Submitted:
13 January 2025
Posted:
14 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Apparatus
2.3. Drugs
2.4. Catheter Implantation Surgery
2.5. METH Self-Administration and Assessment of Individual Demand for METH
2.6. Tissue Collection, RNA Extraction, and Quality Control
2.7. Library Construction and Sequencing
2.8. Transcriptome Assembly and Bioinformatics Analysis
2.9. Behavioral Data Analyses
2.10. Proof of Concept: Individual Effects
3. Results
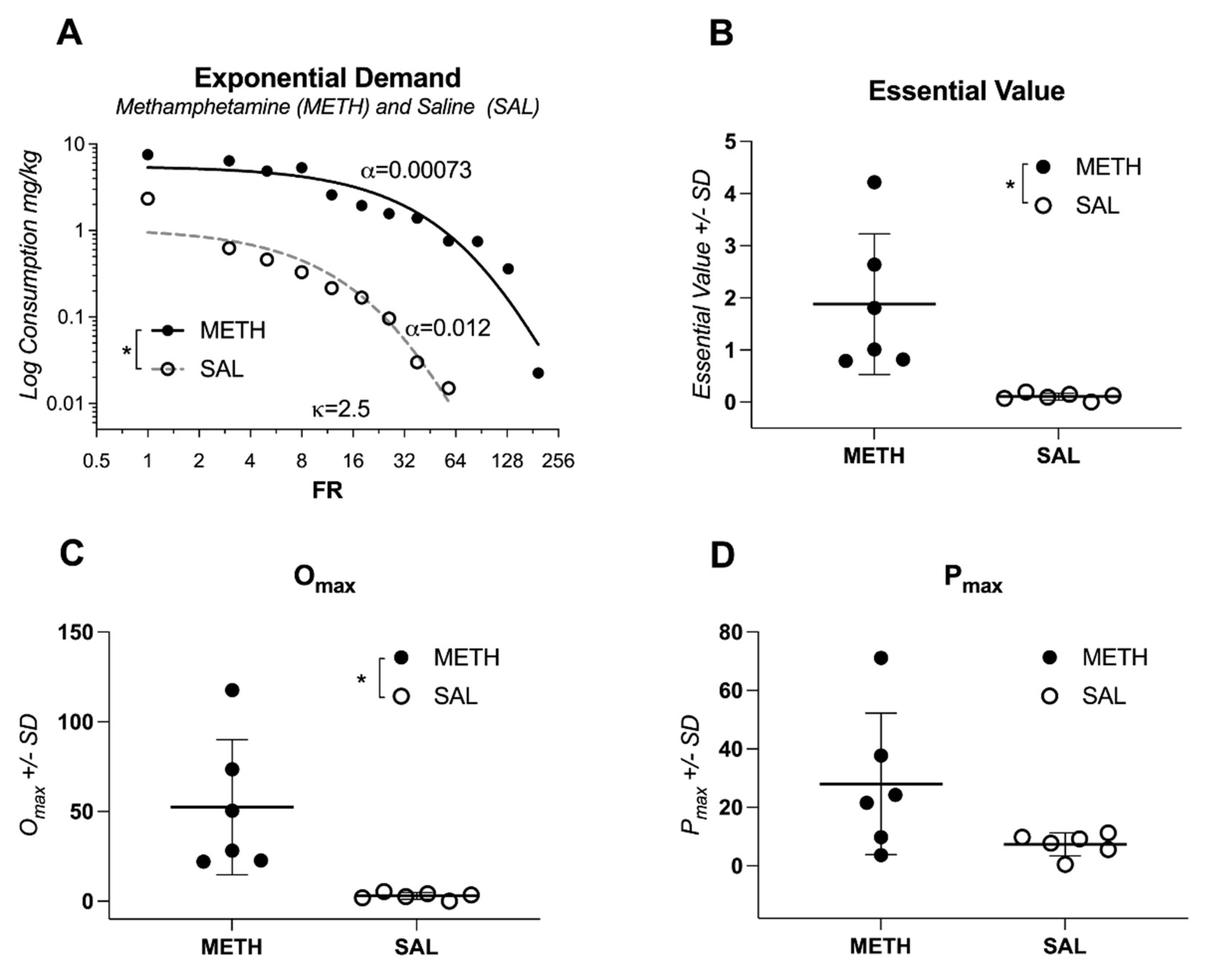
3.1. Acquisition and Economic Demand Analysis of METH Self-Administration
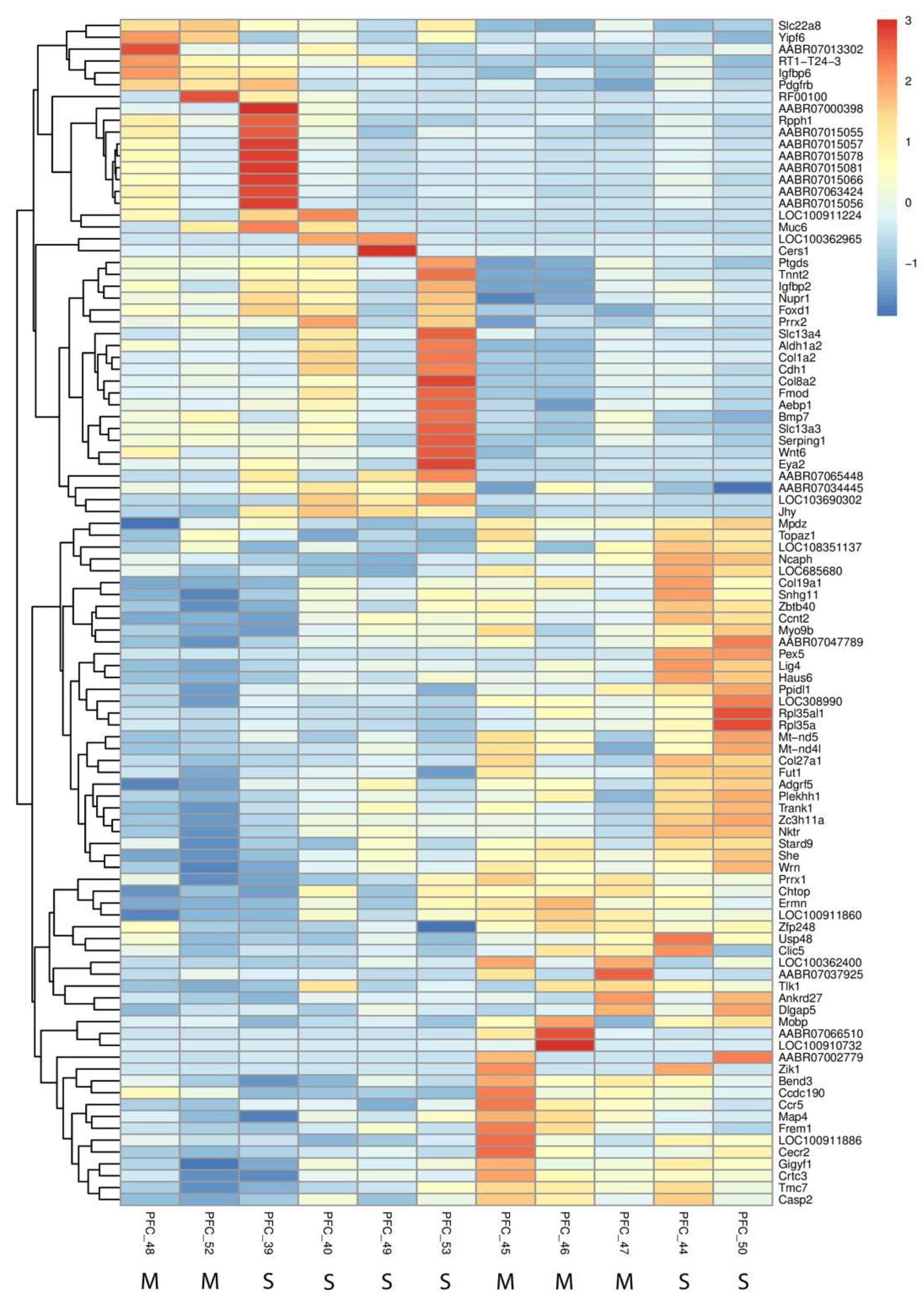
3.2. Transcriptomic Changes in the PFC: Differential Gene Expression
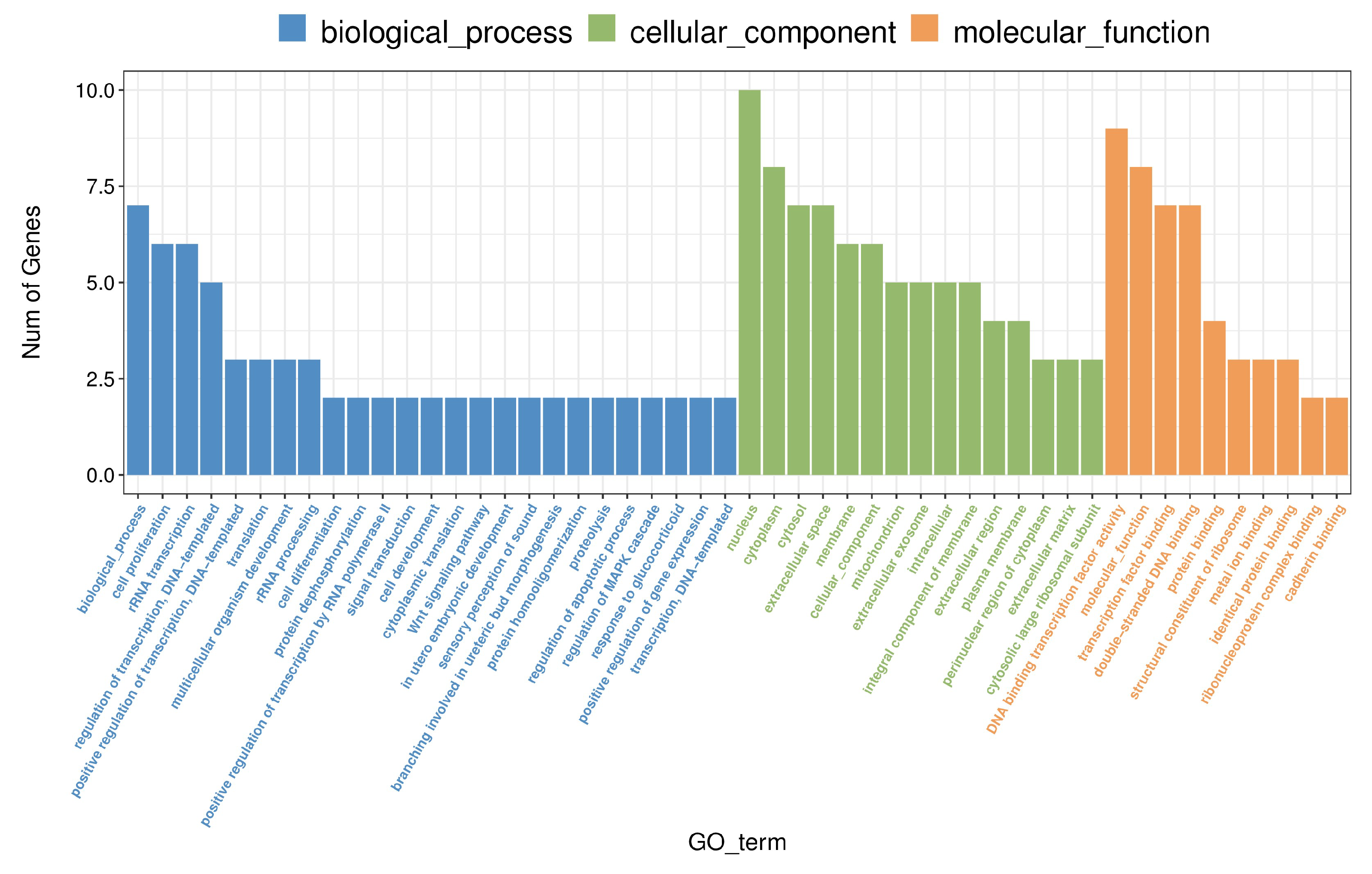
3.3. Differential Gene Expression in the Prefrontal Cortex: Insights from Gene Ontology Enrichment Following Methamphetamine Exposure
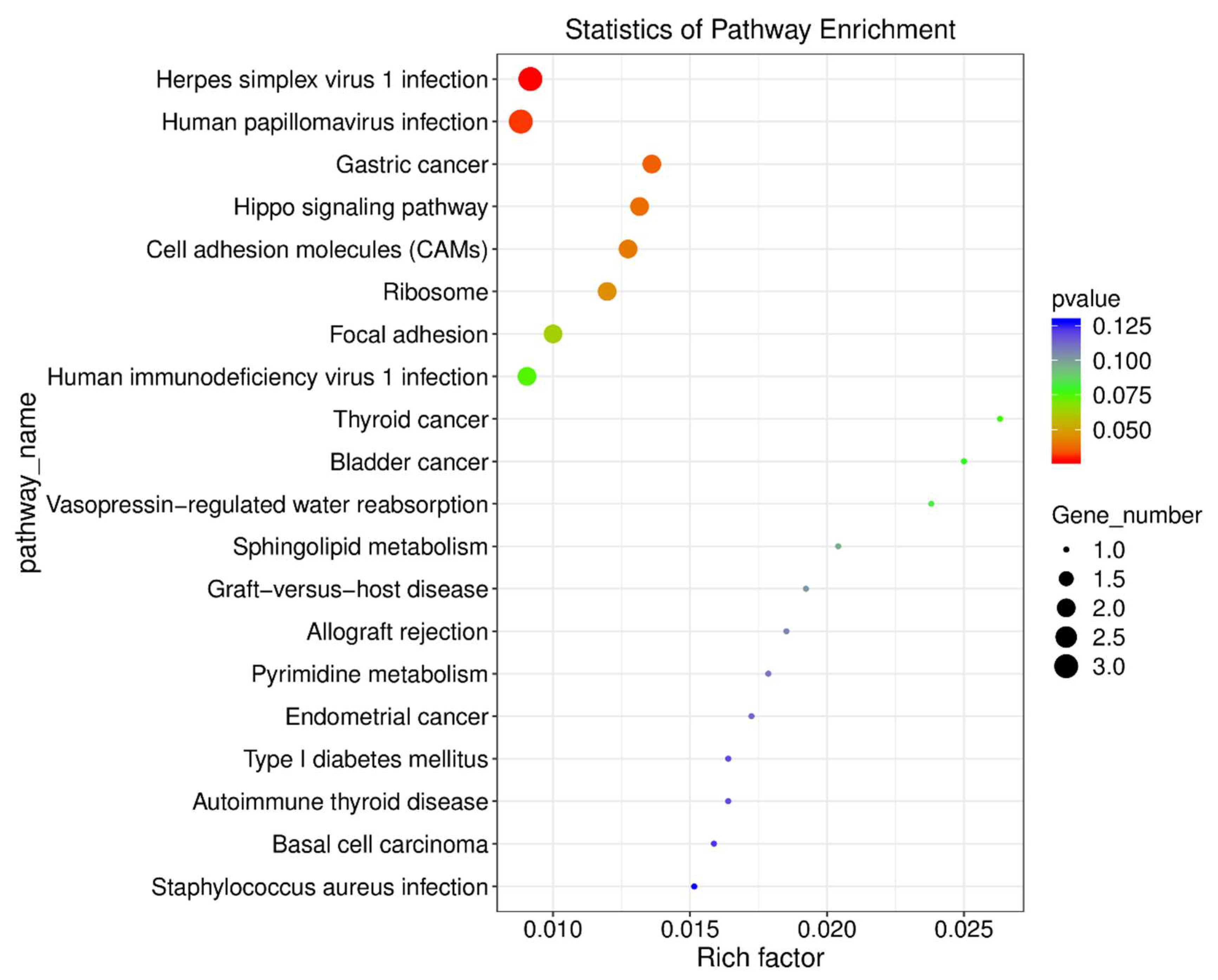
3.4. Comparative Analysis of KEGG Pathway Enrichment in the Prefrontal Cortex: Methamphetamine versus Saline Treatment
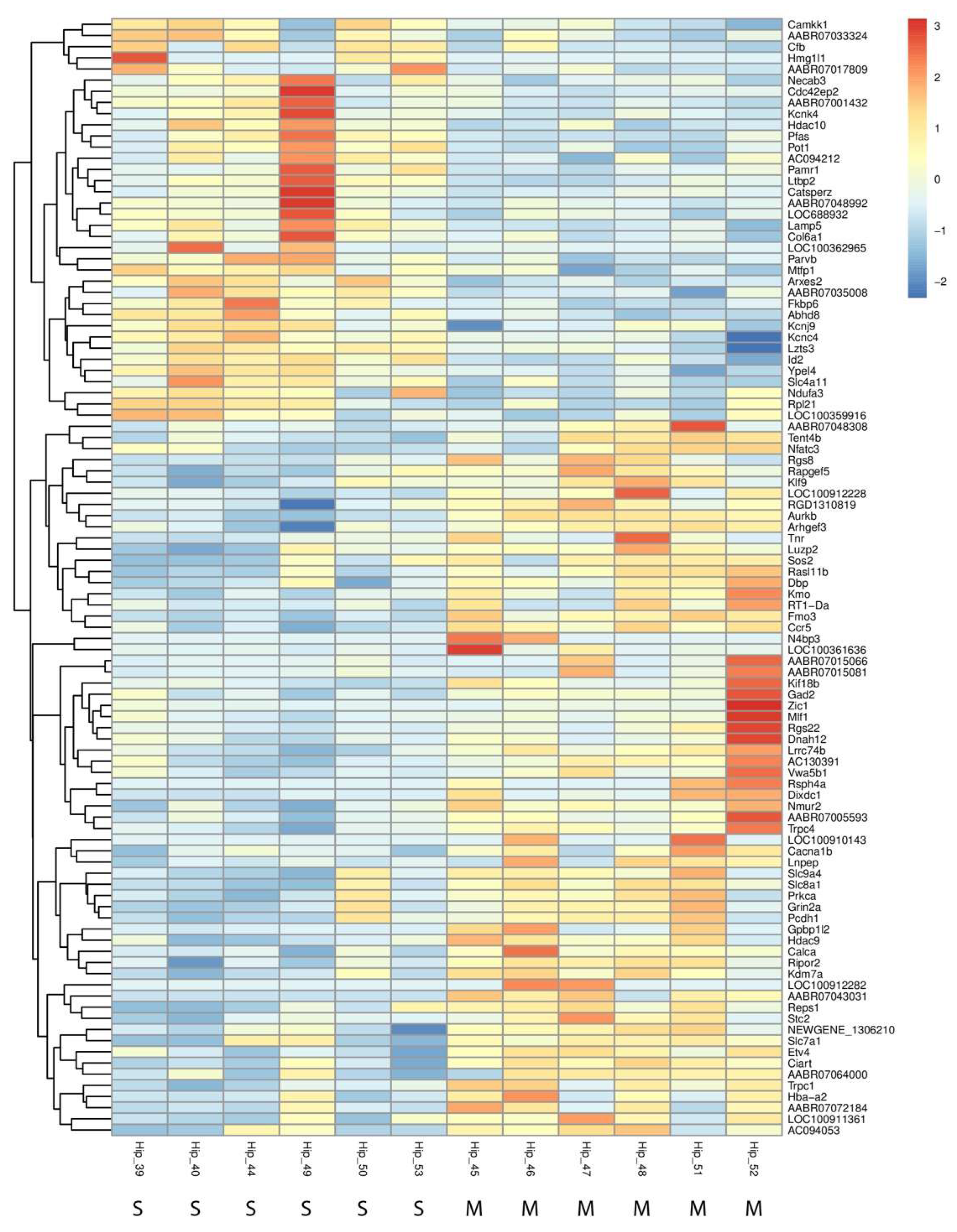
3.5. Transcriptomic Changes in the Hippocampus: Differential Gene Expression
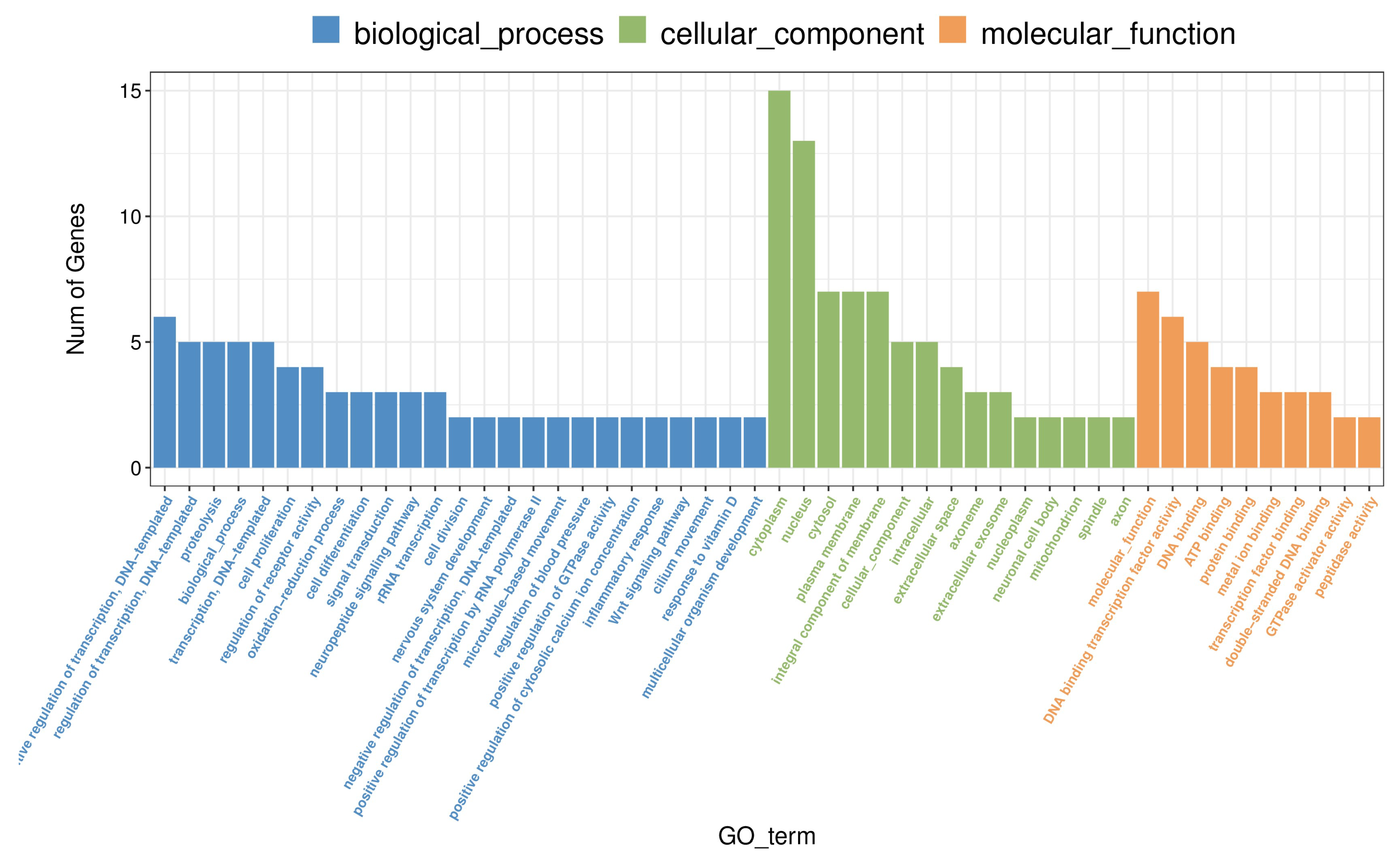
3.6. Gene Ontology Enrichment Analysis Reveals Differential Gene Expression in the Hippocampus Following Methamphetamine Self-Administration
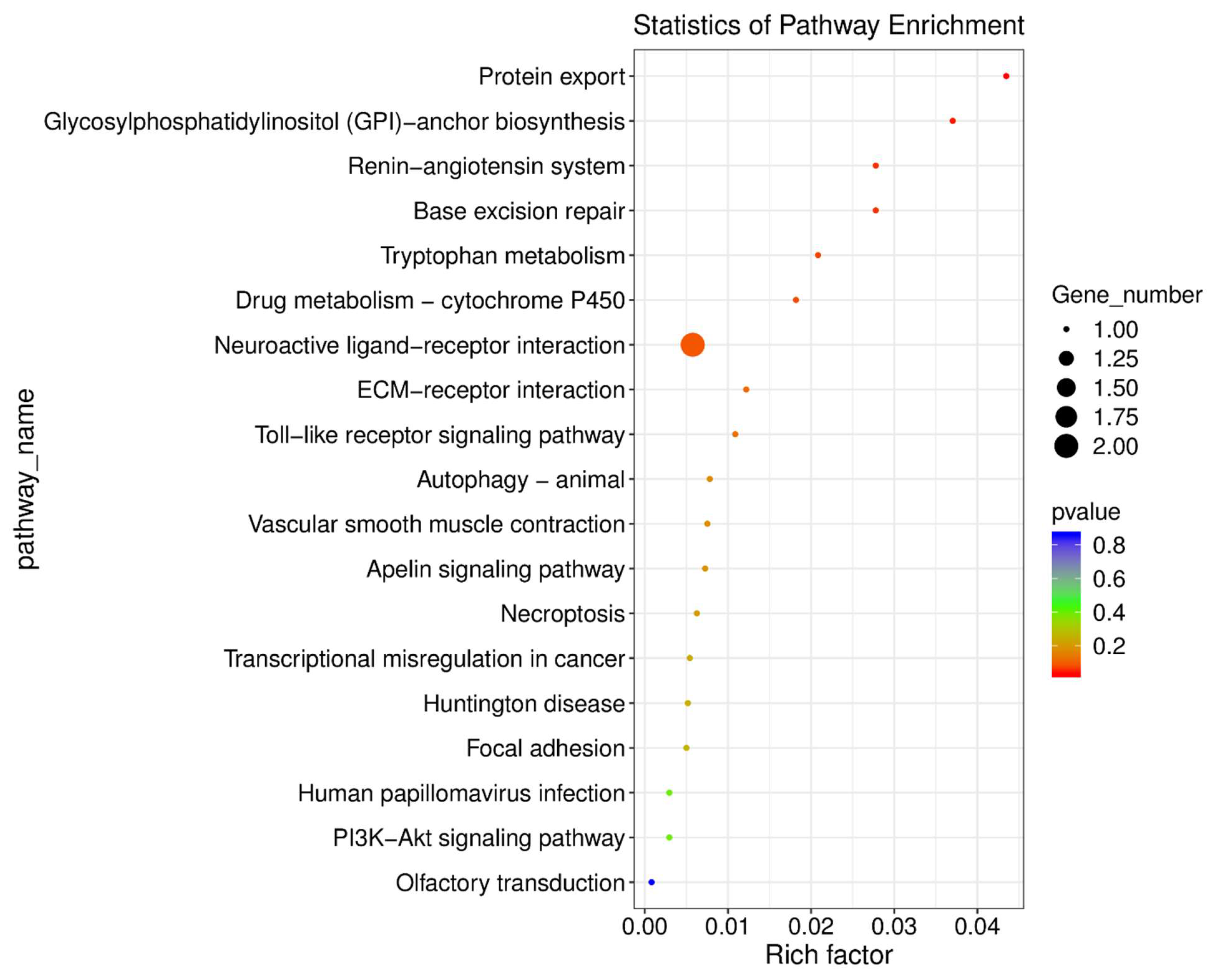
3.7. KEGG Pathway Analysis in the Hippocampus: Differential Enrichment under Methamphetamine and Saline Conditions
3.8. Secondary Analyses: Proof of Concept for Individual Gene Variability (PFC)
3.9. Secondary Analyses: Proof of Concept for Individual Gene Variability (Hippocampus)
4. Discussion
Supplementary Material
Acknowledgments
Conflict of Interest
References
- Ahmed, S. H. 2010. Validation crisis in animal models of drug addiction: Beyond non-disordered drug use toward drug addiction. Neurosci. Biobehav. Rev. 35: 172–184. [Google Scholar] [CrossRef] [PubMed]
- Athota, P., N. M. Nguyen, V. L. Schaal, S. Jagadesan, C. Guda, S. V. Yelamanchili, and G. Pendyala. 2023. Novel RNA-Seq Signatures Post-Methamphetamine and Oxycodone Use in a Model of HIV-Associated Neurocognitive Disorders. Viruses 15: 1948. [Google Scholar] [CrossRef] [PubMed]
