Submitted:
17 December 2024
Posted:
18 December 2024
You are already at the latest version
Abstract
Keywords:
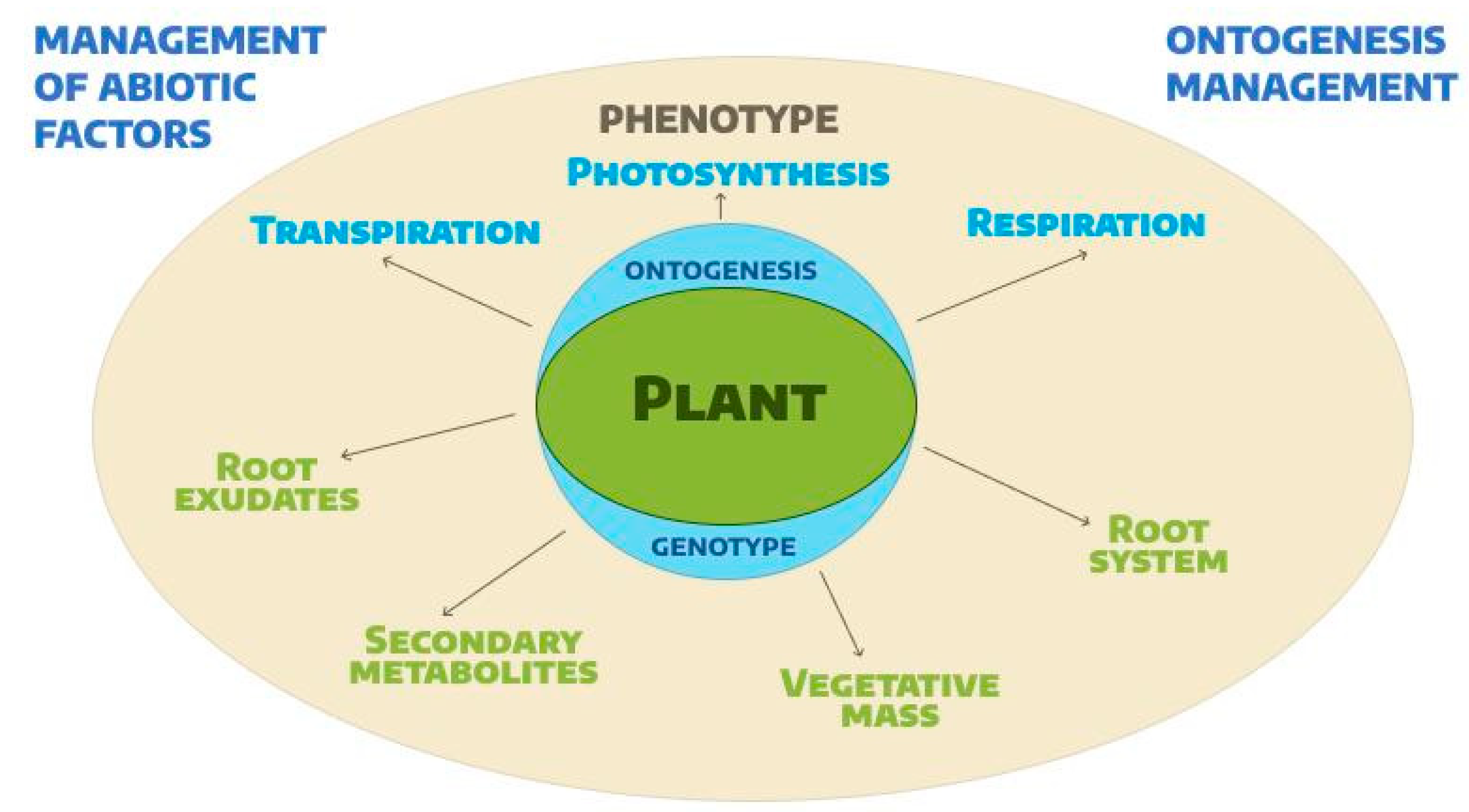
1. Introduction
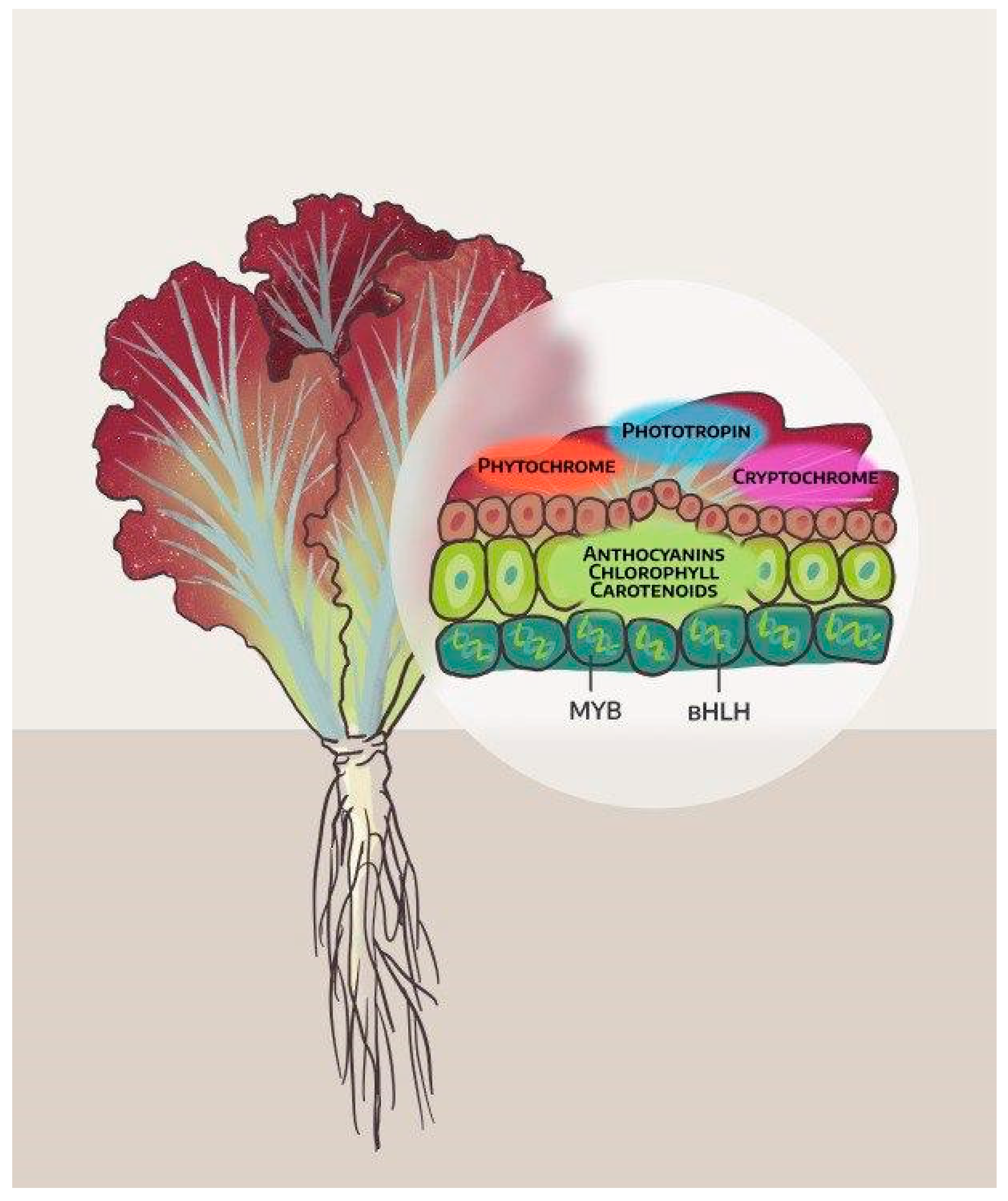
2. The Influence of Light on Leaf Color and Antioxidant Activity of Lettuce
3. Effect of Temperature on Leaf Color and Antioxidant Activity of Lettuce
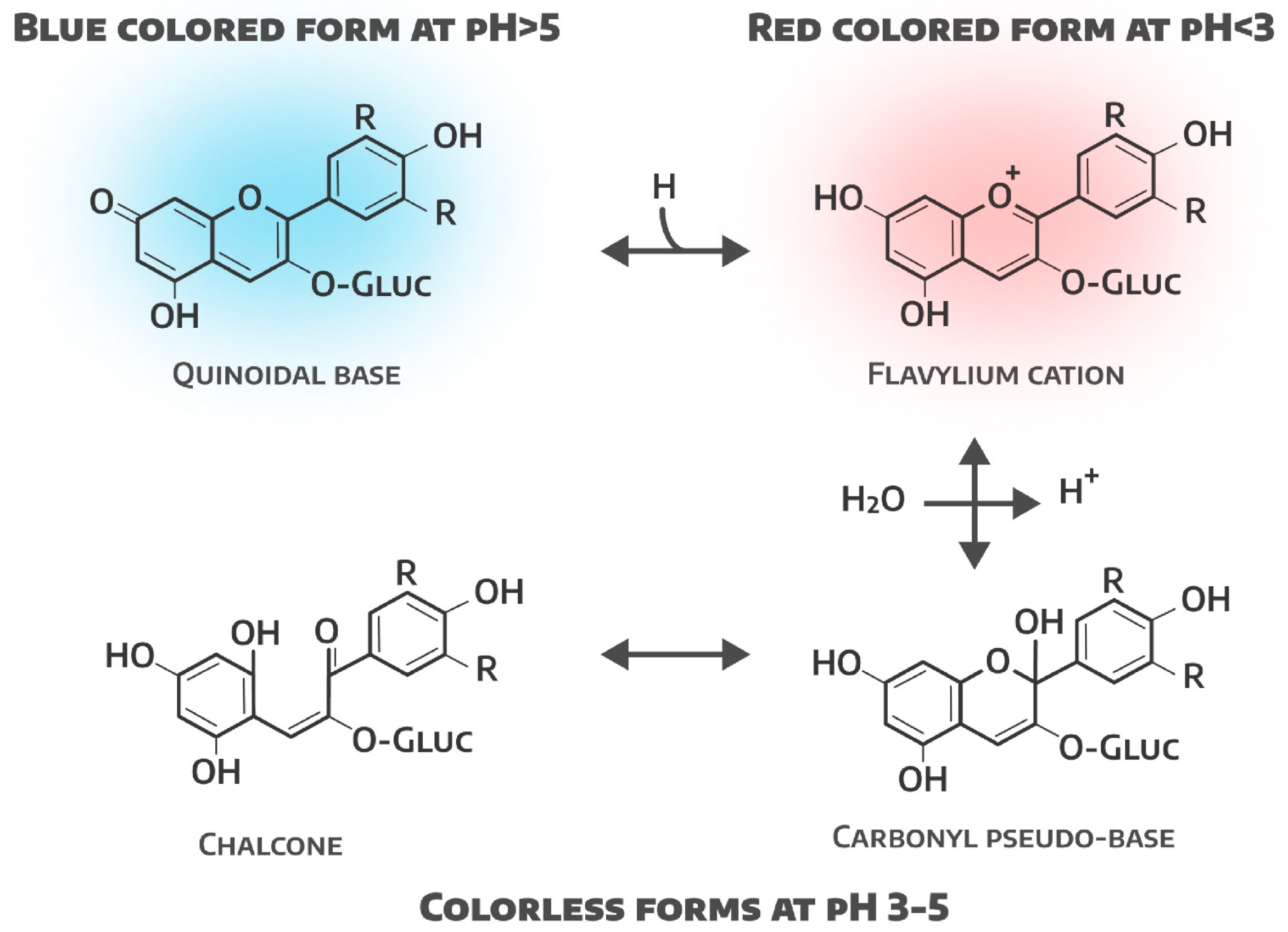
4. Effect of pH on Leaf Color and Antioxidant Activity of Lettuce
5. Bioactive Phytochemicals and Metabolites of Lettuce
6. Genetic Control of Anthocyanin in Lettuces
7. Practical Recommendations for Selection and Cultivation of Red-Leafed Lettuces
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baslam, M.; Morales, F.; Garmendia, I.; Goicoechea, N. Nutritional quality of outer and inner leaves of green and red pigmented lettuces (Lactuca sativa L.) consumed as salads. Sci. Hortic. 2013, 151, 103–111. [Google Scholar] [CrossRef]
- Hung, H.-C.; Joshipura, K.J.; Jiang, R.; Hu, F.B.; Hunter, D.; Smith-Warner, S.A.; Colditz, G.A.; Rosner, B.; Spiegelman, D.; Willett, W.C. Fruit and Vegetable Intake and Risk of Major Chronic Disease. J. Natl. Cancer Inst. 2004, 96, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-E.; Shang, X.; Assefa, A.D.; Keum, Y.-S.; Saini, R.K. Metabolite profiling of green, green/red, and red lettuce cultivars: Variation in health beneficial compounds and antioxidant potential. Food Res. Int. 2018, 105, 361–370. [Google Scholar] [CrossRef]
- Soetan, K.; Olaiya, C.; Oyewole, O. The importance of mineral elements for humans, domestic animals and plants: A review. Afr. J. Food Sci. 2010, 4(5), 200–222. [Google Scholar]
- Huot, B.; Yao, J.; Montgomery, B.L.; He, S.Y. Growth–Defense Tradeoffs in Plants: A Balancing Act to Optimize Fitness. Mol. Plant 2014, 7(8), 1267–1287. [Google Scholar] [CrossRef]
- Shi, M.; Gu, J.; Wu, H.; Rauf, A.; Emran, T.B.; Khan, Z.; Mitra, S.; Aljohani, A.S.M.; Alhumaydhi, F.A.; Al-Awthan, Y.S.; et al. Phytochemicals, Nutrition, Metabolism, Bioavailability, and Health Benefits in Lettuce—A Comprehensive Review. Antioxidants 2022, 11, 1158. [Google Scholar] [CrossRef]
- Mou, B.Q. Genetic variation of beta-carotene and lutein contents in lettuce. J. Am. Soc. Hortic. Sci. 2005, 130, 870–876. [Google Scholar] [CrossRef]
- López, A.; Javier, G.-A.; Fenoll, J.; Hellín, P.; Flores, P. Chemical composition and antioxidant capacity of lettuce: Comparative study of regular-sized (Romaine) and baby-sized (Little Gem and Mini Romaine) types. J. Food Compos. Anal. 2014, 33, 39–48. [Google Scholar] [CrossRef]
