Submitted:
30 December 2024
Posted:
03 January 2025
You are already at the latest version
Abstract
Proteases are hallmarks of various physiological processes and cellular signaling net-works that maintain homeostasis and cellular fate. This regulation is particularly medi-ated by carboxypeptidases, cysteine proteases, and metalloproteases, which function by cleaving peptide bonds, thereby modulating the activity of proteins and influencing their functional outcomes. Proteases play a regulatory role in various cellular processes, in-cluding cell growth, differentiation, cell death, immunity, and inflammation. Protease signaling influences the terminal outcomes of several forms of cell death, including apop-tosis, necroptosis, pyroptosis, and autophagy. Similarly, during inflammatory stimula-tion, proteases orchestrate a cascade of pathways that optimize the immune response to pathogens. Proteases are significant contributors to the development of various human diseases, including cancer, neurological disorders, and inflammatory disorders. The acti-vation of specific proteases determines the outcomes of different forms of cell death high-lighted in this review. Proteases are therefore critical targets for exploring future research on modulating their activity in various diseases and therapeutic targets. This review high-lights the important functions of protease family members in managing different aspects related to cellular balance and disease conditions.
Keywords:
1. Introduction
2. Proteases in Regulating Homeostasis & Cell Death
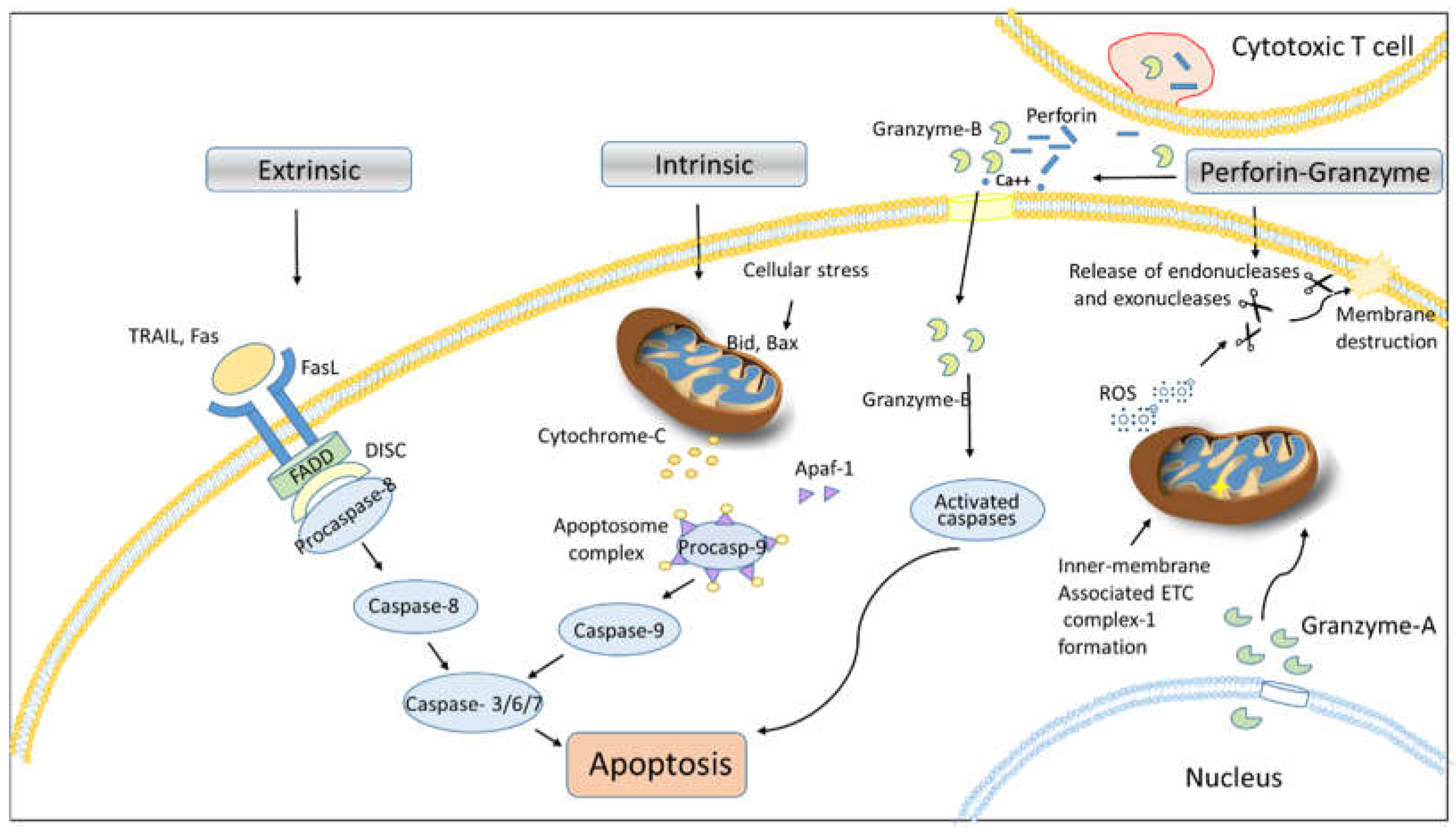
2.1. Proteases in Apoptosis
2.1.1. Calpains in Cell Death
2.1.2. Cathepsin in Cell Death
2.1.3. Perforin-Granzyme Pathway in Cell Death
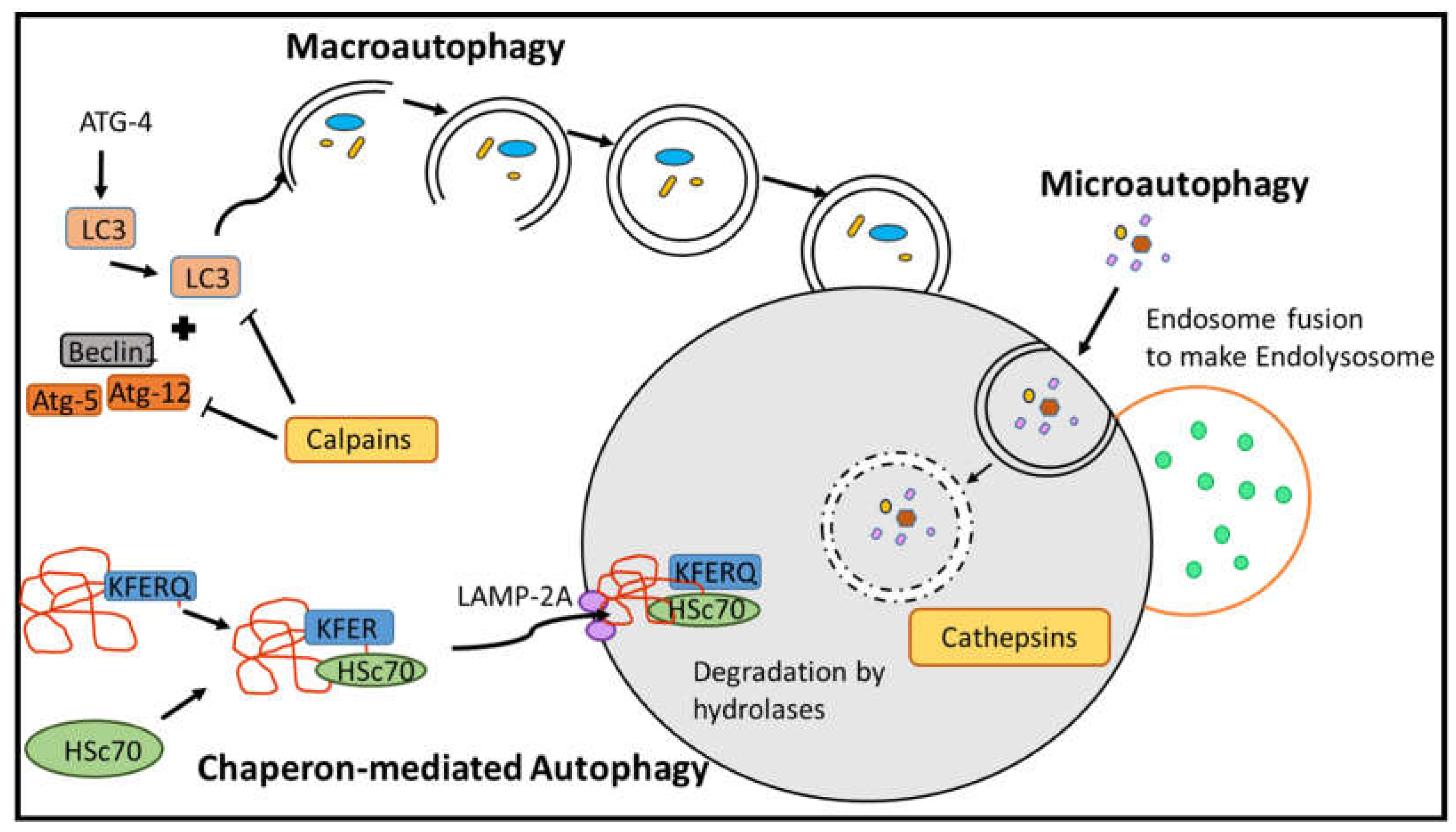
2.2. Proteases in Autophagy Pathways
2.2.1. Macroautophagy
2.2.2. Chaperon-Mediated Autophagy (CMA)
2.2.3. Microautophagy
2.3. Proteases in Necrosis/Necroptosis
3. Proteases in Pyroptosis and Inflammation
3.1. Eryptosis
3.2. Entosis
3.3. Oncosis
4. Proteases in ER- Stress Mediated Cell Death