- Bosch, P. J., M. C. Benton, D. Macartney-Coxson, and B. M. Kivell. 2015. mRNA and microRNA analysis reveals modulation of biochemical pathways related to addiction in the ventral tegmental area of methamphetamine self-administering rats. BMC Neurosci 16: 43. [Google Scholar] [CrossRef]
- Cadet, J. L., C. Brannock, S. Jayanthi, and I. N. Krasnova. 2015. Transcriptional and Epigenetic Substrates of Methamphetamine Addiction and Withdrawal: Evidence from a Long-Access Self-Administration Model in the Rat. Mol. Neurobiol. 51: 696–717. [Google Scholar] [CrossRef]
- Cadet, J. L., C. Brannock, I. N. Krasnova, B. Ladenheim, M. T. McCoy, J. Chou, E. Lehrmann, W. H. Wood, K. G. Becker, and Y. Wang. 2010. Methamphetamine-Induced Dopamine-Independent Alterations in Striatal Gene Expression in the 6-Hydroxydopamine Hemiparkinsonian Rats. PLoS ONE 5: e15643. [Google Scholar] [CrossRef]
- Cadet, J. L., S. Jayanthi, M. T. Mccoy, M. Vawter, and B. Ladenheim. 2001. Temporal profiling of methamphetamine-induced changes in gene expression in the mouse brain: Evidence from cDNA array. Synapse 41: 40–48. [Google Scholar] [CrossRef]
- Charntikov, S., S. T. Pittenger, I. Thapa, D. R. Bastola, R. A. Bevins, and G. Pendyala. 2015. Ibudilast reverses the decrease in the synaptic signaling protein phosphatidylethanolamine-binding protein 1 (PEBP1) produced by chronic methamphetamine intake in rats. Drug Alcohol Depend 152: 15–23. [Google Scholar] [CrossRef]
- Deroche-Gamonet, V., D. Belin, and P. V. Piazza. 2004. Evidence for addiction-like behavior in the rat. Science 305: 1014–1017. [Google Scholar] [CrossRef]
- Deroche-Gamonet, V., and P. V. Piazza. 2014. Psychobiology of cocaine addiction: Contribution of a multi-symptomatic animal model of loss of control. Neuropharmacology 76 Pt B: 437–449. [Google Scholar] [CrossRef]
- Eskandarian Boroujeni, M., T. Peirouvi, F. Shaerzadeh, A. Ahmadiani, M. A. Abdollahifar, and A. Aliaghaei. 2020. Differential gene expression and stereological analyses of the cerebellum following methamphetamine exposure. Addict. Biol. 25: N.PAG–N.PAG. [Google Scholar] [CrossRef]
- Fernandes, N. C., U. Sriram, L. Gofman, J. M. Cenna, S. H. Ramirez, and R. Potula. 2016. Methamphetamine alters microglial immune function through P2X7R signaling. J. Neuroinflammation 13: 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ferrario, C. R., G. Gorny, H. S. Crombag, Y. Li, B. Kolb, and T. E. Robinson. 2005. Neural and behavioral plasticity associated with the transition from controlled to escalated cocaine use. Biol. Psychiatry 58: 751–759. [Google Scholar] [CrossRef] [PubMed]
