Submitted:
27 December 2024
Posted:
30 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
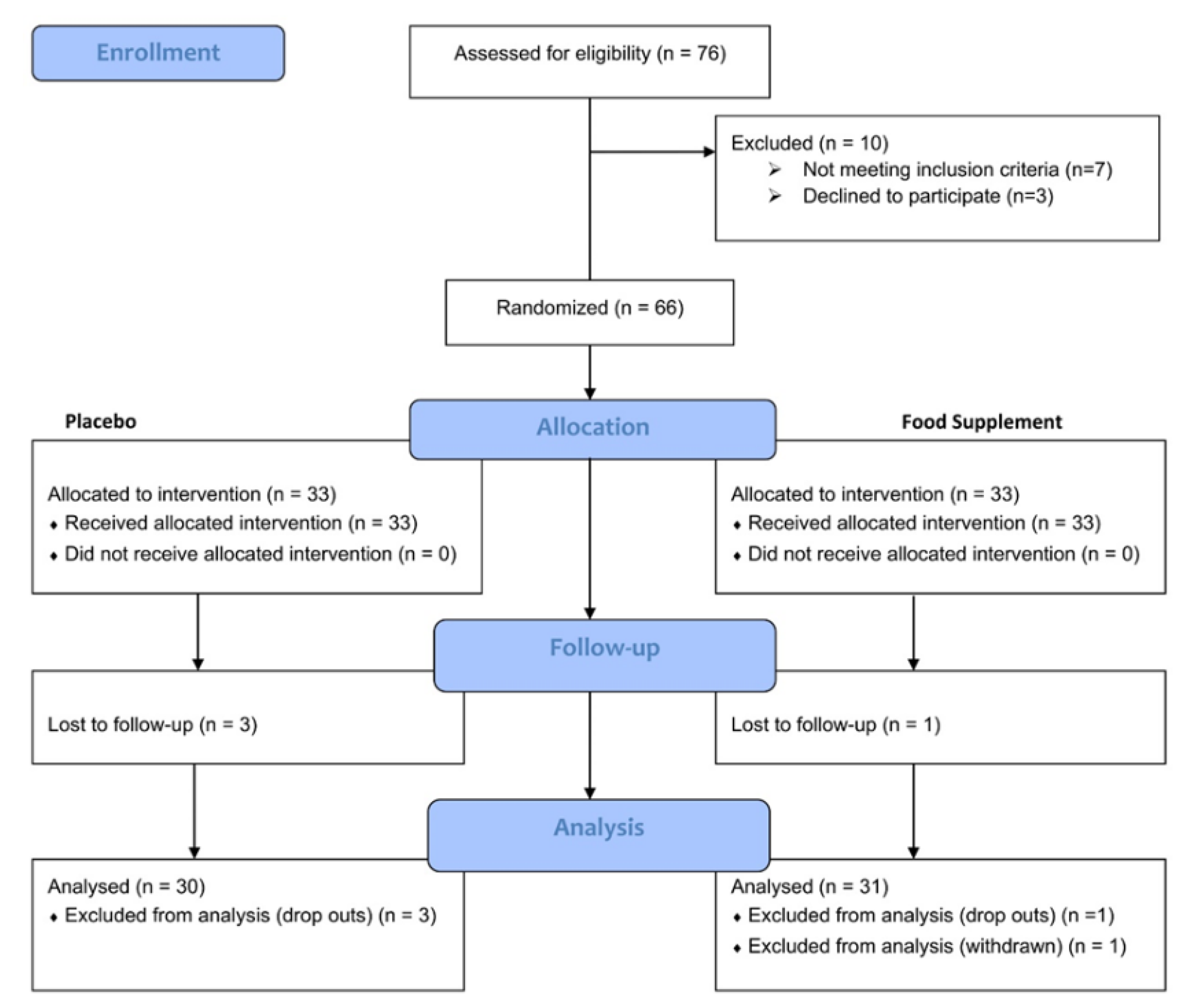
2.1. Study Design and Participants
2.2. Investigational Products, Blinding and Randomization
2.3. Study Flow
2.4. Outcome Measures
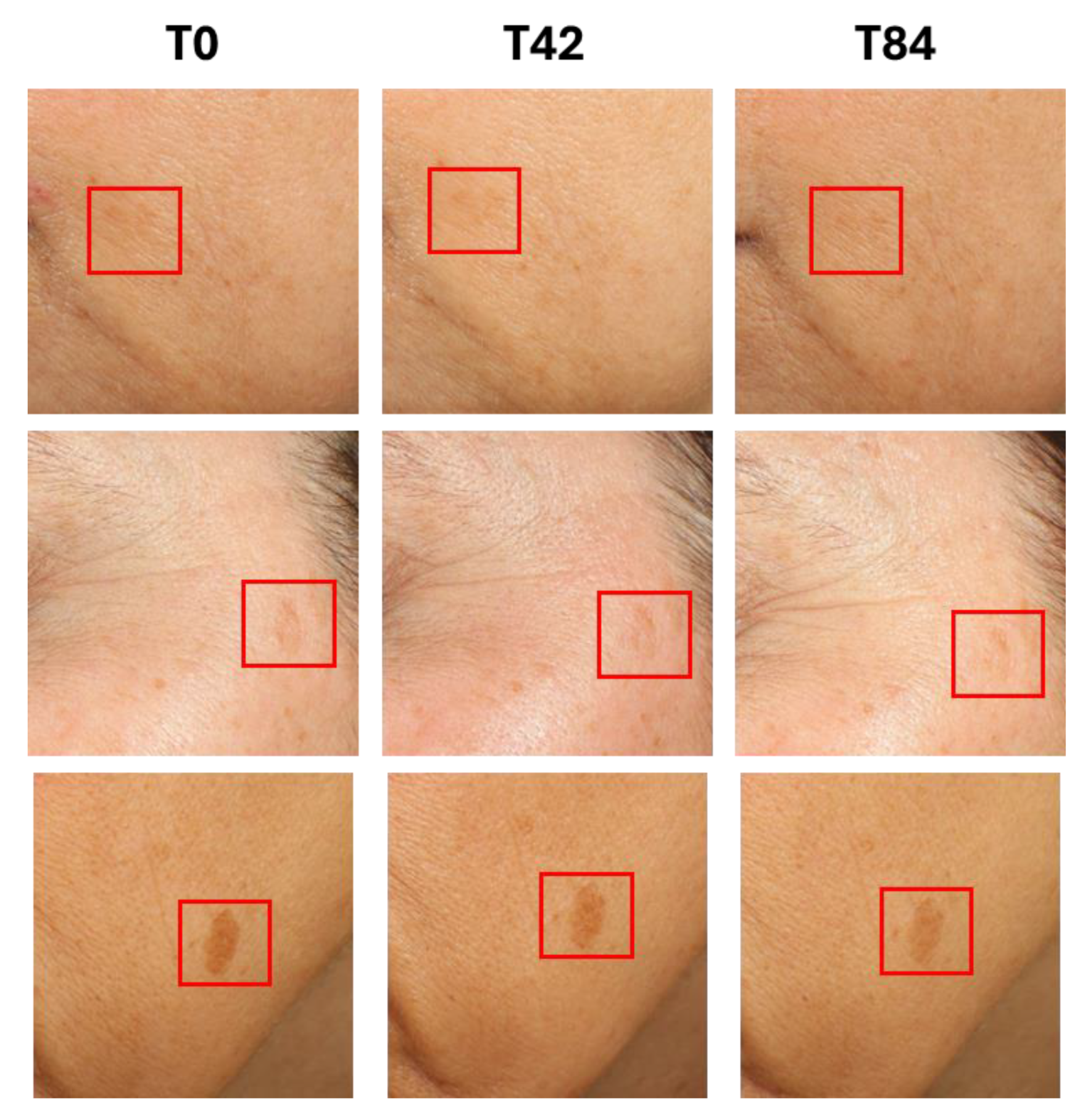
2.4.1. Color of Facial Dark Spots
2.4.2. Dark Spot Surface Measurement
2.4.3. Minimal Erythema Dose (MED)
2.4.4. Skin Redness (a* Parameter)
2.4.5. Clinical Evaluation
2.4.6. Self-Assessment Questionnaire
2.5. Statistical analysis
3. Results
| Placebo | Belight3TM | |||||
|---|---|---|---|---|---|---|
| T0 | T42 | T84 | T0 | T42 | T84 | |
| Skin Complexion evenness % subjects with improvement vs T0 |
1.9±0.1 | 1.0±0.1 3% |
1.2±0.1 23% |
1.8±0.1 | 1.4±0.1## 41% |
1.8±0.1### 72% |
| Dark spot visibility % subjects with improvement vs T0 |
--- | 1.0±0.0 3% |
1.2±0.1 17% |
--- | 1.3±0.1 25% |
1.6±0.1### 56% |
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brenner, M.; Hearing, V. J. The Protective Role of Melanin against UV Damage in Human Skin. Photochem Photobiol 2008, 84, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Zubair, R.; Lyons, A. B.; Vellaichamy, G.; Peacock, A.; Hamzavi, I. What’s New in Pigmentary Disorders. Dermatol Clin 2019, 37, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K. Large Melanosome Complex Is Increased in Keratinocytes of Solar Lentigo. Cosmetics 2017, 4, 49. [Google Scholar] [CrossRef]
- Plensdorf, S.; Livieratos, M.; Dada, N. Pigmentation Disorders: Diagnosis and Management. Am Fam Physician 2017, 96, 797–804. [Google Scholar]
- Cestari, T. F.; Dantas, L. P.; Boza, J. C. Acquired Hyperpigmentations. An Bras Dermatol 2014, 89, 11–25. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Hearing, V. J. Melanocytes and Their Diseases. Cold Spring Harb Perspect Med 2014, 4. [Google Scholar] [CrossRef]
