2. Materials and methods
The present study was approved by the Ethics Committee of the University of Medicine and Pharmacy of Craiova, Romania (approval reference no. 7/20.01.2021).
The study was conducted on Wistar rats, which were given prenatally drugs possibly involved in the production of DDE.
In order to achieve the proposed objectives, 5 batches of 3 adult female Wistar rats were constituted.
The first group was represented by rats in the control group, which were not given drugs. The second group was represented by rats that received 50 mg/kg amoxicillin (standard dose recommended for children). The third group was represented by rats given 100 mg/kg amoxicillin (double dose compared to the standard dose recommended for children). The fourth group was represented by rats that received 8 mg/kg ibuprofen (standard dose recommended for children). The fifth group was represented by rats that were administered 20 mg/kg cefaclor (standard dose recommended for children).
To create the groups, 15 nulliparous females weighing approximately 200 grams were selected and each of them was placed in a cage with a male of the same breed. Every 24 hours, the vagina of the females was inspected by the veterinary technician, to see if mating was achieved. After mating, that day was considered the first day of gestation. The gestational state was also confirmed by weight gain.
On the sixth day of gestation, the control group was separated and drug administration was started for the other 4 groups.
Figure 1.
The groups of rats to which drugs were administered.
Figure 1.
The groups of rats to which drugs were administered.
The cages were ventilated, with a 12-hour light/dark cycle, and were maintained at a temperature of 25±1°C. The rats were fed with standard food - granulated combined fodder and water "ad libitum". For each rat, it was kept a record of the experiment to which it was subjected.
The drugs were administered in a daily dose, orally, with a metal esophageal catheter, until the end of gestation. The animals were weighed daily to calculate the required dose. At the end of the gestation process, the date of birth was recorded for all pups. Pups were kept with their mothers until weaning.
To make up the final batches, 15 pups that had the highest weight from each batch were chosen. At the age of two months, rat pups were sacrificed according to current standards, after administration of an anesthetic overdose of 100 mg/ml Ketamidor 20 IU (0.2 ml) and 0,3 ml Xilazyn Bio 2%. The injection was performed intraperitoneally, slightly to the right of the abdominal white line.
All the phases related to the fertilization of the female rats, the administration of the drugs, the selection of the pups that were sacrificed, the collection of the samples that were examined macroscopically, by optical microscopy and by polarized light microscopy, were carried out within the UMF Craiova Biobase.
After slaughter, the maxilla and mandible of each pup were placed in 10% formalin for 48 hours [
11]. After 48 hours, professional brushing and macroscopic examination of all dental arches (all maxillary and mandibular teeth of the 75 pups) were performed using the consultation kit, air spray, light source from the unit and a magnifying glass with a diameter of 90 mm.
After macroscopic examination and identification of enamel defects, optical and polarized light microscopy examination was performed using a Leica DM2500 microscope. The objective used was 4X, and for obtaining detailed images, 10X. The device used the configuration for reflection analysis of the samples, given that they do not allow transmission analysis due to opacity.
Until the start of the microscopic study, the jaws were preserved in 10% formalin to prevent their denaturation. Before imaging, the samples were left in open air for several minutes to allow the liquid to evaporate from the examined surfaces.
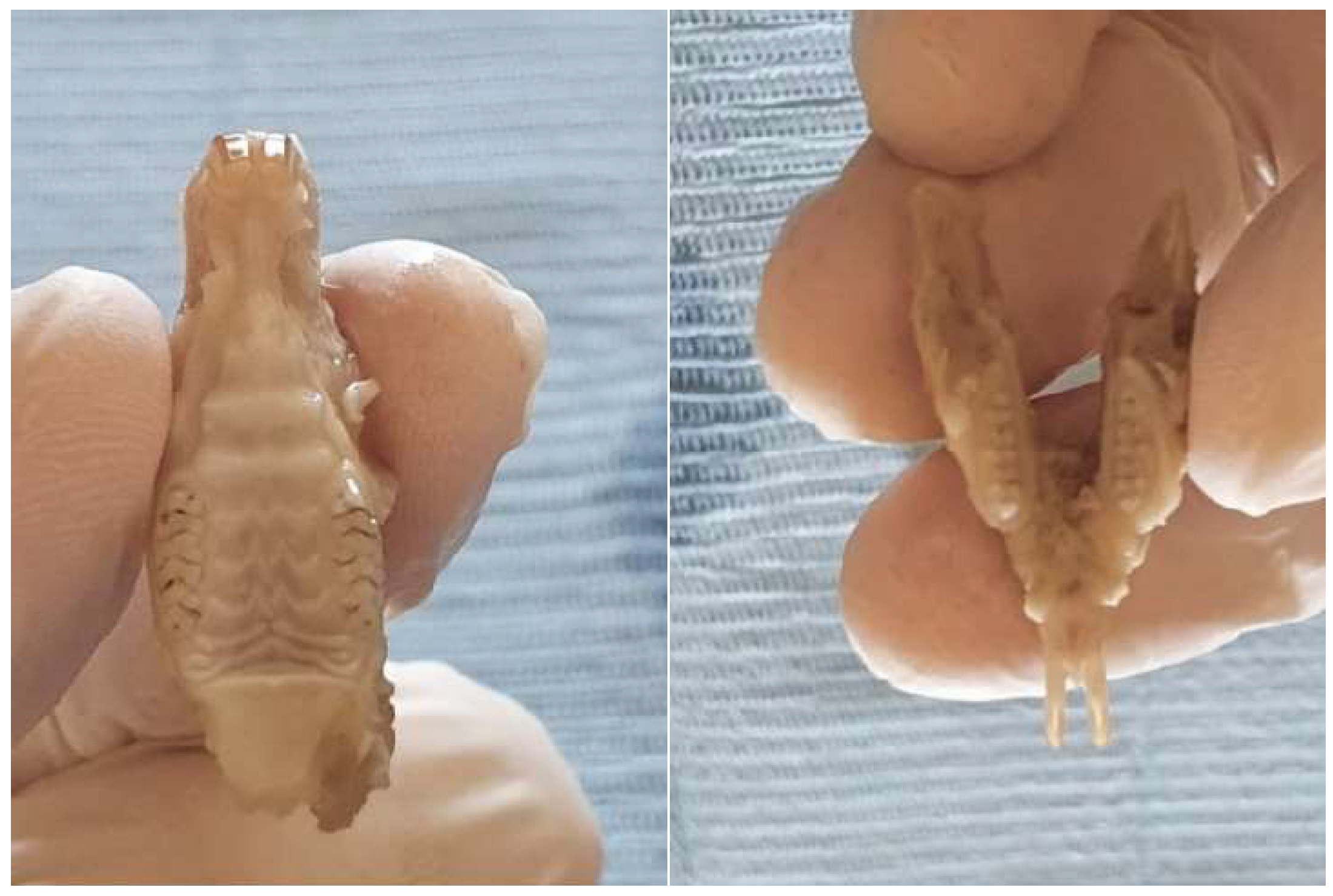
Wiping the samples with different tissues or paper was avoided, so as not to bring new particles to the tooth surfaces. For microscopic examination, the jaws were placed horizontally on the rotating microscope stand, as can be seen in figure 4.
Figure 2.
Examination of rat jaws.
Figure 2.
Examination of rat jaws.
Figure 3.
Leica DM2500 microscope.
Figure 3.
Leica DM2500 microscope.
Figure 4.
Image of the mandible under the Leica DM2500 microscope.
Figure 4.
Image of the mandible under the Leica DM2500 microscope.
