Submitted:
14 January 2025
Posted:
14 January 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Palladium Nanoparticles Supported on Biopolymers for Cross-Coupling Reactions
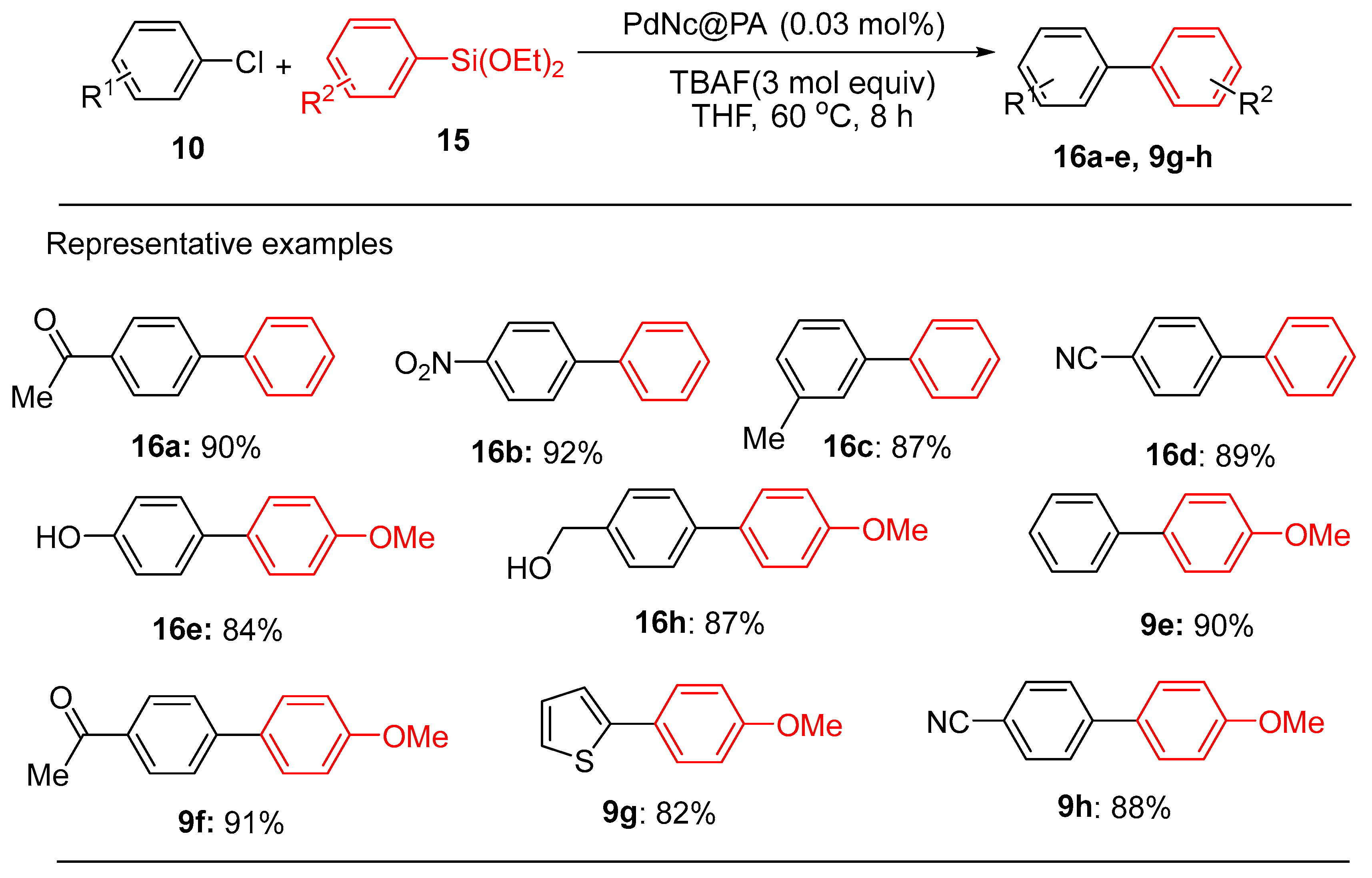
2.1. Palladium Nanoparticles Supported on on Chitosan
2.2. Palladium Nanoparticles Supported on Kenaf-Cellulose Modified with Poly(amidoxime) Ligands
3. Palladium Nanoparticles Supported on Polymers
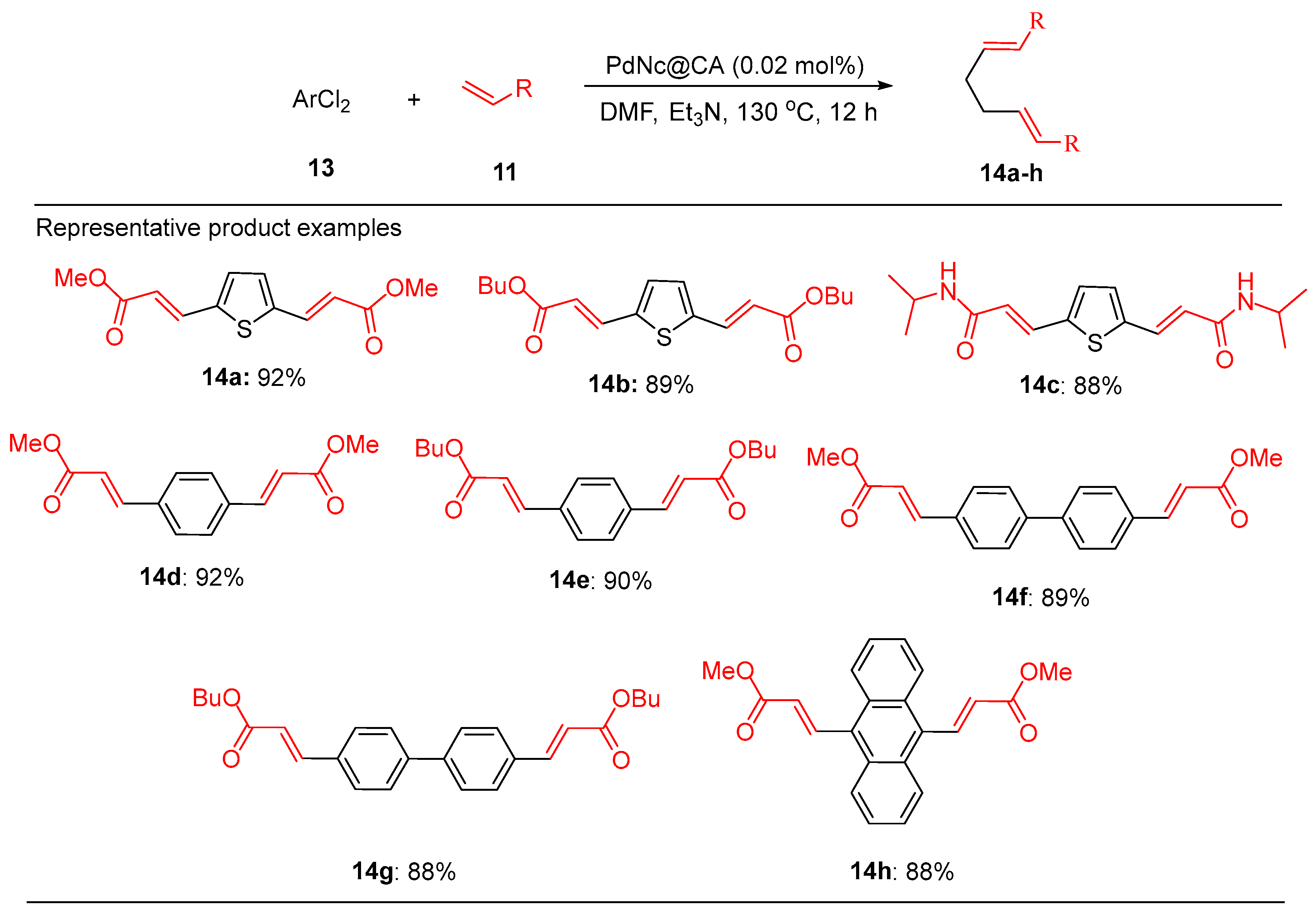
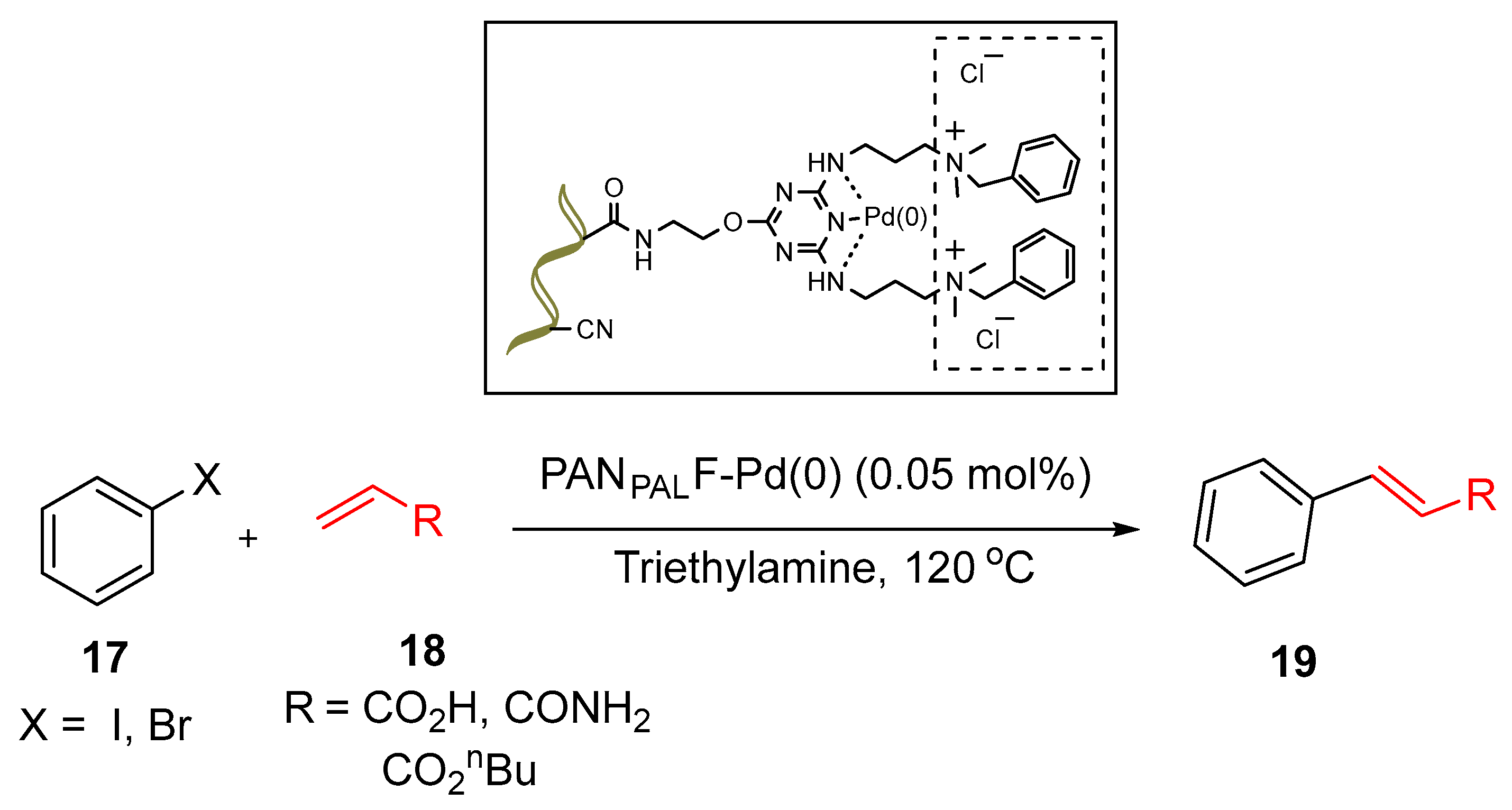
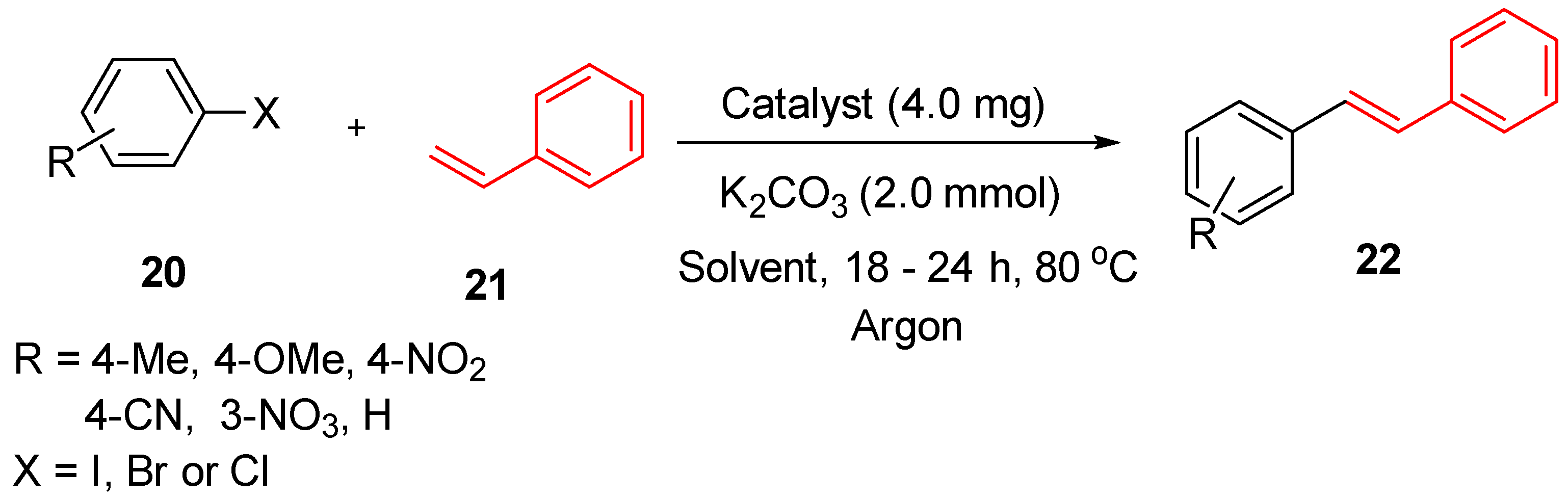
3.1. Palladium Nanoparticles Supported on Functionalized Polyacrylonitrile Fiber
