Submitted:
03 January 2025
Posted:
06 January 2025
You are already at the latest version
Abstract
The enhancement of drug solubility and bioavailability is a significant challenge in pharmaceutical sciences. Solid dispersions have emerged as a promising strategy to address this issue. This research provides a comprehensive review of the advancements in solid dispersion technologies, focusing on the classification and characteristics of different generations of solid dispersions. The study highlights the evolution from first-generation solid dispersions, utilizing crystalline carriers, to fourth-generation systems that incorporate water-insoluble polymers and surfactants to improve drug release and bioavailability. The efficacy of various carriers and techniques, such as hot melt extrusion and solvent evaporation, are critically analyzed. Additionally, the impact of these advancements on poorly soluble anticancer drugs and herbal medicines is discussed, providing insights into future research directions and potential therapeutic applications.
Keywords:
Introduction
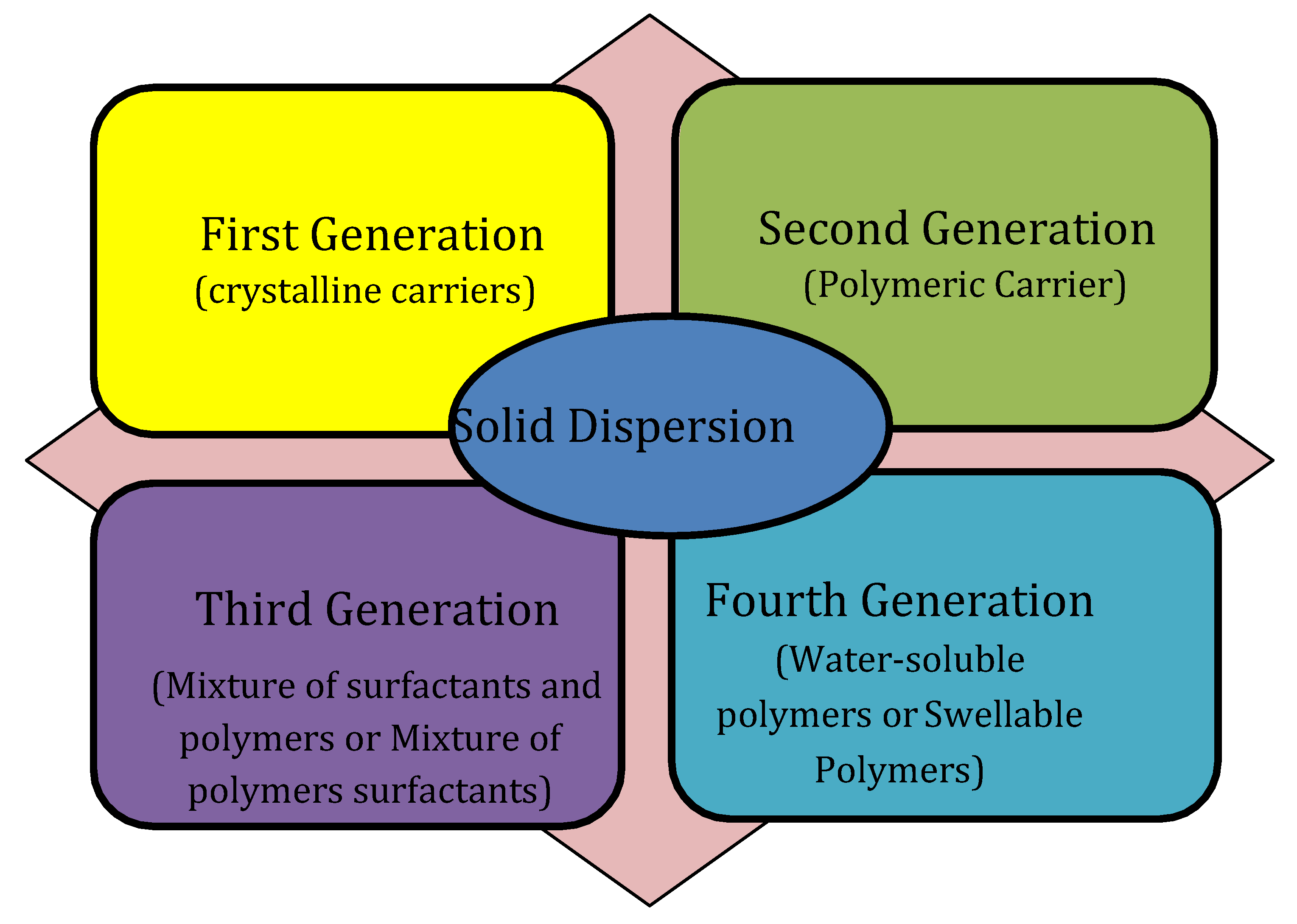
Classification of Solid Dispersions
First-Generation Solid Dispersions
Second-Generation Solid Dispersions
Third-Generation Solid Dispersions
Fourth-Generation Solid Dispersions
Factors Influencing SDs in Drug Development
Drug Release from SDs
Advantages of Solid Dispersions
- [1]
- Drugs that interact with water-soluble transporters can speed up absorption and increase bioavailability by reducing aggregation and releasing in a supersaturation situation [23].