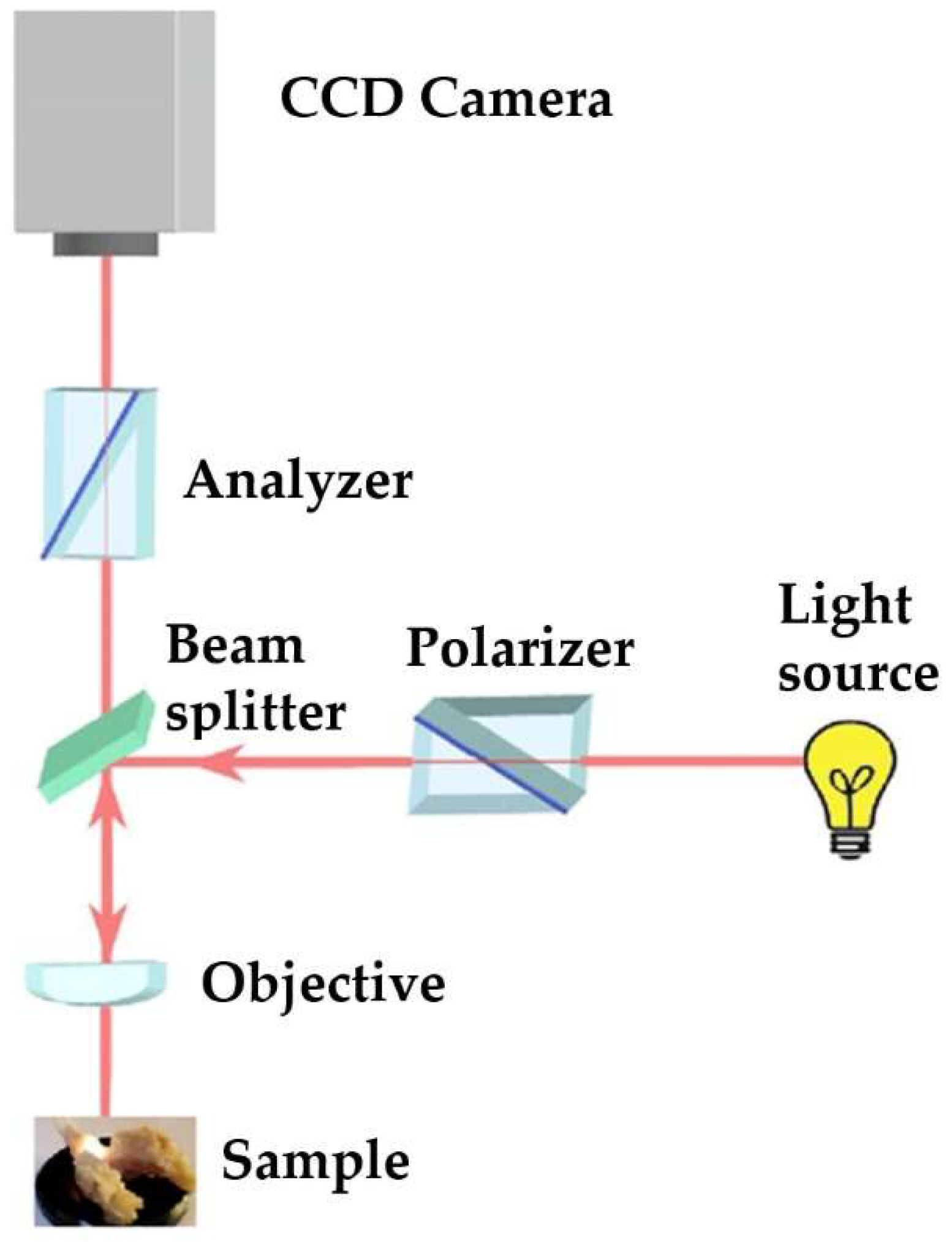
Figure 5.
Optical scheme of polarized light microscopy.
Figure 5.
Optical scheme of polarized light microscopy.
3. Results
Wistar rats have 2 central incisors and 6 molars in each jaw, for a total of 8 teeth. Following the macroscopic examination of the dental arches, no variations of the normal anatomical features were identified: no dental agenesis, abnormalities of position or size of the teeth were identified. Macroscopically, opacities present on the tooth surfaces (demineralization) and enamel discontinuities with a punctiform or linear appearance (hypoplasia) were considered DDE.
Examination by optical microscopy did not allow the highlighting of all structural defects, while polarized light microscopy revealed a greater number of enamel defects. The data to be presented represent the number of DDEs recorded by polarized light microscopy (
Table 1).
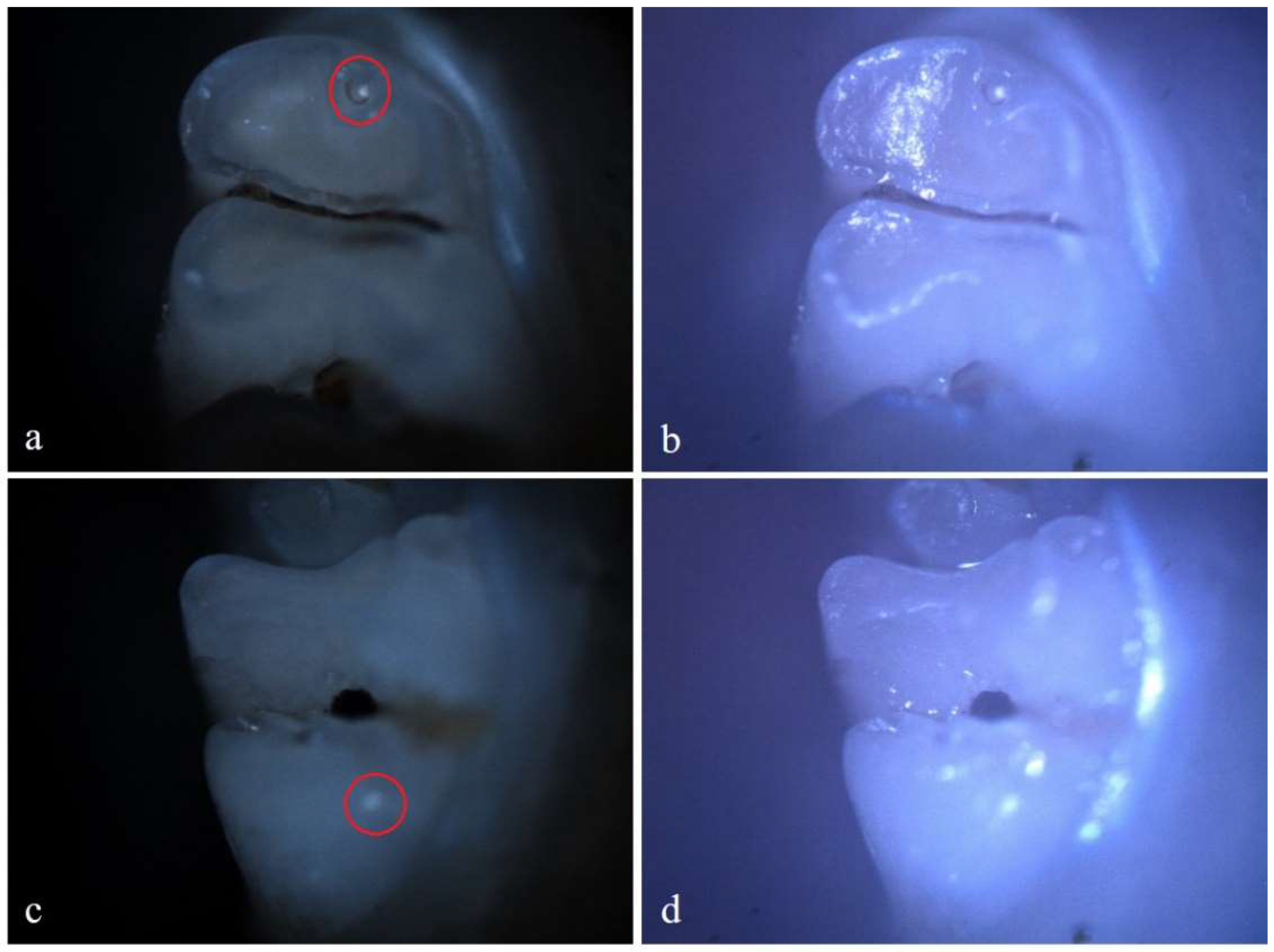
From the control group, 5 rats (33.33%) showed reduced demineralization in extent. Of these, 3 rats showed demineralization in the incisors and 2 rats showed demineralization in the molars. No rats from the control group showed hypoplasia. Within this lot, out of a total number of 240 teeth, 10 teeth (4.17%), 4 incisors and 6 molars showed demineralization. For each of the 10 affected teeth, the lesion was localized on a single surface resulting in a total of 10 tooth surfaces with DDE.
