Submitted:
04 January 2025
Posted:
06 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. DNA Vaccine Plasmid Construction and Verification
2.3. Mammalian Cell Transfection
2.4. Western Blot
2.5. Vaccine Administration
2.6. Enzyme-Linked Immunosorbent Assay (ELISA)
2.7. Lymphocyte Isolation
2.8. T-Cell Stimulation and Flow Cytometry
2.9. Statistics
3. Results
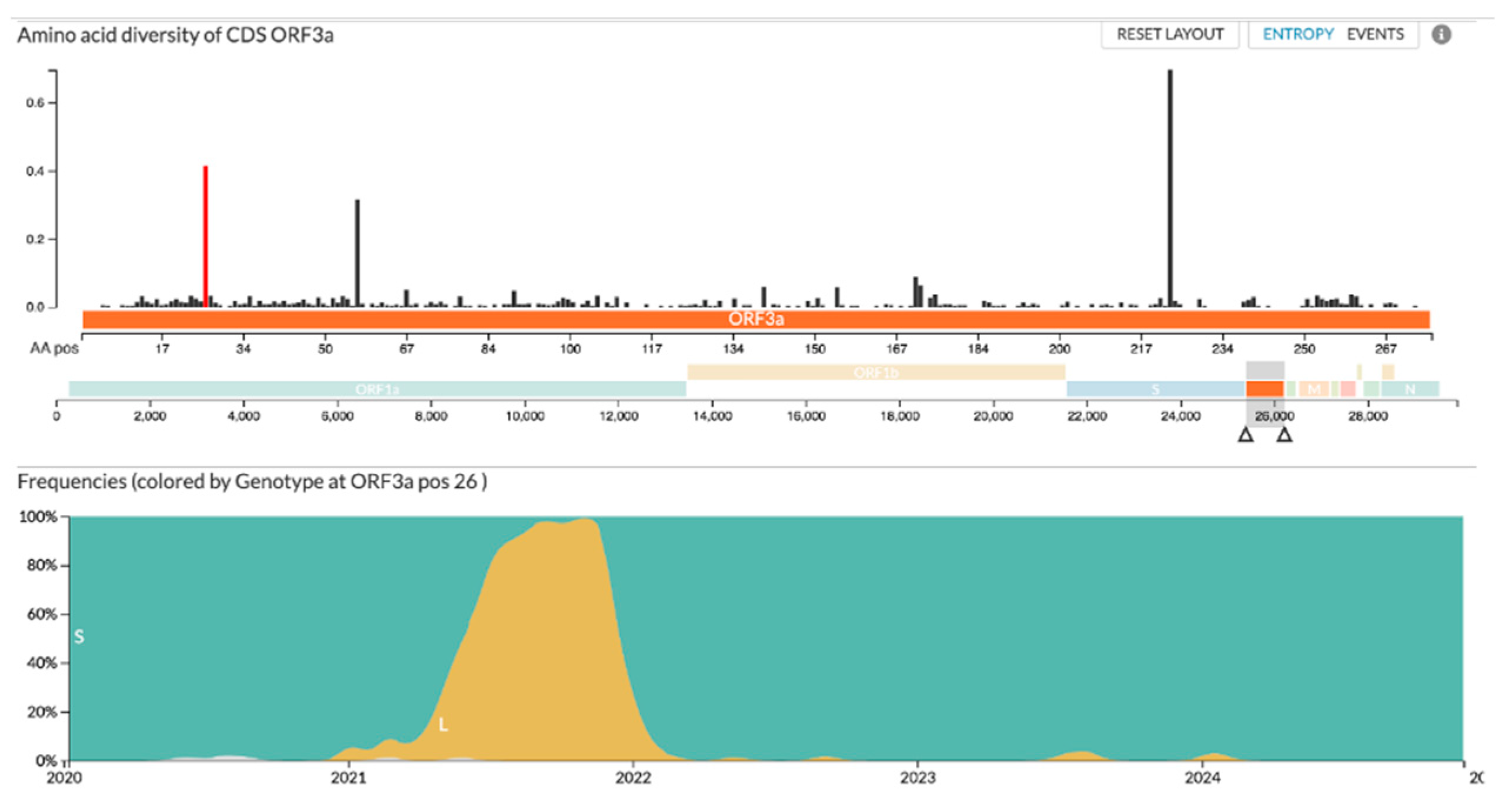
3.1. Antigen Evolution
3.2. DNA Vaccine Creation
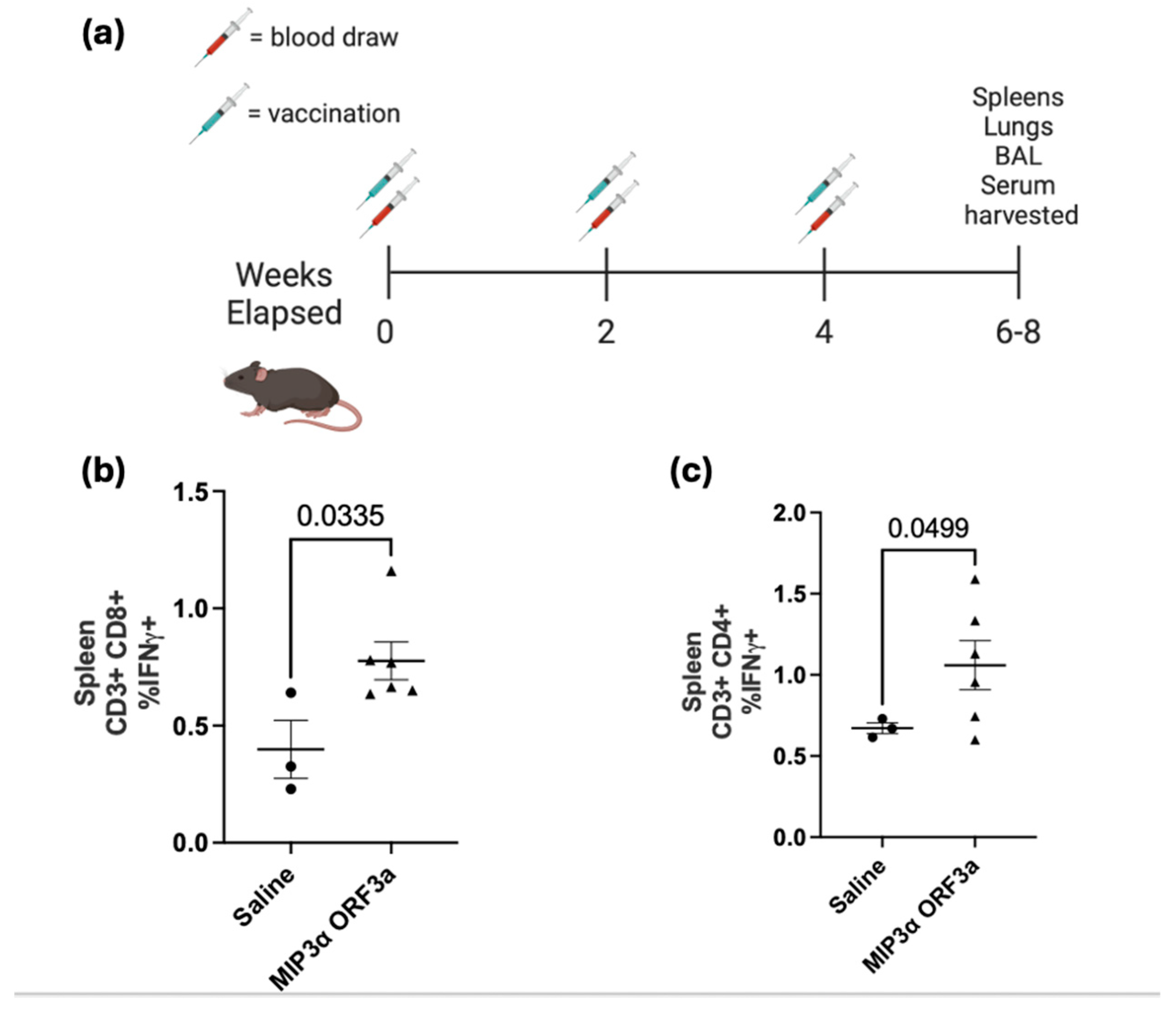
3.3. DNA Intramuscular Vaccine Immunogenicity
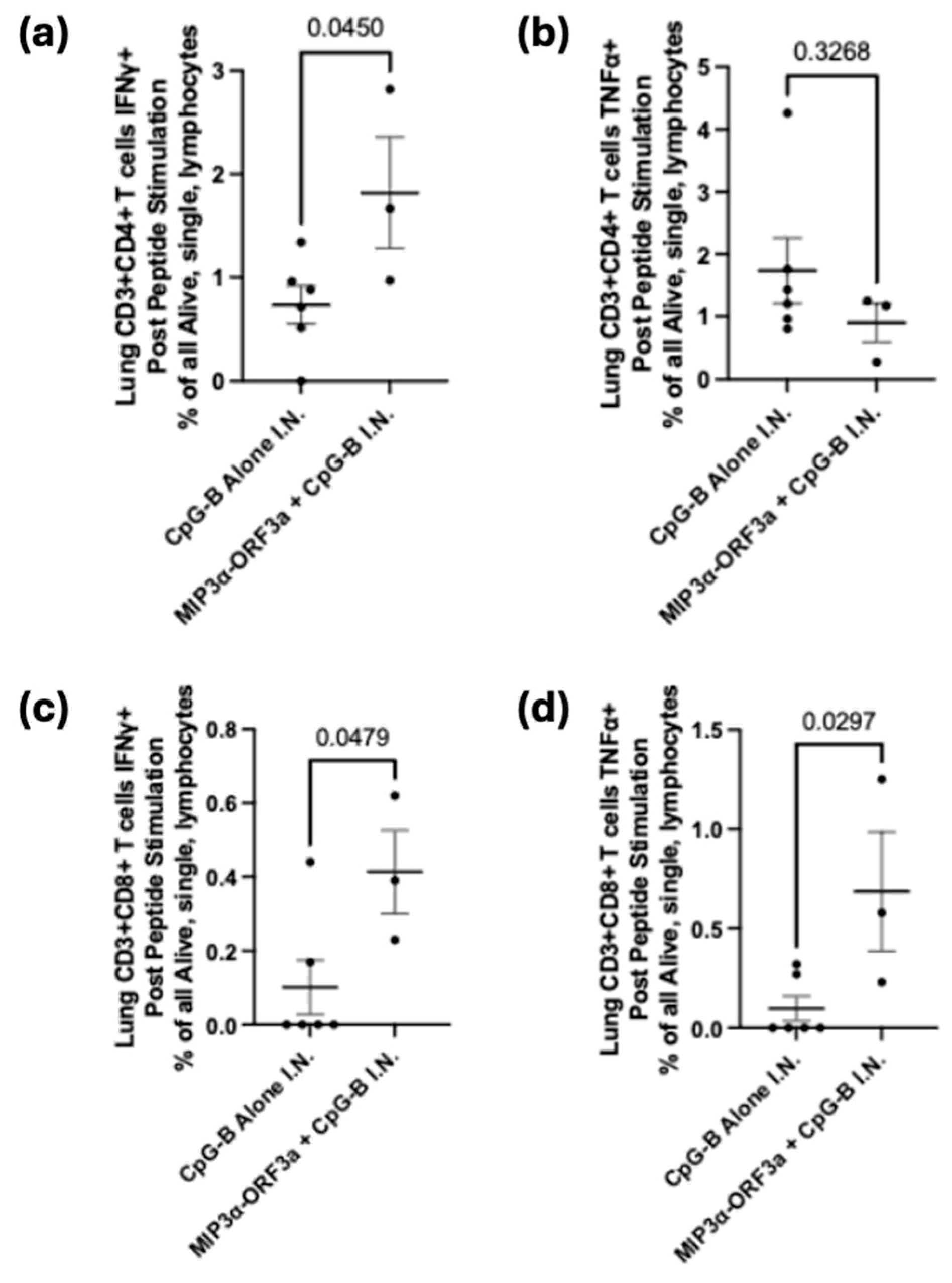
3.4. DNA Intranasal Vaccine Immunogenicity
