Submitted:
20 December 2024
Posted:
20 December 2024
You are already at the latest version
Abstract
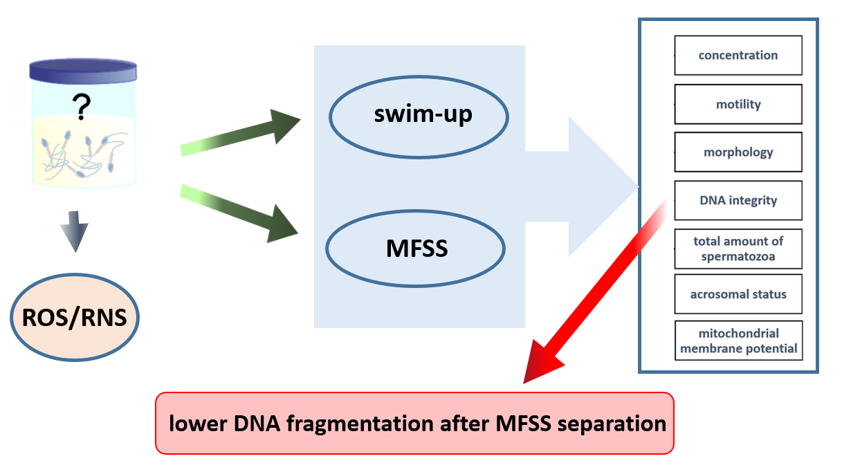
Microfluidic sperm selection systems (MFSS) are increasingly being used and appear to be a promising adjunct in assisted reproductive treatment (ART). They are practical, easy to use, and often presented as highly effective. In this study, we assessed the effectiveness of Ca0 microfluidic chip compared to the swim-up (SU) technique with respect to oxygen radicals and spermiogram parameters. Sample from each patients was separated by both methods and after that sperm was evaluated for sperm concentration, motility, morphology, DNA integrity, acrosomal status and mitochondrial membrane potential. Two subpopulations were selected on basic of spermiogram evaluation: normozoospermic (n=25) and non-normozoospermic (n=20). During analyses when we compare male characteristics between technics of separation, no significant differences were observed except for a lower DNA fragmented sperm in the CA0 group compared to SU (SU: 9,94 vs. CA0: 4,55, p = 0.016) in the non-normozoospermic cohort but not in normozoospermic cohort. The Ca0 method is simple, frequently used in laboratories, and gives good results but does not provide much benefit over the swim-up. In patients who do not have a normal spermiogram and have other problems (e.g. a higher proportion of sperm with fragmented DNA), it may improve the effectiveness of infertility treatment.