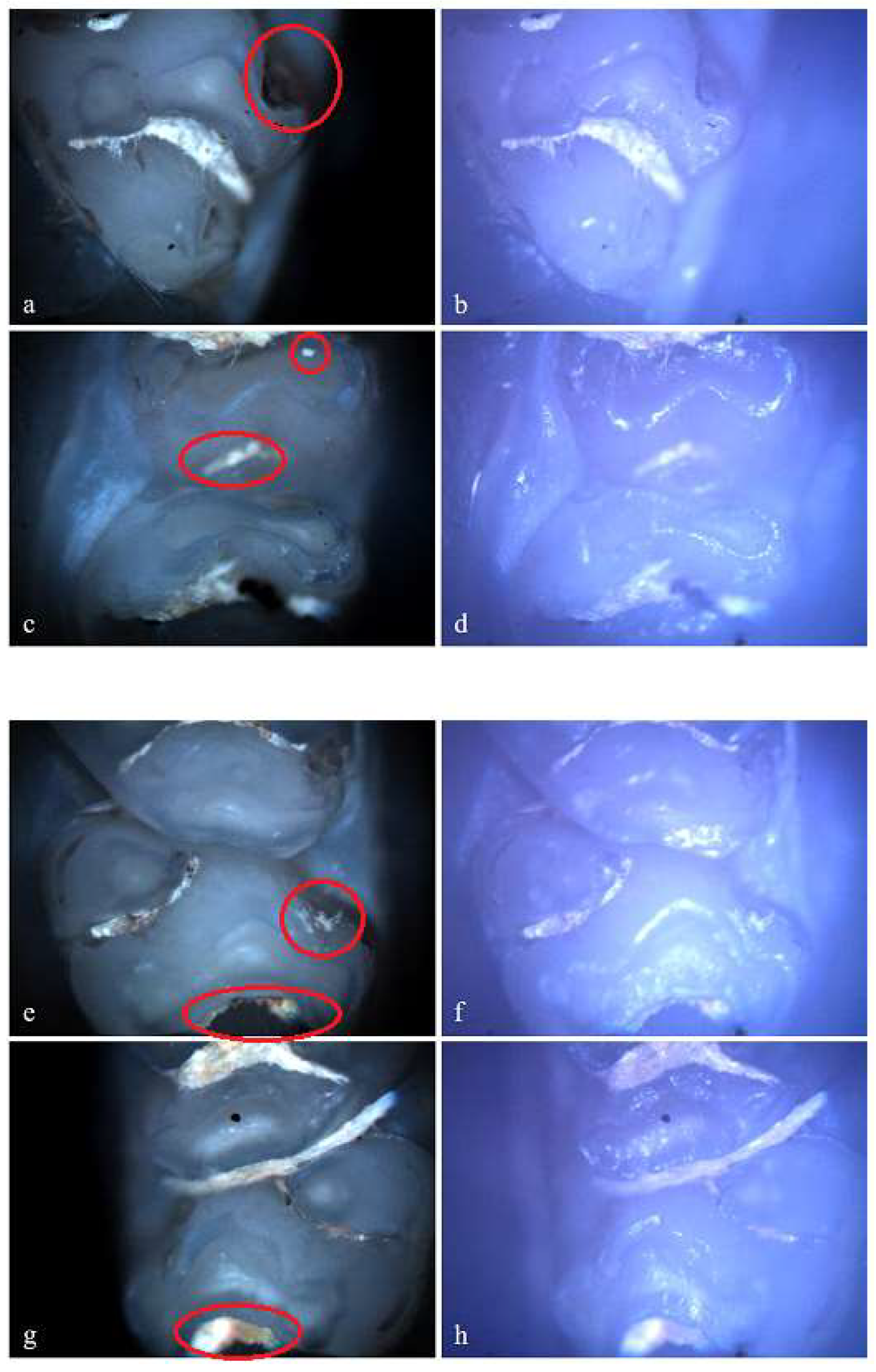
Figure 6.
Demineralization on the occlusal surface of 2 molars (a, c - Polarized microscopy image, 4X objective; b, d - Optical microscopy image, 4X objective). .
Figure 6.
Demineralization on the occlusal surface of 2 molars (a, c - Polarized microscopy image, 4X objective; b, d - Optical microscopy image, 4X objective). .