- Mampholo, B.M.; Maboko, M.M.; Soundy, P.; Sivakumar, D. Phytochemicals and Overall Quality of Leafy Lettuce (Lactuca sativa L.) Varieties Grown in Closed Hydroponic System. J. Food Qual. 2016, 39, 805–815. [Google Scholar] [CrossRef]
- Das, P.R.; Del Moro, D.S.; Givens, S.R.; Armstrong, S.P.; Walters, K.J. Propagation light intensity influences yield, morphology, and phytochemistry of purple-leaf butterhead lettuce (Lactuca sativa). J. Agric. Food Res. 2024, 16, 101210. [Google Scholar] [CrossRef]
- Assefa, A.D.; Hur, O.-S.; Hahn, B.-S.; Kim, B.; Ro, N.-Y.; Rhee, J.-H. Nutritional Metabolites of Red Pigmented Lettuce (Lactuca sativa) Germplasm and Correlations with Selected Phenotypic Characters. Foods 2021, 10(10), 2504. [Google Scholar] [CrossRef] [PubMed]
- Mekouar, M.A. Food and Agriculture Organization of the United Nations (FAO). Yearb. Int. Environ. Law 2023, 34(1), yvae031. [Google Scholar] [CrossRef]
- Altunkaya, A.; Becker, E.M.; Gökmen, V.; Skibsted, L.H. Antioxidant activity of lettuce extract (Lactuca sativa) and synergism with added phenolic antioxidants. Food Chem. 2009, 115, 163–168. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutrition Res. 2017, 61, 1361779. [Google Scholar] [CrossRef]
- Alappat, B.; Alappat, J. Anthocyanin Pigments: Beyond Aesthetics. Molecules 2020, 25, 5500. [Google Scholar] [CrossRef]
- Anum, H.; Li, K.; Tabusam, J.; Saleh, S.A.A.; Cheng, R.-f.; Tong, Y.-x. Regulation of anthocyanin synthesis in red lettuce in plant factory conditions: A review. Food Chem. 2024, 458, 140111. [Google Scholar] [CrossRef] [PubMed]
- Belwal, T.; Singh, G.; Jeandet, P.; Pandey, A.; Giri, L.; Ramola, S.; Bhatt, I.D.; Venskutonis, P.R.; Georgiev, M.I.; Clément, C.; et al. Anthocyanins, multi-functional natural products of industrial relevance: Recent biotechnological advances. Biotechnology Advances 2020, 43, 107600. [Google Scholar] [CrossRef] [PubMed]
- Colombo, R.; Ferron, L.; Papetti, A. Colored Corn: An Up-Date on Metabolites Extraction, Health Implication, and Potential Use. Molecules 2021, 26, 199. [Google Scholar] [CrossRef]
- Petroni, K.; Tonelli, C. Recent advances on the regulation of anthocyanin synthesis in reproductive organs. Plant Sci. 2011, 181, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Morais, C.A.; de Rosso, V.V.; Estadella, D.; Pisani, L.P. Anthocyanins as inflammatory modulators and the role of the gut microbiota. J. Nutr. Biochem. 2016, 33, 1–7. [Google Scholar] [CrossRef]
- Giampieri, F.; Cianciosi, D.; Alvarez-Suarez, J.M.; Quiles, J.L.; Forbes-Hernández, T.Y.; Navarro-Hortal, M.D.; Machì, M.; Casanova, R.d.J.P.; Espinosa, J.C.M.; Chen, X. Anthocyanins: What do we know until now? J. Berry Res. 2023, 13, 1–6. [Google Scholar] [CrossRef]
