1. Introduction
1.1. Brief Overview of MPS VII and Its Rarity
Mucopolysaccharidosis type VII (MPS VII), also known as Sly syndrome, is an ultrarare autosomal recessive lysosomal storage disorder caused by pathogenic variants in the GUSB gene, which induces a deficiency in the β-glucuronidase enzyme [
1,
2]. This enzyme deficiency promotes the progressive accumulation of glycosaminoglycans (GAGs), including chondroitin sulfate, dermatan sulfate, and heparan sulfate, in multiple tissues and organs [
3]. The prevalence of MPS VII varies from 0.02 to 0.24 per 100,000 live births worldwide [
4]. Moreover, its clinical manifestations can range from severe forms presenting with non-immune hydrops fetalis to attenuated phenotypes, with common features including coarse facial features, skeletal dysplasia, hepatosplenomegaly, cognitive impairment, and cardiopulmonary complications [
5]. Around half of the patients with severe MPS VII die before 1 year of age, highlighting the critical importance of early diagnosis and intervention [
2].
1.2. Current Treatment Options with a Focus on Enzyme Replacement Therapy (ERT)
Current treatment options for MPS VII primarily include ERT and hematopoietic stem cell transplantation (HSCT). ERT with vestronidase alfa (Mepsevii), which had been approved by the Food and Drug Administration in November 2017, represents a significant advancement in the treatment of MPS VII [
5]. Clinical studies have demonstrated ERT’s effectiveness in reducing urinary GAG levels and improving organomegaly [
6]. However, ERT has certain limitations, one of which is the inability of the enzyme to effectively cross the blood–brain barrier, making it less effective for neurological manifestations [
7]. In contrast, HSCT offers the potential advantage of enzyme delivery across the blood–brain barrier through donor-derived microglial cells but carries significant procedural risks [
8,
9]. Early initiation of treatment appears crucial for optimal outcomes, as demonstrated in clinical studies showing better response when therapy begins before extensive tissue damage occurs [
10].
1.3. Importance of Long-Term Follow-up Data
Long-term follow-up data for patients with MPS VII are crucial for several reasons. Given the exceeding rarity of this disease, with prevalence rates ranging from 0.02 to 0.24 per 100,000 live births [
4], each patient’s longitudinal data contributes significantly to our understanding of disease progression and treatment effectiveness. Extended follow-up studies have revealed varying clinical trajectories and treatment responses, which have helped establish optimal therapeutic strategies [
3]. Furthermore, long-term monitoring is essential for detecting potential late-onset complications and evaluating the sustained efficacy of treatments [
2]. Such data are particularly valuable for understanding developmental outcomes and quality of life over time, especially in patients who begin treatment early in life [
11]. Cumulative evidence from long-term follow-up studies has also proven instrumental in improving clinical guidelines and supporting the establishment of healthcare policies regarding the management of
this rare disease [
2].
1.4. Significance of the First Case in Taiwan
The documentation of the first MPS VII case in Taiwan represents a significant milestone in research and management of this rare disease in Asia. As the first documented case among Taiwan’s population of approximately 23 million [
12], it provides valuable insights into the genetic and clinical manifestations of MPS VII in the Asian population. This case contributes to the global understanding of the ethnic distribution of MPS VII and its potential genetic variations. Furthermore, as the first case of ERT managed in Taiwan, it establishes an important reference point for regional healthcare systems in treating ultrarare diseases and helps inform healthcare policies regarding orphan drugs in Asian healthcare contexts. The detailed documentation of this case also facilitates better awareness and recognition of MPS VII among healthcare providers in Taiwan and neighboring regions.
2. Case Presentation
2.1. Patient Information
2.1.1. Demographics and Initial Presentation [12]
The patient, a female born in October 2015 at 28 weeks of gestation, presented with significant prenatal complications, including hydrops fetalis requiring four intrauterine ascites aspirations. She was born via cesarean section due to fetal distress, with a birth weight of 1,934 g despite having generalized edema. She was one of twins, with the other fetus having died at 10 weeks of gestation. The initial postnatal period was complicated by respiratory difficulties requiring ventilatory support and chronic lung disease associated with prematurity. Early developmental milestones were delayed, with notable facial dysmorphism becoming progressively apparent during the first year of life. Additional early manifestations included hearing impairment, laryngomalacia, and hepatosplenomegaly, representing the classic multisystemic involvement of MPS VII.