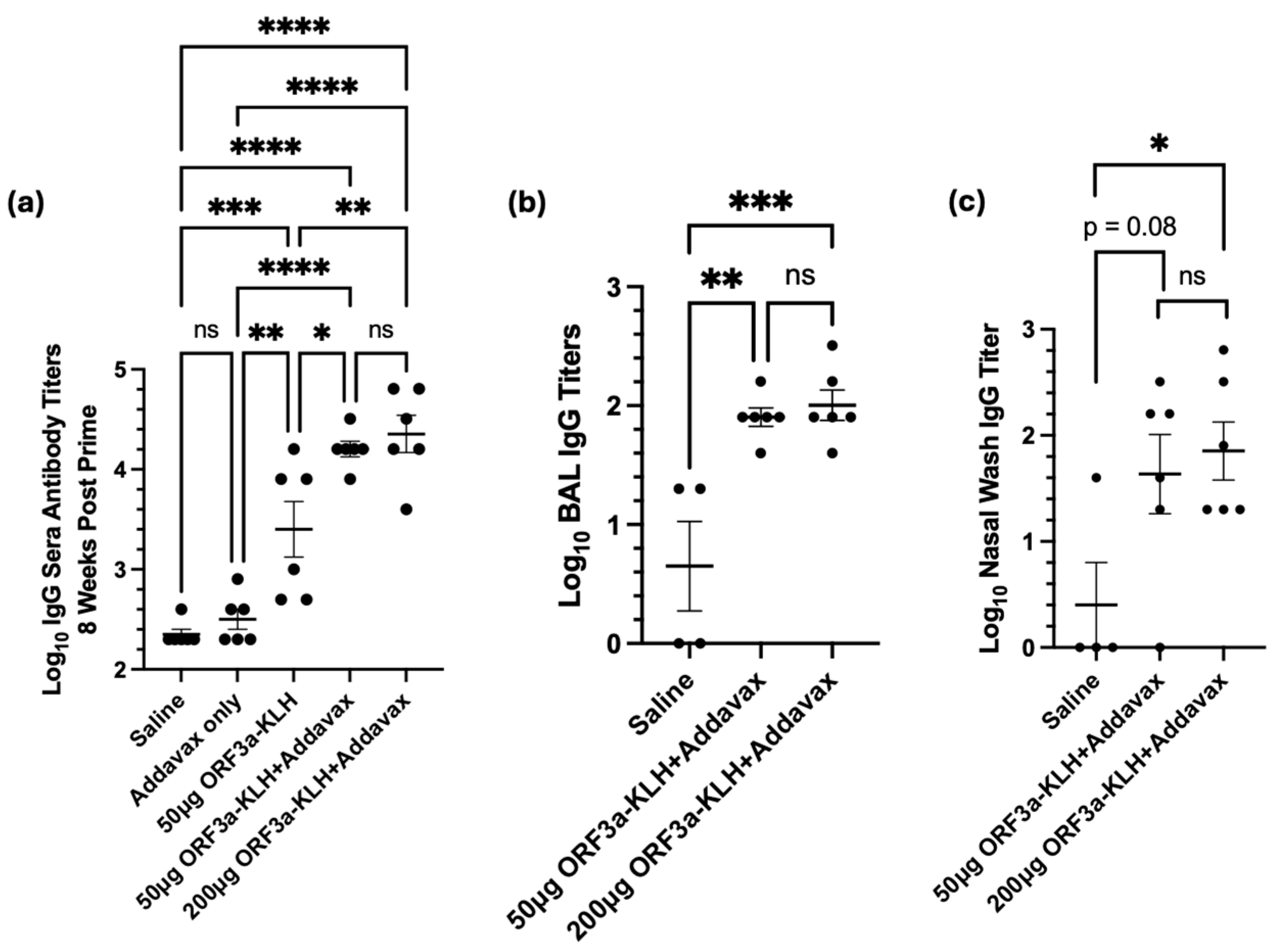
3.5. ORF3a-KLH Peptide Vaccine
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SCV2 | SARS-CoV-2 |
| ORF | Open reading frame |
| MIP3α | Macrophage-inflammatory protein 3 alpha |
| IM | Intramuscular |
| IN | Intranasal |
| KLH | Keyhole limpet hemocyanin |
| iDC | Immature dendritic cell |
| IFNγ | Interferon gamma |
| TNFα | Tumor necrosis factor alpha |
| STING | Stimulator of interferon genes |
| BAL | Bronchoalveolar lavage |
References
- COVID (SARS-CoV-2) Vaccine. Available online: http://www.ncbi.nlm.nih.gov/books/NBK567793/ (accessed on 2 August 2024).