4. Palladium Nanoparticles Supported on Nitrogen-Doped Materials
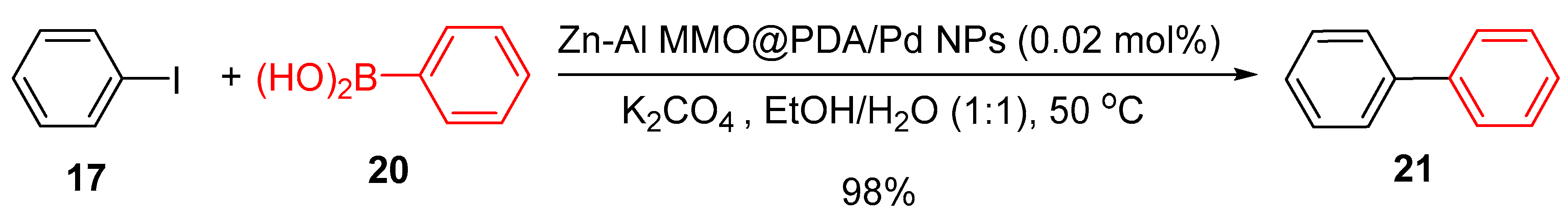
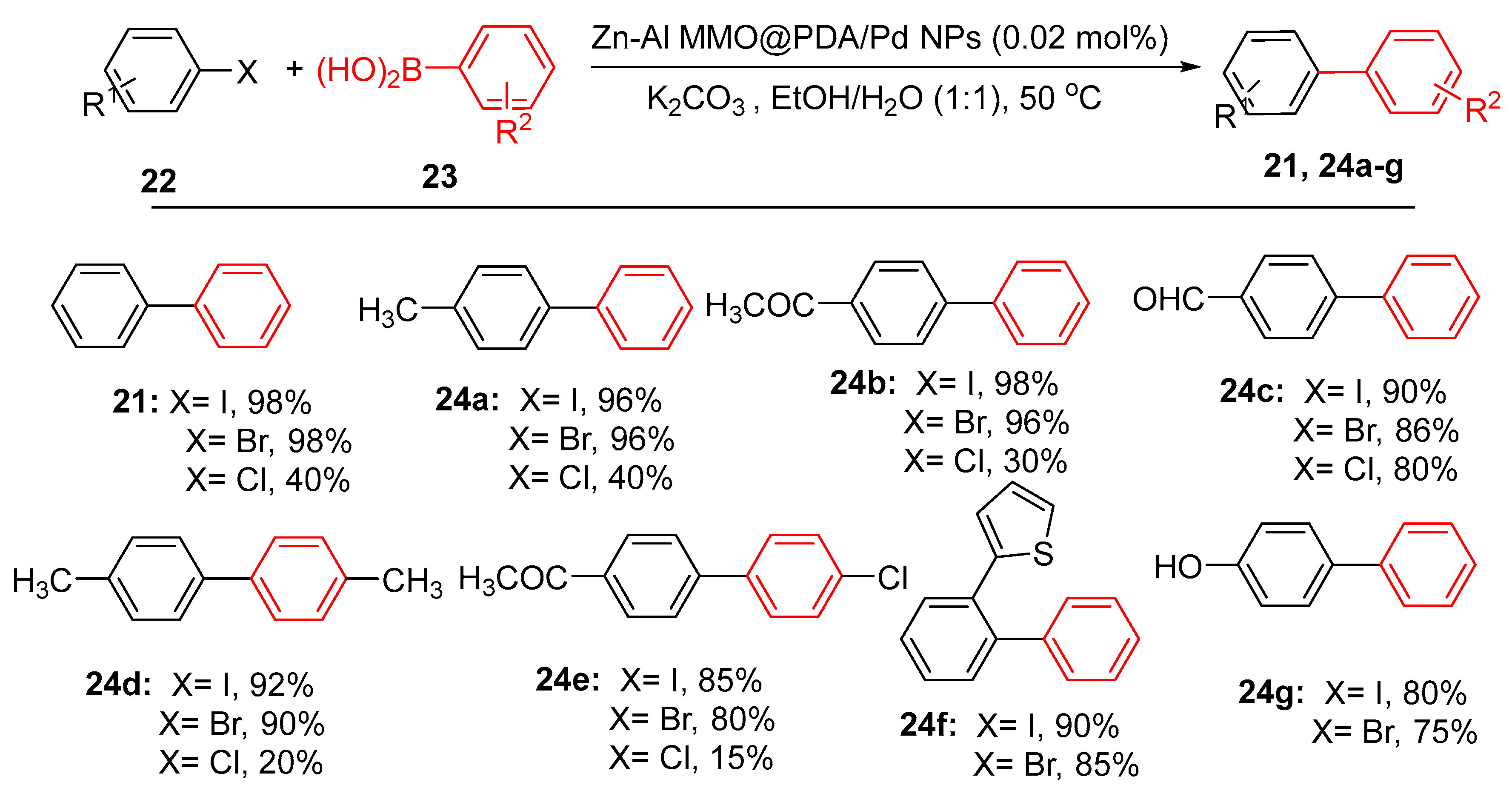
4.1. Palladium Nanoparticles Supported on Polydopamine-Functionalized Zn-Al Mixed Metal Oxides (Zn-Al MMO)
4.2. Palladium Nanoparticles Supported on 3D-Printed Silicon Oxycarbide (SiOC) Monoliths
4.3. Palladium Nanoparticles Supported on Nitrogen-Doped Carbon Nanosheets (N-CNS)
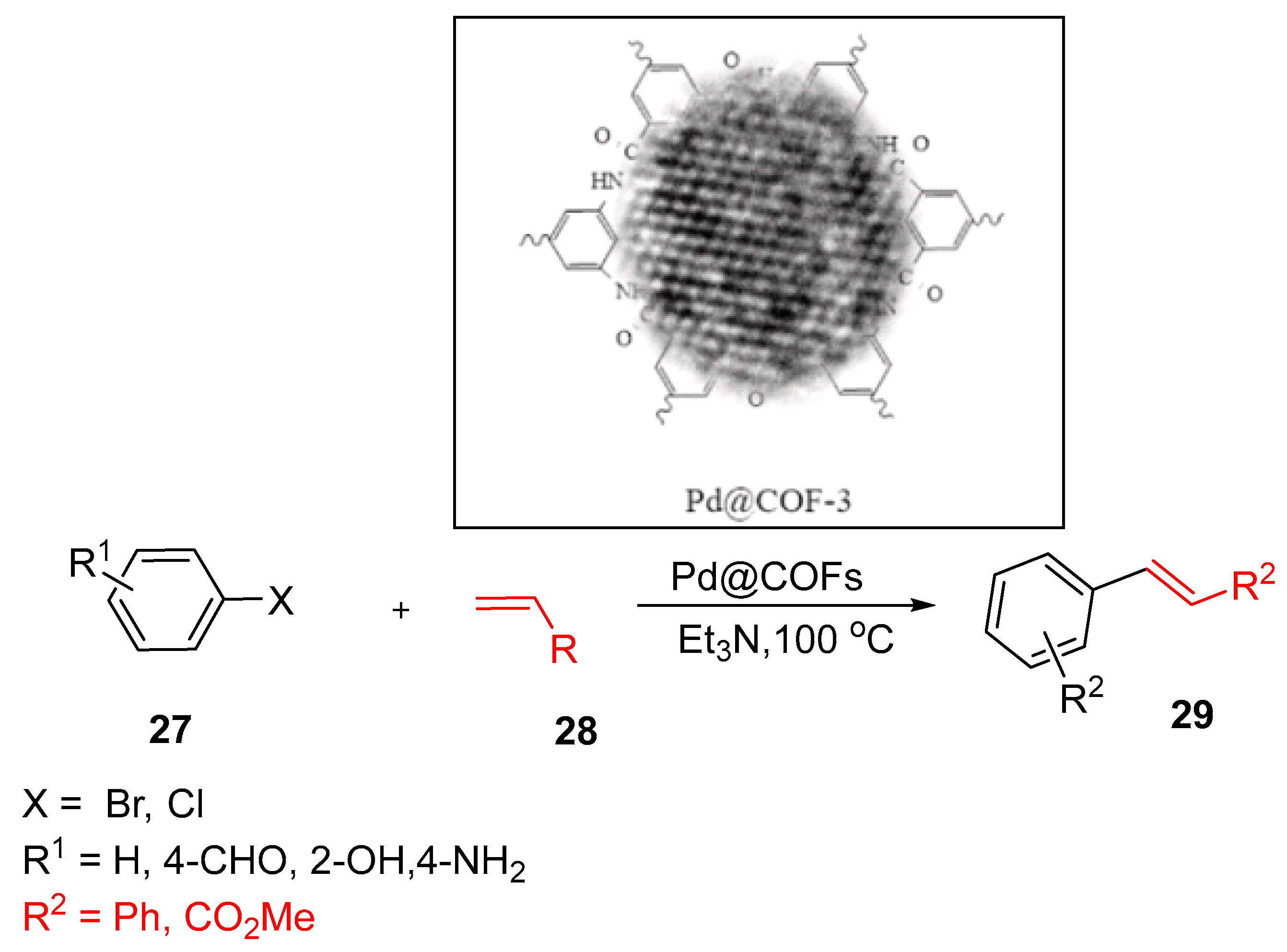
4.4. Palladium Nanocrystals-Embedded Covalent Organic Framework (Pd@COF)
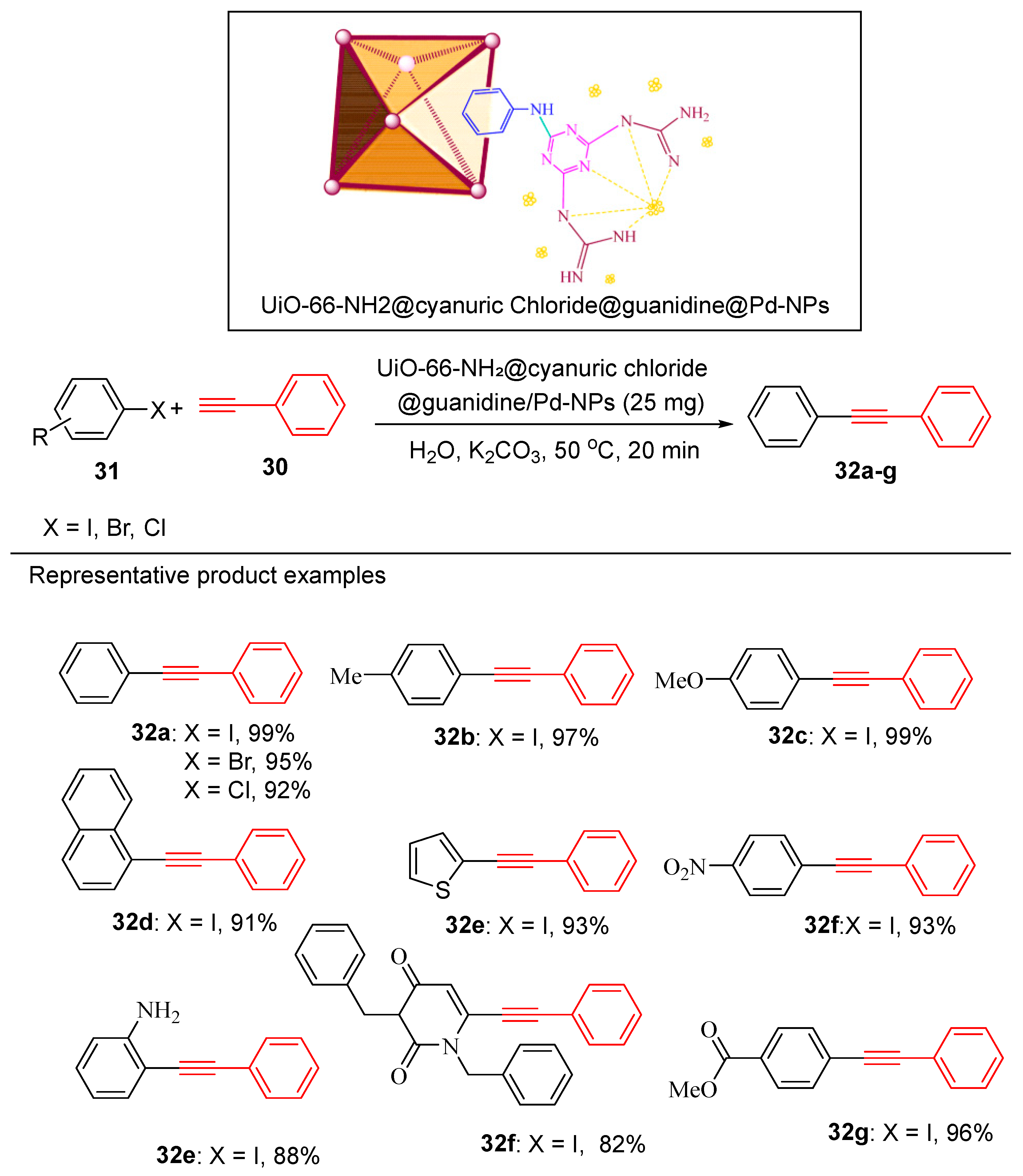
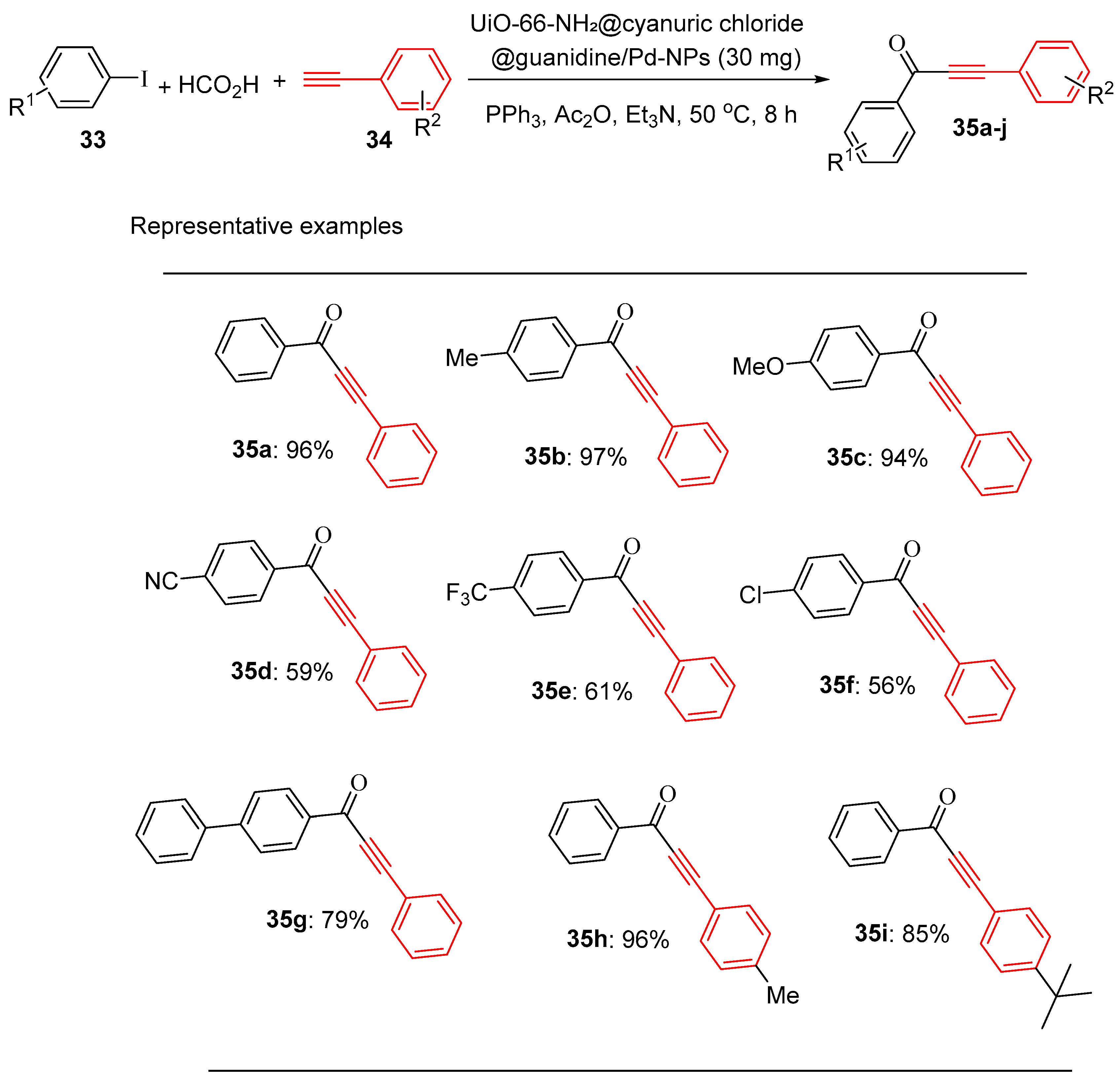
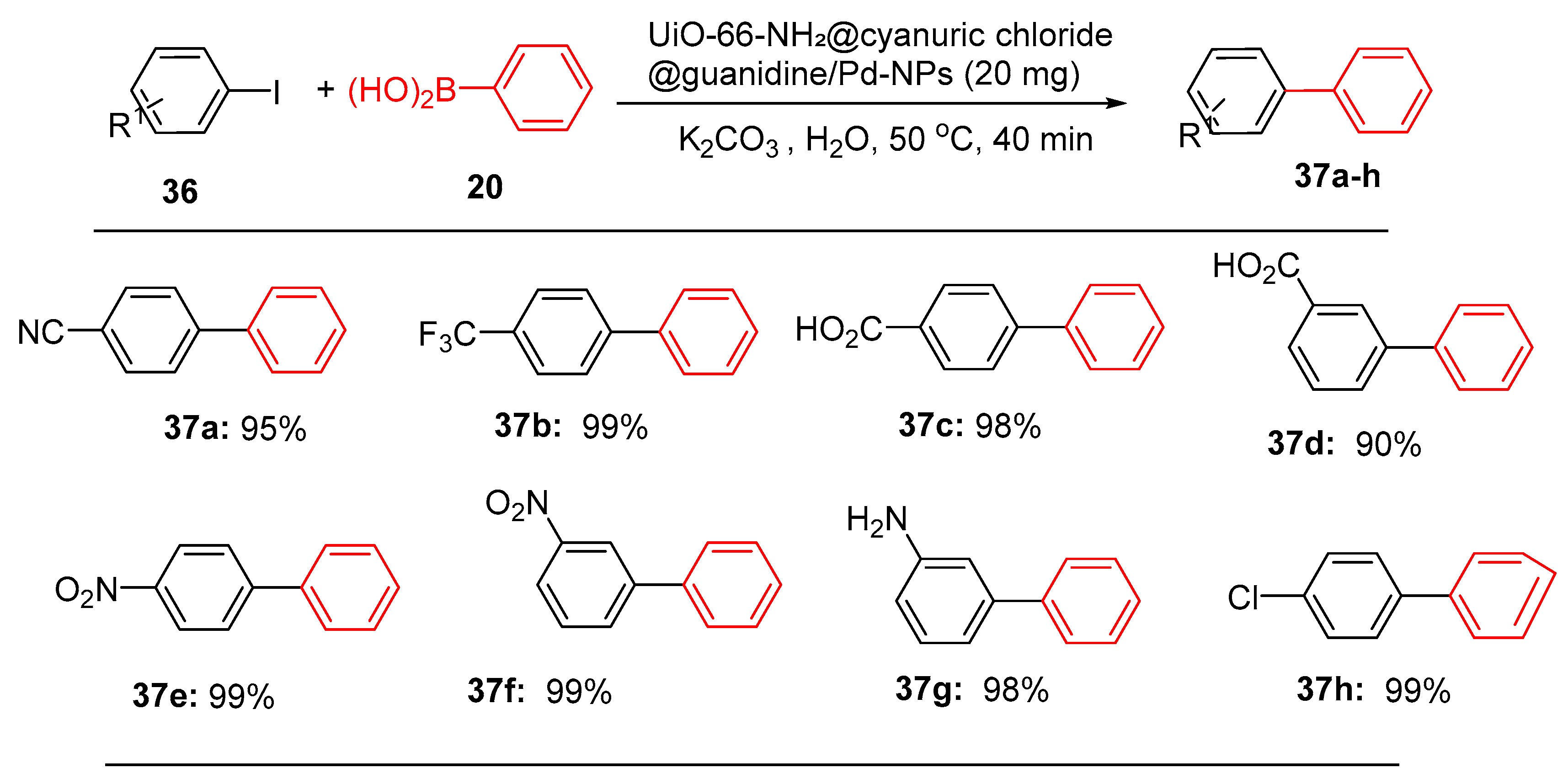