2.1.2. Key Clinical Manifestations [12]
The patient exhibited multiple characteristic features of MPS VII. By age 4, she had developed distinctive facial dysmorphism with midface hypoplasia, a flat nasal bridge, and hypertelorism. Musculoskeletal manifestations included skeletal dysplasia with paddle-shaped ribs, dysplastic vertebral bodies, and bilateral hip dysplasia. Respiratory complications were significant, requiring BiPAP support during sleep and oxygen supplementation during daily activities. Cardiopulmonary evaluation revealed ASD II and mild valvular abnormalities. Growth parameters consistently tracked below normal, with height and weight falling below the 3rd percentile. Neurologically, she exhibited developmental delays but maintained the ability to walk independently. Ophthalmological examination revealed progressive corneal clouding, with hearing impairment persisting despite interventions.
2.1.3. Diagnostic Journey (Genetic Testing Results) [12]
The diagnostic journey of our patient began at age 4 when she was referred for the evaluation of progressive dysmorphic features and developmental delays. Initial screening revealed elevated urinary GAGs (33.68 mg/mmol creatinine; normal range <20.98 mg/mmol creatinine). Enzyme analysis showed normal activities for MPS I, II, IIIB, IVA, and VI. Next-generation sequencing subsequently identified the following compound heterozygous variants in the GUSB gene: c.104C > A (p. Ser35Ter), a known pathogenic variant, and c.1454C > T (p. Ser485Phe), a novel variant inherited from the mother. Segregation analysis confirmed the maternal inheritance of c.1454C > T and paternal inheritance of c.104C > A through grandmother testing. The diagnosis was confirmed by demonstrating severely reduced β-glucuronidase activity (<0.00 μmol/g protein/h; normal range: 255.31–681.29 μmol/g protein/h).
2.2. Clinical Course and Treatment
2.2.1. Pre-ERT Status (2015–2021) [12]
Prior to ERT initiation, the patient experienced progressive multisystemic manifestations. Her respiratory status was significantly compromised, requiring continuous oxygen supplementation (1–1.5 L/min) and BiPAP support for approximately 10 h daily. Growth parameters remained poor, with her height tracking consistently below the 3rd percentile (95 cm at age 6). Musculoskeletal involvement progressed with the development of thoracolumbar scoliosis and bilateral hip dysplasia requiring surgical intervention. Cardiac evaluation revealed ASD II with valve thickening, whereas hepatic assessment showed progressive hepatosplenomegaly with coarse liver texture on ultrasound. Regular monitoring of urinary GAG levels showed persistent elevation (DMB ratio: 33.68 mg/mmol creatinine), and enzyme activity remained undetectable. Despite these challenges, she maintained limited ambulatory ability with support but had significantly reduced exercise tolerance.
2.2.2. ERT Initiation and Regimen
ERT was initiated in June 2021 after confirmation of diagnosis and coverage approval. The patient received biweekly intravenous infusions of vestronidase alfa (Mepsevii) at the standard dose of 4 mg/kg [
10]. Before each infusion, premedication with antipyretics and antihistamines was administered according to established protocol. The initial infusions were conducted under close monitoring in the hospital setting, with infusion rates being gradually increased starting at 2.5% of the total volume for the first hour, increasing to 5% for the second hour, and then up to 10% for the remaining duration, if well tolerated, following the recommended protocol. After establishing safety and tolerability throughout the first 3 months, subsequent infusions were administered over a duration of 4 h. The treatment schedule was consistently maintained with >95% compliance over the 3-year period. The patient’s clinical features and laboratory findings before and after ERT initiation are summarized in
Table 1.
