Submitted:
24 December 2024
Posted:
26 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
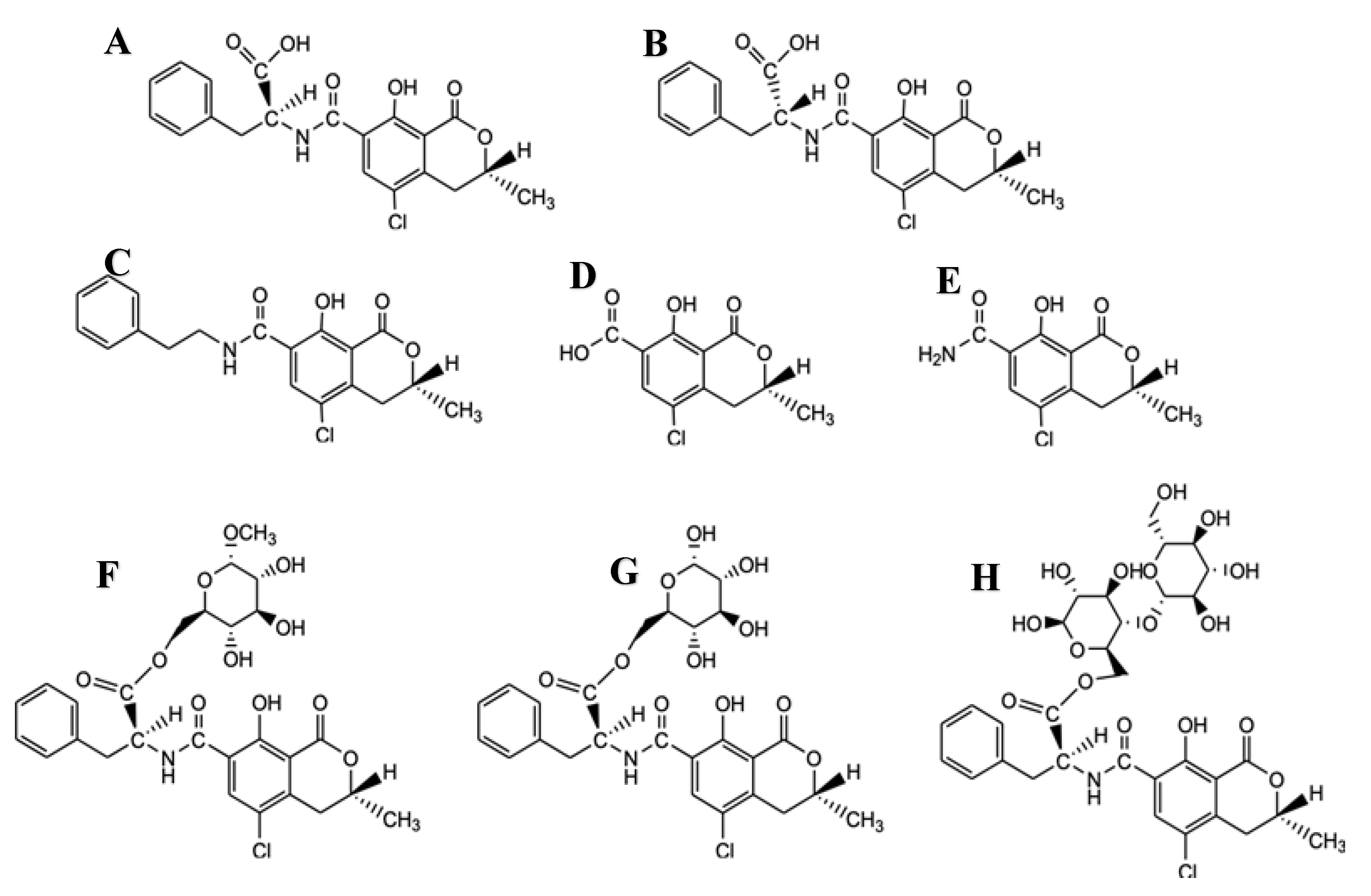
2. Chemistry and Properties of OTA
3. Sources of Exposure
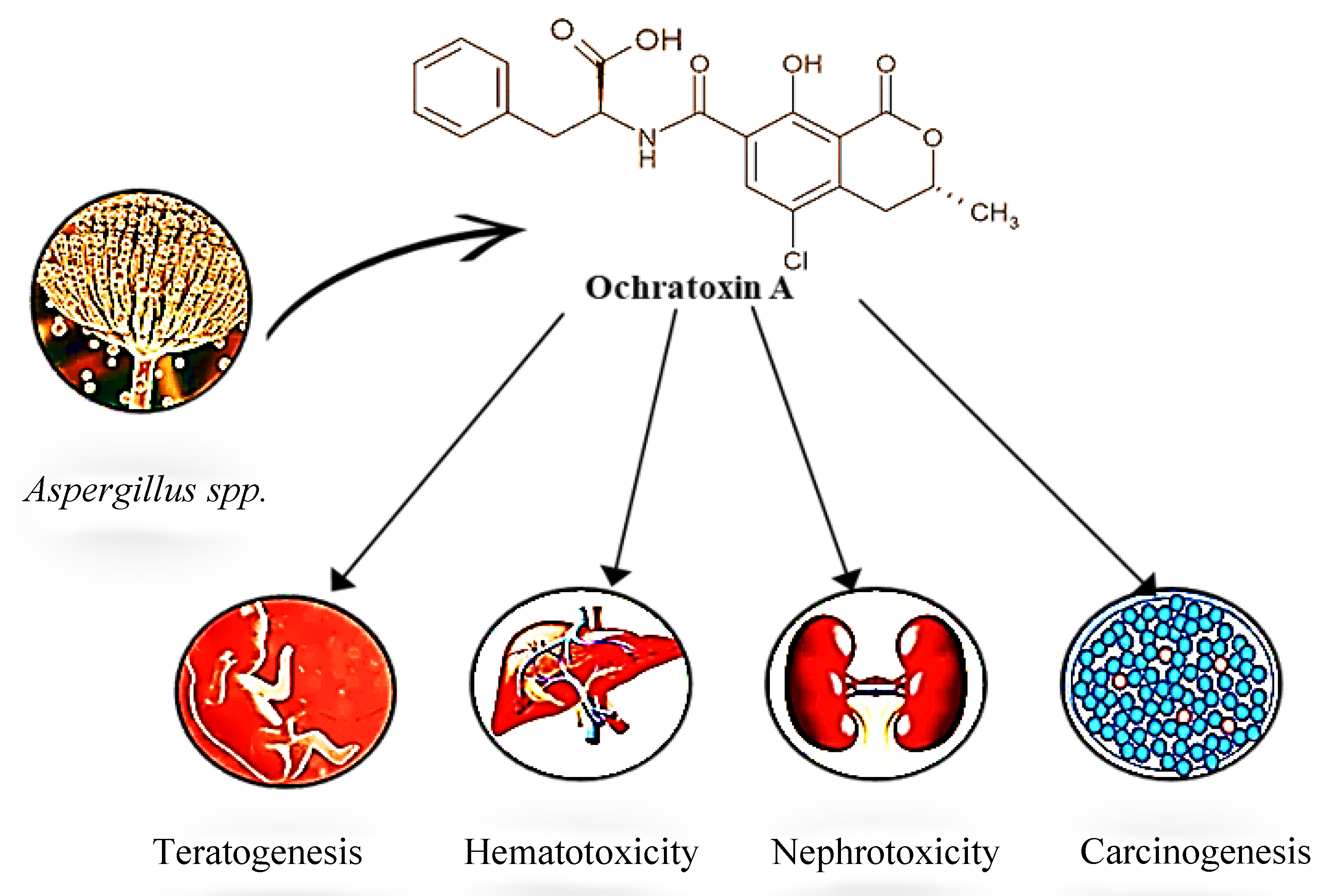
4. Toxicokinetics and Toxicity of OTA
4.1. Mechanisms of Toxicity
4.2. Toxicokinetics
4.3. Organ-Specific Toxicity
4.4. Carcinogenicity and Genotoxicity
4.5. Immunotoxicity
5. Risk Assessment and Regulatory Standards
6. Decontamination Strategies for OTA in Foods
6.1. Physical Methods
6.1.1. Gamma Radiation
6.1.2. Electron Beam Radiation
6.1.3. Ultraviolet Radiation
6.2. Non-Thermal and Bioprocessing Methods
6.2.1. Separation and Cleaning
6.2.2. Milling
6.2.3. Brewing/Fermentation
6.2.4. Cold Plasma
6.3. Thermal Methods
6.3.1. Roasting
6.3.2. Coffee Brewing
6.3.3. Extrusion
6.4. Chemical Methods
6.4.1. Ozone Treatment
6.4.2. Plant Extracts
6.4.3. Adsorbents
6.4.4. Additives
Combined Additives
6.5. Biological Methods
6.5.1. Decontamination by Bacteria
6.5.2. Decontamination by Yeast
6.5.3. Decontamination by Enzymes
6.5.4. Decontamination by Macrofungi
7. Consideration of Decontamination Methods
8. Future Prospects
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pallarés, N.; Berrada, H.; Tolosa, J.; Ferrer, E. Effect of high hydrostatic pressure (HPP) and pulsed electric field (PEF) technologies on reduction of aflatoxins in fruit juices. LWT 2021, 142. [Google Scholar] [CrossRef]
- El Khoury, A.; Atoui, A. Ochratoxin A: General Overview and Actual Molecular Status. Toxins 2010, 2, 461–493. [Google Scholar] [CrossRef] [PubMed]
- Malir, F.; Ostry, V.; Novotna, E. Toxicity of the mycotoxin ochratoxin A in the light of recent data. Toxin Rev. 2013, 32, 19–33. [Google Scholar] [CrossRef]
- Khan, R.; Ghazali, F.M.; Mahyudin, N.A.; Samsudin, N.I.P. Aflatoxin Biosynthesis, Genetic Regulation, Toxicity, and Control Strategies: A Review. J. Fungi 2021, 7, 606. [Google Scholar] [CrossRef]
- Khaneghah, A.M.; Moosavi, M.H.; Oliveira, C.A.F.; Vanin, F.; Sant'Ana, A.S. Electron beam irradiation to reduce the mycotoxin and microbial contaminations of cereal-based products: An overview. Food Chem. Toxicol. 2020, 143, 111557. [Google Scholar] [CrossRef]
- Cappozzo, J.; Jackson, L.; Lee, H.J.; Zhou, W.; Al-Taher, F.; Zweigenbaum, J.; Ryu, D. Occurrence of Ochratoxin A in Infant Foods in the United States. J. Food Prot. 2017, 80, 251–256. [Google Scholar] [CrossRef]
- Khan, R.; Anwar, F.; Ghazali, F.M. A comprehensive review of mycotoxins: Toxicology, detection, and effective mitigation approaches. Heliyon 2024, 10, e28361. [Google Scholar] [CrossRef]
- Khan, R.; Ghazali, F.M.; Mahyudin, N.A.; Samsudin, N.I.P. Chromatographic Analysis of Aflatoxigenic Aspergillus flavus Isolated from Malaysian Sweet Corn. Separations 2021, 8, 98. [Google Scholar] [CrossRef]
- Mitchell, N.J.; Chen, C.; Palumbo, J.D.; Bianchini, A.; Cappozzo, J.; Stratton, J.; Ryu, D.; Wu, F. A risk assessment of dietary Ochratoxin a in the United States. Food Chem. Toxicol. 2017, 100, 265–273. [Google Scholar] [CrossRef]
- Liu W.C, Pushparaj K, Meyyazhagan A, Arumugam V.A, Pappuswamy M, Bhotla H.K. Ochratoxin-A as an alarming health threat for livestock and human: A review on molecular interactions, mechanism of toxicity, detection, detoxification, and dietary prophylaxis. Toxicon, 2022, 213, 59-75.
