Submitted:
15 January 2025
Posted:
16 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Leftwick, A.P. Parnell E.W., New pesticides derived from cyclohexadiene, OA5232A, 1981. https://patents.google.com/patent/OA5232A/en?oq=OA5232A.
- Takegawa, B. , Steroid Compound, JPH0267296A, 1988. https://patents.google.com/patent/JPH0267296A/en?oq=JPH0267296.
- Mukawa, F. , 10β-Chloro-17β-hydroxyestra-1,4-dien-3-one and its related compounds. J. Chem. Soc., Perkin Trans. 1988, 1, 457–460. [Google Scholar] [CrossRef]
- Kaiser, K.L.E. , Niculescu S. P., On the PNN modeling of estrogen receptor binding data for carboxylic acid esters and organochlorine compounds, Water Qual. Res. J. Canada. 2001, 36, 619–630. [Google Scholar] [CrossRef]
- Kato, T. , Ichinose I. , Selective bromination of polyenes by 2,4,4,6-tetrabromocyclohexa-2,5-dienone, J. Chem. Soc. Perkin Trans. 1980, 1, 1051–1056. [Google Scholar] [CrossRef]
- Guy, A.; Lemaire, M.; Guetté, J.P.; Halogenation regioselective en serie, a.r.o.m.a.t.i.q.u.e.-I.I. Chloration des naphtols et de leurs ethers a l’aide de reactifs mettant en jeu des interactions du type donneur-accepteur. Tetrahedron 1982, 38, 2347–2354. [Google Scholar] [CrossRef]
- Mamaghani, M. , Zolfigol M. A., Shojaei M., N-Chloro-2,3,4,4,5,6-hexachlorocyclohexa-2,5-dienylideneamine as a mild and highly regioselective chlorinating reagent. Synth. Commun. 2002, 32, 735–740. [Google Scholar] [CrossRef]
- Fischer, A. , Henderson G. N., Ipso chlorination of 4-alkylphenols. Formation of 4-alkyl-4-chlorocyclohexa-2,5-dienones. Can. J. Chem. 1979, 57, 552–557. [Google Scholar] [CrossRef]
- Bergquist, K.-E. , Nilsson A. , Ronlán A., Norin T., Hjeds H., Electrophilic Chlorination of 4-Methylphenols with Molecular Chlorine. Synthesis of Dimethoxy Aromatics by Methanolysis of 4-Chloro-4-methylcyclohexa-2,5-dienones. Acta Chem. Scand. 1982, 36b, 675–683. [Google Scholar] [CrossRef]