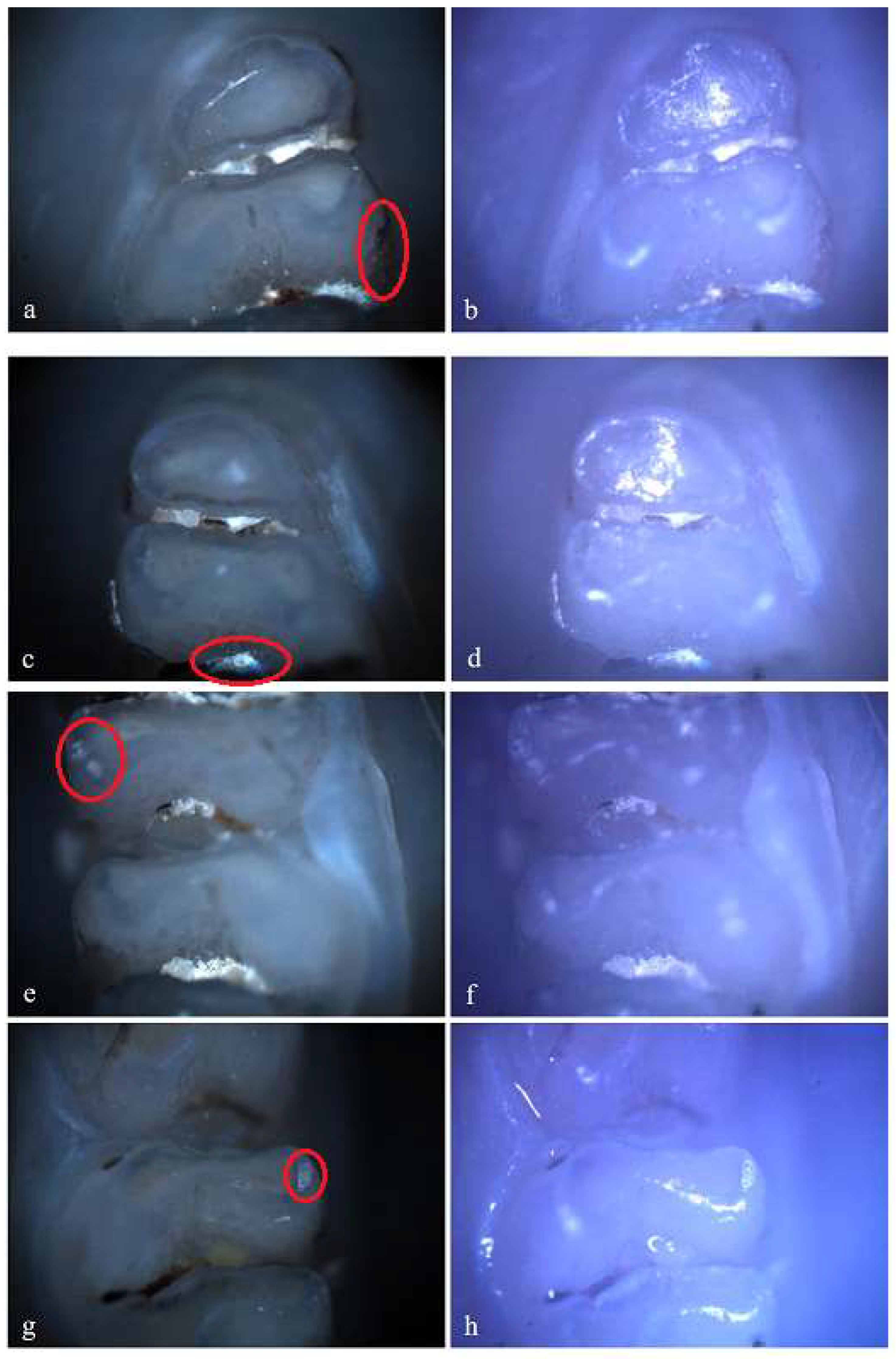
In the standard dose amoxicillin group (50 mg/kg), 10 rats (66.67%) showed at least one clinical form of DDE. Of these, 6 rats showed hypoplasia and 3 rats showed demineralization. One rat showed both clinical forms of DDE. Within this group, out of a total number of 240 teeth, 25 teeth (10.42%) showed at least one clinical form of DDE. Among them, 8 teeth (32%) were incisors and 17 teeth (68%) were molars, and as a clinical form of DDE, 16 teeth (64%) showed hypoplasia (5 incisors and 11 molars) and 9 teeth (36%) showed demineralization (3 incisors and 6 molars). Among the 25 teeth with DDE, 12 teeth (48%) presented lesions that were located on 2 surfaces, thus resulting in a total of 37 tooth surfaces with DDE.
Figure 7.
Hypoplasia on the palatal surface of a molar (a - Polarized microscopy image, 4X objective; b - Optical microscopy image, 4X objective) and on the distal surface of a molar (c - Polarized microscopy image, 4X objective; d - Optical microscopy image, 4X objective); Demineralization on the occlusal surface of 2 molars (e, g - Polarized microscopy image, 4X objective; f, h - Optical microscopy image, 4X objective). .
Figure 7.
Hypoplasia on the palatal surface of a molar (a - Polarized microscopy image, 4X objective; b - Optical microscopy image, 4X objective) and on the distal surface of a molar (c - Polarized microscopy image, 4X objective; d - Optical microscopy image, 4X objective); Demineralization on the occlusal surface of 2 molars (e, g - Polarized microscopy image, 4X objective; f, h - Optical microscopy image, 4X objective). .
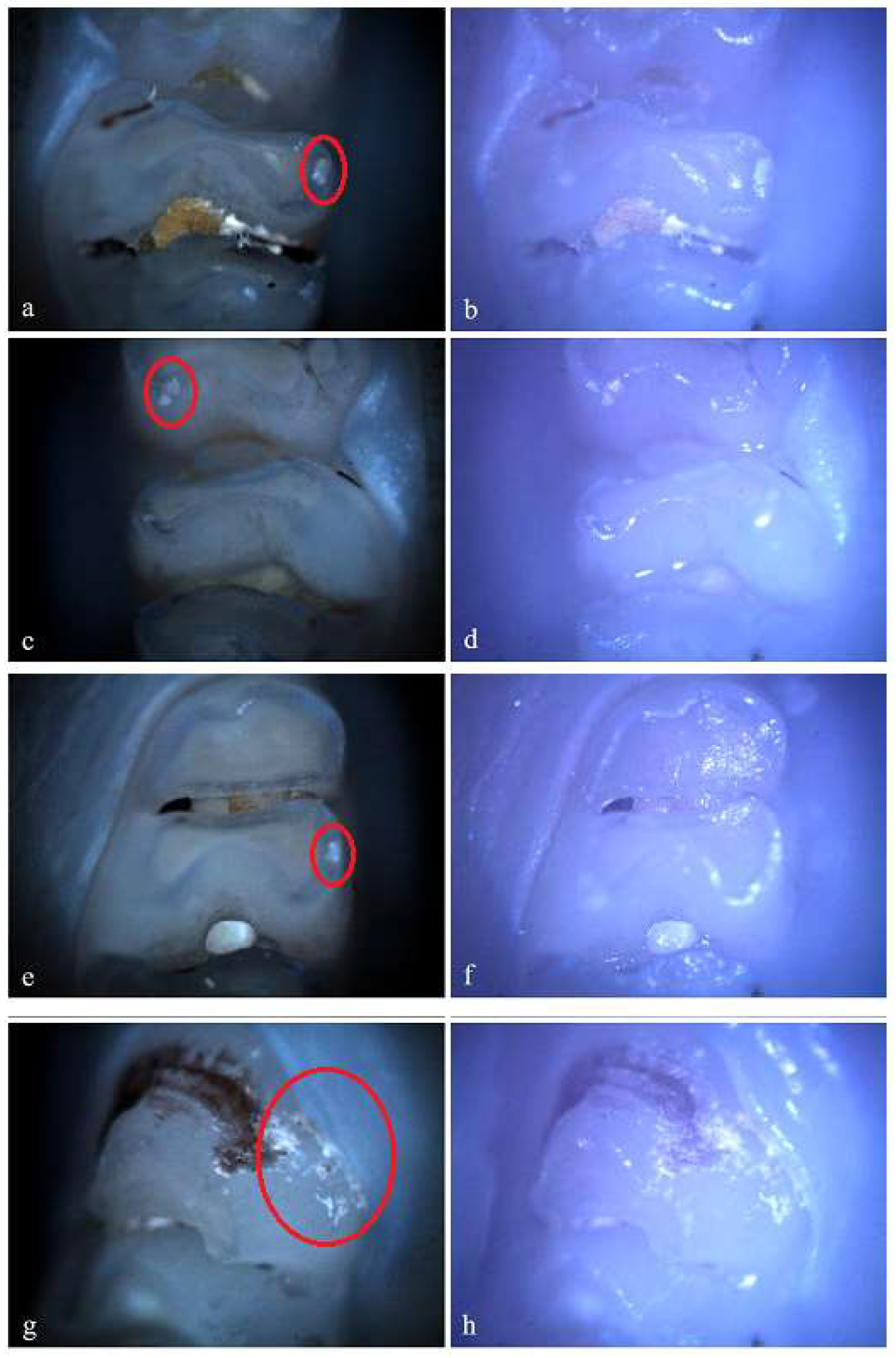
In the group receiving double-dose amoxicillin (100 mg/kg), 13 rats (86.67%) showed at least one clinical form of DDE. Of these, 7 rats were affected by hypoplasia and 5 rats were affected by demineralization. One rat showed both clinical forms of DDE. Within this group, out of a total number of 240 teeth, 43 teeth (17.92%) had at least one lesion. Among them, 13 teeth (30.23%) were incisors and 30 teeth (69.77%) were molars, and as a clinical form of DDE, 25 teeth (58.14%) showed hypoplasia (5 incisors and 20 molars) and 18 teeth (41.86%) showed demineralization (8 incisors and 10 molars). Among the 43 teeth with DDE, 3 teeth showed DDE on 2 surfaces, resulting in 46 affected tooth surfaces.
