Submitted:
08 January 2025
Posted:
14 January 2025
You are already at the latest version
Abstract
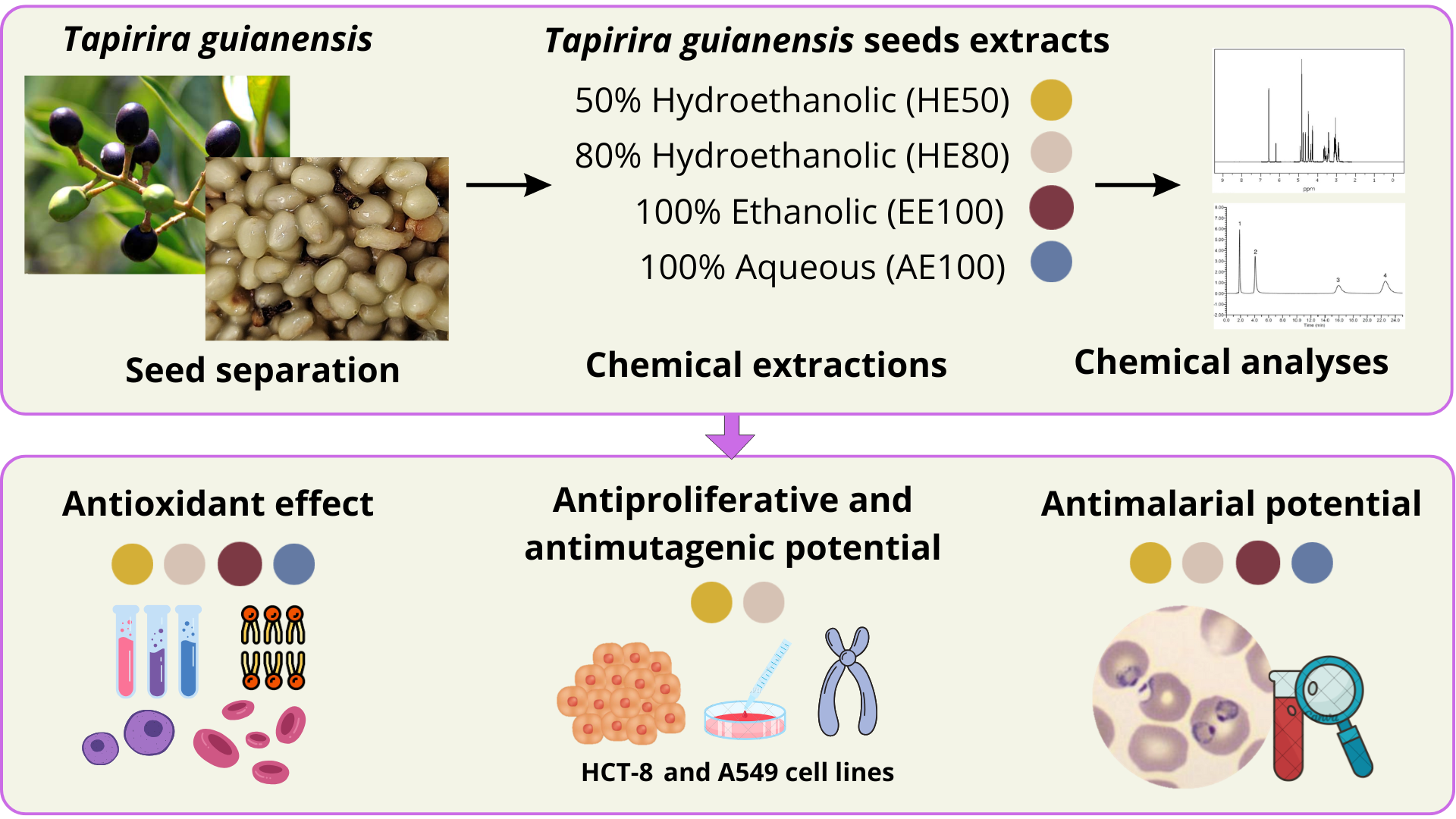
Keywords:
Introduction
2. Materials and Methods
2.1. Chemical Reagents and Cell Lines
2.2. Plant Source and Ultrasound-Assisted Solvent Extraction
2.3. Chemical Profile and Antioxidant Capacity
2.3.1. Phenolic Compounds
2.3.2. Ultra-Performance Liquid Chromatography Coupled with Quadrupole Time-of-Flight Mass Spectrometry (UPLC-QToF/MS) Analyses
2.3.3. Nuclear Magnetic Resonance (NMR) Analysis
2.3.4. Chemical Antioxidant Capacity
2.3.5. Protection Against Lipoperoxidation
2.4. Cell Culture: Cytotoxicity and Antioxidant Activity
2.4.1. Cell Viability Assessment
