1. Introduction
Immunoglobulin-related/light chain amyloidosis (AL amyloidosis) or primary amyloidosis is the most common type of a rare systemic disease known as amyloidosis. According to the 2022 World Health Organization (WHO) classification this disease falls into the plasma cell neoplasms and related disorders category, specifically within the subgroup of diseases with monoclonal immunoglobulin deposition, as immunoglobulin-related amyloidosis [
1]. The International Consensus Classification (ICC) is also in use to classify lymphoid neoplasms, in this system this disease is placed in the same subgroup as in the WHO classification, but is referred to as immunoglobulin light chain amyloidosis (AL amyloidosis) [
2]. In AL amyloidosis the amyloidogenic proteins are monoclonal immunoglobulin light chains (kappa, lambda or both types, sometimes just their fragments) produced by plasma cells.
The symptoms of the disease are heterogeneous due to its multi-organ manifestations. The most commonly affected organs are the liver, kidneys, gastrointestinal tract and heart [
3]. As for to life expectancy, cardiac manifestation of amyloidosis can lead to early death in patients [
4].
The diagnosis is based on microscopic evaluation of tissue samples of affected organs stained with Congo red and further immunohistochemical examinations to clarify the type of amyloid light chain involved. Fluorescence in situ hybridization (FISH) is also important in the diagnostic procedure as around 50% of patients with AL amyloidosis have t(11;14) translocation [
5]. This chromosomal translocation results in the overexpression of cyclin D1 and B-cell lymphoma 2 (BCL-2) proteins in plasma cells, enabling them to avoid apoptosis [
6].
Cure of AL amyloidosis is still unresolved; however, all patients require therapy to prevent further deposition of amyloid in affected organs. Venetoclax is a selective, oral inhibitor of the anti-apoptotic protein BCL-2. By inhibiting BCL-2 this drug promotes programmed cell death, leading to the natural destruction of plasma cells that produce abnormal proteins [
6]. Since the first report of using venetoclax-based regimen in t(11;14) AL amyloidosis multiple case reports have demonstrated the efficacy of venetoclax in this indication [
7], [
8].
Hereby we present the case history of one of our patients with t(11;14) immunoglobulin-related amyloidosis who was effectively treated with the combination of venetoclax, cyclophosphamide, bortezomib and dexamethasone.
2. Case Report
The examination of a 61-year-old male patient started in September 2022. at the Gastroenterology Department of University of Debrecen due to weight loss and abdominal pain under the right costal margin. His medical history included hypertension, duodenal ulcer, gastritis and reflux disease, his Eastern Cooperative Oncology Group Performance Status was classified as ECOG PS 1. Laboratory tests and imaging studies (colonoscopy, gastroscopy) revealed cholestasis, fatty liver disease and cholecystolithiasis. Shear wave elastography showed cirrhosis (87 kPa; stage F4, IQR: 6,5 kPa). Endoscopic retrograde cholangiopancreatography (ERCP) detected stenosis of the papilla of Vater, bile duct lavage was performed resulting in the initiation of bile drainage.
Examinations aimed at detecting malignancy were negative, however CEA and CA 19-9 values were elevated. His personal and family histories were both negative for malignancy.