- Ferreres, F.; Gil, M.I.; Castañer, M.; Tomás-Barberán, F.A. Phenolic metabolites in red pigmented lettuce (Lactuca sativa). Changes with minimal processing and cold storage. J. Agric. Food Chem. 1997, 45, 4249–4254. [Google Scholar] [CrossRef]
- Stintzing, F.C.; Stintzing, A.S.; Carle, R.; Frei, B.; Wrolstad, R.E. Color and Antioxidant Properties of Cyanidin-Based Anthocyanin Pigments. J. Agric. Food Chem. 2002, 50, 6172–6181. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef]
- Mulabagal, V.; Ngouajio, M.; Nair, A.; Zhang, Y.; Gottumukkala, A.L.; Nair, M.G. In vitro evaluation of red and green lettuce (Lactuca sativa) for functional food properties. Food Chem. 2010, 118, 300–306. [Google Scholar] [CrossRef]
- Grotewold, E. The genetics and biochemistry of floral pigments. Ann. Rev. Plant Biol. 2006, 57, 761–780. [Google Scholar] [CrossRef]
- Liu, W.K.; Yang, Q.C. Effects of short-term treatment with various light intensities and hydroponic solutions on nitrate concentration of lettuce. Acta Agric. Scand. Sect. B - Soil Plant Sci. 2012, 62, 109–113. [Google Scholar] [CrossRef]
- Tsormpatsidis, E.; Henbest, R.G.C.; Davis, F.J.; Battey, N.H.; Hadley, P.; Wagstaffe, A. UV irradiance as a major influence on growth, development and secondary products of commercial importance in Lollo Rosso lettuce ‘Revolution’ grown under polyethylene films. Environ. Exp. Bot. 2008, 63, 232–239. [Google Scholar] [CrossRef]
- Viacava, G.E.; Gonzalez-Aguilar, G.; Roura, S.I. Determination of Phytochemicals and Antioxidant Activity in Butterhead Lettuce Related to Leaf Age and Position. J. Food Biochem. 2014, 38, 352–362. [Google Scholar] [CrossRef]
- Ozgen, S.; Sekerci, S. Effect of leaf position on the distribution of phytochemicals and antioxidant capacity among green and red lettuce cultivars. Span. J. Agric. Res. 2011, 9, 801–809. [Google Scholar] [CrossRef]
- Liang, Y.; Weng, X.; Ling, H.; Mustafa, G.; Yang, B.; Lu, N. Transcriptomic Insights into Molecular Response of Butter Lettuce to Different Light Wavelengths. Plants 2024, 13, 1582. [Google Scholar] [CrossRef] [PubMed]
- Bunning, M.L.; Kendall, P.A.; Stone, M.B.; Stonaker, F.H.; Stushnoff, C. Effects of Seasonal Variation on Sensory Properties and Total Phenolic Content of 5 Lettuce Cultivars. J. Food Sci. 2010, 75, S156–S161. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Iwamoto, T.; Carey, E.E. Antioxidant capacity of leafy vegetables as affected by high tunnel environment, fertilisation and growth stage. J. Sci. Food Agric. 2007, 87, 2692–2699. [Google Scholar] [CrossRef] [PubMed]