2.4.2. Intracellular Reactive Oxygen Species (ROS) Generation
2.4.3. Protection Against Chromosomal Aberration
2.4.4. Erythrocyte Cellular Antioxidant Activity and Protection
2.5. In Vitro Antimalarial Properties
2.6. Statistical Analysis
3. Results and Discussion
3.1. Chemical Profile
3.2. Chemical Antioxidant Capacity
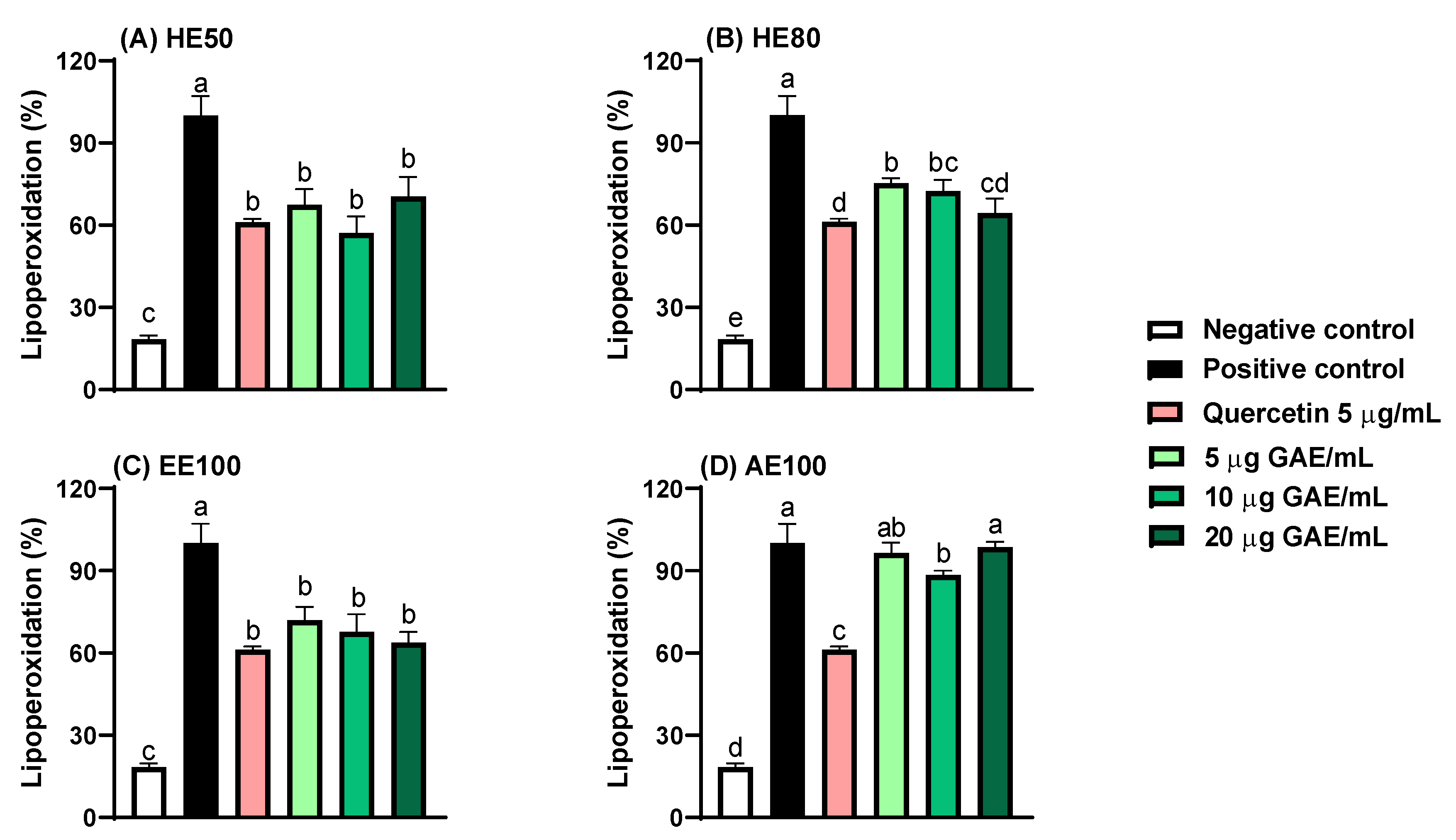
3.2. Protection Against Lipoperoxidation
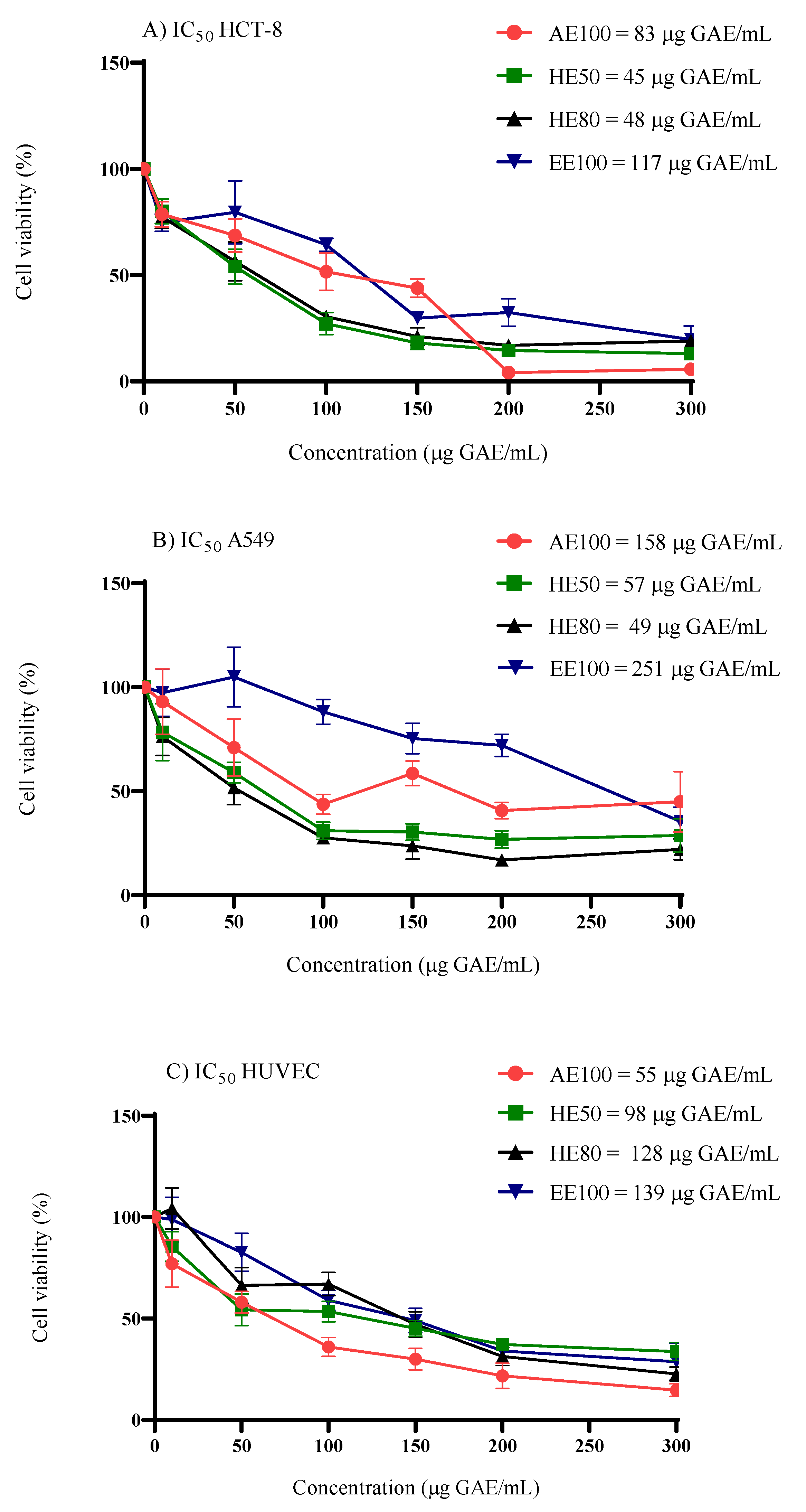
3.3. Cytotoxicity Activity in Cell Culture
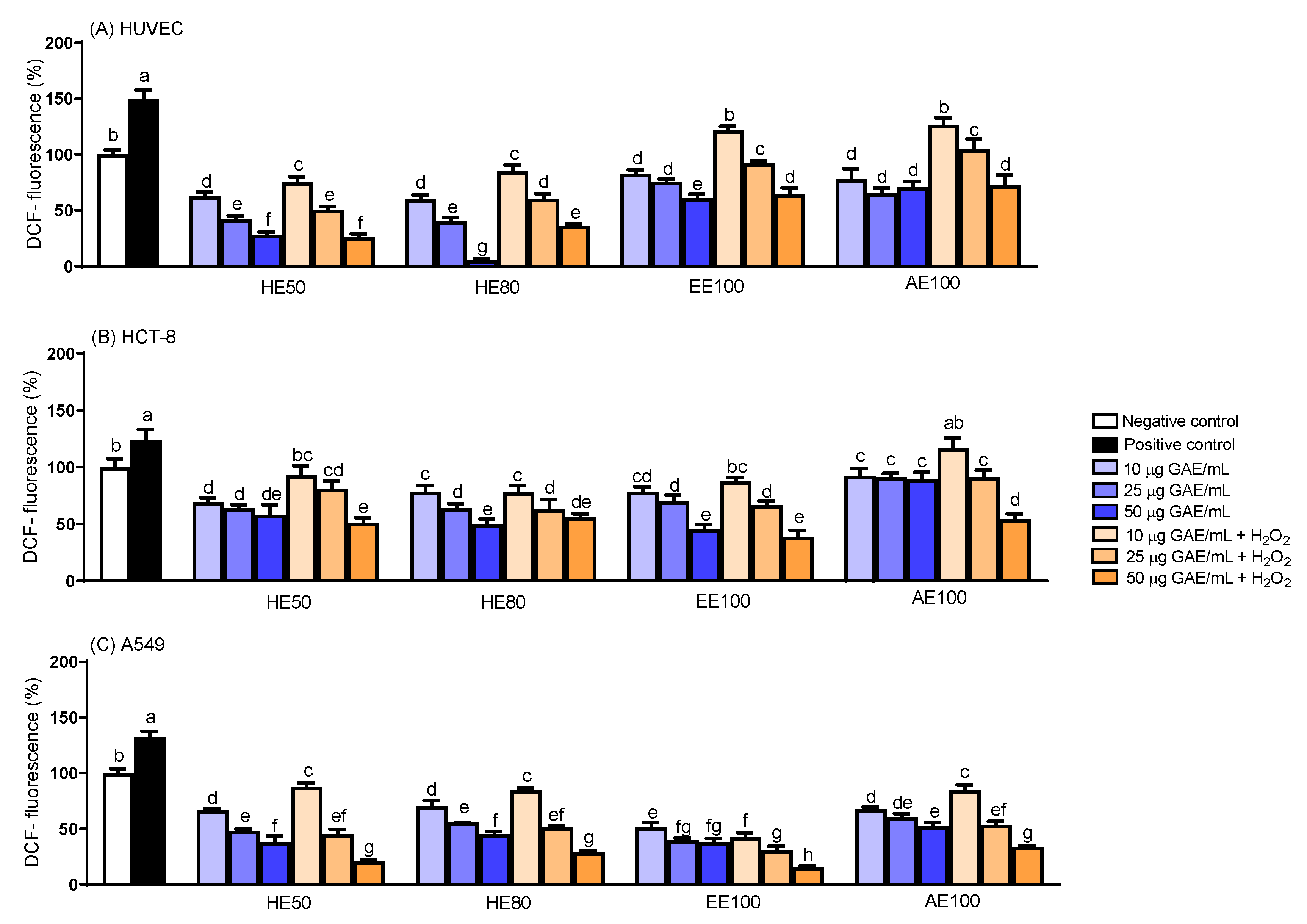
3.4. Intracellular Reactive Oxygen Species (ROS) Generation
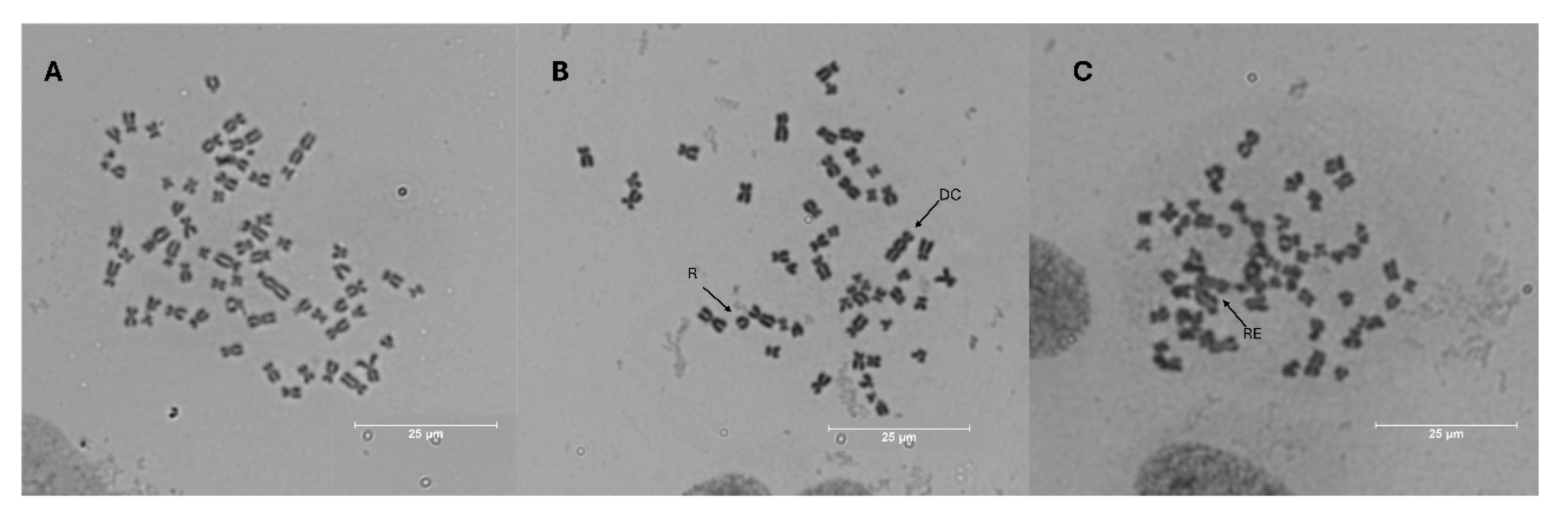
3.5. Chromosomal Aberration
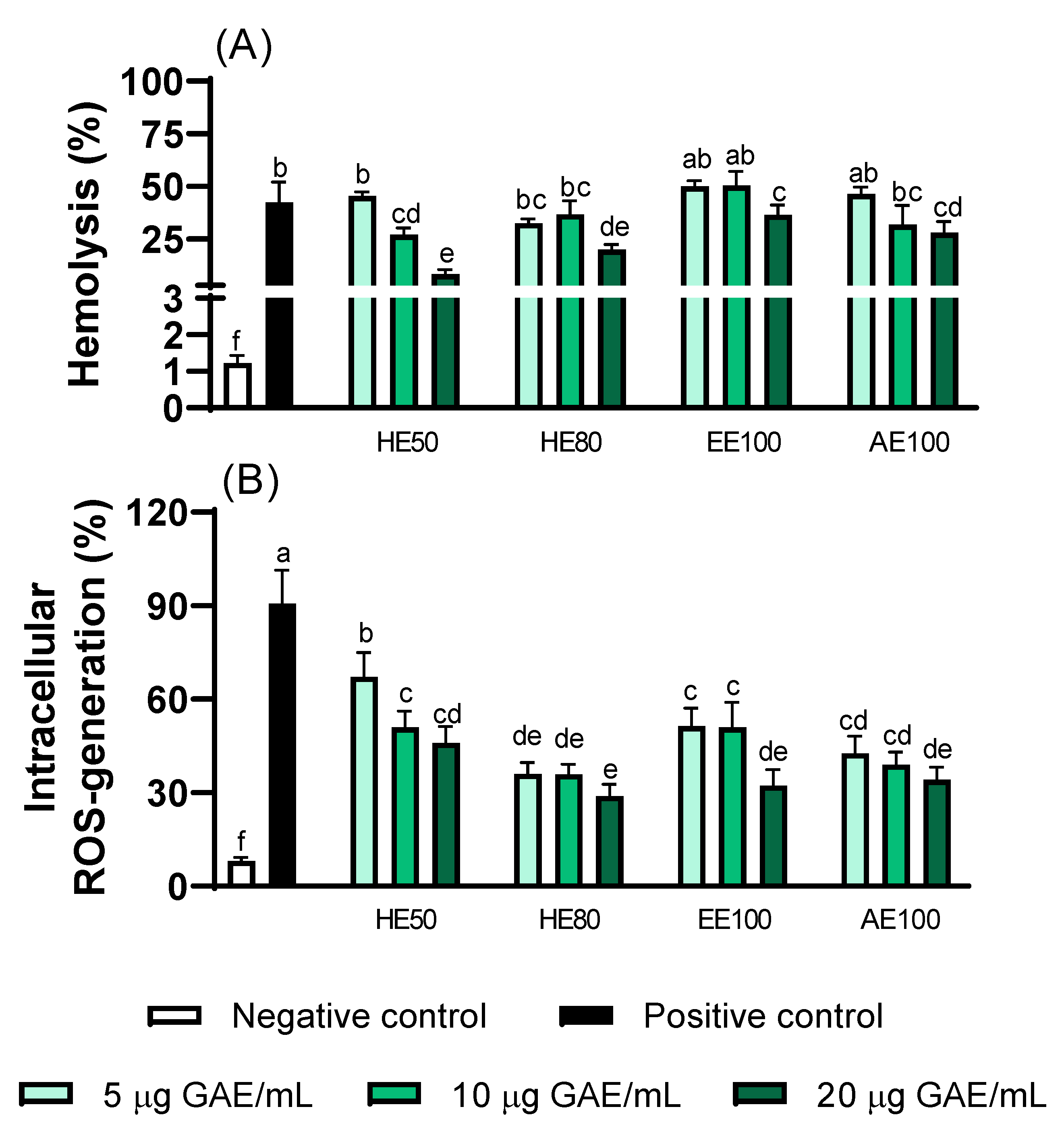
3.6. Effects of T. guianensis Extracts on Erythrocyte Protection
