Submitted:
16 December 2024
Posted:
16 December 2024
You are already at the latest version
Abstract
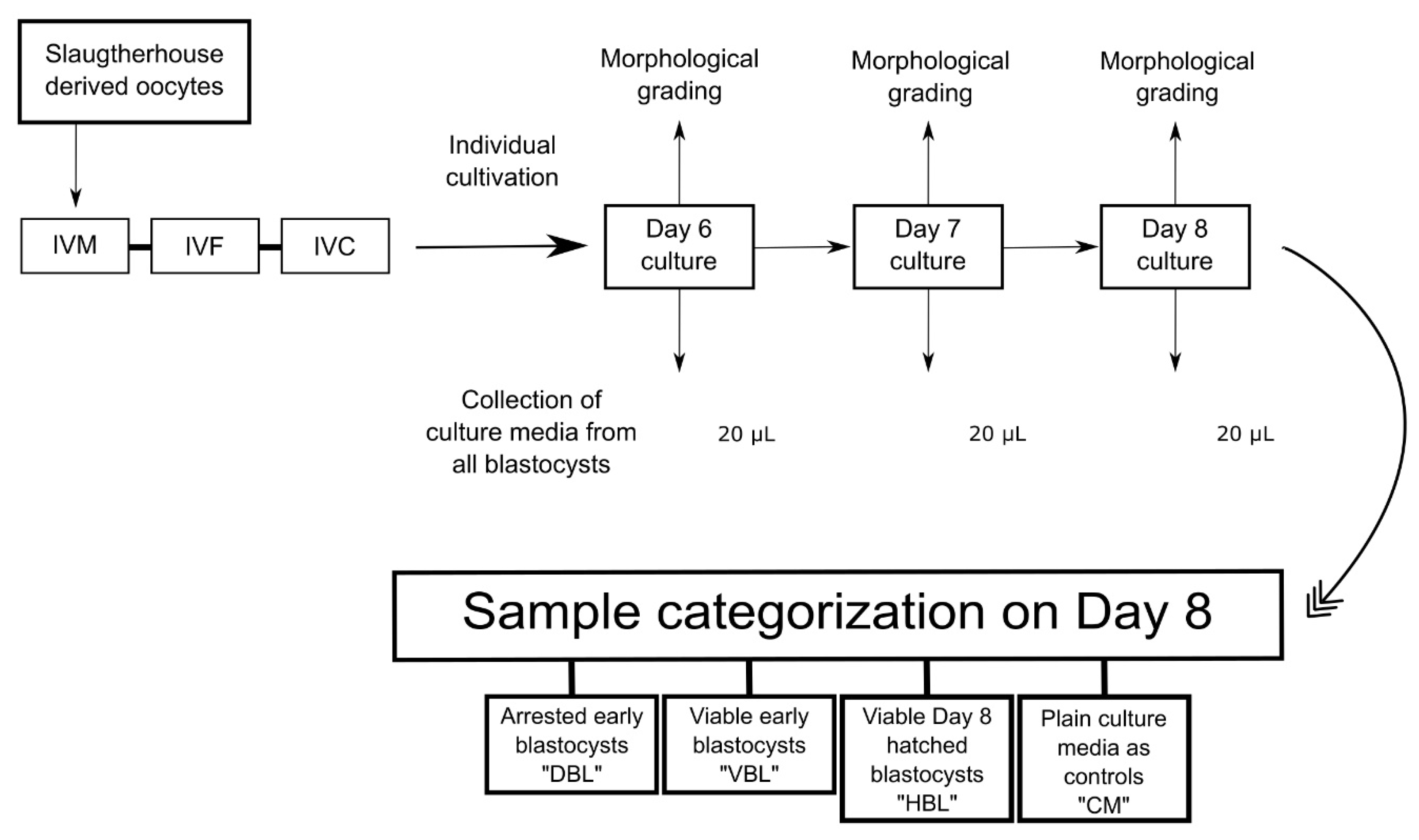
We recently demonstrated that the metabolome of single embryo culture media is associated with successful pregnancy [13]. In this study, we expanded the analysis to compare the metabolomes of viable and nonviable early-stage embryos, as well as to examine metabolomic markers associated with hatching in viable embryos. We analyzed the metabolic profile of 43 spent bovine embryo culture media samples using liquid chromatography-mass spectrometry, covering 189 metabolites, including 40 acylcarnitines, 42 amino acids/biogenic amines, 91 phospholipids, 15 sphingolipids, and the sum of hexoses. The embryos were produced from abattoir-derived oocytes, and the culture media samples were derived from Grade 1 early blastocysts that progressed to hatching (n = 10), non-viable early blastocysts that developed to the blastocyst stage but failed to hatch (n = 12), Grade 1 hatched blastocysts (n = 16), and plain growth media for control (n = 5). We found that methionine sulfoxide (Met-SO) and Lysophosphatidylcholine C24:0 concentrations were significantly lower in the culture media from viable blastocysts compared to those from non-viable blastocysts (p < 0.001). Additionally, blastocysts that resulted in successful hatching had significantly lower levels of phospholipid, arginine and methionine-related metabolites that significantly differentiated the control and viable blastocyst culture media from the media containing non-viable embryos. Building on previous studies, there appears to be an overlap in metabolites released during hatching that are also associated with successful pregnancy. The identified biomarkers can aid in assessing an embryo's developmental potential and enhance embryo selection for transfer or cryopreservation.