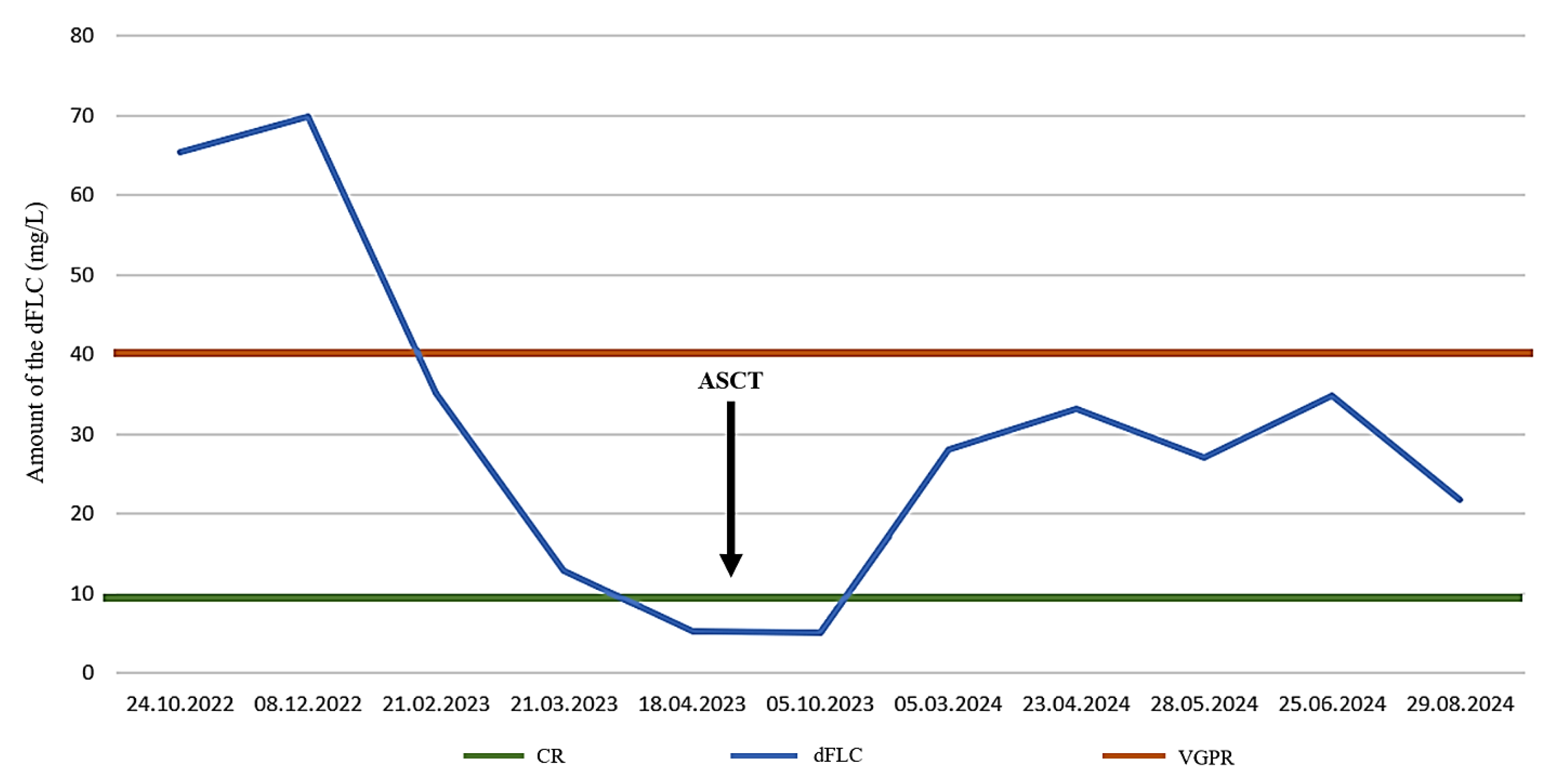
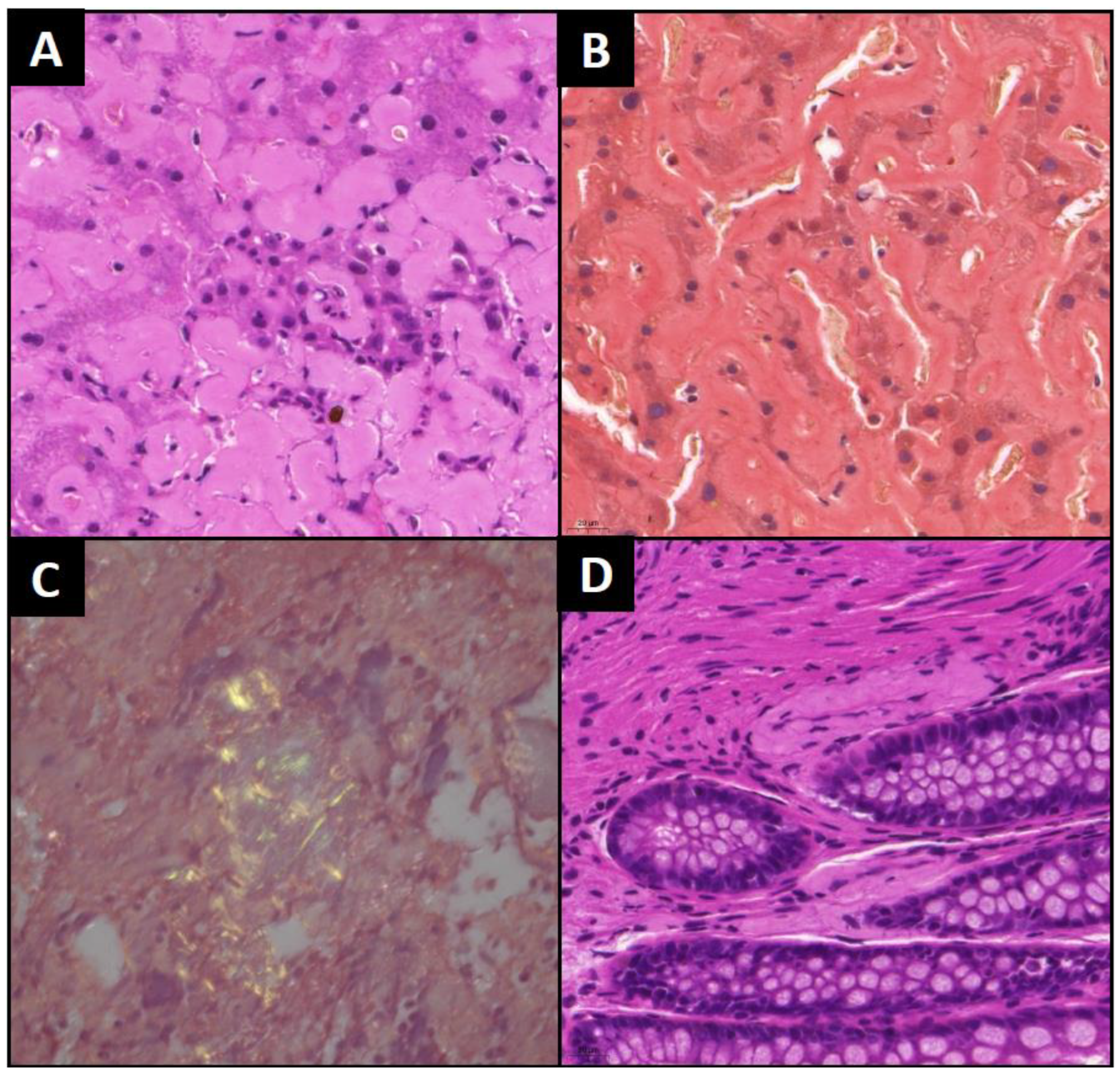
In October 2022 abdominal computer tomography (CT) showed mediastinal, hilar and abdominal lymphadenopathy. The patient was referred to our clinic on 24 October 2022 due to B symptoms and lymphadenopathy. Bone marrow biopsy (posterior iliac crest) showed massive amorphous interstitial deposition (60% of the parenchyma) consistent with amyloidosis without significant plasma cell infiltration (4-5% CD138 positivity with immunohistochemical staining). The sample faintly positive for Congo red exhibited apple-green birefringence under polarized light microscopy. While serum protein electrophoresis did not detect monoclonal gammopathy (M component), free light chain lambda and kappa were 74,8 mg/L and 9,46 mg/L respectively, with a ratio (serum free lambda/kappa light chain) of 7,9. The difference between involved and uninvolved free light chains (dFLC) was 65,34 mg/L (
Figure 3). At the same time biopsies from the liver and the colon were also taken. The liver sample showing amorphous, faint eosinophilic deposition and apple-green birefringence with Congo red staining under polarized light. Tissue sample from the colon was negative at this time, but another bone marrow biopsy we performed in December clearly confirmed the diagnosis of amyloidosis (
Figure 1). 80% of the parenchyma was filled with the same homogenous deposition mentioned above.