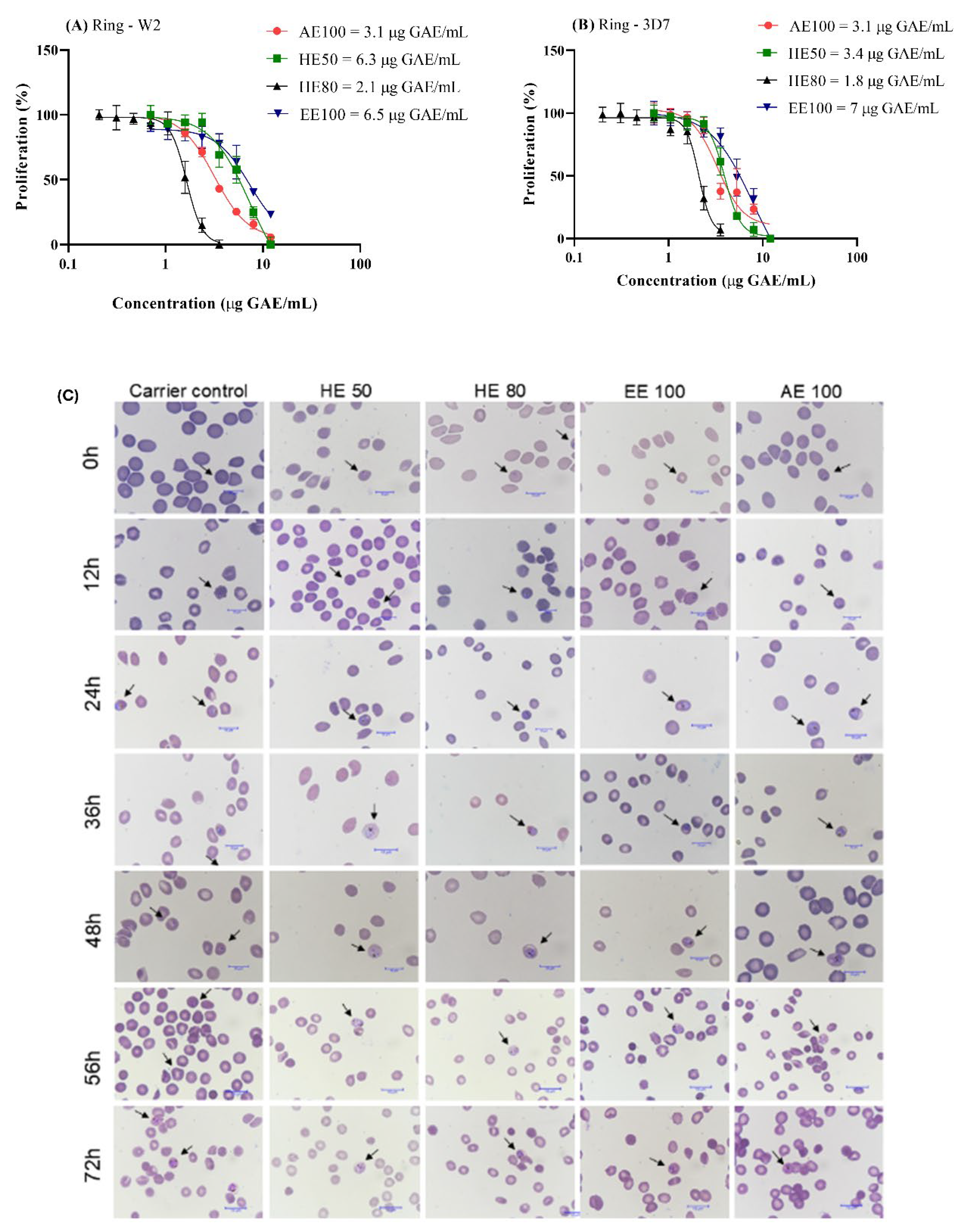
3.7. T. guianensis Extracts Present Antimalarial Activity Against P. falciparum
Conclusions
Authorship contribution statement
Conflicts of interest
Ethics Statements
Acknowledgments
References
- Silva E, David J, David JP, Garcia G, Silva M. Chemical composition of biological active extracts of Tapirira guianensis (Anacardiaceae). Quim Nova 2020. [CrossRef]
- Mar JM, Corrêa RF, Ramos A da S, Kinupp VF, Sanches EA, Campelo PH; et al. Enhancing Bioactive Compound Bioaccessibility in Tapirira guianensis Juices through Ultrasound-Assisted Applications. Processes 2023, 11, 2718. [Google Scholar] [CrossRef]
- Silva E, David J, David JP, Garcia G, Silva M. CHEMICAL COMPOSITION OF BIOLOGICAL ACTIVE EXTRACTS OF Tapirira guianensis (ANACARDIACEAE). Quim Nova 2020. [CrossRef]
- Roumy V, Fabre N, Portet B, Bourdy G, Acebey L, Vigor C; et al. Four anti-protozoal and anti-bacterial compounds from Tapirira guianensis. Phytochemistry 2009, 70, 305–11. [Google Scholar] [CrossRef] [PubMed]
- Zhang H, Tsao R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr Opin Food Sci 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Zeb, A. Concept, mechanism, and applications of phenolic antioxidants in foods. J Food Biochem 2020, 44. [Google Scholar] [CrossRef]
- World Health Organization. The Global Burden of Disease: 2020 Update. Geneva: World Health Organization 2022.