- Cardinali, G.; Kovacs, D.; Picardo, M. Mechanisms Underlying Post-Inflammatory Hyperpigmentation: Lessons from Solar Lentigo. Ann Dermatol Venereol 2012, 139 Suppl 4, S148–152. [Google Scholar] [CrossRef]
- Panzella, L.; Napolitano, A. Natural Phenol Polymers: Recent Advances in Food and Health Applications. Antioxidants (Basel) 2017, 6. [Google Scholar] [CrossRef]
- Syder, N. C.; Quarshie, C.; Elbuluk, N. Disorders of Facial Hyperpigmentation. Dermatol Clin 2023, 41, 393–405. [Google Scholar] [CrossRef]
- Tschachler, E.; Morizot, F. Ethnic Differences in Skin Aging. In Skin Aging; Gilchrest, B. A., Krutmann, J., Eds.; Springer: Berlin, Heidelberg, 2006. [Google Scholar] [CrossRef]
- Goh, S. H. The Treatment of Visible Signs of Senescence: The Asian Experience. Br J Dermatol 1990, 122 Suppl 35, 105–109. [Google Scholar] [CrossRef]
- Kang, H. Y. Melasma and Aspects of Pigmentary Disorders in Asians. Ann Dermatol Venereol 2012, 139 Suppl 4, S144–147. [Google Scholar] [CrossRef]
- Platsidaki, E.; Efstathiou, V.; Markantoni, V.; Kouris, A.; Kontochristopoulos, G.; Nikolaidou, E.; Rigopoulos, D.; Stratigos, A.; Gregoriou, S. Self-Esteem, Depression, Anxiety and Quality of Life in Patients with Melasma Living in a Sunny Mediterranean Area: Results from a Prospective Cross-Sectional Study. Dermatol Ther (Heidelb) 2023, 13, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Duperray, J.; Sergheraert, R.; Chalothorn, K.; Tachalerdmanee, P.; Perin, F. The Effects of the Oral Supplementation of L-Cystine Associated with Reduced L-Glutathione-GSH on Human Skin Pigmentation: A Randomized, Double-Blinded, Benchmark- and Placebo-Controlled Clinical Trial. J