Submitted:
09 January 2025
Posted:
13 January 2025
You are already at the latest version
Abstract
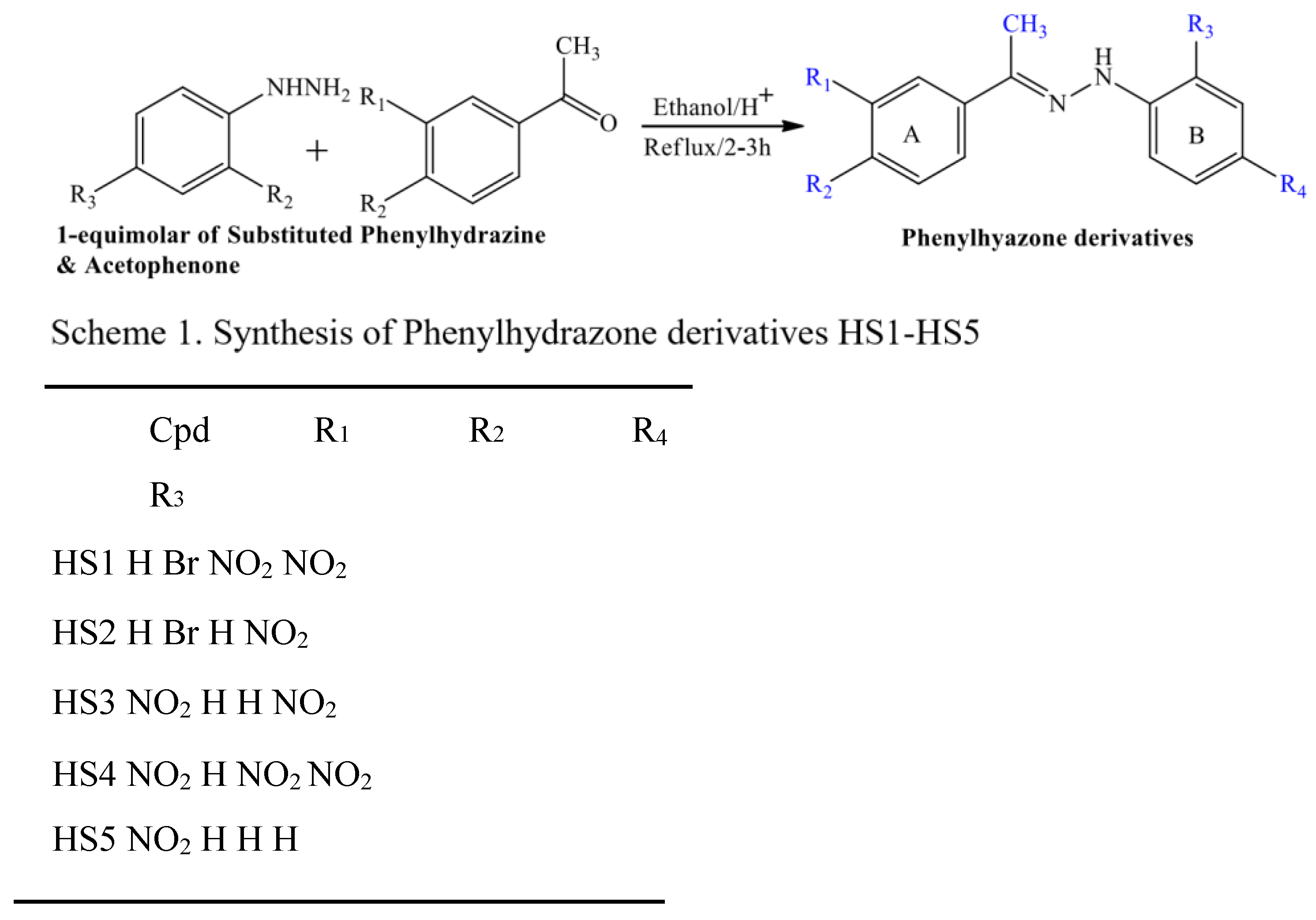
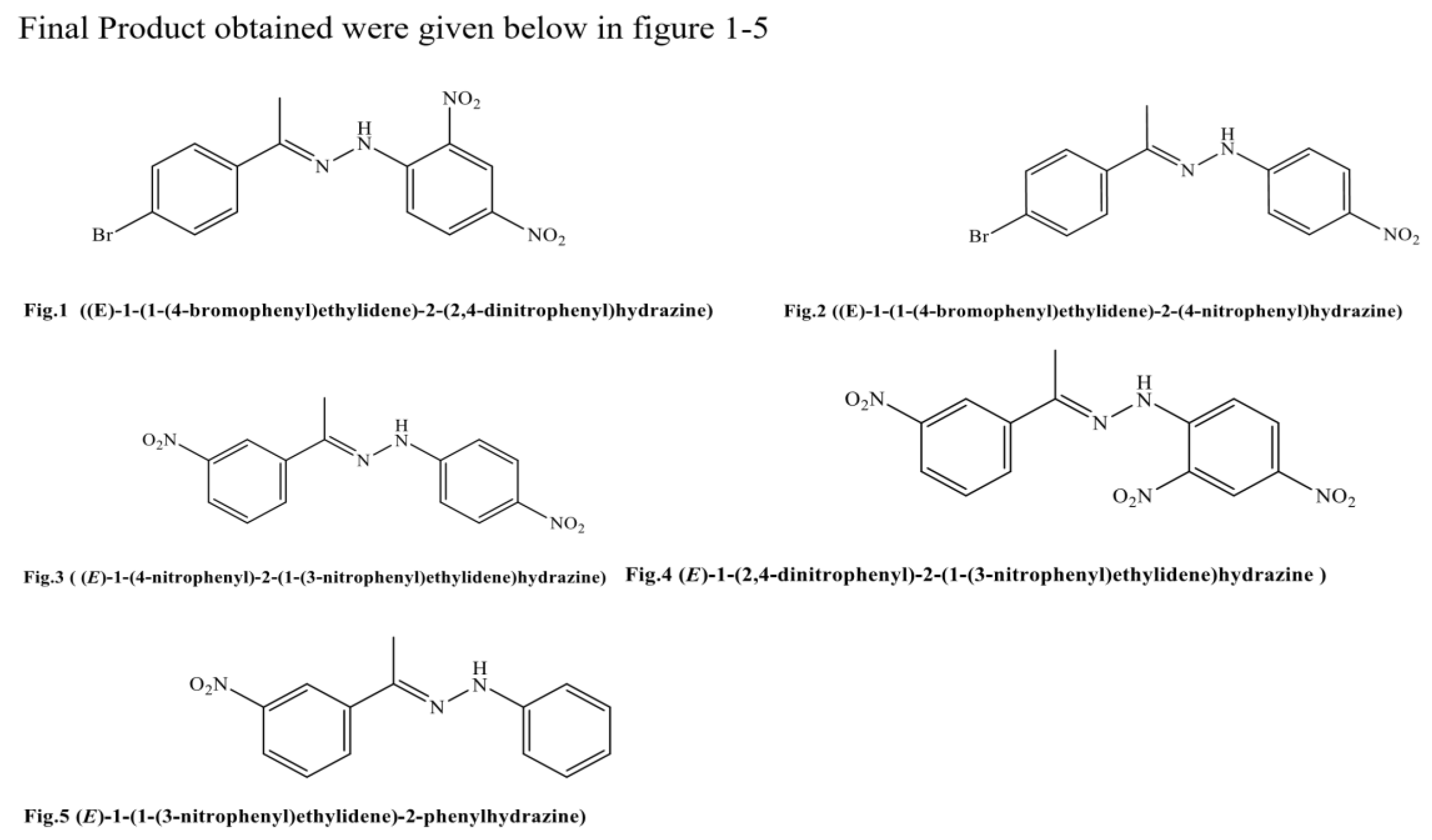
Antimicrobial chemotherapeutic failure as a result of pathogenic resistance stain is great concern across the globe, there is need to search for an effective antimicrobial agent from synthetic sources to wipe microbial resistant in clinical practice. The phenylhydrazone derivatives were scientifically found to have wide application in area of drug discovery due to their anticancer, anti-tubercular, anti-bacterial and anti-fungal activities. Five (5) novel (E)-Substituted-N-(phenylhydrazones) derivatives’ were obtained by condensation reaction through one step reaction, here are the five compounds, HS1 (E)-1-(1-(4-bromophenyl)ethylidene)-2-(2,4dinitrophenyl)hydrazine),HS2 (E)-1-(1-(4-bromophenyl)ethylidene)-2-(4-nitrophenyl)hydrazine), HS3(E)-1-(4nitrophenyl)-2-(1-(3-nitrophenyl)ethylidene)hydrazine), HS4 (E)-1-(2,4-dinitrophenyl)-2-(1-(3nitrophenyl)ethylidene)hydrazine), and HS5 (E)-1-(1-(3-nitrophenyl)ethylidene)-2-phenylhydrazine) and