Keywords:
1. Introduction
2. Results
2.1. Metabolites Differing in Culture Media of Viable and Non-Viable Blastocysts
2.2. Metabolite Sums and Ratios Differing Between Culture Media of Viable and Non-viable Blastocysts
2.3. Metabolites Differing between the Culture Media of Viable Early and Hatched Blastocysts
3. Discussion
3.1. Lipid Metabolism
3.2. Monosaccharide Metabolism
3.3. Amino Acid and Derivative Metabolism
3.4. Polyamine Metabolism
4.5. Future Research Directions
4. Materials and Methods
4.1. Media
4.2. Experimental Design
4.3. Oocyte Collection and In vitro Maturation
4.4. In vitro Fertilization and Cultivation
4.5. Collection of Media for LC-MS/MS and Categorization of Samples
4.6. Preparation of Culture Media Samples for LC-MS/MS and Spectrometry
4.7. Statistical Analysis
5. Conclusions
Supplementary material
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Rabel, R.A.C.; Marchioretto, P. V.; Bangert, E.A.; Wilson, K.; Milner, D.J.; Wheeler, M.B. Pre-Implantation Bovine Embryo Evaluation—From Optics to Omics and Beyond. Animals 2023, 13. [Google Scholar] [CrossRef]
- Gimeno, I.; García-Manrique, P.; Carrocera, S.; López-Hidalgo, C.; Valledor, L.; Martín-González, D.; Gómez, E. The Metabolic Signature of in Vitro Produced Bovine Embryos Helps Predict Pregnancy and Birth after Embryo Transfer. Metabolites 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Viana, JHM. 2022 Statistics of Embryo Production and Transfer in Domestic Farm Animals. Embryo Technol. Newsl. 2023 41:4.
- Gómez, E.; Carrocera, S.; Martín, D.; Pérez-Jánez, J.J.; Prendes, J.; Prendes, J.M.; Vázquez, A.; Murillo, A.; Gimeno, I.; Muñoz, M. Efficient One-Step Direct Transfer to Recipients of Thawed Bovine Embryos Cultured in Vitro and Frozen in Chemically Defined Medium. Theriogenology 2020, 146, 39–47. [Google Scholar] [CrossRef] [PubMed]
- De Vries, A.; Kaniyamattam, K. A Review of Simulation Analyses of Economics and Genetics for the Use of In-Vitro Produced Embryos and Artificial Insemination in Dairy Herds. Anim Reprod 2020, 17. [Google Scholar] [CrossRef] [PubMed]
- Magata, F. Time-Lapse Monitoring Technologies for the Selection of Bovine in Vitro Fertilized Embryos with High Implantation Potential. J Reprod Dev 2023, 69(2), 57–64. [Google Scholar] [CrossRef] [PubMed]
- Imakawa, K.; Matsuno, Y.; Fujiwara, H. New Roles for EVs, MiRNA and LncRNA in Bovine Embryo Implantation. Front Vet Sci 2022, 9. [Google Scholar] [CrossRef] [PubMed]
- Pallisco, R.; Lazzarino, G.; Bilotta, G.; Marroni, F.; Mangione, R.; Saab, M.W.; Brundo, M.V.; Pittalà, A.; Caruso, G.; Capoccia, E.; et al. Metabolic Signature of Energy Metabolism Alterations and Excess Nitric Oxide Production in Culture Media Correlate with Low Human Embryo Quality and Unsuccessful Pregnancy. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Lechniak, D.; Sell-Kubiak, E.; Warzych, E. The Metabolic Profile of Bovine Blastocysts Is Affected by in Vitro Culture System and the Pattern of First Zygotic Cleavage. Theriogenology 2022, 188, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Cheredath, A.; Uppangala, S.; Asha, C.S.; Jijo, A.; Vani Lakshmi, R.; Kumar, P.; Joseph, D.; Nagana, N.G.; Kalthur, G.; Adiga, S.K. Combining Machine Learning with Metabolomic and Embryologic Data Improves Embryo Implantation Prediction. Reproductive Sciences 2023, 30, 984–994. [Google Scholar] [CrossRef]