His recent serum protein electrophoresis did not show monoclonal gammopathy (M component). Free light chain assay detected lambda and kappa concentrations of 80,1 mg/L and 10,2 mg/L respectively, the lambda/kappa ratio was 7,85, the dFLC was 69,9. Immunohistochemistry (IHC) revealed strong CD138 and lambda, weak kappa and transthyretin positivity of the amyloid plaques, and the samples were also MUM 1 (multiple myeloma oncogene 1) and cyclin D1 positive. These results urged us to revise the first colon biopsy IHC, this time a small amount of amyloid deposition was found with Congo red staining. On the lymphoid gene panel no abnormality was detected. Positron emission tomography-computed tomography (PET-CT) showed lymph nodes unsuitable for sampling with a low standardized uptake value (SUVmax was 1,8, with a reference liver SUVmax of 2,8).
We referred our patient for cardiac evaluation to assess heart involvement. Transthoracic echocardiography (TTE) examination was performed, showing diastolic dysfunction (Stage I) and left ventricular enlargement.
The cardiac muscle of the interventricular septum and posterior wall were both 14-15 mm thick; the level of serum cardiac troponin T (cTnT) was 40,85 ng/L (normal range: <30 ng/L) and the level of serum N-terminal portion of pro-brain natriuretic peptide type B (NT-proBNP) was also elevated, exactly 409 ng/L (normal range:<176,8 ng/L). Concurrently we performed fluorescent in situ hybridization (FISH) assay to check the presence of t(11;14), the most common cytogenetic abnormality, also a prognostic and predictive biomarker in AL amyloidosis. The presence of this translocation associated with more pronounced cardiac involvement, lower hematologic response and reduced survival rates. The t(11;14) translocation was confirmed by FISH in our patient. Additionally, gene rearrangement in the immunoglobulin VH-VJ regions was also detected.
Based on results of the above detailed evaluation and as per Mayo 2012 criteria the diagnosis of stage II. immunoglobulin-related amyloidosis could be established.
The 28-day CyBorD regimen was initiated on 27 December 2022. This combination chemotherapy included cyclophosphamide (300 mg per on days 1, 8, 15), bortezomib (2,2 mg per day, administered subcutaneously on days 1, 4, 8, 11) and dexamethasone (20 mg days 1-2, 8-9, 15-16, 22-23). Due to t(11;14) venetoclax was also administered from day 1 of the second CyBorD cycle. On course of a venetoclax dose ramp-up at dose level 200 mg per day the patient developed grade II thrombocytopenia, subsequently 100 mg per day venetoclax was administered throughout in induction and maintenance. During the treatment we did not observe any other hematological toxicities or side effects necessitating intervention.
In AL amyloidosis venetoclax is an off-label therapy. Regulatory permission to use venetoclax in this indication was granted by the National Institute of Pharmacy and Nutrition and treatment costs were covered by the Hungarian National Health Insurance Fund following named-patient based reimbursement application.