- [2]
- Solid dispersion can increase the surface area and increase the wettability of medicines, enhancing their aqueous solubility.
- [3]
- Compared to other forms, including liquid goods, solid dispersion could be created in the form of a solid oral dosage, making it more practical for patients.
- [4]
- Additionally, solid dispersion outperformed co-crystallization, salt formulation, and other techniques. For instance, cationic or anionic ionized active pharmaceutical ingredients (APIs) are included in salt formulations. These formulations are common in the pharmaceutical industry since there are several methods to build them such that they have the desired pharmacological properties. The phase of the dissociation or stability issue is intrinsic in salt generation or co-crystallization because not all the drugs can ionize with all cations or anions. Better regulatory inspection for sturdy acid salts derived from alkyl alcohols; decreased solubility and dissolution rate; decreased relative bioavailability (common ions effect for HCl salts); and increased hygroscopicity; for example, spray-drying/lyophilization can isolate strong acid salts for Na+ and K+ salts. The problems could be avoided by using solid dispersion to make salt formulation.
- [5]
- Practically speaking, total absorption is required for medications to dissolve before they may have the intended therapeutic impact when taken orally. The majority of anticancer medications have poor water solubility, which results in low bioavailability and considerable blood concentration variability. Solid dispersion, a method that encourages supersaturated drug dissolution and, as a result, increases in vivo absorption, can help with the limiting of drug dissolution.
Disadvantages of Solid Dispersions
- (1)
- As solid dispersions age, their crystallinity changes and their rate of disintegration slows down.
- (2)
- Solid dispersion is temperature and humidity sensitive during storage due to its thermodynamic instability. By raising general molecular mobility, lowering the transition glass temperature (Tg), or interference with interacting between the drug and the transporter, these components may encourage separation of phases and crystalline formation of solid dispersion, which lowers the drug's ability to dissolve and dissolving rate.
- (3)
- Cancer patients should continue taking anticancer medications while receiving treatment. However, the quality and efficacy of medications may be impacted by the instability of solid dispersion during storage.
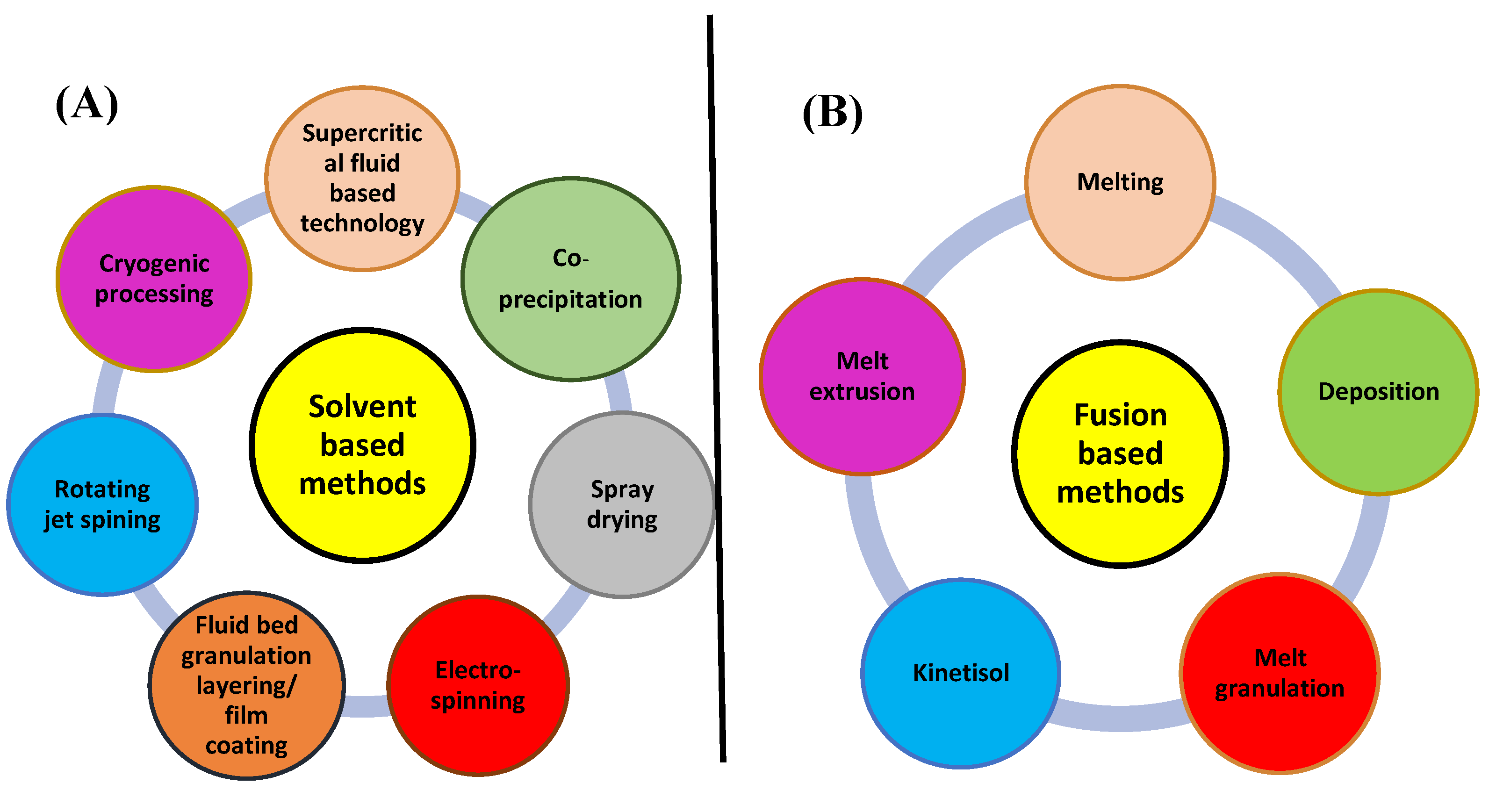
Methods for Solid Dispersion Preparation
Melting Methods
Technique of Solvent Evaporation
Melting Solvent Method (Melt Evaporation)
Melt Agglomeration Process
Hot-Melt Extrusion Method
Lyophilization Techniques
Electrospinning Method
Co-Precipitation
Supercritical Fluid (SCF) Technology
Spray-Drying Technique
Kneading Technique
Characterization of Solid Dispersion
Physical Composition
Drug-Transporter Defeasibility
Stability
Uncrystalline Content
Dissolution Augmentation
Industrial and Laboratory Scale Manufacturing Procedures
Development of Poorly Soluble Anticancer Medicines by the Application of Solid Dispersion
Solid Herbal Medicine Dispersion
Practical Limitations in Solid Dispersion Technique [94,95,96]:
- A.