- European Food Safety Authority (EFSA) Panel on Contaminants in the Food Chain (CONTAM), Schrenk D, Bodin L, Chipman J.K, del Mazo J, Grasl-Kraupp B, Hogstrand, C., Hoogenboom, L., Leblanc, J C., Nebbia, C.S. Risk assessment of ochratoxin A in food. EFSA Journal. 2020, 18(5), e06113.
- Eskola, M.; Kos, G.; Elliott, C.T.; Hajšlová, J.; Mayar, S.; Krska, R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nutr. 2020, 60, 2773–2789. [Google Scholar] [CrossRef] [PubMed]
- Scottt, P.M.; Kanhere, S.R.; Lau, B.P.; Levvis, D.A.; Hayward, S.; Ryan, J.J.; Kuiper-Goodman, T. Survey of Canadian human blood plasma for ochratoxin A. Food Addit. Contam. 1998, 15, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Studer-Rohr, I.; Schlatter, J.; Dietrich, D.R. Kinetic parameters and intraindividual fluctuations of ochratoxin A plasma levels in humans. Arch. Toxicol. 2000, 74, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Tola, M.; Kebede, B. Occurrence, importance and control of mycotoxins: A review. Cogent Food Agric. 2016, 2. [Google Scholar] [CrossRef]
- Wang, L.; Hua, X.; Shi, J.; Jing, N.; Ji, T.; Lv, B.; Liu, L.; Chen, Y. Ochratoxin A: Occurrence and recent advances in detoxification. 2022, 210, 11–18,. [CrossRef]
- Campi, M.; Dueñas, M.; Fagiolo, G. Specialization in food production affects global food security and food systems sustainability. World Dev. 2021, 141. [Google Scholar] [CrossRef]
- Khalil, O.A.; Hammad, A.A.; Sebaei, A.S. Aspergillus flavus and Aspergillus ochraceus inhibition and reduction of aflatoxins and ochratoxin A in maize by irradiation. Toxicon 2021, 198, 111–120. [Google Scholar] [CrossRef]
- Woldemariam, H.W.; Kießling, M.; Emire, S.A.; Teshome, P.G.; Töpfl, S.; Aganovic, K. Influence of electron beam treatment on naturally contaminated red pepper (Capsicum annuum L.) powder: Kinetics of microbial inactivation and physicochemical quality changes. Food Sci. Emerg. Technol. 2020, 67, 102588. [Google Scholar] [CrossRef]
- Ferreira, C.D.; Lang, G.H.; Lindemann, I.d.S.; Timm, N.d.S.; Hoffmann, J.F.; Ziegler, V.; de Oliveira, M. Postharvest UV-C irradiation for fungal control and reduction of mycotoxins in brown, black, and red rice during long-term storage. Food Chem. 2020, 339, 127810. [Google Scholar] [CrossRef]
- Sirohi, R.; Tarafdar, A.; Gaur, V.K.; Singh, S.; Sindhu, R.; Rajasekharan, R.; Madhavan, A.; Binod, P.; Kumar, S.; Pandey, A. Technologies for disinfection of food grains: Advances and way forward. Food Res. Int. 2021, 145, 110396. [Google Scholar] [CrossRef]
- Gonzalez, A.L.; Lozano, V.A.; Escandar, G.M.; Bravo, M.A. Determination of ochratoxin A in coffee and tea samples by coupling second-order multivariate calibration and fluorescence spectroscopy. Talanta 2020, 219, 121288. [Google Scholar] [CrossRef]
- Shukla, S.; Park, J.H.; Kim, M. Efficient, safe, renewable, and industrially feasible strategy employing Bacillus subtilis with alginate bead composite for the reduction of ochratoxin A from wine. J. Clean. Prod. 2019, 242, 118344. [Google Scholar] [CrossRef]
- Pereira, C.; Cunha, S.C.; Fernandes, J.O. Mycotoxins of Concern in Children and Infant Cereal Food at European Level: Incidence and Bioaccessibility. Toxins 2022, 14, 488. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ma, W.; Ma, Z.; Zhang, Q.; Li, H. The Occurrence and Contamination Level of Ochratoxin A in Plant and Animal-Derived Food Commodities. Molecules 2021, 26, 6928. [Google Scholar] [CrossRef] [PubMed]
- Kőszegi, T.; Poór, M. Ochratoxin A: Molecular Interactions, Mechanisms of Toxicity and Prevention at the Molecular Level. Toxins 2016, 8, 111. [Google Scholar] [CrossRef]
- Abrunhosa, L.; Paterson, R.R.; Venâncio, A. Biodegradation of Ochratoxin A for Food and Feed Decontamination. Toxins 2010, 2, 1078–1099. [Google Scholar] [CrossRef]
- Dahal, S.; Lee, H.J.; Gu, K.; Ryu, D. Heat Stability of Ochratoxin A in an Aqueous Buffered Model System. J. Food Prot. 2016, 79, 1748–1752. [Google Scholar] [CrossRef]
- Cramer, B.; Osteresch, B.; Muñoz, K.A.; Hillmann, H.; Sibrowski, W.; Humpf, H. Biomonitoring using dried blood spots: Detection of ochratoxin A and its degradation product 2’R-ochratoxin A in blood from coffee drinkers*. Mol. Nutr. Food Res. 2015, 59, 1837–1843. [Google Scholar] [CrossRef]
- Huff, J. Value, validity, and historical development of carcinogenesis studies for predicting and confirming carcinogenic risks to humans. In Carcinogenicity: CRC Press; 2021, p.
- Baan R.A, Straif K. The Monographs program of the International Agency for Research on Cancer: A brief history of its Preamble. ALTEX-Alternatives to Animal Experimentation. 2022, 39(3), 443-450.
- Zhai, S.; Zhu, Y.; Feng, P.; Li, M.; Wang, W.; Yang, L.; Yang, Y. Ochratoxin A: its impact on poultry gut health and microbiota, an overview. Poult. Sci. 2021, 100, 101037. [Google Scholar] [CrossRef]
- Bui-Klimke, T. R, Wu F. Ochratoxin A, and human health risk: A review of the evidence. Critical Reviews in Food Science and Nutrition, 2015, 55(13), 1860-1869.
- Lautert C, Ferreiro L, Zimmermann C.E, Castilhos L.G, de Jesus F.P, Zanette R.A, Leal, D. B., & Santurio, J. M. In vitro effects of ochratoxin A, deoxynivalenol, and zearalenone on cell viability and E-ADA activity in broiler chickens’ lymphocytes. Pesquisa Veterinária Brasileira.
- Wang, G.; Li, E.; Gallo, A.; Perrone, G.; Varga, E.; Ma, J.; Yang, B.; Tai, B.; Xing, F. Impact of environmental factors on ochratoxin A: From natural occurrence to control strategy. Environ. Pollut. 2022, 317. [Google Scholar] [CrossRef]
- Ravelo Abreu A, Rubio Armendáriz C, Gutiérrez Fernández A, Hardisson De la Torre A. La ocratoxina A en alimentos de consumo humano: revisión. Nutrición Hospitalaria, 2011, 26(6),1215-1226.