- Pierik, R.; de Wit, M. Shade avoidance: phytochrome signalling and other aboveground neighbour detection cues. J.Exp. Bot. 2014, 65(11), 2815–2824. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-J.; Ha, J.-H.; Kim, S.-G.; Choi, H.-K.; Kim, Z.H.; Han, Y.-J.; Kim, J.-I.; Oh, Y.; Fragoso, V.; Shin, K.; et al. Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots. Sci. Signal. 2016, 9(452), ra106. [Google Scholar] [CrossRef]
- Briggs, W.R.; Christie, J.M. Phototropins 1 and 2: versatile plant blue-light receptors. Trends Plant Sci. 2002, 7, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M. Photoregulation of the composition, function, and structure of thylakoid membranes. Annu. Rev. Plant Physiol. 1986, 37, 93–136. [Google Scholar] [CrossRef]
- Spalholz, H.; Perkins-Veazie, P.; Hernández, R. Impact of sun-simulated white light and varied blue:red spectrums on the growth, morphology, development, and phytochemical content of green- and red-leaf lettuce at different growth stages. Sci. Hortic. 2020, 264, 109195. [Google Scholar] [CrossRef]
- Yudina, L.; Sukhova, E.; Gromova, E.; Mudrilov, M.; Zolin, Y.; Popova, A.; Nerush, V.; Pecherina, A.; Grishin, A.A.; Dorokhov, A.A.; et al. Effect of Duration of LED Lighting on Growth, Photosynthesis and Respiration in Lettuce. Plants 2023, 12, 442. [Google Scholar] [CrossRef]
- Wong, C.E.; Teo, Z.W.N.; Shen, L.; Yu, H. Seeing the lights for leafy greens in indoor vertical farming. Trends Food Sci. Technol. 2020, 106, 48–63. [Google Scholar] [CrossRef]
- Samuolienė, G.; Brazaitytė, A.; Sirtautas, R.; Novičkovas, A.; Duchovskis, P. Supplementary red-LED lighting affects phytochemicals and nitrate of baby leaf lettuce. J. Agric. Food Environ. 2011, 9, 271–274. [Google Scholar] [CrossRef]
- Inoue, S.-i.; Kinoshita, T. Blue Light Regulation of Stomatal Opening and the Plasma Membrane H+-ATPase. Plant Physiol. 2017, 174, 531–538. [Google Scholar] [CrossRef]
- Fylladitakis, E.D. Controlled LED lighting for horticulture: a review. Open J. Appl. Sci. 2023, 13, 175–188. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, S.; Cheng, Y.; Peng, Z.; Han, J. Transcriptome profiling of anthocyanin-related genes reveals effects of light intensity on anthocyanin biosynthesis in red leaf lettuce. PeerJ 2018, 6, e4607. [Google Scholar] [CrossRef]
- Blokhina, O.V.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, Oxidative Damage and Oxygen Deprivation Stress: a Review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Noda, K.-i.; Glover, B.J.; Linstead, P.; Martin, C. Flower color intensity depends on specialized cell shape controlled by a Myb-related transcription factor. Nature 1994, 369, 661–664. [Google Scholar] [CrossRef]