their structural elucidation were established on the basis of FTIR, 1D and 2D NMR spectra and the Insilco prediction of physicochemical properties found within the lipinski’s rule of five and the newly synthesized compounds were evaluated for antimicrobial assessment via an in-vitro test model using inhibition zone technique, MIC, MBC and MFC.
Keywords:
1. Introduction
2. Materials and Methods
2.10. Reagents, Solvents and Standard Drug
2.20. Chemistry
3.0. Antimicrobial Activity
3.1. Antimicrobial Evaluation of the Synthesized Compounds
3.2. Source of Test Organisms
3.3. Procedure Preparation of Inoculum:
3.4. Preparation of Test Sample:
3.5. Standard References
3.6. Determination of Antimicrobial Activity (Zone of Inhibition)
3.7. Determination of Minimum Inhibitory Concentration (MIC)
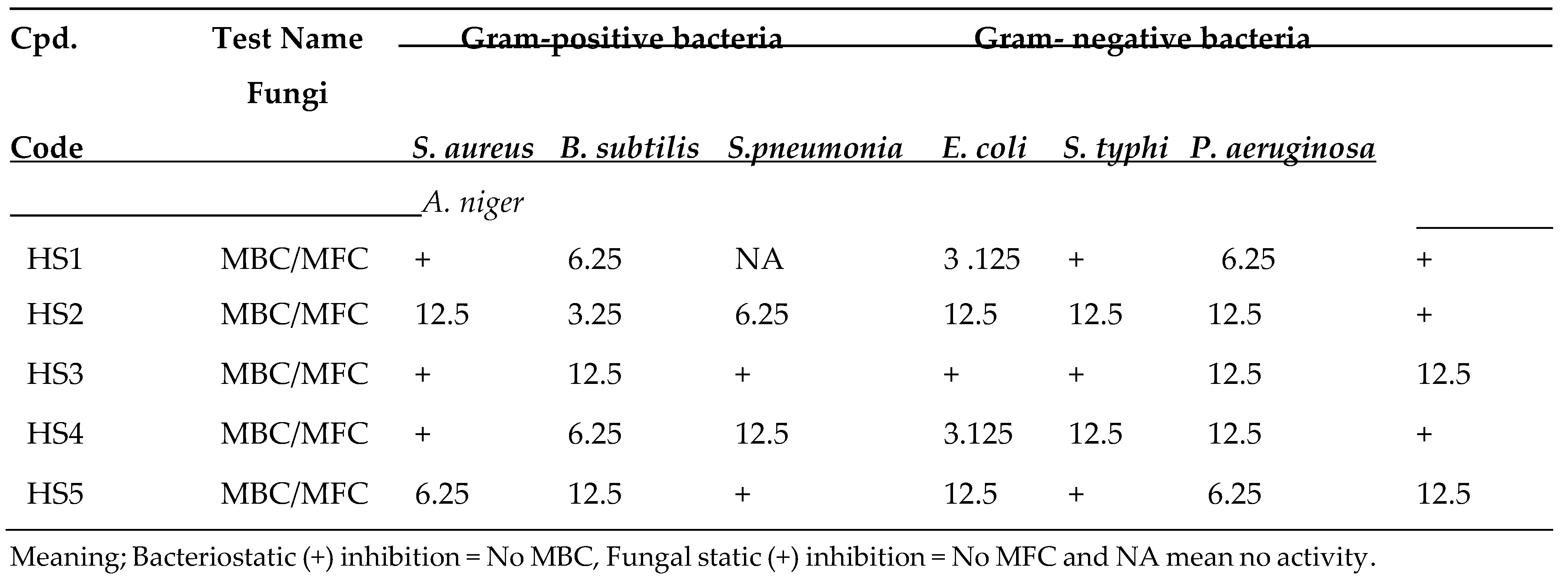
4.0. Results and Discusion
|
Cpd Code |
Conc. (µg/ml) |
Inhibition zone represented by (mm) | ||||||
| Gram-positive bacteria | Gram- negative bacteria | Fungi | ||||||
| S. aureus | B. subtilis | S.pneumonia | E. coli | S. typhi | P. aeruginosa | A. niger | ||