- Heussner, A.H.; Bingle, L.E.H. Comparative Ochratoxin Toxicity: A Review of the Available Data. Toxins 2015, 7, 4253–4282. [Google Scholar] [CrossRef] [PubMed]
- Roncada, P.; Altafini, A.; Fedrizzi, G.; Guerrini, A.; Polonini, G.; Caprai, E. Ochratoxin A contamination of the casing and the edible portion of artisan salamis produced in two Italian regions. World Mycotoxin J. 2020, 13, 553–562. [Google Scholar] [CrossRef]
- Heintz, M.M.; Doepker, C.L.; Wikoff, D.S.; Hawks, S.E. Assessing the food safety risk of ochratoxin A in coffee: A toxicology-based approach to food safety planning. J. Food Sci. 2021, 86, 4799–4810. [Google Scholar] [CrossRef] [PubMed]
- Mounjouenpou, P.; Gueule, D.; Fontana-Tachon, A.; Guyot, B.; Tondje, P.R.; Guiraud, J.-P. Filamentous fungi producing ochratoxin a during cocoa processing in Cameroon. Int. J. Food Microbiol. 2008, 121, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Ringot, D.; Chango, A.; Schneider, Y.-J.; Larondelle, Y. Toxicokinetics and toxicodynamics of ochratoxin A, an update. Chem. -Biol. Interact. 2005, 159, 18–46. [Google Scholar] [CrossRef]
- de Almeida, A.P.; Alaburda, J.; Shundo, L.; Ruvieri, V.; Navas, S.A.; Lamardo, L.C.A.; Sabino, M. Ochratoxin A in brazilian instant coffee. Braz. J. Microbiol. 2007, 38, 300–303. [Google Scholar] [CrossRef]
- Twarużek, M.; Kosicki, R.; Kwiatkowska-Giżyńska, J.; Grajewski, J.; Ałtyn, I. Ochratoxin A and citrinin in green coffee and dietary supplements with green coffee extract. Toxicon 2020, 188, 172–177. [Google Scholar] [CrossRef]
- Klingelhöfer, D.; Braun, M.; Schöffel, N.; Oremek, G.M.; Brüggmann, D.; Groneberg, D.A. Ochratoxin – Characteristics, influences and challenges of global research. Food Control. 2020, 114, 107230. [Google Scholar] [CrossRef]
- Serra, R.; Mendonça, C.; Venâncio, A. Ochratoxin A occurrence and formation in Portuguese wine grapes at various stages of maturation. Int. J. Food Microbiol. 2006, 111, S35–S39. [Google Scholar] [CrossRef]
- Pena, A.; Cerejo, F.; Silva, L.; Lino, C. Ochratoxin A survey in Portuguese wine by LC–FD with direct injection. Talanta 2010, 82, 1556–1561. [Google Scholar] [CrossRef]
- Farbo M.G, Urgeghe P.P, Fiori S, Marceddu S, Jaoua S, Migheli Q. Adsorption of ochratoxin A from grape juice by yeast cells immobilized in calcium alginate beads. International Journal of Food Microbiology. 2016, 217, 29-34.
- Kizis, D.; Vichou, A.-E.; Natskoulis, P.I. Recent Advances in Mycotoxin Analysis and Detection of Mycotoxigenic Fungi in Grapes and Derived Products. Sustainability 2021, 13, 2537. [Google Scholar] [CrossRef]
- Pleadin, J.; Staver, M.M.; Vahčić, N.; Kovačević, D.; Milone, S.; Saftić, L.; Scortichini, G. Survey of aflatoxin B 1 and ochratoxin A occurrence in traditional meat products coming from Croatian households and markets. Food Control. 2015, 52, 71–77. [Google Scholar] [CrossRef]
- Fan, K.; Cheng, X.; Guo, W.; Liu, X.; Zhang, Z.; Yao, Q.; Nie, D.; Yao, B.; Han, Z. Ochratoxin A in human blood plasma samples from apparently healthy volunteers in Nanjing, China. Mycotoxin Res. 2020, 36, 269–276. [Google Scholar] [CrossRef]
- Hassan, A.M.; Sheashaa, H.A.; Fattah, M.F.A.; Ibrahim, A.Z.; Gaber, O.A.; Sobh, M.A. Study of ochratoxin A as an environmental risk that causes renal injury in breast-fed Egyptian infants. Pediatr. Nephrol. 2005, 21, 102–105. [Google Scholar] [CrossRef]
- Turconi G, Guarcello M, Livieri C, Comizzoli S, Maccarini L, Castellazzi A. M, Pietri, A., Piva, G., Roggi, C. Evaluation of xenobiotics in human milk and ingestion by the newborn: An epidemiological survey in Lombardy (Northern Italy). European Journal of Nutrition. 2004, 43, 191–197.
- Martins, H.M.; Almeida, I.; Camacho, C.; Costa, J.M.; Bernardo, F. A survey on the occurrence of ochratoxin A in feeds for swine and laying hens. Mycotoxin Res. 2011, 28, 107–110. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, S.; Guo, X.; Xun, Z.; Wang, L.; Liu, Y.; Mou, W.; Qin, T.; Xu, Z.; Wang, L.; et al. Fast, portable, selective, and ratiometric determination of ochratoxin A (OTA) by a fluorescent supramolecular sensor. J. Hazard. Mater. 2023, 465, 133104. [Google Scholar] [CrossRef]
- Woo C.S.J, El-Nezami H. Maternal-fetal cancer risk assessment of ochratoxin A during pregnancy. Toxins. 2016, 8(4), 87.
- Schaaf, G.; Nijmeijer, S.; Maas, R.; Roestenberg, P.; de Groene, E.; Fink-Gremmels, J. The role of oxidative stress in the ochratoxin A-mediated toxicity in proximal tubular cells. Biochim. et Biophys. Acta (BBA) - Mol. Basis Dis. 2002, 1588, 149–158. [Google Scholar] [CrossRef]
- Lee H.J, Pyo M.C, Shin H.S, Ryu D, Lee K.W. Renal toxicity through AhR, PXR, and Nrf2 signaling pathway activation of ochratoxin A-induced oxidative stress in kidney cells. Food and Chemical Toxicology. 2018, 122, 59-68.
- Shin H.S, Lee H.J, Pyo M.C, Ryu D, Lee K.W. Ochratoxin A-induced hepatotoxicity through phase I and phase II reactions regulated by AhR in liver cells. Toxins, 2019, 11(7), 377.
- Cavin, C.; Delatour, T.; Marin-Kuan, M.; Holzhäuser, D.; Higgins, L.; Bezençon, C.; Guignard, G.; Junod, S.; Richoz-Payot, J.; Gremaud, E.; et al. Reduction in Antioxidant Defenses may Contribute to Ochratoxin A Toxicity and Carcinogenicity. Toxicol. Sci. 2006, 96, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Marin-Kuan, M.; Nestler, S.; Verguet, C.; Bezençon, C.; Piguet, D.; Mansourian, R.; Holzwarth, J.; Grigorov, M.; Delatour, T.; Mantle, P.; et al. A Toxicogenomics Approach to Identify New Plausible Epigenetic Mechanisms of Ochratoxin A Carcinogenicity in Rat. Toxicol. Sci. 2005, 89, 120–134. [Google Scholar] [CrossRef] [PubMed]
- Boesch-Saadatmandi, C.; Loboda, A.; Jozkowicz, A.; Huebbe, P.; Blank, R.; Wolffram, S.; Dulak, J.; Rimbach, G. Effect of ochratoxin A on redox-regulated transcription factors, antioxidant enzymes and glutathione-S-transferase in cultured kidney tubulus cells. Food Chem. Toxicol. 2008, 46, 2665–2671. [Google Scholar] [CrossRef] [PubMed]
- Boesch-Saadatmandi, C.; Wagner, A.E.; Graeser, A.C.; Hundhausen, C.; Wolffram, S.; Rimbach, G. Ochratoxin A impairs Nrf2-dependent gene expression in porcine kidney tubulus cells. J. Anim. Physiol. Anim. Nutr. 2009, 93, 547–554. [Google Scholar] [CrossRef] [PubMed]
- García-Pérez E, Ryu D, Kim H.Y, Kim H.D, Lee H.J. Human proximal tubule epithelial cells (HK-2) as a sensitive in vitro system for ochratoxin A-induced oxidative stress. Toxins, 2021, 13(11), 787.
- García-Pérez, E.; Ryu, D.; Lee, C.; Lee, H.J. Ochratoxin A Induces Oxidative Stress in HepG2 Cells by Impairing the Gene Expression of Antioxidant Enzymes. Toxins 2021, 13, 271. [Google Scholar] [CrossRef]
- Descamps-Latscha, B.; Witko-Sarsat, V. Importance of oxidatively modified proteins in chronic renal failure. Kidney Int. 2001, 59, 108–113. [Google Scholar] [CrossRef]
- Beisswenger, P.J.; Drummond, K.S.; Nelson, R.G.; Howell, S.K.; Szwergold, B.S.; Mauer, M. Susceptibility to Diabetic Nephropathy Is Related to Dicarbonyl and Oxidative Stress. Diabetes 2005, 54, 3274–3281. [Google Scholar] [CrossRef]
- Coşkun, C.; Kural, A.; Döventaş, Y.; Koldaş, M.; Özturk, H.; İnal, B.B.; Gümüş, A. Hemodialysis and Protein Oxidation Products. Ann. New York Acad. Sci. 2007, 1100, 404–408. [Google Scholar] [CrossRef]
- Jonker, J.W.; Merino, G.; Musters, S.; E van Herwaarden, A.; Bolscher, E.; Wagenaar, E.; Mesman, E.; Dale, T.C.; Schinkel, A.H. The breast cancer resistance protein BCRP (ABCG2) concentrates drugs and carcinogenic xenotoxins into milk. Nat. Med. 2005, 11, 127–129. [Google Scholar] [CrossRef]
- Schrickx, J.; Lektarau, Y.; Fink-Gremmels, J. Ochratoxin A secretion by ATP-dependent membrane transporters in Caco-2 cells. Arch. Toxicol. 2005, 80, 243–249. [Google Scholar] [CrossRef]
- Walker, R.; Larsen, J.C. Ochratoxin A: Previous risk assessments and issues arising. Food Addit. Contam. 2005, 22, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Nogueira W.V, Buffon J.G. Micotoxinas em pescado: ocorrência e risco de exposição. REALIDADES E PERSPECTIVAS. 2020:30.