4.5. Palladium Nanoparticle-Decorated Porous Metal–Organic-Framework (Zr)@Guanidine
5. Palladium Nanoparticles Supported on Functionalized Synthetic Material
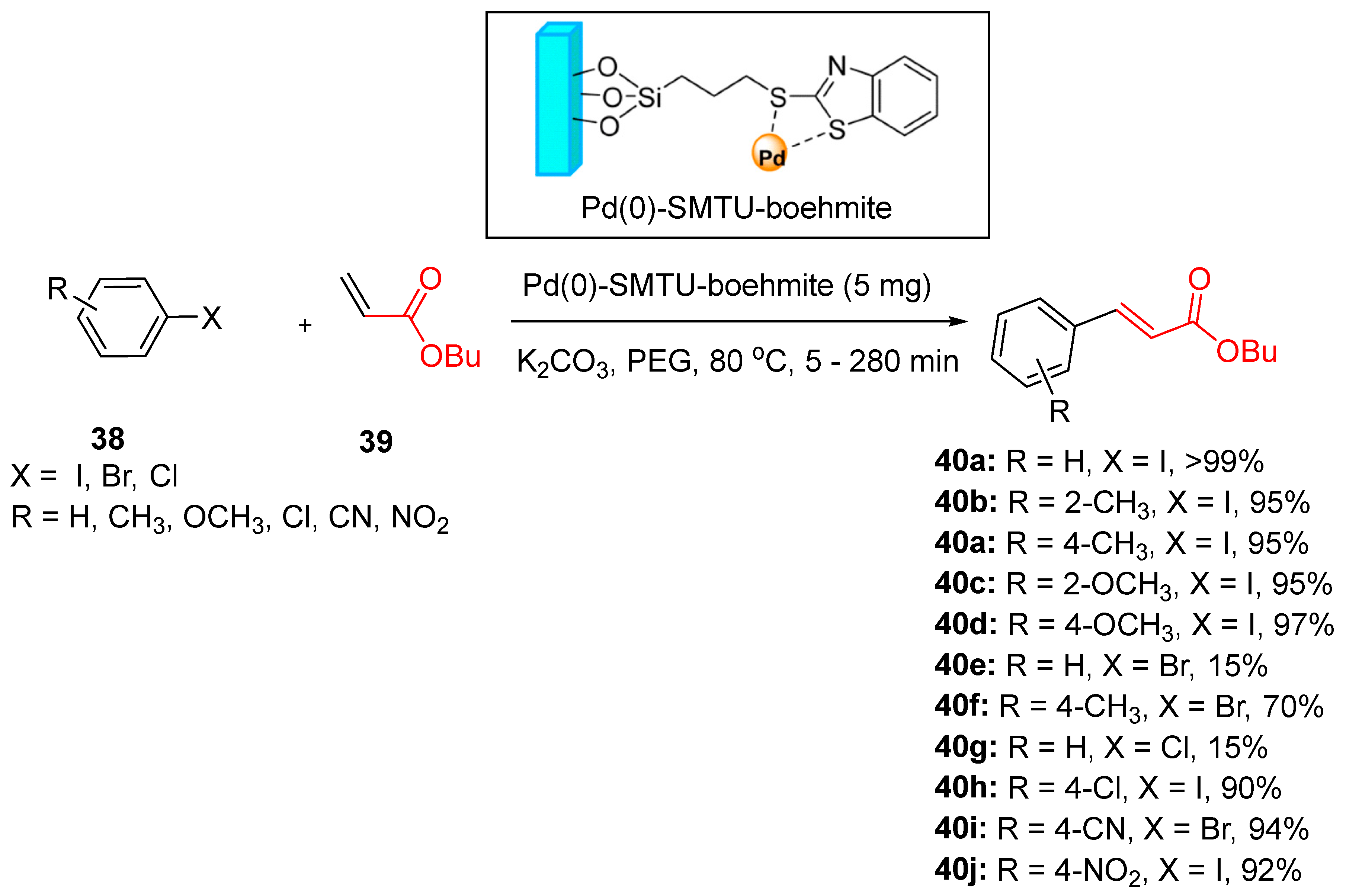
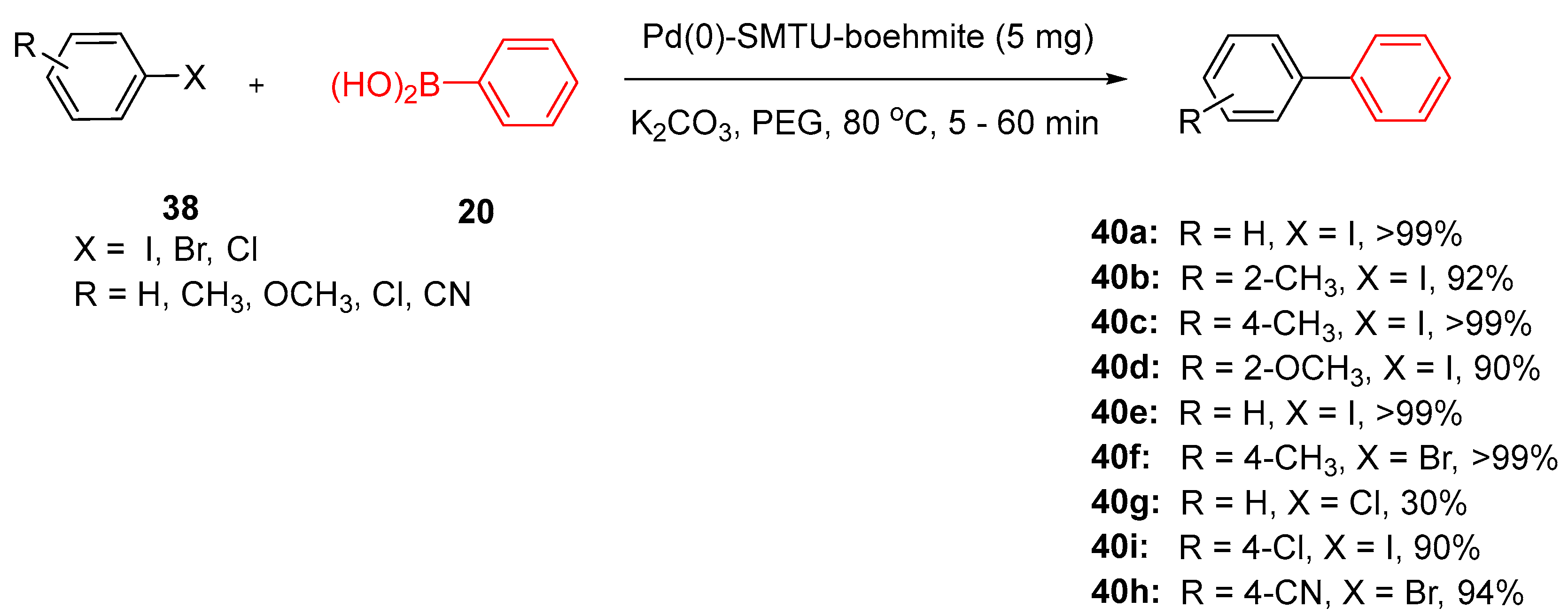
5.1. Palladium Nanoparticles Supported on Boehmite Nanoparticles (Pd(0)-SMTU-Boehmite)
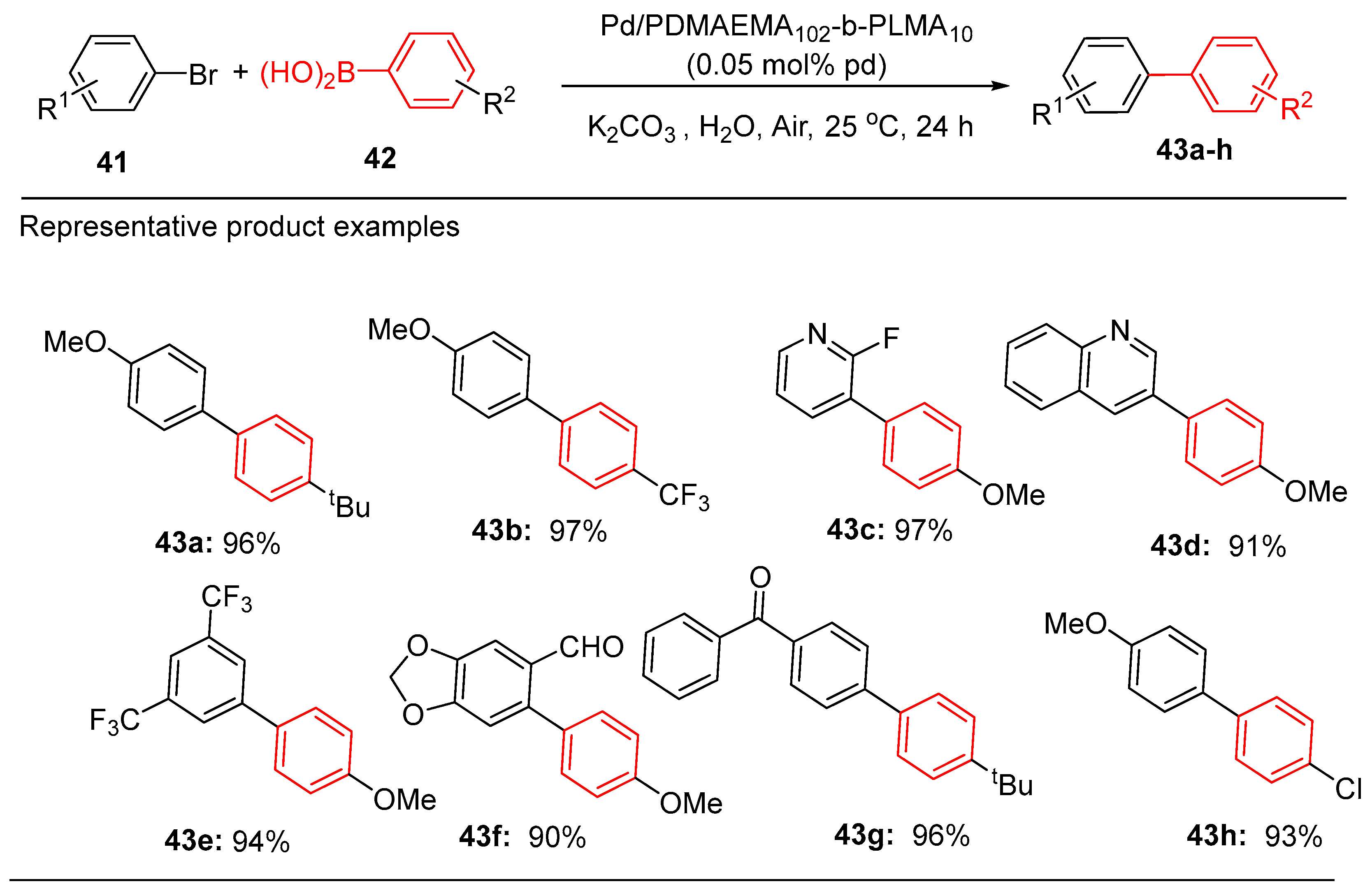
5.2. Palladium Nanoparticles Supported on Micellular Structures
5.3. Palladium Nanoparticles Supported on Polydimethylsiloxane Film
5.4. Palladium Nanoparticles Supported Chiral Surfactants
6. Palladium Nanoparticles Supported on Aerobic Bacterial Cells (Paracoccus yeei)
