Submitted:
13 December 2024
Posted:
17 December 2024
You are already at the latest version
Abstract
Ear reconstruction surgeries, including those addressing congenital deformities like microtia and trauma-induced damages, are relatively common. Given the demand, seeking new approaches to cartilage regeneration is crucial to improving surgical outcomes, reducing recovery times, and enhancing the quality of life for patients. The natural and cost-effective protein lactoferrin (LF) draws significant attention. It is renowned for its anti-inflammatory, antimicrobial properties, and its ability to stimulate chondrogenesis of the hyaline cartilage. This study investigates the effects of LF on the viability, proliferation, and chondrogenesis of rabbit auricular chondrocytes using both in vitro and in vivo models. In vitro, LF significantly increased the metabolic activity of chondrocytes in a dose-dependent manner, with an optimal concentration of 10 μg/ml enhancing cell prolifera-tion and metabolic activity. At this concentration, it also stimulated chondrogenesis. For in vivo studies, a rabbit ear defect model was used. Macroscopic and histological analyses revealed that LF-treated groups exhibited more mature cartilage regeneration, with higher density and organi-zation of elastic fibers compared to control. These findings suggest that LF promotes auricular chondrocyte chondrogenesis and could be beneficial for tissue engineering and clinical applications in cartilage repair.
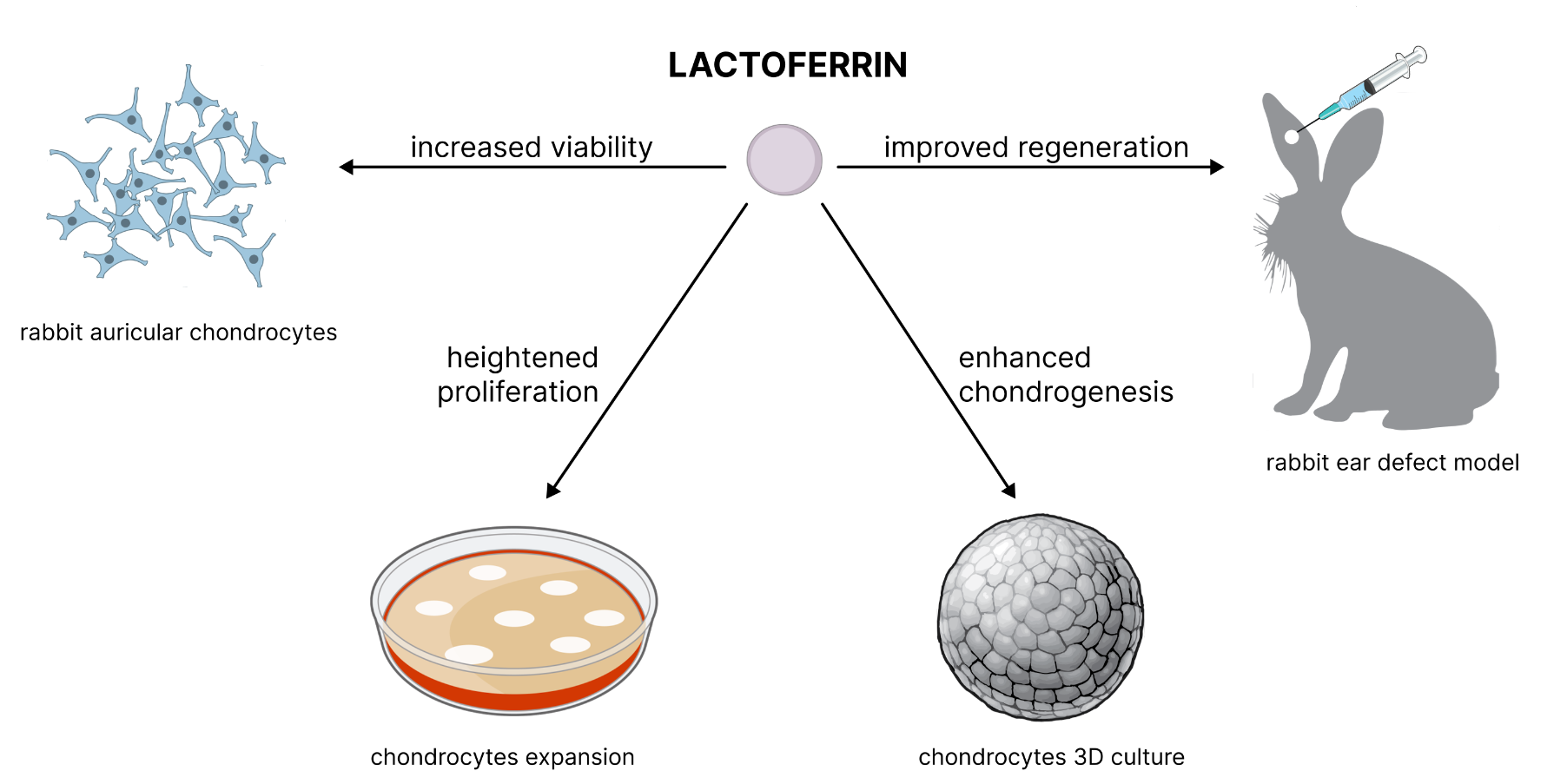
Keywords:
1. Introduction
2. Results
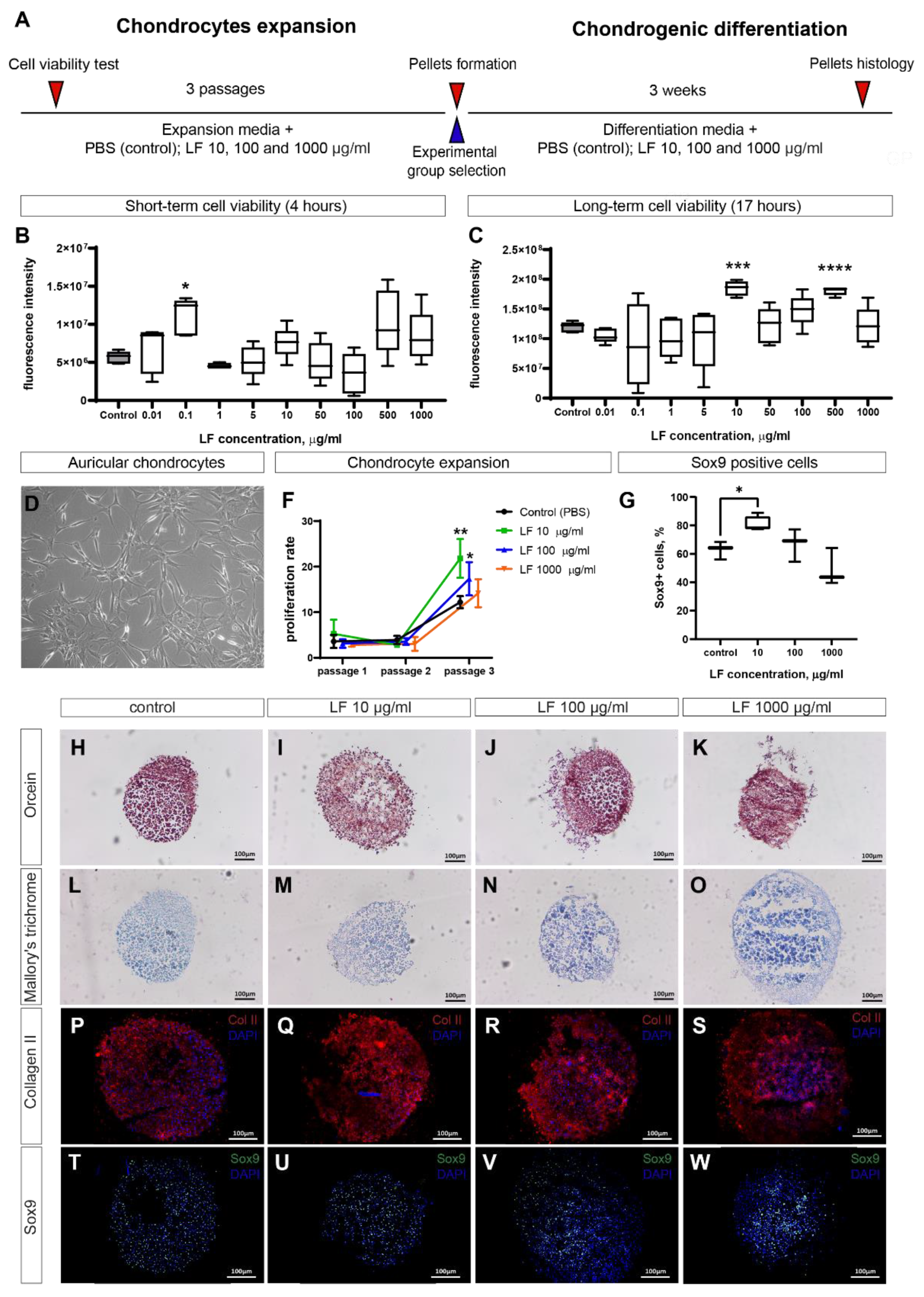
2.1. LF increases viability and proliferation of auricular chondrocytes
2.2. LF stimulates chondrogenesis in 3D pellets culture of auricular chondrocytes