Keywords:
1. Introduction
2. Results
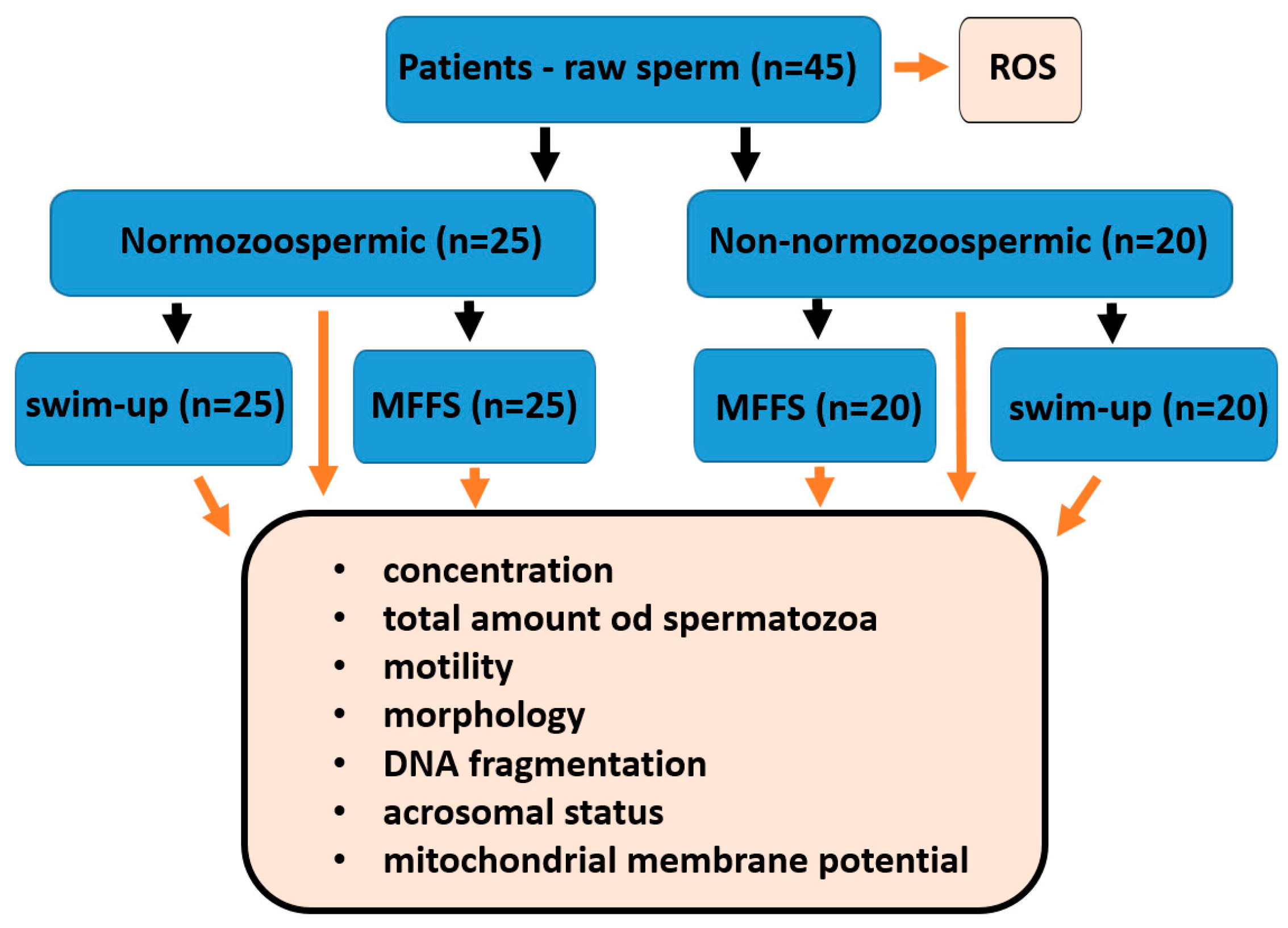
2.1. Patients
2.2. Semen Analyses
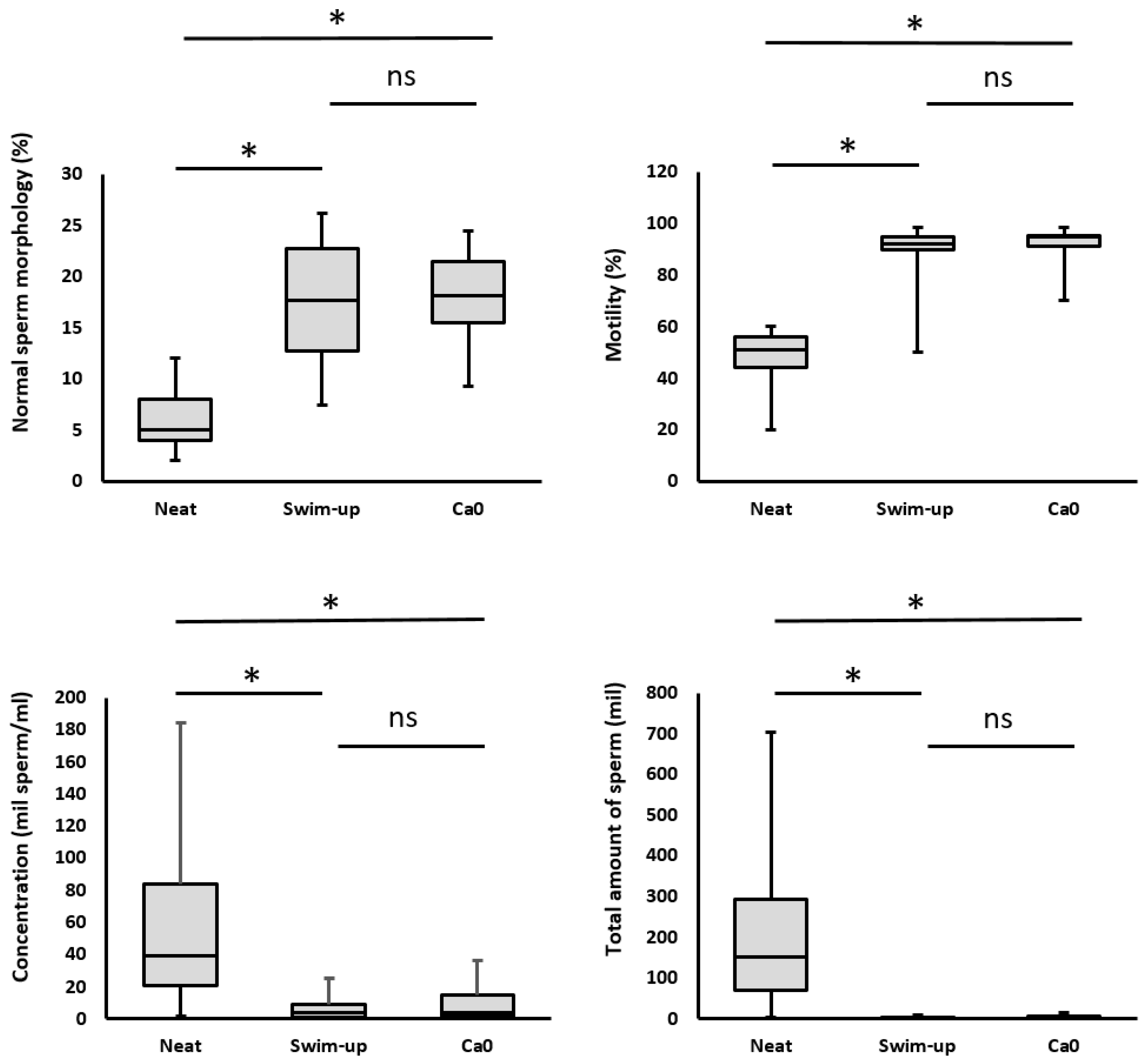
2.2.1. Sperm Concentration
2.2.2. Total Amount of Spermatozoa
2.2.3. Sperm Motility
2.2.4. Morphology of Sperm
2.2.5. DNA Integrity
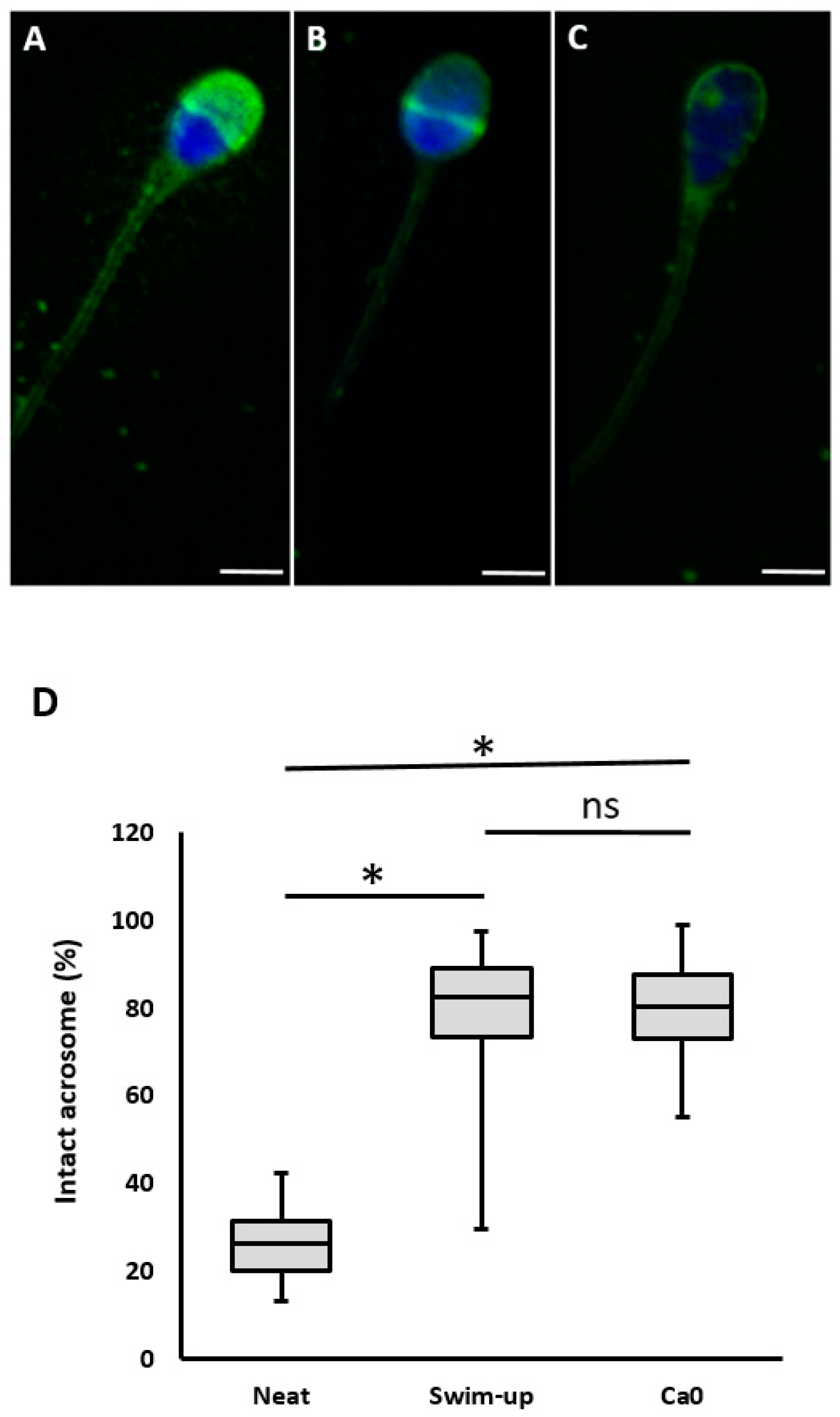
2.2.6. Acrosomal Status
2.2.7. Mitochondrial Activity
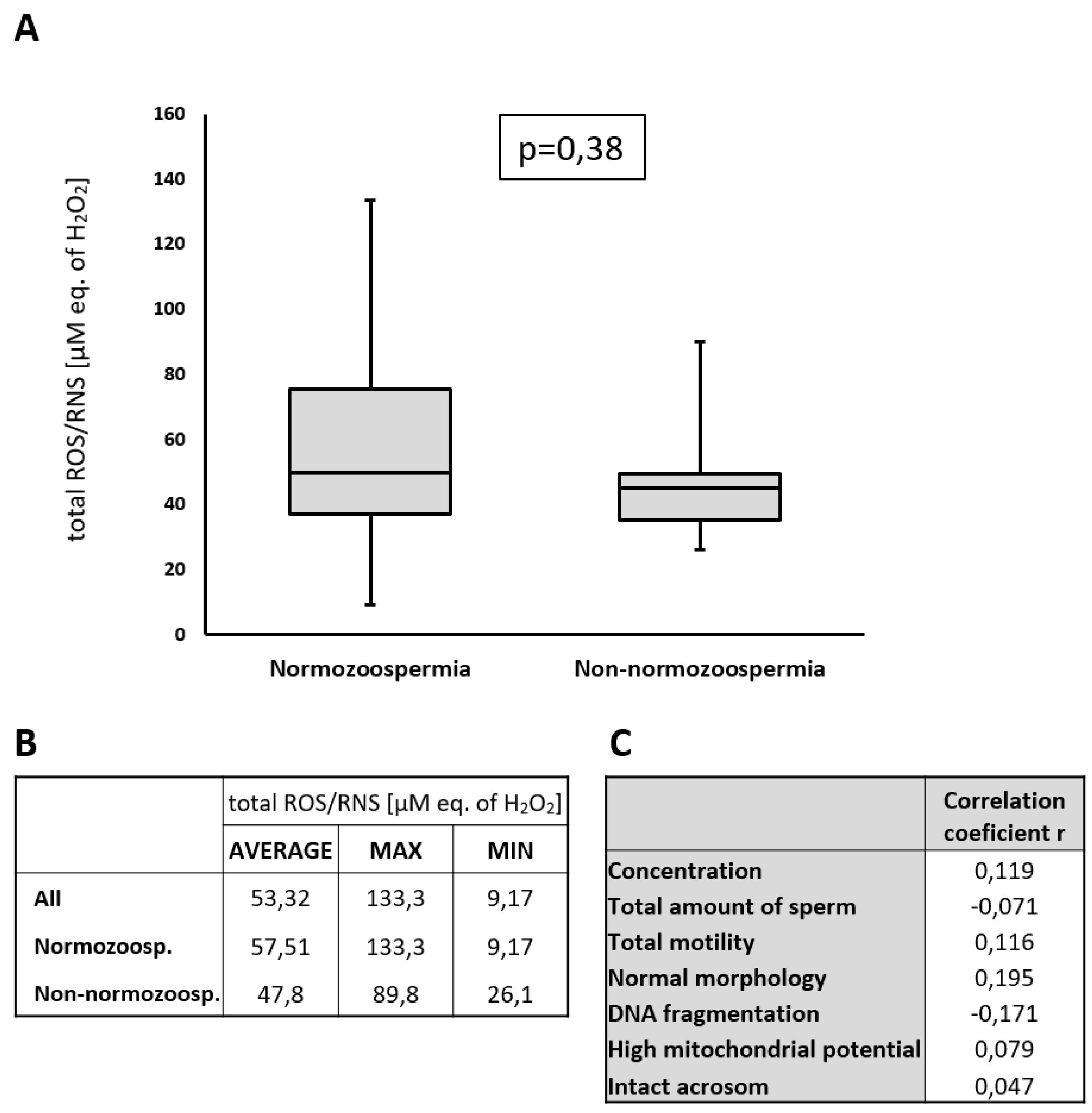
2.3. Detection of ROS/RNS in Seminal Plasma
2.4. Normozoospermic and Non-Normozoospermic Patients
3. Discussion
4. Materials and Methods
4.1. Patients
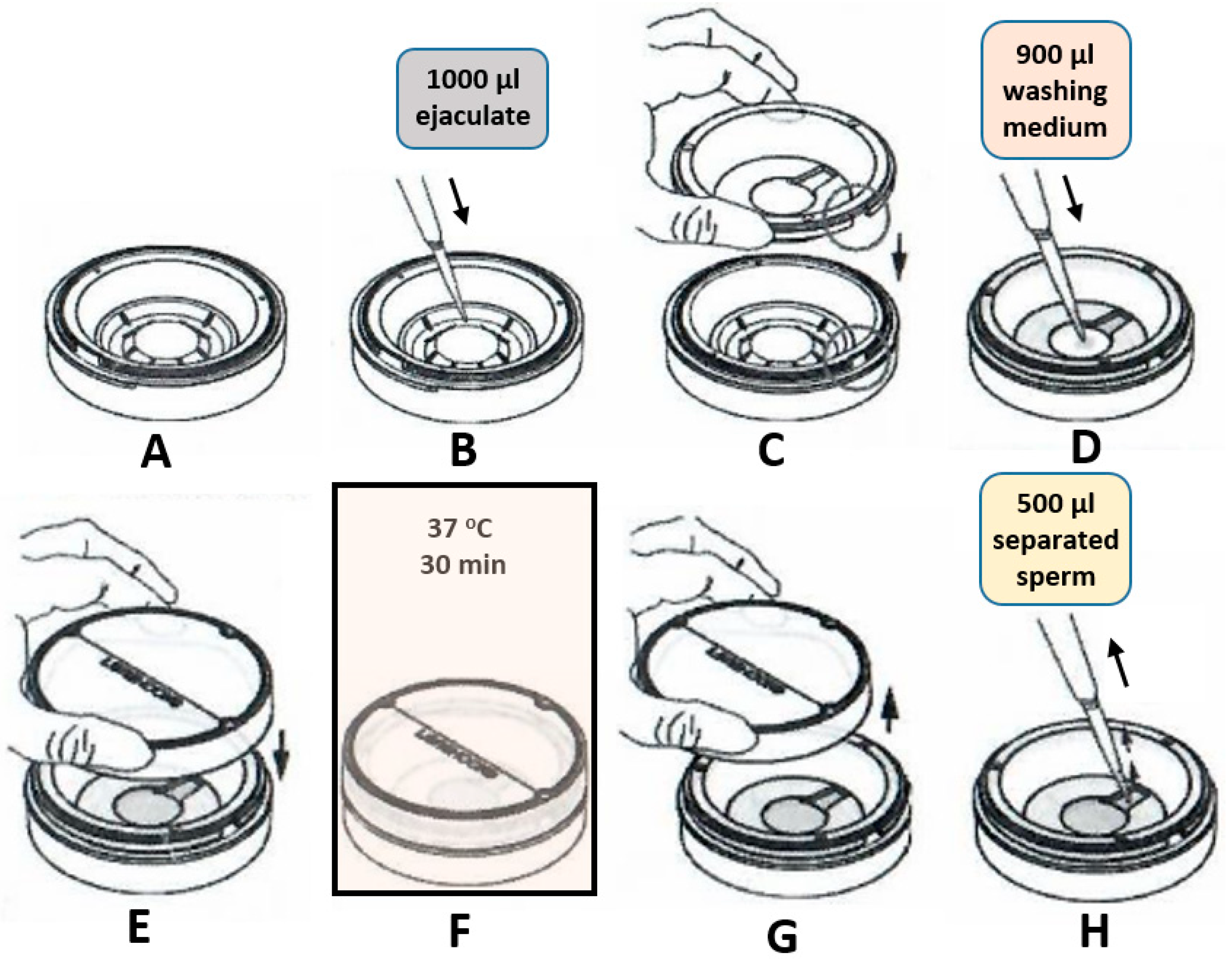
4.2. Sperm Selection
4.2.1. Microfluidic Sperm Selection
4.2.2. Swim-Up Method
4.3. Semen Analyses
4.4. DNA Integrity Assessment
4.5. Evaluation of Acrosomal Status
4.6. Mitochondrial Evaluation
4.7. ROS/RNS Assessment
4.8. Statistical Evaluation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vaughan, D. A., Leung, A., Resetkova, N., Ruthazer, R., Penzias, A. S., Sakkas, D., and Alper, M. M. How many oocytes are optimal to achieve multiple live births with one stimulation cycle? The one-and-done approach. Fertil Steril 2017, 107 (2):397-404.e3. [CrossRef]
- Giojalas, L. C., and Guidobaldi, H. A. Getting to and away from the egg, an interplay between several sperm transport mechanisms and a complex oviduct physiology. Mol Cell Endocrinol 2020, 518:110954. [CrossRef]
- Williams, M., Thompson, L. A., Li, T. C., Mackenna, A., Barratt, C. L., and Cooke, I. D. Uterine flushing: a method to recover spermatozoa and leukocytes. Hum Reprod 1993, 8 (6):925-8. [CrossRef]