2.2.3. Three-Year Follow-up Data (2021–2024)
2.2.3.1. Improvement in Respiratory Function
Following ERT initiation, significant improvements in respiratory function were observed over the 3-year follow-up period. The most notable change was the elimination of daytime oxygen dependency after the first year of treatment. The patient’s continuous oxygen requirement of 1–1.5 L/min was gradually reduced and eventually discontinued for daytime activities. BiPAP support was optimized and required only during sleep, with the average usage decreasing from 10 h to approximately 8 h daily. Pulmonary function parameters showed steady improvement, with a reduction in the frequency of respiratory infections and episodes of sleep-disordered breathing. The patient’s exercise tolerance also improved significantly, allowing participation in daily activities without respiratory distress or oxygen desaturation. Notably, progression from trivial to mild aortic regurgitation and mild to moderate mitral regurgitation was observed over the follow-up period (
Table 2).
2.2.3.2. Growth Parameters
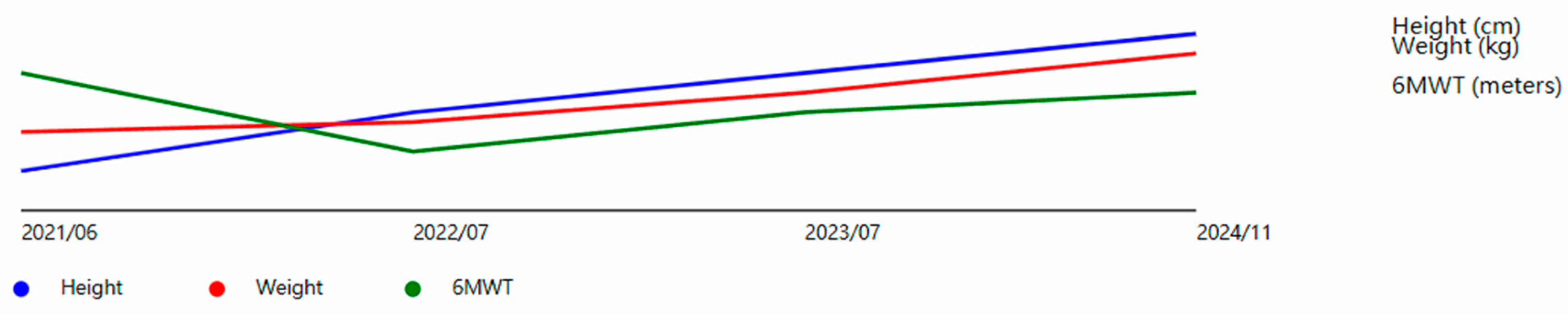
During the 3-year follow-up period, the patient demonstrated steady improvement in growth parameters. Height increased from 110 to 118 cm, despite remaining below the 3rd percentile for her age. Body weight gradually improved from 16 to 22 kg, corresponding to an increase from the 3rd to 15th percentile. Body mass index improved from 13.2 to 15.8, reflecting better nutritional status and overall health. Head circumference grew steadily from 49.1 to 50.1 cm, tracking along the 25th percentile. The annual growth velocity increased from 2.71 cm/year pre-ERT to an average of 3.5 cm/year after treatment initiation, indicating improved growth dynamics. These modest improvements suggest that ERT had a positive impact on physical development (
Figure 1).
2.2.3.4. Involvement of Other Organ Systems (Cardiac, Hepatic, Skeletal)
Over the 3-year follow-up period, the patient showed varying responses across different organ systems. Cardiac evaluation revealed persistent mild structural abnormalities, including ASD II (0.35–0.40 cm) with a left-to-right shunt, thickened interventricular septum, and mild valvular involvement (mitral and aortic valves). The ejection fraction remained stable between 53% and 77%, indicating preserved systolic function. Notably, progression from trivial to mild aortic regurgitation and mild to moderate mitral regurgitation was observed over the follow-up period.
Hepatic assessment demonstrated persistent coarse liver texture and stable hepatosplenomegaly. Liver stiffness measured using acoustic radiation force impulse fluctuated between 0.68 and 1.40 m/s, suggesting variable hepatic involvement. Spleen size remained consistently enlarged, ranging from 9.92 to 10.93 cm, with recent evaluations showing the development of tortuous splenic veins.
Skeletal manifestations remained significant, necessitating multiple surgical interventions. The patient underwent bilateral Dega acetabuloplasty using a bone bank allograft in 2021, followed by bilateral intertrochanteric shortening extension osteotomy in 2022 and corrective osteotomy of the right femur in 2024. Despite these interventions, the patient maintained ambulatory ability with support. Regular imaging revealed persistent dysostosis multiplex with bullet-shaped vertebral bodies and thoracolumbar scoliosis.