- Levine, C.P.; Hayashi, S.; Ohmori, Y.; Kusano, M.; Kobayashi, M.; Nishizawa, T.; Kurimoto, I.; Kawabata, S.; Yamori, W. Controlling root zone temperature improves plant growth and pigments in hydroponic lettuce. Ann. Bot. 2023, 132, 455–470. [Google Scholar] [CrossRef]
- He, J.; Qin, L.; Lee, S.K. Root-zone CO2 and root-zone temperature effects on photosynthesis and nitrogen metabolism of aeroponically grown lettuce (Lactuca sativa L.) in the tropics. Photosynthetica 2013, 51, 330–340. [Google Scholar] [CrossRef]
- Paulsen, G.M. High Temperature Responses of Crop Plants. In Physiology and Determination of Crop Yield; 1994; pp. 365–389. [Google Scholar]
- Porter, J.R.; Gawith, M. Temperatures and the growth and development of wheat: a review. European Journal of Agronomy 1999, 10, 23–36. [Google Scholar] [CrossRef]
- Allen, D.J.; Ort, D.R. Impacts of chilling temperatures on photosynthesis in warm-climate plants. Trends Plant Sci. 2001, 6, 36–42. [Google Scholar] [CrossRef]
- Akula, R.; Ravishankar, G.A. Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal. Behav. 2011, 6, 1720–1731. [Google Scholar] [CrossRef] [PubMed]
- Bumgarner, N.R.; Scheerens, J.C.; Mullen, R.W.; Bennett, M.A.; Ling, P.P.; Kleinhenz, M.D. Root-zone temperature and nitrogen affect the yield and secondary metabolite concentration of fall- and spring-grown, high-density leaf lettuce. J. Sci. Food Agric. 2012, 92(1), 116–124. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-S.; Kim, J.-B.; Cho, K.-J.; Cheon, C.-I.; Sung, M.-K.; Choung, M.-G.; Roh, K.-H. Arabidopsis R2R3-MYB transcription factor AtMYB60 functions as a transcriptional repressor of anthocyanin biosynthesis in lettuce (Lactuca sativa). Plant Cell Rep. 2008, 27, 985–994. [Google Scholar] [CrossRef]
- Boo, H.-O.; Heo, B.-G.; Gorinstein, S.; Chon, S.-U. Positive effects of temperature and growth conditions on enzymatic and antioxidant status in lettuce plants. Plant Sci. 2011, 181, 479–484. [Google Scholar] [CrossRef]
- Rivas-San Vicente, M.; Plasencia, J. Salicylic acid beyond defence: its role in plant growth and development. J. Exp. Bot. 2011, 62(10), 3321–3338. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, M.; Swann, J.R.; Gawthrop, F.; Michelmore, R.; Scaglione, D.; Jose-Truco, M.; Wagstaff, C. Mapping taste and flavour traits to genetic markers in lettuce Lactuca sativa. Food Chem. Mol. Sci. 2024, 9, 100215. [Google Scholar] [CrossRef]
- Takahashi, S.; Murata, N. How do environmental stresses accelerate photoinhibition? Trends Plant Sci. 2008, 13, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Sairam, R.K.; Srivastava, G.C.; Saxena, D.C. Increased Antioxidant Activity under Elevated Temperatures: A Mechanism of Heat Stress Tolerance in Wheat Genotypes. Biol. Plant. 2000, 43, 245–251. [Google Scholar] [CrossRef]