- Antinori, G.; Baciocchi, E.; Illuminati, G. ; Non-conventional paths in electrophilic aromatic reactions Part, V.I. Chlorination of 3,5-dichloro-2,4,6-trimethylanisole and related compounds. J. Chem. Soc. B Phys. Org. 1969, 373. [Google Scholar] [CrossRef]
- Benedikt, R. , Schmidt M. V., Notizen über Halogenderivate. Monatshefte Für Chemie. 1983, 4, 604–609. [Google Scholar] [CrossRef]
- Mukawa, F. , Suzuki T. , Ishibashi M., Yamada F., Estrogen and androgen receptor binding affinity of 10β-chloro-estrenen derivatives, J. Steroid Biochem. 1988, 31, 867–870. [Google Scholar] [CrossRef]
- Mukawa, F. The anomalous chlorination of estradiol 17β-acetate with isocyanuric chloride, Tetrahedron Lett. 1959, 1, 17–20. [Google Scholar] [CrossRef]
- Ferron, B. , Jacquesy JC., Jouannetaud M.P., Karam O., Coustard J.M., Ipso-chlorination of 4-alkylphenols ethers a novel route to 4-chlorocyclohexa-2,5-dienones., Tetrahedron Lett. 1993, 34, 2949–2952. [Google Scholar] [CrossRef]
- Uyanik, M. , Sasakura N, Kuwahata M., Ejima Y., Ishihara K., Practical oxidative dearomatization of phenols with sodium hypochlorite pentahydrate, Chem. Lett. 2015, 44, 381–383. [Google Scholar] [CrossRef]
- Zhang, Z. , Sun Q, Xu D., Xia C., Sun W., Direct halogenative dearomatization of 2-naphthols by NXS (X = Cl, Br) in water, Green Chem. 2016, 18, 5485–5492. [Google Scholar] [CrossRef]
- Yin, Q. , Wang SG., Liang X.W., Gao D.W., Zheng J., You S.L., Organocatalytic asymmetric chlorinative dearomatization of naphthols, Chem. Sci. 2015, 6, 4179–4183. [Google Scholar] [CrossRef] [PubMed]
- Uyanik, M. , Sahara N, Ishihara K., Regioselective Oxidative Chlorination of Arenols Using NaCl and Oxone, Euro. J. Org. Chem. 2019, 2019, 27–31. [Google Scholar] [CrossRef]
- Tilstam, U. , Weinmann H, Trichloroisocyanuric Acid: A Safe and Efficient Oxidant, Org. Process Res. Dev. 2002, 6, 384–393. [Google Scholar] [CrossRef]