2.3. Laboratory Findings
2.3.1. Serial Urinary GAG Levels
Urinary GAG levels, a key biomarker of the therapeutic response, were monitored regularly throughout the 3-year follow-up period. At baseline (June 2020), the patient’s urinary GAG level was markedly elevated at 33.68 mg/mmol creatinine (reference: <20.98 mg/mmol creatinine). Initial liquid chromatography–tandem mass spectrometry analysis revealed elevated dermatan sulfate (DS) at 3.33 μg/mmol creatinine (reference: <1.56 μg/mmol creatinine), with other GAG species within normal ranges.
Following ERT initiation, urinary GAG levels gradually declined with some fluctuation. By September 2021, the DMB ratio decreased to 19.34 mg/mmol creatinine, though still above the age-specific reference range. The most recent measurements in October 2023 showed further stabilization at 15.21 mg/mmol creatinine, representing a 54.8% reduction from baseline. Notably, DS levels normalized over time, whereas heparan sulfate and keratan sulfate consistently remained within reference ranges throughout the treatment period.
This pattern of GAG reduction, which was particularly evident during the first year of therapy, followed by stabilization aligns with previously reported responses to vestronidase alfa in MPS VII patients. The persistent elevation above the normal range, despite significant reduction, suggests ongoing disease activity despite therapeutic control [
10,
13].
2.3.2. Measurement of Enzyme Activity
Serial measurements of β-glucuronidase enzyme activity over the 3-year treatment period demonstrated a complex response pattern to ERT. Before treatment initiation, the patient’s leukocyte β-glucuronidase activity was undetectable (<0.00 μmol/gm protein/h; reference range: 255.31–681.29 μmol/gm protein/h). After commencing ERT, enzyme activity levels gradually improved but remained significantly below the reference range.
The first significant increase was observed 3 months after treatment, with activity rising to 42.59 μmol/gm protein/h. Subsequent measurements showed fluctuating levels, ranging from 23.62 to 166.3 μmol/gm protein/h, with the highest activity having been recorded 18 months after treatment initiation. The most recent measurement in late 2023 showed stabilized activity at 57.42 μmol/gm protein/h, corresponding to approximately 22.5% of the lower reference range limit.
This pattern of partial enzyme activity restoration, characterized by significant improvement from baseline but persistent subnormal levels, is consistent with the known pharmacodynamics of biweekly ERT administration. The fluctuating enzyme levels likely reflect the timing of sampling relative to ERT infusions, as well as variability in individual responses [
14,
15].
2.3.3. Other Relevant Biochemical Markers
Regular monitoring of additional biochemical markers provided valuable insights into the multisystemic involvement and treatment responses of this disease. Liver function tests remained relatively stable throughout the 3-year follow-up period, with mild elevations in transaminases (aspartate aminotransferase, 41 IU/L and alanine aminotransferase, 31 IU/L) and gamma-GT (18 IU/L). Total bilirubin levels remained within the normal range (0.6 mg/dL), suggesting preserved hepatic synthetic function despite the underlying storage disease.
Hematological parameters were stable, with hemoglobin levels remaining within the adequate range (13.7 g/dL in recent measurements), although mild leukopenia was noted (WBC 3.9 × 103/µL). Renal function remained preserved throughout the follow-up period, with serum creatinine consistently tracking below age-appropriate reference ranges, likely reflecting the reduced muscle mass associated with MPS VII.
Cardiac biomarkers and lipid profile showed a favorable pattern, with total cholesterol (109 mg/dL) and triglycerides (62 mg/dL) remaining within normal limits. Although not disease-specific, these biochemical parameters provide important complementary information about organ function and therapeutic safety during long-term ERT administration.
3. Discussion
3.1. Clinical Significance of Improvements After ERT
The 3-year follow-up data obtained from the current case demonstrates several clinically significant improvements following ERT initiation, providing valuable insights into the effectiveness of treatments for MPS VII. The most remarkable improvement observed was that in respiratory function, with the elimination of daytime oxygen dependency representing a major therapeutic milestone. This improvement not only enhanced the patient’s quality of life but also suggested successful reduction of GAG accumulation in respiratory tissues [
16,
17].
