Submitted:
02 January 2025
Posted:
03 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Report
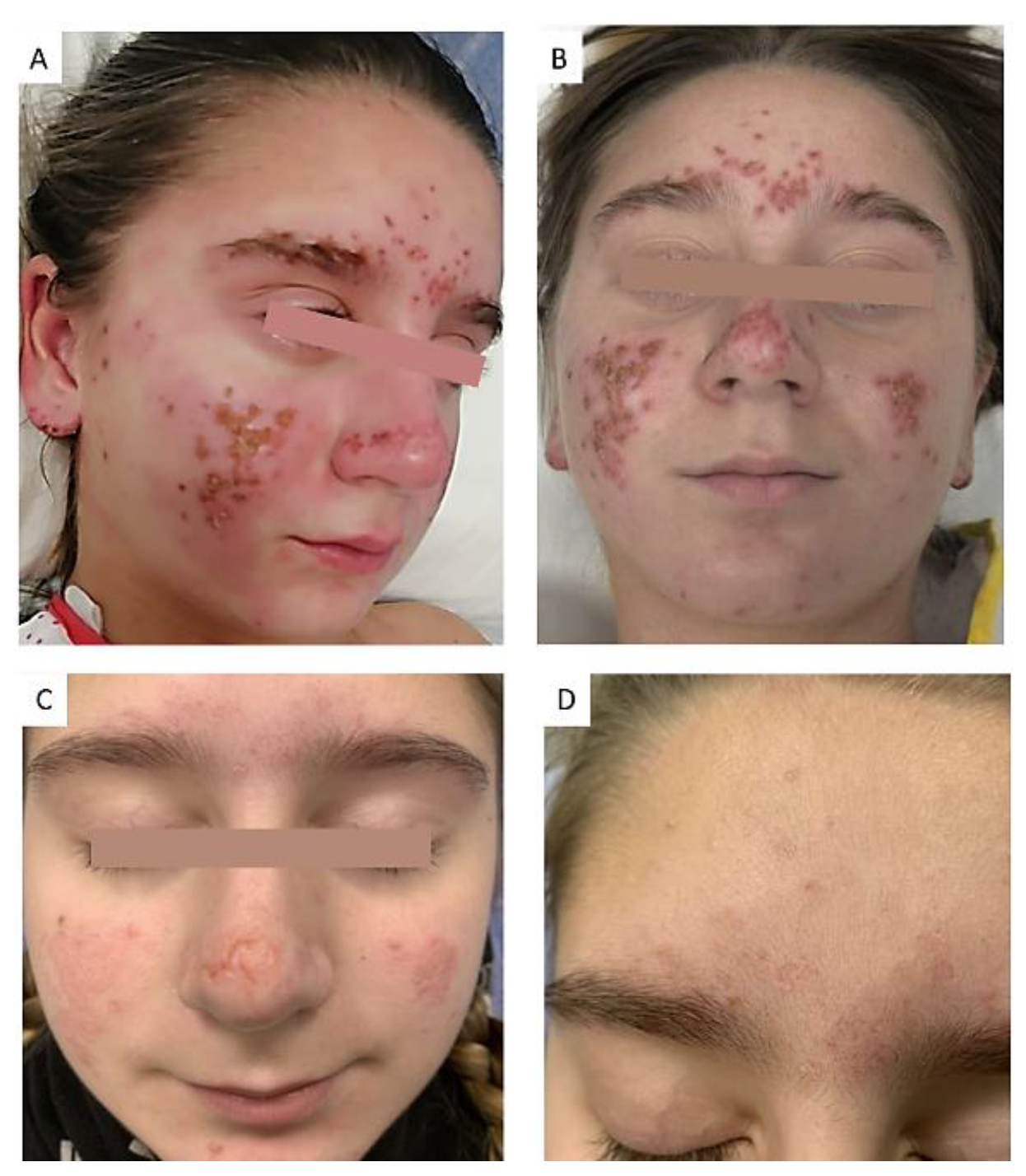
Clinical Presentation
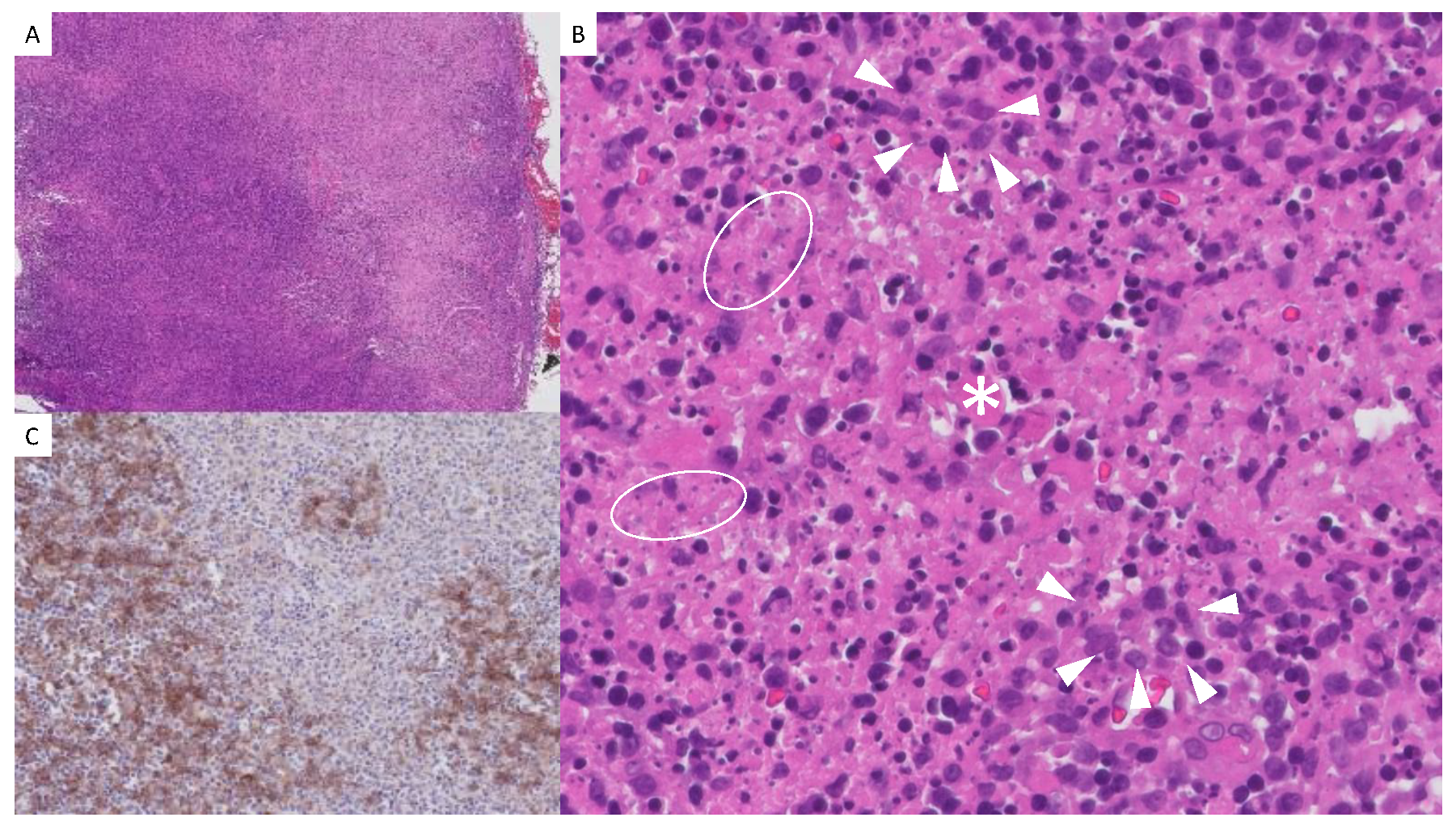
Histopathologic Findings
3. Literature Review
Cutaneous Manifestations
Histopathology
Treatment
Complications
| Study (date) |
N; sex; age. | Sample |
Histopathology | Cutaneous manifestations | Possible Trigger | Treatment | Associated complications |
|---|---|---|---|---|---|---|---|
| Heldenberg et al [21] 1996 |
1; male; 10. |
Lymph node excisional biopsy Lymph node excisional biopsy Lymph node excisional biopsy |
Cortical or paracortical necrosis. Karyorrhexis. Histiocytes (CD68+), plasmacytoid dendritic cells (CD123+), T-cell activation. Absence of neutrophils, eosinophils and granulomas Cortical or paracortical necrosis. Karyorrhexis. Histiocytes (CD68+), plasmacytoid dendritic cells (CD123+), T-cell activation. Absence of neutrophils, eosinophils and granulomas Cortical or paracortical necrosis. Karyorrhexis. Histiocytes (CD68+), plasmacytoid dendritic cells (CD123+), T-cell activation. Absence of neutrophils, eosinophils and granulomas |
No | No | No specific therapy |
No |
| Gonzálvez-Piñera et al.[8] 2000 |
1;female;7 | Generalized maculopapular rash | No | No specific therapy | No | ||
| Emir et al.[42] 2001 |
1;female;14 | No | No | No specific therapy | No |
||
| Kim et al.[9] 2003 |
1;female;13 | Generalized maculopapular rash | EBV | IVIG IV methylprednisolone |
Hemophagocytic syndrome | ||
| Chiang et al.[22] 2004 |
1;male;8 | No | No | No specific therapy | No | ||
| Chuang et al.[10] 2005 |
64; 35 males; 29 females; Mean age 16. |
Generalized maculopapular rash (4/64) | CMV (2/64) |
No specific therapy | Developed SLE (1/64) | ||
| Lin et al.[6] 2005 |
23; 15 males; 8 females; Mean age 12.8 |
NA | NA | No specific therapy | Developed SLE (1/23) | ||
| Park et al.[30] 2007 |
16; 8 males; 8 females. Mean age 10.6 | No | EBV (2) ANA (1) | Oral prednisone (6/16) | No | ||
| Zou et al.[11] 2009 |
36; 23 males, 13 females; mean age 10.1 | Generalized maculopapular rash (5/36) | EBV (3/36) HSV (2/36) ANA (3/36) |
No specific therapy | No | ||
| Gómez et al.[31] 2010. |
1; female;10 | No | No | Oral prednisone | No | ||
| Lee et al.[5] 2012 |
9; 8 males;1 female Mean age 11.3 | Generalized maculopapular rash (2/9) | No ANA (1/9) |
High-dose corticosteroids, IVIG, plasmapheresis | Developed SLE (1/9) | ||