- Mortimer, D., Leslie, E. E., Kelly, R. W., and Templeton, A. A. Morphological selection of human spermatozoa in vivo and in vitro. J Reprod Fertil 1982, 64 (2):391-9. [CrossRef]
- Pérez-Cerezales, S., Ramos-Ibeas, P., Acuña, O. S., Avilés, M., Coy, P., Rizos, D., and Gutiérrez-Adán, A. The oviduct: from sperm selection to the epigenetic landscape of the embryo. Biol Reprod 2018, 98 (3):262-276. [CrossRef]
- Sakkas, D., Ramalingam, M., Garrido, N., and Barratt C. L. Sperm selection in natural conception: what can we learn from mother nature to improve assisted reproduction outcomes? Hum Reprod Update 2015, 21 (6):711-26. [CrossRef]
- Baldini, D., Ferri, D., Baldini, G. M., Lot D., Catino, A., Vizziello, D., and Vizziello, G. Sperm selection for ICSI: Do we have a winner? Cells 2021, 10 (12). [CrossRef]
- Henkel, R. Sperm preparation: state-of-the-art-physiological aspects and application of advanced sperm preparation methods. Asian J Androl 2012, 14 (2):260-9. [CrossRef]
- Dai, C., Zhang, Z., Shan, G., Chu, L. T., Huang, Z., Moskovtsev, S., Librach, C., Jarvi, K., and Sun, Y. Advances in sperm analysis: techniques, discoveries and applications. Nat Rev Urol 2021, 18 (8):447-467. [CrossRef]
- Gandini, L., Lombardo, F., Paoli, D., Caruso, F., Eleuteri, P., Leter, G., Ciriminna, R., Culasso, F., Dondero, F., Lenzi, A., and Spanò, M. Full-term pregnancies achieved with ICSI despite high levels of sperm chromatin damage. Hum Reprod 2004, 19 (6):1409-17. [CrossRef]