- Naing, A.H.; Kim, C.K. Abiotic stress-induced anthocyanins in plants: Their role in tolerance to abiotic stresses. Physiol. Plant. 2021, 172, 1711–1723. [Google Scholar] [CrossRef] [PubMed]
- Biswas, T.; Mathur, A. Plant Anthocyanins: Biosynthesis, Bioactivity and in vitro Production from tissue cultures. Adv. Biotech. Micro. 2017, 5(5), 555672. [Google Scholar] [CrossRef]
- Jung, H.; Kwak, H.-K.; Hwang, K.T. Antioxidant and antiinflammatory activities of cyanidin-3-glucoside and cyanidin-3-rutinoside in hydrogen peroxide and lipopolysaccharide-treated RAW264.7 cells. Food Sci. Biotechnol. 2014, 23, 2053–2062. [Google Scholar] [CrossRef]
- Assefa, A.D.; Jeong, Y.-J.; Kim, D.-J.; Jeon, Y.-A.; Ok, H.-C.; Baek, H.-J.; Sung, J.-S. Characterization, identification, and quantification of phenolic compounds using UPLC-Q-TOF-MS and evaluation of antioxidant activity of 73 Perilla frutescens accessions. Food Res. Int. 2018, 111, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Llorach, R.; Martínez-Sánchez, A.; Tomás-Barberán, F.A.; Gil, M.I.; Ferreres, F. Characterisation of polyphenols and antioxidant properties of five lettuce varieties and escarole. Food Chem. 2008, 108, 1028–1038. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, U.; Pinzino, C.; Quartacci, M.F.; Ranieri, A.; Sgherri, C. Phenolic Composition and Related Antioxidant Properties in Differently Colored Lettuces: A Study by Electron Paramagnetic Resonance (EPR) Kinetics. J. Agric. Food Chem. 2014, 62, 12001–12007. [Google Scholar] [CrossRef]
- Su, W.; Tao, R.; Liu, W.; Yu, C.; Yue, Z.; He, S.; Lavelle, D.; Zhang, W.; Zhang, L.; An, G.; et al. Characterization of four polymorphic genes controlling red leaf color in lettuce that have undergone disruptive selection since domestication. Plant Biotechnol. J. 2020, 18, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Chung, G.C.; Jang, J.Y.; Ahn, S.J.; Zwiazek, J.J. Overexpression of PIP2;5 Aquaporin Alleviates Effects of Low Root Temperature on Cell Hydraulic Conductivity and Growth in Arabidopsis. Plant Physiol. 2012, 159, 479–488. [Google Scholar] [CrossRef]
- Hong, Y.; Yang, L.-w.; Li, M.-l.; Dai, S.-l. Comparative analyses of light-induced anthocyanin accumulation and gene expression between the ray florets and leaves in chrysanthemum. Plant Physiol. Biochem. 2016, 103, 120–132. [Google Scholar] [CrossRef]
- Yang, X.; Wei, S.; Liu, B.; Guo, D.; Zheng, B.; Feng, L.; Liu, Y.; Tomás-Barberán, F.A.; Luo, L.; Huang, D. A novel integrated non-targeted metabolomic analysis reveals significant metabolite variations between different lettuce (Lactuca sativa. L) varieties. Hortic. Res. 2018, 5, 33. [Google Scholar] [CrossRef]