-
Issue with the creation of the dosage form
- Bad flow and compressibility: Grinding and sifting are difficult when solid dispersion is present. It also shows poor stability and compressibility. Drug granulation in-situ is used to address this problem.
- Pasting the solid dispersion grains to die and punches: Adhering to die and punch was noticed during compression. This obstacle could be resolved by placing tiny pieces of grease-proof paper among the surface of the metal and the granules. The granules and metal surface were therefore not in direct touch.
- B.
-
Issue with production and scalability
- Chance of condensation of moisture over solid dispersion during cooling: Moisture can condense above solid dispersion during evaporation. The revolving or surface-moving belt utilized in continually cooling operations can be employed to address this problem.
- Reliability of physicochemical parameters: The preparation circumstances, including the heating rate, maximum temperature employed, cooling rate and technique, pulverization process, and size of particles, significantly affect the physicochemical characteristics of solid dispersions.
- C.
- Issue related with stability
Some Challenges in the Solid Dispersion Development
Novel Technologies Associated with Solid Dispersions
Potential for Solid Dispersion in the Future
Conclusion
References
- Wadke, D.A.; Serajuddin, A.T.M.; Jacobson, H. Preformulation testing. Pharm Dos forms Tablets. 1989, 1, 1–73. [Google Scholar]
- Verma, S.; Jain, D.; Shukla, S.B.; Yadav, P.; Sonkar, N.; Soloman, J.A. Role of Modular in Effective Drug Delivery System: A Review. Res J Pharm Dos Forms Technol. 2010, 2, 370–373. [Google Scholar]
- Pawar, S.R.; Barhate, S.D. Solubility enhancement (Solid Dispersions) novel boon to increase bioavailability. J Drug Deliv Ther. 2019, 9, 583–590. [Google Scholar] [CrossRef]
- Pudipeddi, M.; Serajuddin, A.T.M.; Shah, A.V.; Mufson, D. Integrated drug product development: From lead candidate selection to life-cycle management. In: Drug Discovery and Development, Third Edition. CRC Press; 2019. p. 223–61.
- Vasconcelos, T.; Sarmento, B.; Costa, P. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov Today. 2007, 12, 1068–1075. [Google Scholar] [CrossRef] [PubMed]
- Sekiguchi, K.; Obi, N. Studies on Absorption of Eutectic Mixture. I. A Comparison of the Behavior of Eutectic Mixture of Sulfathiazole and that of Ordinary Sulfathiazole in Man. Chem Pharm Bull. 1961, 9, 866–872. [Google Scholar]
- LEVYG Effect of particle size on dissolution and gastrointestinal absorption rates of pharmaceuticals. Am J Pharm Sci Support Public Health. 1963, 135, 78–92.