Growth parameters showed modest but steady improvement, with height increasing by 8 cm over 3 years and BMI improving from 13.2 to 15.8. Despite remaining below the normal percentiles, these improvements represent a significant departure from the natural disease course, wherein growth typically plateaus or declines [
5]. Although modest in absolute terms, the improvement in exercise capacity, as evidenced by the increase in 6MWT distance and DSP, demonstrates meaningful functional gains in the context of this progressive disease.
Furthermore, the stabilization of cardiac and hepatic parameters, despite some progression in valve involvement, suggests that ERT may effectively slow disease progression in certain organ systems. The preservation of ambulatory function, albeit with support, represents another significant achievement, particularly considering the natural trajectory of MPS VII, wherein progressive physical deterioration is common. These improvements, while variable across different organ systems, collectively indicate that ERT can modify the disease course and potentially improve long-term outcomes in MPS VII patients [
5].
3.2. Comparison with Previously Reported Cases
The current case demonstrates both similarities and notable differences when compared to previously reported MPS VII cases treated with ERT. The initial presentation with hydrops fetalis aligns with approximately 40% of reported cases [
18]. However, our patient’s survival beyond the critical first year, despite the severe presentation, represents a more favorable outcome than that typically reported in the literature, wherein early mortality is common in severe cases [
19].
The response to ERT in our case showed patterns comparable to those reported in a phase III clinical trial of vestronidase alfa, particularly regarding urinary GAG reduction and respiratory improvement [
15]. However, our patient’s mobility improvements, while modest, appear more substantial than those reported in some adult-onset cases, possibly reflecting the advantages of earlier treatment initiation. The documented increase in the 6MWT distance from 70 to 95 m mirrors improvements reported in younger patients, though the absolute values remained lower than those reported in the pivotal trial (mean improvement 18–25 m at 24 weeks) [
15].
Notably, our patient’s cardiac manifestations progressed more slowly than those reported in untreated cases, while skeletal involvement required surgical interventions similar to those documented in other cases. The preservation of ambulatory function at age 9, albeit with support, represents a better outcome than that reported in many cases with similar initial severity. This case also provides unique insights into treatment responses among the Asian population, for whom long-term ERT data have been limited [
19].
3.3. Treatment Challenges and Decisions
Over the 3-year follow-up period, several key challenges requiring careful clinical decisions complicated the management of our patient.
First, optimizing respiratory support posed an initial challenge. Through careful titration of BiPAP settings and monitoring, daytime oxygen dependency was successfully eliminated while maintaining BiPAP support only during sleep. This adjustment significantly improved the patient’s quality of life while ensuring adequate oxygenation [
10].
Second, the management of progressive skeletal manifestations required multiple surgical interventions. The decision to proceed with bilateral Dega acetabuloplasty in 2021, followed by bilateral intertrochanteric shortening extension osteotomy in 2022 and corrective osteotomy of the right femur in 2024, was based on the need to address both functional mobility and pain management [
17]. These interventions were carefully timed between ERT infusions to optimize healing.
Third, balancing ERT dosing with the patient’s growth and clinical response required ongoing assessment. The standard dose of 4 mg/kg every 2 weeks was maintained, with careful monitoring of biomarkers, including urinary GAG levels and enzyme activity. Although urinary GAG levels showed a significant initial reduction (from 33.68 to 15.21 mg/mmol creatinine), some fluctuation in enzyme activity levels were observed (ranging from 23.62 to 166.3 μmol/gm protein/h), necessitating continued close monitoring [
13,
14].
3.4. Impact of Treatment Timing and Long-Term Therapeutic Strategy
Initiating ERT at age 6 in our patient provides important insights into the optimal timing of treatment intervention. While earlier treatment initiation might have potentially prevented some irreversible manifestations, our patient’s positive response, particularly in respiratory function and mobility, suggests that treatment initiation during early childhood can still provide meaningful benefits [
16]. This observation aligns with recent studies indicating that while early treatment is ideal, therapeutic benefits can still be realized even after some tissue damage has occurred [
20,
21].