Figure 8.
Hypoplasia on the palatal surface of a molar (a - Polarized microscopy image, 4X objective; b - Optical microscopy image, 4X objective) and on the distal surface of some molars (e, g - Polarized microscopy image, 4X objective; f, h - Optical microscopy image, 4X objective); Demineralization on the occlusal surface of a molar (c - Polarized microscopy image, 4X objective; d - Optical microscopy image, 4X objective) and on the buccal surface of a molar (e - Polarized microscopy image, 4X objective; f - Optical microscopy image, 4X objective).
Figure 8.
Hypoplasia on the palatal surface of a molar (a - Polarized microscopy image, 4X objective; b - Optical microscopy image, 4X objective) and on the distal surface of some molars (e, g - Polarized microscopy image, 4X objective; f, h - Optical microscopy image, 4X objective); Demineralization on the occlusal surface of a molar (c - Polarized microscopy image, 4X objective; d - Optical microscopy image, 4X objective) and on the buccal surface of a molar (e - Polarized microscopy image, 4X objective; f - Optical microscopy image, 4X objective).
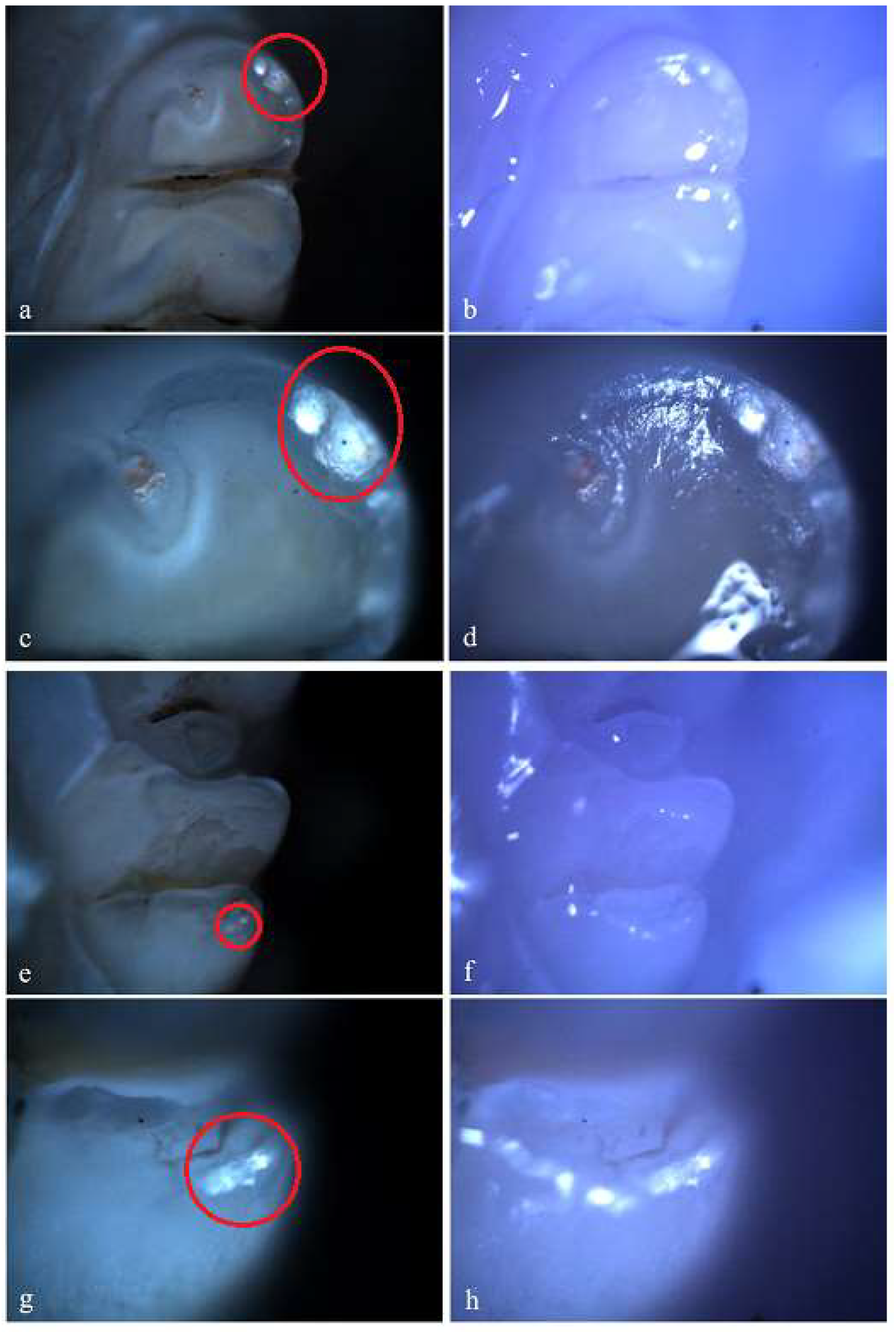
In the standard dose ibuprofen group (8 mg/kg), 11 rats (73.33%) showed at least one clinical form of DDE. Of these, 2 rats showed hypoplasia and 5 rats showed demineralization. Also, 3 rats showed both clinical forms of DDE. Within this group, from a total number of 240 teeth, 33 teeth (13.75%) presented at least one clinical form of DDE, respectively 15 incisors (45.45%) and 18 molars (54.55%), and according to the clinical form of DDE, 7 teeth (21.21%) showed hypoplasia (molars) and 26 teeth (78.79%) showed demineralization (15 incisors and 11 molars). Each tooth with DDE had only one affected surface, resulting in a total of 33 affected tooth surfaces.
Figure 9.
Demineralization on the occlusal surface of some molars (a, c, e - Polarized microscopy image, 4X objective; b, d, f - Optical microscopy image, 4X objective) and on the mesial surface of a molar (g - Polarized microscopy image, 4X objective; h - Optical microscopy image, 4X objective).
Figure 9.
Demineralization on the occlusal surface of some molars (a, c, e - Polarized microscopy image, 4X objective; b, d, f - Optical microscopy image, 4X objective) and on the mesial surface of a molar (g - Polarized microscopy image, 4X objective; h - Optical microscopy image, 4X objective).