- Sakkas, D., and Alvarez, J. G. Sperm DNA fragmentation: mechanisms of origin, impact on reproductive outcome, and analysis. Fertil Steril 2010, 93 (4):1027-36. [CrossRef]
- Gotsiridze, K., Nana, M., Mariam, M. and Tamar, J. Live motile sperm sorting device improves embryo aneuploidy: A Retrospective Cohort Study. Fertility & Reproduction 2024, 06 (03):117-122. [CrossRef]
- Quinn, M. M., Jalalian, L., Ribeiro, S., Ona, K., Demirci, U., Cedars, M. I., and Rosen, M. P. Microfluidic sorting selects sperm for clinical use with reduced DNA damage compared to density gradient centrifugation with swim-up in split semen samples. Hum Reprod 2018, 33 (8):1388-1393. [CrossRef]
- Sheibak, N., Amjadi, F., Shamloo, A., Zarei, F., and Zandieh, Z. Microfluidic sperm sorting selects a subpopulation of high-quality sperm with a higher potential for fertilization. Hum Reprod 2024, 39 (5):902-911. [CrossRef]
- Banti, M., Van Zyl, E., and Kafetzis, D. Suprem preparation with microfluidic sperm sorting chip may improve intracytoplasmic sperm injection outcomes compared to density gradient centrifugation. Reprod Sci 2024, 31 (6):1695-1704. [CrossRef]
- Anbari, F., Khalili, M. A., Sultan Ahamed, A. M., Mangoli, E., Nabi, A., Dehghanpour, F., and Sabour, M. Microfluidic sperm selection yields higher sperm quality compared to conventional method in ICSI program: A pilot study. Syst Biol Reprod Med 2021, 67 (2):137-143. [CrossRef]