Cosmet Dermatol 2022, 21, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Dimitrov, D.; Kroumpouzos, G. Beauty Perception: A Historical and Contemporary Review. Clin Dermatol 2023, 41, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Ruthruff, E.; Tybur, J. M.; Gaspelin, N.; Miller, G. Perception of Facial Attractiveness Requires Some Attentional Resources: Implications for the “Automaticity” of Psychological Adaptations. Evolution and Human Behavior 2012, 33, 241–250. [Google Scholar] [CrossRef]
- Nautiyal, A.; Wairkar, S. Management of Hyperpigmentation: Current Treatments and Emerging Therapies. Pigment Cell Melanoma Res 2021, 34, 1000–1014. [Google Scholar] [CrossRef]
- Pérez-Bernal, A.; Muñoz-Pérez, M. A.; Camacho, F. Management of Facial Hyperpigmentation. Am J Clin Dermatol 2000, 1, 261–268. [Google Scholar] [CrossRef]
- Davis, E. C.; Callender, V. D. Postinflammatory Hyperpigmentation: A Review of the Epidemiology, Clinical Features, and Treatment Options in Skin of Color. J Clin Aesthet Dermatol 2010, 3, 20–31. [Google Scholar]
- Makino, E. T.; Kadoya, K.; Sigler, M. L.; Hino, P. D.; Mehta, R. C. Development and Clinical Assessment of a Comprehensive Product for Pigmentation Control in Multiple Ethnic Populations. J Drugs Dermatol 2016, 15, 1562–1570. [Google Scholar]
- Rossi, A. M.; Perez, M. I. Treatment of Hyperpigmentation. Facial Plast Surg Clin North Am 2011, 19, 313–324. [Google Scholar] [CrossRef]
- Scarcella, G.; Gerber, P. A.; Edge, D.; Nielsen, M. C. E. Effective Removal of Solar Lentigines by Combination of Pre- And Post-Fluorescent Light Energy Treatment with Picosecond Laser Treatment. Clin Case Rep 2020, 8, 1429–1432. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, A.; Madan, R. Post-Inflammatory Hyperpigmentation: A Review of Treatment Strategies. J Drugs Dermatol 2020, 19, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Moolla, S.; Miller-Monthrope, Y. Dermatology: How to Manage Facial Hyperpigmentation in Skin of Colour. Drugs Context 2022, 11, 2021–11. [Google Scholar] [CrossRef] [PubMed]
- Riahi, R. R.; Bush, A. E.; Cohen, P. R. Topical Retinoids: Therapeutic Mechanisms in the Treatment of Photodamaged Skin. Am J Clin Dermatol 2016, 17, 265–276. [Google Scholar] [CrossRef]
- Nguyen, Q. H.; Bui, T. P. Azelaic Acid: Pharmacokinetic and Pharmacodynamic Properties and Its Therapeutic Role in Hyperpigmentary Disorders and Acne. Int J Dermatol 1995, 34, 75–84. [Google Scholar] [CrossRef]
- Zolghadri, S.; Bahrami, A.; Hassan Khan, M. T.; Munoz-Munoz, J.; Garcia-Molina, F.; Garcia-Canovas, F.; Saboury, A. A. A Comprehensive Review on Tyrosinase Inhibitors. J Enzyme Inhib Med Chem 2019, 34, 279–309. [Google Scholar] [CrossRef]
- Banihashemi, M.; Zabolinejad, N.; Jaafari, M. R.; Salehi, M.; Jabari, A. Comparison of Therapeutic Effects of Liposomal Tranexamic Acid and Conventional Hydroquinone on Melasma. J Cosmet Dermatol 2015, 14, 174–177. [Google Scholar] [CrossRef]
- Ebrahimi, B.; Naeini, F. F. Topical Tranexamic Acid as a Promising Treatment for Melasma. J Res Med Sci 2014, 19, 753–757. [Google Scholar]
- Karrabi, M.; David, J.; Sahebkar, M. Clinical Evaluation of Efficacy, Safety and Tolerability of Cysteamine 5% Cream in Comparison with Modified Kligman’s Formula in Subjects with Epidermal Melasma: A Randomized, Double-Blind Clinical Trial Study. Skin Res Technol 2021, 27, 24–31. [Google Scholar] [CrossRef]
- Selwyn, A.; Govindaraj, S. Study of Plant-Based Cosmeceuticals and Skin Care. South African Journal of Botany 2023, 158, 429–442. [Google Scholar] [CrossRef]