- Emergency Use Authorization. Available online: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#vaccines (accessed on 31 July 2024).
- Zheng, C.; Shao, W.; Chen, X.; Zhang, B.; Wang, G.; Zhang, W. Real-world effectiveness of COVID-19 vaccines: a literature review and meta-analysis. International journal of infectious diseases 2022, 114, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ejikemeuwa, A.; Gerzanich, V.; Nasr, M.; Tang, Q.; Simard, J.M.; Zhao, R.Y. Understanding the Role of SARS-CoV-2 ORF3a in Viral Pathogenesis and COVID-19. Frontiers in microbiology 2022, 13, 854567. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Hom, K.; Zhang, C.; Nasr, M.; Gerzanich, V.; Zhang, Y.; Tang, Q.; Xue, F.; Simard, J.M.; Zhao, R.Y. SARS-CoV-2 ORF3a Protein as a Therapeutic Target against COVID-19 and Long-Term Post-Infection Effects. Pathogens (Basel) 2024, 13, 75. [Google Scholar] [CrossRef] [PubMed]
- Moss, P. The T cell immune response against SARS-CoV-2. Nat Immunol 2022, 23, 186–193. [Google Scholar] [CrossRef]
- Vardhana, S.; Baldo, L. ; Morice, 2.,William G.; Wherry, E.J. Understanding T cell responses to COVID-19 is essential for informing public health strategies. Science immunology 2022, 7, eabo1303. [Google Scholar] [CrossRef]
- Gong, W.; Parkkila, S.; Wu, X.; Aspatwar, A. SARS-CoV-2 variants and COVID-19 vaccines: Current challenges and future strategies. Int Rev Immunol 2023, ahead-of-print. 1–22. [Google Scholar] [CrossRef]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; Marrama, D.; de Silva, A.M.; Frazier, A.; Carlin, A.F.; Greenbaum, J.A.; Peters, B.; Krammer, F.; Smith, D.M.; Crotty, S.; Sette, A. Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell 2020, 181, 1489–1501.e15. [Google Scholar] [CrossRef] [PubMed]
- Tarke, A.; Sidney, J.; Kidd, C.K.; Dan, J.M.; Ramirez, S.I.; Yu, E.D.; Mateus, J.; da Silva Antunes, R.; Moore, E.; Rubiro, P.; Methot, N.; Phillips, E.; Mallal, S.; Frazier, A.; Rawlings, S.A.; Greenbaum, J.A.; Peters, B.; Smith, D.M.; Crotty, S.; Weiskopf, D.; Grifoni, A.; Sette, A. Comprehensive analysis of T cell immunodominance and immunoprevalence of SARS-CoV-2 epitopes in COVID-19 cases. Cell reports.Medicine 2021, 2, 100204. [Google Scholar] [CrossRef]
- Saini, S.K.; Hersby, D.S.; Tamhane, T.; Povlsen, H.R.; Amaya Hernandez, S.P.; Nielsen, M.; Gang, A.O.; Hadrup, S.R. SARS-CoV-2 genome-wide T cell epitope mapping reveals immunodominance and substantial CD8 + T cell activation in COVID-19 patients. Science immunology 2021, 6. [Google Scholar] [CrossRef]
- Aziz, N.; Detels, R.; Chang, L.C.; Butch, A.W. Macrophage Inflammatory Protein-3 Alpha (MIP-3alpha)/CCL20 in HIV-1-Infected Individuals. J AIDS Clin Res 2016, 7, 587, Epub 2016 Jun 14. [Google Scholar] [CrossRef]
- Schutyser, E.; Struyf, S.; Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev 2003, 14, 409–426. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, R.; Baatar, D.; Olkhanud, P.; Indig, F.E.; Restifo, N.; Taub, D.; Biragyn, A. Chemokine receptor targeting efficiently directs antigens to MHC class I pathways and elicits antigen-specific CD8+ T-cell responses. Blood 2006, 107, 4597–4605. [Google Scholar] [CrossRef]