- Godino, A., S. Jayanthi, and J. L. Cadet. 2015. Epigenetic landscape of amphetamine and methamphetamine addiction in rodents. Epigenetics 10: 574–580. [Google Scholar] [CrossRef] [PubMed]
- Golsorkhdan, S. A., M. E. Boroujeni, A. Aliaghaei, M. A. Abdollahifar, A. Ramezanpour, R. Nejatbakhsh, I. J. Anarkooli, E. Barfi, and M. J. Fridoni. 2020. Methamphetamine administration impairs behavior, memory and underlying signaling pathways in the hippocampus. Behav. Brain Res. 379: 112300. [Google Scholar] [CrossRef]
- Gould, T. J. 2010. Addiction and Cognition. Addict. Sci. Clin. Pract. 5: 4–14. [Google Scholar]
- Hart, E., D. Hertia, S. T. Barrett, and S. Charntikov. 2021. Varenicline rescues nicotine-induced decrease in motivation for sucrose reinforcement. Behav. Brain Res. 397: 112887. [Google Scholar] [CrossRef]
- Hitzemann, R., O. D. Iancu, C. Reed, H. Baba, D. R. Lockwood, and T. J. Phillips. 2019. Regional Analysis of the Brain Transcriptome in Mice Bred for High and Low Methamphetamine Consumption. Brain Sci 9: 155. [Google Scholar] [CrossRef]
- Huang, X., Y.-Y. Chen, Y. Shen, X. Cao, A. Li, Q. Liu, Z. Li, L.-B. Zhang, W. Dai, T. Tan, O. Arias-Carrion, Y.-X. Xue, H. Su, and T.-F. Yuan. 2017. Methamphetamine abuse impairs motor cortical plasticity and function. Mol. Psychiatry 22: 1274–1281. [Google Scholar] [CrossRef]
- Hursh, S. R., C. M. Galuska, G. Winger, and J. H. Woods. 2005. The economics of drug abuse: a quantitative assessment of drug demand. Mol. Interv. 5: 20–28. [Google Scholar] [CrossRef]
- Hursh, S.R., and P.G. Roma. 2016. Behavioral Economics and the Analysis of Consumption and Choice. Manag. Decis. Econ. 37: 224–238. [Google Scholar] [CrossRef]
- Jayanthi, S., A.P. Daiwile, and J.L. Cadet. 2021. Neurotoxicity of methamphetamine: main effects and mechanisms. Exp. Neurol. 344: 113795. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.M., D. Houry, B. Han, G. Baldwin, A. Vivolo-Kantor, and W.M. Compton. 2022. Methamphetamine use in the United States: epidemiological update and implications for prevention, treatment, and harm reduction. Ann. N. Y. Acad. Sci. 1508: 3–22. [Google Scholar] [CrossRef] [PubMed]