- Gomez, E.; Canela, N.; Herrero, P.; Cereto, A.; Gimeno, I.; Carrocera, S.; Martin-gonzalez, D.; Murillo, A.; Muñoz, M. Metabolites Secreted by Bovine Embryos in Vitro Predict Pregnancies That the Recipient Plasma Metabolome Cannot, and Vice Versa. Metabolites 2021, 11. [Google Scholar] [CrossRef]
- Lipinska, P.; Pawlak, P.; Warzych, E. Species and Embryo Genome Origin Affect Lipid Droplets in Preimplantation Embryos. Front Cell Dev Biol 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Tsopp, E.; Kilk, K.; Taalberg, E.; Pärn, P.; Viljaste-Seera, A.; Kavak, A.; Jaakma, Ü. Associations of the Single Bovine Embryo Growth Media Metabolome with Successful Pregnancy. Metabolites 2024. [Google Scholar] [CrossRef]
- Cabello-Pinedo, S.; Abdulla, H.; Mas, S.; Fraire, A.; Maroto, B.; Seth-Smith, M.; Escriba, M.; Teruel, J.; Crespo, J.; Munné, S.; et al. Development of a Novel Non-Invasive Metabolomics Assay to Predict Implantation Potential of Human Embryos. Reproductive Sciences 2024. [Google Scholar] [CrossRef] [PubMed]
- Melo-Sterza, F. de A.; Poehland, R. Lipid Metabolism in Bovine Oocytes and Early Embryos under in Vivo, in Vitro, and Stress Conditions. Int J Mol Sci 2021, 22. [Google Scholar]
- Dambrova, M.; Makrecka-Kuka, M.; Kuka, J.; Vilskersts, R.; Nordberg, D.; Attwood, M.M.; Smesny, S.; Sen, Z.D.; Guo, A.C.; Oler, E.; et al. Acylcarnitines: Nomenclature, Biomarkers, Therapeutic Potential, Drug Targets, and Clinical Trials. Pharmacol Rev 2022, 74, 506–551. [Google Scholar] [CrossRef] [PubMed]
- Dunning, K.R.; Cashman, K.; Russell, D.L.; Thompson, J.G.; Norman, R.J.; Robker, R.L. Beta-Oxidation Is Essential for Mouse Oocyte Developmental Competence and Early Embryo Development. Biol Reprod 2010, 83, 909–918. [Google Scholar] [CrossRef] [PubMed]
- Taghizadeh, H.; Emamgholipour, S.; Hosseinkhani, S.; Arjmand, B.; Rezaei, N.; Dilmaghani-Marand, A.; Ghasemi, E.; Panahi, N.; Dehghanbanadaki, H.; Ghodssi-Ghassemabadi, R.; et al. The Association between Acylcarnitine and Amino Acids Profile and Metabolic Syndrome and Its Components in Iranian Adults: Data from STEPs 2016. Front Endocrinol (Lausanne) 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zeng, X.; Ren, M.; Mao, X.; Qiao, S. Novel Metabolic and Physiological Functions of Branched Chain Amino Acids: A Review. J Anim Sci Biotechnol 2017, 8. [Google Scholar] [CrossRef]
- Wongsrikeao, P.; Otoi, T.; Taniguchi, M.; Karja, N.W.K.; Agung, B.; Nii, M.; Nagai, T. Effects of Hexoses on in Vitro Oocyte Maturation and Embryo Development in Pigs. Theriogenology 2006, 65, 332–343. [Google Scholar] [CrossRef]