- Feyzioglu, B. S., and Avul, Z. Effects of sperm separation methods before intrauterine insemination on pregnancy outcomes and live birth rates: Differences between the swim-up and microfluidic chip techniques. Medicine (Baltimore) 2023, 102 (46):e36042. [CrossRef]
- Pujianto, D. A., Oktarina, M., Sharma Sharaswati, I. A., and Yulhasri. Hydrogen peroxide has adverse effects on human sperm quality parameters, induces apoptosis, and reduces survival. J Hum Reprod Sci 2021, 14 (2):121-128. [CrossRef]
- Hsu, C. T., Lee, C. I., Lin, F. S., Wang, F. Z., Chang, H. C., Wang, T. E., Huang, C. C., Tsao, H. M., Lee, M. S., and Agarwal, A. Live motile sperm sorting device for enhanced sperm-fertilization competency: comparative analysis with density-gradient centrifugation and microfluidic sperm sorting. J Assist Reprod Genet 2023, 40 (8):1855-1864. [CrossRef]
- Andrabi, S. W., Ara, A., Saharan, A., Jaffar, M., Gugnani, N., and Esteves, S. C. Sperm DNA fragmentation: causes, evaluation and management in male infertility. JBRA Assist Reprod 2024, 28 (2):306-319. [CrossRef]
- Wright, C., Milne, S., and Leeson, H. Sperm DNA damage caused by oxidative stress: modifiable clinical, lifestyle and nutritional factors in male infertility. Reprod Biomed Online 2014, 28 (6):684-703. [CrossRef]
- Vasilescu, S. A., Ding, L., Parast, F. Y., Nosrati, R., and Warkiani. Sperm quality metrics were improved by a biomimetic microfluidic selection platform compared to swim-up methods. Microsyst Nanoeng 2023, 9:37. [CrossRef]
