Submitted:
07 January 2025
Posted:
08 January 2025
You are already at the latest version
Abstract
Keywords:
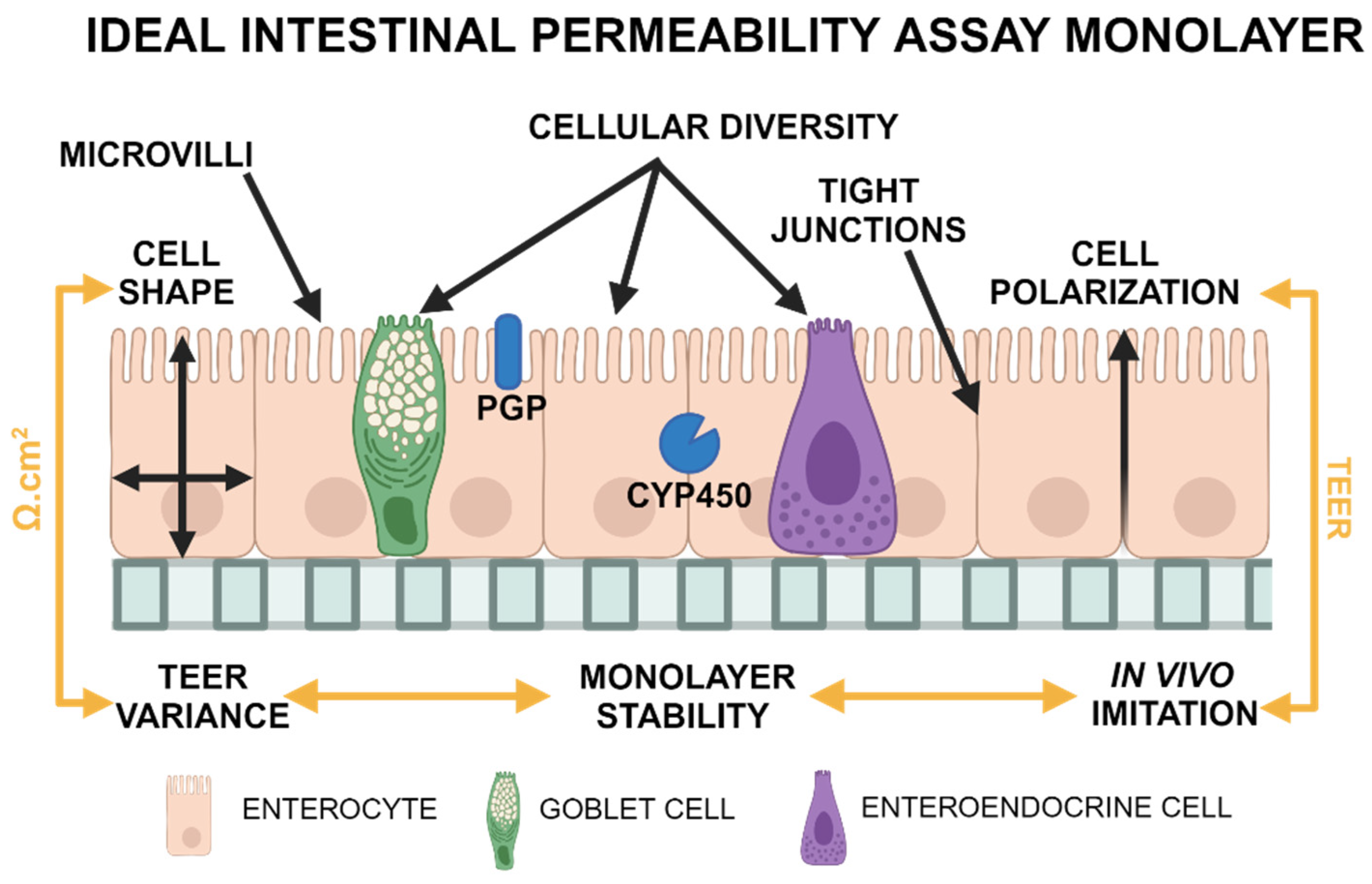
1. Introduction
- The presence of mucin-producing and secretory cells (1); a polarized (2), columnar (3) cellular monolayer and typical microanatomical features of enterocytes such as microvilli (4);
- The presence of tight junctions as evaluated through transmission electron microscopy (TEM) (5) and TEER measurements. TEER values should also replicate those measured in vivo (6), with minimal variations between replicates (7). The focus on TEER values can be justified as this method is the only quantifiable variable obtained in real-time during cell culture.