- World Health Organization. World Malaria Report 2024: Addressing Inequity in the Global Malaria Response; WHO: Geneva, Switzerland, 2024; Available online: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2024 (accessed on 12/12/2024).
- Rosenthal, PJ. The interplay between drug resistance and fitness in malaria parasites. Mol Microbiol 2013, 89, 1025–38. [Google Scholar] [CrossRef]
- Haldar K, Bhattacharjee S, Safeukui I. Drug resistance in Plasmodium. Nat Rev Microbiol 2018, 16, 156–70. [Google Scholar] [CrossRef]
- Cragg GM, Newman DJ. Natural products: A continuing source of novel drug leads. Biochimica et Biophysica Acta (BBA) - General Subjects 2013, 1830, 3670–95. [Google Scholar] [CrossRef]
- Do Carmo M, Granato D, Azevedo L. Antioxidant/pro-oxidant and antiproliferative activities of phenolic-rich foods and extracts: A cell-based point of view. Adv Food Nutr Res. 1st ed., Elsevier Inc.; 2021, p. 1–28. [CrossRef]
- Wen C, Zhang J, Zhang H, Dzah CS, Zandile M, Duan Y; et al. Advances in ultrasound assisted extraction of bioactive compounds from cash crops – A review. Ultrason Sonochem 2018, 48, 538–49. [Google Scholar] [CrossRef]
- Lima A dos S, de Oliveira Pedreira FR, Bento NA, Novaes RD, dos Santos EG, de Almeida Lima GD; et al. Digested galactoglucomannan mitigates oxidative stress in human cells, restores gut bacterial diversity, and provides chemopreventive protection against colon cancer in rats. Int J Biol Macromol 2024, 277, 133986. [Google Scholar] [CrossRef]
- do Carmo MAV, Fidelis M, Sanchez CA, Castro AP, Camps I, Colombo FA; et al. Camu-camu (Myrciaria dubia) seeds as a novel source of bioactive compounds with promising antimalarial and antischistosomicidal properties. Food Research International 2020, 136, 109334. [Google Scholar] [CrossRef]
- Granato D, Reshamwala D, Korpinen R, Azevedo L, Vieira do Carmo MA, Cruz TM; et al. From the forest to the plate – Hemicelluloses, galactoglucomannan, glucuronoxylan, and phenolic-rich extracts from unconventional sources as functional food ingredients. Food Chem 2022, 381, 132284. [Google Scholar] [CrossRef] [PubMed]
- de Athayde AE, de Araujo CES, Sandjo LP, Biavatti MW. Metabolomic analysis among ten traditional “Arnica” (Asteraceae) from Brazil. J Ethnopharmacol 2021, 265, 113149. [Google Scholar] [CrossRef] [PubMed]
- Xu L, Liu Y, Wu H, Zhou A. Rapid identification of absorbed components and metabolites of Gandou decoction in rat plasma and liver by UPLC-Q-TOF-MSE. Journal of Chromatography B 2020, 1137, 121934. [Google Scholar] [CrossRef]
- Mohammadi N, Dos Santos Lima A, Azevedo L, Granato D. Bridging the gap in antioxidant activity of flavonoids: Correlating the oxidation of human plasma with chemical and cellular assays. Curr Res Food Sci 2024,8. [CrossRef]
- Fidelis M, Vieira do Carmo MA, Azevedo L, Cruz TM, Marques MB, Myoda T; et al. Response surface optimization of phenolic compounds from jabuticaba (Myrciaria cauliflora [Mart.] O.Berg) seeds: Antioxidant, antimicrobial, antihyperglycemic, antihypertensive and cytotoxic assessments. Food and Chemical Toxicology 2020, 142, 111439. [Google Scholar] [CrossRef]
- Do Carmo MAV, Fidelis M, Pressete CG, Marques MJ, Castro-Gamero AM, Myoda T; et al. Hydroalcoholic Myrciaria dubia (camu-camu) seed extracts prevent chromosome damage and act as antioxidant and cytotoxic agents. Food Research International 2019, 125, 108551. [Google Scholar] [CrossRef]
- Cruz TM, Lima A dos S, Silva AO, Mohammadi N, Zhang L, Azevedo L; et al. High-throughput synchronous erythrocyte cellular antioxidant activity and protection screening of phenolic-rich extracts: Protocol validation and applications. Food Chem 2024, 440, 138281. [Google Scholar] [CrossRef]
- Trager W, Jensen JB. Human Malaria Parasites in Continuous Culture. Science (1979) 1976, 193, 673–5. [Google Scholar] [CrossRef]
- Lambros C, Vanderberg JP. Synchronization of Plasmodium falciparum Erythrocytic Stages in Culture. J Parasitol 1979, 65, 418. [Google Scholar] [CrossRef]
- Alves UV, Jardim e Silva E, dos Santos JG, Santos LO, Lanna E, de Souza Pinto AC; et al. Potent and selective antiplasmodial activity of marine sponges from Bahia state, Brazil. Int J Parasitol Drugs Drug Resist 2021, 17, 80–3. [Google Scholar] [CrossRef]