| HS1 | 50 | 20 | 22 | NA | 18 | 20 | 12 | 15 |
| 25 | 18 | 18 | NA | 15 | 18 | NA | 13 | |
| 12.5 | 16 | NA | NA | NA | 15 | NA | 12 | |
| 6.25 | 16 | NA | NA | NA | NA | NA | NA | |
| HS2 | 50 | 19 | 15 | 15 | 15 | 18 | 17 | 25 |
| 25 | 17 | 13 | 12 | 12 | 15 | 13 | 20 | |
| 12.5 | 15 | 12 | 11 | 11 | 13 | 12 | 17 | |
| 6.25 | 14 | 11 | NA | NA | NA | NA | 15 | |
| HS3 | 50 | 17 | 17 | 20 | 17 | 22 | 14 | 20 |
| 25 | 15 | 18 | 18 | 13 | 20 | 13 | 18 | |
| 12.5 | 14 | 15 | 15 | 12 | 19 | 12 | 15 | |
| 6.25 | 12 | NA | NA | NA | 18 | NA | NA | |
| HS4 | 50 | 19 | 20 | 15 | 15 | 20 | 15 | 20 |
| 25 | 16 | 17 | NA | 13 | 19 | R | 17 | |
| 12.5 | 15 | 15 | NA | 12 | 16 | R | 15 | |
| 6.25 | 13 | 12 | NA | NA | NA | NA | 12 | |
| HS5 | 50 | 20 | 11 | 18 | 22 | 20 | 22 | 18 |
| 25 | 17 | NA | 15 | 19 | 19 | 18 | 16 | |
| 12.5 | 15 | NA | 12 | 16 | 16 | 16 | 13 | |
| 6.25 | NA | NA | NA | NA | NA | NA | NA | |
| CF | 10 | - | - | - | - | - | - | - |
| TN | 30 | - | - | - | - | - | - | - |
|
Test Name |
Gram-positive bacteria | Gram- negative bacteria | Fungi | ||||||