- Karbalivand, M., L.L. Almada, S.M. Ansell, M.E. Fernandez-Zapico, and S.F. Elsawa. 2022. MLL1 inhibition reduces IgM levels in Waldenström macroglobulinemia. Leuk. Res. 116: 106841. [Google Scholar] [CrossRef] [PubMed]
- Kays, J.S., and B.K. Yamamoto. 2019. Evaluation of Microglia/Macrophage Cells from Rat Striatum and Prefrontal Cortex Reveals Differential Expression of Inflammatory-Related mRNA after Methamphetamine. Brain Sci 9: 340. [Google Scholar] [CrossRef]
- Kazan, T., C.L. Robison, N. Cova, V.M. Madore, and S. Charntikov. 2020. Assessment of individual differences in response to acute bupropion or varenicline treatment using a long-access nicotine self-administration model and behavioral economics in female rats. Behav. Brain Res. 385: 112558. [Google Scholar] [CrossRef]
- Kim, B., J. Yun, and B. Park. 2020. Methamphetamine-Induced Neuronal Damage: Neurotoxicity and Neuroinflammation. Biomol. Ther. 28: 381–388. [Google Scholar] [CrossRef]
- Kim, D., B. Langmead, and S.L. Salzberg. 2015. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12: 357–360. [Google Scholar] [CrossRef]
- Koob, G.F. 2008. A Role for Brain Stress Systems in Addiction. Neuron 59: 11–34. [Google Scholar] [CrossRef]
- Koob, G.F., C.L. Buck, A. Cohen, S. Edwards, P.E. Park, E. Joel, B. Schmeichel, L.F. Vendruscolo, C.L. Wade, W. Timothy, W. Jr, O. George, J.E. Schlosburg, B. Schmeichel, L.F. Vendruscolo, C.L. Wade, T.W. Whitfield, O. George, E. Joel, B. Schmeichel, L.F. Vendruscolo, C.L. Wade, W. Timothy, W. Jr, and O. George. 2014. Addiction as a stress surfeit disorder. Neuropharmacology 76: 370–382. [Google Scholar] [CrossRef]
- Koob, G.F., and M. Le Moal. 2001. Drug Addiction, Dysregulation of Reward, and Allostasis. Neuropsychopharmacology 24: 97–129. [Google Scholar] [CrossRef]
- Kosten, T.R., T.P. George, and T.A. Kosten. 2002. The potential of dopamine agonists in drug addiction. Expert Opin. Investig. Drugs 11: 491–499. [Google Scholar] [CrossRef] [PubMed]
- Limanaqi, F., S. Gambardella, F. Biagioni, C.L. Busceti, and F. Fornai. 2018. Epigenetic Effects Induced by Methamphetamine and Methamphetamine-Dependent Oxidative Stress. Oxid. Med. Cell. Longev., 4982453. [Google Scholar] [CrossRef] [PubMed]