- Kocur, O. M., Xie P., Cheung, S., Souness, S., McKnight, M., Rosenwaks, Z., and Palermo, G. D. Can a sperm selection technique improve embryo ploidy? Andrology 2023, 11 (8):1605-1612. [CrossRef]
- Parrella, A., Keating, D., Cheung, S., Xie, P., Stewart, J. D., Rosenwaks, Z., and Palermo, G. D. A treatment approach for couples with disrupted sperm DNA integrity and recurrent ART failure. J Assist Reprod Genet 2019, 36 (10):2057-2066. [CrossRef]
- Shirota, K., Yotsumoto, F., Itoh, H., Obama, H., Hidaka, N., Nakajima, K., and Miyamoto, S. Separation efficiency of a microfluidic sperm sorter to minimize sperm DNA damage. Fertil Steril 2016, 105 (2):315-21.e1. [CrossRef]
- Muratori, M., Tarozzi, N., Cambi, M., Boni, L., Iorio, A. L., Passaro, C., Luppino, B., Nadalini, M., Marchiani, S., Tamburrino, L., Forti, G., Maggi, M., Baldi, E., and Borini, A. Variation of DNA fragmentation levels during density gradient sperm selection for assisted reproduction techniques: a possible new male predictive parameter of pregnancy? Medicine (Baltimore) 2016, 95 (20):e3624. [CrossRef]
- akeshima, T., Yumura, Y., Kuroda, S., Kawahara, T., Uemura, H., and Iwasaki, A.. Effect of density gradient centrifugation on reactive oxygen species in human semen. Syst Biol Reprod Med 2017, 63 (3):192-198. [CrossRef]