| S. aureus | B. subtilis | S.pneumonia | E. coli | S. typhi | P. aeruginosa | A. niger | |||
| HS1 | MIC | 12.5 | 3.125 | NA | 1.56 | 12.5 | 3.125 | 12.5 | |
| HS2 | MIC | 12.5 | 12.5 | 6.25 | 12.5 | 12.5 | 6.25 | 12.5 | |
| HS3 | MIC | 6.25 | 3.125 | 3.125 | 1.56 | 6.25 | 6.25 | 12.5 | |
| HS4 | MIC | 12.5 | 3.125 | 6.25 | 3.125 | 6.25 | 6.25 | 12.5 | |
| HS5 | MIC | 3.125 | 6.25 | 12.5 | 6.25 | 12.5 | 3.125 | 6.25 | |
 |
| Code | Mol. Formula | Mol.Wt. g/mol | Color | M.P. (0C) | Rf Value | Yield (%) |
| HS1 | C14H11BrN4O4 | 379.17 | Pink | 213-215 | 0.91 | 40.00 |
| HS2 | C14H12BrN3O2 | 334.17 | Brown | 220-222 | 0.85 | 67.00 |
| HS3 | C14H12N4O4 | 300.27 | Sandy- brown | 213-215 | 0.83 | 67.00 |
| HS4 | C14H11N5O6 | 345.27 | Yellow | 210-212 | 0.87 | 89.00 |
| HS5 | C14H13N3O2 | 255.27 | Reddish Brown | 110-112 | 0.89 | 81.00 |
5.0. Conclusion
Acknowledgments
Abbreviations
References
- Ade, A. , Amengor, C. D. K., Brobbey, A., Ayensu, I., Harley, B. K., & Boakye, Y. D. (2020). Synthesis and Antimicrobial Resistant Modulatory Activity of 2, 4-Dinitrophenylhydrazone Derivatives as Agents against Some ESKAPE Human Pathogens. 2020.