- Kanlayavattanakul, M.; Lourith, N. Plants and Natural Products for the Treatment of Skin Hyperpigmentation - A Review. Planta Med 2018, 84, 988–1006. [Google Scholar] [CrossRef] [PubMed]
- Hollinger, J. C.; Angra, K.; Halder, R. M. Are Natural Ingredients Effective in the Management of Hyperpigmentation? A Systematic Review. J Clin Aesthet Dermatol 2018, 11, 28–37. [Google Scholar] [PubMed]
- Panzella, L.; Napolitano, A. Natural and Bioinspired Phenolic Compounds as Tyrosinase Inhibitors for the Treatment of Skin Hyperpigmentation: Recent Advances. Cosmetics 2019, 6, 57. [Google Scholar] [CrossRef]
- Grimes, P.; Bhawan, J.; Howell, M.; Desai, S.; Coryell, E.; Einziger, M.; Simpson, A.; Yaroshinsky, A.; McCraw, T. Histopathological Changes Induced by Malassezin: A Novel Natural Microbiome Indole for Treatment of Facial Hyperpigmentation. J Drugs Dermatol 2022, 21, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Gao, J. The Use of Botanical Extracts as Topical Skin-Lightening Agents for the Improvement of Skin Pigmentation Disorders. J Investig Dermatol Symp Proc 2008, 13, 20–24. [Google Scholar] [CrossRef]
- Ertam, I.; Mutlu, B.; Unal, I.; Alper, S.; Kivçak, B.; Ozer, O. Efficiency of Ellagic Acid and Arbutin in Melasma: A Randomized, Prospective, Open-Label Study. J Dermatol 2008, 35, 570–574. [Google Scholar] [CrossRef]
- Bala, H. R.; Lee, S.; Wong, C.; Pandya, A. G.; Rodrigues, M. Oral Tranexamic Acid for the Treatment of Melasma: A Review. Dermatol Surg 2018, 44, 814–825. [Google Scholar] [CrossRef]
- Pillaiyar, T.; Manickam, M.; Namasivayam, V. Skin Whitening Agents: Medicinal Chemistry Perspective of Tyrosinase Inhibitors. J Enzyme Inhib Med Chem 2017, 32, 403–425. [Google Scholar] [CrossRef]