- Biragyn, A.; Ruffini, P.A.; Coscia, M.; Harvey, L.K.; Neelapu, S.S.; Baskar, S.; Wang, J.; Kwak, L.W. Chemokine receptor-mediated delivery directs self-tumor antigen efficiently into the class II processing pathway in vitro and induces protective immunity in vivo. Blood 2004, 104, 1961–1969. [Google Scholar] [CrossRef] [PubMed]
- Luo, K.; Zhang, H.; Zavala, F.; Biragyn, A.; Espinosa, D.A.; Markham, R.B.; Kumar, N. Fusion of antigen to a dendritic cell targeting chemokine combined with adjuvant yields a malaria DNA vaccine with enhanced protective capabilities. PloS one 2014, 9, e90413. [Google Scholar] [CrossRef]
- Scheinecker, C.; Castellino, F.; Germain, R.N.; Altan-Bonnet, G.; Stoll, S.; Huang, A.Y. Chemokines enhance immunity by guiding naive CD8 + T cells to sites of CD4 + T cell-dendritic cell interaction. Nature 2006, 440, 890–895. [Google Scholar] [CrossRef]
- Guo, J.H.; Fan, M.W.; Sun, J.H.; Jia, R. Fusion of antigen to chemokine CCL20 or CXCL13 strategy to enhance DNA vaccine potency. Int Immunopharmacol 2009, 9, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Abe, N.; Hirano, T.; Suzuki, M. A single nasal dose of CCL20 chemokine induces dendritic cell recruitment and enhances nontypable Haemophilus influenzae-specific immune responses in the nasal mucosa. Acta Otolaryngol 2011, 131, 989–996. [Google Scholar] [CrossRef]
- Gordy, J.T.; Luo, K.; Zhang, H.; Biragyn, A.; Markham, R.B. Fusion of the dendritic cell-targeting chemokine MIP3α to melanoma antigen Gp100 in a therapeutic DNA vaccine significantly enhances immunogenicity and survival in a mouse melanoma model. J Immunother Cancer 2016, 4, 96, Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5168589/ (accessed on Jul 20, 2024). [Google Scholar] [CrossRef] [PubMed]
- Karanika, S.; Gordy, J.T.; Neupane, P.; Karantanos, T.; Ruelas Castillo, J.; Quijada, D.; Comstock, K.; Sandhu, A.K.; Kapoor, A.R.; Hui, Y.; Ayeh, S.K.; Tasneen, R.; Krug, S.; Danchik, C.; Wang, T.; Schill, C.; Markham, R.B.; Karakousis, P.C. An intranasal stringent response vaccine targeting dendritic cells as a novel adjunctive therapy against tuberculosis. Frontiers in immunology 2022, 13, 972266. [Google Scholar] [CrossRef] [PubMed]
- Luo, K.; Gordy, J.T.; Zavala, F.; Markham, R.B. A chemokine-fusion vaccine targeting immature dendritic cells elicits elevated antibody responses to malaria sporozoites in infant macaques. Scientific reports 2021, 11, 1220. [Google Scholar] [CrossRef] [PubMed]