2.3. LF promotes healing of cartilage defects in rabbit ear
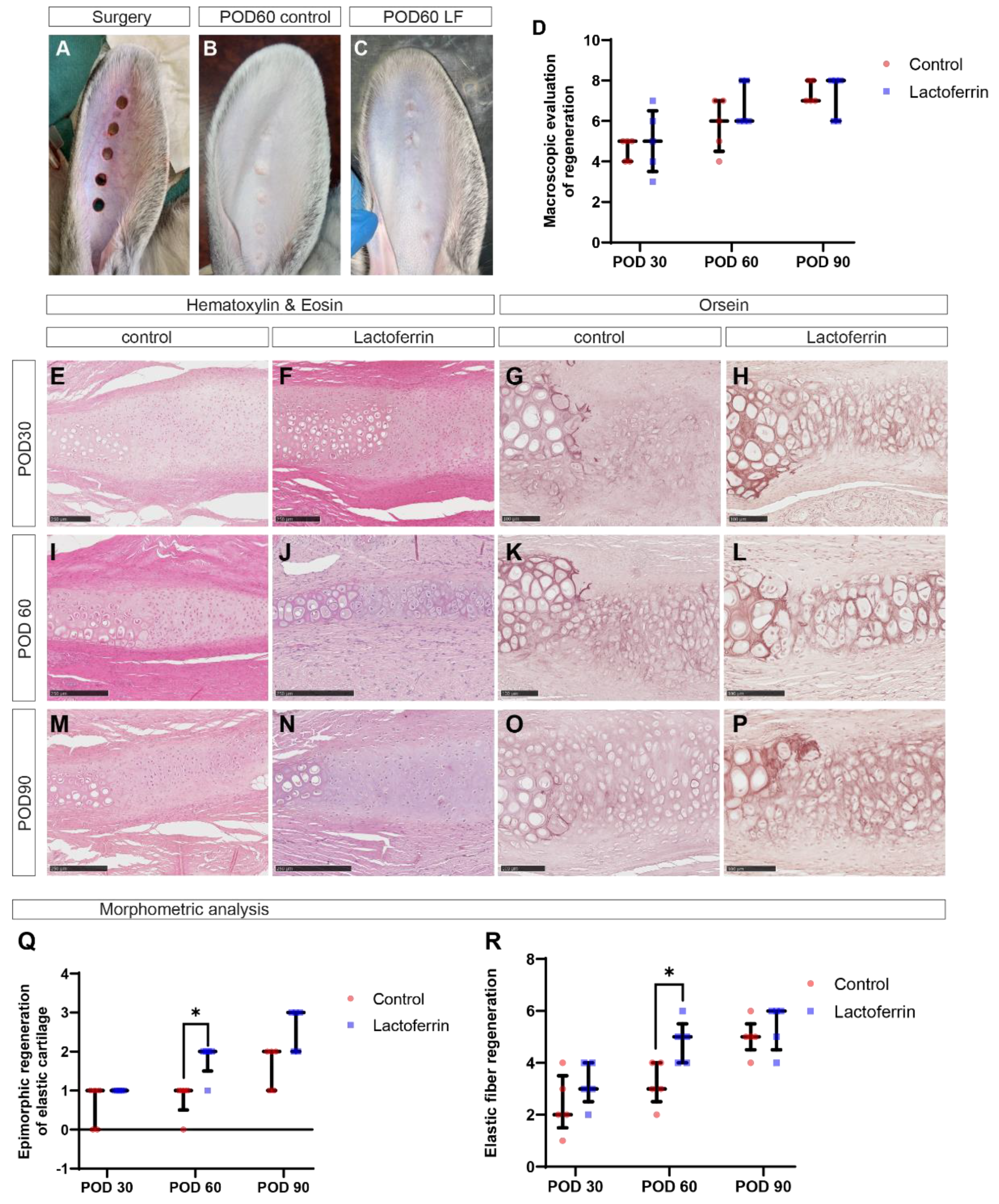
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Auricular chondrocytes isolation
4.3. Cell culture
4.4. Cell viability test
4.5. Surgery
4.6. Macroscopic evaluation
4.7. Histology
4.8. Morphometric analysis
4.9. Statistical analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sterodimas, A.; de Faria, J.; Correa, W.E.; Pitanguy, I. Tissue Engineering and Auricular Reconstruction: A Review. J. Plast. Reconstr. Aesthetic Surg. 2009, 62, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Humphries, S.; Joshi, A.; Webb, W.R.; Kanegaonkar, R. Auricular Reconstruction: Where Are We Now? A Critical Literature Review. Eur. Arch. Oto-Rhino-Laryngol. 2022, 279, 541–556. [Google Scholar] [CrossRef] [PubMed]
- Bos, E.J.; Doerga, P.; Breugem, C.C.; van Zuijlen, P.P. The Burned Ear; Possibilities and Challenges in Framework Reconstruction and Coverage. Burns 2016, 42, 1387–1395. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Zhao, H.; Wang, Y.; Bi, S.; Zhou, K.; Li, H.; Zhou, C.; Wang, Y.; Wu, W.; Peng, B.; et al. The Application and Progress of Tissue Engineering and Biomaterial Scaffolds for Total Auricular Reconstruction in Microtia. Front. Bioeng. Biotechnol. 2023, 11, 1–13. [Google Scholar] [CrossRef]
- Dai, W.; Sun, M.; Leng, X.; Hu, X.; Ao, Y. Recent Progress in 3D Printing of Elastic and High-Strength Hydrogels for the Treatment of Osteochondral and Cartilage Diseases. Front. Bioeng. Biotechnol. 2020, 8, 1–19. [Google Scholar] [CrossRef]
- Jeyaraman, M.; Jeyaraman, N.; Nallakumarasamy, A.; Ramasubramanian, S.; Yadav, S. Critical Challenges and Frontiers in Cartilage Tissue Engineering. Cureus 2024, 16, 1–9. [Google Scholar] [CrossRef]