- Juhasz, M. L. W.; Levin, M. K. The Role of Systemic Treatments for Skin Lightening. J Cosmet Dermatol 2018, 17, 1144–1157. [Google Scholar] [CrossRef]
- Abdelnour, A.; Adlam, T.; Potts, G. A. Effects of Plant-Derived Dietary Supplements on Skin Health: A Review. Cureus 2023, 15. [Google Scholar] [CrossRef]
- Chan, L. K. W.; Lee, K. W. A.; Lee, C. H.; Lam, K. W. P.; Lee, K. F. V.; Wu, R.; Wan, J.; Shivananjappa, S.; Sky, W. T. H.; Choi, H.; Yi, K.-H. Cosmeceuticals in Photoaging: A Review. Skin Res Technol 2024, 30. [Google Scholar] [CrossRef] [PubMed]
- Pouchieu, C.; Pourtau, L.; Gaudout, D.; Gille, I.; Chalothorn, K.; Perin, F. Effect of an Oral Formulation on Skin Lightening: Results from In Vitro Tyrosinase Inhibition to a Double-Blind Randomized Placebo-Controlled Clinical Study in Healthy Asian Participants. Cosmetics 2023, 10, 143. [Google Scholar] [CrossRef]
- Petit, L.; Piérard, G. E. Skin-Lightening Products Revisited. Int J Cosmet Sci 2003, 25, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Ly, B. C. K.; Dyer, E. B.; Feig, J. L.; Chien, A. L.; Del Bino, S. Research Techniques Made Simple: Cutaneous Colorimetry: A Reliable Technique for Objective Skin Color Measurement. J Invest Dermatol 2020, 140, 3–12e1. [Google Scholar] [CrossRef]
- Heckman, C. J.; Chandler, R.; Kloss, J. D.; Benson, A.; Rooney, D.; Munshi, T.; Darlow, S. D.; Perlis, C.; Manne, S. L.; Oslin, D. W. Minimal Erythema Dose (MED) Testing. J Vis Exp 2013, No. 75, e50175. [Google Scholar] [CrossRef]
| Skin complexion evenness at T0 | Score | Improvement vs. T0 | Score |
|---|---|---|---|
| Skin complexion is not uniform (discolorations all over the face) | 1 | No variation | 1 |
| Skin complexion is uneven (discolorations on some parts of the face) | 2 | Slight improvement | 2 |