- Castañeda-Valbuena D, Ayora-Talavera T, Luján-Hidalgo C, Álvarez-Gutiérrez P, Martínez-Galero N, Meza-Gordillo R. Ultrasound extraction conditions effect on antioxidant capacity of mango by-product extracts. Food and Bioproducts Processing 2021, 127, 212–24. [Google Scholar] [CrossRef]
- Cruz TM, Santos JS, do Carmo MAV, Hellström J, Pihlava J-M, Azevedo L; et al. Extraction optimization of bioactive compounds from ora-pro-nobis (Pereskia aculeata Miller) leaves and their in vitro antioxidant and antihemolytic activities. Food Chem 2021, 361, 130078. [Google Scholar] [CrossRef]
- Gil-Martín E, Forbes-Hernández T, Romero A, Cianciosi D, Giampieri F, Battino M. Influence of the extraction method on the recovery of bioactive phenolic compounds from food industry by-products. Food Chem 2022, 378, 131918. [Google Scholar] [CrossRef]
- Ma J-Y, Zhou X, Fu J, He C-Y, Feng R, Huang M; et al. In Vivo Metabolite Profiling of a Purified Ellagitannin Isolated from Polygonum capitatum in Rats. Molecules 2016, 21, 1110. [Google Scholar] [CrossRef] [PubMed]
- Dubrow GA, Tello E, Schwartz E, Forero DP, Peterson DG. Identification of non-volatile compounds that impact consumer liking of strawberry preserves: Untargeted LC–MS analysis. Food Chem 2022, 378, 132042. [Google Scholar] [CrossRef]
- Correia S de J, DavidI JM, Silva EP da, David JP, Lopes LMX, Guedes MLS. Flavonóides, norisoprenóides e outros terpenos das folhas de Tapirira guianensis. Quim Nova 2008, 31, 2056–9. [Google Scholar] [CrossRef]
- Oliveira ESC, Pontes FLD, Acho LDR, do Rosário AS, da Silva BJP, de A. Bezerra J; et al. qNMR quantification of phenolic compounds in dry extract of Myrcia multiflora leaves and its antioxidant, anti-AGE, and enzymatic inhibition activities. J Pharm Biomed Anal 2021, 201, 114109. [Google Scholar] [CrossRef]
- Xiao F, Xu T, Lu B, Liu R. Guidelines for antioxidant assays for food components. Food Front 2020, 1, 60–9. [Google Scholar] [CrossRef]
- Taylor PG, Cesari IM, Arsenak M, Ballen D, Abad MJ, Fernández A; et al. Evaluation of Venezuelan Medicinal Plant Extracts for Antitumor and Antiprotease Activities. Pharm Biol 2006, 44, 349–62. [Google Scholar] [CrossRef]
- Silva-Oliveira R, Lopes G, Camargos L, Ribeiro A, Santos F, Severino R; et al. Tapirira guianensis Aubl. Extracts Inhibit Proliferation and Migration of Oral Cancer Cells Lines. Int J Mol Sci 2016, 17, 1839. [Google Scholar] [CrossRef]
- Kato NN, Stavis VK, Boaretto AG, Castro DTH, Alves FM, de Picoli Souza K; et al. Application of the metabolomics approach to the discovery of active compounds from Brazilian trees against resistant human melanoma cells. Phytochemical Analysis 2021, 32, 992–1002. [Google Scholar] [CrossRef]
- Hussein-Al-Ali SH, Al-Qubaisi M, Rasedee A, Hussein MZ. Evaluation of the Cytotoxic Effect of Ellagic Acid Nanocomposite in Lung Cancer A549 Cell Line and RAW 264.9 Cells. Journal of Bionanoscience 2017, 11, 578–83. [Google Scholar] [CrossRef]
- Liu Q, Liang X, Niu C, Wang X. Ellagic acid promotes A549 cell apoptosis via regulating the phosphoinositide 3-kinase/protein kinase B pathway. Exp Ther Med 2018. [CrossRef]
- Ko E-B, Jang Y-G, Kim C-W, Go R-E, Lee HK, Choi K-C. Gallic Acid Hindered Lung Cancer Progression by Inducing Cell Cycle Arrest and Apoptosis in A549 Lung Cancer Cells via PI3K/Akt Pathway. Biomol Ther (Seoul) 2022, 30, 151–61. [Google Scholar] [CrossRef] [PubMed]
- Tsantila EM, Esslinger N, Christou M, Papageorgis P, Neophytou CM. Antioxidant and Anticancer Activity of Vitis vinifera Extracts in Breast Cell Lines. Life 2024, 14, 228. [Google Scholar] [CrossRef] [PubMed]
- Ghose K, Selvaraj K, McCallum J, Kirby CW, Sweeney-Nixon M, Cloutier SJ; et al. Identification and functional characterization of a flax UDP-glycosyltransferase glucosylating secoisolariciresinol (SECO) into secoisolariciresinol monoglucoside (SMG) and diglucoside (SDG). BMC Plant Biol 2014, 14, 82. [Google Scholar] [CrossRef]
- Polat Kose L, Gulcin İ. Evaluation of the Antioxidant and Antiradical Properties of Some Phyto and Mammalian Lignans. Molecules 2021, 26, 7099. [Google Scholar] [CrossRef]