| Tchidjou et al.[43] 2004 |
1; male; 11 | No | No | IV methylprednisolone | |||
| Gonçalves et al.[40] 2014 |
1; male;9 | No | No | IV Dexamethasone | CNS involvement | ||
| Martins et al.[18] 2014 |
1;female;12 | malar rash | ANAs | Oral prednisone Hidroxychloroquine |
Developed SLE | ||
| Ojeda et al.[32] 2015 |
1:female;9 | No | ANAs | Oral Prednisone | No | ||
| Rossetti et al.[44] 2015 |
1;male;11 | No | No | Corticosterois | Upper airway obstruction | ||
| Altinel et al.[35] 2018 |
1;male;12 | No | Salmonella | IVIG | Auto-immune thyroiditis and papilloedema. | ||
| Chowdhury et al.[39] 2019 |
1;female;14 | No | No | IV methylprednisolone | CNS involvement | ||
| Handa et al.[45] 2016 |
1;male;15 | No | No | Oral corticosteroids | No | ||
| Lelii et al.[23] 2018 |
2;male;12;female;16 | No | CMV (1/2) ANA (1/2) |
No specific Therapy (1/2) Oral prednisone and cyclosporine (1/2) |
CNS involvement in 1 patient | ||
| Singh et al.[24] 2019 |
1;male;11 | No | No | No specific Therapy |
No | ||
| Quadir et al.[25] 2020 |
2; females;12 and 16 (dizygotic twins) |
Generalized maculopapular rash (2/2) |
Mycoplasma (1/2) | No specific Therapy (1/2) Oral prednisone (1/2) |
Developed alopecia areata (1/2) |
||
| Guleria et al.[37] 2020 |
6; 4 males; 2 females; Mean age 10.8 |
No | NA | Oral prednisolone (2/6) | Concomitant SLE (1/6) | ||
| Arslan et al.[46] 2020 |
1;female;12 | No | No | Oral prednisone | Optic Neuritis | ||
| Kim et al.[26] 2020 |
1;female;7 | No | Parvovirus B19 | No specific Therapy |
Severe neutropenia | ||
| Al Mosawi et al.[12] 2020 |
11; 6 males;5females; Mean age 10 | Generalized maculopapular rash (4/11) | No | Hidroxychloroquine (7/11) Corticosteroids (1/11) No specific Therapy (3/11) |
No | ||
| Cannon et al.[27] 2020 |
1;female;11 | No | No | No specific Therapy |
No | ||
| Hua et al.[38] 2021 |
1;male;14 | No | No | IV methylprednisolone |
Appendectomy (mimicked appendicitis) | ||
| Yang et al.[14] 2021 |
13; 9 males, 4 females. | Generalized maculopapular rash in 10 patients |
ANAs in 4 patients |
IVIG + Methylprednisolone: (6/13) Methylprednisolone + Cyclosporine A: (1/13) Cyclosporine A + Dexamethasone + Etoposide: (1/13) Methylprednisolone + Cyclosporine A + Cyclophosphamide: (1/13) Ruxolitinib + Methylprednisolone: (1/13) Methylprednisolone + Dexamethasone: (1/13) Symptomatic treatment: (3/13) Intrathecal Injection + Methotrexate: (1/13) IV Immunoglobulin: (1/13) |