- Patel, R.D.; Raval, M.K.; Sheth, N.R. Formation of Diacerein− fumaric acid eutectic as a multi-component system for the functionality enhancement. J Drug Deliv Sci Technol. 2020, 58, 101562. [Google Scholar] [CrossRef]
- Van Den Mooter, G. The use of amorphous solid dispersions: A formulation strategy to overcome poor solubility and dissolution rate. Drug Discov Today Technol. 2012, 9, e79–85. [Google Scholar] [CrossRef]
- Mishra, D.K.; Dhote, V.; Bhargava, A.; Jain, D.K.; Mishra, P.K. Amorphous solid dispersion technique for improved drug delivery: basics to clinical applications. Drug Deliv Transl Res. 2015, 5, 552–565. [Google Scholar] [CrossRef]
- Vemula, V.R.; Lagishetty, V.; Lingala, S. Solubility enhancement techniques. Int J Pharm Sci Rev Res. 2010, 5, 41–51. [Google Scholar]
- Qi, S.; Belton, P.; Nollenberger, K.; Clayden, N.; Reading, M.; Craig, D.Q.M. Characterisation and prediction of phase separation in hot-melt extruded solid dispersions: A thermal, microscopic and NMR relaxometry study. Pharm Res. 2010, 27, 1869–1883. [Google Scholar] [CrossRef] [PubMed]
- Vasanthavada, M.; Tong, W.Q.; Joshi, Y.; Kislalioglu, M.S. Phase behavior of amorphous molecular dispersions I: Determination of the degree and mechanism of solid solubility. Pharm Res. 2004, 21, 1598–1606. [Google Scholar] [CrossRef] [PubMed]
- Konno, H.; Handa, T.; Alonzo, D.E.; Taylor, L.S. Effect of polymer type on the dissolution profile of amorphous solid dispersions containing felodipine. Eur J Pharm Biopharm. 2008, 70, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Inbar, P.; Chokshi, H.P.; Malick, A.W.; Choi, D.S. Prediction of the thermal phase diagram of amorphous solid dispersions by flory-huggins theory. J Pharm Sci. 2011, 100, 3196–3207. [Google Scholar] [CrossRef]
- Marsac, P.J.; Li, T.; Taylor, L.S. Estimation of drug-polymer miscibility and solubility in amorphous solid dispersions using experimentally determined interaction parameters. Pharm Res. 2009, 26, 139–151. [Google Scholar] [CrossRef]
- Rumondor, A.C.F.; Ivanisevic, I.; Bates, S.; Alonzo, D.E.; Taylor, L.S. Evaluation of drug-polymer miscibility in amorphous solid dispersion systems. Pharm Res. 2009, 26, 2523–2534. [Google Scholar] [CrossRef]
- Sun, Y.E.; Tao, J.; Zhang, G.G.Z.; Yu, L. Solubilities of crystalline drugs in polymers: An improved analytical method and comparison of solubilities of indomethacin and nifedipine in PVP, PVP/VA, and PVAc. J Pharm Sci. 2010, 99, 4023–4031. [Google Scholar] [CrossRef]
- Lin, D.; Huang, Y. A thermal analysis method to predict the complete phase diagram of drug-polymer solid dispersions. Int J Pharm. 2010, 399, 109–115. [Google Scholar] [CrossRef]
- Qian F, Huang J, Zhu Q, Haddadin R, Gawel J, Garmise R, et al. Is a distinctive single Tg a reliable indicator for the homogeneity of amorphous solid dispersion? Int J Pharm. 2010, 395, 232–235.
- Dannenfelser, R.M.; He, H.; Joshi, Y.; Bateman, S.; Serajuddin, A.T.M. Development of Clinical Dosage Forms for a Poorly Water Soluble Drug I: Application of Polyethylene Glycol-Polysorbate 80 Solid Dispersion Carrier System. J Pharm Sci. 2004, 93, 1165–1175. [Google Scholar] [CrossRef]
- Ghebremeskel, A.N.; Vemavarapu, C.; Lodaya, M. Use of surfactants as plasticizers in preparing solid dispersions of poorly soluble API: Selection of polymer-surfactant combinations using solubility parameters and testing the processability. Int J Pharm. 2007, 328, 119–129. [Google Scholar] [CrossRef]
- Vo, C.L.N.; Park, C.; Lee, B.J. Current trends and future perspectives of solid dispersions containing poorly water-soluble drugs. Eur J Pharm Biopharm. 2013, 85, 799–813. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, B.; Kaur, R.; Kour, S.; Behl, H.; Kour, S. Solid dispersion: an evolutionary approach for solubility enhancement of poorly water soluble drugs. Int J Recent Adv Pharm Res. 2012, 2, 1–16. [Google Scholar]
- Guo, S.; Wang, G.; Wu, T.; Bai, F.; Xu, J.; Zhang, X. Solid dispersion of berberine hydrochloride and Eudragit® S100: Formulation, physicochemical characterization and cytotoxicity evaluation. J Drug Deliv Sci Technol. 2017, 40, 21–27. [Google Scholar] [CrossRef]
- Allen, L.V.; Popovich, N.G.; Ansel, H.C. Ansel’s pharmaceutical dosage forms and drug delivery systems: Ninth edition. Ansel’s Pharmaceutical Dosage Forms and Drug Delivery Systems: Ninth Edition. Lippincott Williams & Wilkins; 2012. 1–710 p.