- Nakamura H, Takada K. Reactive oxygen species in cancer: Current findings and future directions. Cancer Sci 2021, 112, 3945–52. [Google Scholar] [CrossRef]
- Nogueira V, Hay N. Molecular Pathways: Reactive Oxygen Species Homeostasis in Cancer Cells and Implications for Cancer Therapy. Clinical Cancer Research 2013, 19, 4309–14. [Google Scholar] [CrossRef]
- Santibáñez-Andrade M, Quezada-Maldonado EM, Rivera-Pineda A, Chirino YI, García-Cuellar CM, Sánchez-Pérez Y. The Road to Malignant Cell Transformation after Particulate Matter Exposure: From Oxidative Stress to Genotoxicity. Int J Mol Sci 2023, 24, 1782. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress: Concept and Some Practical Aspects. Antioxidants 2020, 9, 852. [Google Scholar] [CrossRef]
- Tchounwou PB, Dasari S, Noubissi FK, Ray P, Kumar S. Advances in Our Understanding of the Molecular Mechanisms of Action of Cisplatin in Cancer Therapy. J Exp Pharmacol 2021, 13, 303–28. [Google Scholar] [CrossRef]
- Hazafa A, Rehman KU, Jahan N, Jabeen Z. The Role of Polyphenol (Flavonoids) Compounds in the Treatment of Cancer Cells. Nutr Cancer 2020, 72, 386–97. [Google Scholar] [CrossRef] [PubMed]
- do Carmo MAV, Pressete CG, Marques MJ, Granato D, Azevedo L. Polyphenols as potential antiproliferative agents: Scientific trends. Curr Opin Food Sci 2018, 24, 26–35. [Google Scholar] [CrossRef]
- Bonassi S, Norppa H, Ceppi M, Stromberg U, Vermeulen R, Znaor A; et al. Chromosomal aberration frequency in lymphocytes predicts the risk of cancer: Results from a pooled cohort study of 22 358 subjects in 11 countries. Carcinogenesis 2008, 29, 1178–83. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen C, Alonso P, Ringwald P. Current and emerging strategies to combat antimalarial resistance. Expert Rev Anti Infect Ther 2022, 20, 353–72. [Google Scholar] [CrossRef]
- Villarreal W, Castro W, González S, Madamet M, Amalvict R, Pradines B; et al. Copper (I)-Chloroquine Complexes: Interactions with DNA and Ferriprotoporphyrin, Inhibition of β-Hematin Formation and Relation to Antimalarial Activity. Pharmaceuticals 2022, 15, 921. [Google Scholar] [CrossRef]
- Jansen O, Tits M, Angenot L, Nicolas J-P, De Mol P, Nikiema J-B; et al. Anti-plasmodial activity of Dicoma tomentosa (Asteraceae) and identification of urospermal A-15-O-acetate as the main active compound. Malar J 2012, 11, 289. [Google Scholar] [CrossRef]
- Mohammed WSA, Yasin K, Mahgoub NS, Abdel Hamid MM. Cross sectional study to determine chloroquine resistance among Plasmodium falciparum clinical isolates from Khartoum, Sudan. F1000Res 2018, 7, 208. [Google Scholar] [CrossRef]
- Foguim FT, Bogreau H, Gendrot M, Mosnier J, Fonta I, Benoit N; et al. Prevalence of mutations in the Plasmodium falciparum chloroquine resistance transporter, PfCRT, and association with ex vivo susceptibility to common anti-malarial drugs against African Plasmodium falciparum isolates. Malar J 2020, 19, 201. [Google Scholar] [CrossRef]
- Ali AH, Agustar HK, Hassan NI, Latip J, Embi N, Sidek HM. Data on antiplasmodial and stage-specific inhibitory effects of Aromatic (Ar)-Turmerone against Plasmodium falciparum 3D7. Data Brief 2020, 33, 106592. [Google Scholar] [CrossRef]
- Kennedy L, Sandhu JK, Harper ME, Cuperlovic-culf M. Role of glutathione in cancer: From mechanisms to therapies. Biomolecules 2020, 10, 1–27. [Google Scholar] [CrossRef]
- Painter HJ, Morrisey JM, Vaidya AB. Mitochondrial Electron Transport Inhibition and Viability of Intraerythrocytic Plasmodium falciparum. Antimicrob Agents Chemother 2010, 54, 5281–7. [Google Scholar] [CrossRef] [PubMed]
- Aminake MN, Schoof S, Sologub L, Leubner M, Kirschner M, Arndt H-D; et al. Thiostrepton and Derivatives Exhibit Antimalarial and Gametocytocidal Activity by Dually Targeting Parasite Proteasome and Apicoplast. Antimicrob Agents Chemother 2011, 55, 1338–48. [Google Scholar] [CrossRef] [PubMed]
- Biosca A, Ramírez M, Gomez-Gomez A, Lafuente A, Iglesias V, Pozo OJ; et al. Characterization of Domiphen Bromide as a New Fast-Acting Antiplasmodial Agent Inhibiting the Apicoplastidic Methyl Erythritol Phosphate Pathway. Pharmaceutics 2022, 14, 1320. [Google Scholar] [CrossRef]