- Loftis, J.M., and A. Janowsky. 2014. Neuroimmune Basis of Methamphetamine Toxicity. Int. Rev. Neurobiol. 118: 165–197. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. 2011. Cutadapt removes adapter sequences from high-throughput sequencing reads. Embnet J. 17. [Google Scholar]
- Matissek, S.J., M. Karbalivand, W. Han, A. Boutilier, E. Yzar-Garcia, L.L. Kehoe, D.S. Gardner, A. Hage, K. Fleck, V. Jeffers, R. Rajsbaum, and S.F. Elsawa. 2022. A novel mechanism of regulation of the oncogenic transcription factor GLI3 by toll-like receptor signaling. Oncotarget 13: 944–959. [Google Scholar] [CrossRef]
- Miao, B., X. Xing, V. Bazylianska, P. Madden, A. Moszczynska, and B. Zhang. 2023. Methamphetamine-induced region-specific transcriptomic and epigenetic changes in the brain of male rats. Commun. Biol. 6: 1–17. [Google Scholar] [CrossRef]
- Munoz, C., S. Jayanthi, B. Ladenheim, and J.L. Cadet. 2023a. Compulsive methamphetamine self-administration in the presence of adverse consequences is associated with increased hippocampal mRNA expression of cellular adhesion molecules. Front. Mol. Neurosci. 15. [Google Scholar] [CrossRef]
- Munoz, C., S. Jayanthi, B. Ladenheim, and J.L. Cadet. 2023b. Compulsive methamphetamine self-administration in the presence of adverse consequences is associated with increased hippocampal mRNA expression of cellular adhesion molecules. Front. Mol. Neurosci. 15: 1104657. [Google Scholar] [CrossRef]
- Nestler, E.J. 2014. Epigenetic mechanisms of drug addiction. Neuropharmacology 76 Pt B: 259–268. [Google Scholar] [CrossRef]
- Paterson, N.E., and A. Markou. 2003. Increased motivation for self-administered cocaine after escalated cocaine intake. Neuroreport 14: 2229–32. [Google Scholar] [CrossRef]
- Pertea, M., G.M. Pertea, C.M. Antonescu, T.-C. Chang, J.T. Mendell, and S.L. Salzberg. 2015. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 33: 290–295. [Google Scholar] [CrossRef]
- Polvat, T., T. Prasertporn, P. Na Nakorn, S. Pannengpetch, W. Suwanjang, J. Panmanee, S. Ngampramuan, J.L. Cornish, and B. Chetsawang. 2023. Proteomic Analysis Reveals the Neurotoxic Effects of Chronic Methamphetamine Self-Administration-Induced Cognitive Impairments and the Role of Melatonin-Enhanced Restorative Process during Methamphetamine Withdrawal. J. Proteome Res. 22: 3348–3359. [Google Scholar] [CrossRef] [PubMed]