- Peraica, M.; Flajs, D.; Domijan, A.-M.; Ivić, D.; Cvjetković, B. Ochratoxin A Contamination of Food from Croatia. Toxins 2010, 2, 2098–2105. [Google Scholar] [CrossRef] [PubMed]
- Bando E, Gonçales L, Tamura N. Machinski Junior, M. J Bras Patol Med Lab. 2007, 43, 175.
- Sekiyama, B.L.; Ribeiro, A.B.; Machinski, P.A.; Junior, M.M. Aflatoxins, ochratoxin A and zearalenone in maize-based food products. Braz. J. Microbiol. 2005, 36, 289–294. [Google Scholar] [CrossRef]
- Bakker M, Pieters M.N. Risk assessment of ochratoxin A in the Netherlands. 2003.
- Rousseau J. Ochratoxin A in wines: current knowledge. Vinidea net Wine Internet Technical J. 2004;(5).
- Walker, R. Risk assessment of ochratoxin: current views of the European Scientific Committee on Food, the JECFA, and the Codex Committee on Food Additives and Contaminants. Mycotoxins and Food Safety. 2002, 249–255. [Google Scholar]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to ochratoxin A in food. EFSA Journal. 2006, 4(6), 365.
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific opinion on the risk for public health related to the presence of mercury and methylmercury in food. EFSA Journal. 2012, 10(12), 2985.
- Khodaei, D.; Javanmardi, F.; Khaneghah, A.M. The global overview of the occurrence of mycotoxins in cereals: a three-year survey. Curr. Opin. Food Sci. 2020, 39, 36–42. [Google Scholar] [CrossRef]
- Ismail, A.; Naeem, I.; Gong, Y.Y.; Routledge, M.N.; Akhtar, S.; Riaz, M.; Ramalho, L.N.Z.; de Oliveira, C.A.F.; Ismail, Z. Early life exposure to dietary aflatoxins, health impact and control perspectives: A review. Trends Food Sci. Technol. 2021, 112, 212–224. [Google Scholar] [CrossRef]
- Maatouk, I.; Mehrez, A.; Ben Amara, A.; Chayma, R.; Abid, S.; Jerbi, T.; Landoulsi, A. Effects of Gamma Irradiation on Ochratoxin A Stability and Cytotoxicity in Methanolic Solutions and Potential Application in Tunisian Millet Samples. J. Food Prot. 2019, 82, 1433–1439. [Google Scholar] [CrossRef]
- Calado, T.; Fernández-Cruz, M.L.; Verde, S.C.; Venâncio, A.; Abrunhosa, L. Gamma irradiation effects on ochratoxin A: Degradation, cytotoxicity and application in food. Food Chem. 2018, 240, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Kalagatur N.K, Gurunathan S, Kamasani J.R, Gunti L, Kadirvelu K, Mohan C.D, Rangappa S, Prasad R, Almeida F, Mudili V. Inhibitory effect of C. zeylanicum, C. longa, O. basilicum, Z. officinale, and C. martini essential oils on growth and ochratoxin A content of A. ochraceous and P. verrucosum in maize grains. Biotechnology Reports. 2020, 27, e00490.
- Popović, V.; Fairbanks, N.; Pierscianowski, J.; Biancaniello, M.; Zhou, T.; Koutchma, T. Feasibility of 3D UV-C treatment to reduce fungal growth and mycotoxin loads on maize and wheat kernels. Mycotoxin Res. 2018, 34, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Shanakhat, H.; Sorrentino, A.; Raiola, A.; Reverberi, M.; Salustri, M.; Masi, P.; Cavella, S. Technological properties of durum wheat semolina treated by heating and UV irradiation for reduction of mycotoxin content. J. Food Process. Eng. 2019, 42. [Google Scholar] [CrossRef]
- Casas-Junco, P.P.; Solís-Pacheco, J.R.; Ragazzo-Sánchez, J.A.; Aguilar-Uscanga, B.R.; Bautista-Rosales, P.U.; Calderón-Santoyo, M. Cold Plasma Treatment as an Alternative for Ochratoxin A Detoxification and Inhibition of Mycotoxigenic Fungi in Roasted Coffee. Toxins 2019, 11, 337. [Google Scholar] [CrossRef]
- Guo, Y.; Zhao, L.; Ma, Q.; Ji, C. Novel strategies for degradation of aflatoxins in food and feed: A review. Food Res. Int. 2020, 140, 109878. [Google Scholar] [CrossRef]
- Pereira, E.; Barros, L.; Antonio, A.L.; Verde, S.C.; Santos-Buelga, C.; Ferreira, I.C.F.R.; Rodrigues, P. Is Gamma Radiation Suitable to Preserve Phenolic Compounds and to Decontaminate Mycotoxins in Aromatic Plants? A Case-Study with Aloysia citrodora Paláu. Molecules 2017, 22, 347. [Google Scholar] [CrossRef]
- Kumar S, Kunwar A, Gautam S, Sharma A. Inactivation of A. ochraceus spores and detoxification of ochratoxin A in coffee beans by gamma irradiation. Journal of Food Science, 2012, 77(2), T44-T51.
- Refai M, Aziz N, El-Far F, Hassan A. Detection of ochratoxin produced by A. ochraceus in feedstuffs and its control by γ radiation. Applied Radiation and Isotopes, 1996, 47(7), 617-621.
- Deng L.Z, Tao Y, Mujumdar A.S, Pan Z, Chen C, Yang X.H, Liu Z.L, Wang H, Xiao H.W. Recent advances in non-thermal decontamination technologies for microorganisms and mycotoxins in low-moisture foods. Trends in Food Science & Technology. 2020, 106, 104-412.
- Mehrez, A.; Maatouk, I.; Romero-González, R.; Ben Amara, A.; Kraiem, M.; Frenich, A.G.; Landoulsi, A. Assessment of ochratoxin A stability following gamma irradiation: experimental approaches for feed detoxification perspectives. World Mycotoxin J. 2016, 9, 289–298. [Google Scholar] [CrossRef]
- Liu, R.; Wang, R.; Lu, J.; Chang, M.; Jin, Q.; Du, Z.; Wang, S.; Li, Q.; Wang, X. Degradation of AFB1 in aqueous medium by electron beam irradiation: Kinetics, pathway and toxicology. Food Control. 2016, 66, 151–157. [Google Scholar] [CrossRef]
- Pankaj, S.K.; Wan, Z.; Keener, K.M. Effects of Cold Plasma on Food Quality: A Review. Foods 2018, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Qi, L.; Liu, Y.; Wang, R.; Yang, D.; Li, K.; Wang, L.; Li, Y.; Zhang, Y.; Chen, Z. Effects of Electron Beam Irradiation on Zearalenone and Ochratoxin A in Naturally Contaminated Corn and Corn Quality Parameters. Toxins 2017, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.; Wang, J. Degradation of 3-chloro-4-hydroxybenzoic acid in biological treated effluent by gamma irradiation. Radiat. Phys. Chem. 2016, 119, 194–199. [Google Scholar] [CrossRef]
- Rifna, E.; Ramanan, K.R.; Mahendran, R. Emerging technology applications for improving seed germination. Trends Food Sci. Technol. 2019, 86, 95–108. [Google Scholar] [CrossRef]
- Falguera V, Pagán J, Garza S, Garvín A, Ibarz A. Ultraviolet processing of liquid food: A review: Part 2: Effects on microorganisms and food components and properties. Food Research International, 2011, 44(6), 1580-1588.
- Food, Drug Administration. Irradiation in the production, processing, and handling of food. Final rule. Federal Register, 2012, 77(112), 34212-3415.
- Garg N, Aggarwal M, Javed S, Khandal R.K. Studies for optimization of conditions for reducing aflatoxin contamination in peanuts using ultraviolet radiations. International Journal of Drug Development and Research. 2013, 5(3), 408-424.
- Akhila, P.P.; Sunooj, K.V.; Aaliya, B.; Navaf, M.; Sudheesh, C.; Sabu, S.; Sasidharan, A.; Mir, S.A.; George, J.; Khaneghah, A.M. Application of electromagnetic radiations for decontamination of fungi and mycotoxins in food products: A comprehensive review. Trends Food Sci. Technol. 2021, 114, 399–409. [Google Scholar] [CrossRef]
- Byun K.H, Park S.Y, Lee D.U, Chun H.S, Ha S.D. Effect of UV-C irradiation on inactivation of Aspergillus flavus and Aspergillus parasiticus and quality parameters of roasted coffee bean (Coffea arabica L.). Food Additives & Contaminants: Part A. 2020, 37(3), 507-518.