Hemophagocytic syndrome (13/13) CNS involvement (8/13) Developed SLE (1/13) Developed SS (2/13) |
||
| Danai et al.[13] 2021 |
1;female;10 | Generalized maculopapular rash and oral ulcers | ANAs | Prednisone Hidroxychloroquine Methotrexate |
Concomitant SLE | ||
| Öztürk et al. [47] 2021 | 1;male;5 | No | COVID-19 | No specific Therapy |
No | ||
| Chisholm et al.[28] 2022 |
14;NA;NA | NA | NA | NA | NA | ||
| Takahashi et al.[15] 2023 |
2; female,13; male;11 Brothers |
Plantar annular erythema and chilblain lupus-like rashes. | No (later met the criteria for SLE) | Corticosteroid+ Hydroxychloroquine (1/2) Hydroxychloroquine (1/2) |
Developed SLE Developed SS (2/2) |
||
| Sevrin et al.[48] 2023 |
1;male,12 |
No | COVID-19 vaccination | Corticosterois |
No | ||
| Choi Sujin et al.[17] 2023 |
114;62 males;52 females; Mean age 12 | Oral ulcer (12/114) Generalized rash (11/114) |
NA | Corticosteroids (46/114) | No | ||
| Zhou et al[49] 2024 |
1;male;4 | No | No | Oral prednisone | CASPR2 antibody-associated encephalitis | ||
| Lu et al.[50] 2024 |
1;male;13 | No | No |
Corticosterois Rituximab |
occlusive retinal vasculitis with near total central retinal artery occlusion | ||
| Bao et al.[36] 2024 |
1;male;13 | No | No | IVIG, Oral Prednisone |
Optic Neuritis |
||
| Harrison et al.[16] 2024 |
1;female;16 | Generalized rash, sparing the nasolabial folds. | No (later met the criteria for SLE) | Hydroxychloroquine, prednisone, and mycophenolate mofetil |
Developed SLE |
||
| Camacho-Badilla et al.[20] 2005 |
1;male;10 | Lymph node excisional biopsy | Granulomas characterized by central necrosis with abundant karyorrhexis, surrounded by histiocytes, lymphocytes and giant multinucleated cells, without neutrophils. | No | No | No specific therapy |
No |
| Burns et al.[29] 2020 |
1;female;16 | FNAC | Necrotizing lymphadenitis | No | EBV, CMV | No specific Therapy |
No |
| Sierra et al.[7] 1999 |
1;male;14. | 1. Lymph node excisional biopsy 2. Cutenaous biopsy |
1. Histiocytic necrotizing lymphadenitis (see previous) 2. Lymphohistiocytic infiltrates, predominantly perivascular, without any necrosis of the adjacent vascular wall |
Generalized maculopapular rash | No | IV Methylprednisolone | Multisystemic involvement Adverse reaction to drugs |
| Chen et al.[33] 2000 |
1;female;13 | 1. Lymph node excisional biopsy 2.Bone marrow aspirate |
1. Histiocytic necrotizing lymphadenitis (see previous) 2.hemophagocytic histiocytes |