- Agrawal, P., & Jeyabalan, G. Indian Journal of Pharmaceutical and Biological Research ( IJPBR ) Synthesis and antimicrobial activity of some newer semicarbazone analogues. 2017, 5, 12–17.
- Al-otibi, F. , Alkhudhair, S. K., Alharbi, R. I., Al-askar, A. A., Aljowaie, R. M., & Al-shehri, S. (2021). The Antimicrobial Activities of Silver Nanoparticles from and Fungi. Molecules, 26(6081).
- Arshad, M. , Shoeb, M., Shahab, K., Asghar, A., & Dabeer, N. (2019). benzeneamine : synthesis, characterization, antibacterial, and MTT assessment. SN Applied Sciences, 1(6), 1–8.
- https://doi.org/10.1007/s42452-019-0571-8. [CrossRef]
- Elhady, H. A., Al-nathali, H. S., & El-sayed, R. (2017). ISSN : 2320-5407 Manuscript Info Abstract.
- Introduction : - ISSN : 2320-5407 Results and discussion : -. 5(10), 1716–1725.
- https://doi.org/10.21474/IJAR01/5694. [CrossRef]
- Facts, K. E. Y. (2015). WHO, “Antimicrobials: Handle with Care” in 2020. 1–6.
- Fen, K., Dergisi, B., & Derivatives, H. (2023). Karadeniz Fen Bilimleri Dergisi. 13(1), 135–152.
- https://doi.org/10.31466/kfbd.1184337. [CrossRef]
- Ii, Z. N. , Al-qadsy, I., Saeed, W. S., Alrabie, A., & Al-adhreai, A. (2021). Synthesis, Characterization, SingleCrystal X-ray Structure and Biological Activities of [(Z)-N -(4-Methoxybenzylidene)benzohydrazide– Nickel(II)] Complex. Ii, 1–16.
- Moussa, Z., Al-mamary, M., Al-juhani, S., & Ahmed, S. A. (2020). Heliyon Preparation and biological assessment of some aromatic hydrazones derived from hydrazides of phenolic acids and aromatic aldehydes. Heliyon, 6(April), e05019. [CrossRef]
- Nesterkina, M. , Barbalat, D., & Kravchenko, I. (2020). Design, synthesis and pharmacological pro fi le of ( − ) - verbenone hydrazones. 943–950.
- Thi, N. , Thoa, L., Nam, P. C., & Nhat, D. M. (2015). Antibacterial Activities of The Extracts of Mimosa pudica L. An in-vitro Study. 5(5), 358–361.
- You, A., Be, M. A. Y., & In, I. (2021). Microwave-assisted synthesis and antioxidant activity of an imine , ( E ) -1- ( 3- bromobenzylidene ) -2-phenylhydrazine Microwave-Assisted Synthesis and Antioxidant Activity of 040041(June 2020).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