7. Palladium Nanoparticles Supported on Porous Silica Materials
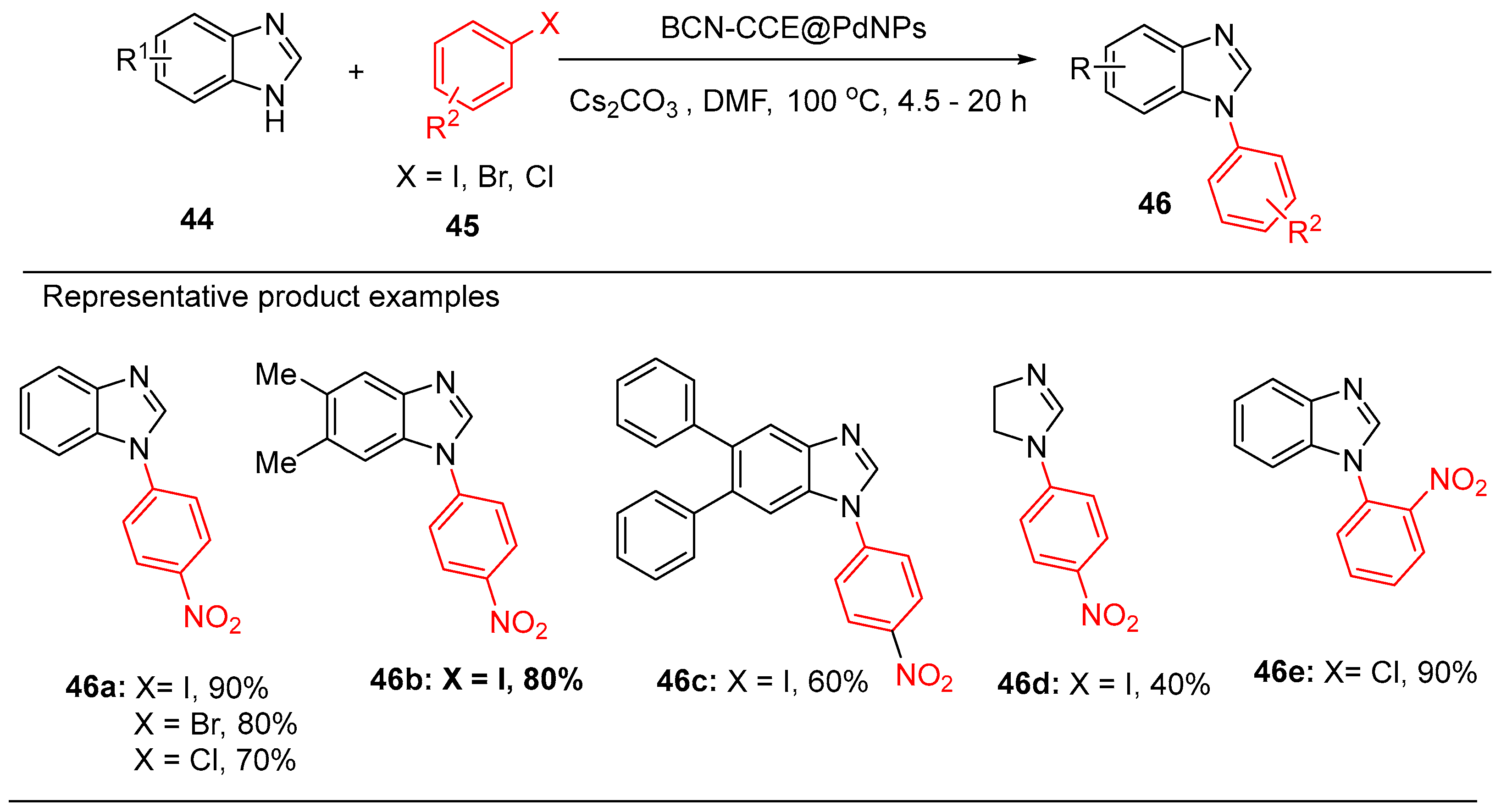
8. Palladium Nanoparticle Immobilized on Coconut Coir Extract Coated Boron Carbon Nitride
9. Palladium Nanoparticles Supported on Metal Oxides and Metal Oxide Hybrid Materials as Catalyst for Cross Coupling Reaction
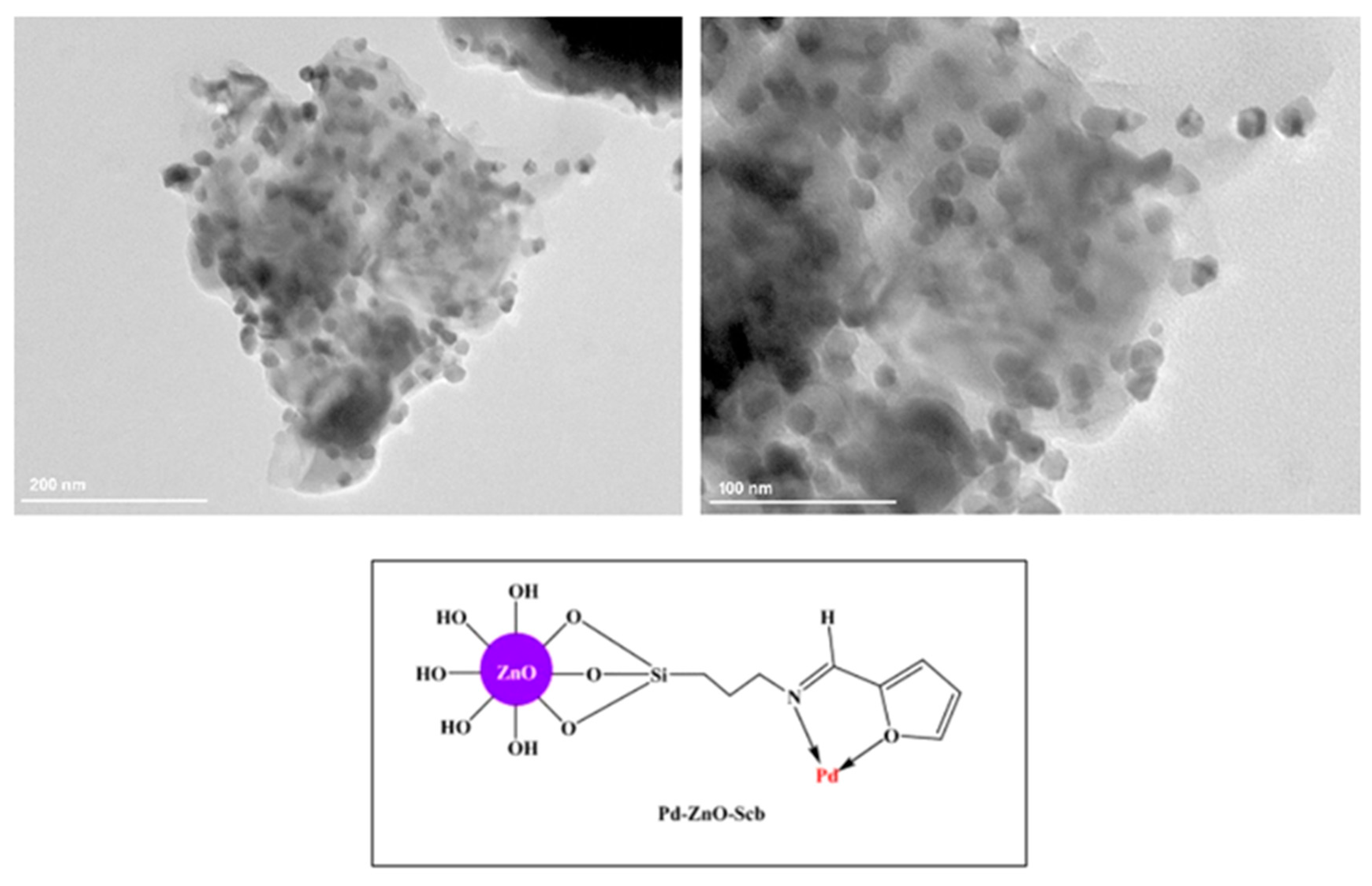
9.1. Immobilised Palladium Nanoparticles on Schiff Base-Modified ZnO Particles (Pd–ZnO–Scb)
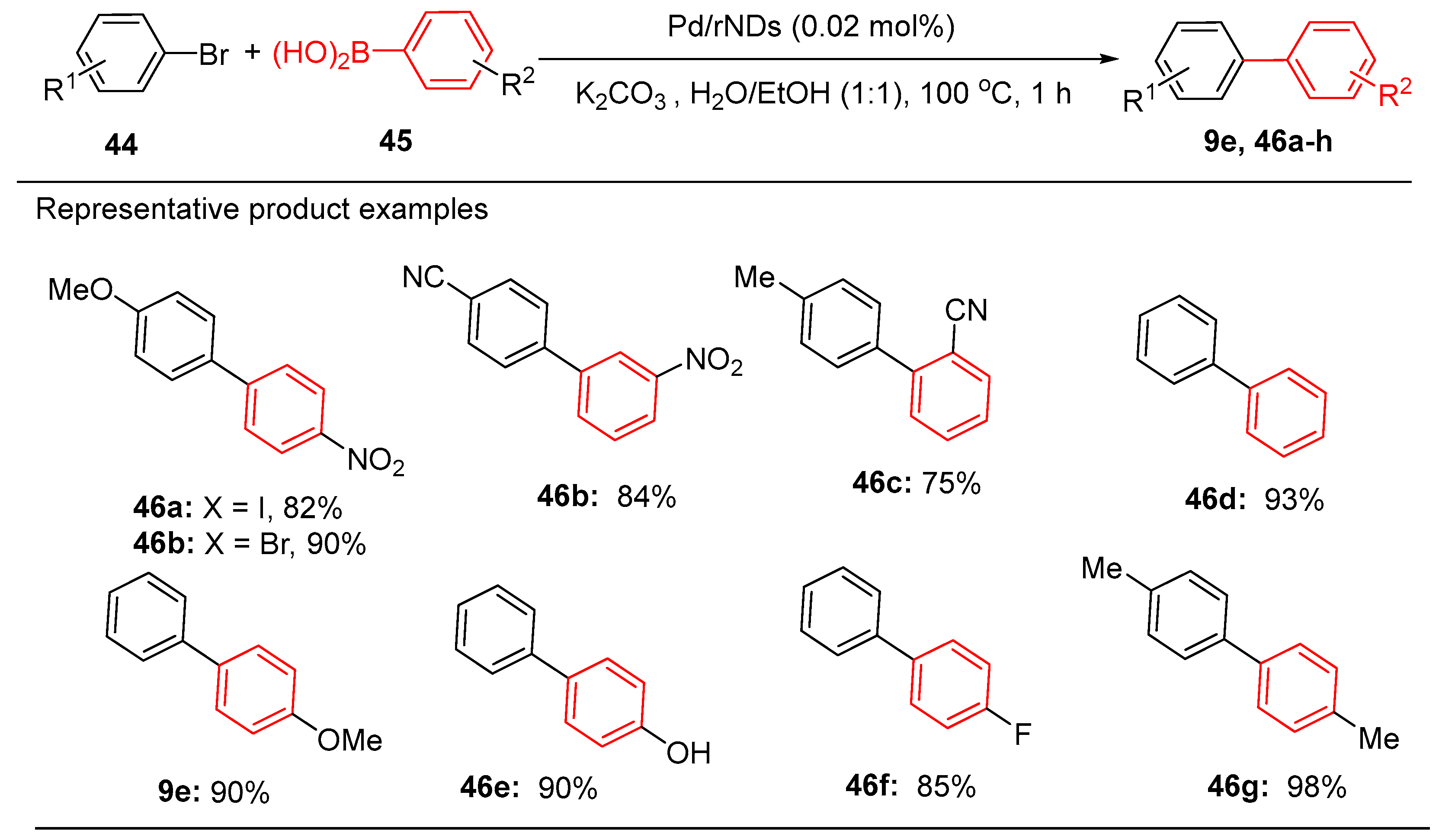
9.2. Pd Nanoparticles Supported by Atomically Dispersed Metal Oxide
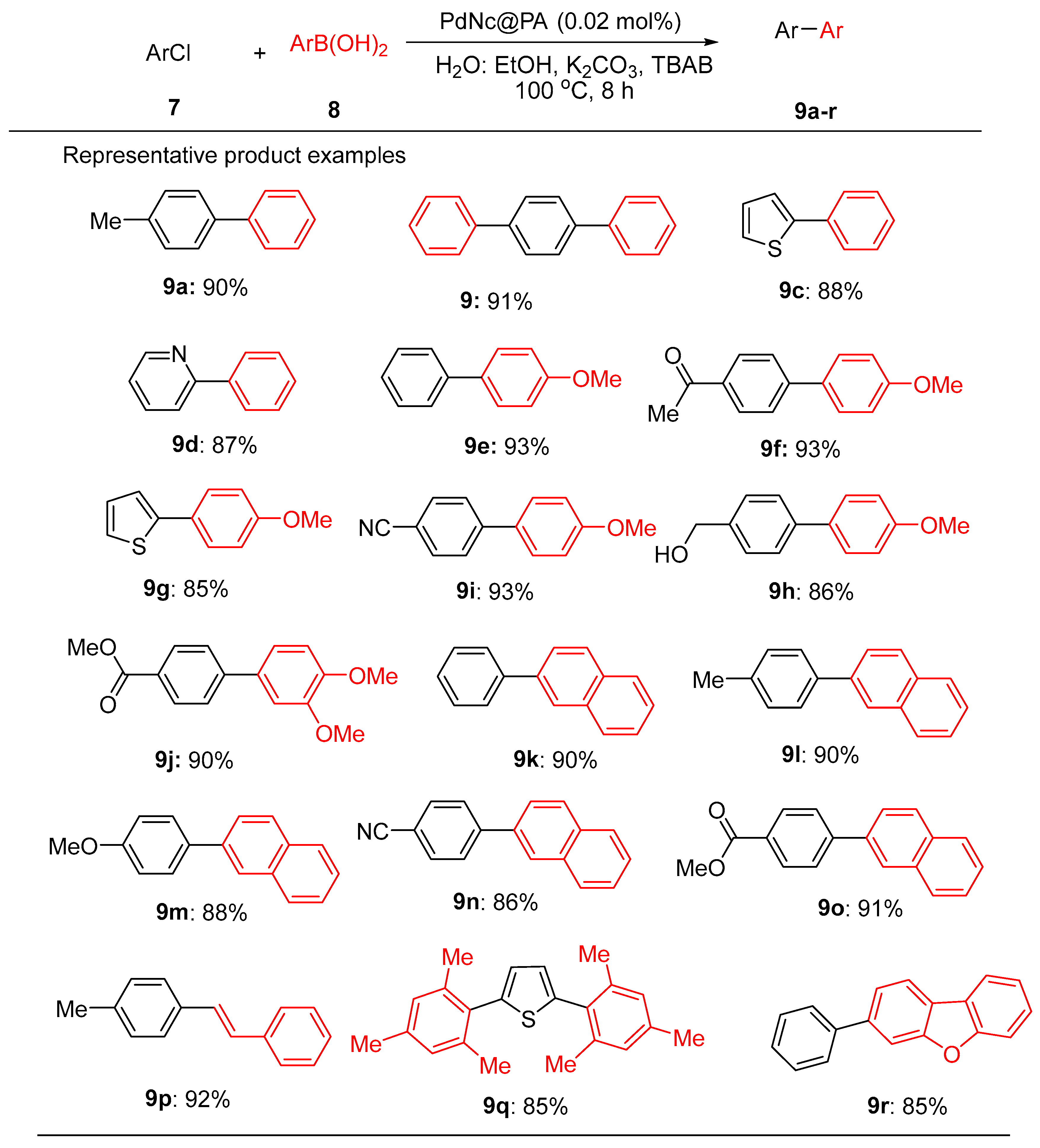
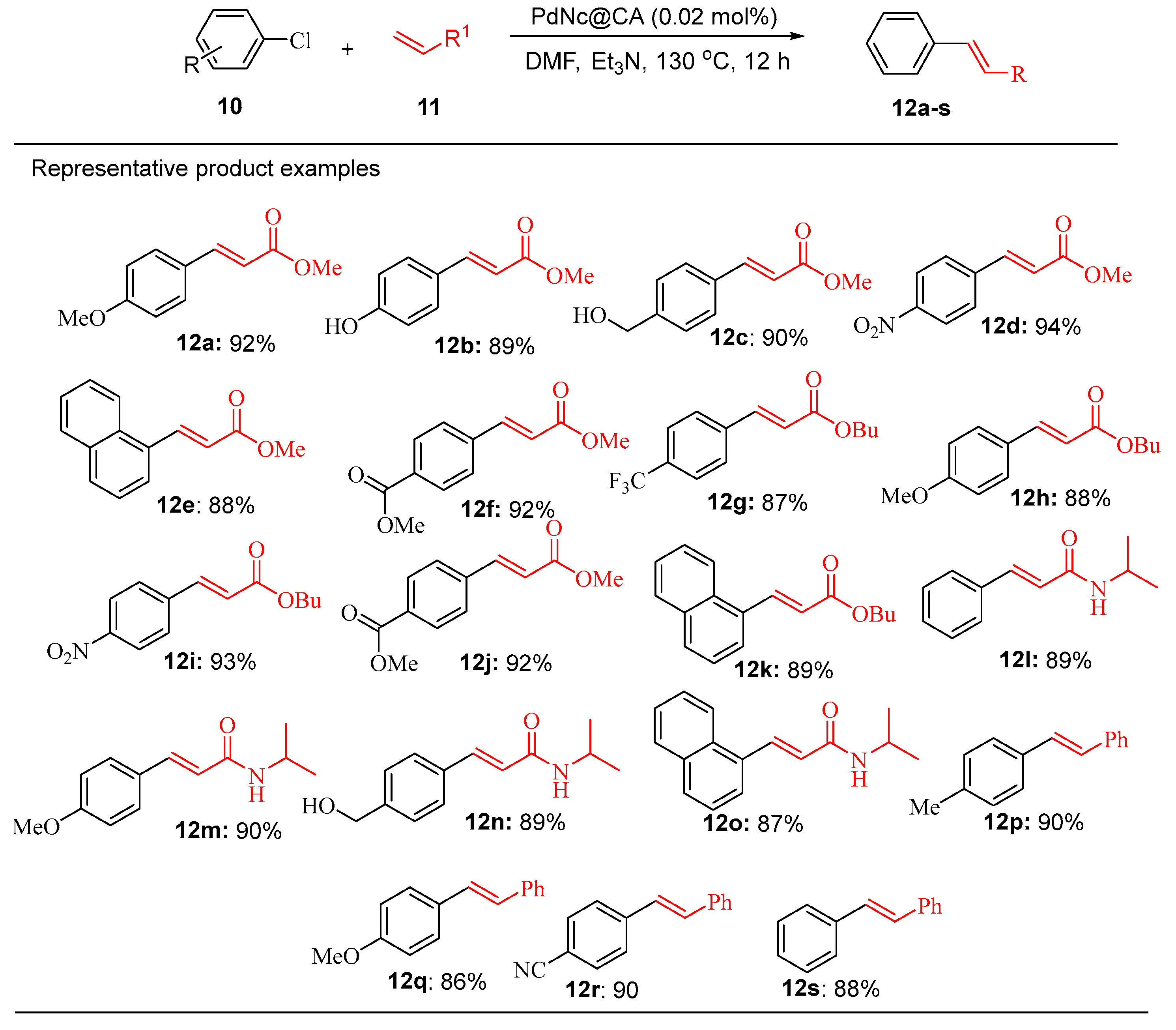