5. Proteases and Cancer
5.1. Proteases in Tumor Progression
5.1.1. Serine Proteases
5.1.2. Cysteine Proteases
5.1.3. Aspartyl Proteases
5.1.4. Metalloproteases (MMPs) in Cancer
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Lopez-Otin, C.; Bond, J.S. Proteases: multifunctional enzymes in life and disease. J Biol Chem 2008, 283, 30433-30437. [CrossRef]
- Bialas, A.; Kafarski, P. Proteases as Anti-Cancer Targets - Molecular and Biological Basis for Development of Inhibitor-Like Drugs Against Cancer. Anti-Cancer Agent Me 2009, 9, 728-762. [CrossRef]
- Noël, A.; Jost, M.; Maquoi, E. Matrix metalloproteinases at cancer tumor-host interface. Semin Cell Dev Biol 2008, 19, 52-60. [CrossRef]
- Pérez-Silva, J.G.; Español, Y.; Velasco, G.; Quesada, V. The Degradome database: expanding roles of mammalian proteases in life and disease. Nucleic Acids Res 2016, 44, D351-D355. [CrossRef]
- Gurumallesh, P.; Alagu, K.; Ramakrishnan, B.; Muthusamy, S. A systematic reconsideration on proteases. Int J Biol Macromol 2019, 128, 254-267. [CrossRef]
- Neurath, H.; Walsh, K.A. Role of proteolytic enzymes in biological regulation (a review). Proc Natl Acad Sci U S A 1976, 73, 3825-3832. [CrossRef]
- Vicencio, J.M.; Galluzzi, L.; Tajeddine, N.; Ortiz, C.; Criollo, A.; Tasdemir, E.; Morselli, E.; Ben Younes, A.; Maiuri, M.C.; Lavandero, S.; et al. Senescence, apoptosis or autophagy? When a damaged cell must decide its path--a mini-review. Gerontology 2008, 54, 92-99. [CrossRef]
- Ranjan, K.; Pathak, C. Cellular Dynamics of Fas-Associated Death Domain in the Regulation of Cancer and Inflammation. Int J Mol Sci 2024, 25. [CrossRef]
- Ranjan, K.; Surolia, A.; Pathak, C. Apoptotic potential of Fas-associated death domain on regulation of cell death regulatory protein cFLIP and death receptor mediated apoptosis in HEK 293T cells. J Cell Commun Signal 2012, 6, 155-168. [CrossRef]
- Sukharev, S.A.; Pleshakova, O.V.; Sadovnikov, V.B. Role of proteases in activation of apoptosis. Cell Death Differ 1997, 4, 457-462, doi:DOI 10.1038/sj.cdd.4400263.