- Amézqueta, S.; González-Peñas, E.; Murillo, M.; de Cerain, A.L. Occurrence of ochratoxin A in cocoa beans: Effect of shelling. Food Addit. Contam. 2005, 22, 590–596. [Google Scholar] [CrossRef]
- Scudamore, K.A.; Banks, J.; MacDonald, S.J. Fate of ochratoxin A in the processing of whole wheat grains during milling and bread production. Food Addit. Contam. 2003, 20, 1153–1163. [Google Scholar] [CrossRef]
- Park, J. W, Chung S. B. Fate of ochratoxin A during cooking of naturally contaminated polished rice. Journal of Food Protection. 2005, 68(10), 2107–2111. [Google Scholar]
- Mansouri-Nasrabadi, R.; Milani, J.M.; Nazari, S.S.J. Optimization of washing and cooking processes of rice for Ochratoxin A decrement by RSM. Food Sci. Nutr. 2018, 6, 2523–2529. [Google Scholar] [CrossRef]
- Blanc, M.; Pittet, A.; Muñoz-Box, R.; Viani, R. Behavior of Ochratoxin A during Green Coffee Roasting and Soluble Coffee Manufacture. J. Agric. Food Chem. 1998, 46, 673–675. [Google Scholar] [CrossRef] [PubMed]
- Jalili, M.; Jinap, S.; Son, R. The effect of chemical treatment on reduction of aflatoxins and ochratoxin A in black and white pepper during washing. Food Addit. Contam. Part A 2011, 28, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Iha, M.; Trucksess, M.; Tournas, V. Effect of processing on ochratoxin A content in dried beans. Food Addit. Contam. Part A 2009, 26, 1389–1395. [Google Scholar] [CrossRef] [PubMed]
- Amézqueta, S.; González-Peñas, E.; Lizarraga, T.; Murillo-Arbizu, M.; de Cerain, A.L. A Simple Chemical Method Reduces Ochratoxin A in Contaminated Cocoa Shells. J. Food Prot. 2008, 71, 1422–1426. [Google Scholar] [CrossRef]
- Osborne, B.G.; Ibe, F.; Brown, G.L.; Petagine, F.; Scudamore, K.A.; Banks, J.N.; Hetmanski, M.T.; Leonard, C.T. The effects of milling and processing on wheat contaminated with ochratoxin A. Food Addit. Contam. 1996, 13, 141–153. [Google Scholar] [CrossRef]
- Peng, C.; Wang, L.; An, F.; Zhang, L.; Wang, Y.; Li, S.; Wang, C.; Liu, H. Fate of ochratoxin A during wheat milling and some Chinese breakfast processing. Food Control. 2015, 57, 142–146. [Google Scholar] [CrossRef]
- Scott P, Kanhere S, Lawrence G, Daley E, Farber J. Fermentation of wort containing added ochratoxin A and fumonisins B1 and B2. Food Additives & Contaminants, 1995, 12(1), 31-40.
- Kuruc, J.; Hegstad, J.; Lee, H.J.; Simons, K.; Ryu, D.; Wolf-Hall, C. Infestation and Quantification of Ochratoxigenic Fungi in Barley and Wheat Naturally Contaminated with Ochratoxin A. J. Food Prot. 2015, 78, 1350–1356. [Google Scholar] [CrossRef]
- Krogh P, Hald B, Gjertsen P, Myken F. Fate of ochratoxin A and citrinin during malting and brewing experiments. Applied Microbiology, 1974, 28(1), 31-34.
- Chu F, Chang C, Ashoor S.H, Prentice N. Stability of aflatoxin B1 and ochratoxin A in brewing. Applied Microbiology. 1975, 29(3), 313-316.
- Nip, W.K.; Chang, F.C.; Chu, F.S.; Prentice, N. Fate of ochratoxin A in brewing. Appl. Microbiol. 1975, 30, 1048–1049. [Google Scholar] [CrossRef]
- Inoue, T.; Nagatomi, Y.; Uyama, A.; Mochizuki, N. Fate of Mycotoxins during Beer Brewing and Fermentation. Biosci. Biotechnol. Biochem. 2013, 77, 1410–1415. [Google Scholar] [CrossRef]
- Delage, N.; D’harlingue, A.; Ceccaldi, B.C.; Bompeix, G. Occurrence of mycotoxins in fruit juices and wine. Food Control. 2003, 14, 225–227. [Google Scholar] [CrossRef]
- Zimmerli, B.; Dick, R. Ochratoxin A in table wine and grape-juice: Occurrence and risk assessment∗. Food Addit. Contam. 1996, 13, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Freire, L.; Braga, P.A.; Furtado, M.M.; Delafiori, J.; Dias-Audibert, F.L.; Pereira, G.E.; Reyes, F.G.; Catharino, R.R.; Sant’ana, A.S. From grape to wine: Fate of ochratoxin A during red, rose, and white winemaking process and the presence of ochratoxin derivatives in the final products. Food Control. 2020, 113, 107167. [Google Scholar] [CrossRef]
- Csutorás, C.; Rácz, L.; Rácz, K.; Fűtő, P.; Forgó, P.; Kiss, A. Monitoring of ochratoxin A during the fermentation of different wines by applying high toxin concentrations. Microchem. J. 2012, 107, 182–184. [Google Scholar] [CrossRef]
- Cecchini, F.; Morassut, M.; Moruno, E.G.; Di Stefano, R. Influence of yeast strain on ochratoxin A content during fermentation of white and red must. Food Microbiol. 2006, 23, 411–417. [Google Scholar] [CrossRef]
- Yu, J.; Smith, I.N.; Mikiashvili, N. Reducing Ochratoxin A Content in Grape Pomace by Different Methods. Toxins 2020, 12, 424. [Google Scholar] [CrossRef] [PubMed]
- Valle-Algarra, F.; Mateo, E.; Medina, .; Mateo, F.; Gimeno-Adelantado, J.; Jiménez, M. Changes in ochratoxin A and type B trichothecenes contained in wheat flour during dough fermentation and bread-baking. Food Addit. Contam. Part A 2009, 26, 896–906. [CrossRef]
- Milani, J.; Heidari, S. Stability of Ochratoxin a During Bread Making Process. J. Food Saf. 2016, 37. [Google Scholar] [CrossRef]
- Hojnik, N.; Cvelbar, U.; Tavčar-Kalcher, G.; Walsh, J.L.; Križaj, I. Mycotoxin Decontamination of Food: Cold Atmospheric Pressure Plasma versus “Classic” Decontamination. Toxins 2017, 9, 151. [Google Scholar] [CrossRef]
- Ekezie F.G.C, Sun D.W, Cheng J.H. A review on recent advances in cold plasma technology for the food industry: Current applications and future trends. Trends In Food Science & Technology, 2017, 69, 46-58.
- Ten Bosch L, Pfohl K, Avramidis G, Wieneke S, Viöl W, Karlovsky P. Plasma-based degradation of mycotoxins produced by Fusarium, Aspergillus and Alternaria species. Toxins. 2017, 9(3), 97.
- Ouf S.A, Basher A.H, Mohamed A.A.H. Inhibitory effect of double atmospheric pressure argon cold plasma on spores and mycotoxin production of Aspergillus niger contaminating date palm fruits. Journal of the Science of Food and Agriculture.
- Devi, Y.; Thirumdas, R.; Sarangapani, C.; Deshmukh, R.; Annapure, U. Influence of cold plasma on fungal growth and aflatoxins production on groundnuts. Food Control. 2017, 77, 187–191. [Google Scholar] [CrossRef]
- Kabak, B. The fate of mycotoxins during thermal food processing. J. Sci. Food Agric. 2009, 89, 549–554. [Google Scholar] [CrossRef]
- Boudra, H.; Le Bars, P.; Le Bars, J. Thermostability of Ochratoxin A in wheat under two moisture conditions. Appl. Environ. Microbiol. 1995, 61, 1156–8. [Google Scholar] [CrossRef]
- Khan, R.; Ghazali, F.M.; Mahyudin, N.A.; Samsudin, N.I.P. Biocontrol of Aflatoxins Using Non-Aflatoxigenic Aspergillus flavus: A Literature Review. J. Fungi 2021, 7, 381. [Google Scholar] [CrossRef]
- Nehad, E.A.; Farag, M.M.; Kawther, M.S.; Abdel-Samed, A.K.M.; Naguib, K. Stability of ochratoxin A (OTA) during processing and decaffeination in commercial roasted coffee beans. Food Addit. Contam. Part A 2005, 22, 761–767. [Google Scholar] [CrossRef]
- Castellanos-Onorio, O.; Gonzalez-Rios, O.; Guyot, B.; Fontana, T.A.; Guiraud, J.; Schorr-Galindo, S.; Durand, N.; Suárez-Quiroz, M. Effect of two different roasting techniques on the Ochratoxin A (OTA) reduction in coffee beans (Coffea arabica). Food Control. 2011, 22, 1184–1188. [Google Scholar] [CrossRef]
- Romani, S.; Pinnavaia, G.G.; Dalla Rosa, M. Influence of roasting levels on ochratoxin a content in coffee. J. Agric. Food Chem. 2003, 51, 5168–5171. [Google Scholar] [CrossRef]
- Tsubouchi, H.; Yamamoto, K.; Hisada, K.; Sakabe, Y.; Udagawa, S.I. Effect of roasting on ochratoxin A level in green coffee beans inoculated with Aspergillus ochraceus. Mycopathologia 1987, 97, 111–115. [Google Scholar] [CrossRef]
- Suárez-Quiroz, M.; De Louise, B.; Gonzalez-Rios, O.; Barel, M.; Guyot, B.; Schorr-Galindo, S.; Guiraud, J. The impact of roasting on the ochratoxin A content of coffee. Int. J. Food Sci. Technol. 2005, 40, 605–611. [Google Scholar] [CrossRef]
- Perez de Obanos A, Gonzalez-Penas E, Lopez de Cerain A. Influence of roasting and brew preparation on the ochratoxin A content in coffee infusion. Food Additives and Contaminants. 2005, 22(5), 463–471.
- Viani, R. Fate of ochratoxin A (OTA) during the processing of coffee. 1996.
- Lee, H.J. Stability of ochratoxin A in oats during roasting with reducing sugars. Food Control. 2020, 118, 107382. [Google Scholar] [CrossRef]
- Carbon, H.; Lee, H. Varied reduction of ochratoxin A in brown and white rice during roasting. World Mycotoxin J. 2022, 15, 361–368. [Google Scholar] [CrossRef]
- Manda, P.; Dano, D.S.; Kouadio, J.H.; Diakité, A.; Sangaré-Tigori, B.; Ezoulin, M.J.M.; Soumahoro, A.; Dembele, A.; Fourny, G. Impact of industrial treatments on ochratoxin A content in artificially contaminated cocoa beans. Food Addit. Contam. Part A 2009, 26, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Lee H.J, Gu B.J, Ganjyal G, Ryu D. Reduction of ochratoxin A in direct steam injected oat-based infant cereals with baking soda. Food Control, 2019, 96, 441-444.