- Oliveira ESC, Pontes FLD, Acho LDR; et al. NMR and multivariate methods: Identification of chemical markers in extracts of pedra-ume-caá and their antiglycation, antioxidant, and enzymatic inhibition activities. Phytochem Anal. 2024, 35, 552–566 https://. [Google Scholar] [CrossRef] [PubMed]
- Pereira ZC, Cruz JMA, Castro DRG; et al. Passiflora nitida Kunth fruit: Chemical analysis, antioxidant capacity, and cytotoxicity. Food Science and Technology, v. 44, p. 1–8, 2024. [CrossRef]
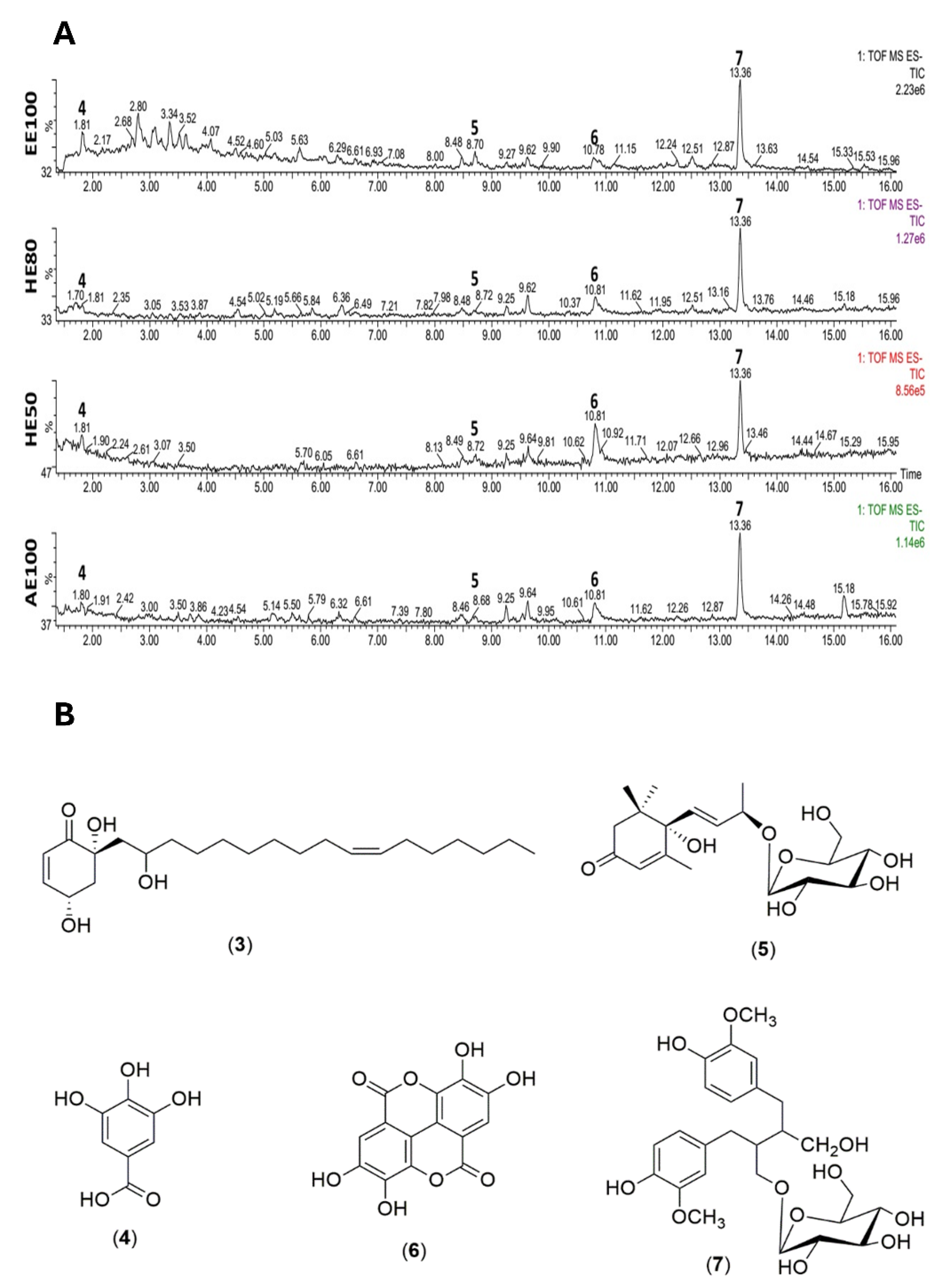
| Compounds | HE50 | HE80 | EE100 | AE100 |
|---|---|---|---|---|
| TPC (mg GAE/g) | 53 ± 1a | 23 ± 1c | 14 ± 0.6d | 32 ± 0.7b |
| TFC (mg CE/g) | 158 ± 11a | 113 ± 2c | 32 ± 4d | 132 ± 2b |
| Total flavonol content | 16 ± 1a | 4 ± 0.3b | 6 ± 0.7b | 5 ± 1b |
| (mg QE/g) | ||||
|
Ortho-diphenols (mg CAE/g) |
33 ± 1a | 15 ± 0.5b | 7 ± 0.4c | 15 ± 1b |
| Antioxidant activity | ||||
| DPPH (mg AAE/g) | 103 ± 3b | 78 ± 1c | 27 ± 2d | 119 ± 1a |
| FRAP (mg AAE/g) | 87 ± 2a | 54 ± 1b | 24 ± 1d | 50 ± 0.3c |
| Hydroxyl radical scavenging activity (mg GAE/g) | 16 ± 0.4d | 44 ± 1b | 23 ± 0.5c | 53 ± 2a |
| No | RT (min) | Adduct | m/z | Identified mass |
Calculated mass | Fragmentations (m/z) |
Compound (Empirical formula, error in ppm) |
δ 1H in ppm (J, Hz) | δ 13C in ppm | References |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | - | - | - | - | - | - | α-glucose | δ 5.08 (d; J=3.7 Hz, H-1) | 93.8 (C-1), 71.8 (C-2), 73.5 (C3). | [62] |
| 2 | - | - | - | - | - | - | β-glucose | δ 4.45 (d; J=7.7 Hz, H-1) δ 3.31 (dd; J=9.2; 7.8 Hz, H-3) |
98.0 (C-1), 77.8 (C-3), 70.3 (C-4). | [62] |
| 3 | - | - | - | - | - | - | 4,6,2’-trihydroxi-6-[10’(Z)-heptadecenyl]-1-cyclohexene-2-one (C23H40O4) |
5.88 (dd, J = 10.2; 2.0 Hz, H-2); 6.92 (m, H-3); 1.90 (m, H-1’), 4.02 (m, H-2’); 5.31 (t, J = 5.0 Hz, H-10’, H-11’); 2.0 (m, H-12’), 0.88 (t, J = 7.0 Hz, H-17’). | 126.2 (C-2), 153.6 (C-3), 64.7 (C-4), 43.3 (C-1’), 70.7 (C-2’), 130.5 (C10’, C11’), 27.8 (C12’), 14.0 (C-17’). | [4] |
| 4 | 1.8 | [M-H]- | 169.0133 | 170.0211 | 170.0215 | 125.0245 | Gallic acid (C7H5O5, -2.35) |
7.03 (s, H-2,6) | 108.8 (C-2, 6), 145.0 (C-3,5), 138.5 (C-4), 168.8 (C-7). |
[1,61] |
| 5 | 8.7 | [M+Formic Acid-H]- | 385.1892 | 386,1970 | 386.1940 | 431.1931 223.1338 205.1258 |
(6S,7E,9S)-6,9-dihydroxy-megastigma-4,7-dien-3-one 9-O-β-glucopyranoside (C19H30O8, 7.77) |
2.44 (m, H-2ax) 2.56 (m, H-2eq) |
50.5 (C-2), 200.0 (C-3), 131.3 (C-7), 71.8 (C-4’). | [31] |
| 6 | 10.8 | [M-H]- | 300.9990 | 302.00683 | 302.00626 | 283.9960 229.0141 185.0220 |
Ellagic acid (C14H5O8, 1.88) |
7.53 (s, H-5’, H-5’). | 113.0 (C-1, C-1’), 139.7 (C-3, C-3’), 148.0 (C-4, C-4’), 110.5 (C5, C-5’), 108.1 (C6, C-6’), 160.0 (C-7, C-7’). | [29,61] |
| 7 | 13.3 | [M-H]- | 523.2188 | 524.2266 | 524.2257 | 361.1647 | (−)-Secoisolariciresinol-9’-O-β-d-glucopyranoside (C26H36O11, 1.71) |
6.56 (d, J = 1.8 Hz, H-2); 6.63 (d, J = 8.0 Hz, H-6); 3.49 (d, J = 5.1 Hz, H-9); 2.49 – 2.54 (m, H-7, H-7’); 1.91 (m, H-8); 6.58 (d, J = 1.8 Hz, H-2’); 6.63 (d, J = 8.0 Hz, H-5’); 6.53 (m, H-6’); 4.20 (d, J = 7.8 Hz, H-1’’); 3.84 (d, J = 1.8 Hz, H-6’’); 3.63 (m, H-6’’); 3.82 (m, OCH3-3); 3.71 (s, OCH3-3’). |
113.3 (C-2), 147.2 (C-4), 122.4 (C-6), 61.1 (C9), 36.5 (C-7, C-7’); 43.5 (C-8), 132.7 (C-1’), 115.8 (C-2’), 147.4 (C-3’), 144.2 (C-4’), 115.5 (C-5’), 122.4 (C-6’), 104.3 (C-1’’) 62.5 (C-6”), 56.4 (OCH3-3) 56.0 (OCH3-3’) |
[30] |
| Extracts | HUVEC / P. falciparum | HUVEC / Cancer cells | ||||
| 3D7 | W2 | A549 | HCT-8 | |||
| HE50 | 28.4 | 15.5 | 1.7 | 2.2 | ||
| HE80 | 70.5 | 60.2 | 2.6 | 2.7 | ||
| EE100 | 20 | 21.3 | 0.6 | 1.2 | ||
| AE100 | 17.6 | 18 | 0.3 | 0.7 | ||
| HE50 Dosage (µg GAE/mL) |
CIS | TC | Aberrant Type | TNCA | CA rate (%) | |||||||
| R | DC | FR | CB | CEB | TC | QC | RE | |||||
| NC | 4315 | 2 | 31 | 0 | 0 | 0 | 1 | 1 | 7 | 42b | 1 | |
| PC | 4 μM | 5239 | 8 | 40 | 0 | 0 | 0 | 3 | 5 | 22 | 78a | 1.5 |
| 20 | 4343 | 3 | 19 | 1 | 0 | 0 | 1 | 7 | 9 | 40b | 1 | |
| 50 | 4367 | 0 | 15 | 1 | 0 | 0 | 1 | 2 | 2 | 21c | 0.5 | |
| 5 | 4 μM | 4045 | 7 | 29 | 1 | 1 | 1 | 7 | 2 | 6 | 41b | 1 |
| 10 | 4 μM | 4038 | 5 | 22 | 3 | 4 | 1 | 2 | 6 | 4 | 34b | 1 |
| 20 | 4 μM | 4269 | 3 | 30 | 3 | 1 | 1 | 2 | 4 | 4 | 35bc | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