- Robbins, T.W., K.D. Ersche, and B.J. Everitt. 2008. Drug addiction and the memory systems of the brain. Ann. N. Y. Acad. Sci. 1141: 1–21. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D., D.J. McCarthy, and G.K. Smyth. 2010. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26: 139–140. [Google Scholar] [CrossRef] [PubMed]
- Robison, C.L., N. Cova, V. Madore, T. Allen, S. Barrett, and S. Charntikov. 2023. Assessment of ethanol and nicotine interactions using a reinforcer demand modeling with grouped and individual levels of analyses in a long-access self-administration model using male rats. Front. Behav. Neurosci. 17. [Google Scholar] [CrossRef]
- Robison, C.L., V. Madore, N. Cova, R.C. Drugan, and S. Charntikov. 2024. Individual corticosterone response to intermittent swim stress predicts a shift in economic demand for ethanol from pre-stress to post-stress in male rats. Front. Behav. Neurosci. 18. [Google Scholar] [CrossRef]
- Robson, M.J., R.C. Turner, Z.J. Naser, C.R. McCurdy, J.D. Huber, and R.R. Matsumoto. 2013. SN79, a sigma receptor ligand, blocks methamphetamine-induced microglial activation and cytokine upregulation. Exp. Neurol. 247: 134. [Google Scholar] [CrossRef]
- Salamanca, S.A., E.E. Sorrentino, J.D. Nosanchuk, and L.R. Martinez. 2015. Impact of methamphetamine on infection and immunity. Front. Neurosci. 8. [Google Scholar] [CrossRef]
- Serra, M., N. Simola, A.E. Pollack, and G. Costa. 2023. Brain dysfunctions and neurotoxicity induced by psychostimulants in experimental models and humans: an overview of recent findings. Neural Regen. Res. 19: 1908–1918. [Google Scholar] [CrossRef]
- Shaerzadeh, F., W.J. Streit, S. Heysieattalab, and H. Khoshbouei. 2018. Methamphetamine neurotoxicity, microglia, and neuroinflammation. J. Neuroinflammation 15: 1–6. [Google Scholar] [CrossRef]
- Stafford, N.P., T.N. Kazan, C.M. Donovan, E.E. Hart, R.C. Drugan, and S. Charntikov. 2019. Individual Vulnerability to Stress Is Associated With Increased Demand for Intravenous Heroin Self-administration in Rats. Front. Behav. Neurosci. 13: 134. [Google Scholar] [CrossRef]
