Submitted:
23 December 2024
Posted:
24 December 2024
You are already at the latest version
Abstract
The present work addresses a method, based on multilamination, for the fabrication of microfluidic devices or analytical microsystems using only commercially available 3D printers and photocurable resins. The created devices were used for the colorimetric measurement of copper ions in aqueous solutions, yielding results comparable to traditional cyclic olefin copolymer (COC) systems but with a significant cost reduction. The microfluidic platforms showed stability and functionality over a twelve-week testing period. This study highlights the potential of 3D printing as a flexible, efficient, and cost-effective alternative for the fabrication of customized microfluidic devices, promoting its use in research with limited resources.
Keywords:
1. Introduction
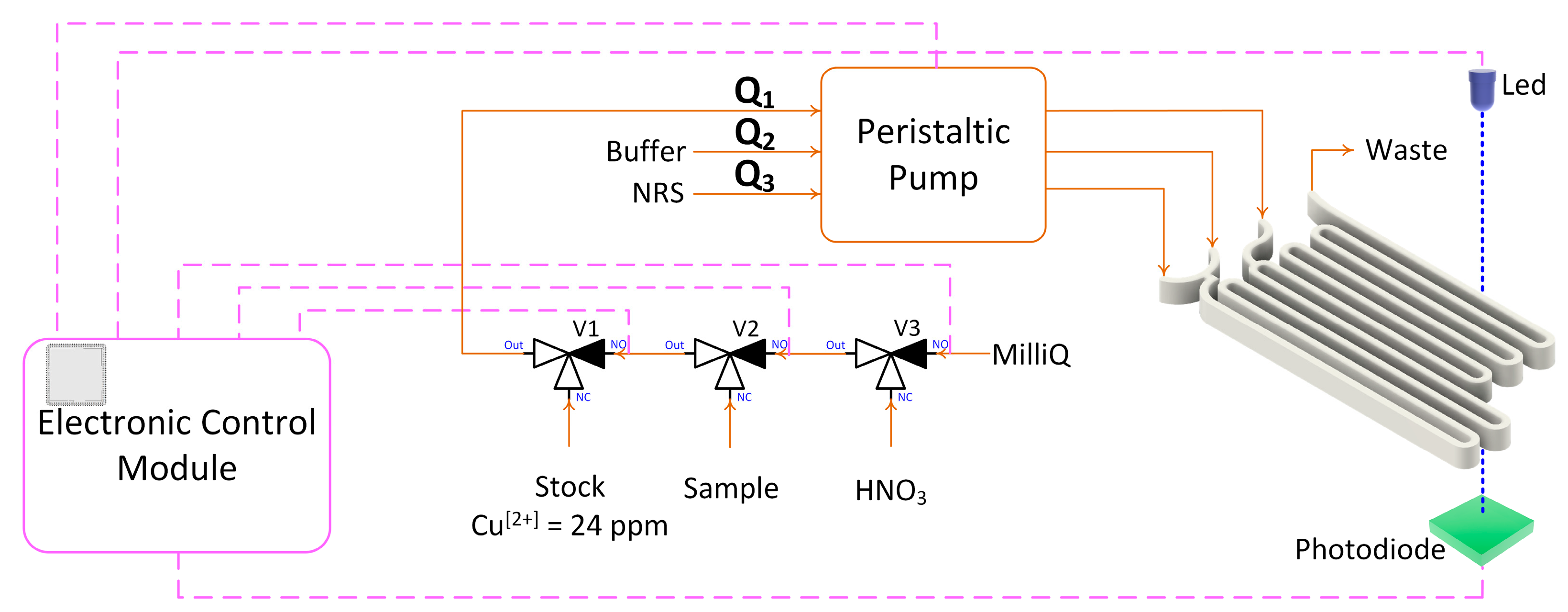
2. Materials and Methods
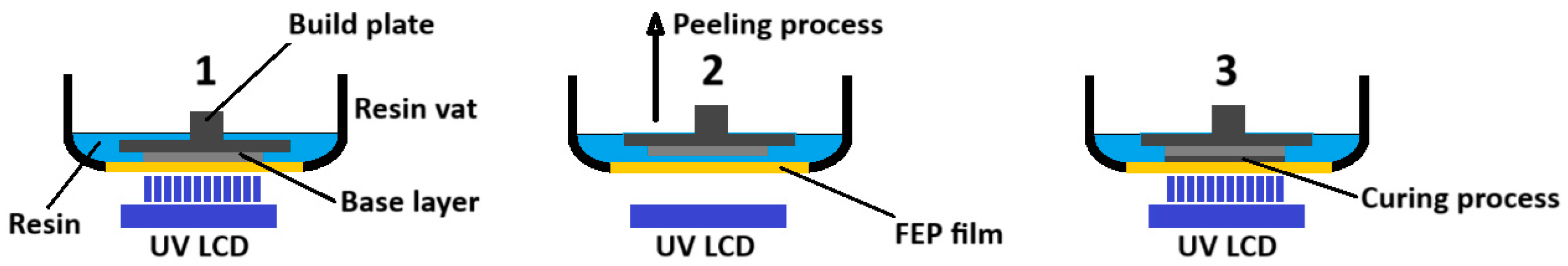
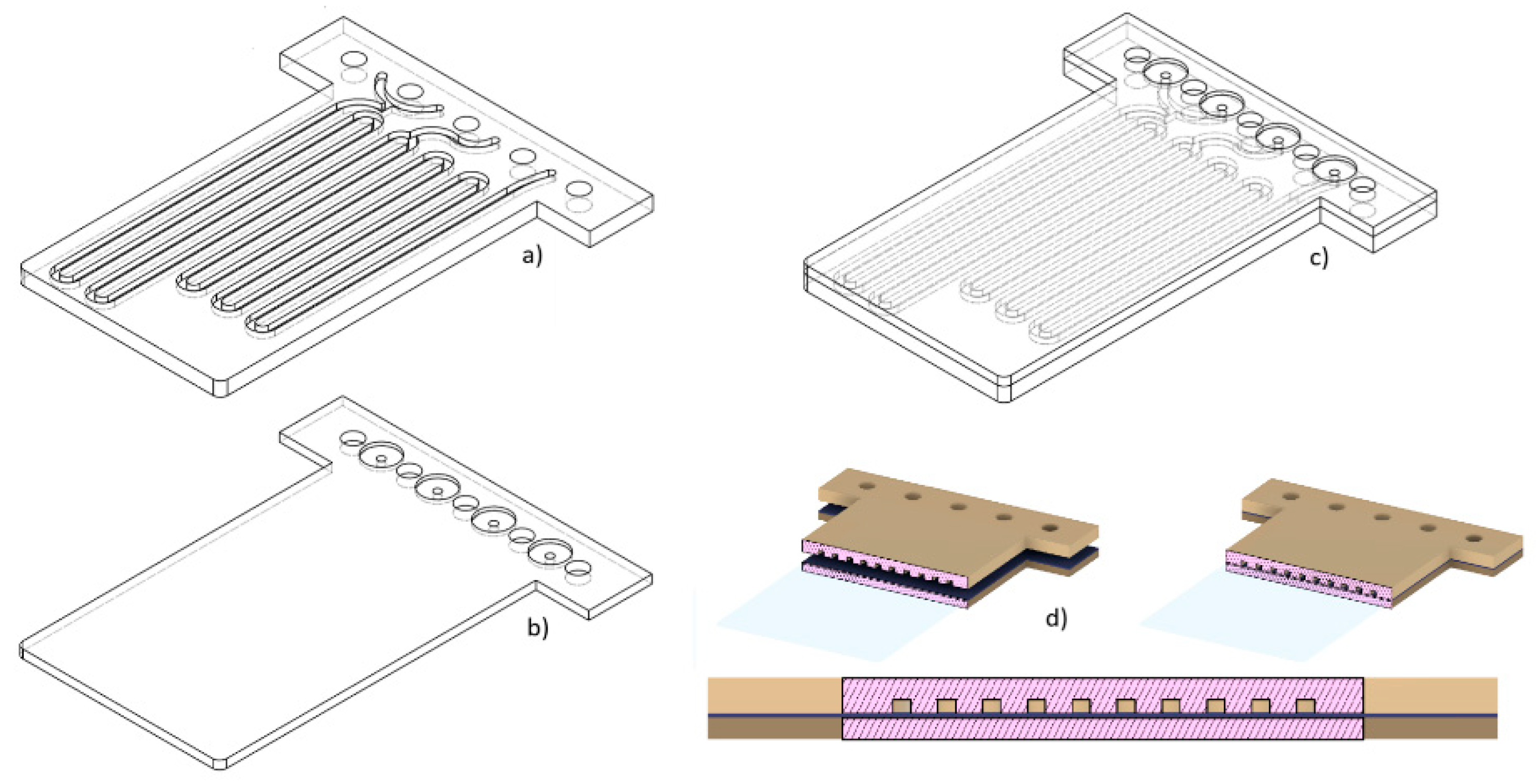
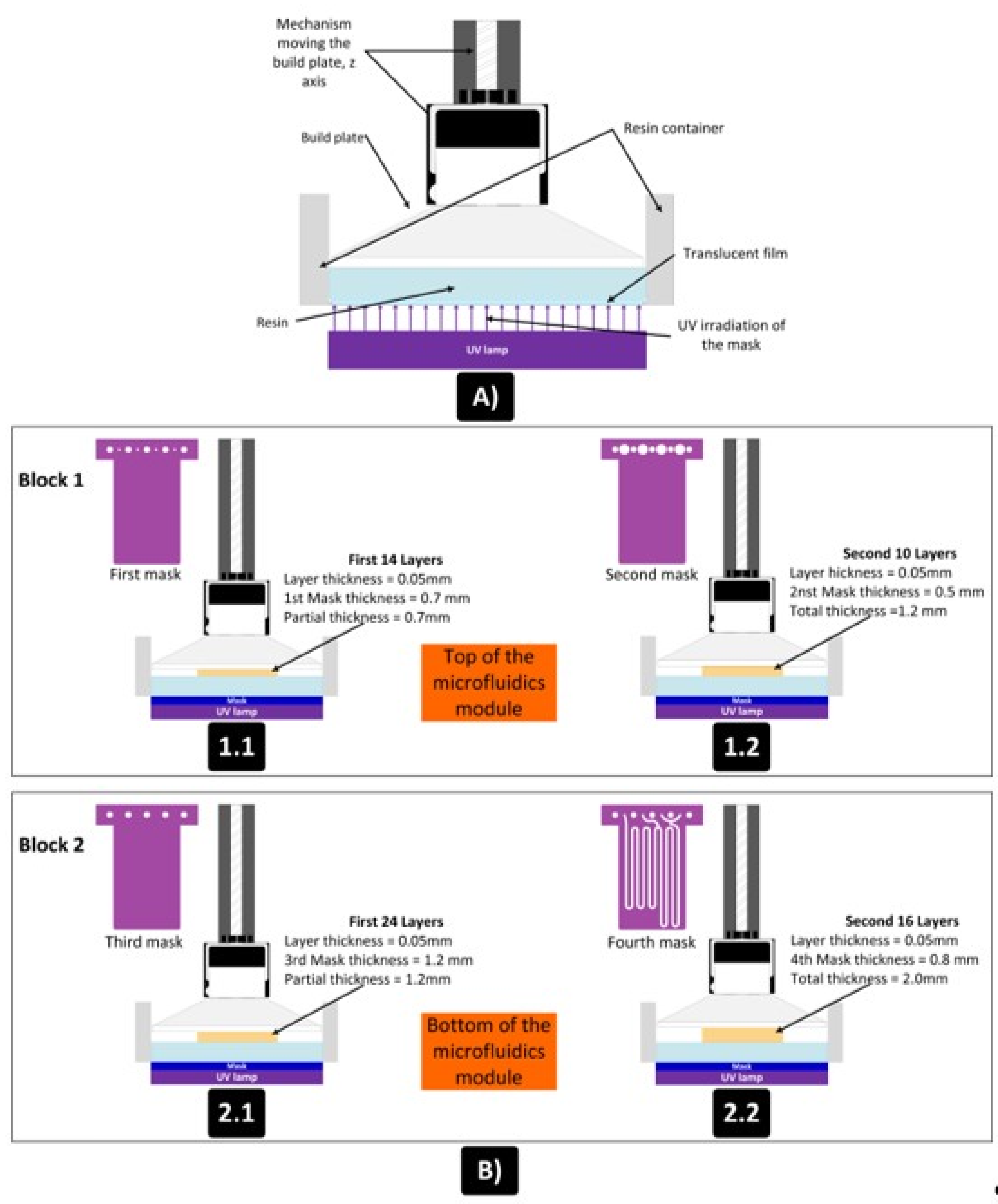
Microfluidic platform fabrication method
- The full 3D design was made using computer aided design (CAD) software.