The challenges associated with maintaining optimal therapeutic outcomes highlight the need for an integrated treatment approach. Our experience demonstrated that ERT alone, while effective for certain manifestations, required complementary interventions, including surgical procedures and supportive therapies, to achieve optimal outcomes. This multimodal approach proved particularly important in addressing skeletal manifestations and maintaining functional mobility [
22].
3.5. Biomarker Correlation and Monitoring Challenges
The relationship between biomarkers and clinical response in our case revealed interesting patterns. Although urinary GAG levels were consistently reduced, their correlation with clinical improvements varied across different organ systems. The fluctuating enzyme activity levels (23.62–166.3 μmol/gm protein/h) despite the stable clinical status suggest that current biomarkers may not fully reflect therapeutic efficacy [
23]. This observation supports recent proposals for developing new biomarkers that might better reflect tissue-specific responses to therapy [
24].
3.6. Considerations for Asian Populations and Implications for Healthcare Policy
As the first documented case of MPS VII treated with ERT in Taiwan, our experience provides valuable insights into treatment response patterns among Asian populations. The positive therapeutic response observed in our patient, particularly in terms of growth parameters and respiratory function, suggests that the efficacy profile of vestronidase alfa may be comparable across ethnic groups [
5]. However, our patient’s specific genetic variants and clinical course highlight the importance of considering population-specific factors during treatment planning [
12].
Our case also demonstrates the critical role of healthcare policy in allowing access to treatments for ultrarare diseases. The successful implementation of ERT in the current case was facilitated by Taiwan’s comprehensive rare disease policy framework, which could serve as a model for other Asian healthcare systems [
25].
4. Conclusions
This 3-year follow-up study of the first Taiwanese MPS VII patient treated with ERT demonstrated clinically meaningful improvements across multiple organ systems, with the most notable improvements being the progression from continuous oxygen dependency to requiring BiPAP support only during sleep, along with the modest but significant gains in growth parameters and physical performance. Our experience highlights three crucial aspects of MPS VII management: (1) the value of therapeutic intervention even after tissue damage has occurred, as evidenced by meaningful clinical benefits despite ERT initiation at age 6; (2) the necessity of a comprehensive, multimodal treatment approach given the variable responses across organ systems; and (3) the need for developing more refined monitoring tools based on observed biomarker patterns. Although our findings should be interpreted within the context of a single case report, this detailed longitudinal documentation of the first documented case of MPS VII treated with ERT in Taiwan provides valuable insights into treatment response patterns among Asian populations, demonstrates the feasibility of implementing ultrarare disease treatments in Asian healthcare systems under Taiwan’s comprehensive rare disease policy framework, and contributes significantly to the global understanding of long-term ERT outcomes.
Author Contributions
The manuscript was prepared by C.-L.L., with contributions in patient follow-up and manuscript preparation from S.-P.L. and H.-Y.L. Manuscript revisions were performed by C.-K.C., R.-Y.T., Y.-T.L., Y.-H.C., and H.-C.C. All authors diligently reviewed and provided their approval for the final version of the manuscript.
Funding
This study was funded by MacKay Memorial Hospital grant numbers (MMH-MM-113-13, MMH-E-113-13, MMH-MM-112-14, MMH-E-112-13, and MMH-E-111-13) and the Ministry of Science and Technology, Executive Yuan, Taiwan grant numbers (NSTC-113-2314-B-195-003, NSTC-113-2314-B-195-004, NSTC-113-2314-B-715-002, NSTC-113-2314-B-195-021, NSTC-113-2811-B-195-001, NSTC-112-2314-B-195-014-MY3, NSTC-112-2811-B-195-001, NSTC-112-2314-B-195-003, NSTC-111-2314-B-195-017, NSTC-111-2811-B-195-002, NSTC-111-2811-B-195-001, NSTC-110-2314-B-195-014, NSTC-110-2314-B-195-010-MY3, and NSTC-110-2314-B-195-029).
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Mackay Memorial Hospital Institutional Review Board (Reference number: 21MMHIS109e, approval date: 1 October 2021) and was permitted to be published. Written informed consent to have the case details and accompanying images published was obtained from the patient’s parents. All clinical investigations were conducted following the principles expressed in the Declaration of Helsinki.