- Thomas, D.M., D.M. Francescutti-Verbeem, X. Liu, and D.M. Kuhn. 2004. Identification of differentially regulated transcripts in mouse striatum following methamphetamine treatment–an oligonucleotide microarray approach. J. Neurochem. 88: 380–393. [Google Scholar] [CrossRef] [PubMed]
- Uphoff, K.W., S.D. Bell, A.D. Ellington, C. Opin, S. Biol, M.G. Wallis, P. Biophys, M. Biol, S.H. Ahmed, and G.F. Koob. 1998. Transition from moderate to excessive drug intake: Change in hedonic set point. Science 282: 298–300. [Google Scholar]
- Venables, W.N., and B.D. Ripley. 2002. Modern Applied Statistics with S, Statistics and Computing. Springer: New York, NY. [Google Scholar] [CrossRef]
- Wang, H., X. Dong, M.U.N. Awan, and J. Bai. 2022. Epigenetic mechanisms involved in methamphetamine addiction. Front. Pharmacol. 13. [Google Scholar] [CrossRef] [PubMed]
- Yang, X., Y. Wang, Q. Li, Y. Zhong, L. Chen, Y. Du, J. He, L. Liao, K. Xiong, C. Yi, and J. Yan. 2018. The Main Molecular Mechanisms Underlying Methamphetamine-Induced Neurotoxicity and Implications for Pharmacological Treatment. Front. Mol. Neurosci. 11. [Google Scholar] [CrossRef]
- Zeng, K., X. Yu, Z. Wei, Y. Wu, J. Wang, R. Liu, Y. Li, and X. Wang. 2023. Single-nucleus transcriptome profiling of prefrontal cortex induced by chronic methamphetamine treatment. Gen. Psychiatry 36: e101057. [Google Scholar] [CrossRef]
- Zhu, L., J. Zhu, Y. Liu, Y. Chen, Y. Li, S. Chen, T. Li, Y. Dang, and T. Chen. 2015a. Chronic methamphetamine regulates the expression of MicroRNAs and putative target genes in the nucleus accumbens of mice. J. Neurosci. Res. 93: 1600–1610. [Google Scholar] [CrossRef]
- Zhu, L., J. Zhu, Y. Liu, Y. Chen, Y. Li, L. Huang, S. Chen, T. Li, Y. Dang, and T. Chen. 2015b. Methamphetamine induces alterations in the long non-coding RNAs expression profile in the nucleus accumbens of the mouse. BMC Neurosci 16: 18. [Google Scholar] [CrossRef]
- Zhu, R., T. Yang, F. Kobeissy, T.H. Mouhieddine, M. Raad, A. Nokkari, M.S. Gold, K.K. Wang, and Y. Mechref. 2016. The Effect of Chronic Methamphetamine Exposure on the Hippocampal and Olfactory Bulb Neuroproteomes of Rats. PLoS ONE 11: e0151034. [Google Scholar] [CrossRef]
| Behavioral Economics Parameters | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Brain Area | Sample Name | Quality (RNA IQ) | Conc. (µg/µL) | Group Name | EV | Q0 | Omax | Pmax | ||