9.3. Palladium Nanoparticles Supported on Solid Matrices
10. Future Directions and Challenges
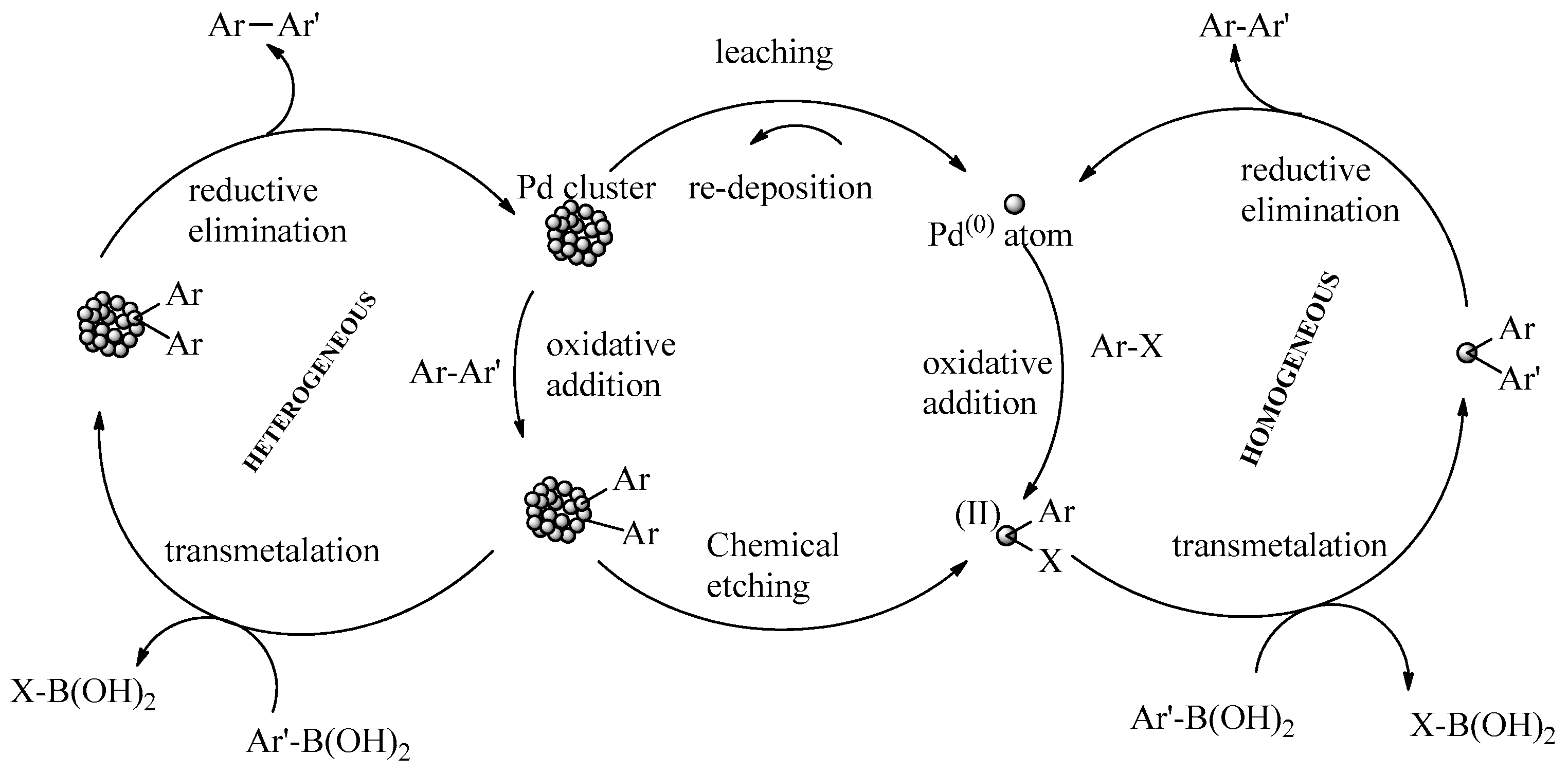
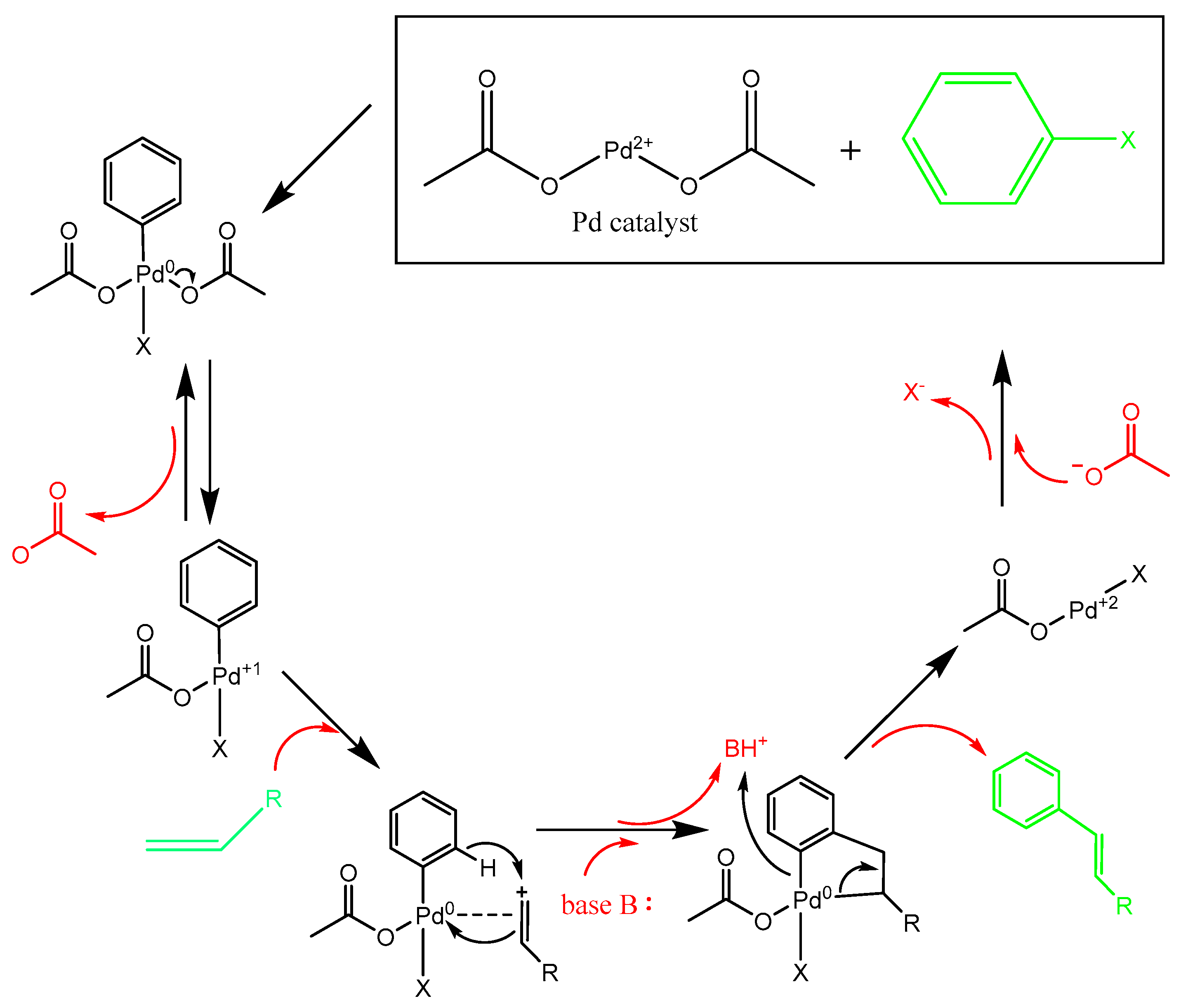
11. Proposed Reaction Mechanisms
12. Conclusion
Funding
Acknowledgments
Conflicts of Interest
References
- Sheldon, R.A., Fundamentals of green chemistry: efficiency in reaction design. Chem. Soc. Rev., 2012. 41(4): p. 1437-1451. [CrossRef]
- Gómez-López, P., et al., Nanomaterials and catalysis for green chemistry. Curr. Opin. Green Sustain. Chem., 2020. 24: p. 48-55. [CrossRef]
- Centi, G. and S. Perathoner, Catalysis and sustainable (green) chemistry. Catal. Today, 2003. 77(4): p. 287-297.