No | EBV | IVIG IV methylprednisolone oral dexamethasone |
hemophagocytic syndrome |
| Jun-Fen et al.[34] 2007. |
1; female;7 | 1. Lymph node excisional biopsy 2.Bone marrow aspirate |
1. Histiocytic necrotizing lymphadenitis (see previous) 2. Specific changes of idiopathic thrombocytopenic purpura |
No | No | IVIG IV methylprednisolone Oral dexamethasone |
Idiopathic thrombocytopenic purpura and Mobitz type II atrioventricular block |
| Sykes et al.[41] 2016 |
1;female;26 | 1. Lymph node excisional biopsy 2.Bone marrow aspirate |
1. Cortical or paracortical necrosis with various histiocytes, plasmacytoid monocytes and a variable number of lymphoid cells with caryorrhectic nuclear fragments and absence of neutrophils 2. hemophagocytic histiocytes |
No | No | High-dose corticosteroids, IVIG, and plasmapheresis | Hemophagocytic syndrome |
| Das et al.[51] 2019 |
1;male;NA | 1. Lymph node excisional biopsy 2.Bone marrow aspirate |
1. Cortical or paracortical necrosis with various histiocytes, plasmacytoid monocytes and a variable number of lymphoid cells with caryorrhectic nuclear fragments and absence of neutrophils 2. Reticulo-endothelial activity |
Generalized maculopapular rash | Brucella | Rifampicin and gentamicin, doxycycline, trimethoprim-sulfamethoxazole. Oral prednisolone |
Doxycycline-Induced Intracranial Hypertension |
| Lin et al.[19] 2019 |
40;20 males;20 females;Mean age 13.9 | Lymph node excisional biopsy | 14 patients with proliferative type containing karyorrhectic nuclear fragments, eosinophilic apoptotic debris and various histiocytes 23 patients with necrotizing type showing the existence of coagulative necrosis 1 patient wit xanthomatous type showing foamy histiocytes. 2 patients with NA histopathology. |
Generalized maculopapular rash (2/40) | ANAs (1/40) EBV (1/40) |
Oral prednisone (5/40) Hidroxychloroquine (5/40) No specific Therapy in the rest. |
No |
| Abdu et al.[52] 2022 |
1;female,10 |
1. Lymph node excisionalbiopsy 2. Bone marrow biopsy |
1. Necrotizing his- tiocytic lymphadenitis, 2. Normocellular bone marrow with reactive lymphocytosis and hemophagocytosis |
No | Acute otitis media |
Corticosterois |
No |
4. Discussion
Sources of funding
Ethics statement
Data Availability
Conflicts of Interest
References
- Perry AM, Choi SM. Kikuchi-Fujimoto Disease: A Review. Arch Pathol Lab Med. 2018 Nov;142(11):1341–6. [CrossRef]