- Gaspa, S. , Carraro M, Pisano L., Porcheddu A., De Luca L., Trichloroisocyanuric Acid: a Versatile and Efficient Chlorinating and Oxidizing Reagent, Euro. J. Org. Chem. 2019, 2019, 3544–3552. [Google Scholar] [CrossRef]
- Gambacorta, G. , Sharley JS., Baxendale I.R., A comprehensive review of flow chemistry techniques tailored to the flavours and fragrances industries, Beilstein J. Org. Chem. 2021, 17, 1181–1312. [Google Scholar] [CrossRef] [PubMed]
- Sagmeister, P. , Williams JD., Hone C.A., Kappe C.O., Laboratory of the future: A modular flow platform with multiple integrated PAT tools for multistep reactions, React. Chem. Eng. 2019, 4, 1571–1578. [Google Scholar] [CrossRef]
- Webb, D. , Jamison TF., Continuous flow multi-step organic synthesis, Chem. Sci. 2010, 1, 675–680. [Google Scholar] [CrossRef]
- Pastre, J.C. , Browne DL., Ley S.V., Flow chemistry syntheses of natural products, Chem. Soc. Rev. 2013, 42, 8849–8869. [Google Scholar] [CrossRef]
- Browne, D.L. , Deadman BJ., Ashe R., Baxendale I.R., Ley S.V., Continuous Flow Processing of Slurries: Evaluation of an Agitated Cell Reactor, Org. Process Res. Dev. 2011, 15, 693–697. [Google Scholar] [CrossRef]
- Deadman, B.J. , Browne DL., Baxendale I.R., Ley S.V., Back Pressure Regulation of Slurry-Forming Reactions in Continuous Flow, Chem. Eng. Technol. 2015, 38, 259–264. [Google Scholar] [CrossRef]
- Sharma, M.K. , Suru A., Joshi A., Kulkarni A.A., A Novel Flow Reactor for Handling Suspensions: Hydrodynamics and Performance Evaluation, Ind. Eng. Chem. Res. 2020, 59, 16462–16472. [Google Scholar] [CrossRef]
- Bianchi, P. , Williams JD., Kappe C.O., Oscillatory flow reactors for synthetic chemistry applications, J. Flow Chem. 2020, 10, 475–490. [Google Scholar] [CrossRef]
- Doyle, B.J. , Gutmann B, Bittel M., Hubler T., Macchi A., Roberge D.M., Handling of Solids and Flow Characterization in a Baffleless Oscillatory Flow Coil Reactor. , Ind. Eng. Chem. Res. 2020, 59, 4007–4019. [Google Scholar] [CrossRef]