- Zografi, G.; Newman, A. Interrelationships Between Structure and the Properties of Amorphous Solids of Pharmaceutical Interest. J Pharm Sci. 2017, 106, 5–27. [Google Scholar] [CrossRef]
- Cid, A.G.; Simonazzi, A.; Palma, S.D.; Bermúdez, J.M. Solid dispersion technology as a strategy to improve the bioavailability of poorly soluble drugs. Ther Deliv. 2019, 10, 363–382. [Google Scholar] [CrossRef]
- Craig, D.Q.M. The mechanisms of drug release from solid dispersions in water-soluble polymers. Int J Pharm. 2002, 231, 131–144. [Google Scholar] [CrossRef]
- Fernández-Colino, A.; Bermudez, J.M.; Arias, F.J.; Quinteros, D.; Gonzo, E. Development of a mechanism and an accurate and simple mathematical model for the description of drug release: Application to a relevant example of acetazolamide-controlled release from a bio-inspired elastin-based hydrogel. Mater Sci Eng C. 2016, 61, 286–292. [Google Scholar] [CrossRef]
- Romero, A.I.; Villegas, M.; Cid, A.G.; Parentis, M.L.; Gonzo, E.E.; Bermúdez, J.M. Validation of kinetic modeling of progesterone release from polymeric membranes. Asian J Pharm Sci. 2018, 13, 54–62. [Google Scholar] [CrossRef]
- Simonazzi, A.; Davies, C.; Cid, A.G.; Gonzo, E.; Parada, L.; Bermúdez, J.M. Preparation and Characterization of Poloxamer 407 Solid Dispersions as an Alternative Strategy to Improve Benznidazole Bioperformance. J Pharm Sci. 2018, 107, 2829–2836. [Google Scholar] [CrossRef]
- Tran, P.; Pyo, Y.C.; Kim, D.H.; Lee, S.E.; Kim, J.K.; Park, J.S. Overview of the manufacturing methods of solid dispersion technology for improving the solubility of poorly water-soluble drugs and application to anticancer drugs. Pharmaceutics. 2019, 11, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Alwossabi, A.M.; Elamin, E.S.; Ahmed, E.M.M.; Abdelrahman, M. Solubility enhancement of some poorly soluble drugs by solid dispersion using Ziziphus spina-christi gum polymer: Solubility enhancement of some poorly soluble drugs by solid dispersion. Saudi Pharm J [Internet]. 2022, 30, 711–725. [Google Scholar] [CrossRef] [PubMed]
- Schaber, S.D.; Gerogiorgis, D.I.; Ramachandran, R.; Evans, J.M.B.; Barton, P.I.; Trout, B.L. Economic analysis of integrated continuous and batch pharmaceutical manufacturing: A case study. Ind Eng Chem Res. 2011, 50, 10083–10092. [Google Scholar] [CrossRef]
- Hurter, Patricia, Thomas, Hayden, Nadig, David, Emiabata-Smith, David, Paone A. Implementing Continuous Manufacturing to Streamline and Accelerate Drug Development. AAPS Newsmag. 2013, 16, 15–19.
- Hallouard, F.; Mehenni, L.; Lahiani-Skiba, M.; Anouar, Y.; Skiba, M. Solid Dispersions for Oral Administration: An Overview of the Methods for their Preparation. Curr Pharm Des. 2016, 22, 4942–4958. [Google Scholar] [CrossRef]
- Bley, H.; Fussnegger, B.; Bodmeier, R. Characterization and stability of solid dispersions based on PEG/polymer blends. Int J Pharm. 2010, 390, 165–173. [Google Scholar] [CrossRef]
- Yao, W.W.; Bai, T.C.; Sun, J.P.; Zhu, C.W.; Hu, J.; Zhang, H.L. Thermodynamic properties for the system of silybin and poly(ethylene glycol) 6000. Thermochim Acta. 2005, 437, 17–20. [Google Scholar] [CrossRef]
- Hurley, D.; Potter, C.B.; Walker, G.M.; Higginbotham, C.L. Investigation of Ethylene Oxide-co-propylene Oxide for Dissolution Enhancement of Hot-Melt Extruded Solid Dispersions. J Pharm Sci. 2018, 107, 1372–1382. [Google Scholar] [CrossRef]
- Breitenbach, J. Melt extrusion: from process to drug delivery technology. Eur J Pharm Biopharm. 2002, 54, 107–117. [Google Scholar] [CrossRef]
- Seo, A.; Holm, P.; Kristensen, H.G.; Schæfer, T. The preparation of agglomerates containing solid dispersions of diazepam by melt agglomeration in a high shear mixer. Int J Pharm. 2003, 259, 161–171. [Google Scholar] [CrossRef]
- Crowley, M.M.; Zhang, F.; Repka, M.A.; Thumma, S.; Upadhye, S.B.; Battu, S.K.; et al. Pharmaceutical applications of hot-melt extrusion: Part, I. Drug Dev Ind Pharm. 2007, 33, 909–926. [Google Scholar] [CrossRef]
- Andrews, G.P.; Jones, D.S.; Diak, O.A.; McCoy, C.P.; Watts, A.B.; McGinity, J.W. The manufacture and characterisation of hot-melt extruded enteric tablets. Eur J Pharm Biopharm. 2008, 69, 264–273. [Google Scholar] [CrossRef]
- Todd, D.B. Plastic compounding equipment and processing. Vol. 14, IEEE Electrical Insulation Magazine. Hanser Publishers; 1998. 40 p.
- Tachibana, T.; Nakamura, A. A methode for preparing an aqueous colloidal dispersion of organic materials by using water-soluble polymers: Dispersion of Β-carotene by polyvinylpyrrolidone. Kolloid-Zeitschrift Zeitschrift für Polym. 1965, 203, 130–133. [Google Scholar] [CrossRef]
- Mayersohn, M.; Gibaldi, M. New method of solid-state dispersion for increasing dissolution rates. J Pharm Sci. 1966, 55, 1323–1324. [Google Scholar] [CrossRef] [PubMed]
- Oliveira V da S, Dantas ED, Queiroz AT de S, Oliveira JW de F, da Silva M de S, Ferreira PG, et al. Novel solid dispersions of naphthoquinone using different polymers for improvement of antichagasic activity. Pharmaceutics. 2020, 12, 1–15.