- Gordy, J.T.; Hui, Y.; Schill, C.; Wang, T.; Chen, F.; Fessler, K.; Meza, J.; Li, Y.; Taylor, A.D.; Bates, R.E.; Karakousis, P.C.; Pekosz, A.; Sachithanandham, J.; Li, M.; Karanika, S.; Markham, R.B. A SARS-CoV-2 RBD vaccine fused to the chemokine MIP-3α elicits sustained murine antibody responses over 12 months and enhanced lung T-cell responses. Frontiers in Immunology 2024, 15, Available online: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1292059. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, P.; Yanik, S.; Venkatesh, V.; Gordy, J.; Alameh, M.; Meza, J.; Li, Y.; Glass, E.; Flores-Garcia, Y.; Tam, Y.; Chaiyawong, N.; Sarkar, D.; Weissman, D.; Markham, R. Immature dendritic cell-targeting mRNA vaccine expressing PfCSP enhances protective immune responses against Plasmodium liver infection. 2024. Available online: https://www.researchsquare.com/article/rs-4656309/v1 (accessed on Jul 30, 2024). [Google Scholar] [CrossRef]
- Tollefsen, S.; Vordermeier, M.; Olsen, I.; Storset, A.K.; Reitan, L.J.; Clifford, D.; Lowrie, D.B.; Wiker, H.G.; Huygen, K.; Hewinson, G.; Mathiesen, I.; Tjelle, T.E. DNA Injection in Combination with Electroporation: a Novel Method for Vaccination of Farmed Ruminants. Scand J Immunol 2003, 57, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Babiuk, S.; Baca-Estrada, M.; Foldvari, M.; Middleton, D.M.; Rabussay, D.; Widera, G.; Babiuk, L.A. Increased gene expression and inflammatory cell infiltration caused by electroporation are both important for improving the efficacy of DNA vaccines. J Biotechnol 2004, 110, 1–10. [Google Scholar] [CrossRef]
- Ahlen, G.; Soderholm, J.; Tjelle, T.; Kjeken, R.; Frelin, L.; Hoglund, U.; Blomberg, P.; Fons, M.; Mathiesen, I.; Sallberg, M. In Vivo Electroporation Enhances the Immunogenicity of Hepatitis C Virus Nonstructural 3/4A DNA by Increased Local DNA Uptake, Protein Expression, Inflammation, and Infiltration of CD3+ T Cells. The Journal of immunology (1950) 2007, 179, 4741–4753. [Google Scholar] [CrossRef] [PubMed]
- JINYAN, L.I.U.; KJEKEN, R.; MATHIESEN, I.; BAROUCH, D.H. Recruitment of Antigen-Presenting Cells to the Site of Inoculation and Augmentation of Human Immunodeficiency Virus Type 1 DNA Vaccine Immunogenicity by In Vivo Electroporation. J Virol 2008, 82, 5643–5649. [Google Scholar] [CrossRef]
- Zhong, X.; Guo, Z.; Yang, H.; Peng, L.; Xie, Y.; Wong, T.; Lai, S.; Guo, Z. Amino terminus of the SARS coronavirus protein 3a elicits strong, potentially protective humoral responses in infected patients. J Gen Virol 2006, 87, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Åkerström, S.; Tan, Y.; Mirazimi, A. Amino acids 15–28 in the ectodomain of SARS coronavirus 3a protein induces neutralizing antibodies. FEBS Lett 2006, 580, 3799–3803. [Google Scholar] [CrossRef]
- Lu, B.; Tao, L.; Wang, T.; Zheng, Z.; Li, B.; Chen, Z.; Huang, Y.; Hu, Q.; Wang, H. Humoral and Cellular Immune Responses Induced by 3a DNA Vaccines against Severe Acute Respiratory Syndrome (SARS) or SARS-Like Coronavirus in Mice. Clinical and Vaccine Immunology 2009, 16, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, X.; Liu, J.; Xia, H.; Zou, J.; Muruato, A.E.; Periasamy, S.; Kurhade, C.; Plante, J.A.; Bopp, N.E.; Kalveram, B.; Bukreyev, A.; Ren, P.; Wang, T.; Menachery, V.D.; Plante, K.S.; Xie, X.; Weaver, S.C.; Shi, P. A live-attenuated SARS-CoV-2 vaccine candidate with accessory protein deletions. Nature communications 2022, 13, 4337. [Google Scholar] [CrossRef] [PubMed]