- Cell monolayers must be stable (8) for several days to allow for permeability assay experiments. Stability is defined as a <10% decrease in TEER values in 2 days following the plateau state.
2. Materials and Methods
2.1. Canine 3D Enteroid/Colonoid Culture and Monolayer
2.2. D Cell Culture
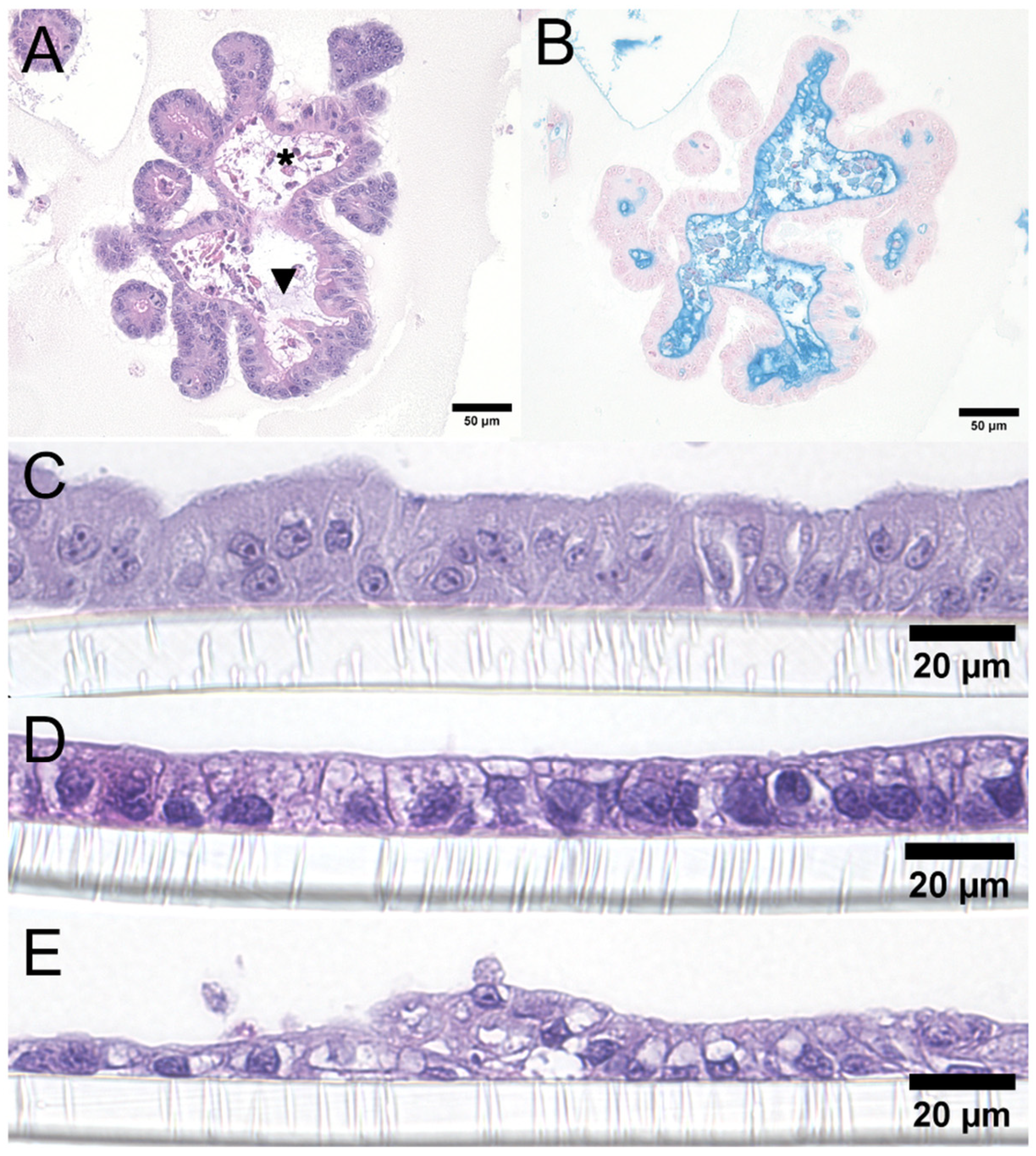
2.3. Histochemical Staining
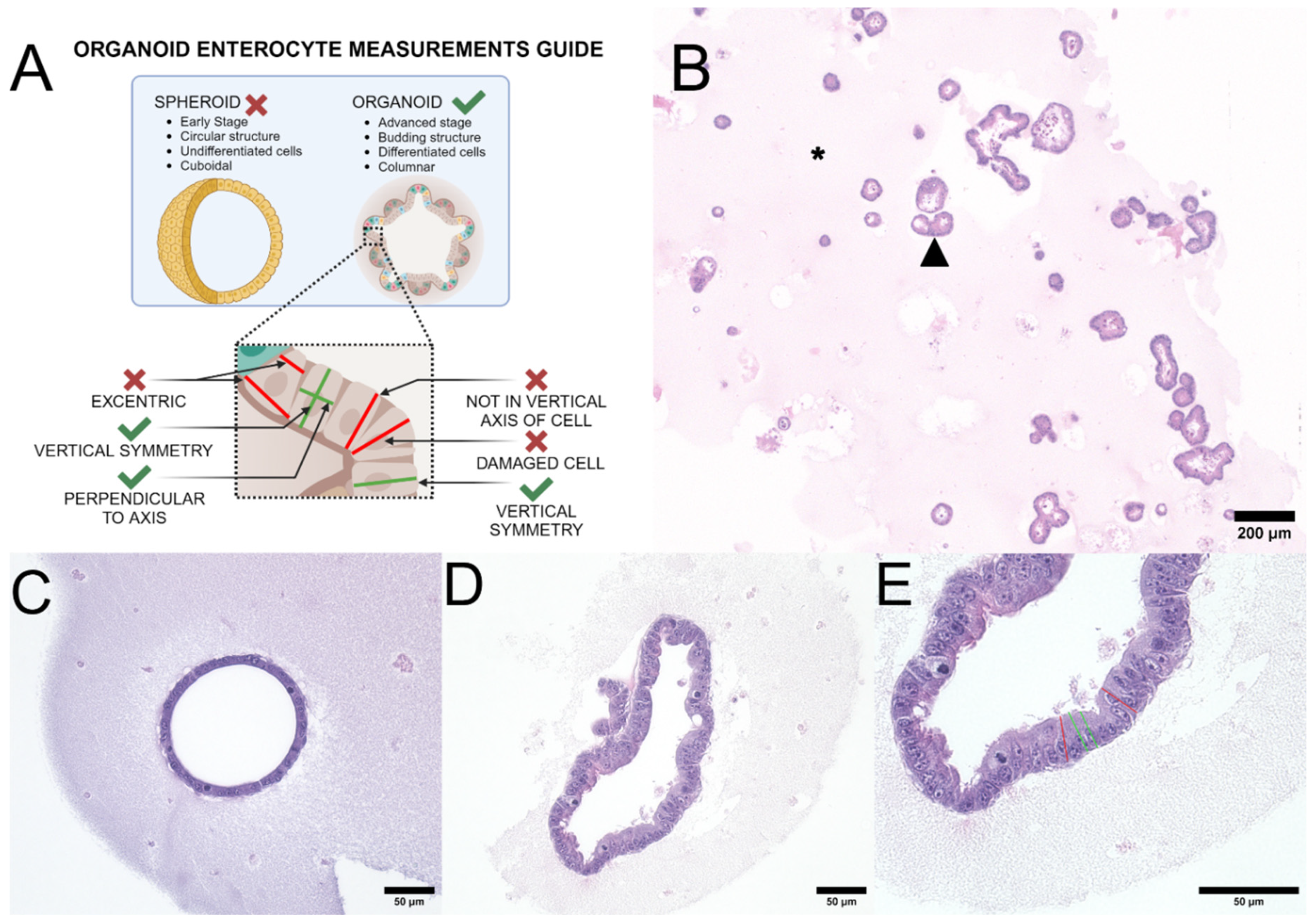
2.4. Cell Measurement
2.5. Statistical Analysis
2.6. Transmission Electron Microscopy (TEM)
3. Results
3.1. Canine Organoid Enterocyte Measurements
3.2. Cell Morphology and Immunofluorescence
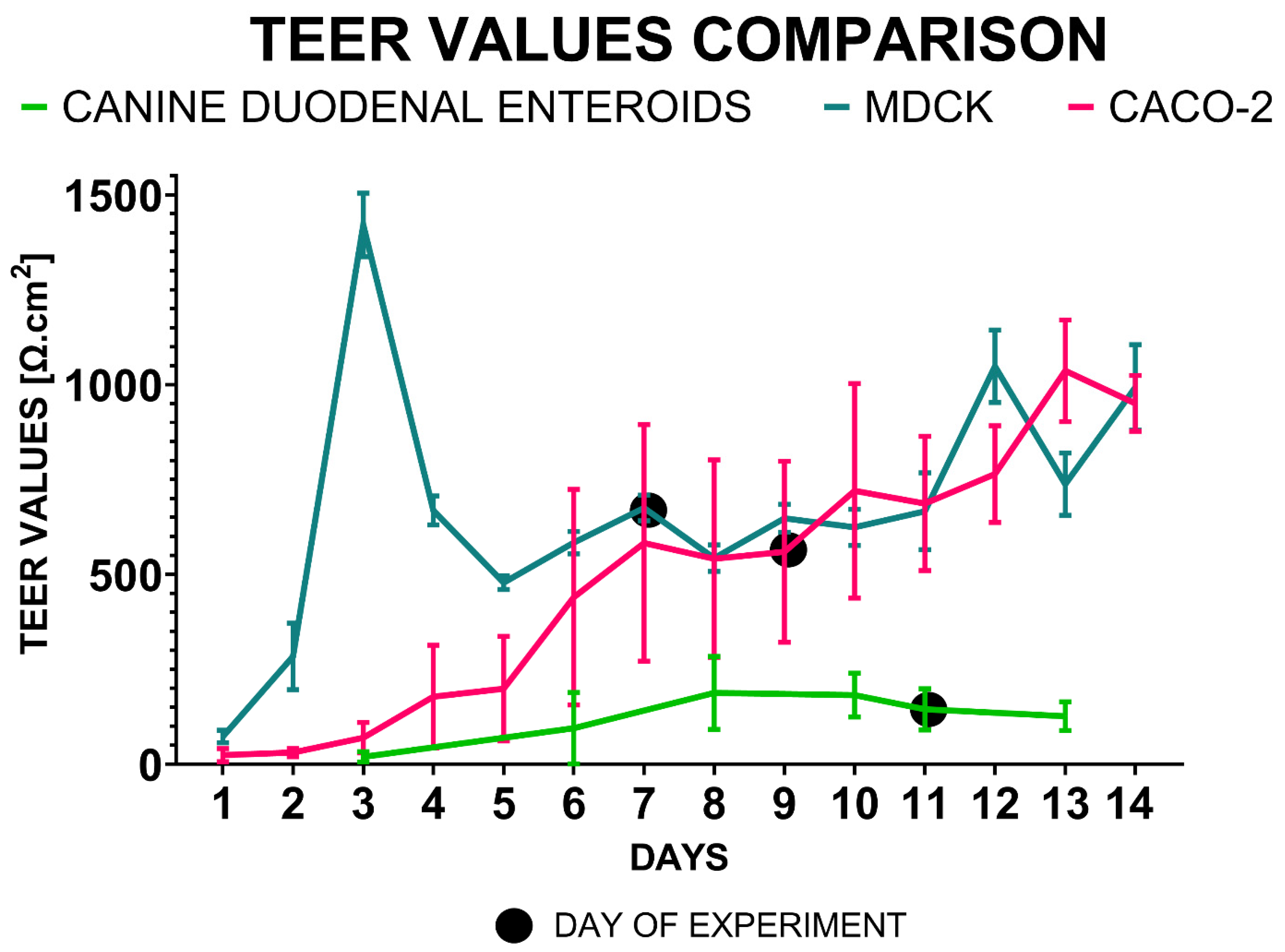
3.3. Transepithelial Electrical Resistance (TEER) Measurements
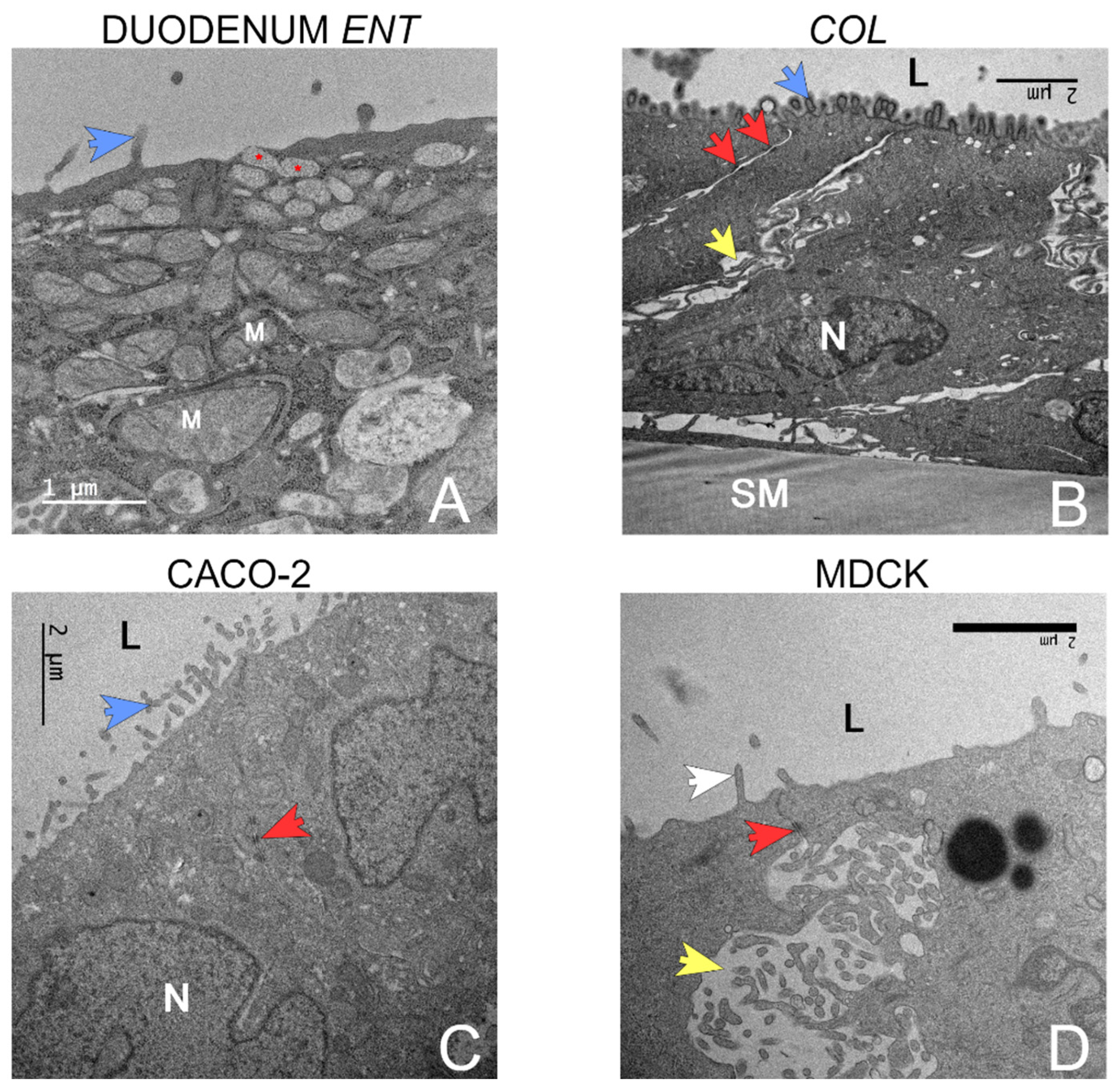
3.4. Transmission Electron Microscopy (TEM)
4. Discussion
4.1. Study Limitations
5. Conclusion
Supplemental Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- König, J.; Wells, J.; Cani, P.D.; et al. Human Intestinal Barrier Function in Health and Disease. Clin Transl Gastroenterol. 2016, 7, e196. [Google Scholar] [CrossRef]