- Adeli, E. Preparation and evaluation of azithromycin binary solid dispersions using various polyethylene glycols for the improvement of the drug solubility and dissolution rate. Brazilian J Pharm Sci. 2016, 52, 1–13. [Google Scholar] [CrossRef]
- Shuai S, Yue S, Huang Q, Wang W, Yang J, Lan K, et al. Preparation, characterization and in vitro/vivo evaluation of tectorigenin solid dispersion with improved dissolution and bioavailability. Eur J Drug Metab Pharmacokinet. 2016, 41, 413–422.
- Daravath, B.; Tadikonda, R.R.; Vemula, S.K. Formulation and pharmacokinetics of gelucire solid dispersions of flurbiprofen. Drug Dev Ind Pharm. 2015, 41, 1254–1262. [Google Scholar] [CrossRef]
- Mustapha O, Kim KS, Shafique S, Kim DS, Jin SG, Seo YG, et al. Comparison of three different types of cilostazol-loaded solid dispersion: Physicochemical characterization and pharmacokinetics in rats. Colloids Surfaces B Biointerfaces. 2017, 154, 89–95.
- Zhang H, Cui D, Wang B, Han Y-H, Balimane P, Yang Z, et al. Pharmacokinetic drug interactions involving 17α-ethinylestradiol: a new look at an old drug. Clin Pharmacokinet. 2007, 46, 133–57.
- Frizon, F.; de Oliveira Eloy, J.; Donaduzzi, C.M.; Mitsui, M.L.; Marchetti, J.M. Dissolution rate enhancement of loratadine in polyvinylpyrrolidone K-30 solid dispersions by solvent methods. Powder Technol. 2013, 235, 532–9. [Google Scholar] [CrossRef]
- Miao L, Liang Y, Pan W, Gou J, Yin T, Zhang Y, et al. Effect of supersaturation on the oral bioavailability of paclitaxel/polymer amorphous solid dispersion. Drug Deliv Transl Res. 2019, 9, 344–356.
- Chen Y, Shi Q, Chen Z, Zheng J, Xu H, Li J, et al. Preparation and characterization of emulsified solid dispersions containing docetaxel. Arch Pharm Res. 2011, 34, 1909–1917.
- DHILLONV; TYAGIR Solid Dispersion: a Fruitful Approach for Improving the Solubility and Dissolution Rate of Poorly Soluble Drugs. J Drug Deliv Ther. 2012, 2, 5–14.
- Goldberg, A.H.; Gibaldi, M.; Kanig, J.L. Increasing dissolution rates and gastrointestinal absorption of drugs via solid solutions and eutectic mixtures III: Experimental evaluation of griseofulvin—succinic acid solid solution. J Pharm Sci. 1966, 55, 487–492. [Google Scholar] [CrossRef]
- Chen, H.; Jiang, G.; Ding, F. Monolithic osmotic tablet containing solid dispersion of 10-hydroxycamptothecin. Drug Dev Ind Pharm. 2009, 35, 131–137. [Google Scholar] [CrossRef]
- Van Drooge, D.J.; Hinrichs, W.L.J.; Visser, M.R.; Frijlink, H.W. Characterization of the molecular distribution of drugs in glassy solid dispersions at the nano-meter scale, using differential scanning calorimetry and gravimetric water vapour sorption techniques. Int J Pharm. 2006, 310, 220–229. [Google Scholar] [CrossRef]
- Vilhelmsen, T.; Eliasen, H.; Schæfer, T. Effect of a melt agglomeration process on agglomerates containing solid dispersions. Int J Pharm. 2005, 303, 132–142. [Google Scholar] [CrossRef]
- Kaur, J.; Aggarwal, G.; Singh, G.; Rana, A.C. Improvement of drug solubility using solid dispersion. Int J Pharm Pharm Sci. 2012, 4, 47–53. [Google Scholar]
- Genina, N.; Hadi, B.; Löbmann, K. Hot Melt Extrusion as Solvent-Free Technique for a Continuous Manufacturing of Drug-Loaded Mesoporous Silica. J Pharm Sci. 2018, 107, 149–155. [Google Scholar] [CrossRef]
- Betageri, G.V.; Makarla, K.R. Enhancement of dissolution of glyburide by solid dispersion and lyophilization techniques. Int J Pharm. 1995, 126, 155–160. [Google Scholar] [CrossRef]
- Altamimi, M.A.; Neau, S.H. Investigation of the in vitro performance difference of drug-Soluplus® and drug-PEG 6000 dispersions when prepared using spray drying or lyophilization. Saudi Pharm J. 2017, 25, 419–439. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.G.; Li, J.J.; Williams, G.R.; Zhao, M. Electrospun amorphous solid dispersions of poorly water-soluble drugs: A review. J Control Release. 2018, 292, 91–110. [Google Scholar] [CrossRef] [PubMed]
- Pamudji JS, Khairurrijal, Mauludin R, Sudiati T, Evita M. PVA-ketoprofen nanofibers manufacturing using electrospinning method for dissolution improvement of ketoprofen. In: Materials Science Forum. Trans Tech Publ; 2013. p. 166–75.