- Kidd, V.J.; Lathi, J.M.; Teitz, T. Proteolytic regulation of apoptosis. Semin Cell Dev Biol 2000, 11, 191-201, doi:DOI 10.1006/scdb.2000.0165.
- Yanumula, A.; Cusick, J.K. Biochemistry, Extrinsic Pathway of Apoptosis. In StatPearls; Treasure Island (FL) ineligible companies. Disclosure: John Cusick declares no relevant financial relationships with ineligible companies., 2024.
- Ranjan, K.; Waghela, B.N.; Vaidya, F.U.; Pathak, C. Cell-Penetrable Peptide-Conjugated FADD Induces Apoptosis and Regulates Inflammatory Signaling in Cancer Cells. Int J Mol Sci 2020, 21. [CrossRef]
- Kesavardhana, S.; Malireddi, R.K.S.; Kanneganti, T.D. Caspases in Cell Death, Inflammation, and Pyroptosis. Annu Rev Immunol 2020, 38, 567-595. [CrossRef]
- Ranjan, K.; Pathak, C. FADD regulates NF-kappaB activation and promotes ubiquitination of cFLIPL to induce apoptosis. Sci Rep 2016, 6, 22787. [CrossRef]
- Ranjan, K.; Pathak, C. Expression of FADD and cFLIP(L) balances mitochondrial integrity and redox signaling to substantiate apoptotic cell death. Mol Cell Biochem 2016, 422, 135-150. [CrossRef]
- Chico, J.F.; Saggau, C.; Adam, D. Proteolytic control of regulated necrosis. Bba-Mol Cell Res 2017, 1864, 2147-2161. [CrossRef]
- McIlwain, D.R.; Berger, T.; Mak, T.W. Caspase Functions in Cell Death and Disease (vol 5, a008656, 2013). Csh Perspect Biol 2015, 7.
- Ranjan, K.; Sharma, A.; Surolia, A.; Pathak, C. Regulation of HA14-1 mediated oxidative stress, toxic response, and autophagy by curcumin to enhance apoptotic activity in human embryonic kidney cells. Biofactors 2014, 40, 157-169. [CrossRef]
- Smith, M.A.; Schnellmann, R.G. Calpains, mitochondria, and apoptosis. Cardiovasc Res 2012, 96, 32-37. [CrossRef]
- Hu, H.X.; Li, X.P.; Li, Y.; Wang, L.F.; Mehta, S.; Feng, Q.P.; Chen, R.Z.; Peng, T.Q. Calpain-1 induces apoptosis in pulmonary microvascular endothelial cells under septic conditions. Microvasc Res 2009, 78, 33-39. [CrossRef]
- Wingrave, J.M.; Schaecher, K.E.; Sribnick, E.A.; Wilford, G.G.; Ray, S.K.; Hazen-Martin, D.J.; Hogan, E.L.; Banik, N.L. Early induction of secondary injury factors causing activation of calpain and mitochondria-mediated neuronal apoptosis following spinal cord injury in rats. J Neurosci Res 2003, 73, 95-104. [CrossRef]
- Berchem, G.; Glondu, M.; Gleizes, M.; Brouillet, J.P.; Vignon, F.; Garcia, M.; Liaudet-Coopman, E. Cathepsin-D affects multiple tumor progression steps: proliferation, angiogenesis and apoptosis. Oncogene 2002, 21, 5951-5955. [CrossRef]
- Liaudet-Coopman, E.; Beaujouin, M.; Derocq, D.; Garcia, M.; Glondu-Lassis, M.; Laurent-Matha, V.; Prébois, C.; Rochefort, H.; Vignon, F. Cathepsin D:: newly discovered functions of a long-standing aspartic protease in cancer and apoptosis. Cancer Lett 2006, 237, 167-179. [CrossRef]
- Vetvicka, V.; Fusek, M. Procathepsin D as a tumor marker, anti-cancer drug or screening agent. Anticancer Agents Med Chem 2012, 12, 172-175. [CrossRef]
- Chwieralski, C.E.; Welte, T.; Bühling, F. Cathepsin-regulated apoptosis. Apoptosis 2006, 11, 143-149. [CrossRef]
- Droga-Mazovec, G.; Bojic, L.; Petelin, A.; Ivanova, S.; Romih, R.; Repnik, U.; Salvesen, G.S.; Stoka, V.; Turk, V.; Turk, B. Cysteine cathepsins trigger caspase-dependent cell death through cleavage of Bid and antiapoptotic Bcl-2 homologues. J Biol Chem 2008, 283, 19140-19150. [CrossRef]
- Trapani, J.A.; Smyth, M.J. Functional significance of the perforin/granzyme cell death pathway. Nat Rev Immunol 2002, 2, 735-747. [CrossRef]
- Lieberman, J. Granzyme A activates another way to die. Immunol Rev 2010, 235, 93-104.