- Raad, G., Bakos, H. W., Bazzi, M., Mourad, Y., Fakih, F., Shayya, S., Mchantaf, L., and Fakih, C. Differential impact of four sperm preparation techniques on sperm motility, morphology, DNA fragmentation, acrosome status, oxidative stress, and mitochondrial activity: A prospective study. Andrology 2021, 9 (5):1549-1559. [CrossRef]
- Esteves, S. C., Sharma, R. K., Thomas, A. J., and Agarwal, A. Effect of swim-up sperm washing and subsequent capacitation on acrosome status and functional membrane integrity of normal sperm. Int J Fertil Womens Med 2000, 45 (5):335-341.
- Paoli, D., Gallo, M., Rizzo, F., Baldi, E., Francavilla, S., Lenzi, A., Lombardo, F., and Gandini, L. Mitochondrial membrane potential profile and its correlation with increasing sperm motility. Fertil Steril 2011, 95 (7):2315-2319. [CrossRef]
- Agarwal, A., Saleh, R.A. Role of oxidants in male infertility: rationale, significance, and treatment. Urol Clin North Am. 2002,29(4):817-27. [CrossRef]
- Ferreira Aderaldo, J., da Silva Maranhão, K., and Ferreira Lanza, D. C. Does microfluidic sperm selection improve clinical pregnancy and miscarriage outcomes in assisted reproductive treatments? A systematic review and meta-analysis. PLoS One 2023, 18 (11):e0292891. [CrossRef]
- Jahangiri, A. R., Ziarati, N., Dadkhah, E., Bucak, M. N., Rahimizadeh, P., Shahverdi, A., Sadighi Gilani, M. A., and Topraggaleh, T. R. Microfluidics: The future of sperm selection in assisted reproduction. Andrology 2024, 12 (6):1236-1252. [CrossRef]
- World Health Organization. 2010. WHO laboratory manual for the examination and processing of human semen. 5th ed. Geneva: World Health Organization.