| Skin complexion is quite uniform | 3 | Moderate improvement | 3 |
| Skin complexion is uniform | 4 | Remarkable improvement | 4 |
| Placebo (n=30) | Belight3TM (n=31) | |
|---|---|---|
| Age, years | 42.7 ± 1.3 (Min 28; Max 52) | 46.1 ± 1.1 (Min 28; Max 52) |
| Female, n (%) | 23 (76.7%) | 25 (80.6%) |
| Skin phototype, n (%) | ||
| I | 2 (6.7%) | 1 (3.2%) |
| II | 17 (56.7%) | 13 (41.9%) |
| II | 11 (36.7%) | 17 (54.8%) |
| Heart rate (bpm) | 76.6 ± 1.6 | 73.7 ± 1.6 |
| Systolic blood pressure (mmHg) | 116.6 ± 2.9 | 115.4 ± 2.8 |
| Diastolic blood pressure (mHg) | 77.3 ± 1.6 | 75.0 ± 1.4 |
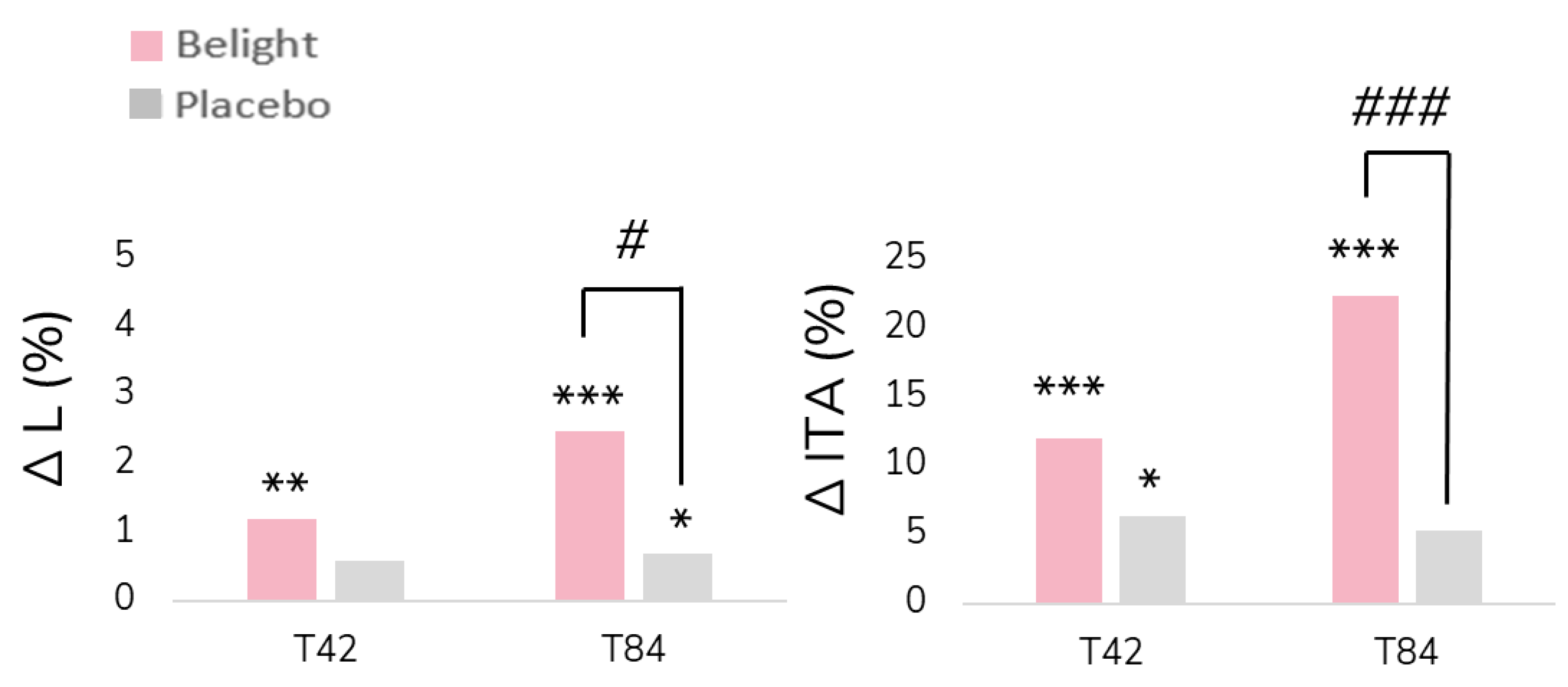
| Placebo | Belight3TM | |||||
|---|---|---|---|---|---|---|
| T0 | T42 | T84 | T0 | T42 | T84 | |
| L* value (a.u.) |
58.07 ± 0.43 |
58.39 ± 0.43 0.6% |
58.49 ± 0.44* 0.7% |
57.58 ± 0.34 | 58.25 ± 0.36** 1.2% |
59.00 ± 0.38*** 2.5%# |
| ITA ° (degrees°) |
26.03 ± 1.32 | 27.41 ± 1.31* 6.4% |
27.18 ± 1.30 5.3% |
23.11 ± 1.11 | 25.67 ± 1.21*** 12.1% |
27.79 ± 1.22*** 22.4%### |
| Spot Surface % | 2.73 ± 0.54 | 2.77 ± 0.54 2.9% |
2.79 ± 0.54 3.5% |
3.68 ± 0.60 | 3.67 ± 0.60 -0.6% |
3.62 ± 0.58 -1.0% |
| Placebo | Belight3TM | |||||
|---|---|---|---|---|---|---|
| T0 | T42 | T84 | T0 | T42 | T84 | |
| Minimal Erythema Dose | 595.8 ± 35.0 | 577.4 ± 34.5 –2.3% |
545.5 ± 34.5* –7.6% |
634.6 ± 31.7 | 618.2.4 ± 31.5 –2.3% |
606.3 ± 34.4 –4.4% |
| T0 | T0+24h | T42 | T42+24h | T84 | T84+24h | |
|---|---|---|---|---|---|---|
| Placebo | 8.97 ± 0.36 |
11.80 ± 0.34*** 34.4% |
8.66 ± 0.36 |
11.15 ± 0.34*** 34.0% |
8.53 ± 0.40 |
10.66 ± 0.31*** 30.5% |
| Belight3TM | 8.44 ± 0.32 |
11.06 ± 0.25*** 35.9% |
8.23 ± 0.26 |
10.89 ± 0.32*** 35.0% |
8.50 ± 0.27 |
10.95 ± 0.28*** 32.2% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