- Sonali, D.; Tejal, S.; Vaishali, T.; Tejal, G. Silymarin-solid dispersions: Characterization and influence of preparation methods on dissolution. Acta Pharm. 2010, 60, 427–443. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liu, Y.; Liu, T.; Zhao, L.; Zhao, J.; Feng, N. Development and in-vivo assessment of the bioavailability of oridonin solid dispersions by the gas anti-solvent technique. Int J Pharm. 2011, 411, 172–177. [Google Scholar] [CrossRef]
- Riekes MK, Caon T, da Silva J, Sordi R, Kuminek G, Bernardi LS, et al. Enhanced hypotensive effect of nimodipine solid dispersions produced by supercritical CO2 drying. Powder Technol. 2015, 278, 204–10.
- Jun SW, Kim M-S, Jo GH, Lee S, Woo JS, Park J-S, et al. Cefuroxime axetil solid dispersions prepared using solution enhanced dispersion by supercritical fluids. J Pharm Pharmacol. 2010, 57, 1529–1537.
- Abuzar SM, Hyun SM, Kim JH, Park HJ, Kim MS, Park JS, et al. Enhancing the solubility and bioavailability of poorly water-soluble drugs using supercritical antisolvent (SAS) process. Int J Pharm. 2018, 538, 1–13.
- Singh, A.; Van den Mooter, G. Spray drying formulation of amorphous solid dispersions. Adv Drug Deliv Rev. 2016, 100, 27–50. [Google Scholar] [CrossRef]
- Herbrink, M.; Schellens, J.H.M.; Beijnen, J.H.; Nuijen, B. Improving the solubility of nilotinib through novel spray-dried solid dispersions. Int J Pharm. 2017, 529, 294–302. [Google Scholar] [CrossRef]
- Dhandapani, N.V.; El-gied, A.A. Solid dispersions of cefixime using β-cyclodextrin : Characterization and in vitro evaluation. Int J Pharmacol Pharm Sci. 2016, 10, 1523–1527. [Google Scholar]
- Liu, X.; Feng, X.; Williams, R.O.; Zhang, F. Characterization of amorphous solid dispersions. J Pharm Investig. 2018, 48, 19–41. [Google Scholar] [CrossRef]
- Lamm, M.S.; DiNunzio, J.; Khawaja, N.N.; Crocker, L.S.; Pecora, A. Assessing mixing quality of a copovidone-TPGS hot melt extrusion process with atomic force microscopy and differential scanning calorimetry. Aaps Pharmscitech. 2016, 17, 89–98. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Gornstein, E.L.; Schwarz, T.L. Neurotoxic mechanisms of paclitaxel are local to the distal axon and independent of transport defects. Exp Neurol. 2017, 288, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Gréen, H.; Khan, M.S.; Jakobsen-Falk, I.; Åvall-Lundqvist, E.; Peterson, C. Impact of CYP3A5*3 and CYP2C8-HapC on paclitaxel/carboplatin-induced myelosuppression in patients with ovarian cancer. J Pharm Sci. 2011, 100, 4205–4209. [Google Scholar] [CrossRef]
- Tétu, P.; Hamelin, A.; Moguelet, P.; Barbaud, A.; Soria, A. Management of hypersensitivity reactions to Tocilizumab. Clin Exp Allergy. 2018, 48, 749–752. [Google Scholar] [CrossRef]
- Thanki, K.; Gangwal, R.P.; Sangamwar, A.T.; Jain, S. Oral delivery of anticancer drugs: Challenges and opportunities. J Control Release. 2013, 170, 15–40. [Google Scholar] [CrossRef]
- Sawicki, E.; Schellens, J.H.M.; Beijnen, J.H.; Nuijen, B. Inventory of oral anticancer agents: Pharmaceutical formulation aspects with focus on the solid dispersion technique. Cancer Treat Rev. 2016, 50, 247–263. [Google Scholar] [CrossRef]
- Licciardi, P.V.; Underwood, J.R. Plant-derived medicines: A novel class of immunological adjuvants. Int Immunopharmacol. 2011, 11, 390–398. [Google Scholar] [CrossRef]
- Choudhary, N.; Sekhon, B. An overview of advances in the standardization of herbal drugs. J Pharm Educ Res. 2011, 2, 55–70. [Google Scholar]
- Atmakuri, L.R.; Dathi, S. Available online through Current Trends in Herbal Medicines. J fo Pharm Res. 2010, 3, 109–113. [Google Scholar]
- Yadav, N.P.; Dixit, V.K. Recent approaches in herbal drug standardization. Int J Integr Biol. 2008, 2, 195–203. [Google Scholar]
- Liang, Y.-Z.; Xie, P.-S.; Chan, K. Chromatographic Fingerprinting and Metabolomics for Quality Control of TCM. Comb Chem High Throughput Screen. 2010, 13, 943–953. [Google Scholar] [CrossRef]
- Musthaba, S.M.; Baboota, S.; Ahmed, S.; Ahuja, A.; Ali, J. Status of novel drug delivery technology for phytotherapeutics. Expert Opin Drug Deliv. 2009, 6, 625–637. [Google Scholar] [CrossRef]
- Ireson C, Orr S, Jones DJL, Verschoyle R, Lim CK, Luo JL, et al. Characterization of metabolites of the chemopreventive agent curcumin in human and rat hepatocytes and in the rat in vivo, and evaluation of their ability to inhibit phorbol ester-induced prostaglandin E2production. Cancer Res. 2001, 61, 1058–1064.