- Volpe, D.A. Variability in Caco-2 and MDCK cell-based intestinal permeability assays. J Pharm Sci. 2008, 97, 712–725. [Google Scholar] [CrossRef] [PubMed]
- Fogh, J.; Fogh, J.M.; Orfeo, T. One hundred and twenty-seven cultured human tumor cell lines producing tumors in nude mice. J Natl Cancer Inst. 1977, 59, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Gaush, C.R.; Hard, W.L.; Smith, T.F.; Read, W.O. Characterization of an established line of canine kidney cells (MDCK). Proc Soc Exp Biol Med. 1966, 122, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Volpe, D.A. Application of method suitability for drug permeability classification. AAPS J. 2010, 12, 670–678. [Google Scholar] [CrossRef] [PubMed]
- Mochel, J.P.; Jergens, A.E.; Kingsbury, D.; Kim, H.J.; Martín, M.G.; Allenspach, K. Intestinal Stem Cells to Advance Drug Development, Precision, and Regenerative Medicine: A Paradigm Shift in Translational Research. AAPS Journal. 2018, 20, 17. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Vries, R.G.; Snippert, H.J.; et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009, 459, 262–265. [Google Scholar] [CrossRef] [PubMed]
- Chandra, L.; Borcherding, D.C.; Kingsbury, D.; et al. Derivation of adult canine intestinal organoids for translational research in gastroenterology. BMC Biol. 2019, 17, 33. [Google Scholar] [CrossRef]
- Drost, J.; Clevers, H. Translational applications of adult stem cell-derived organoids. Development. 2017, 144, 968–975. [Google Scholar] [CrossRef]
- Gabriel, V.; Zdyrski, C.; Sahoo, D.K.; et al. Canine Intestinal Organoids in a Dual-Chamber Permeable Support System. Journal of Visualized Experiments 2022. [Google Scholar] [CrossRef]
- Zhao, J.; Zeng, Z.; Sun, J.; et al. A Novel Model of P-Glycoprotein Inhibitor Screening Using Human Small Intestinal Organoids. Basic Clin Pharmacol Toxicol. 2017, 120, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Biological Transport Phenomena in the Gastrointestinal Tract: Cellular Mechanisms. In Transport Processes in Pharmaceutical Systems; CRC Press, 1999; pp. 163–200. [CrossRef]
- Srinivasan, B.; Kolli, A.R.; Esch, M.B.; Abaci, H.E.; Shuler, M.L.; Hickman, J.J. TEER Measurement Techniques for In Vitro Barrier Model Systems. J Lab Autom. 2015, 20, 107–126. [Google Scholar] [CrossRef]