- Rousalova, I.; Krepela, E. Granzyme B-induced apoptosis in cancer cells and its regulation (Review). Int J Oncol 2010, 37, 1361-1378. [CrossRef]
- Kaminskyy, V.; Zhivotovsky, B. Proteases in autophagy. Biochim Biophys Acta 2012, 1824, 44-50. [CrossRef]
- Ranjan, K.; Pathak, C. Expression of cFLIPL Determines the Basal Interaction of Bcl-2 With Beclin-1 and Regulates p53 Dependent Ubiquitination of Beclin-1 During Autophagic Stress. J Cell Biochem 2016, 117, 1757-1768. [CrossRef]
- Shibutani, S.T.; Saitoh, T.; Nowag, H.; Munz, C.; Yoshimori, T. Autophagy and autophagy-related proteins in the immune system. Nat Immunol 2015, 16, 1014-1024. [CrossRef]
- Kumar, M.; Ranjan, K.; Singh, V.; Pathak, C.; Pappachan, A.; Singh, D.D. Hydrophilic Acylated Surface Protein A (HASPA) of Leishmania donovani: Expression, Purification and Biophysico-Chemical Characterization. Protein J 2017, 36, 343-351. [CrossRef]
- Duque, T.L.A.; Serrao, T.; Goncalves, A.; Pinto, E.F.; Oliveira-Neto, M.P.; Pirmez, C.; Pereira, L.O.R.; Menna-Barreto, R.F.S. Leishmania (V.) braziliensis infection promotes macrophage autophagy by a LC3B-dependent and BECLIN1-independent mechanism. Acta Trop 2021, 218, 105890. [CrossRef]
- Kang, J.W.; Yan, J.; Ranjan, K.; Zhang, X.; Turner, J.R.; Abraham, C. Myeloid Cell Expression of LACC1 Is Required for Bacterial Clearance and Control of Intestinal Inflammation. Gastroenterology 2020, 159, 1051-1067. [CrossRef]
- Bandyopadhyay, U.; Kaushik, S.; Varticovski, L.; Cuervo, A.M. The chaperone-mediated autophagy receptor organizes in dynamic protein complexes at the lysosomal membrane. Mol Cell Biol 2008, 28, 5747-5763. [CrossRef]
- Kaushik, S.; Bandyopadhyay, U.; Sridhar, S.; Kiffin, R.; Martinez-Vicente, M.; Kon, M.; Orenstein, S.J.; Wong, E.; Cuervo, A.M. Chaperone-mediated autophagy at a glance. J Cell Sci 2011, 124, 495-499. [CrossRef]
- Mortimore, G.E.; Lardeux, B.R.; Adams, C.E. Regulation of Microautophagy and Basal Protein-Turnover in Rat-Liver - Effects of Short-Term Starvation. J Biol Chem 1988, 263, 2506-2512.
- Pathak, C.; Vaidya, F.U.; Waghela, B.N.; Jaiswara, P.K.; Gupta, V.K.; Kumar, A.; Rajendran, B.K.; Ranjan, K. Insights of Endocytosis Signaling in Health and Disease. Int J Mol Sci 2023, 24. [CrossRef]
- Numrich, J.; Péli-Gulli, M.P.; Arlt, H.; Sardu, A.; Griffith, J.; Levine, T.; Engelbrecht-Vandré, S.; Reggiori, F.; De Virgilio, C.; Ungermann, C. The I-BAR protein Ivy1 is an effector of the Rab7 GTPase Ypt7 involved in vacuole membrane homeostasis. J Cell Sci 2015, 128, 2278-2292. [CrossRef]
- Tekirdag, K.; Cuervo, A.M. Chaperone-mediated autophagy and endosomal microautophagy: Jointed by a chaperone. J Biol Chem 2018, 293, 5414-5424. [CrossRef]
- Sendler, M.; Mayerle, J.; Lerch, M.M. Necrosis, Apoptosis, Necroptosis, Pyroptosis: It Matters How Acinar Cells Die During Pancreatitis. Cell Mol Gastroenterol Hepatol 2016, 2, 407-408. [CrossRef]
- Cai, Z.Y.; Zhang, A.L.; Choksi, S.; Li, W.H.; Li, T.; Zhang, X.M.; Liu, Z.G. Activation of cell-surface proteases promotes necroptosis, inflammation and cell migration. Cell Res 2016, 26, 886-900. [CrossRef]
- Vasudevan, S.O.; Behl, B.; Rathinam, V.A. Pyroptosis-induced inflammation and tissue damage. Semin Immunol 2023, 69, 101781. [CrossRef]
- Patel, P.; Karch, J. Regulation of cell death in the cardiovascular system. Int Rev Cel Mol Bio 2020, 353, 153-209. [CrossRef]
- Pathak, C.; Vaidya, F.U.; Waghela, B.N.; Chhipa, A.S.; Tiwari, B.S.; Ranjan, K. Advanced Glycation End Products Mediated Oxidative Stress and Regulated Cell Death Signaling in Cancer. In Handbook of Oxidative Stress in Cancer: Mechanistic Aspects, Chakraborti, S., Ray, B.K., Roychowdhury, S., Eds.; Springer Singapore: Singapore, 2020; pp. 1-16.