- Osborne, B.G. Reversed-phase high performance liquid chromatography determination of ochratoxin a in flour and bakery products. J. Sci. Food Agric. 1979, 30, 1065–1070. [Google Scholar] [CrossRef]
- Milanez, T.V.; Leitão, M.F.F. The effect of cooking on ochratoxin A content of beans, variety ‘Carioca’. Food Addit. Contam. 1996, 13, 89–93. [Google Scholar] [CrossRef]
- Trenk, H. L, Butz M. S. Production of ochratoxins in different cereal products by Aspergillus ochraceus. Applied Microbiology. 1971, 21(6), 1032–1035. [Google Scholar]
- Lee, H. J, Lee C, Ryu D. Effects of baking soda and fructose in reduction of ochratoxin A in rice and oat porridge during retorting process. Food Control. 2020, 116, 107325. [Google Scholar]
- Lee, H. J, Li S, Gu K, Ryu D. Reduction of ochratoxin A during the preparation of porridge with sodium bicarbonate and fructose. Toxins. 2021, 13(3), 224. [Google Scholar]
- Romani, S.; Sacchetti, G.; López, C.C.; Pinnavaia, G.G.; Rosa, M.D. Screening on the Occurrence of Ochratoxin A in Green Coffee Beans of Different Origins and Types. J. Agric. Food Chem. 2000, 48, 3616–3619. [Google Scholar] [CrossRef]
- Gahlawat P, Sehgal S. Phytic acid, saponins, and polyphenols in weaning foods prepared from oven-heated green gram and cereals. Cereal Chemistry. 1992, 69(4), 463-464.
- Cramer, B.; Königs, M.; Humpf, H.-U. Identification and in Vitro Cytotoxicity of Ochratoxin A Degradation Products Formed during Coffee Roasting. J. Agric. Food Chem. 2008, 56, 5673–5681. [Google Scholar] [CrossRef] [PubMed]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef] [PubMed]
- Malir, F.; Ostry, V.; Pfohl-Leszkowicz, A.; Toman, J.; Bazin, I.; Roubal, T. Transfer of Ochratoxin A into Tea and Coffee Beverages. Toxins 2014, 6, 3438–3453. [Google Scholar] [CrossRef] [PubMed]
- Harper, J. A comparative analysis of single and twin-screw extruders. Food Extrusion Science and Technology. 1992, 139–148. [Google Scholar]
- Scudamore, K.A.; Banks, J.N.; Guy, R.C.E. Fate of ochratoxin A in the processing of whole wheat grain during extrusion. Food Addit. Contam. 2004, 21, 488–497. [Google Scholar] [CrossRef]
- Castells, M.; Pardo, E.; Ramos, A.J.; Sanchis, V.; Marín, S.; MIREN CASTELLS, ESTER PARDO, ANTONIO J. RAMOS, VICENTE SANCHIS, and SONIA MARÍN*Food Technology Department, University of Lleida, Rovira Roure 191, 25198 Lleida, Spain; Lee, H.J.; Dahal, S.; Perez, E.G.; Kowalski, R.J.; et al. Reduction of Ochratoxin A in Extruded Barley Meal. J. Food Prot. 2006, 69, 1139–1143. [CrossRef]
- Lee, H.J.; Dahal, S.; Perez, E.G.; Kowalski, R.J.; Ganjyal, G.M.; Ryu, D. Reduction of Ochratoxin A in Oat Flakes by Twin-Screw Extrusion Processing. J. Food Prot. 2017, 80, 1628–1634. [Google Scholar] [CrossRef]
- Ryu, D.; Kowalski, R.J.; Ganjyal, G.; Lee, H.J. Reduction of ochratoxin A in oats and rice by twin-screw extrusion processing with baking soda. Food Control. 2019, 105, 21–28. [Google Scholar] [CrossRef]
- Laika, J.; Viteritti, E.; Molina-Hernandez, J.B.; Sergi, M.; Neri, L.; Laurita, R.; Tappi, S.; Ricci, A.; Chaves-López, C. Efficiency of cold atmospheric plasma under ozone (O3) and nitrogen oxide (NOx) regimes on the degradation of aflatoxins and ochratoxin A in solid state and in spiked pistachio kernels. Food Control. 2024, 159. [Google Scholar] [CrossRef]
- Mayookha, V.; Pandiselvam, R.; Kothakota, A.; Ishwarya, S.P.; Khanashyam, A.C.; Kutlu, N.; Rifna, E.; Kumar, M.; Panesar, P.S.; El-Maksoud, A.A.A. Ozone and cold plasma: Emerging oxidation technologies for inactivation of enzymes in fruits, vegetables, and fruit juices. Food Control. 2022, 144. [Google Scholar] [CrossRef]
- Loffredo, E.; Scarcia, Y.; Parlavecchia, M. Removal of ochratoxin A from liquid media using novel low-cost biosorbents. Environ. Sci. Pollut. Res. 2020, 27, 34484–34494. [Google Scholar] [CrossRef] [PubMed]
- Mohos, V.; Faisal, Z.; Fliszár-Nyúl, E.; Szente, L.; Poór, M. Testing the extraction of 12 mycotoxins from aqueous solutions by insoluble beta-cyclodextrin bead polymer. Environ. Sci. Pollut. Res. 2021, 29, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Ponzilacqua, B.; Rottinghaus, G.; Landers, B.; Oliveira, C. Effects of medicinal herb and Brazilian traditional plant extracts on in vitro mycotoxin decontamination. Food Control. 2019, 100, 24–27. [Google Scholar] [CrossRef]
- Karaca, H.; Velioglu, Y.S. Ozone Applications in Fruit and Vegetable Processing. Food Rev. Int. 2007, 23, 91–106. [Google Scholar] [CrossRef]
- Zhu, F. Effect of ozone treatment on the quality of grain products. 2018, 264, 358–366. [CrossRef]
- Mohammadi Kouchesfahani M, Alimohammadi M, Jahed Khaniki G, Nabizadeh Nodehi R, Aghamohseni Z, Moazeni M, Rezaie S. Antifungal effects of ozonated water on Aspergillus parasiticus: A new approach to prevent wheat contamination. Journal of Food Safety. 2015, 35(3), 295-302.
- Savi, G.D.; Piacentini, K.C.; Bittencourt, K.O.; Scussel, V.M. Ozone treatment efficiency on Fusarium graminearum and deoxynivalenol degradation and its effects on whole wheat grains (Triticum aestivum L.) quality and germination. J. Stored Prod. Res. 2014, 59, 245–253. [Google Scholar] [CrossRef]
- Piemontese, L.; Messia, M.C.; Marconi, E.; Falasca, L.; Zivoli, R.; Gambacorta, L.; Perrone, G.; Solfrizzo, M. Effect of gaseous ozone treatments on DON, microbial contaminants and technological parameters of wheat and semolina. Food Addit. Contam. Part A 2018, 35, 761–772. [Google Scholar] [CrossRef]
- Li, M.; Guan, E.; Bian, K. Structure Elucidation and Toxicity Analysis of the Degradation Products of Deoxynivalenol by Gaseous Ozone. Toxins 2019, 11, 474. [Google Scholar] [CrossRef]
- Sun, C.; Ji, J.; Wu, S.; Sun, C.; Pi, F.; Zhang, Y.; Tang, L.; Sun, X. Saturated aqueous ozone degradation of deoxynivalenol and its application in contaminated grains. Food Control. 2016, 69, 185–190. [Google Scholar] [CrossRef]
- Iacumin, L.; Manzano, M.; Comi, G. Prevention of Aspergillus ochraceus growth on and Ochratoxin a contamination of sausages using ozonated air. Food Microbiol. 2011, 29, 229–232. [Google Scholar] [CrossRef]
- Qi, L.; Li, Y.; Luo, X.; Wang, R.; Zheng, R.; Wang, L.; Li, Y.; Yang, D.; Fang, W.; Chen, Z. Detoxification of zearalenone and ochratoxin A by ozone and quality evaluation of ozonised corn. Food Addit. Contam. Part A 2016, 33, 1700–1710. [Google Scholar] [CrossRef]
- Torlak, E. Use of gaseous ozone for reduction of ochratoxin A and fungal populations on sultanas. Aust. J. Grape Wine Res. 2018, 25, 25–29. [Google Scholar] [CrossRef]
- Jafarzadeh, S.; Hadidi, M.; Forough, M.; Nafchi, A.M.; Khaneghah, A.M. The control of fungi and mycotoxins by food active packaging: a review. Crit. Rev. Food Sci. Nutr. 2022, 63, 6393–6411. [Google Scholar] [CrossRef] [PubMed]
- Hossain, F.; Follett, P.; Salmieri, S.; Vu, K.D.; Fraschini, C.; Lacroix, M. Antifungal activities of combined treatments of irradiation and essential oils (EOs) encapsulated chitosan nanocomposite films in in vitro and in situ conditions. Int. J. Food Microbiol. 2019, 295, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Chen, B.; Rao, J. Natural compounds of plant origin in the control of fungi and mycotoxins in foods. Curr. Opin. Food Sci. 2023, 52. [Google Scholar] [CrossRef]
- Manzoor, A.; Yousuf, B.; Pandith, J.A.; Ahmad, S. Plant-derived active substances incorporated as antioxidant, antibacterial or antifungal components in coatings/films for food packaging applications. Food Biosci. 2023, 53. [Google Scholar] [CrossRef]
- Jafarzadeh S, Yildiz Z, Yildiz P, Strachowski P, Forough M, Esmaeili Y, Naebe M, Abdollahi M. Advanced macromolecular technologies in biodegradable packaging using intelligent sensing to fight food waste; A review. International Journal of Biological Macromolecules. 2024, 129647.