- Hufnagel, A.; Grant, I.D.; Aiken, C.E.M. Glucose and Oxygen in the Early Intrauterine Environment and Their Role in Developmental Abnormalities. Semin Cell Dev Biol 2022, 131, 25–34. [Google Scholar] [CrossRef]
- Ferrick, L.; Lee, Y.S.L.; Gardner, D.K. Metabolic Activity of Human Blastocysts Correlates with Their Morphokinetics, Morphological Grade, KIDScore and Artificial Intelligence Ranking. Human Reproduction 2020, 35, 2004–2016. [Google Scholar] [CrossRef]
- Cai, S.; Ye, Q.; Zeng, X.; Yang, G.; Ye, C.; Chen, M.; Yu, H.; Wang, Y.; Wang, G.; Huang, S.; et al. CBS and MAT2A Improve Methionine-Mediated DNA Synthesis through SAMTOR/MTORC1/S6K1/CAD Pathway during Embryo Implantation. Cell Prolif 2021, 54. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Kang, J.; Su, J.; Zhang, J.; Zhang, L.; Liu, X.; Zhang, J.; Wang, F.; Lu, Z.; Xing, X.; et al. Methionine Adenosyltransferase 2A Regulates Mouse Zygotic Genome Activation and Morula to Blastocyst Transition. Biol Reprod 2019, 100, 601–617. [Google Scholar] [CrossRef]
- Hugentobler, S.A.; Diskin, M.G.; Leese, H.J.; Humpherson, P.G.; Watson, T.; Sreenan, J.M.; Morris, D.G. Amino Acids in Oviduct and Uterine Fluid and Blood Plasma during the Estrous Cycle in the Bovine. Mol Reprod Dev 2007, 74, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Drazic, A.; Winter, J. The Physiological Role of Reversible Methionine Oxidation. Biochim Biophys Acta Proteins Proteom 2014, 1844, 1367–1382. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Kim, G.; Liu, C.; Levine, R.L. Transgenic Mice Overexpressing Methionine Sulfoxide Reductase A: Characterization of Embryonic Fibroblasts. Free Radic Biol Med 2010, 49, 641–648. [Google Scholar] [CrossRef]
- Gouge, R.C.; Marshburn, P.; Gordon, B.E.; Nunley, W.; Huet-Hudson, Y.M. Nitric Oxide as a Regulator of Embryonic Development. Biol Reprod 1998, 58, 875–9. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.-H.; Wu, M.-Y.; Chen, M.-J.; Chao, K.-H.; Ho, H.-N.; Yang, Y.-S. Nitric Oxide Is Associated with Poor Embryo Quality and Pregnancy Outcome in in Vitro Fertilization Cycles. Fertil Steril. 2004, 82(1), 126–31. [Google Scholar] [CrossRef] [PubMed]
- Tranguch, S.; Steuerwald, N.; Huet-Hudson, Y.M. Nitric Oxide Synthase Production and Nitric Oxide Regulation of Preimplantation Embryo Development. Biol Reprod 2003, 68, 1538–1544. [Google Scholar] [CrossRef]
- Christophorou, M.A.; Castelo-Branco, G.; Halley-Stott, R.P.; Oliveira, C.S.; Loos, R.; Radzisheuskaya, A.; Mowen, K.A.; Bertone, P.; Silva, J.C.R.; Zernicka-Goetz, M.; et al. Citrullination Regulates Pluripotency and Histone H1 Binding to Chromatin. Nature 2014, 507, 104–108. [Google Scholar] [CrossRef]
- Read, C.C.; Edwards, L.; Schrick, N.; Rhinehart, J.D.; Payton, R.R.; Campagna, S.R.; Castro, H.F.; Klabnik, J.L.; Horn, E.J.; Moorey, S.E. Correlation between Pre-Ovulatory Follicle Diameter and Follicular Fluid Metabolome Profiles in Lactating Beef Cows. Metabolites 2021, 11. [Google Scholar] [CrossRef]