- Masab M, Surmachevska N, Farooq H. Kikuchi-Fujimoto Disease. In Treasure Island (FL); 2024.
- Cuglievan B, Miranda RN. Kikuchi-Fujimoto disease. Blood. 2017 Feb;129(7):917.
- Kim TY, Ha K-S, Kim Y, Lee J, Lee K, Lee J. Characteristics of Kikuchi-Fujimoto disease in children compared with adults. Eur J Pediatr. 2014 Jan;173(1):111–6. [CrossRef]
- Lee BC, Patel R. Kikuchi-Fujimoto disease: a 15-year analysis at a children’s hospital in the United States. Clin Pediatr (Phila). 2013 Jan;52(1):92–5.
- Lin H-C, Su C-Y, Huang S-C. Kikuchi’s disease in Asian children. Pediatrics. 2005 Jan;115(1):e92-6. [CrossRef]
- Sierra ML, Vegas E, Blanco-González JE, GonzálezA, Martínez P, Calero MA. Kikuchi’s disease with multisystemic involvement and adverse reaction to drugs. Pediatrics. 1999 Aug;104(2):e24. [CrossRef]
- Gonzálvez Piñera J, Ruiz Cano R, García Rodríguez MR, Fernández Córdoba MS, Crusat Braña S, Sango Lamban M. [Necrotizing histiocytic lymphadenitis (Kikuchi-Fujimoto disease) in a 7-year-old girl]. Cir Pediatr. 2000 Jul;13(3):126–8.
- Kim YM, Lee YJ, Nam SO, Park SE, Kim JY, Lee EY. Hemophagocytic syndrome associated with Kikuchi’s disease. J Korean Med Sci. 2003 Aug;18(4):592–4. [CrossRef]
- Chuang C-H, Yan D-C, Chiu C-H, Huang Y-C, Lin P-Y, Chen C-J, et al. Clinical and laboratory manifestations of Kikuchi’s disease in children and differences between patients with and without prolonged fever. Pediatr Infect Dis J. 2005 Jun;24(6):551–4. [CrossRef]
- Zou CC, Zhao ZY, Liang L. Childhood Kikuchi-Fujimoto disease. Indian J Pediatr. 2009 Sep;76(9):959–62. [CrossRef]
- Al Mosawi Z, Ebrahim B, Baik S, Madan W, Aljufairi E. Kikuchi-Fujimoto Disease: Eleven Pediatric Cases and Literature Review. Arch Rheumatol. 2020 Jun;35(2):303–7. [CrossRef]
- Danai MM, Kaye SP, Gong R, McKnight H. A Rare Pediatric Case of Persistent Kikuchi-Fujimoto Disease and Underlying Systemic Lupus Erythematosus. J Clin Rheumatol Pract reports Rheum Musculoskelet Dis. 2021 Apr;27(3):e111–2. [CrossRef]
- Yang Y, Lian H, Ma H, Zhao Y, Zhang Q, Zhang L, et al. Hemophagocytic Lymphohistiocytosis Associated with Histiocytic Necrotizing Lymphadenitis: A Clinical Study of 13 Children and Literature Review. J Pediatr. 2021 Feb;229:267-274.e3. [CrossRef]
- Takahashi K, Okura Y, Shimomura M, Taniguchi K, Nawate M, Takahashi Y, et al. Development of Sjögren’s syndrome following Kikuchi-Fujimoto disease - A sibling case. Pediatr Int. 2023;65(1):e15524.