- Gabriel, V.; Zdyrski, C.; Sahoo, D.K.; et al. Standardization and Maintenance of 3D Canine Hepatic and Intestinal Organoid Cultures for Use in Biomedical Research. J Vis Exp 2022. [Google Scholar] [CrossRef]
- Meyerholz, D.K.; Beck, A.P. Principles and approaches for reproducible scoring of tissue stains in research. Lab Invest. 2018, 98, 844–855. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Zdyrski, C.; Gabriel, V.; Gessler, T.B.; et al. Establishment and characterization of turtle liver organoids provides a potential model to decode their unique adaptations. Communications Biology 2024, 7, 1–19. [Google Scholar] [CrossRef]
- Díaz-Regañón, D.; Gabriel, V.; Livania, V.; et al. Changes of Enterocyte Morphology and Enterocyte: Goblet Cell Ratios in Dogs with Protein-Losing and Non-Protein-Losing Chronic Enteropathies. Vet Sci. 2023, 10, 417. [Google Scholar] [CrossRef] [PubMed]
- Thompson, F.M.; Catto-Smith, A.G.; Moore, D.; Davidson, G.; Cummins, A.G. Epithelial growth of the small intestine in human infants. J Pediatr Gastroenterol Nutr. 1998, 26, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Irvine, J.D.; Takahashi, L.; Lockhart, K.; et al. MDCK (Madin-Darby canine kidney) cells: A tool for membrane permeability screening. J Pharm Sci. 1999, 88, 28–33. [Google Scholar] [CrossRef]
- Cereijido, M.; Robbins, E.S.; Dolan, W.J.; Rotunno, C.A.; Sabatini, D.D. Polarized monolayers formed by epithelial cells on a permeable and translucent support. J Cell Biol. 1978, 77, 853–880. [Google Scholar] [CrossRef] [PubMed]
- Van Breemen, R.B.; Li, Y. Caco-2 cell permeability assays to measure drug absorption. Expert Opin Drug Metab Toxicol. 2005, 1, 175–185. [Google Scholar] [CrossRef]
- Artursson, P. Cell cultures as models for drug absorption across the intestinal mucosa. Crit Rev Ther Drug Carrier Syst. 1991, 8, 305–330. [Google Scholar] [PubMed]
- Hilgendorf, C.; Spahn-Langguth, H.; Regårdh, C.G.; Lipka, E.; Amidon, G.L.; Langguth, P. Caco-2 versus Caco-2/HT29-MTX Co-cultured Cell Lines: Permeabilities Via Diffusion, Inside- and Outside-Directed Carrier-Mediated Transport. J Pharm Sci. 2000, 89, 63–75. [Google Scholar] [CrossRef]