- Bovo, S.; Mazzoni, G.; Galimberti, G.; Calò, D.G.; Fanelli, F.; Mezzullo, M.; Schiavo, G.; Manisi, A.; Trevisi, P.; Bosi, P.; et al. Metabolomics Evidences Plasma and Serum Biomarkers Differentiating Two Heavy Pig Breeds. Animal 2016, 10, 1741–1748. [Google Scholar] [CrossRef] [PubMed]
- Muccini, A.M.; Tran, N.T.; de Guingand, D.L.; Philip, M.; Gatta, P.A.D.; Galinsky, R.; Sherman, L.S.; Kelleher, M.A.; Palmer, K.R.; Berry, M.J.; et al. Creatine Metabolism in Female Reproduction, Pregnancy and Newborn Health. Nutrients 2021, 13, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Lenis, Y.Y.; Johnson, G.A.; Wang, X.; Tang, W.W.; Dunlap, K.A.; Satterfield, M.C.; Wu, G.; Hansen, T.R.; Bazer, F.W. Functional Roles of Ornithine Decarboxylase and Arginine Decarboxylase during the Peri-Implantation Period of Pregnancy in Sheep. J Anim Sci Biotechnol 2018, 9. [Google Scholar] [CrossRef]
- Aguila, L.; Treulen, F.; Therrien, J.; Felmer, R.; Valdivia, M.; Smith, L.C. Oocyte Selection for in Vitro Embryo Production in Bovine Species: Noninvasive Approaches for New Challenges of Oocyte Competence. Animals 2020, 10, 1–24. [Google Scholar] [CrossRef]
- Abraham, M.C.; Gustafsson, H.; Ruete, A.; Brandt, Y.C. Breed Influences on in Vitro Development of Abattoir-Derived Bovine Oocytes. Acta Vet Scand 2012, 54, 36. [Google Scholar] [CrossRef]
- Merton, J.S.; Vermeulen, Z.L.; Otter, T.; Mullaart, E.; de Ruigh, L.; Hasler, J.F. Carbon-Activated Gas Filtration during in Vitro Culture Increased Pregnancy Rate Following Transfer of in Vitro-Produced Bovine Embryos. Theriogenology 2007, 67, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Farin, P.W.; Farin, C.E. Transfer of Bovine Embryos Produced In Vivo or In Vitro: Survival and Fetal Development. Biol Reprod 1995, 52, 676–82. [Google Scholar] [CrossRef] [PubMed]
- Yagi, A.; Miyanaga, S.; Shrestha, R.; Takeda, S.; Kobayashi, S.; Chiba, H.; Kamiya, H.; Hui, S.P. A Fatty Acid Profiling Method Using Liquid Chromatography-High Resolution Mass Spectrometry for Improvement of Assisted Reproductive Technology. Clinica Chimica Acta 2016, 456, 100–106. [Google Scholar] [CrossRef]
| Metabolites | Concentrations in culture media | ||||
|---|---|---|---|---|---|
| Metabolite | DBL | VBL | HBL | CM | p-Value |
| Met-SO (µM) | 0.58 (0.45-4.68) a | 0.09 (0.059-0.144) b | 0.34 (0.17-0.53) a | 0.22 (0.14-0.62) ab | 0.00012 |
| LysoPC a C24:0 (nM) | 86 ± 32 a | 45 ± 13 b | 70 ± 20 a | 83 ± 5 a | 0.00075 † |
| LysoPC a C20:4 (nM) | 30 ± 11 ab | 18 ± 10 a | 36 ± 17 b | 15 ± 2 a | 0.0021 † |
| Cit | 5 (41.67%) a | 0 (0%) b | 0 (0%) b | 0 (0%) b | 0.0022 # |
| PC.aa.C36.5 (nM) | 3.2 (2-7) a | 1.7 (1.3-2.0) b | 1.3 (0.9-2.0) ab | 2.0 (1.0-2.0) a | 0.00242 |
| Ac-Orn (µM) | 0.18 (0.14-0.87) a | 0.12 (0.11-0.12) b | 0.13 (0.11-0.19) ab | 0.13 (0.12-0.19) ab | 0.00437 |
| PC aa C42:4 (nM) | 4 ± 2 a | 2 ± 1 b | 3 ± 1 ab | 2 ± 1 ab | 0.0062 † |