- Harrison J, Sukumaran S, Vijayan V. Systemic Lupus Erythematosus Lymphadenopathy Presenting as Kikuchi-Fujimoto Disease in an Adolescent. Vol. 15, Cureus. United States; 2023. p. e35304. [CrossRef]
- Choi S, Choi HS, Ryu YJ, Kim JY, Paik JH, Ahn S, et al. Characterization of Kikuchi-Fujimoto Disease in Children and Risk Factors Associated with Its Course. J Pediatr. 2023 Sep;260:113515. [CrossRef]
- Martins SS, Buscatti IM, Freire PS, Cavalcante EG, Sallum AM, Campos LMA, et al. [Kikuchi-Fujimoto disease prior to childhood-systemic lupus erythematosus diagnosis]. Rev Bras Reumatol. 2014;54(5):400–3. [CrossRef]
- Lin Y-C, Huang H-H, Nong B-R, Liu P-Y, Chen Y-Y, Huang Y-F, et al. Pediatric Kikuchi-Fujimoto disease: A clinicopathologic study and the therapeutic effects of hydroxychloroquine. J Microbiol Immunol Infect. 2019 Jun;52(3):395–401. [CrossRef]
- Camacho-Badilla K, Cañas-Coto A, Soriano-Fallas A, Carranza-Portocarrero A, Avila-Agüero ML. [Kikuchi-Fujimoto disease in pediatric population: a case report and a review of literature]. Gac Med Mex. 2005;141(1):53–6.
- Heldenberg D, Amar M, Ben-Arie Y, Iuchtman M. Axillary involvement in pediatric Kikuchi’s disease. Eur J Pediatr Surg Off J Austrian Assoc Pediatr Surg. [et al] = Zeitschrift fur Kinderchirurgie. 1996 Feb;6(1):32–4.
- Chiang Y-C, Chen RMY, Chao P-Z, Yang T-H, Lee F-P. Pediatric Kikuchi-Fujimoto disease masquerading as a submandibular gland tumor. Int J Pediatr Otorhinolaryngol. 2004 Jul;68(7):971–4. [CrossRef]
- Lelii M, Senatore L, Amodeo I, Pinzani R, Torretta S, Fiori S, et al. Kikuchi-Fujimoto disease in children: two case reports and a review of the literature. Ital J Pediatr. 2018 Jul;44(1):83. [CrossRef]
- Singh JM, Shermetaro CB. Kikuchi-Fujimoto Disease in Michigan: A Rare Case Report and Review of the Literature. Clin Med insights Ear, nose throat. 2019;12:1179550619828680. [CrossRef]
- Quadir A, Peacock K, Hsu P, Singh-Grewal D, Alexander S. A familial case of Kikuchi-Fujimoto disease in dizygotic twins. Pediatr Rheumatol Online J. 2020 Aug;18(1):62. [CrossRef]
- Kim L, Tatarina-Numlan O, Yin YD, John M, Sundaram R. A Case of Kikuchi-Fujimoto Disease in a 7-Year-Old African American Patient: A Case Report and Review of Literature. Am J Case Rep. 2020 Aug;21:e922784. [CrossRef]
- Cannon L, Wang E, Becker M. Diagnosis of Kikuchi-Fujimoto disease in an 11-year-old girl with fever and sickle cell disease. BMJ Case Rep. 2020 Aug;13(8). [CrossRef]
- Chisholm KM, Bohling SD, Tsuchiya KD, Paulson VA. A Malignant Mimicker: Features of Kikuchi-Fujimoto Disease in the Pediatric Population. Pediatr Dev Pathol Off J Soc Pediatr Pathol Paediatr Pathol Soc. 2022;25(5):538–47. [CrossRef]
- Burns J, Pathania S, Griesser R, Norelli J, Gilgeours K, Tugetman J, et al. Evaluation of a unilateral neck mass in a 16-year-old female: Kikuchi-Fujimoto disease with superimposed bacterial lymphadenitis. Vol. 8, SAGE open medical case reports. England; 2020. p. 2050313X20962640. [CrossRef]
- Park HS, Sung MJ, Park SE, Lim YT. Kikuchi-Fujimoto disease of 16 children in a single center of Korea. Pediatr allergy Immunol Off Publ Eur Soc Pediatr Allergy Immunol. 2007 Mar;18(2):174–8. [CrossRef]
- Gómez C, Eraso RM, Aguirre CA, Pérez MDP. [Kikuchi-Fujimoto disease: pediatric case presentation]. Biomedica. 2010;30(4):465–72.