- Doostabadi, M. R., Mangoli, E., Marvast, L. D., Dehghanpour, F., Maleki, B., Torkashvand, H., and Talebi, A. R.. Microfluidic devices employing chemo- and thermotaxis for sperm selection can improve sperm parameters and function in patients with high DNA fragmentation. Andrologia 2022, 54 (11):e14623. [CrossRef]
- Gangwar, C., Kharche, S. D., Mishra, A. K., Saraswat, S., Kumar, N., and Sikarwar A.K. Effect of diluent sugars on capacitation status and acrosome reaction of spermatozoa in buck semen at refrigerated temperature. Trop Anim Health Prod 2020, 52 (6):3409-3415. [CrossRef]
- Smiley, S. T., Reers, M., Mottola-Hartshorn, C., Lin, M., Chen, A., Smith, T. W., Steele, G. D., and Chen, L.B. Intracellular heterogeneity in mitochondrial membrane potentials revealed by a J-aggregate-forming lipophilic cation JC-1. Proc Natl Acad Sci U S A 1991, 88 (9):3671-3675. [CrossRef]
- Maleki, B., Khalili M. A., Gholizadeh L., Mangoli E., and Agha-Rahimi, A. Single sperm vitrification with permeable cryoprotectant-free medium is more effective in patients with severe oligozoospermia and azoospermia. Cryobiology 2022, 104:15-22. [CrossRef]
| Parameters | Mean ± SD |
|---|---|
| Number of patients Age Volume (ml) |
45 32.2 ± 7.2 4.19 ± 1.7 |
| Concentration (x106 cell/ml) Progressive motility (%) Total motility (%) Normal morphology (%) High MMP (%) DFI (%) Intact acrosome (%) ROS (µM, H2O2) |
46.3 ± 35.17 42.08 ± 13.74 51.11 ± 13.74 5.13 ± 2.37 87.56 ± 3.96 18.33 ± 8.71 26.8 ± 5.12 53.32 ± 25.36 |
| Parameters | Ejaculate | Separation methods | mean ± SD |
|---|---|---|---|
| Concentration (x106 sperm/ml) | 46.37 ± 35.17 | Swim-up | 6.3 ± 5.9a |
| MFSS | 8.6 ± 8.9a | ||
| Total amount of sperm (x106) | 180.59 ± 146.94 | Swim-up | 4.02 ± 4.17a |
| MFSS | 2.9 ± 2.8a | ||
| Total motility (%) | 51.11 ± 13.74 | Swim-up | 89.71 ± 10.18a |
| MFSS | 92.84 ± 4.82a | ||
| Normal morphology (%) | 5.13 ± 2.37 | Swim-up | 17.23 ± 6.07a |
| MFSS | 18.15 ± 5.26a | ||
| DNA fragmentation (%) | 18.33 ± 8.71 | Swim-up | 9.85 ± 7.95a |
| MFSS | 5.99 ± 6.34b | ||
| High mitochondrial potential (%) | 87.56 ± 3.96 | Swim-up | 93.43 ± 2.32a |
| MFSS | 93.37 ± 2.75a | ||
| Intact acrosome (%) | 26.8 ± 8.11 | Swim-up | 79.09 ± 14.21a |
| MFSS | 79.72 ± 10.45a |
| Parameters | Swim-up | MFSS | p-value |
|---|---|---|---|
| Concentration (x106 sperm/ml) | 5.58 ± 5.73 | 12.03 ± 9.31 | 0.11 |
| Total amount of sperm (x106) | 3.92 ± 2.67 | 5.56 ± 4.34 | 0.1 |
| Total motility (%) | 91.34 ± 5.95 | 92.76 ± 5.28 | 0.36 |
| Normal morphology (%) | 18.65 ± 6.02 | 19.44 ± 5.08 | 0.54 |
| DNA fragmentation (%) | 9.78 ± 7.85 | 7.03 ± 7.43 | 0.2 |
| High mitochondrial potential (%) | 93.18 ± 2.14 | 93.88 ± 1.74 | 0.2 |
| Intact acrosom (%) | 81.2 ± 13.52 | 82.47 ± 10.84 | 0.7 |
| Parameters | Swim-up | MFSS | p-value |
|---|---|---|---|
| Concentration (x106 sperm/ml) | 3.2 ± 4.9 | 4.1 ± 6.1 | 0.6 |
| Total amount of sperm (x106) | 1.5 ± 2.39 | 1,9 ± 2.88 | 0.63 |
| Total motility (%) | 87.47 ± 13.97 | 92,94 ± 4.24 | 0.11 |
| Normal morphology (%) | 15.28 ± 5.75 | 16,36 ± 5.09 | 0.54 |
| DNA fragmentation (%) | 9.94 ± 8.3 | 4,56 ± 4.23 | 0.016 |
| High mitochondrial potential (%) | 93.78 ± 2.31 | 92.68 ± 3.56 | 0.29 |
| Intact acrosom (%) | 76.15 ± 14.96 | 75.95 ± 8.83 | 0.96 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