- The two blocks (halves) forming the device were defined. In one of them there are the open microchannels that were printed. The open channels facilitate the removal of the uncured resin. The other block will be used to seal the microchannnels
- The individual blocks can have a different thickness since they can have a different number of layers.
- Both blocks are joined by multilamination using a photocurable resin.
- The spatula is loaded by dipping it no more than 1 mm perpendicular to the surface of the resin.
- The spatula is withdrawn and left to drain the excess resin.
- The block with the largest surface area is chosen. This is generally the one that acts as a lid closing the microchannels on the other block.
- The spatula is dragged at approximately 45 degrees respective to the surface where the resin is applied, taking care to spread it uniformly.
- Both BOCKS are aligned and joined without pressure to prevent the uncured resin from going into the microchannels.
- The microfluidic device is irradiated on both faces to attain a uniform and hermetic sealing.
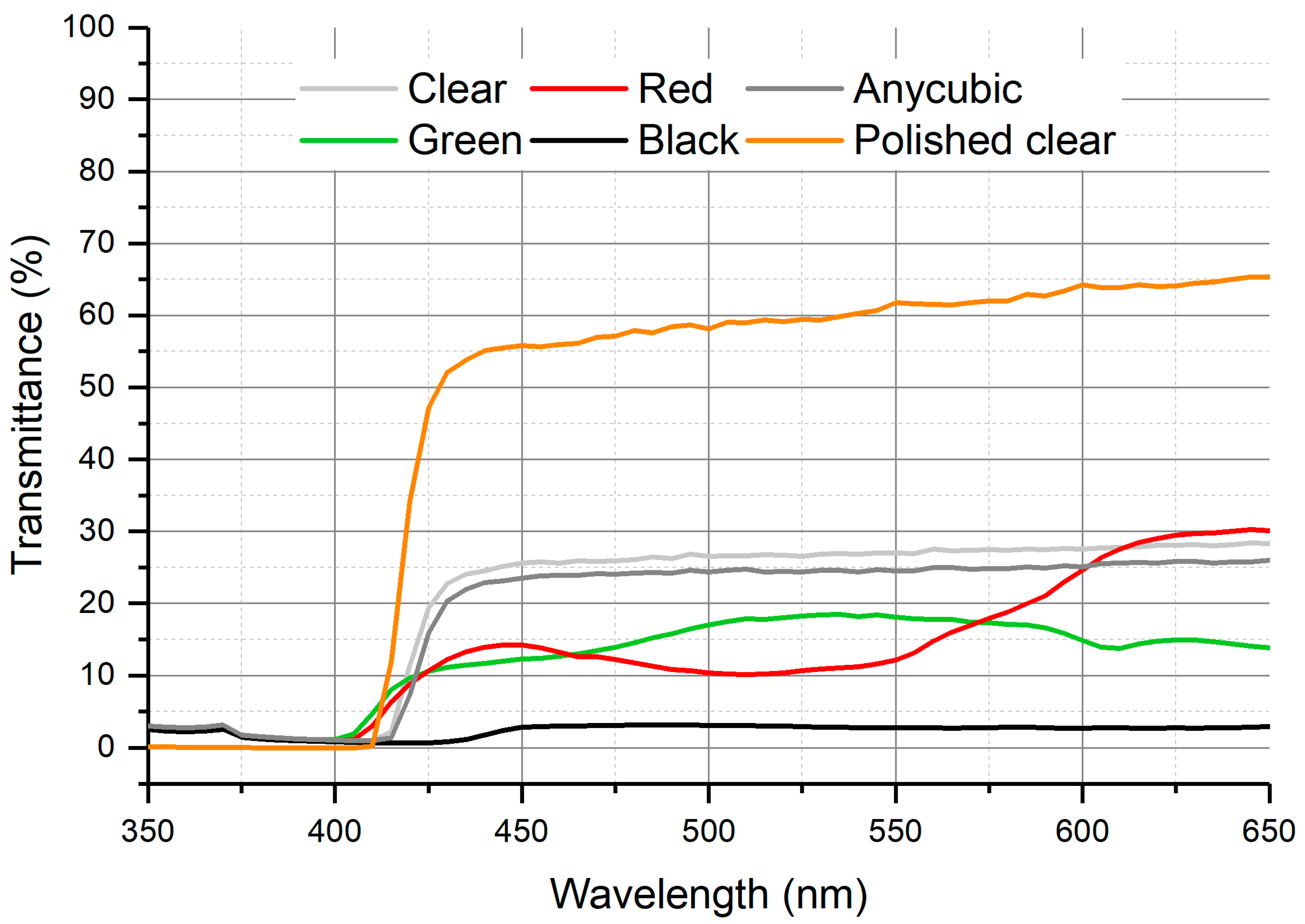
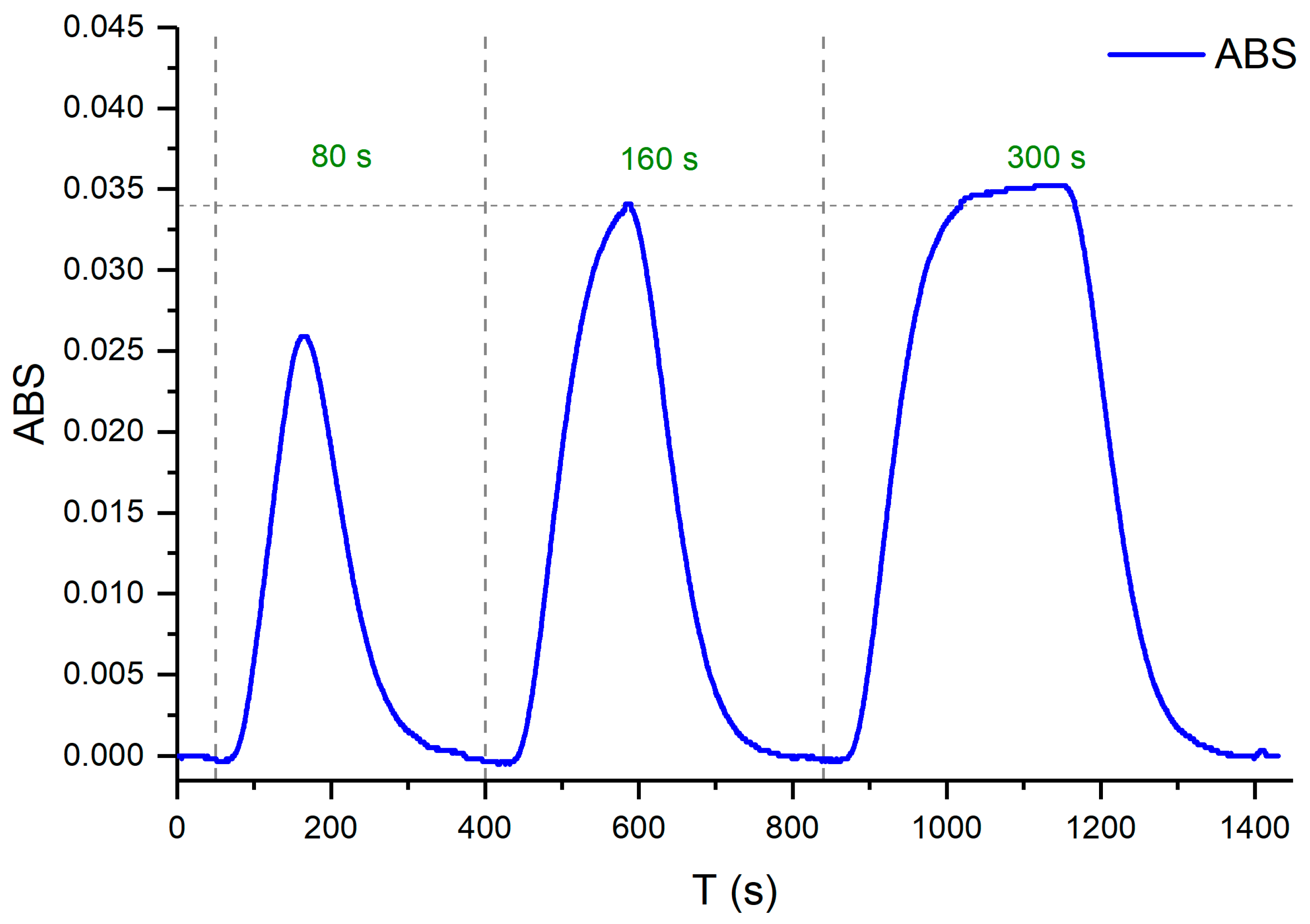
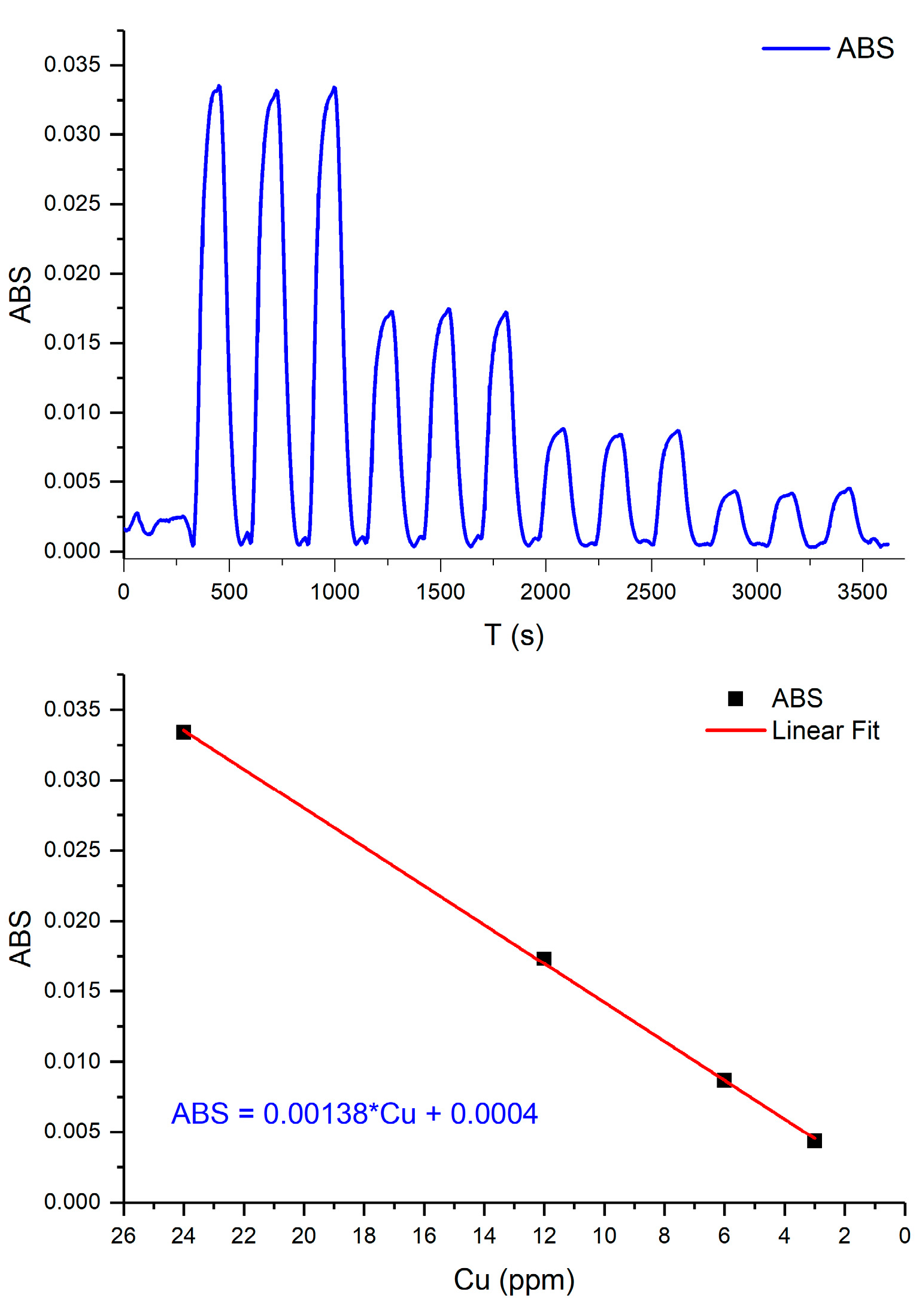
3. Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanders, G.; Manz, A. Chip-based microsystems for genomic and proteomic analysis. TrAC 2000, 19, 364–378. [Google Scholar] [CrossRef]
- Ibáñez-García, N.; Baeza, M.; Puyol, M.; Gómez, R.; Batlle, M.; Alonso-Chamarro, J. Biparametric Potentiometric Analytical Microsystem Based on the Green Tape Technology. Electroanalysis 2010, 22, 2376–2382. [Google Scholar] [CrossRef]
- Amatón, K.V.G.; et al. Microanalyser Prototype for On-Line Monitoring of Copper (II) Ion in Mining Industrial Processes. Sensors 2019, 19, 3382. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.; Ali, Z. Fabrication Methods for Microfluidic Devices: An Overview. Micromachines 2021, 12, 319. [Google Scholar] [CrossRef] [PubMed]
- Juang, Y.-J.; Chiu, Y.-J. Fabrication of Polymer Microfluidics: An Overview. Polymers 2022, 14, 2028. [Google Scholar] [CrossRef] [PubMed]
- Couceiro, P.; Alonso-Chamarro, J. Microfabrication of monolithic microfluidic platform using low temperature co-fire ceramics suitable fluorescence imaging. Anal. Chem. 2017, 89, 9147–9153. [Google Scholar] [CrossRef] [PubMed]
- Berenguel-Alonso, M.; et al. Rapid prototyping of a cyclic olefin copolymer microfluidic device for automated oocyte culturing. SLAS Technol. 2017, 22, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Lopez, *!!! REPLACE !!!*; Ymbern, O.; Puyol, M.; Casalta, J.M.; Alonso-Chamarro, J. Potentiometric analytical microsystem based on the integration of gas-difussion step for on-line ammonium determination in water recycling processes in manned space missions. Anal. Chim. Acta 2015, 87, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Prada, J.; Cordes, C.; Harms, C.; Lang, W. Design and manufacturing of a disposable, cyclo-olefin copolymer, microfluidic device for biosensor. Sensors 2019, 19, 1178. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, N.; Urrios, A.; Kang, S.; Folch, A. The upcoming 3D-printing revolution in microfluidics. Lab. Chip 2016, 16, 1720–1742. [Google Scholar] [CrossRef] [PubMed]
- Au, K.; Huynh, W.; Horowitz, L.F.; Folch, A. 3D-printed microfluidics. Angew. Chem. Int. Ed. 2016, 55, 3862–3881. [Google Scholar] [CrossRef] [PubMed]
- Gross, C.; Erkal, J.L.; Lockwood, S.Y.; Chen, C.; Spence, D.M. Evaluation of 3D printing and its potential impact on biotechnology and the chemical sciences. 2014.