- Waghela, B.N.; Vaidya, F.U.; Ranjan, K.; Chhipa, A.S.; Tiwari, B.S.; Pathak, C. AGE-RAGE synergy influences programmed cell death signaling to promote cancer. Mol Cell Biochem 2021, 476, 585-598. [CrossRef]
- Chandrasekar, S.A.; Palaniyandi, T.; Parthasarathy, U.; Surendran, H.; Viswanathan, S.; Wahab, M.R.A.; Baskar, G.; Natarajan, S.; Ranjan, K. Implications of Toll-like receptors (TLRs) and their signaling mechanisms in human cancers. Pathol Res Pract 2023, 248, 154673. [CrossRef]
- Jia, C.; Chen, H.W.; Zhang, J.; Zhou, K.L.; Zhuge, Y.Z.; Niu, C.; Qiu, J.X.; Rong, X.; Shi, Z.W.; Xiao, J.; et al. Role of pyroptosis in cardiovascular diseases. Int Immunopharmacol 2019, 67, 311-318. [CrossRef]
- Cheng, K.T.; Xiong, S.Q.; Ye, Z.M.; Hong, Z.G.; Di, A.K.; Tsang, K.M.; Gao, X.P.; An, S.J.; Mittal, M.; Vogel, S.M.; et al. Caspase-11-mediated endothelial pyroptosis underlies endotoxemia-induced lung injury. J Clin Invest 2017, 127, 4124-4135. [CrossRef]
- Oltra, S.S.; Colomo, S.; Sin, L.; Perez-Lopez, M.; Lazaro, S.; Molina-Crespo, A.; Choi, K.H.; Ros-Pardo, D.; Martinez, L.; Morales, S.; et al. Distinct GSDMB protein isoforms and protease cleavage processes differentially control pyroptotic cell death and mitochondrial damage in cancer cells. Cell Death Differ 2023, 30, 1366-1381. [CrossRef]
- Zou, J.; Zheng, Y.; Huang, Y.; Tang, D.; Kang, R.; Chen, R. The Versatile Gasdermin Family: Their Function and Roles in Diseases. Front Immunol 2021, 12, 751533. [CrossRef]
- Wang, K.; Sun, Q.; Zhong, X.; Zeng, M.; Zeng, H.; Shi, X.; Li, Z.; Wang, Y.; Zhao, Q.; Shao, F.; et al. Structural Mechanism for GSDMD Targeting by Autoprocessed Caspases in Pyroptosis. Cell 2020, 180, 941-955 e920. [CrossRef]
- Clancy, D.M.; Sullivan, G.P.; Moran, H.B.T.; Henry, C.M.; Reeves, E.P.; McElvaney, N.G.; Lavelle, E.C.; Martin, S.J. Extracellular Neutrophil Proteases Are Efficient Regulators of IL-1, IL-33, and IL-36 Cytokine Activity but Poor Effectors of Microbial Killing. Cell Rep 2018, 22, 2937-2950. [CrossRef]
- Shi, J.; Gao, W.; Shao, F. Pyroptosis: Gasdermin-Mediated Programmed Necrotic Cell Death. Trends Biochem Sci 2017, 42, 245-254. [CrossRef]
- Wei, X.; Xie, F.; Zhou, X.X.; Wu, Y.C.; Yan, H.Y.; Liu, T.; Huang, J.; Wang, F.W.; Zhou, F.F.; Zhang, L. Role of pyroptosis in inflammation and cancer. Cell Mol Immunol 2022, 19, 971-992. [CrossRef]
- Qadri, S.M.; Bissinger, R.; Solh, Z.; Oldenborg, P.A. Eryptosis in health and disease: A paradigm shift towards understanding the (patho)physiological implications of programmed cell death of erythrocytes. Blood Rev 2017, 31, 349-361. [CrossRef]
- Föller, M.; Huber, S.M.; Lang, F. Erythrocyte programmed cell death. Iubmb Life 2008, 60, 661-668. [CrossRef]
- Lang, F.; Gulbins, E.; Lerche, H.; Huber, S.M.; Kempe, D.S.; Föller, M. Eryptosis, a Window to Systemic Disease. Cell Physiol Biochem 2008, 22, 373-380. [CrossRef]
- Dreischer, P.; Duszenko, M.; Stein, J.; Wieder, T. Eryptosis: Programmed Death of Nucleus-Free, Iron-Filled Blood Cells. Cells-Basel 2022, 11. [CrossRef]
- Matarrese, P.; Straface, E.; Pietraforte, D.; Gambardella, L.; Vona, R.; Maccaglia, A.; Minetti, M.; Malorni, W. Peroxynitrite induces senescence and apoptosis of red blood cells through the activation of aspartyl and cysteinyl proteases. Faseb J 2005, 19, 416-+. [CrossRef]
- Velásquez, F.C.; Maté, S.; Bakás, L.; Herlax, V. Induction of eryptosis by low concentrations of alpha-hemolysin. Bba-Biomembranes 2015, 1848, 2779-2788. [CrossRef]
- Berg, C.P.; Engels, I.H.; Rothbart, A.; Lauber, K.; Renz, A.; Schlosser, S.F.; Schulze-Osthoff, K.; Wesselborg, S. Human mature red blood cells express caspase-3 and caspase-8, but are devoid of mitochondrial regulators of apoptosis. Cell Death Differ 2001, 8, 1197-1206, doi:DOI 10.1038/sj.cdd.4400905.