- Antoshin, A.A.; Shpichka, A.I.; Huang, G.; Chen, K.; Lu, P.; Svistunov, A.A.; Lychagin, A. V.; Lipina, M.M.; Sinelnikov, M.Y.; Reshetov, I.V.; et al. Lactoferrin as a Regenerative Agent: The Old-New Panacea? Pharmacol. Res. 2021, 167, 105564. [Google Scholar] [CrossRef]
- Xue, H.; Tu, Y.; Ma, T.; Liu, X.; Wen, T.; Cai, M.; Xia, Z.; Mei, J. Lactoferrin Inhibits IL-1β-Induced Chondrocyte Apoptosis through AKT1-Induced CREB1 Activation. Cell. Physiol. Biochem. 2015, 36, 2456–2465. [Google Scholar] [CrossRef]
- Brandl, N.; Zemann, A.; Kaupe, I.; Marlovits, S.; Huettinger, P.; Goldenberg, H.; Huettinger, M. Signal Transduction and Metabolism in Chondrocytes Is Modulated by Lactoferrin. Osteoarthr. Cartil. 2010, 18, 117–125. [Google Scholar] [CrossRef]
- Yan, D.; Chen, D.; Shen, J.; Xiao, G.; van Wijnen, A.J.; Im, H. Bovine Lactoferricin Is Anti-inflammatory and Anti-catabolic in Human Articular Cartilage and Synovium. J. Cell. Physiol. 2013, 228, 447–456. [Google Scholar] [CrossRef]
- Rasheed, N.; Alghasham, A.; Rasheed, Z. Lactoferrin from Camelus Dromedarius Inhibits Nuclear Transcription Factor-Kappa B Activation, Cyclooxygenase-2 Expression and Prostaglandin E2 Production in Stimulated Human Chondrocytes. Pharmacogn. Res. 2016, 8, 135–141. [Google Scholar] [CrossRef]
- Yan, D.; Kc, R.; Chen, D.; Xiao, G.; Im, H.J. Bovine Lactoferricin-Induced Anti-Inflammation Is, in Part, via up-Regulation of Interleukin-11 by Secondary Activation of STAT3 in Human Articular Cartilage. J. Biol. Chem. 2013, 288, 31655–31669. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Y.; Tang, W.; Kamiya, N.; Kim, H. Lactoferrin Activates BMP7 Gene Expression through the Mitogen-Activated Protein Kinase ERK Pathway in Articular Cartilage. Biochem. Biophys. Res. Commun. 2013, 431, 31–35. [Google Scholar] [CrossRef]
