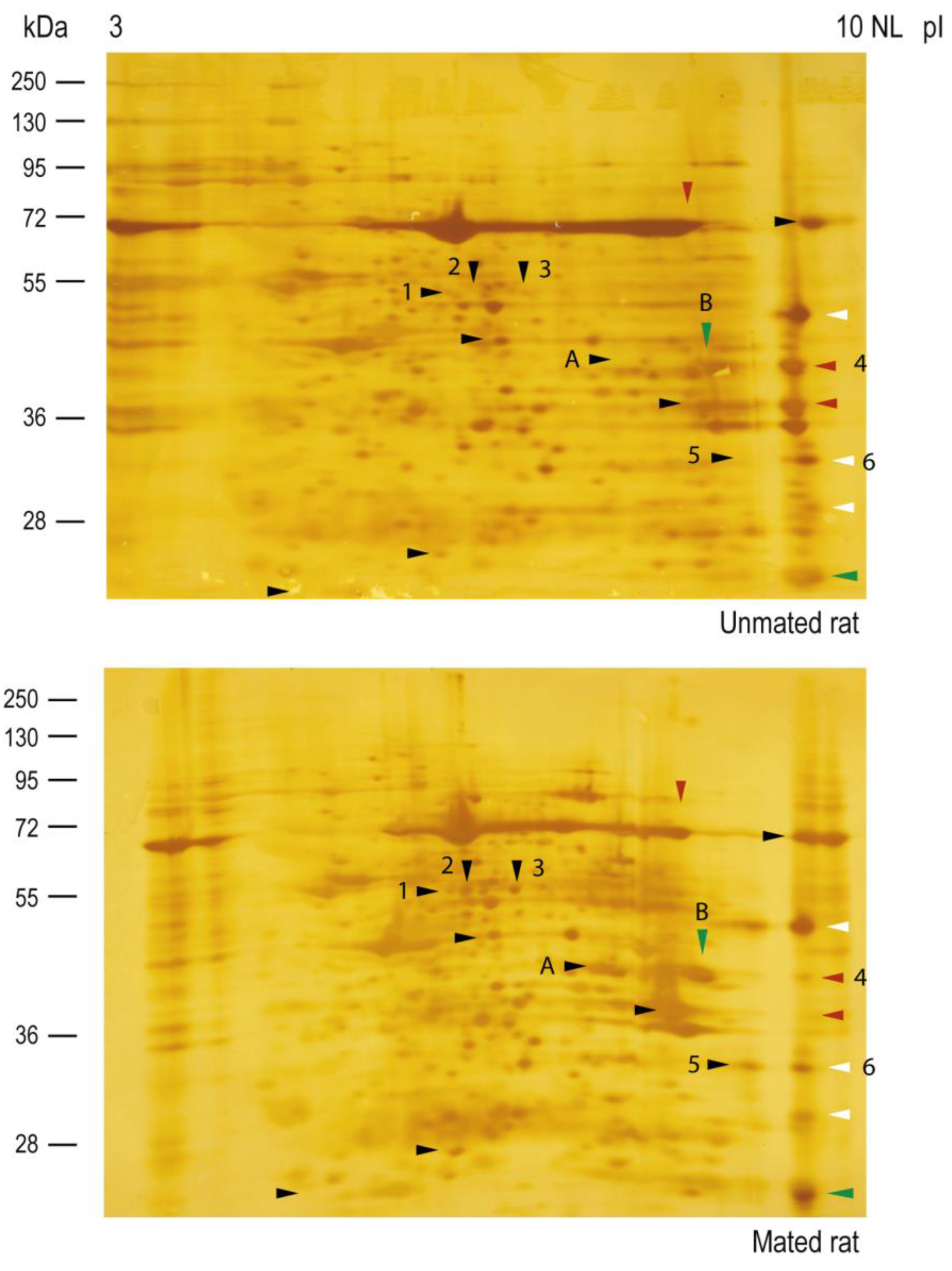
| Spot number | Protein | Gene | Sequence coverage (%) | Molecular mass (kDa) |
| 2 | Aldehyde dehydrogenase 9 family, member A1 | Aldh9a1 | 37.2 | 54.1 |
| 3 | Aldehyde dehydrogenase 9 family, member A1 | Aldh9a1 | 45.3 | 54.1 |
| 4 | Fructose bisphosphate aldolase A | Aldoa | 42.0 | 39.4 |
| 5 | Four and a half LIM domains protein 1 | Fhl1 | 59.3 | 31.9 |
| 6 | Four and a half LIM domains protein 1 | Fhl1 | 68.2 | 31.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