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An Iron-Dependent Form of Nonapoptotic Cell Death. Cell 2012, 149, 1060-1072. [CrossRef]
- Nagakannan, P.; Islam, M.I.; Conrad, M.; Eftekharpour, E. Cathepsin B is an executioner of ferroptosis. Bba-Mol Cell Res 2021, 1868. [CrossRef]
- Chen, X.; Yu, C.H.; Kang, R.; Kroemer, G.; Tang, D.L. Cellular degradation systems in ferroptosis. Cell Death Differ 2021, 28, 1135-1148. [CrossRef]
- Overholtzer, M.; Mailleux, A.A.; Mouneimne, G.; Normand, G.; Schnitt, S.J.; King, R.W.; Cibas, E.S.; Brugge, J.S. A nonapoptotic cell death process, entosis, that occurs by cell-in-cell invasion. Cell 2007, 131, 966-979. [CrossRef]
- Krishna, S.; Overholtzer, M. Mechanisms and consequences of entosis. Cell Mol Life Sci 2016, 73, 2379-2386. [CrossRef]
- Durgan, J.; Florey, O. Cancer cell cannibalism: Multiple triggers emerge for entosis. Bba-Mol Cell Res 2018, 1865, 831-841. [CrossRef]
- Guo, J.; Yang, W.T.; Mai, F.Y.; Liang, J.R.; Luo, J.; Zhou, M.C.; Yu, D.D.; Wang, Y.L.; Li, C.G. Unravelling oncosis: morphological and molecular insights into a unique cell death pathway. Front Immunol 2024, 15, 1450998. [CrossRef]
- Liu, X.L.; Van Vleet, T.; Schnellmann, R.G. The role of calpain in oncotic cell death. Annu Rev Pharmacol 2004, 44, 349-370, doi:DOI 10.1146/annurev.pharmtox.44.101802.121804.
- Ranjan, K.; Hedl, M.; Abraham, C. The E3 ubiquitin ligase RNF186 and RNF186 risk variants regulate innate receptor-induced outcomes. Proc Natl Acad Sci U S A 2021, 118. [CrossRef]
- Ranjan, K.; Hedl, M.; Sinha, S.; Zhang, X.; Abraham, C. Ubiquitination of ATF6 by disease-associated RNF186 promotes the innate receptor-induced unfolded protein response. J Clin Invest 2021, 131. [CrossRef]
- Kim, I.; Xu, W.J.; Reed, J.C. Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities. Nat Rev Drug Discov 2008, 7, 1013-1030. [CrossRef]
- Huang, C.; Hedl, M.; Ranjan, K.; Abraham, C. LACC1 Required for NOD2-Induced, ER Stress-Mediated Innate Immune Outcomes in Human Macrophages and LACC1 Risk Variants Modulate These Outcomes. Cell Rep 2019, 29, 4525-4539 e4524. [CrossRef]
- Ranjan, K. Intestinal Immune Homeostasis and Inflammatory Bowel Disease: A Perspective on Intracellular Response Mechanisms. Gastrointest Disord 2020, 2, 246-266. [CrossRef]
- Ferri, K.F.; Kroemer, G. Organelle-specific initiation of cell death pathways. Nat Cell Biol 2001, 3, E255-E263, doi:DOI 10.1038/ncb1101-e255.
- Momoi, T. Caspases involved in ER stress-mediated cell death. J Chem Neuroanat 2004, 28, 101-105. [CrossRef]
- Nakagawa, T.; Yuan, J.Y. Cross-talk between two cysteine protease families: Activation of caspase-12 by calpain in apoptosis. J Cell Biol 2000, 150, 887-894, doi:DOI 10.1083/jcb.150.4.887.
- Saleh, M.; Vaillancourt, J.P.; Graham, R.K.; Huyck, M.; Srinivasula, S.M.; Alnemri, E.S.; Steinberg, M.H.; Nolan, V.; Baldwin, C.T.; Hotchkiss, R.S.; et al. Differential modulation of endotoxin responsiveness by human caspase-12 polymorphisms. Nature 2004, 429, 75-79, doi:DOI 10.1038/nature02451.
- Koblinski, J.E.; Ahram, M.; Sloane, B.F. Unraveling the role of proteases in cancer. Clin Chim Acta 2000, 291, 113-135. [CrossRef]
- Kos, J. Proteases: Role and Function in Cancer. Int J Mol Sci 2022, 23. [CrossRef]
- Tagirasa, R.; Yoo, E. Role of Serine Proteases at the Tumor-Stroma Interface. Front Immunol 2022, 13. [CrossRef]
- Martin, C.E.; List, K. Cell surface-anchored serine proteases in cancer progression and metastasis. Cancer Metast Rev 2019, 38, 357-387. [CrossRef]
- Goretzki, L.; Schmitt, M.; Mann, K.; Calvete, J.; Chucholowski, N.; Kramer, M.; Gunzler, W.A.; Janicke, F.; Graeff, H. Effective Activation of the Proenzyme Form of the Urokinase-Type Plasminogen-Activator (Pro-Upa) by the Cysteine Protease Cathepsin-L. Febs Lett 1992, 297, 112-118, doi:Doi 10.1016/0014-5793(92)80339-I.
- Hua, T.; Robitaille, M.; Roberts-Thomson, S.J.; Monteith, G.R. The intersection between cysteine proteases, Ca2+signalling and cancer cell apoptosis*. Bba-Mol Cell Res 2023, 1870. [CrossRef]
- Zhou, M.; Liu, X.J.; Li, Z.H.; Huang, Q.; Li, F.; Li, C.Y. Caspase-3 regulates the migration, invasion and metastasis of colon cancer cells. Int J Cancer 2018, 143, 921-930. [CrossRef]
- Huang, K.H.; Fang, W.L.; Li, A.F.Y.; Liang, P.H.; Wu, C.W.; Shy, Y.M.; Yang, M.H. Caspase-3, a key apoptotic protein, as a prognostic marker in gastric cancer after curative surgery. Int J Surg 2018, 52, 266-271. [CrossRef]
- Amptoulach, S.; Lazaris, A.C.; Giannopoulou, I.; Kavantzas, N.; Patsouris, E.; Tsavaris, N. Expression of caspase-3 predicts prognosis in advanced noncardia gastric cancer. Med Oncol 2015, 32. [CrossRef]
- Nakajima, Y.I.; Kuranaga, E. Caspase-dependent non-apoptotic processes in development. Cell Death Differ 2017, 24, 1422-1430. [CrossRef]
- Kimura, Y.; Koga, H.; Araki, N.; Mugita, N.; Fujita, N.; Takeshima, H.; Nishi, T.; Yamashima, T.; Saido, T.C.; Yamasaki, T.; et al. The involvement of calpain-dependent proteolysis of the tumor suppressor NF2 (merlin) in schwannomas and meningiomas. Nat Med 1998, 4, 915-922. [CrossRef]
- Lakshmikuttyamma, A.; Selvakumar, P.; Kanthan, R.; Kanthan, S.C.; Sharma, R.K. Overexpression of -calpain in human colorectal adenocarcinomas. Cancer Epidem Biomar 2004, 13, 1604-1609.