- VanDussen, K.L.; Sonnek, N.M.; Stappenbeck, T.S. L-WRN conditioned medium for gastrointestinal epithelial stem cell culture shows replicable batch-to-batch activity levels across multiple research teams. Stem Cell Res. 2019, 37, 101430. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, D.K.; Martinez, M.N.; Dao, K.; et al. Canine Intestinal Organoids as a Novel In Vitro Model of Intestinal Drug Permeability: A Proof-of-Concept Study. Cells. 2023, 12, 1–31. [Google Scholar] [CrossRef]
| Adult Duodenum | Juvenile Duodenum | Adult Ileum | Juvenile Ileum | Adult Colon | Juvenile Colon | Caco-2 | MDCK | |
|---|---|---|---|---|---|---|---|---|
| Number of values | 290 | 400 | 459 | 300 | 687 | 493 | 99 | 99 |
| Median | 14.2d | 10.8f | 17.4c | 13.5e | 19.7a | 17.7b | 13.8de | 7.5g |
| Interquartile range (IQR) | 6.2 | 3.6 | 5.9 | 7.2 | 6.1 | 5.4 | 6.6 | 2.6 |
| Mean ranks | 1149.0 | 582.1 | 1659.0 | 1078.0 | 2007.0 | 1715.0 | 1070.0 | 161.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