- McGrath, M.E.; Xue, Y.; Dillen, C.; Oldfield, L.; Assad-Garcia, N.; Zaveri, J.; Singh, N.; Baracco, L.; Taylor, L.J.; Vashee, S.; Frieman, M.B. SARS-CoV-2 variant spike and accessory gene mutations alter pathogenesis. Proceedings of the National Academy of Sciences - PNAS 2022, 119, e2204717119. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Wu, R.; Shen, M.; Huang, J.; Li, T.; Hu, C.; Luo, F.; Song, S.; Mu, S.; Hao, Y.; Han, X.; Wang, Y.; Li, L.; Li, S.; Chen, Q.; Wang, W.; Jin, A. Extended SARS-CoV-2 RBD booster vaccination induces humoral and cellular immune tolerance in mice. iScience 2022, 25, 105479. [Google Scholar] [CrossRef]
- Pérez-Then, E.; Lucas, C.; Monteiro, V.S.; Miric, M.; Brache, V.; Cochon, L.; Vogels, C.B.F.; Malik, A.A.; De la Cruz, E.; Jorge, A.; De los Santos, M.; Leon, P.; Breban, M.I.; Billig, K.; Yildirim, I.; Pearson, C.; Downing, R.; Gagnon, E.; Muyombwe, A.; Razeq, J.; Campbell, M.; Ko, A.I.; Omer, S.B.; Grubaugh, N.D.; Vermund, S.H.; Iwasaki, A. Neutralizing antibodies against the SARS-CoV-2 Delta and Omicron variants following heterologous CoronaVac plus BNT162b2 booster vaccination. Nat Med 2022, 28, 481–485. [Google Scholar] [CrossRef]
- van der Ploeg, K.; Kirosingh, A.S.; Mori, D.A.M.; Chakraborty, S.; Hu, Z.; Sievers, B.L.; Jacobson, K.B.; Bonilla, H.; Parsonnet, J.; Andrews, J.R.; Press, K.D.; Ty, M.C.; Ruiz-Betancourt, D.; de la Parte, L.; Tan, G.S.; Blish, C.A.; Takahashi, S.; Rodriguez-Barraquer, I.; Greenhouse, B.; Singh, U.; Wang, T.T.; Jagannathan, P. TNF-α+ CD4+ T cells dominate the SARS-CoV-2 specific T cell response in COVID-19 outpatients and are associated with durable antibodies. Cell reports.Medicine 2022, 3, 100640. [Google Scholar] [CrossRef]
- Harris, J.R.; Markl, J. Keyhole limpet hemocyanin (KLH): a biomedical review. Micron 1999, 30, 597–623, Available online: https://www.sciencedirect.com/science/article/pii/S0968432899000360. [Google Scholar] [CrossRef] [PubMed]
- Wherry, E.J.; Barouch, D.H. T cell immunity to COVID-19 vaccines. Science (American Association for the Advancement of Science) 2022, 377, 821–822. [Google Scholar] [CrossRef]
- Liu, J.; Chandrashekar, A.; Sellers, D.; Barrett, J.; Jacob-Dolan, C.; Lifton, M.; McMahan, K.; Sciacca, M.; VanWyk, H.; Wu, C.; Yu, J.; Collier, A.Y.; Barouch, D.H. Vaccines elicit highly conserved cellular immunity to SARS-CoV-2 Omicron. Nature 2022, 603, 493–496. [Google Scholar] [CrossRef]
- Hadfield, J.; Megill, C.; Bell, S.M.; Huddleston, J.; Potter, B.; Callender, C.; Sagulenko, P.; Bedford, T.; Neher, R.A. Nextstrain: real-time tracking of pathogen evolution. Bioinformatics 2018, 34, 4121. [Google Scholar] [CrossRef] [PubMed]
- Sagulenko, P.; Puller, V.; Neher, R.A. TreeTime: Maximum-likelihood phylodynamic analysis. Virus Evol 2018, 4, vex042. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