- Xu, F.K.; Gu, J.; Lu, C.L.; Mao, W.; Wang, L.; Zhu, Q.L.; Liu, Z.H.; Chu, Y.W.; Liu, R.H.; Ge, D. Calpain-2 Enhances Non-Small Cell Lung Cancer Progression and Chemoresistance to Paclitaxel via EGFR-pAKT Pathway. Int J Biol Sci 2019, 15, 127-137. [CrossRef]
- Storr, S.J.; Carragher, N.O.; Frame, M.C.; Parr, T.; Martin, S.G. The calpain system and cancer. Nat Rev Cancer 2011, 11, 364-374. [CrossRef]
- Vasiljeva, O.; Turk, B. Dual contrasting roles of cysteine cathepsins in cancer progression: Apoptosis versus tumour invasion. Biochimie 2008, 90, 380-386. [CrossRef]
- Soond, S.M.; Kozhevnikova, M.V.; Townsend, P.A.; Zamyatnin, A.A. Cysteine Cathepsin Protease Inhibition: An update on its Diagnostic, Prognostic and Therapeutic Potential in Cancer. Pharmaceuticals-Base 2019, 12. [CrossRef]
- Vizovisek, M.; Fonovic, M.; Turk, B. Cysteine cathepsins in extracellular matrix remodeling: Extracellular matrix degradation and beyond. Matrix Biol 2019, 75-76, 141-159. [CrossRef]
- Foghsgaard, L.; Wissing, D.; Mauch, D.; Lademann, U.; Bastholm, L.; Boes, M.; Elling, F.; Leist, M.; Jäättelä, M. Cathepsin B acts as a dominant execution protease in tumor cell apoptosis induced by tumor necrosis factor. J Cell Biol 2001, 153, 999-1009, doi:DOI 10.1083/jcb.153.5.999.
- Nagaraj, N.S.; Vigneswaran, N.; Zacharias, W. Cathepsin B mediates TRAIL-induced apoptosis in oral cancer cells. J Cancer Res Clin 2006, 132, 171-183. [CrossRef]
- Rodríguez, J.; Vázquez, J.; Corte, M.D.; Lamelas, M.; Bongera, M.; Corte, M.G.; Alvarez, A.; Allende, M.; Gonzalez, L.; Sánchez, M.; et al. Clinical significance of cathepsin D concentration in tumor cytosol of primary breast cancer. Int J Biol Marker 2005, 20, 103-111, doi:Doi 10.1177/172460080502000204.
- Zhu, S.; Li, Z. Proteases and Cancer Development. In Role of Proteases in Cellular Dysfunction, Dhalla, N.S., Chakraborti, S., Eds.; Springer New York: New York, NY, 2014; pp. 129-145.
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2002, 2, 161-174. [CrossRef]
- Parks, W.C.; Wilson, C.L.; López-Boado, Y.S. Matrix metalloproteinases as modulators of inflammation and innate immunity. Nat Rev Immunol 2004, 4, 617-629. [CrossRef]
- Page-McCaw, A.; Ewald, A.J.; Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Bio 2007, 8, 221-233. [CrossRef]
- Cal, S.; López-Otín, C. ADAMTS proteases and cancer. Matrix Biol 2015, 44-46, 77-85. [CrossRef]
- Rocks, N.; Paulissen, G.; El Hour, M.; Quesada, F.; Crahay, C.; Gueders, M.; Foidart, J.M.; Noel, A.; Cataldo, D. Emerging roles of ADAM and ADAMTS metalloproteinases in cancer. Biochimie 2008, 90, 369-379. [CrossRef]
- Decock, J.; Hendrickx, W.; Thirkettle, S.; Gutiérrez-Fernández, A.; Robinson, S.D.; Edwards, D.R. Pleiotropic functions of the tumor- and metastasis-suppressing matrix metalloproteinase-8 in mammary cancer in MMTV-PyMT transgenic mice. Breast Cancer Res 2015, 17. [CrossRef]
- Deryugina, E.I.; Quigley, J.P. Pleiotropic roles of matrix metalloproteinases in tumor angiogenesis: Contrasting, overlapping and compensatory functions. Bba-Mol Cell Res 2010, 1803, 103-120. [CrossRef]
- Shay, G.; Lynch, C.C.; Fingleton, B. Moving targets: Emerging roles for MMPs in cancer progression and metastasis. Matrix Biol 2015, 44-46, 200-206. [CrossRef]
- Gialeli, C.; Theocharis, A.D.; Karamanos, N.K. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. Febs J 2011, 278, 16-27. [CrossRef]
- Willbold, R.; Wirth, K.; Martini, T.; Sultmann, H.; Bolenz, C.; Wittig, R. Excess hepsin proteolytic activity limits oncogenic signaling and induces ER stress and autophagy in prostate cancer cells. Cell Death Dis 2019, 10, 601. [CrossRef]
- Suzuki, Y.; Imai, Y.; Nakayama, H.; Takahashi, K.; Takio, K.; Takahashi, R. A serine protease, HtrA2, is released from the mitochondria and interacts with XIAP, inducing cell death. Mol Cell 2001, 8, 613-621. [CrossRef]
- Kummari, R.; Dutta, S.; Chaganti, L.K.; Bose, K. Discerning the mechanism of action of HtrA4: a serine protease implicated in the cell death pathway. Biochem J 2019, 476, 1445-1463. [CrossRef]
- Kajiwara, Y.; Schiff, T.; Voloudakis, G.; Gama Sosa, M.A.; Elder, G.; Bozdagi, O.; Buxbaum, J.D. A critical role for human caspase-4 in endotoxin sensitivity. J Immunol 2014, 193, 335-343. [CrossRef]
- Shalini, S.; Dorstyn, L.; Dawar, S.; Kumar, S. Old, new and emerging functions of caspases. Cell Death Differ 2015, 22, 526-539. [CrossRef]
- Page-McCaw, A.; Ewald, A.J.; Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Biol 2007, 8, 221-233. [CrossRef]
| Protease | Role in cell death | References |
|---|---|---|
| Serine Protease | ||