- Ojeda Lewis E, Vásquez Paz H. Kikuchi-Fujimoto disease: pediatric case report. Medwave. 2015 Sep;15(8):e6270.
- Chen JS, Chang KC, Cheng CN, Tsai WH, Su IJ. Childhood hemophagocytic syndrome associated with Kikuchi’s disease. Vol. 85, Haematologica. Italy; 2000. p. 998–1000.
- Jun-Fen F, Chun-Lin W, Li L, Dayan C, Guan-Ping D, Fang H. Kikuchi-Fujimoto disease manifesting as recurrent thrombocytopenia and Mobitz type II atrioventricular block in a 7-year-old girl: a case report and analysis of 138 Chinese childhood Kikuchi-Fujimoto cases with 10 years of follow-up in 97 patients. Acta Paediatr. 2007 Dec;96(12):1844–7. [CrossRef]
- Altinel Açoğlu E, Sari E, Şahin G, Oğuz MM, Akçaboy M, Zorlu P, et al. Kikuchi-Fujimoto disease triggered by Salmonella enteritidis in a child with concurrent auto-immune thyroiditis and papilloedema. Paediatr Int Child Health. 2018 Nov;38(4):298–301.
- Bao S, Huang H, Jin Y, Ding F, Yang Z, Xu X, et al. Optic Neuritis in a Pediatric Patient with Kikuchi-Fujimoto Disease: A Case Report and Review of the Literature. Vol. 17, Journal of inflammation research. New Zealand; 2024. p. 2889–95. [CrossRef]
- Guleria S, Gupta A, Pilania RK, Pandiarajan V, Rawat A, Saikia UN, et al. Kikuchi-Fujimoto Disease: An Under Recognized Cause of Fever with Lymphadenopathy. Vol. 87, Indian journal of pediatrics. India; 2020. p. 85. [CrossRef]
- Hua C-Z, Chen Y-K, Chen S-Z, Gu W-Z, Shu Q. Histiocytic Necrotizing Lymphadenitis Mimicking Acute Appendicitis in a Child: A Case Report. Vol. 9, Frontiers in pediatrics. Switzerland; 2021. p. 682738. [CrossRef]
- Chowdhury SR. Extra-nodal Kikuchi Disease and Kikuchi Encephalitis. Vol. 56, Indian pediatrics. India; 2019. p. 881–2.
- Gonçalves LF, Debelenko L V, Bhambhani KJ, Scheid A, Altinok D. Histiocytic necrotizing lymphadenitis (Kikuchi-Fujimoto disease) with CNS involvement in a child. Pediatr Radiol. 2014 Feb;44(2):234–8. [CrossRef]
- Sykes JA, Badizadegan K, Gordon P, Sokol D, Escoto M, Ten I, et al. Simultaneous Acquired Self-limited Hemophagocytic Lymphohistiocytosis and Kikuchi Necrotizing Lymphadenitis in a 16-Year-Old Teenage Girl: A Case Report and Review of the Literature. Pediatr Emerg Care. 2016 Nov;32(11):792–8.
- Emir S, Gögüş S, Güler E, Büyükpamukçu M. Kikuchi-Fujimoto disease (histiocytic necrotizing lymphadenitis) confused with lymphoma in a child. Med Pediatr Oncol. 2001 Dec;37(6):546–8. [CrossRef]
- Tchidjou HK, Macchiaiolo M, Ariganello P, Carducci FC, De Vito R, De Benedetti F, et al. Kikuchi-Fujimoto disease in patient with systemic phacomatosis pigmentovascularis. Blood Coagul fibrinolysis an Int J Haemost Thromb. 2014 Oct;25(7):783–5. [CrossRef]
- Rossetti E, Appierto L, Bianchi R, Picardo S. Kikuchi-Fujimoto disease and life-threatening upper airway obstruction. Vol. 25, Paediatric anaesthesia. France; 2015. p. 864–5.
- Handa A, Nozaki T, Hosoya Y, Sakoda A, Suzuki K. Multiple migratory recurrence of Kikuchi-Fujimoto disease. Pediatr Int. 2016 Dec;58(12):1360–2. [CrossRef]
- Arslan A, Kraus CL, Izbudak I. Optic Neuritis as an Isolated Presentation of Kikuchi–Fujimoto Disease in a Pediatric Patient. Vol. 37, Balkan medical journal. Turkey; 2020. p. 172–3. [CrossRef]
- Öztürk N, Kılıç İ, Göçün PU, Kaya Z. Kikuchi-Fujimoto disease in a child who had a high suspicion of COVID-19 infection. J Hematop. 2022 Sep;15(3):197–8. [CrossRef]
- Sevrin F, Beaujot J, Jaillard A, Fayard C, Forestier A, Defachelles A-S. Kikuchi-Fujimoto disease following SARS-CoV-2 vaccination in a patient with previous history of rhabdomyosarcoma. Pediatric blood & cancer. United States; 2023. p. e30345.