- Turner, N.J., Sustainable catalysis. 2016, Beilstein-Institut. p. 1778-1779. [CrossRef]
- Habib, U., et al., Sustainable Catalysis: Navigating Challenges and Embracing Opportunities for a Greener Future. J. Chem. Environ. 2023. 2(2): p. 14-53. [CrossRef]
- McCarthy, S., D.C. Braddock, and J.D. Wilton-Ely, Strategies for sustainable palladium catalysis. Coord. Chem. Rev. 2021. 442: p. 213925. [CrossRef]
- Fiorio, J.L., et al., Recent advances in the use of nitrogen-doped carbon materials for the design of noble metal catalysts. Coord. Chem. Rev., 2023. 481: p. 215053. [CrossRef]
- Ashraf, M., et al., Transition metal nanoparticles as nanocatalysts for Suzuki, Heck and Sonogashira cross-coupling reactions. Coord. Chem. Rev., 2023. 476: p. 214928. [CrossRef]
- Liu, L. and A. Corma, Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles. Chem. Rev., 2018. 118(10): p. 4981-5079. [CrossRef]
- Hong, K., et al., Palladium nanoparticles on assorted nanostructured supports: applications for Suzuki, Heck, and Sonogashira cross-coupling reactions. ACS Appl. Nano Mater., 2020. 3(3): p. 2070-2103. [CrossRef]
- Swain, S., et al., Computational and experimental design of the octahedral PdFe alloy nanocatalyst for hiyama cross-coupling and environmental pollutant degradation. ACS Appl. Nano Mater., 2023. 6(5): p. 3254-3267. [CrossRef]
- Wang, J., et al., Facile Assembly of C–N Bond-Containing Polymer Electrolytes Enabled by Lithium Salt-Catalyzed Aza-Michael Addition. Macromolecules, 2023. 56(6): p. 2484-2493. [CrossRef]
- Ghorbani-Choghamarani, A., B. Tahmasbi, and P. Moradi, Palladium–S-propyl-2-aminobenzothioate immobilized on Fe3O4 magnetic nanoparticles as catalyst for Suzuki and Heck reactions in water or poly (ethylene glycol). Appl. Organomet. Chem., 2016. 30(6): p. 422-430.
- Aryanasab, F., et al., Immobilizing palladium on melamine-functionalized magnetic nanoparticles: An efficient and reusable phosphine-free catalyst for Mizoroki–Heck reaction. Appl. Organomet. Chem., 2021. 35(5): p. e6198. [CrossRef]
- Wolfson, A. and O. Levy-Ontman, Development and application of palladium nanoparticles on renewable polysaccharides as catalysts for the Suzuki cross-coupling of halobenzenes and phenylboronic acids. Mol. Catal., 2020. 493: p. 111048. [CrossRef]
- Heravi, M.M. and E. Hashemi, Recent applications of the Suzuki reaction in total synthesis. Tetrahedron, 2012. 68(45): p. 9145-9178. [CrossRef]
- Maity, P., et al., Development of a scalable synthesis of BMS-978587 featuring a stereospecific Suzuki coupling of a cyclopropane carboxylic acid. Org. Process Res. Dev., 2018. 22(7): p. 888-897. [CrossRef]
- Sirindil, F., et al., Total synthesis of Rhazinilam through gold-catalyzed cycloisomerization–sulfonyl migration and palladium-catalyzed Suzuki–Miyaura coupling of pyrrolyl sulfonates. Org. Lett., 2019. 21(14): p. 5542-5546. [CrossRef]
- Otte, F. and B. Schmidt, Matsuda–Heck arylation of glycals for the stereoselective synthesis of aryl C-glycosides. J. Org. Chem., 2019. 84(22): p. 14816-14829. [CrossRef]
- Ashworth, I.W., et al., Process development of a Suzuki reaction used in the manufacture of lanabecestat. Org. Process Res. Dev., 2018. 22(12): p. 1801-1808. [CrossRef]
- Luo, J. and J.A. May, Enantioselective Total Synthesis of Cannabinoids via a Tandem Conjugate Addition/Enolate Alkylation Annulation with Ambiphilic Organoboronates. Org. Lett., 2023. 25(4): p. 708-713. [CrossRef]
- Qin, Y., et al., Silacyclization through palladium-catalyzed intermolecular silicon-based C (sp 2)–C (sp 3) cross-coupling. Chem. Sci., 2021. 12(42): p. 14224-14229.
- Paul, D., et al., Intramolecular heck reaction in total synthesis of natural products: an update. Eur. J. Org. Chem., 2021. 2021(14): p. 2057-2076. [CrossRef]
- Stanway-Gordon, H.A., J.A. Odger, and M.J. Waring, Development of a micellar-promoted heck reaction for the synthesis of DNA-encoded libraries. Bioconjugate Chem., 2023. 34(4): p. 756-763. [CrossRef]
- Hayama, N., Y. Kobayashi, and Y. Takemoto, Asymmetric hetero-Michael addition to α, β-unsaturated carboxylic acids using thiourea–boronic acid hybrid catalysts. Tetrahedron, 2021. 89: p. 132089.
- Evano, G., J. Wang, and A. Nitelet, Metal-mediated C–O bond forming reactions in natural product synthesis. Org. Chem. Front., 2017. 4(12): p. 2480-2499. [CrossRef]
- Vásquez-Céspedes, S., et al., Heterogeneous catalysis for cross-coupling reactions: an underutilized powerful and sustainable tool in the fine chemical industry? Org. Process Res. Dev., 2021. 25(4): p. 740-753.
- Hegedus, L.S., Transition metals in the synthesis of complex organic molecules. 1999: University Science Books.
- Wu, T.-C., Process for preparing olefins. 1996, Google Patents.
- Beller, M., A. Zapf, and W. Mägerlein, Efficient Synthesis of Fine Chemicals and Organic Building Blocks Applying Palladium-Catalyzed Coupling Reactions. Chemical Engineering & Technology: Industrial Chemistry-Plant Equipment-Process Engineering-Biotechnology, 2001. 24(6): p. 575-582. [CrossRef]
- Shinkai, I., A. King, and R. Larsen, A practical asymmetric synthesis of LTD4 antagonist. Pure Appl, Chem., 1994. 66(7): p. 1551-1556. [CrossRef]
- Macor, J.E. and M.J. Wythes, Indole derivatives. 1998, Google Patents.
- Shen, H.C., Selected Applications of Transition Metal-Catalyzed Carbon–Carbon Cross-Coupling Reactions in the Pharmaceutical Industry. 2012, Wiley Online Library. p. 25-43.
- Perkins, J., Eur. Patent 1088817 (2001); JE Macor and MJ Wythes, Int. Patent WO 9206973 (1992), to Pfizer Inc.; CQ Meng. Curr. Opin. Cent. Peripher. Nerv. Syst. Invest. Drugs, 2000. 2: p. 186.
- King, A., et al., An efficient synthesis of LTD4 antagonist L-699,392. J. Org. Chem., 1993. 58(14): p. 3731-3735. [CrossRef]
- Hervé, G. and C. Len, First ligand-free, microwave-assisted, Heck cross-coupling reaction in pure water on a nucleoside–Application to the synthesis of antiviral BVDU. RSC Adv., 2014. 4(87): p. 46926-46929.
- Shen, C., et al., A novel D-glucosamine-derived pyridyl-triazole@ palladium catalyst for solvent-free Mizoroki–Heck reactions and its application in the synthesis of Axitinib. Green Chem., 2015. 17(1): p. 225-230. [CrossRef]
- Herkes, F.E., Catalysis of organic reactions. Vol. 75. 1998: CRC Press.
- Polshettiwar, V. and T.N. Asefa, Synthesis and Applications. 2013: Wiley Online Library.
- Luo, S., et al., Perspectives on palladium-based nanomaterials: green synthesis, ecotoxicity, and risk assessment. Environ. Sci. Nano, 2021. 8(1): p. 20-36. [CrossRef]
- Piermatti, O., Green synthesis of Pd nanoparticles for sustainable and environmentally benign processes. Catalysts, 2021. 11(11): p. 1258. [CrossRef]
- Law, C.K.Y., et al., Biogenic synthesis of palladium nanoparticles: New production methods and applications. Nanotechnol. Rev., 2022. 11(1): p. 3104-3124. [CrossRef]
- Kamel, S. and T.A. Khattab, Recent advances in cellulose supported metal nanoparticles as green and sustainable catalysis for organic synthesis. Cellulose, 2021. 28(8): p. 4545-4574. [CrossRef]
- Ayogu, J.I. and E.A. Onoabedje, Prospects and Applications of Palladium Nanoparticles in the Cross-coupling of (hetero) aryl Halides and Related Analogues. Chem. Open, 2021. 10(4): p. 430-450. [CrossRef]
- Aksoy, M., et al., Recent advances in the development of palladium nanocatalysts for sustainable organic transformations. Inorg. Chem. Front., 2021. 8(2): p. 499-545. [CrossRef]
- Srivastava, A., et al., Optimal exploitation of supported heterogenized Pd nanoparticles for CC cross-coupling reactions. Coord. Chem. Rev., 2024. 507: p. 215763. [CrossRef]
- Palem, R.R., et al., Biogenic palladium nanoparticles: An effectual environmental benign catalyst for organic coupling reactions. J. Ind. Eng. Chem., 2022. 106: p. 52-68. [CrossRef]
- Dong, Z., et al., Palladium supported on urea-containing porous organic polymers as heterogeneous catalysts for C–C cross coupling reactions and reduction of nitroarenes. J. Saudi Chem. Soc., 2021. 25(9): p. 101317. [CrossRef]
- Van Vaerenbergh, B., et al., Towards high-performance heterogeneous palladium nanoparticle catalysts for sustainable liquid-phase reactions. React. Chem. Eng., 2020. 5(9): p. 1556-1618.
- Xiao, Q., et al., Visible light-driven cross-coupling reactions at lower temperatures using a photocatalyst of palladium and gold alloy nanoparticles. ACS Catal., 2014. 4(6): p. 1725-1734. [CrossRef]
- Hamdi, J., et al., Room-temperature aqueous Suzuki–Miyaura cross-coupling reactions catalyzed via a recyclable palladium@ halloysite nanocomposite. Org. Lett., 2019. 21(10): p. 3471-3475. [CrossRef]
- Gautam, P., et al., KCC-1 supported palladium nanoparticles as an efficient and sustainable nanocatalyst for carbonylative Suzuki–Miyaura cross-coupling. Green Chem., 2016. 18(21): p. 5890-5899. [CrossRef]
- Doherty, S., et al., Heteroatom donor-decorated polymer-immobilized ionic liquid stabilized palladium nanoparticles: efficient catalysts for room-temperature Suzuki-Miyaura cross-coupling in aqueous media. Adv. Synth. Catal., 2018. 360(19): p. 3716-3731.
- Rezapour, E., M. Jafarpour, and A. Rezaeifard, Palladium niacin complex immobilized on starch-coated maghemite nanoparticles as an efficient homo-and cross-coupling catalyst for the synthesis of symmetrical and unsymmetrical biaryls. Catal. Lett., 2018. 148: p. 3165-3177. [CrossRef]
- Astruc, D., Palladium nanoparticles as efficient green homogeneous and heterogeneous carbon− carbon coupling precatalysts: A unifying view. Inorg. Chem., 2007. 46(6): p. 1884-1894. [CrossRef]
- Ricciardi, R., J. Huskens, and W. Verboom, Dendrimer-encapsulated Pd nanoparticles as catalysts for C–C cross-couplings in flow microreactors. Org. Biomol. Chem., 2015. 13(17): p. 4953-4959. [CrossRef]
- Ricciardi, R., et al., Dendrimer-Encapsulated Palladium Nanoparticles for Continuous-Flow Suzuki–Miyaura Cross-Coupling Reactions. ChemCatChem, 2015. 7(6): p. 936-942. [CrossRef]
- Benyettou, F., et al., Palladium-Loaded Cucurbit [7] uril-Modified Iron Oxide Nanoparticles for C− C Cross-Coupling Reactions. Chem. Eur. J., 2018. 24(10): p. 2349-2353. [CrossRef]
- Cui, S., et al., Pd Nanoparticles Immobilized on Pyridinic N-Rich Carbon Nanosheets for Promoting Suzuki Cross-Coupling Reactions. Nanomater., 2024. 14(21): p. 1690. [CrossRef]
- Chandrashekharan, B., et al., Palladium nanoparticle immobilized on coconut coir extract coated boron carbon nitride: A green and sustainable nanocatalyst for cross-coupling reactions and HER studies. Diamond Relat. Mater., 2024: p. 111261. [CrossRef]
- Islam, M.S., et al., Applications of bio-resource based sustainable heterogeneous Pd-Nanocatalyst for cross-coupling and michael addition reactions. Chem. Eng. J., 2024. 483: p. 149271.