| Granzyme B | Catalyse the cleavage and activation of various downstream caspases, leading to apoptotic changes in target cell. | [30] |
| Granzyme H | Found to induce the release of pro-apoptotic proteins from the cell mitochondria. It can also catalyse DFF45/ICAD directly by proteolytic process. | [84] |
| Granzyme A | GrA does not triggers caspase-cascade, but appears to take part in cell death by targeting nuclear envelope protein and chromatin structural proteins | [84] |
| Granzyme K | Accelerates rapid ROS generation and collapse inner membrane potential of mitochondria. It targets mitochondria by activating Bid into t-Bid which disrupts the outer membrane of mitochondria leading to cytochrome C release. | [28] |
| Granzyme M | Asscoaited with both caspase-dependent and caspase-independent forms of apoptotic cell death in humans. It executes its cytotoxic function by cleaving Fas-associated protein with death domain (FADD). | [28] |
| Hepsin | Using cyclin B, cyclin A, and a p53-dependent mechanism, hepsin causes cell cycle arrest at the G2/M phase. Hepsin also shows inhibitory effect on tumor cell growth. |
[83,112] |
| HtrA2 | Increases apoptosis through a caspase-mediated mechanism that depends on protease activity, leading to the degradation of XIAP, a crucial anti-apoptotic molecule.Promotes cell death by two distinct pathways. One involves direct interaction with IAPs and inhibition of those molecules, which is accompanied by a marked increase in caspase activity. The other is via a serine protease activity-dependent, caspase-independent, and IAP inhibition-independent pathway. |
[102] [113] |
| HtrA4 | HtrA4 cleaves XIAP and induce apoptosis. [XIAP (anti-apoptotic protein) is a caspase-3 blocker.] | [114] |
| Cysteine Protease | ||
| Cathepsins (B, C, F, H, K, L, O, S, V, X, W) |
Known to cleave Bid protein involved in apoptosis, also cysteine cathepsins acts on anti-apoptotic family members of Bcl-2. Known to degrade E-cadherin a cell adhesion. | [27,98] |
| Caspase-1 | An inflammatory caspase, that trigger pyroptosis Cleave pro-IL-1β during initiation of pyroptosis |
[14,21] |
| Caspase-2 | Involved in initiation of GTP-depletion induced apoptosis, in pancreatic β cells, also has a role in cancer cell death. Plays as an effector enzyme in activation of caspase 3 in DNA damage induced apoptosis. |
[18] |
| Caspase 3 | Plays a mojor role in both extrinsic and intrinsic pathway, it can cleave more than 500 cellular substrates. Also helps in apoptotic chromatin condensation and cell dismantling. | [89,90] |
| Caspase 4 | Belong to family of Inflammatory caspases, play a critical role in IL-1β and IL18 secretion, also associated with cell death. | [115] |
| Caspase 5 | A class of cytosolic cysteine protease, may play a role in innate immune response and inflammation. | [116] |
| Caspase 6 | An executioner caspase that mediates innate immunity and inflammasome activation. It is known to play role in activation of pyroptosis, apoptosis and necroptosis. |
|
| Caspase 7 | In intrinsic cell death pathway caspase 7 may be responsible for ROS production, accumulation and cell detachment. | |
| Caspase 8 | Play a role in execution of extrinsic apoptosis, inflammasome formation and inhibition of necroptosis | |
| Caspase 9 | Increase ROS production and mitochondrial uncoupling in intrinsic pathway. | |
| Caspase-12 | Also an inflammatory caspases that seems to mediate ER-stress-induced apoptosis. Exact function of Cas-12 is poorly understood | |
| Aspartic Proteases | ||
| Cathepsin D | CD has shown to mediate apoptosis in P53-dependent tumor suppression. Overexpression of cath-D has been known to activate growth factors and promote angiogenesis | [23,24] |
| Matrix metalloproteinases (MMPs) | ||
| MMP-1 (Collagenase 1) | Can kill cells of CNS when activated through mechanism of S-nitrosylation. | [117] |
| MMP-2 (Gelatinase A) | Role in triggering neuronal apoptosis. | |
| MMP-3 (Stromelysin-1) | Involved in neuronal apoptosis. Increased expression of MMP-3 may have anti-apoptotic effect. | |
| MMP-7 (Matrilysin) | Release membranebound Fas Ligand (FasL) and induces apoptosis of neighbouring cells. | |
| MMP-9 (Gelatinase B) | Involved in degradation of ECM proteins (Laminins, fibronectin, vitronectin) to induce apoptosis in developing cerebellum and retinal ganglion cells. | |
| MMP-11 (Stromelysin 3) | Increases apoptosis during tissue remodelling and development or may inhibit apoptosis of cancer cells in animal models, promote tumor generation and metastasis. | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