Informed Consent Statement
Written informed consent to have the case details and accompanying images published was obtained from the patient’s legal guardian.
Data Availability Statement
Not applicable
Acknowledgments
We are grateful for the tireless efforts and dedication of the clinical staff and research laboratory personnel, without whom this study would not have been possible.
Conflicts of Interest
The authors declare that they have no competing interests or potential conflicts of interest in this study.
References
- Sly, W.S.; Quinton, B.A.; McAlister, W.H.; Rimoin, D.L. Beta glucuronidase deficiency: Report of clinical, radiologic, and biochemical features of a new mucopolysaccharidosis. J. Pediatr. 1973, 82, 249–257.
- Montaño, A.M.; Lock-Hock, N.; Steiner, R.D.; Graham, B.H.; Szlago, M.; Greenstein, R.; Pineda, M.; Gonzalez-Meneses, A.; Çoker, M.; Bartholomew, D.; Sands, M.S.; Wang, R.; Giugliani, R.; Macaya, A.; Pastores, G.; Ketko, A.K.; Ezgü, F.; Tanaka, A.; Arash, L.; Beck, M.; Falk, R.E.; Bhattacharya, K.; Francom J.; White, K.K.; Mitchell, G.A.; Cimbalistiene, L.; Holtz, M.; Sly, W.S. Clinical course of sly syndrome (mucopolysaccharidosis type VII). J. Med. Genet. 2016, 53, 403–418.
- Zielonka, M.; Garbade, S.F.; Kölker, S.; Hoffmann, G.F.; Ries, M. Quantitative clinical characteristics of 53 patients with MPS VII: A cross-sectional analysis. Genet. Med. 2017, 19, 983–988.
- Khan, S.A.; Peracha, H.; Ballhausen, D.; Wiesbauer, A.; Rohrbach, M.; Gautschi, M.; Mason, R.W.; Giugliani, R.; Suzuki, Y.; Orii, K.E.; Orii, T.; Tomatsu, S. Epidemiology of mucopolysaccharidoses. Mol. Genet. Metab. 2017, 121, 227–240.
- McCafferty, E.H.; Scott, L.J. Vestronidase alfa: A review in mucopolysaccharidosis VII. BioDrugs 2019, 33, 233–240.
- Fox, J.E.; Volpe, L.; Bullaro, J.; Kakkis, E.D.; Sly, W.S. First human treatment with investigational rhGUS enzyme replacement therapy in an advanced stage MPS VII patient. Mol. Genet. Metab. 2015, 114, 203–208.
- Concolino, D.; Deodato, F.; Parini, R. Enzyme replacement therapy: Efficacy and limitations. Ital. J. Pediatr. 2018, 44, 120.
- Taylor, M.; Khan, S.; Stapleton, M.; Wang, J.; Chen, J.; Wynn, R.; Yabe, H.; Chinen, Y.; Boelens, J.J.; Mason, R.W.; Kubaski, F.; Horovitz, D.D.G.; Barth, A.L.; Serafini, M.; Bernardo, M.E.; Kobayashi, H.; Orii, K.E.; Suzuki, Y.; Orii, T.; Tomatsu, S. Hematopoietic stem cell transplantation for mucopolysaccharidoses: Past, present, and future. Biol. Blood Marrow Transplant. 2019, 25, e226–e246.
- Boelens, J.J.; Prasad, V.K.; Tolar, J.; Wynn, R.F.; Peters, C. Current international perspectives on hematopoietic stem cell transplantation for inherited metabolic disorders. Pediatr. Clin. North Am. 2010, 57, 123–145.
- Harmatz, P.; Whitley, C.B.; Wang, R.Y.; Bauer, M.; Song, W.; Haller, C.; Kakkis, E. A novel Blind Start study design to investigate vestronidase alfa for mucopolysaccharidosis VII, an ultra-rare genetic disease. Mol. Genet. Metab. 2018, 123, 488–494.