- Koca, C.G.; Yıldırım, B.; Özmen, Ö.; Çiçek, M.F.; İğneci, M.; Kırarslan, Ö.; Erdil, A. Comparison of the Efficacy of Intra-Articular Injections of Hyaluronic Acid and Lactoferrin in Mono-Iodoacetate-Induced Temporomandibular Joint Osteoarthritis: A Histomorphometric, Immunohistochemistry, and Micro-Computed Tomography Analysis. Jt. Dis. Relat. Surg. 2023, 34, 166–175. [Google Scholar] [CrossRef]
- Tu, Y.; Xue, H.; Francis, W.; Davies, A.P.; Pallister, I.; Kanamarlapudi, V.; Xia, Z. Lactoferrin Inhibits Dexamethasone-Induced Chondrocyte Impairment from Osteoarthritic Cartilage through up-Regulation of Extracellular Signal-Regulated Kinase 1/2 and Suppression of FASL, FAS, and Caspase 3. Biochem. Biophys. Res. Commun. 2013, 441, 249–255. [Google Scholar] [CrossRef]
- Kim, J.; Ellman, M.B.; Yan, D.; An, H.S.; KC, R.; Li, X.; Chen, D.; Xiao, G.; Cs-Szabo, G.; Hoskin, D.W.; et al. Lactoferricin Mediates Anti-inflammatory and Anti-catabolic Effects via Inhibition of IL-1 and LPS Activity in the Intervertebral Disc. J. Cell. Physiol. 2013, 228, 1884–1896. [Google Scholar] [CrossRef]
- Ellman, M.B.; Kim, J.; An, H.S.; Chen, D.; Kc, R.; Li, X.; Xiao, G.; Yan, D.; Suh, J.; van Wjnen, A.J.; et al. Lactoferricin Enhances BMP7-Stimulated Anabolic Pathways in Intervertebral Disc Cells. Gene 2013, 524, 282–291. [Google Scholar] [CrossRef]
- Longhin, E.M.; El Yamani, N.; Rundén-Pran, E.; Dusinska, M. The Alamar Blue Assay in the Context of Safety Testing of Nanomaterials. Front. Toxicol. 2022, 4, 1–10. [Google Scholar] [CrossRef]
- Nguyen, D.N.; Jiang, P.; Stensballe, A.; Bendixen, E.; Sangild, P.T.; Chatterton, D.E.W. Bovine Lactoferrin Regulates Cell Survival, Apoptosis and Inflammation in Intestinal Epithelial Cells and Preterm Pig Intestine. J. Proteomics 2016, 139, 95–102. [Google Scholar] [CrossRef]