- Rybochkin, P.V., et al., Aerobic bacteria-supported biohybrid palladium catalysts for efficient cross-coupling reactions. J. Catal., 2024. 429: p. 115238. [CrossRef]
- Campelo, J.M., et al., Sustainable preparation of supported metal nanoparticles and their applications in catalysis. ChemSusChem, 2009. 2(1): p. 18-45. [CrossRef]
- Karimi-Maleh, H., et al., Recent advances in using of chitosan-based adsorbents for removal of pharmaceutical contaminants: A review. J. Clean. Prod., 2021. 291: p. 125880. [CrossRef]
- Naghipour, A. and A. Fakhri, Heterogeneous Fe3O4@ chitosan-Schiff base Pd nanocatalyst: Fabrication, characterization and application as highly efficient and magnetically-recoverable catalyst for Suzuki–Miyaura and Heck–Mizoroki C–C coupling reactions. Catal. Commun., 2016. 73: p. 39-45. [CrossRef]
- Sinha, V., et al., Chitosan microspheres as a potential carrier for drugs. Int. J. Pharm., 2004. 274(1-2): p. 1-33. [CrossRef]
- Bao, Y., et al., Cobalt, nickel and iron embedded chitosan microparticles as efficient and reusable catalysts for Heck cross-coupling reactions. Int. J. Biol. Macromol., 2019. 130: p. 203-212. [CrossRef]
- Hajipour, A.R. and Z. Tavangar-Rizi, Methionine-functionalized chitosan–Pd (0) complex: A novel magnetically separable catalyst for Heck reaction of aryl iodides and aryl bromides at room temperature in water as only solvent. Appl. Organomet. Chem., 2017. 31(7): p. e3638. [CrossRef]
- Jadhav, S., A. Kumbhar, and R. Salunkhe, Palladium supported on silica–chitosan hybrid material (Pd-CS@ SiO2) for Suzuki–Miyaura and Mizoroki–Heck cross-coupling reactions. Appl. Organomet. Chem., 2015. 29(6): p. 339-345.
- Orooji, Y., et al., Polysaccharide-based (nano) materials for Cr (VI) removal. Int. J. Biol. Macromol., 2021. 188: p. 950-973. [CrossRef]
- Baran, T. and M. Nasrollahzadeh, Facile synthesis of palladium nanoparticles immobilized on magnetic biodegradable microcapsules used as effective and recyclable catalyst in Suzuki-Miyaura reaction and p-nitrophenol reduction. Carbohydr. Polym., 2019. 222: p. 115029. [CrossRef]
- Guibal, E., Heterogeneous Catalysis on Chitosan-Based Materials. ChemInform, 2006. 37(2): p. no-no. [CrossRef]
- Leonhardt, S.E., et al., Chitosan as a support for heterogeneous Pd catalysts in liquid phase catalysis. Appl Catal., A, 2010. 379(1-2): p. 30-37. [CrossRef]
- Zheng, X., et al., Preparation of porous chitosan/reduced graphene oxide microspheres supported Pd nanoparticles catalysts for Heck coupling reactions. Carbohydr. Polym., 2020. 230: p. 115583. [CrossRef]
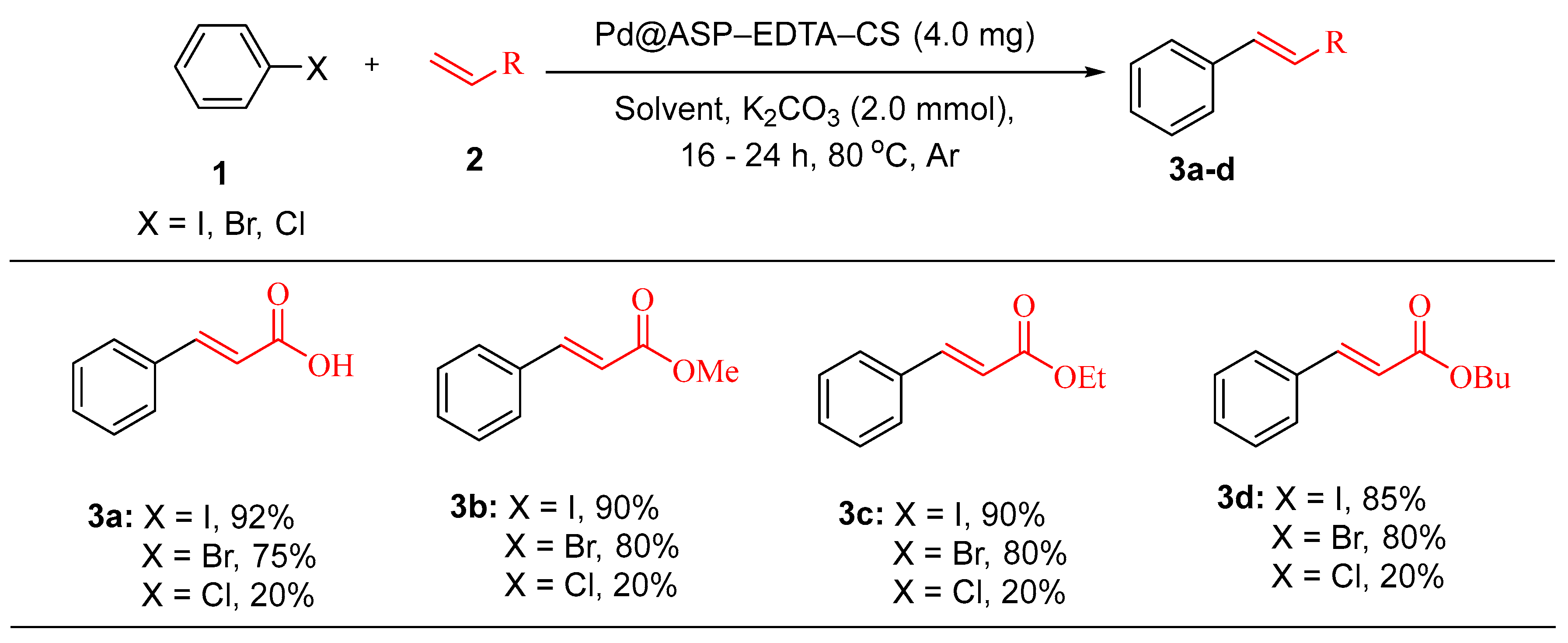
- Dohendou, M., M.G. Dekamin, and D. Namaki, Pd@ l-asparagine–EDTA–chitosan: a highly effective and reusable bio-based and biodegradable catalyst for the Heck cross-coupling reaction under mild conditions. Nanoscale Adv., 2023. 5(9): p. 2621-2638. [CrossRef]
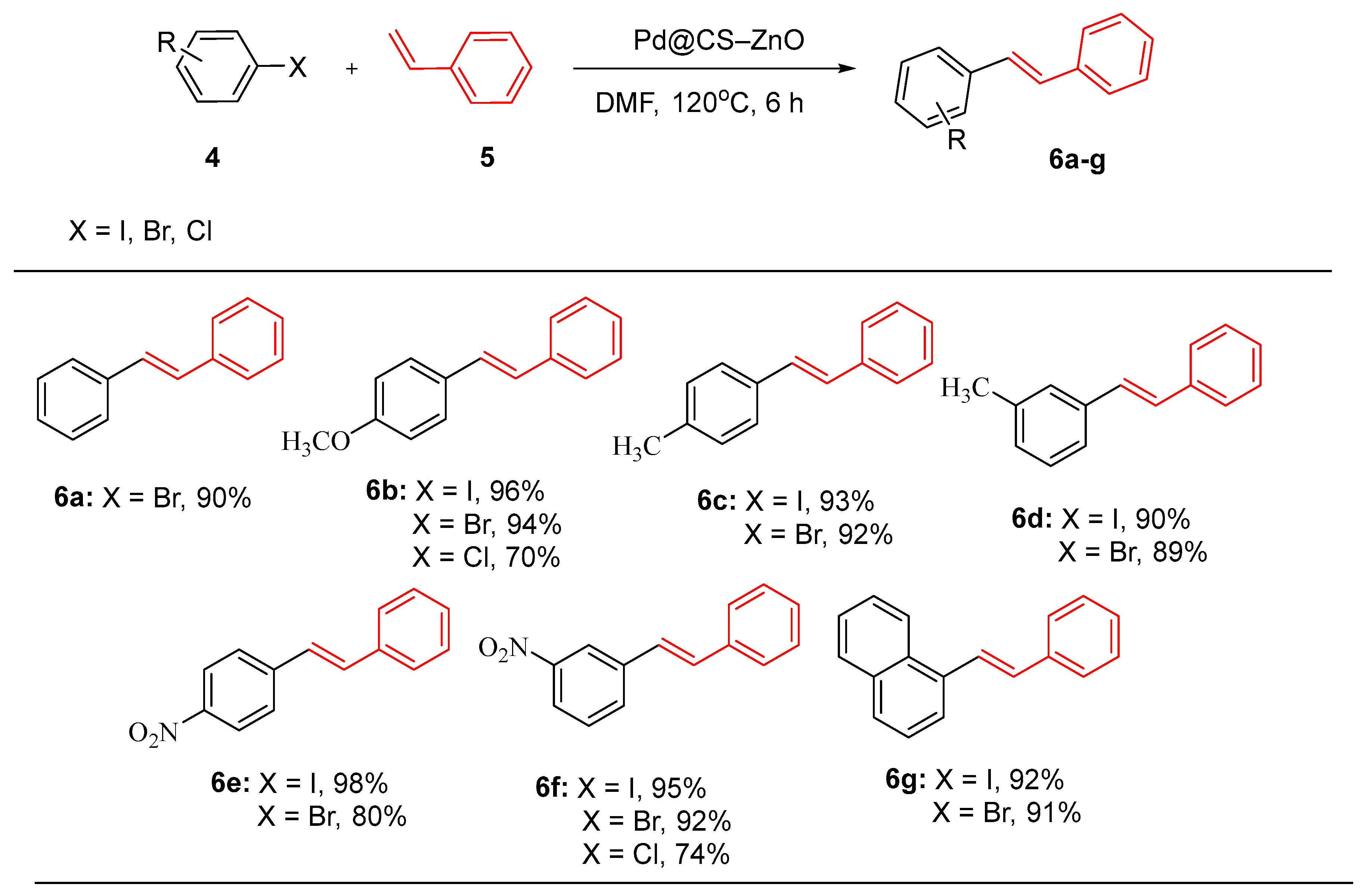
- Baran, N.Y., T. Baran, and M. Nasrollahzadeh, Synthesis of palladium nanoparticles stabilized on Schiff base-modified ZnO particles as a nanoscale catalyst for the phosphine-free Heck coupling reaction and 4-nitrophenol reduction. Sci. Rep., 2023. 13(1): p. 12008. [CrossRef]
- Çalışkan, M. and T. Baran, Nanoscaled reusable palladium catalyst supported on chitosan hybrid composite microcapsules reinforced with ZnO nanoparticles for Heck coupling reactions. Cellulose, 2022. 29(14): p. 7789-7802.
- Albano, G., A. Petri, and L.A. Aronica, Palladium Supported on Bioinspired Materials as Catalysts for C–C Coupling Reactions. Catalysts, 2023. 13(1): p. 210. [CrossRef]
- Dahl, J.A., B.L. Maddux, and J.E. Hutchison, Toward greener nanosynthesis. Chem. Rev., 2007. 107(6): p. 2228-2269. [CrossRef]
- Astruc, D., F. Lu, and J.R. Aranzaes, Nanoparticles as recyclable catalysts: the frontier between homogeneous and heterogeneous catalysis. Angew. Chem. Intl Ed., 2005. 44(48): p. 7852-7872. [CrossRef]
- Panziera, N., et al., MVS-derived palladium nanoparticles deposited on polydimethylphosphazene as recyclable catalysts for Heck-type reactions: Preparation, structural study, and catalytic activity. J. Catal., 2007. 246(2): p. 351-361. [CrossRef]
- Caporusso, A.M., et al., Functional resins in palladium catalysis: promising materials for Heck reaction in aprotic polar solvents. J. Catal., 2005. 234(1): p. 1-13. [CrossRef]
- Boisselier, E., et al., Encapsulation and stabilization of gold nanoparticles with “click” polyethyleneglycol dendrimers. J. Am. Chem. Soc., 2010. 132(8): p. 2729-2742. [CrossRef]
- Wu, Z., et al., Immobilization of Pd (0) nanoparticles on polyaza-ligand-functionalized polyacrylonitrile fiber as highly active catalyst for the Heck reaction. J. Catal., 2023. 427: p. 115121. [CrossRef]
- Cao, Y., et al., Metal/porous carbon composites for heterogeneous catalysis: old catalysts with improved performance promoted by N-doping. ACS Catal., 2017. 7(12): p. 8090-8112. [CrossRef]
- Xu, X., et al., Synthesis of palladium nanoparticles supported on mesoporous N-doped carbon and their catalytic ability for biofuel upgrade. J. Am. Chem. Soc., 2012. 134(41): p. 16987-16990. [CrossRef]
- Gao, Y., et al., Nitrogen-Doped sp 2-Hybridized Carbon as a Superior Catalyst for Selective Oxidation. Angew. Chem. Intl. Ed., 2013. 52(7).