- Zhou Y, Shu S. Histiocytic necrotizing lymphadenitis with autoimmune encephalitis in a child: a case report. Transl Pediatr. 2024 Oct;13(10):1892–8. [CrossRef]
- Lu ES, Hoyek S, Yuan M, El Khatib BA, Gonzalez E, Rothermel H, et al. Occlusive Retinal Vasculitis in a Pediatric Patient With Kikuchi-Fujimoto Disease and Sickle Cell Trait. Ophthalmic Surg Lasers Imaging Retina. 2024 Apr;55(4):235–9. [CrossRef]
- Das A, Bhave SJ, Pal B, Arun I, Goel G, Bhattacharya S, et al. Brucellosis Complicated by Kikuchi-Fujimoto Disease and Doxycycline-Induced Intracranial Hypertension. Vol. 86, Indian journal of pediatrics. India; 2019. p. 1063–4. [CrossRef]
- Abdu A, Pajkrt D, Bijker EM. Kikuchi Disease in Children: Case Report and Review of the Literature. Pediatr Infect Dis J. 2022 Mar;41(3):199–204.
- Seo J-H, Lee JS, Lee E-J, Lee J-H, Kim JP, Woo SH, et al. Comparison of clinical features and EBV expression in histiocytic necrotizing lymphadenitis of children and adults. Int J Pediatr Otorhinolaryngol. 2014 May;78(5):748–52. [CrossRef]
- Yen H-R, Lin P-Y, Chuang W-Y, Chang M-L, Chiu C-H. Skin manifestations of Kikuchi-Fujimoto disease: case report and review. Eur J Pediatr. 2004 Apr;163(4–5):210–3. [CrossRef]
- Yasukawa K, Matsumura T, Sato-Matsumura KC, Takahashi T, Fujioka Y, Kobayashi H, et al. Kikuchi’s disease and the skin: case report and review of the literature. Br J Dermatol. 2001 Apr;144(4):885–9. [CrossRef]
- Kim KJ, Jee MS, Chang SE, Choi JH, Sung KJ, Moon KC, et al. Kikuchi-Fujimoto disease with papulopustular skin manifestations. Clin Exp Dermatol. 2003 Mar;28(2):142–4. [CrossRef]
- Spies J, Foucar K, Thompson CT, LeBoit PE. The histopathology of cutaneous lesions of Kikuchi’s disease (necrotizing lymphadenitis): a report of five cases. Am J Surg Pathol. 1999 Sep;23(9):1040–7.
- Paradela S, Lorenzo J, Martínez-Gómez W, Yebra-Pimentel T, Valbuena L, Fonseca E. Interface dermatitis in skin lesions of Kikuchi-Fujimoto’s disease: a histopathological marker of evolution into systemic lupus erythematosus? Lupus. 2008 Dec;17(12):1127–35. [CrossRef]
- Frankel AM, Frenkel S, Aminlari A, Chan T. Kikuchi Disease: A Case Report. J Emerg Med. 2020 Dec;59(6):927–30. [CrossRef]
- Yen A, Fearneyhough P, Raimer SS, Hudnall SD. EBV-associated Kikuchi’s histiocytic necrotizing lymphadenitis with cutaneous manifestations. J Am Acad Dermatol. 1997 Feb;36(2 Pt 2):342–6. [CrossRef]
- Kuo TT. Kikuchi’s disease (histiocytic necrotizing lymphadenitis). A clinicopathologic study of 79 cases with an analysis of histologic subtypes, immunohistology, and DNA ploidy. Am J Surg Pathol. 1995 Jul;19(7):798–809. [CrossRef]
- Yu R-B, Chen Y-J, Chang C-H, Chen Y-L, Chen J-W. Kikuchi-Fujimoto Disease Associated With Mycoplasma Pneumoniae Infection. Ear Nose Throat J. 2024 Apr;103(4):NP223–5. [CrossRef]
- Müller CSL, Vogt T, Becker SL. Kikuchi-Fujimoto Disease Triggered by Systemic Lupus Erythematosus and Mycoplasma pneumoniae Infection-A Report of a Case and a Review of the Literature. Am J Dermatopathol. 2021 Mar;43(3):202–8. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