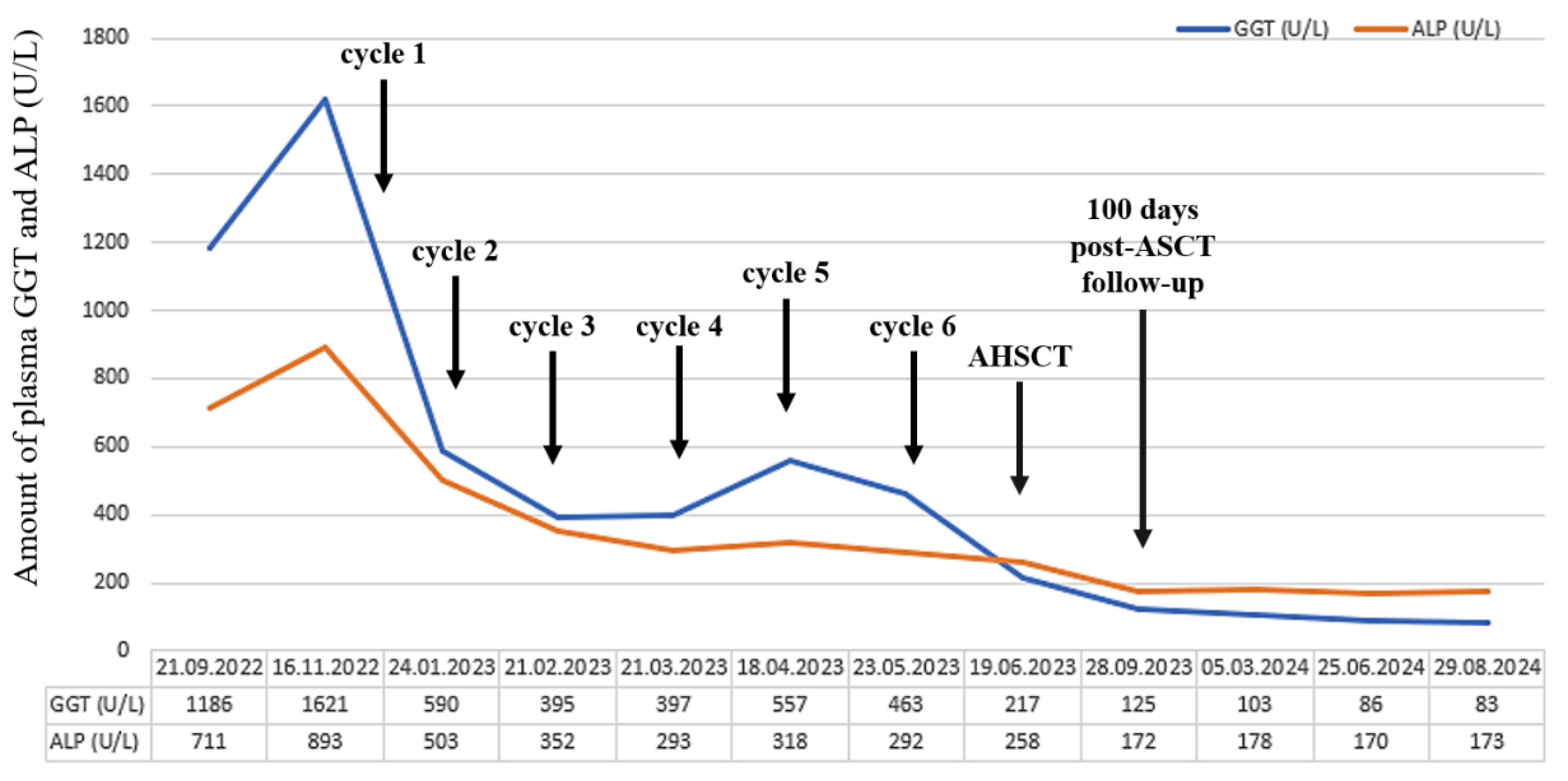
Shortly after treatment initiation, our patient’s clinical condition improved significantly. Furthermore, liver-function tests also showed positive changes after the first cycle of therapy, inasmuch the amount of GGT was reduced to a quarter and ALP also decreased by almost half (
Figure 2).
Shear wave elastography was repeated in February 2023, unfortunately no improvement was observed (139,4 kPa; stage F4). After the third treatment cycle a control iliac crest bone marrow biopsy was performed. Despite clinical improvement, no significant quantitative or qualitative changes were observed. However, concurrent serum electrophoresis showed the normalization of free light chain levels (serum lambda light chain: 22,4 mg/L, kappa light chain: 9,53 mg/L, lambda/kappa light chain ratio: 2,35, dFLC: 12,87), and no M component was detected.
Ursodeoxycholic acid was also initiated due to significant liver involvement signified by elevated serum alkaline phosphatase and γ-glutamyl transferase levels.