- Chen, S., et al., Platinum nanoparticles supported on N-doped carbon nanotubes for the selective oxidation of glycerol to glyceric acid in a base-free aqueous solution. RSC Adv., 2015. 5(40): p. 31566-31574. [CrossRef]
- Zhang, L., et al., N-Doped porous carbon supported palladium nanoparticles as a highly efficient and recyclable catalyst for the Suzuki coupling reaction. Chin. Chem. Lett., 2016. 27(1): p. 149-154. [CrossRef]
- Joshani, Z., et al., Palladium nanoparticles (Pd NPs) immobilized over polydopamine-functionalized Zn-Al mixed metal oxide as a novel nanocatalyst for synthesis of CC bond via Suzuki-Miyaura coupling reactions. Inorg. Chem. Commun., 2024. 163: p. 112308. [CrossRef]
- Dory, H., et al., Monolith Catalyst Design by Combining 3D Printing and Atomic Layer Deposition: Toward Green Palladium-Catalyzed Cross-Coupling Reactions. Adv. Eng. Mater., 2024: p. 2401546. [CrossRef]
- Abdellah, A.R., et al., Palladium nanocrystals-embedded covalent organic framework as an efficient catalyst for Heck cross-coupling reaction. Microporous and Mesoporous Mater., 2022. 339: p. 111961. [CrossRef]
- Zhou, J. and D. Astruc, Recent Trends and Perspectives in Palladium Nanocatalysis: From Nanoparticles to Frameworks, Atomically Precise Nanoclusters and Single-Atom Catalysts. J. Inorg. Organomet. Polym. Mater., 2024: p. 1-23. [CrossRef]
- Salemi, H., et al., Covalent organic framework supported palladium catalysts. J. Mate. Chem., A, 2022. 10(39): p. 20707-20729. [CrossRef]
- Dong, Z., et al., Palladium Immobilized on a Polyimide Covalent Organic Framework: An Efficient and Recyclable Heterogeneous Catalyst for the Suzuki–Miyaura Coupling Reaction and Nitroarene Reduction in Water. Catal. Lett., 2022. 152(1): p. 299-306. [CrossRef]
- Shukla, F., et al., Palladium nanoparticles-confined pore-engineered urethane-linked thiol-functionalized covalent organic frameworks: a high-performance catalyst for the Suzuki Miyaura cross-coupling reaction. Dalton Trans., 2023. 52(8): p. 2518-2532. [CrossRef]
- Xiao, S., et al., Ultrahigh stable covalent organic framework-derived carbon–nitrogen-supported palladium nanoparticles for highly efficient electrocatalytic methanol and ethanol oxidation reactions. Green Chem., 2022. 24(15): p. 5813-5821. [CrossRef]
- Zhu, Q.-L. and Q. Xu, Metal–organic framework composites. Chem. Soc. Rev., 2014. 43(16): p. 5468-5512.
- Luo, S., et al., Metal organic frameworks as robust host of palladium nanoparticles in heterogeneous catalysis: synthesis, application, and prospect. ACS Appl. Mater. Interfaces, 2019. 11(36): p. 32579-32598. [CrossRef]
- Hajek, J., et al., Mechanistic studies of aldol condensations in UiO-66 and UiO-66-NH2 metal organic frameworks. J. Catal., 2015. 331: p. 1-12. [CrossRef]
- Zhang, Q., et al., Covalent construction of sustainable hybrid UiO-66-NH2@ Tb-CP material for selective removal of dyes and detection of metal ions. ACS Sustainable chem. Eng., 2019. 7(3): p. 3203-3212. [CrossRef]
- Mohammadi, L. and M.R. Vaezi, Palladium Nanoparticle-Decorated Porous Metal–Organic-Framework (Zr)@ Guanidine: Novel Efficient Catalyst in Cross-Coupling (Suzuki, Heck, and Sonogashira) Reactions and Carbonylative Sonogashira under Mild Conditions. ACS Omega, 2023. 8(18): p. 16395-16410.
- Yuan, S., et al., Stable metal–organic frameworks with group 4 metals: current status and trends. ACS Cent. Sci., 2018. 4(4): p. 440-450. [CrossRef]
- Wang, Q. and D. Astruc, State of the art and prospects in metal–organic framework (MOF)-based and MOF-derived nanocatalysis. Chem. Rev., 2019. 120(2): p. 1438-1511. [CrossRef]
- Stock, N. and S. Biswas, Synthesis of metal-organic frameworks (MOFs): routes to various MOF topologies, morphologies, and composites. Chem. Rev., 2012. 112(2): p. 933-969. [CrossRef]
- Bai, Y., et al., Zr-based metal–organic frameworks: design, synthesis, structure, and applications. Chem. Soc. Rev., 2016. 45(8): p. 2327-2367.
- D’Amato, R., et al., “Shake ‘n Bake” Route to Functionalized Zr-UiO-66 Metal–Organic Frameworks. Inorg. Chem., 2021. 60(18): p. 14294-14301.
- Hendrickx, K., et al., Understanding intrinsic light absorption properties of UiO-66 frameworks: a combined theoretical and experimental study. Inorg. Chem., 2015. 54(22): p. 10701-10710. [CrossRef]
- Katz, M.J., et al., A facile synthesis of UiO-66, UiO-67 and their derivatives. Chem. Commun., 2013. 49(82): p. 9449-9451. [CrossRef]
- Hajighasemi, Z., et al., Efficient and biocompatible new palladium-supported boehmite nanoparticles: synthesis, characterization and application in Suzuki–Miura and Mizoroki–Heck coupling reactions. Nanoscale Adv., 2023. 5(18): p. 4925-4933. [CrossRef]
- Majhi, S., et al., Palladium nanoparticles on polydimethylsiloxane film for C− C coupling reactions, and catalytic reduction of organic pollutants in water. ChemistrySelect, 2023. 8(42): p. e202303057.
- Gupta, P.K., N. Kumar, and K.V. Ranganath, Asymmetric Suzuki Cross-Coupling Reactions Catalyzed by Chiral Surfactant-Stabilized Palladium Nanoparticles. Asian J. Org. Chem., 2022. 11(8): p. e202200362. [CrossRef]
- Wang, J., et al., Pd nanoparticles supported in PDMAEMA-b-PLMA micelles: A superb catalytic platform for Suzuki-Miyaura cross-coupling in water. Eur. Polym. J., 2024. 202: p. 112650. [CrossRef]
- Shi, W., et al., Nano-sized alumina supported palladium catalysts for methane combustion with excellent thermal stability. J. Environ. Sci., 2023. 126: p. 333-347. [CrossRef]
- Kalishyn, Y.Y., et al., Synthesis and Thermal Stability of Palladium Nanoparticles Supported on γ-Al2O3. Curr. Nanomater., 2020. 5(1): p. 79-90.
- Ghorbani-Choghamarani, A., B. Tahmasbi, and P. Moradi, Synthesis of a new Pd (0)-complex supported on boehmite nanoparticles and study of its catalytic activity for Suzuki and Heck reactions in H 2 O or PEG. RSC Adv., 2016. 6(49): p. 43205-43216. [CrossRef]
- Liu, J., X. Liao, and B. Shi, Pd nanoparticles immobilized on boehmite by using tannic acid as structure-directing agent and stabilizer: a high performance catalyst for hydrogenation of olefins. Res. Chem. Intermed., 2014. 40: p. 249-258. [CrossRef]
- Tahmasbi, B., M. Darabi, and M. Nikoorazm, A new Schiff-base complex of palladium nanoparticles on modified boehmite with di (pyridin-2-yl) methanone as a robust, reusable, and selective nanocatalyst in the C-C coupling reaction. Appl. Organomet. Chem., 2024. 38(3): p. e7348.
- Wu, S., et al., Micelle-Derived palladium nanoparticles for suzuki–miyaura coupling reactions in water at room temperature. ACS Appl. Nano Mater., 2023. 6(3): p. 1592-1602. [CrossRef]
- Nalakath, T.A.A. (2022), Sustainable nanocatalysis in water for C–C and C–N Cross–Couplings [Doctoral Dissertation, The Univerdsity of Louisville's].
- Mastalir, Á. and Á. Molnár, Palladium Nanoparticles Supported on Porous Silica Materials as Heterogeneous Catalysts of C− C Coupling and Cross-Coupling Reactions. ChemCatChem, 2023. 15(19): p. e202300643.
- Kumbhar, A., Palladium catalyst supported on zeolite for cross-coupling reactions: an overview of recent advances. Top. Curr. Chem., 2017. 375(1): p. 2. [CrossRef]
- Dutta, P. and A. Sarkar, Palladium Nanoparticles Immobilized on Chemically Modified Silica Gel: Efficient Heterogeneous Catalyst for Suzuki, Stille and Sonogashira Cross-Coupling Reactions. Adv. Synth. Catal., 2011. 353(14-15): p. 2814-2822. [CrossRef]
- Saleh, M.Y., et al., Palladium fabricated on Fe3O4 as an organic-inorganic hybrid nanocatalyst for the Suzuki and Stille coupling reactions. J. Mol. Struct., 2025. 1321: p. 139597. [CrossRef]
- Wilson, K.A., L.A. Picinich, and A.R. Siamaki, Nickel–palladium bimetallic nanoparticles supported on multi-walled carbon nanotubes; versatile catalyst for Sonogashira cross-coupling reactions. RSC Adv., 2023. 13(12): p. 7818-7827. [CrossRef]
- Pocklanová, R., et al., Nanodiamond Supported Ultra-Small Palladium Nanoparticles as an Efficient Catalyst for Suzuki Cross-Coupling Reactions. Catalysts, 2024. 14(1): p. 53. [CrossRef]
- Chen, Y., et al., Dynamic structural evolution of MgO-supported palladium catalysts: from metal to metal oxide nanoparticles to surface then subsurface atomically dispersed cations. Chem. Sci., 2024. 15(17): p. 6454-6464.
- Kumar, M., M.S. Solanki, and S. Sharma, Graphene Supported Palladium Nanocatalyst for Cross-Coupling Reactions, in Graphene-based Carbocatalysis: Synthesis, Properties and Applications: Volume 1. 2023, Bentham Science Publishers. p. 263-296.
- Fatahi, Y., et al., Palladium supported aminobenzamide modified silica coated superparamagnetic iron oxide as an applicable nanocatalyst for Heck cross-coupling reaction. J. Organomet. Chem., 2021. 936: p. 121711. [CrossRef]
- Tran, T.P.N., et al., Tailoring graphene oxide framework with N-and S-containing organic ligands for the confinement of Pd nanoparticles towards recyclable catalyst systems. Catal. Lett., 2021. 151: p. 247-254. [CrossRef]
- Lawrence, A.S., et al., Palladium-Based Metal Organic Frameworks as Heterogeneous Catalysts for C− C Couplings. ChemCatChem, 2022. 14(18): p. e202200403.
- Monteil, C., et al., Phosphonated Polyethylenimine Maghemite Nanoparticles: A Convenient Support of Palladium for Cross-Coupling Reactions. Organics, 2022. 3(4): p. 491-501. [CrossRef]
- Pérez-Lorenzo, M., Palladium nanoparticles as efficient catalysts for Suzuki cross-coupling reactions. The Journal of Physical Chem. Lett., 2012. 3(2): p. 167-174. [CrossRef]
- Costa, P., D. Sandrin, and J.C. Scaiano, Real-time fluorescence imaging of a heterogeneously catalysed Suzuki–Miyaura reaction. Nat. Catal., 2020. 3(5): p. 427-437. [CrossRef]
- Trzeciak, A. and A. Augustyniak, The role of palladium nanoparticles in catalytic C–C cross-coupling reactions. Coord. Chem. Rev., 2019. 384: p. 1-20. [CrossRef]
- Kempasiddaiah, M., et al., Palladium-catalyzed denitrogenative cross-coupling of aryl halides with arylhydrazines under mild reaction conditions. Transition Met. Chem., 2021. 46: p. 273-281. [CrossRef]
- Wang, Y., et al., Zeolite-enhanced sustainable Pd-catalyzed C–C cross-coupling reaction: Controlled release and capture of palladium. ACS Appl. Mater. Interfaces, 2020. 12(10): p. 11419-11427. [CrossRef]
- Zhou, T. and M. Szostak, Palladium-catalyzed cross-couplings by C–O bond activation. Catal. Sci. & technology, 2020. 10(17): p. 5702-5739. [CrossRef]
- Sengupta, T., et al., Experimental and computational study of the catalytic activity of Pd and PdCu nanoparticle catalysts and clusters supported on reduced graphene oxide and graphene acid for the suzuki cross-coupling reaction. Appl. Catal., A, 2023. 667: p. 119448. [CrossRef]
- Choudhary, A., et al., Pd nanoparticles supported on magnetic CoTiMgLDH with enhanced catalytic activity in Suzuki cross-coupling and reductive degradation of dyes. Appl. Organomet. Chem., 2023. 37(7): p. e7112. [CrossRef]
- Balanta, A., C. Godard, and C. Claver, Pd nanoparticles for C–C coupling reactions. Chem. Soc. Rev., 2011. 40(10): p. 4973-4985.
- Mpungose, P.P., et al., The current status of heterogeneous palladium catalysed Heck and Suzuki cross-coupling reactions. Molecules, 2018. 23(7): p. 1676. [CrossRef]

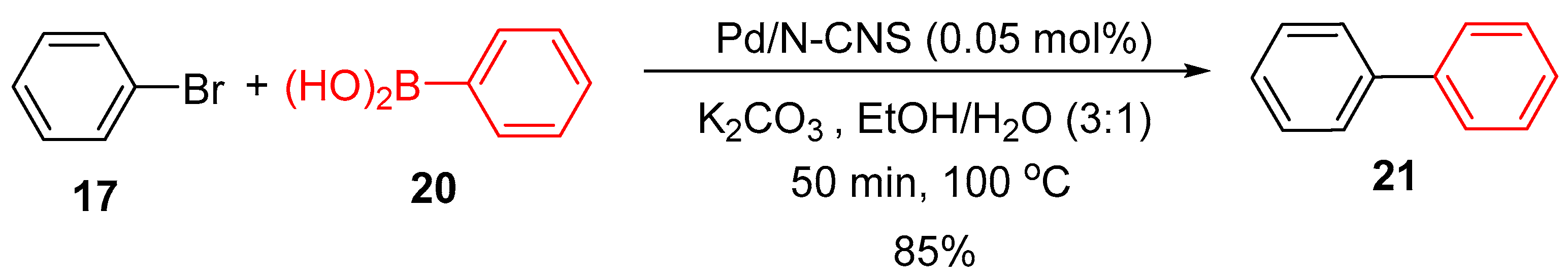
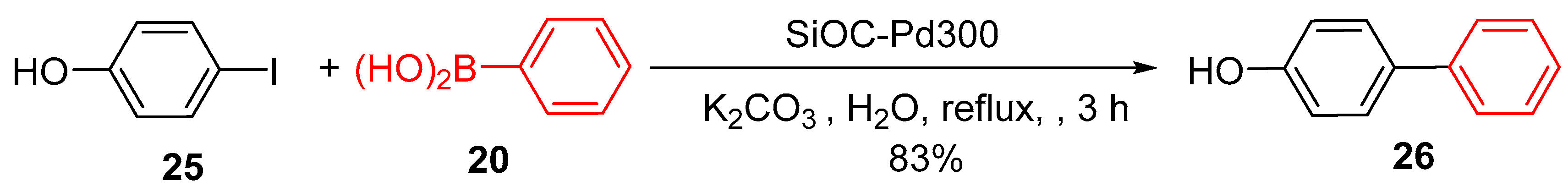
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