- Hartman, R.L. , Naber JR., Zaborenko N., Buchwald S.L., Jensen K.F., Overcoming the Challenges of Solid Bridging and Constriction during Pd-Catalyzed C−N Bond Formation in Microreactors, Org. Process Res. Dev. 2010, 14, 1347–1357. [Google Scholar] [CrossRef]
- Schoenitz, M. , Grundemann L, Augustin W., Scholl S., Fouling in microstructured devices: A review, Chem. Commun. 2015, 51, 8213–8228. [Google Scholar] [CrossRef] [PubMed]
- Wu, K. , Kuhn S, Strategies for solids handling in microreactors, Chim. Oggi/Chemistry Today. 2014, 32, 62–66. [Google Scholar]
- Dong, Z. , Zhao S, Zhang Y., Yao C., Yuan Q., Chen G., Mixing and residence time distribution in ultrasonic microreactors, AIChE J. 2017, 63, 1404–1418. [Google Scholar] [CrossRef]
- Rivas D., F. , Kuhn S, Synergy of Microfluidics and Ultrasound: Process Intensification Challenges and Opportunities, Top. Curr. Chem. 2016, 374, 1–30. [Google Scholar] [CrossRef]
- Dong, Z. , Delacour C, Carogher K.M., Udepurkar A.P., Kuhn S., Continuous ultrasonic reactors: Design, mechanism and application, Materials 2020, 13, 344–1. [Google Scholar] [CrossRef]
- Husain, S. , Kifayatullah, M., Phenol-dienone rearrangement in the chlorination of p-cresol with tert-butyl hypochlorite: Indian. J. Chem., Section B: Org. Chem. Incl. Med. Chem. 1985, 24B, 711–714. [Google Scholar]
- Chip, G. K. , Grossert, J. S. Can. J. Chem. 1972, 50, 1233–1240. [Google Scholar] [CrossRef]
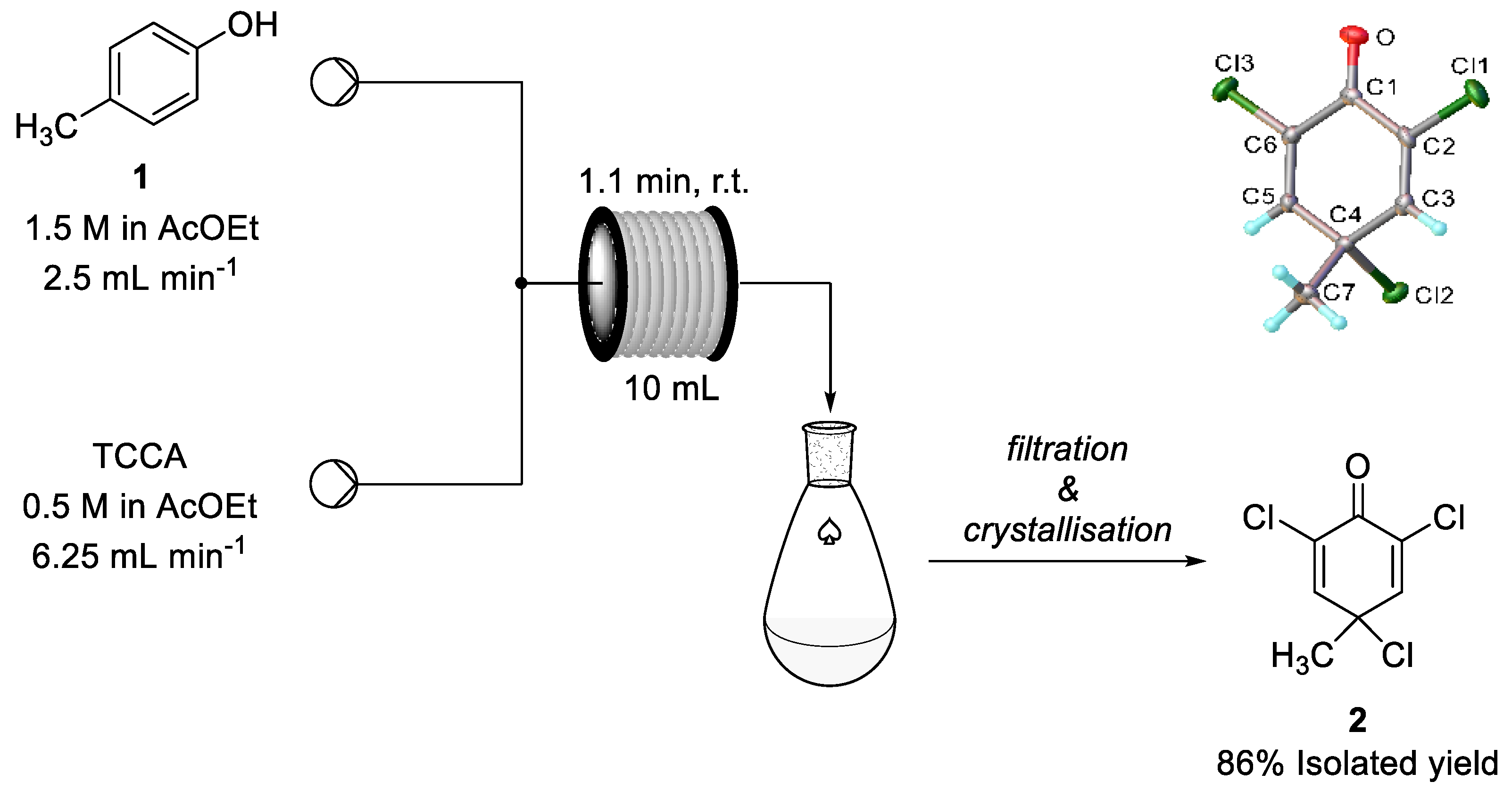

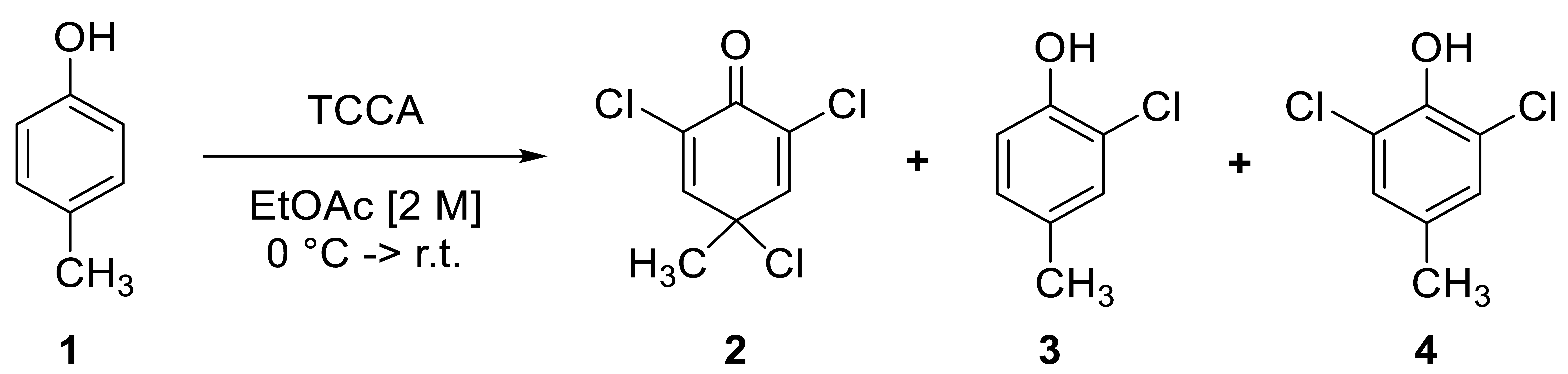 | |||||
|---|---|---|---|---|---|
| Entry1 | TCCA (equiv.) | Residence Time (min) | GC-MS yield (%)2 3 4 | ||
| 1 | 2 | 20 | 57.7 | 42.3 | - |
| 2 | 2 | 30 | 75.0 | 25.0 | - |
| 3 | 2 | 60 | 67.0 | 20.9 | 11.9 |
| 4 | 2.2 | 30 | 75.0 | 25.0 | - |
| 5 | 2.5 | 30 | 84.9 | 9.4 | 5.7 |
| 6 | 2.7 | 30 | 83.7 | 16.3 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