Between 27th of December 2022 and 23rd of May 2023 the patient received six cycles of CyBorD and venetoclax as a result complete remission was achieved by April 2023. Control echocardiography performed in June 2023 indicated some improvement insofar the thickness of the left atrium wall decreased from 47 mm to 38 mm. Follow-up bone marrow biopsies showed less than 5% of the initial clonal plasma cells. Serum free light chain levels decreased by nearly 90%, the lambda/kappa ratio was 1,85, and dFLC decreased to 5,33. Cholestasis tests also showed improvement (
Figure 2 and
Figure 3). The patient reaching ECOG PS 0 and being in a sufficiently deep of remission by April 2023, autologous stem cell transplantation (ASCT) has become a viable step forward. Autologous stem cell transplantation is considered highly effective in eligible patients, especially with pretransplant CyBorD induction followed by melphalan conditioning. Solo granulocyte-colony stimulating factor (G-CSF) was used for stem cells mobilization. The collection of CD34 positive stem cells from the peripheral blood was successfully performed between the 2nd and 4th of June, we collected 4,62x10
6 per kg of body weight without any complications. The conditioning regimen included 350 mg melphalan (200 mg/m
2), it was followed by the reinfusion of 2,8x10
6 per kg of body weight cryopreserved stem cells on the 30th of June.
Figure 3.
Difference between involved and uninvolved free light chains (dFLC) during treatment before and after autologous stem cell transplantation (ASCT, 20.06.2023). VGPR is defined as dFLC less than 40 mg/L, while CR is defined as dFLC less than 10 mg/L. CR: complete remission; VGPR: very good partial remission.
Figure 3.
Difference between involved and uninvolved free light chains (dFLC) during treatment before and after autologous stem cell transplantation (ASCT, 20.06.2023). VGPR is defined as dFLC less than 40 mg/L, while CR is defined as dFLC less than 10 mg/L. CR: complete remission; VGPR: very good partial remission.
Bone marrow sample was collected on the day 100 check-up on the 28th of September from the iliac crest: though the amorphous, homogeneous, pale eosinophilic deposit continues to occupy a significant portion of the parenchyma (50 %), although its quantity has somewhat decreased. This time a monoclonal immunoglobulin G (IgG) lambda component was detectable in the sample (1,2 g/L). The levels of serum lambda and kappa free light chains improved and their difference further decreased to 5,11 (
Figure 3).
With his disease in hematological remission the patient’s clinical status was excellent. Maintenance treatment with venetoclax at a dose of 100 mg per day was initiated, he tolerated it well without any hematological toxicities or major infective episodes.
We continue to see our patient on regular follow-up visits. Liver function tests (GGT and ALP) show further improvement (
Figure 2). At last check-up he was in a good clinical condition and his disease was in very good partial remission. The last serum protein electrophoresis performed on 29 August 2024, detected oligoclonal lambda component at the gamma region, and revealed the following-free light values: lambda 32,1 mg/L, kappa 10,4 mg/L, dFLC of 21,7 mg/L. The lambda/kappa ratio was 3,08.
3. Conclusions
Immunoglobulin-related or light chain amyloidosis (AL amyloidosis) is a rare and potentially life-threatening disorder caused by the deposition of abnormal light chain proteins in various tissues of various organs, leading to organ dysfunction and damage. Current therapies focus on eliminating the plasma cell clone, that produce the amyloidogenic protein, thus preventing the formation of additional amyloid deposits and further organ damage [
9].
Treatment guidelines, such as those provided by the International Society of Amyloidosis (ISA) and the European Hematology Association (EHA) can also be helpful in special cases.
Most chemotherapy regimens used in AL amyloidosis are extrapolated from multiple myeloma (MM), justified by similarities [
10]. A non-exhaustive list of agents used include corticosteroids (dexamethasone), alkylating agents (cyclophosphamide), immunomodulatory drugs (thalidomide), proteasome inhibitors (bortezomib) and daratumumab [
11]. With the combination of these drugs the hematologic response rate achieved is in the range of 60-94 % [
8].