- Lin, H.Y.; Lee, C.L.; Chang, C.Y.; Chiu, P.C.; Chien, Y.H.; Niu, D.M.; Tsai, F.J.; Lin, S.J.; Lin, J.L.; Chao, M.C.; Chang, T.M.; Tsai, W.H.; Wang, T.J.; Chuang, C.K.; Lin, S.P. Survival and diagnostic age of 175 Taiwanese patients with mucopolysaccharidoses (1985-2019). Orphanet J. Rare Dis. 2020, 15, 314.
- Lee, C.L.; Chuang, C.K.; Hsu, C.H.; Lin, S.P. The first mucopolysaccharidosis type VII in a Taiwanese girl: A case report and review of the literature. J. Formos. Med. Assoc. 2022, 121, 712–717.
- Khan, S.A.; Mason, R.W.; Giugliani, R.; Orii, K.; Fukao, T.; Suzuki, Y.; Yamaguchi, S.; Kobayashi, H.; Orii, T.; Tomatsu, S. Glycosaminoglycans analysis in blood and urine of patients with mucopolysaccharidosis. Mol. Genet. Metab. 2018, 125, 44–52.
- Qi, Y.; McKeever, K.; Taylor, J.; Haller, C.; Song, W.; Wang, R.Y.; Jones, S.A.; Shi J. Pharmacokinetic and pharmacodynamic modeling to optimize the dose of vestronidase alfa, an enzyme replacement therapy for treatment of patients with mucopolysaccharidosis type VII: Results from three trials. Clin. Pharmacokinet. 2019, 58, 673–683.
- Wang, R.Y.; da Silva Franco, J.F.; López-Valdez, J.; Martins, E.; Sutton, V.R.; Whitley, C.B.; Zhang, L.; Cimms, T.; Marsden, D.; Jurecka, A.; Harmatz, P. The long-term safety and efficacy of vestronidase alfa, rhGUS enzyme replacement therapy, in subjects with mucopolysaccharidosis VII. Mol. Genet. Metab. 2020, 129, 219–227.
- Sawamoto, K.; Stapleton, M.; Alméciga-Díaz, C.J.; Espejo-Mojica, A.J.; Losada, J.C.; Suarez, D.A.; Tomatsu, S. Therapeutic options for mucopolysaccharidoses: Current and emerging treatments. Drugs 2019, 79, 1103–1134.
- Oussoren, E.; Brands, M.M.; Ruijter, G.J.; der Ploeg, A.T.; Reuser, A.J. Bone, joint and tooth development in mucopolysaccharidoses: Relevance to therapeutic options. Biochim. Biophys. Acta 2011, 1812, 1542–1556.
- Giugliani, R.; Barth, A.L.; Dumas, M.R.C.; Lopez-Valdez, J.; Horovitz, D.D.G.; Gonzalez-Gutierrez-Solana, L.; Klinge, L.; Belmatoug, N.; Harmatz, P. Mucopolysaccharidosis VII in Brazil: Natural history and clinical findings. Orphanet J. Rare Dis. 2021, 16, 238.
- Grant, C.L.; López-Valdez, J.; Marsden, D.; Ezgü, F. Mucopolysaccharidosis type VII (Sly syndrome) - What do we know? Mol. Genet. Metab. 2024, 141, 108145.
- Muenzer, J. Early initiation of enzyme replacement therapy for the mucopolysaccharidoses. Mol. Genet. Metab. 2014, 111, 63–72.
- Dewalska, A.; Rombel-Bryzek, A. Clinical trials of new therapies for mucopolysaccharidoses. Med. Sci. Pulse 2024, 18, 1–11.
- Noh, H.; Lee, J.I. Current and potential therapeutic strategies for mucopolysaccharidoses. J. Clin. Pharm. Ther. 2014, 39, 215–224.
- Mandolfo, O.; Parker, H.; Bigger, B. Innate immunity in mucopolysaccharide diseases. Int. J. Mol. Sci. 2022, 23, 1999.
- Pandey, M.K. Exploring pro-inflammatory immunological mediators: Unraveling the mechanisms of neuroinflammation in lysosomal storage diseases. Biomedicines 2023, 11, 1067.
- Chung, C.C.Y.; Hong Kong Genome Project; Chu, A.T.W.; Chung, B.H.Y. Rare disease emerging as a global public health priority. Front. Public Health 2022, 10, 1028545.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).