- de Pascual-Teresa, S.; Sanchez-Ballesta, M.T. Anthocyanins: from plant to health. Phytochem. Rev. 2008, 7, 281–299. [Google Scholar] [CrossRef]
- Khan, R.A.; Abbas, N. Role of epigenetic and post-translational modifications in anthocyanin biosynthesis: A review. Gene 2023, 887, 147694. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Sugumaran, K.; Al-Roumi, A.; Shajan, A. De-novo transcriptome assembly and analysis of lettuce plants grown under red, blue or white light. Scie. Rep. 2022, 12, 22477. [Google Scholar] [CrossRef] [PubMed]
- Khusnutdinov, E.; Sukhareva, A.; Panfilova, M.; Mikhaylova, E. Anthocyanin Biosynthesis Genes as Model Genes for Genome Editing in Plants. Int. J. Mol. Sci. 2021, 22, 8752. [Google Scholar] [CrossRef]
- Shi, L.; Li, X.; Fu, Y.; Li, C. Environmental Stimuli and Phytohormones in Anthocyanin Biosynthesis: A Comprehensive Review. Int. J. Mol. Sci. 2023, 24(22), 16415. [Google Scholar] [CrossRef] [PubMed]
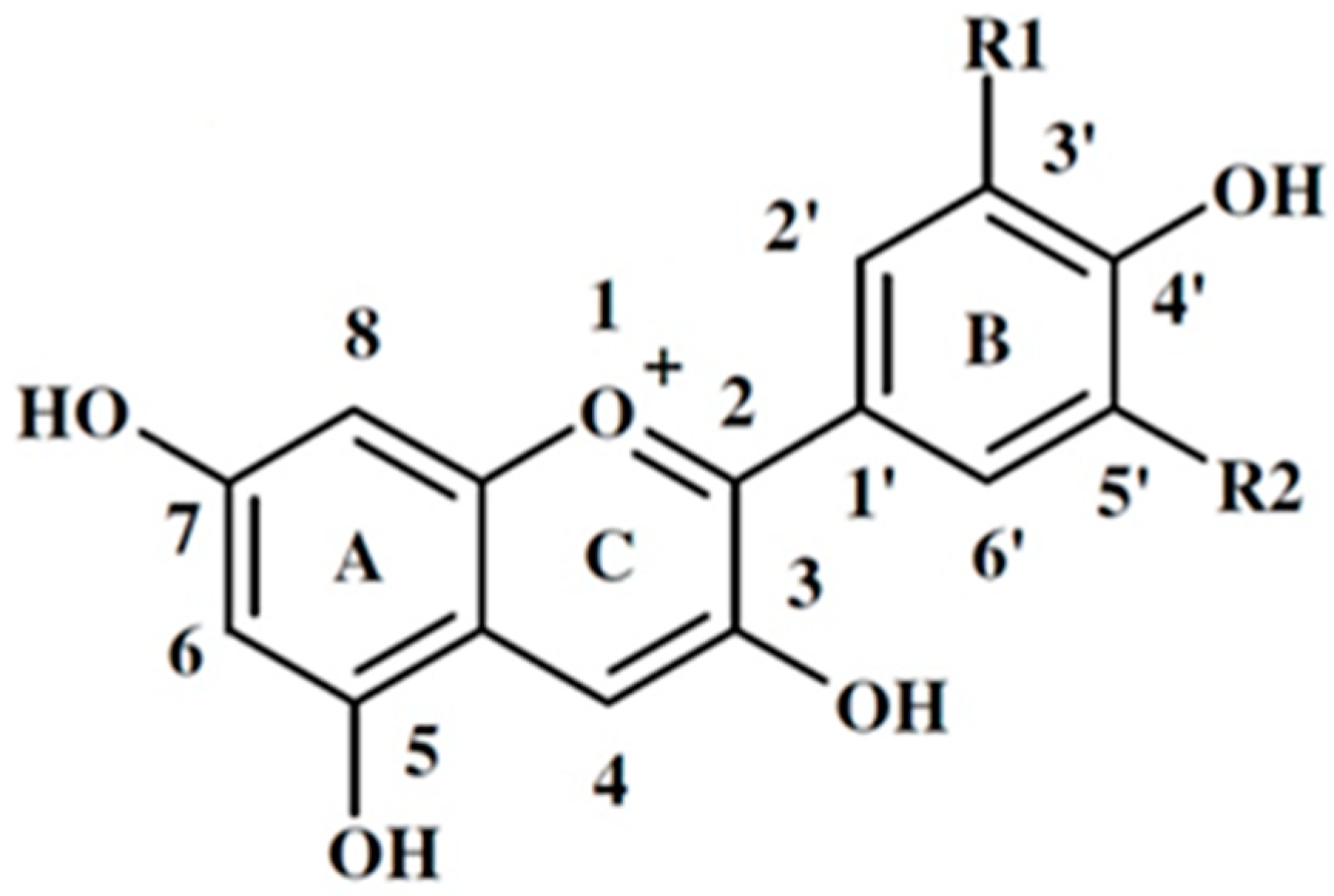
| Anthocyanidin | Side radicals of the B-ring | Color | Present in red leaf lettuce | |
|---|---|---|---|---|
| R1 | R2 | |||
| Anthocyanidins | ||||
| Cyanidin (Cy) | HE | N | orange red | Cyanidin 3-0-(6'-0- malonylglucoside) – 97% of total anthocyanin content ([17]; Belwal et al., 2020) or 251-928 mg/kg wet weight ([18]; Colombo et al., 2021) |
| Peonidin (Pn) | OSN 3 | N | red | |
| Pelargonidine (Pg) | N | N | orange | |
| Malvidin (Mv) | OSN 3 | OSN 3 | purple | |
| Delphinidin (Dp) | HE | HE | blue red | |
| Petunidin (Pt) | OSN 3 | HE | purple | |
| Carajurin | H | H | - | |
| Arrabidin | H | H | - | |
| 5- methylcyanidin | HE | OCH3 | orange red | |
| Peonidin | HE | HE | orange | |
| Capensinidin | OH | OCH3 | blue red | |
| Eupinidin | OH | OCH3 | blue red | |
| Pulchellidin | OH | OCH3 | blue red | |
| 6-hydroxydelphinidin | OH | OH | blue red | |
| Aurantinidin | OH | OH | orange | |
| 30-hydroxyarrabidine | H | H | blue red | |
| Tricetinidine | H | OH | red | |
| 6-hydroxycyanidin | OH | OH | red | |
| Rosinidin | Red | Red | red | |
| other substances that color plants | ||||
| Quercetin | 3-0-(6'-0- malonyl)glycoside ([11]; Assefa et al., 2021) | |||
| Tartaric acid | 2,3 – di-O- caffeoyltartaric acid | |||
| Gene name | Low-light FPKM | High-light FPKM | Log 2 Fold Change (hiht /low light) | FDR | Up/down |
|---|---|---|---|---|---|
| LsCHS Unigene12000_All CL4608.Contig2_All |
1 ,68 11.36 |
858.26 97.28 |
9.0 3.1 |
0 5.33e -259 |
Up |
| LsCHI Unigene10166_All |
10.6 |
68,72 |
2.70 |
1 ,43 e - 96 |
Up |
| LsF3H Unigene8465_All |
14.21 |
408,44 |
4.85 |
0 |
Up |