Approximately 50% of patients with light chain amyloidosis have t(11;14) with the consequential overexpression of cyclin D1 and the B-cell lymphoma 2 (BCL-2) protein in plasma cells, allowing them to avoid apoptosis [
6]. Venetoclax is a BH3 mimetic selectively inhibiting the anti-apoptotic protein BCL-2. By BCL-2 inhibition this drug promotes programmed cell death in susceptible cells via intrinsic apoptosis this time that of plasma cells that produce abnormal proteins [
6]. T(11;14) is also present in MM where venetoclax is already recognized as an effective (though not registered) treatment option for patients with this cytogenetic abnormality and is used in monotherapy or in combinations [
12], [
13]. Most of the time, venetoclax proves to be an effectively salvage therapy in relapses or refractory disease patients reaching complete remission (CR) or at least a very good partial remission (VGPR).
Results from the randomized double-blind multicenter Phase 3 BELLINI study have raised many questions regarding the safety of venetoclax. Later, it was discovered that the higher mortality rate associated with venetoclax treatment was due to the very high efficacy of this targeted therapy. Inhibition of BCL-2 occurs not only in abnormal plasma cells but also in healthy ones. Severe hypogammaglobulinemia can develop, which favors the spread of the infectious agents [
10].
Considering the presence of the t(11;14) in both MM and AL amyloidosis and the key role of plasma cells in both diseases, venetoclax could be as useful in AL-amyloidosis as it is in MM for those patients who have this cytogenetic abnormality [
10]. The first report of the activity of a venetoclax-based combination treatment (venetoclax, bortezomib, dexamethasone) in AL amyloidosis patients with a partial hematologic response to CyBorD, was published by Leung et al. [
7], [
8]. Since then, several new reports have emerged on using this salvage treatment, and what is more, not only in patients whose response plateaued on CyBorD, or those who are relapsed and refractory, but also in patients with severe cardiac or kidney involvement. These patients achieved CR or VGPR with venetoclax as a second-line therapy [
14].
In a Multicenter International Retrospective Real-World Study, the combination of venetoclax with daratumumab or dexamethasone did not result in more favorable outcomes compared to venetoclax monotherapy [
15]. On the other hand, the combination of daratumumab with CyBorD demonstrated positive results in the phase III ANDROMEDA trial, indicating its effectiveness for patients with AL amyloidosis [
16].
As we have already seen, venetoclax is most often used as a second-line or salvage therapy, however we have less knowledge about its effectiveness when used in the early stages of these diseases [
17]. Unfortunately, there are only scarce reports with limited value on venetoclax as maintenance treatment for immunoglobulin-related amyloidosis. The maintenance treatment of AL amyloidosis is not a well-studied area and there are no firmly established evidence-based treatment options in clinical practice yet [
18]. There are two phase 2 trials on consolidation therapy post-autologous stem cell transplantation in patients with light chain amyloidosis who did not achieve at least partial remission after previous therapies. In these studies, after 12 months of treatment with either a combination of thalidomide and dexamethasone or bortezomib and dexamethasone, 83 out of 95 patients showed significant improvements [
19].
Our patient underwent a comprehensive medical evaluation and the results encouraged us to administer CyBorD (the standard therapy for AL amyloidosis) combined with venetoclax. He received 6 cycles of this protocol with venetoclax at a dose of 100 mg per day. We also initiated ursodeoxycholic acid based on promising studies demonstrating successful use in AL amyloidosis patients with severe hepatic involvement, marked by elevated alkaline phosphatase and γ-glutamyl transferase levels [
20]. Our patient achieved complete remission with significant improvement in liver test results. Autologous stem cell transplantation was successfully performed. Venetoclax maintenance therapy was reinitiated on the 100th post-transplant day. According to ongoing regular follow-up examinations our patient is in a state of complete hematological remission, the level of dFLC and serum free light chain have almost normalized.
Our case report confirms the notion that venetoclax can be highly effective in selected patients, not only as salvage or second line treatment, but also as part of a first line induction therapy in t(11;14) AL amyloidosis. Furthermore, venetoclax maintenance can also be useful in these cases. Our positive, though limited experiences with venetoclax emphasize the importance of conducting larger studies to explore its further potential.