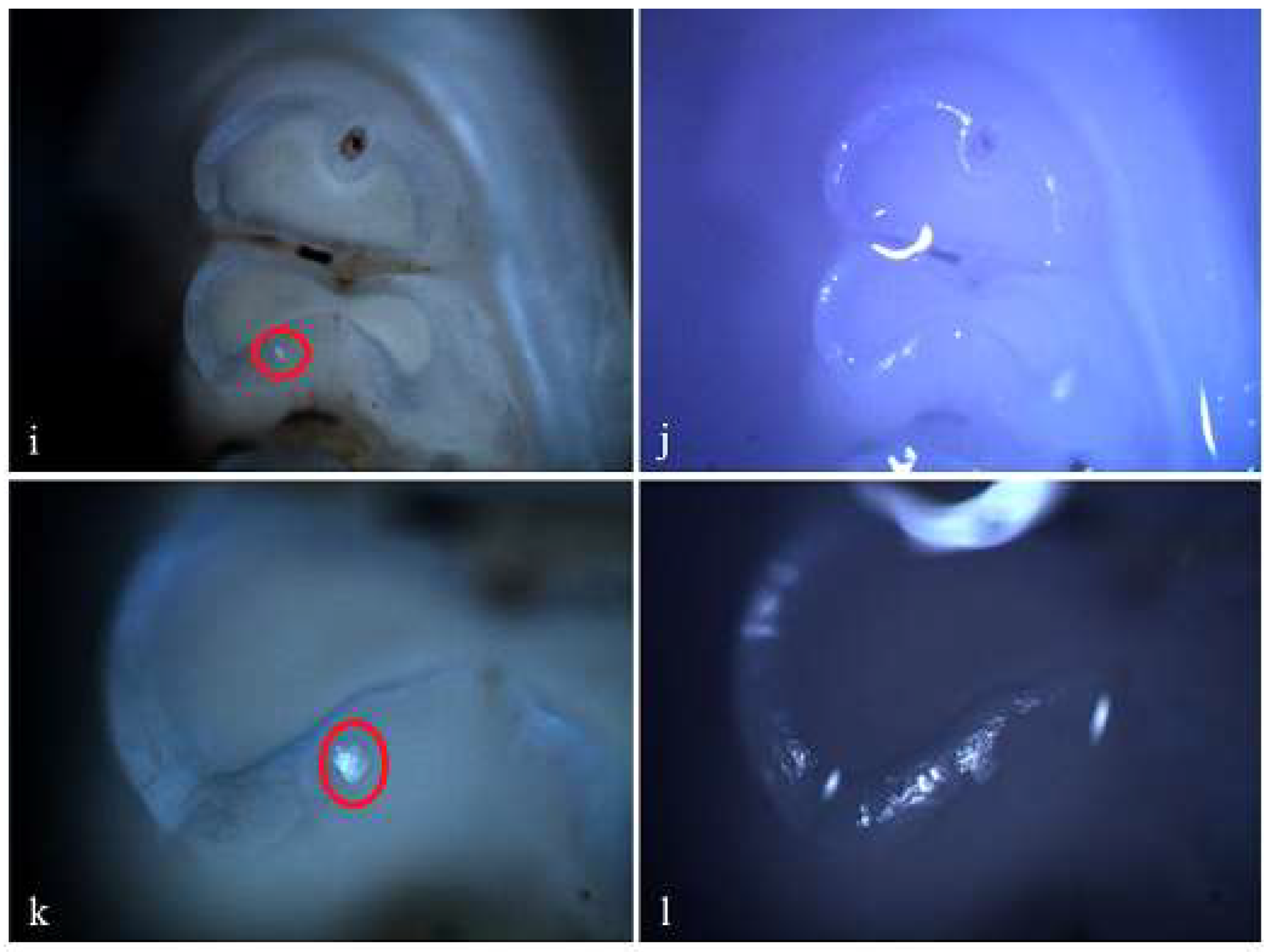
In the standard dose cefaclor group (20 mg/kg), 14 rats (93.33%) showed at least one clinical form of DDE. Of these, 5 rats showed demineralization and 7 rats showed both clinical forms of DDE. Within this group, from a total number of 240 teeth, 45 teeth (18.75%) presented at least one type of DDE, respectively 20 incisors (44.44%) and 25 molars (55.56 %), and as a clinical form of DDE, 15 teeth (33.33%) showed hypoplasia (2 incisors and 13 molars) and 30 teeth (66.67%) showed demineralization (18 incisors and 12 molars). Among the 45 affected teeth, 10 teeth showed DDE on 2 surfaces, resulting in 55 affected tooth surfaces.
Figure 10.
Demineralization on the occlusal surface of some molars (a, e, i - Polarized microscopy image, 4X objective; c, g, k - Polarized microscopy image, 10X objective; b, f, j - Optical microscopy image, 4X objective; d, h, l - Optical microscopy image, 10X objective).
Figure 10.
Demineralization on the occlusal surface of some molars (a, e, i - Polarized microscopy image, 4X objective; c, g, k - Polarized microscopy image, 10X objective; b, f, j - Optical microscopy image, 4X objective; d, h, l - Optical microscopy image, 10X objective).
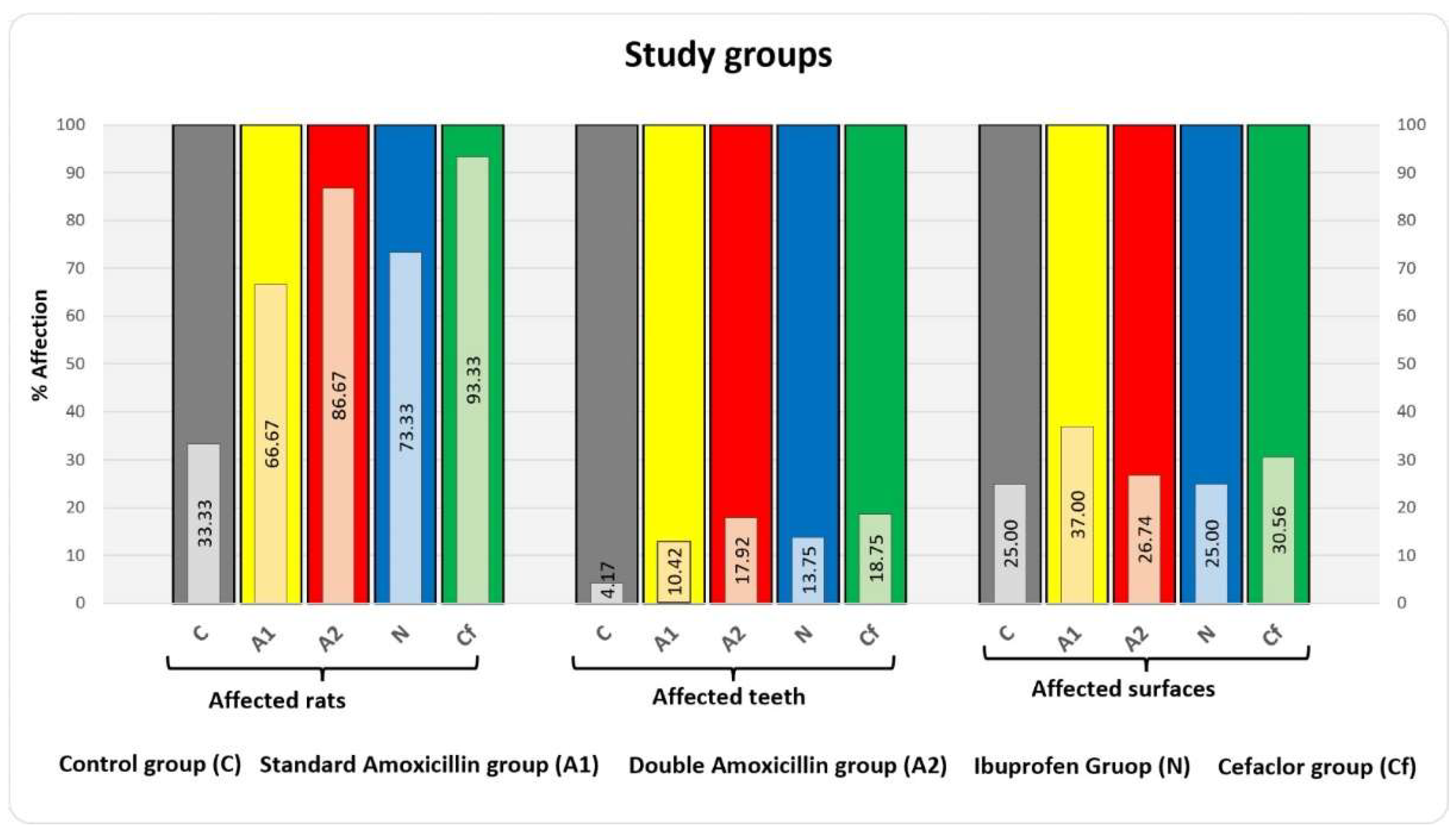
Figure 11.