| Hip | Hip 45 | 7 | 2.3208 | METH | 0.82 | 19.00 | 22.77 | 3.69 | ||
| Hip | Hip 46 | 7 | 2.4656 | METH | 0.79 | 2.80 | 22.06 | 24.28 | ||
| Hip | Hip 47 | 8.2 | 1.48624 | METH | 2.64 | 6.00 | 73.54 | 37.77 | ||
| Hip | Hip 48 | 7.6 | 1.95136 | METH | 1.81 | 7.20 | 50.43 | 21.58 | ||
| Hip | Hip 51 | 7.4 | 1.80288 | METH | 1.01 | 8.90 | 28.24 | 9.78 | ||
| Hip | Hip 52 | 9.1 | 0.85248 | METH | 4.22 | 5.10 | 117.66 | 71.10 | ||
| PFC | PFC 45 | 7.6 | 2.14736 | METH | 0.82 | 19.00 | 22.77 | 3.69 | ||
| PFC | PFC 46 | 7.2 | 2.6532 | METH | 0.79 | 2.80 | 22.06 | 24.28 | ||
| PFC | PFC 47 | 7.4 | 2.3032 | METH | 2.64 | 6.00 | 73.54 | 37.77 | ||
| PFC | PFC 48 | 8.3 | 1.44056 | METH | 1.81 | 7.20 | 50.43 | 21.58 | ||
| PFC | PFC 51* | 7.6 | 2.29496 | METH | 1.01 | 8.90 | 28.24 | 9.78 | ||
| PFC | PFC 52 | 7.6 | 3.15064 | METH | 4.22 | 5.10 | 117.66 | 71.10 | ||
| Hip | Hip 39 | 6.8 | 2.19064 | SAL | 0.09 | 0.87 | 2.61 | 9.26 | ||
| Hip | Hip 40 | 7.2 | 2.26464 | SAL | 0.00 | 0.74 | 0.11 | 0.46 | ||
| Hip | Hip 44 | 7.2 | 2.26144 | SAL | 0.13 | 1.40 | 3.53 | 7.77 | ||
| Hip | Hip 49 | 7.8 | 1.41472 | SAL | 0.15 | 2.30 | 4.15 | 5.56 | ||
| Hip | Hip 50 | 7.8 | 1.70512 | SAL | 0.19 | 1.70 | 5.43 | 9.84 | ||
| Hip | Hip 53 | 8.1 | 1.73056 | SAL | 0.07 | 0.52 | 1.91 | 11.31 | ||
| PFC | PFC 39 | 8 | 1.51104 | SAL | 0.09 | 0.87 | 2.61 | 9.26 | ||
| PFC | PFC 40 | 7.5 | 2.19352 | SAL | 0.00 | 0.74 | 0.11 | 0.46 | ||
| PFC | PFC 44 | 7.5 | 2.18944 | SAL | 0.13 | 1.40 | 3.53 | 7.77 | ||
| PFC | PFC 49 | 7.7 | 2.88296 | SAL | 0.15 | 2.30 | 4.15 | 5.56 | ||
| PFC | PFC 50 | 7.9 | 1.84504 | SAL | 0.19 | 1.70 | 5.43 | 9.84 | ||
| PFC | PFC 53 | 7.9 | 1.9628 | SAL | 0.07 | 0.52 | 1.91 | 11.31 | ||
| Hip - Hippocampus; PFC - prefrontal cortex; * sample excluded from the analyses because it was compromised during transport; METH - methamphetamine; SAL - saline; EV, Q0, Omax, and Pmax are parameters derived from the exponential economic demand model of consumption. | ||||||||||
| Parameter | t | df | p-value | METH mean | SAL mean |
|---|---|---|---|---|---|
| EV | 3.215 | 10 | 0.0093 | 1.879 | 0.105 |
| Q0 | 2.956 | 10 | 0.0144 | 8.166 | 1.255 |
| Pmax | 2.064 | 10 | 0.0659 | 28.034 | 7.368 |
| Omax | 3.215 | 10 | 0.0093 | 52.448 | 2.957 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