| LsF'3H CL524.Contig1_All |
9.08 |
193.8 |
4.42 |
0 |
Up |
| LsDFR Unigene2105_All |
2,3 |
473 |
7.68 |
0 |
Up |
| LsANS CL1994.Contig1_All |
4.25 |
269.69 |
5.99 |
0 |
Up |
| Ls3GT CL4808.Contig1_All CL4808.Contig2_All |
5.84 3.31 |
158.28 84.6 |
4.76 4.66 |
0 0 |
Up |
| LsGST Unigene10814_All |
1.84 |
245.41 |
7.06 |
0 |
Up |
| LsMATE Unigene12020_All |
8.42 |
18.14 |
1.11 |
9.25 e - 15 |
Up |
| LsMYB Unigene12430_All Unigene12294_All Unigene23058_All Unigene24751_All CL6440.Contig1_All |
2.21 3.14 0.11 0.56 0.72 |
8.30 37.14 7.25 22.95 4.60 |
1.91 3.56 6.04 5.36 2.66 |
4.36 e - 10 5.02 e - 85 5 ,68 e - 17 7.05 e - 86 9.14 e - 11 |
Up |
| LsbHLH Unigene13011_All |
3.64 |
21.95 |
2.59 |
3.37 e - 84 |
Up |
| LsHY5 Unigene19629_All |
3.18 |
10.42 |
1.71 |
3.15 e - 5 |
Up |
| Factor | Description of the factor | Effect on coloring | Mechanism of action | Reference |
|---|---|---|---|---|
| Genetic factors | ||||
| Mutations in MYB/ bHLH genes | Mutations that result in gain or loss of gene function | Strengthening or weakening of red color | Changes in the transcriptional activity of genes, controlling anthocyanin biosynthesis | ([66]; Su et al., 2020) |
| Gene changes and losses during domestication | Loss of function of wild-type genes during cultivation | Loss of red color, appearance of green varieties | Decreased or stopped synthesis of anthocyanins | ([27]; Liu et al., 2012) |
| Genetic diversity of varieties | Differences between varieties and genotypes | Variation in the content of anthocyanins and phenolic compounds | Genetic predisposition to accumulation of phytochemicals | ([64]; Llorach et al., 2008) |
| Environmental factors | ||||
| Light spectrum | Exposure to different light spectra (blue, red, UV-A) | Strengthening or weakening of red color | Regulation of anthocyanin biosynthesis gene expression through photomorphogenesis | ([28]; Tsormpatsidis et al. 2008) |
| Root zone temperature | Changing the temperature of the nutrient solution (low or high) | Enhanced anthocyanin accumulation at low temperature; variable effect at high temperature | Induction of stress reactions, increase in antioxidant synthesis | ([47]; Levine et al., 2023) |
| pH of the environment | Changing the acidity of the nutrient medium | Effect on antioxidant activity and possible color change | Modification of element bioavailability and enzyme activity | ([16]; Anum et al. 2024) |
| Position of the leaf on the plant (ecological) | Inner, middle or outer leaves | Change in color intensity and pigment content | Differences in light exposure and tissue age | ([28]; Tsormpatsidis et al. 2008) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