Centralizing graph of the 5 study groups.
Figure 11.
Centralizing graph of the 5 study groups.
Centralizing these data, it resulted that the group to which cefaclor was administered was the most affected (14 rats, respectively 45 teeth with 55 affected surfaces), followed by the group to which amoxicillin was administered in a double dose (13 rats, respectively 43 teeth with 46 affected surfaces), the group to which ibuprofen was administered (11 rats, respectively 33 teeth with 33 affected surfaces), the group to which amoxicillin was administered in a standard dose (10 rats, respectively 25 teeth with 37 affected surfaces) and the control group (5 rats, respectively 10 teeth with 10 affected surfaces).
We make it clear that in the case of administration of cefaclor and amoxicillin in double dose and in standard dose, there were also teeth with more than one affected surface.
4. Discussion
The present study is a follow-up of the statistical study we previously carried out and represents, in fact, an "in vivo" validation of the results obtained in the etiological study [
10].
The choice of the drugs that were administered to reproduce in the laboratory the conditions that can generate DDE during the period of odontogenesis was made according to the results of the previously performed etiological study.
Similar studies in rats have also been conducted, but generally fewer drugs or smaller number of rats were used. Thus, Kameli used in 2019 standard and double-dose amoxicillin and standard-dose tetracycline, Serna Muñoz used in 2018 amoxicillin, amoxicillin with clavulanic acid, erythromycin, paracetamol, ibuprofen and celecoxib, de Souza used in 2015 standard and 5 times higher dose amoxicilin, Gottberg used in 2014 standard and double-dose amoxicillin and standard-dose tetracycline and Sahlberg used in 2013 amoxicillin [
11,
12,
13,
14,
15].
The identification of DDE was carried out in the present study by macroscopic examination, optical and polarized light microscopy examination of the teeth surface.
Polarized light microscopy offers a better contrast and quality of the images compared to conventional microscopy and at the same time knowledge about the optically active properties (change the polarization direction of the polarized light) of the analyzed sample. Optically active substances are different proteins, lipids or carbohydrates, which contain an asymmetric carbon in the molecule. The polarized light that is reflected on the surface of the dental samples has its polarization plane changed, if the respective area has optically active properties. A minor influence is also the inclination of the surfaces in relation to the normal incidence of the polarized light beam and its polarization direction.
The configuration used for the analysis of the jaws was that of crossed polarizers ("dark field"), thus ensuring a higher resolution regarding the optically active areas of the different dental samples. Therefore, the light linearly polarized by the polarizer (polarizing filter) and sent to the sample through the microscopic objective, following the reflection on the surface of the dental samples, undergoes a change in the polarization direction. Thus, in case of crossed polarizers, optically active areas will appear bright, and those that do not present such properties will appear dark.
Compared to optical microscopy, polarized light microscopy allows easier identification of DDE, as well as their delimitation.
Gottberg used optical microscopy to identify DDE [
11].
In the present study, by macroscopic examination of the teeth, no variations of the normal anatomical features were identified. Similar results were reported by Gottberg, in a similar study [
11].
The results obtained in the laboratory, regarding the effects of drugs administered during odontogenesis, were much more conclusive than in the etiological study, thus demineralization and even hypoplasia were identified in a greater proportion.
DDE present in the control group (in a reduced proportion) should be noted, suggesting a number of other factors involved in the development of DDE.
Administration of standard and double dose amoxicillin to pregnant female rats determined the presence of hypoplasia and demineralization in their offspring.
Amoxicillin is a semi-synthetic penicillin of the Beta-lactam family, with a broad spectrum, with bactericidal activity against gram-positive and gram-negative bacteria [
11]. Amoxicillin is prescribed as the antibiotic of choice for the treatment of respiratory, gastrointestinal, genital, skin and neurological infections [
16]. In children, it is the most commonly prescribed antibiotic [
17]. In the dental field, it is administered against microorganisms associated with dental infections, being at the same time the antibiotic of choice in the prophylaxis of bacterial endocarditis [
18,
19].
The most common side effects of amoxicillin are hypersensitivity reactions and gastrointestinal disorders [
16].
Although in the specialized literature amoxicillin is frequently associated with demineralization, in the present study, the groups to which this antibiotic was administered developed a higher percentage of hypoplasia. In a review on the etiology of DDE in temporary teeth, amoxicillin was mentioned as a risk factor for hypoplasia [
20].
A series of studies have proven that amoxicillin affects the thickness of the ameloblastic layer and implicitly the thickness of the enamel, hypoplasia being quantitative defects [
12,
14,
15].
De Souza observed vacuole-like structures in the ameloblastic layer of amoxicillin-treated rats and concluded that these structures arose because of the antibiotic's interference with the transmitters environment, leading to reduced protein secretion and transmission [
14].
Reduction in enamel thickness was also reported by Sahlberg, who, in an "in vitro" study, exposed the erupted teeth of rats to amoxicillin. His explanation was that the antibiotic prevented ameloblastic differentiation [
15].
The results of the present study, regarding the demineralization that occurred as a result of amoxicillin administration, were also confirmed by other "in vitro" studies [
11,
21].
Following the histological examination, Gottberg identified demineralization in the enamel of all examined samples from the 100 mg/kg amoxicillin group. Only half of the examined samples from the 50 mg/kg amoxicillin group showed demineralization. These results coincided with the present study's results, which demonstrates that the drug's effects on teeth are directly proportional to the administered dose [
15].
Incorporation of amoxicillin into developing teeth is favored by the fat-solubility of the antibiotic (superior to other penicillins) to the large number of blood vessels found in the intermediate layer of the molars intercuspid regions and by the low affinity of this drug due to plasma proteins [
16].
The mechanism by which some antibiotics affect tooth enamel has been attributed to the fact that these drugs alter protein synthesis [
11]. Recently, Sahlberg demonstrated that amoxicillin decreases the level of the MMP 20 metalloproteinase, which has an important role in the degradation and removal of enamel proteins [
15]. Gottberg and Laisi concluded that the antibiotic alters the normal temporal sequence of tooth enamel development [
11,
21].
The widespread administration of amoxicillin to both pregnant women and children, in the absence of clear evidence of the antibiotic effect on the developing dentition, suggests the need to determine whether there is a connection between the antibiotic and DDE [
11].