- Taczała, J.; Czepułkowska, W.; Konieczny, B.; Sokołowski, J.; Kozakiewicz, M.; Szymor, P. Comparison of 3D printing MJP and FDM technology in dentistry. Arch. Mater. Sci. Eng. 2020, 101, 32–40. [Google Scholar] [CrossRef]
- Pagac, M.; et al. A review of vat photopolymerization technology: Materials, applications, challenges, and future trends of 3d printing. Polymers 2021, 13, 598. [Google Scholar] [CrossRef] [PubMed]
- Versatile Lock and Key Assembly for Optical Measurements with Microfluidic Platforms and Cartridges. Oriol Ymbern, Miguel Berenguel-Alonso, Antonio Calvo-López, Sara Gómez-de Pedro, David Izquierdo and Julián Alonso-Chamarro. Anal. Chem. 2015, 87, 1503−1508. [CrossRef]
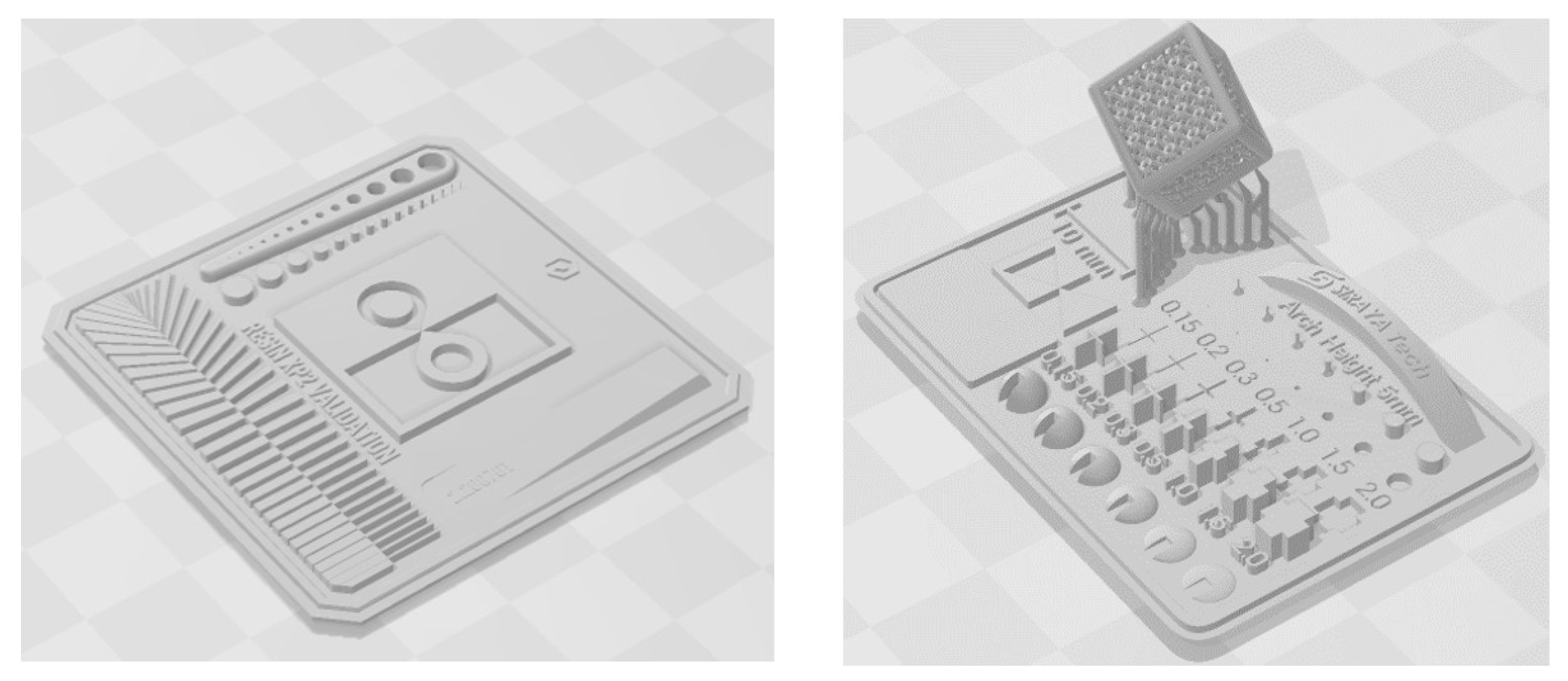
- Tech, S. Siraya Tech Test Model. 2023.
- Electronics, C. Optimal Layer Exposure Time for Perfect Resin Prints - Tutorial Australia. 2023.
- Anycubic. Anycubic Photon Mono X - Google Drive. 2023.





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