- Takayama, Y.; Mizumachi, K. Inhibitory Effect of Lactoferrin on Hypertrophic Differentiation of ATDC5 Mouse Chondroprogenitor Cells. BioMetals 2010, 23, 477–484. [Google Scholar] [CrossRef]
- Kruzel, M.L.; Zimecki, M.; Actor, J.K. Lactoferrin in a Context of Inflammation-Induced Pathology. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Yami, H.A.; Tahmoorespur, M.; Javadmanesh, A.; Tazarghi, A.; Sekhavati, M.H. The Immunomodulatory Effects of Lactoferrin and Its Derived Peptides on NF-ΚB Signaling Pathway: A Systematic Review and Meta-Analysis. Immunity Inflamm. Dis. 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, R.; Yadav, P.K.; Pandey, R.; Mehrotra, D. Auricular Reconstruction via 3D Bioprinting Strategies: An Update. J. Oral Biol. Craniofacial Res. 2022, 12, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Ad-El, D.D.; Selah, J.; Goshen, G.; Dano, I. Induction of Cartilage Growth in a Rabbit Ear Model: A Pilot Study. Eur. J. Plast. Surg. 2006, 28, 513–516. [Google Scholar] [CrossRef]
- Gawriluk, T.R.; Simkin, J.; Thompson, K.L.; Biswas, S.K.; Clare-Salzler, Z.; Kimani, J.M.; Kiama, S.G.; Smith, J.J.; Ezenwa, V.O.; Seifert, A.W. Comparative Analysis of Ear-Hole Closure Identifies Epimorphic Regeneration as a Discrete Trait in Mammals. Nat. Commun. 2016, 7, 1–16. [Google Scholar] [CrossRef]
- Valieva, Y.; Igrunkova, A.; Fayzullin, A.; Serejnikova, N.; Kurkov, A.; Fayzullina, N.; Valishina, D.; Bakulina, A.; Timashev, P.; Shekhter, A. Epimorphic Regeneration of Elastic Cartilage: Morphological Study into the Role of Cellular Senescence. Biology 2023, 12, 565. [Google Scholar] [CrossRef]