- Kurtbay, H.M.; Bekçi, Z.; Merdivan, M.; Yurdakoç, K. Reduction of Ochratoxin A Levels in Red Wine by Bentonite, Modified Bentonites, and Chitosan. J. Agric. Food Chem. 2008, 56, 2541–2545. [Google Scholar] [CrossRef]
- Aguilar-Zuniga, K.; Laurie, V.F.; Moore-Carrasco, R.; Ortiz-Villeda, B.; Carrasco-Sánchez, V. Agro-industrial Waste Products as Mycotoxin Biosorbents: A Review of in Vitro and in Vivo Studies. Food Rev. Int. 2021, 39, 2914–2930. [Google Scholar] [CrossRef]
- La Placa, L.; Tsitsigiannis, D.; Leggieri, M.C.; Battilani, P. From Grapes to Wine: Impact of the Vinification Process on Ochratoxin A Contamination. Foods 2023, 12, 260. [Google Scholar] [CrossRef]
- Howard P.C, Churchwell M.I, Couch L.H, Marques M.M, Doerge D.R. Formation of N-(carboxymethyl) fumonisin B1, following the reaction of fumonisin B1 with reducing sugars. Journal of Agricultural and Food Chemistry. 1998, 46(9), 3546-3557.
- Lu Y, Clifford L, Hauck C.C, Hendrich S, Osweiler G, Murphy P.A. Characterization of fumonisin B1− glucose reaction kinetics and products. Journal of Agricultural and Food Chemistry. 2002, 50(16), 4726-4733.
- Fernández-Surumay, G.; Osweiler, G.D.; Yaeger, M.J.; Rottinghaus, G.E.; Hendrich, S.; Buckley, L.K.; Murphy, P.A. Fumonisin B−Glucose Reaction Products Are Less Toxic When Fed to Swine. J. Agric. Food Chem. 2005, 53, 4264–4271. [Google Scholar] [CrossRef]
- Murphy, P.A.; Hendrich, S.; Hopmans, E.C.; Hauck, C.C.; Lu, Z.; Buseman, G.; Munkvold, G. Effect of processing on fumonisin content of corn. Fumonisins in Food, 1996, 323-334.
- Gu, K.; Ryu, D.; Lee, H.J. Ochratoxin A and its reaction products affected by sugars during heat processing. Food Chem. 2021, 348, 129038. [Google Scholar] [CrossRef]
- Nora N.S, Feltrin A.C.P, Sibaja K.V.M, Furlong E.B, Garda-Buffon J. Ochratoxin A reduction by peroxidase in a model system and grape juice. Brazilian Journal of Microbiology, 2019, 50, 1075-1082.
- Alsalabi, F.A.; Hassan, Z.U.; Al-Thani, R.F.; Jaoua, S. Molecular identification and biocontrol of ochratoxigenic fungi and ochratoxin A in animal feed marketed in the state of Qatar. 9, 1283; e5. [Google Scholar] [CrossRef]
- Mwabulili, F.; Xie, Y.; Li, Q.; Sun, S.; Yang, Y.; Ma, W. Research progress of ochratoxin a bio-detoxification. Toxicon 2022, 222, 107005. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.; Castro, T.; Venâncio, A.; Silva, C. Degradation of ochratoxins A and B by lipases: A kinetic study unraveled by molecular modeling. Heliyon 2023, 9, e19921. [Google Scholar] [CrossRef]
- Kupski, L.; Queiroz, M.I.; Badiale-Furlong, E. Application of carboxypeptidase A to a baking process to mitigate contamination of wheat flour by ochratoxin A. Process. Biochem. 2018, 64, 248–254. [Google Scholar] [CrossRef]
- Du, G.; Liu, L.; Guo, Q.; Cui, Y.; Chen, H.; Yuan, Y.; Wang, Z.; Gao, Z.; Sheng, Q.; Yue, T. Microbial community diversity associated with Tibetan kefir grains and its detoxification of Ochratoxin A during fermentation. Food Microbiol. 2021, 99, 103803. [Google Scholar] [CrossRef]
- Shehata, M.G.; Badr, A.N.; El Sohaimy, S.A.; Asker, D.; Awad, T.S. Characterization of antifungal metabolites produced by novel lactic acid bacterium and their potential application as food biopreservatives. Ann. Agric. Sci. 2019, 64, 71–78. [Google Scholar] [CrossRef]
- Touranlou, F.A.; Noori, S.M.A.; Salari, A.; Afshari, A.; Hashemi, M. Application of kefir for reduction of contaminants in the food industry: A systematic review. Int. Dairy J. 2023, 146. [Google Scholar] [CrossRef]
- Hassan, Z.U.; Al Thani, R.; Atia, F.A.; Alsafran, M.; Migheli, Q.; Jaoua, S. Application of yeasts and yeast derivatives for the biological control of toxigenic fungi and their toxic metabolites. Environ. Technol. Innov. 2021, 22. [Google Scholar] [CrossRef]
- Cecchini, F.; Morassut, M.; Saiz, J.-C.; Garcia-Moruno, E. Anthocyanins enhance yeast’s adsorption of Ochratoxin A during the alcoholic fermentation. Eur. Food Res. Technol. 2018, 245, 309–314. [Google Scholar] [CrossRef]
- Söylemez T, Yamaç M. Screening of macrofungi isolates for aflatoxin B1 and ochratoxin A degradation. Biology Bulletin. 2021, 48, 122–129.
- Yang, Y.; Zhong, W.; Wang, Y.; Yue, Z.; Zhang, C.; Sun, M.; Wang, Z.; Xue, X.; Gao, Q.; Wang, D.; et al. Isolation, identification, degradation mechanism and exploration of active enzymes in the ochratoxin A degrading strain Acinetobacter pittii AP19. J. Hazard. Mater. 2023, 465, 133351. [Google Scholar] [CrossRef] [PubMed]
- Gonaus C, Wieland L, Thallinger G. G, Prasad S. Ochratoxin A degrading enzymes of Stenotrophomonas sp. 043-1a. FEMS Microbiology Letters. 2023, 370, fnad028.
- Domínguez-Gutiérrez, G.; Perraud-Gaime, I.; Escalona-Buendía, H.; Durand, N.; Champion-Martínez, E.; Fernández-Soto, R.; Saucedo-Castañeda, G.; Rodríguez-Serrano, G. Inhibition of Aspergillus carbonarius growth and Ochratoxin A production using lactic acid bacteria cultivated in an optimized medium. Int. J. Food Microbiol. 2023, 404, 110320. [Google Scholar] [CrossRef] [PubMed]
- Mateo, E.M.; Tarazona, A.; Jiménez, M.; Mateo, F. Lactic Acid Bacteria as Potential Agents for Biocontrol of Aflatoxigenic and Ochratoxigenic Fungi. Toxins 2022, 14, 807. [Google Scholar] [CrossRef] [PubMed]
- Lević S, Đorđević V, Kalušević A, Đorđević R, Bugarski B, Nedović V. 18 Immobilized Yeast in Winemaking. Winemaking: Basics and Applied Aspects. 2021, 468.