- Kelloff, G.J.; Boone, C.W.; Crowell, J.A.; Steele, V.E.; Lubet, R.; Sigman, C.C. Chemopreventive Drug Development: Perspectives and Progress. Cancer Epidemiol Biomarkers Prev. 1994, 3, 85–98. [Google Scholar]
- Seo, S.W.; Han, H.K.; Chun, M.K.; Choi, H.K. Preparation and pharmacokinetic evaluation of curcumin solid dispersion using Solutol ® HS15 as a carrier. Int J Pharm. 2012, 424, 18–25. [Google Scholar] [CrossRef]
- Li, B.; Wen, M.; Li, W.; He, M.; Yang, X.; Li, S. Preparation and characterization of baicalin-poly-vinylpyrrolidone coprecipitate. Int J Pharm. 2011, 408, 91–96. [Google Scholar] [CrossRef]
- Serajuddin, A.T.M. Solid dispersion of poorly water-soluble drugs: Early promises, subsequent problems, and recent breakthroughs. J Pharm Sci. 1999, 88, 1058–1066. [Google Scholar] [CrossRef]
- Argade PS, Magar DD, Saudagar RB. 9. Solid Dispersion: Solubility Enhancement Technique for poorly water soluble Drugs. J Adv Pharm Educ Res [Internet]. 2013, 3, 427–439.
- Ramesh, V.; Meenakshi, S. Enhancement of solubility for poorly water soluble drugs by using solid dsipersion technology. Int J Pharm Res Bio-Science. 2016, 5, 47–74. [Google Scholar]
- Janssens, S.; Van den Mooter, G. Review: physical chemistry of solid dispersions. J Pharm Pharmacol. 2009, 61, 1571–1586. [Google Scholar] [CrossRef] [PubMed]
- Sheen, P.C.; Khetarpal, V.K.; Cariola, C.M.; Rowlings, C.E. Formulation studies of a poorly water-soluble drug in solid dispersions to improve bioavailability. Int J Pharm. 1995, 118, 221–227. [Google Scholar] [CrossRef]
- Enose, A.A.; Dasan, P. Formulation, Characterization and Pharmacokinetic Evaluation of Telmisartan Solid Dispersions. J Mol Pharm Org Process Res. 2016, 4, 1. [Google Scholar] [CrossRef]
- Zawar, L.; Bari, S. Microwave Induced Solid Dispersion as a Novel Technique for Enhancing Dissolution Rate of Repaglinide. Adv Pharmacol Pharm. 2013, 1, 95–101. [Google Scholar] [CrossRef]
- Singh G, Sharma S, Gupta G Das. Extensive Diminution of Particle Size and Amorphization of a Crystalline Drug Attained by Eminent Technology of Solid Dispersion: A Comparative Study. AAPS PharmSciTech. 2017, 18, 1770–1784.
- Usmanova LS, Ziganshin MA, Gorbatchuk VV, Ziganshina SA, Bizyaev DA, Bukharaev AA, et al. A study of the formation of magnetically active solid dispersions of phenacetin using atomic and magnetic force microscopy. J Adv Pharm Technol Res. 2017, 8, 2–7.
- Duarte Í, Corvo ML, Serôdio P, Vicente J, Pinto JF, Temtem M. Production of nano-solid dispersions using a novel solvent-controlled precipitation process — Benchmarking their in vivo performance with an amorphous micro-sized solid dispersion produced by spray drying. Eur J Pharm Sci. 2016, 93, 203–214.
- Lee SN, Poudel BK, Tran TH, Marasini N, Pradhan R, Lee YI, et al. A novel surface-attached carvedilol solid dispersion with enhanced solubility and dissolution. Arch Pharm Res. 2013, 36, 79–85.
| Drug | Polymer | Trade name | Dosage form | Processing technology | Manufacturer |
|---|---|---|---|---|---|
| Verapamil | HPC/ HPMC | Isoptin SR ® | Tablet | Hot melt extrusion | Abbot |
| Nabilone | PVP | Cesamet ® | Tablet Capsule | Solvent evaporation | Valeant |
| Itraconazole | HPMC | Sporanox ® | Capsule | Fluid-bed bead | Janssen |
| Troglitazone | PVP | Rezulin ® † | Tablet | Hot melt extrusion | Pfizer |
| Ritonavir/ Lopinavir | PVP-VA 64 | Kaletra ® | Tablet | Hot melt extrusion | Abbot |
| Everolimus | HPMC | Zortress ® | Tablet | Spray drying | Novartis |
| Ritonavir | HPMC | Onmel ® | Tablet | Hot melt extrusion | Stiefel |
| Vemurafenib | HPMC-AS | Zelboraf ® | Tablet | Solvent- controlled precipitation | Roche |
| Ivacaftor | HPMC-AS | Kalydeco ® | Tablet | Spray drying | Vertex |
| Rosuvastatin | HPMC | Crestor ® | Tablet | Spray drying | Astra Zeneca |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