- ten Koppel, P. A New in Vivo Model for Testing Cartilage Grafts and Biomaterials: The “rabbit Pinna Punch-Hole” Model. Biomaterials 2001, 22, 1407–1414. [Google Scholar] [CrossRef]
- Presti, S.; Manti, S.; Parisi, G.F.; Papale, M.; Barbagallo, I.A.; Volti, G.L.; Leonardi, S. Lactoferrin: Cytokine Modulation and Application in Clinical Practice. J. Clin. Med. 2021, 10. [Google Scholar] [CrossRef]
- Sproul, E.P.; Argraves, W.S. A Cytokine Axis Regulates Elastin Formation and Degradation. Matrix Biol. 2013, 32, 86–94. [Google Scholar] [CrossRef]
- Gruden, Š.; Ulrih, N.P. Diverse Mechanisms of Antimicrobial Activities of Lactoferrins, Lactoferricins, and Other Lactoferrin-Derived Peptides. Int. J. Mol. Sci. 2021, 22. [Google Scholar] [CrossRef]
| Score | Wound Consolidation | Density | Color | Surface Texture |
|---|---|---|---|---|
| 0 | ≤25% | Entire defect dense | Red | Deformed with retractions and depressions |
| 1 | 25% - 50% | Wound edges dense | Pale | Smooth and even |
| 2 | 50% - 75% | - | Pigmented | - |
| 3 | ˃ 75% | - | Normal | - |
| Score | Morphological Features |
|---|---|
| 0 | Absence of rounded chondrocytes in regenerating fibrous cartilage |
| 1 | Presence of rounded chondrocytes in regenerating fibrous cartilage |
| 2 | Foci of regenerating elastic cartilage with large lacunae surrounded by elastic fibers |
| 3 | Continuous area of regenerating elastic cartilage with large lacunae surrounded by elastic fibers |
| Score | Morphological Features |
|---|---|
| 0 | Absence of elastic fibers |
| 1 | Isolated foci (islands) of elastic fibers |
| 2 | Elastic fibers close to the edges of the intact cartilage plate |
| 3 | Regenerating cartilage plate with elastic fibers |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