- Yang, Q.; Dhanasekaran, S.; Ngea, G.L.N.; Tian, S.; Li, B.; Zhang, H. Unveiling ochratoxin a controlling and biodetoxification molecular mechanisms: Opportunities to secure foodstuffs from OTA contamination. Food Chem. Toxicol. 2022, 169, 113437. [Google Scholar] [CrossRef]
- Kapetanakou, A.E.; Passiou, K.E.; Chalkou, K.; Skandamis, P.N. Assessment of Spoilage Potential Posed by Alicyclobacillus spp. in Plant-Based Dairy Beverages Mixed with Fruit Juices during Storage. J. Food Prot. 2021, 84, 497–508. [Google Scholar] [CrossRef]
- Dini, I.; Alborino, V.; Lanzuise, S.; Lombardi, N.; Marra, R.; Balestrieri, A.; Ritieni, A.; Woo, S.L.; Vinale, F. Trichoderma Enzymes for Degradation of Aflatoxin B1 and Ochratoxin A. Molecules 2022, 27, 3959. [Google Scholar] [CrossRef]
- Petrova, P.; Arsov, A.; Tsvetanova, F.; Parvanova-Mancheva, T.; Vasileva, E.; Tsigoriyna, L.; Petrov, K. The Complex Role of Lactic Acid Bacteria in Food Detoxification. Nutrients 2022, 14, 2038. [Google Scholar] [CrossRef]
- Lyu, Z.; Ding, S.; Du, D.; Qiu, K.; Liu, J.; Hayashi, K.; Zhang, X.; Lin, Y. Recent advances in biomedical applications of 2D nanomaterials with peroxidase-like properties. Adv. Drug Deliv. Rev. 2022, 185, 114269. [Google Scholar] [CrossRef]
- Ding, L.; Han, M.; Wang, X.; Guo, Y. Ochratoxin A: Overview of Prevention, Removal, and Detoxification Methods. Toxins 2023, 15, 565. [Google Scholar] [CrossRef]
| Food/Beverage | Country | Concentration | References |
|---|---|---|---|
| Cereals | Sweden | 0.12 μg/kg | [39] |
| - | 0.03–27.5 ppm | [39] | |
| Bread | Portugal | Below legal limit | - |
| Beans/peas | Sweden | 442 μg/kg | [41] |
| Peanuts | Argentina | 170 μg/kg | |
| Olives | Greece | 1.86 μg/kg | |
| Cocoa mass | 2.79–2.41 μg/kg | [45] | |
| Cocoa powder | 0.63 μg/kg | ||
| Coffee (grain) | Brazil | Up to 3.3 μg/kg | [42] |
| Coffee (instant) | 6.29 μg/kg | [42] | |
| Coffee drink | Switzerland | 4.2 μg/L | [43] |
| Coffee | Ivory Coast | 56 μg/kg | [43] |
| Roasted coffee | Ethiopia | 2.0 μg/kg | [44] |
| Grapes | Portugal | 115.6 μg/kg | [45] |
| Grapes | Dão region and Madeira Island | 9.5–115.6 µg/kg | [45] |
| Grape juice | 0.337 µg/kg | [47,48] | |
| Red wine | Portugal | 1-1.23 μg/L | [46] |
| White wine | - | 1-2.4 μg/L | |
| Porto | - | 0.05–0.08 μg/L | |
| Red wine | - | 0.2–0.4 μg/L | [45] |
| Beer | - | 0.033 μg/L | |
| Weekly intake | - | 15–60 ng/kg | |
| Serum | Coimbra, Portugal | >0.05 ng/mL | [49] |
| Daily intake of children | Switzerland | 3.6 ng/kg bw | [50] |
| Breast Milk | Egypt | 1.89 ng/L | [51] |
| Breast milk | Italy | 1.2–6.6 ng/L | [33] |
| Breast milk | - | 6.01 ng/L ( | [52] |
| Breast milk | Norway | 10–182 ng/L | [26] |
| Country | Food product | Maximum level of OTA (ng/g) |
|---|---|---|
| EU | Cereal grains | 5.0 |
| Cereal-based products | 3.0 | |
| Raisins, currents, and sultanas | 10.0 | |
| Roasted and ground coffee beans | 5.0 | |
| Instant coffee | 10.0 | |
| Beverages (wine and beer) | 2.0 | |
| Non-concentrated grape juices | 2.0 | |
| Infant cereals | 0.5 | |
| China | Cereal grains and beans | 5.0 |
| Singapore | Cereals, coffee, wine, and beer | 3.0 |
| Brazil | Cereal grains, excluding corn | 10.0 |
| Roasted coffee | 10.0 | |
| Wine | 10.0 | |
| Cereal-based baby food | 2.0 | |
| Dried fruits | 10.0 | |
| India | Cereal and cereal-based food products | 20.0 |
| Russia | Cereal and cereal-based food products | 5.0 |
| Cereal-based products for young children | 0.5 | |
| Korea | Cereal and cereal-based food products | 5.0 |
| Roasted coffee beans and instant coffee | 10.0 | |
| Concentrated grape juices and wine | 2.0 | |
| Dried grapes (raisins) | 10.0 | |
| Cereal-based food for infants | 0.5 | |
| Malaysia | Cereal-based food for infants and young kids | 0.5 |
| Roasted coffee beans and ground coffee | 5.0 | |
| Instant coffee | 10.0 | |
| Indonesia | Cereal-based food products | 5.0 |
| Roasted coffee beans and ground coffee | 5.0 | |
| Instant coffee | 10.0 |
| Technique | Sample | Condition | Reduction (%) | Reference | |
|---|---|---|---|---|---|
| Gamma radiation (37.5%) | Corn | 10 kGy | 40.3 | [18] | |
| 20 kGy | 61.1 | ||||
| 2 kGy | 48 | ||||
| Cornflour | [82] | ||||
| 4 kGy | 62 | ||||
| Wheat flour | 24 | ||||
| Grape juice | 30.5 kGy | 12 | [83] | ||
| Wine | 23 | ||||
| Electron-beam radiation (12.5%) | 10 kGy | 16 | [19] | ||
| Red pepper powder | |||||
| 30 kGy | 25 | ||||
| UV radiation (37.5%) | 60 min | 0 | [84] | ||
| Brown Rice | |||||
| 180 min | 0 | ||||
| Black Rice | 60 min | 54.5 | |||
| 180 min | 92.5 | ||||
| Red Rice | 60 min | 90.2 | |||
| 180 min | 100 | ||||
| Corn grain | 17 | ||||
| 7 min | [85] | ||||
| Wheat grain | |||||
| 6 | |||||
| Wheat Semolina | 15 min | 100 | [86] | ||
| Cold plasma (12.5%) | Coffee bean | 30 min | 50 | [87] |
| Thermal processing | Matrix | Reduction (%) | Reference |
|---|---|---|---|
| Roasting | Coffee | 0–12% 17–86% 67% 86–94% |
[139] [140] [141] [142] |
| Oat | 2–18% | [143] | |
| White/brown rice | 15–60% | [144] | |
| Cocoa beans | 24–41% | [145] | |
| Explosive Puffing | Oat | 38–52% | [146] |
| Rice | 15–28% | ||
| Baking | Wheat (bread) | 0% | [147] |
| Wheat (biscuits) | 67% | [105] | |
| Pressure cooking or autoclaving | Bean | 84% | [148] |
| Oatmeal, rice | 59–89% | [149] | |
| Retorting | Oat | 17% | [150] |
| Rice | 54% | ||
| Direct steam injection | Oat | 20–28% | [146] |
| Indirect steaming | Oat | 13% | [151] |
| Rice | 59% | ||
| Brewing | Beer (barley) | 14–28% | [117] |
| Coffee | 0–97% 15–50% |
[152] [141] |
| Technique | Sample | Condition | Reduction (%) | Reference |
|---|---|---|---|---|
| Adsorbent materials (50%) |
Aqueous solution | Beta-cylodextrin, pH 3.0 | 80 | [165] |
| Alginate with activated charcoal | > 90 | [22] | ||
| Beer | ||||
| Plant extract (50%) | Alginate with pectin | > 90 | ||
| P. alata | 0 | |||
| P. cattleianum | 0 | |||
| In vitro | [166] | |||
| R. officinalis | 0 | |||
| O. vulgare | 0 | |||
| C. zeylanicum, 1500 μg | 100 | |||
| O. basilicum: 3500 μg/g | 100 | |||
| Corn grain | Z. officinale: 3500 μg/g | 100 | [84] | |
| C. longa, 3500 μg | 100 | |||
| C. martini: 2500 μg | 100 |
| Processing method | Matrix | OTA levels | Additive conc. (%) | OTA reduction without additive | OTA reduction with additive | References |
|---|---|---|---|---|---|---|
| Baking soda | ||||||
| Frying | Wheat flour | 93.2 and 248 ng/g | 0.4 | 46.3 ng/g | 38.8 ng/g | [113] |
| Direct steam injection | Oat flour | OTA standard spike, 100 ng/g | 0.5 | 146.5 ng/g | 129.0% | [146] |
| Twin-screw extrusion | Rice flour | OTA standard spike, 100 ng/g | 1 | 19.8-27.9% | 36.1-44.3% | |
| Retorting | Oat flakes | OTA standard spike, 20 ng/g | 0.5 | 77.9–82.2% | 43.4-51.4% | |
| Indirect steaming | Rice flour | OTA standard spike, 20 ng/g | 1 | 39.5–42.7% | 74.9–78.2% | [161] |
| Oat flour | 0.5 | 53.8% | 71.5-77.4% | |||
| Rice flour | 1 | 17.2% | 56.2–57.7% | |||
| 0.5 | 59.4% | 64.6-65.4% | [150] | |||
| 1 | 13.6% | 55.5% | ||||
| 0.5 | 66.4% | |||||
| 1 | 30.3% | |||||
| 0.5 | 40.8% | [151] | ||||
| 1 | 78.1% | |||||
| 0.5 | 68.7% | |||||
| 1 | 57.6% | |||||
| Sugar (glucose, fructose) | ||||||
| Roasting | Oat grain | OTA standard spike, 100 ng/g | 1 | 10.00% | 15.20% | [150] |
| Retorting | Rice flour | OTA standard spike, 20 ng/g | 1 | 53.80% | 38.70% | |
| Indirect steaming | Oat flour | OTA standard spike, 20 ng/g | 0.5 | 17.20% | 18.20% | [150] |
| Rice flour | OTA standard spike, 20 ng/g | 5 | 59.40% | 40.80% | ||
| Oat flour | OTA standard spike, 20 ng/g | 0.5 | 13.60% | 35.50% | [151] | |
| Combined (baking soda and fructose) | ||||||
| Roasting | Rice flour | OTA standard spike, 20 ng/g | 0.5+0.5 | 53.80% | 35.80% | [150] |
| Indirect steaming | Oat flour | OTA standard spike, 20 ng/g | 0.5+0.5 | 17.20% | 78.60% | [151] |
| Technique (prevalence) | Microorganism/ Enzyme | Sample | Condition | Reduction (%) | Reference |
|---|---|---|---|---|---|
| Enzymes (20%) | Carboxypeptidase A | Wheat flour | 30 to 60 min | 16.8 to 78.5 | [194] |
| Peroxidase | Grape juice | After 24 h | 17 | [190] | |
|
Bacteria (50%) |
L. kefiranofaciens | In vitro | After 24 h | 90.94 | [195] |
| Lactobacillus. sp. RM1 | In vitro | 15 mg. mL | 100 | [196] | |
| L. kefiri FR7 | Peanut | - | 25 | [197] | |
| L. kefiri, K. servazzii, and A. syzygii |
Milk |
pH 4.8 |
94 |
[197] |
|
| B. subtilus | Red wine | Encapsulated in sodium alginate | 78.5 | [23] | |
|
Yeasts (20%) |
S. cerevisiae | Aqueous solution | 15 min. with pH 7.0 | 92 | [198] |
| Red wine | - | 51 | |||
| S. cerevisiae | [199] | ||||
| White wine | - | 27 | |||
| Macrofungi (10%) | A. campestres | In vitro | - | 56 | [200] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