Ibuprofen is a drug that belongs to the group of non-steroidal anti-inflammatory drugs, being used in many countries for the treatment of inflammation and conditions manifested by pain and fever [
22]. Ibuprofen is part of several medicines: Nurofen (tablets for adults, syrup for children), Brufen (tablets), Ibufen (tablets), Ibalgin (tablets). All these can be bought over the counter in pharmacies in Romania. The use of ibuprofen in pediatrics has been reviewed, with results reporting that the drug can be administered safely and is effective against acute pain and fever [
22]. In general, for infants and children up to the age of 12, the most used form of presentation of ibuprofen is Nurofen in the form of syrup. It has the effect of relieving fever and pain, being recommended in fever after vaccination or in various other conditions accompanied by fever and pain (pharyngitis, tonsillitis, tooth eruption accidents or complicated caries, inflammatory diseases of the ear, minor traumas). In dentistry, ibuprofen is used to reduce pain and inflammation, especially in teeth diagnosed with acute pulpitis or acute apical periodontitis [
23]. A number of studies have shown that long-term use presents a relatively small risk for hepato-renal or gastrointestinal adverse effects [
22].
In the specialized literature, there is only one study in which the involvement of ibuprofen in the development of DDE was investigated [
13]. The study was carried out on mice, which were administered several drugs, including ibuprofen, for 30 days from the 21st day of life. Following scanning electron microscopy and energy dispersive x-ray analysis, Serna Muñoz found that ibuprofen did not influence the level of calcium and phosphorus, having no role in the occurrence of demineralization [
13].
These results are in contradiction with the present study's results, in which 33 teeth with DDE were identified, most of them in the form of demineralization. However, in the present study, ibuprofen was administered prenatally at a daily dose of 8 mg/kg, while Serna Muñoz administered it postnatally at a dose of 2.5 mg/day. Also, the methods of drug administration were different, as were the teeth chosen for examination. Thus, Serna Muñoz administered ibuprofen in strawberry gelatin, while a metal esophageal catheter was used in the present study. Further, in the present study all teeth were examined, while Serna Muñoz examined only third molars.
Serna Muñoz explained the possible mechanism by which ibuprofen can affect amelogenesis, producing demineralization [
13]. According to her, NSAIDs exert their anti-inflammatory effect by inhibiting cyclooxygenase (COX) and prostaglandin synthesis. Cyclooxygenases are enzymes that catalyze the formation of prostaglandins from arachidonic acid, by mediating physiological processes, including inflammation. At least 2 different enzymes are involved in this mechanism: constitutive COX1 (cyclooxygenase 1) and inductive COX2 (cyclooxygenase 2), closer to inflammatory processes. COX2 expression is transiently increased in response to inflammatory stimuli. Induction of COX2 results in rapid augmentation of intracellular level of nitric oxide, cytokines, and intracellular calcium [
22]. It is possible that the mineralization phase of enamel requires inflammation mediators because ameloblasts require a large influx of ions during crystal formation. Serna Muñoz proved that COX2 is present in the enamel organ. Inhibition of COX2 activity could produce a reduction in prostacyclin, which could decrease the blood flow in the ameloblasts area. During the maturation phase of amelogenesis, the rapid diffusion of nutrients at the level of the ameloblastic layer is necessary, as well as the rapid incorporation of ions, for the correct development of crystals [
24]. Prostacyclin reduction is also likely to affect the buffering capacity of the local tissue involved in maintaining pH homeostasis at the level of the enamel mineralization matrix, essential for healthy enamel [
24]. Therefore, COX2 is involved in the maturation phase of the enamel organ, its inhibition resulting in the disruption of amelogenesis and the appearance of demineralization [
13].
Through immunohistochemistry techniques, Serna Muñoz highlighted the fact that ibuprofen produced an average decrease in the amount of immunoreactive COX2, considered insignificant. The author also reported that another nonsteroidal anti-inflammatory drug, celecoxib, did not reduce the amount of COX2, a contradictory result, since celecoxib is known as a selective inhibitor of COX2 [
13]. In 2008, Vardar-Sengul explained these mechanisms by the fact that celecoxib could reduce COX2 activity, not the expression or amount of the enzyme [
25]. Thus, it can be considered that the demineralization highlighted in the present study, in the group to which ibuprofen was administered, may be based on the inhibition of COX2 activity.
Cefaclor is an antibiotic that is part of the 2nd generation of the Cephalosporin family, being used more and more frequently in the medical practice to treat a wide variety of infections [
26]. Its widespread use is due to the fact that it has low toxicity and allergenic effects, being effective against a wide spectrum of conditions [
27]. Cefaclor has antibacterial activity against gram-positive and gram-negative aerobic microorganisms [
28]. Cefaclor is recommended in the treatment of pneumonia, acute bronchitis, acute sinusitis, pharyngitis, tonsillitis, otitis media, skin and soft tissue infections, urinary tract infections [
28]. Cefaclor is available as capsules for adults and oral suspension for children. In dentistry, the antibiotic is used systemically, as a treatment for abscesses generated by dental infections, and, in endodontics, being a component of the triple antibiotic paste, applied intracanally for disinfectant and pulp regeneration actions [
29]. Among the reported side effects are pruritus, rashes, indigestion and diarrhea [
28,
30].
According to the present study, among the drugs used, cefaclor induced the highest number of DDE, in the form of demineralization, most frequently, and hypoplasia.
Other studies have also associated the administration of cefaclor in children with the development of DDE [
8,
9,
31].
Tariq's 2014 study was based on the examination of children and the filling of a questionnaire by parents [
31]. Tapias conducted a retrospective study in 2001, in which he analyzed the association between various risk factors of DDE on the first permanent molar [
9]. Thus, he followed the medical history of 48 children during the first 5 years of life and found a statistically significant association between the administration of cefaclor at the age of 4 and DDE. In a case report, Simratvir reported in 2011 the existence of hypoplasia in all erupted and unerupted permanent teeth of a 9-year-old girl [
8]. One of the 6-year-old molars was also affected by demineralization. Following the patient's medical history, Simratvir concluded that the only factors possibly involved in the development of DDE were the frequent presence in the first 4 years of life of upper respiratory tract conditions and the medication used as treatment. The parents gave her cefaclor and paracetamol for almost 4 years because of the recurrence of the respiratory infection.
In the specialized literature, however, there are also studies that address the association between antibiotics and DDE, with opposite results compared to the previously presented studies. Thus, in a study carried out in 2009 on 147 children with an average age of 10.7 years, the association of several antibiotics, including cefaclor, with the development of demineralization was investigated. A direct relationship between the use of cefaclor and the presence of demineralization was not identified [
21].
To our knowledge, there are no explanations in the specialized literature of how cefaclor intervenes in odontogenesis and causes the development of DDE.
The differences between the results reported by the studies addressing this topic can be attributed to the different drugs and doses used, the duration and routes of administration, the different moment of pups slaughtering, as well as the teeth chosen for the study [
11].
The present study demonstrates the importance of re-evaluating the adverse effects of the administration of some drugs during pregnancy and in the first years of the child's life, and possibly limiting the prescription of these drugs to pregnant women and children, in cases where it is strictly necessary. Also, the replacement of these drugs with others that are not associated with DDE should be considered.
We consider it useful to educate pregnant women and mothers about the prevention methods of DDE and the risk that these drugs present for the development of DDE.
Although in the previous statistical study the drugs (amoxicillin, ibuprofen, cefaclor) that we highlighted as having a role in the development of DDE were administered postnatally, and in the present study they were administered prenatally, we believe that in both cases, they influenced odontogenesis [
10].
We found no rats studies in the specialty literature that used four drugs to produce DDE, simultaneously, under the same conditions.