| Putrescine | 2 (16.67 %) a | 0 (0 %) a | 9 (56.25 %) b | 3 (60 %) a | 0.0073 # |
| Spermine | 0 (0 %) a | 0 (0 %) a | 6 (37.5 %) b | 0 (0 %) a | 0.0082 # |
| Spermidine | 4 (33.33 %) ab | 0 (0 %) a | 9 (56.25 %) b | 0 (0 %) a | 0.0085 # |
| ADMA, Creatinine, Serotonin, t4-OH-Pro, Taurine | 4 (33.33 %) a | 0 (0 %) b | 0 (0 %) b | 0 (0 %) b | 0.0098 # |
| C2 (µM) | 0.27 (±0.08) a | 0.195 (±0.024) b | 0.23 (±0.034) ab | 0.23 (±0.017) ab | 0.011 † |
| C3 (nM) | 49 (40-130) a | 41 (36-43) b | 50 (44-58) a | 54 (45-57) ab | 0.013 |
| C5 (nM) | 60 ±23 a | 45 ± 7 b | 45 ± 6 b | 42 ± 2 ab | 0.015 † |
| PC aa C34:1 (nM) | 23 (15-96 ) a | 14 (13-15) ab | 17 (13-18) ab | 19 (15-21) ab | 0.015 |
| PC aa C36:3 (nM) | 7.2 (5.4-9.5) a | 4.7 (4.0-5.6) b | 5.0 (3.7-5.6) ab | 4.1 (4.0-5.2) a | 0.017 |
| PC ae C42:0 (nM) | 215 ± 12 ab | 226 ± 11 a | 211 ± 15 b | 223 ± 6 ab | 0.027 † |
| PC ae C40:2 (nM) | 2.7 (2.0-19.5) a | 1.8 (1.4-2.3) b | 2.0 (1.8-3.0) b | 3.0 (3.0-3.0) ab | 0.027 |
| His (µM) | 53.4 (51.3-55.7) a | 44.7 (40.6-49.0) b | 50.4 (45.2-54.4) ab | 49.5 (48.5-54.7) ab | 0.028 |
| PC ae C32:2 (nM) | 15 ± 8 a | 10 ± 3 b | 11 ± 1 ab | 10 ± 1 ab | 0.028 † |
| lysoPC a C26:0 (nM) | 23 ± 10 a | 26 ± 9 a | 17 ± 7 b | 18 ± 4 ab | 0.029 † |
| PC aa C32:1 (nM) | 9.0 (7.1-61) a | 5.9 (5.0-6.9) b | 6.7 (4.5-9.3) ab | 6.3 (6.0-6.4) ab | 0.029 |
| PC aa C38:3 (nM) | 4 (1-240) a | 1.1 (0.9-1.4) b | 1.7 (1.0-4.0) ab | 1.2 (1.0-1.4) ab | 0.0303 |
| PC ae C38:2 (nM) | 4.4 (3.0-24) a | 2.1 (1.5-2.8) b | 3.6 (2.7-5.3) ab | 4.0 (3.5-4.5) ab | 0.0374 |
| PC ae C44:3 (nM) | 10 (9-11) a | 13 (11-22) b | 13 (10-14) ab | 11 (10-17) ab | 0.0381 |
| PC ae C38:1 (nM) | 6.0 (3.8-27) a | 2.8 (2.4-3.4) b | 3.9 (3.0-5.0) ab | 3.0 (2.8-3.8) ab | 0.0423 |
| PC aa C36:2 (nM) | 58 (49-338) a | 48 (45-50) b | 50 (47-54) ab | 55 (54-57) ab | 0.0453 |
| Metabolites | Concentrations in culture media, μM | ||||
|---|---|---|---|---|---|
| Metabolite | DBL | VBL | HBL | CM | p-Value |
| Met-SO/Met | 0.012 (0.009-0.09) a |
0.0016 (0.001-0.003) b |
0.006 (0.003-0.011) a |
0.004 (0.003-0.011) ab |
6.3 × 10−5 |
| Total SM-OH/SM-non-OH | 0.33±0.16 a | 0.54±0.11 b | 0.43±0.13 ab | 0.57±0.044 b | 0.001† |
| C2/C0 | 0.14 (0.13-0.16) a | 0.14 (0.13-0.15) a | 0.16 (0.14-0.18) b | 0.16 (0.16-0.17) b | 0.00448 |
| MUFA PC/SFA PC | 0.37 (0.33-2.14) a | 0.32 (0.31-0.33) b | 0.32 (0.29-0.35) ab | 0.34 (0.31-0.38) a | 0.00602 |
| Total PC ae | 0.82 (0.80-1.48) ab | 0.82 (0.81-0.84) a | 0.79 (0.78-0.81) b | 0.81 (0.80-0.84) b | 0.0122 |
| Hexoses | 705 (670-830) a | 891 (838-946) b | 830 (777-879) ab | 816 (781-821) ab | 0.0221 |
| SFA PC | 0.80 (0.76-1.05) ab | 0.81 (0.79-0.82) a | 0.78(0.76-0.79) b | 0.80 (0.79-0.81) ab | 0.0313 |
| PUFA PC/SFA PC | 0.91 (0.86-2.44) a |
0.84 (0.82-0.86) b |
0.87 (0.84-0.91) a |
0.88 (0.84-0.89) a |
0.0389 |
| Total AC-DC/Total AC | 0.188 (±0.019) a |
0.202 (±0.006) b |
0.192 (±0.009) a |
0.198 (±0.006) a |
0.041† |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




