Submitted:
11 December 2024
Posted:
12 December 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
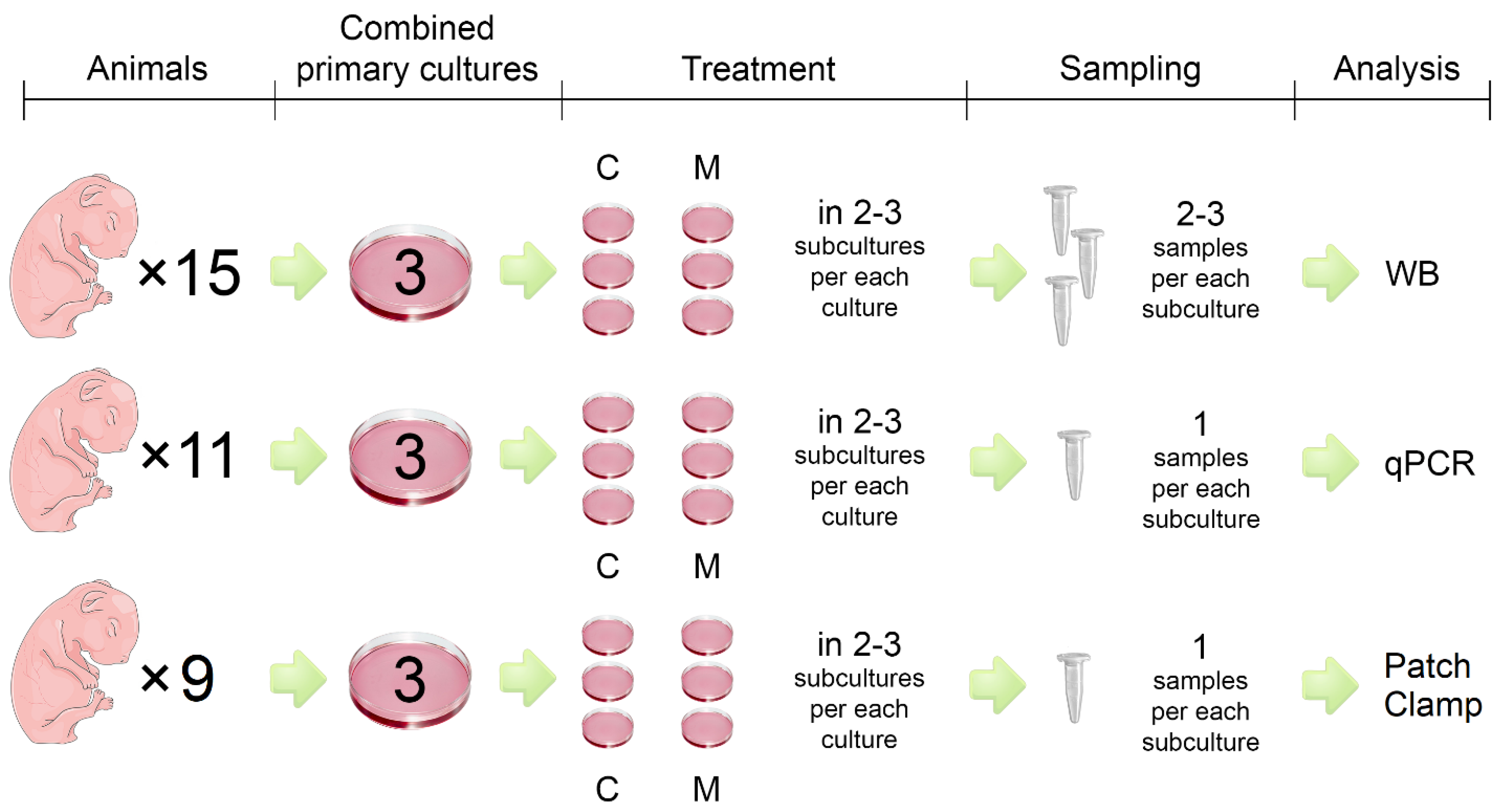
General
Isolation and Cultures of Neonatal Ventricular Cell
PCR Analysis
Western Blotting
Patch-Clamp Electrophysiological Recording
Statistical Analysis
Results
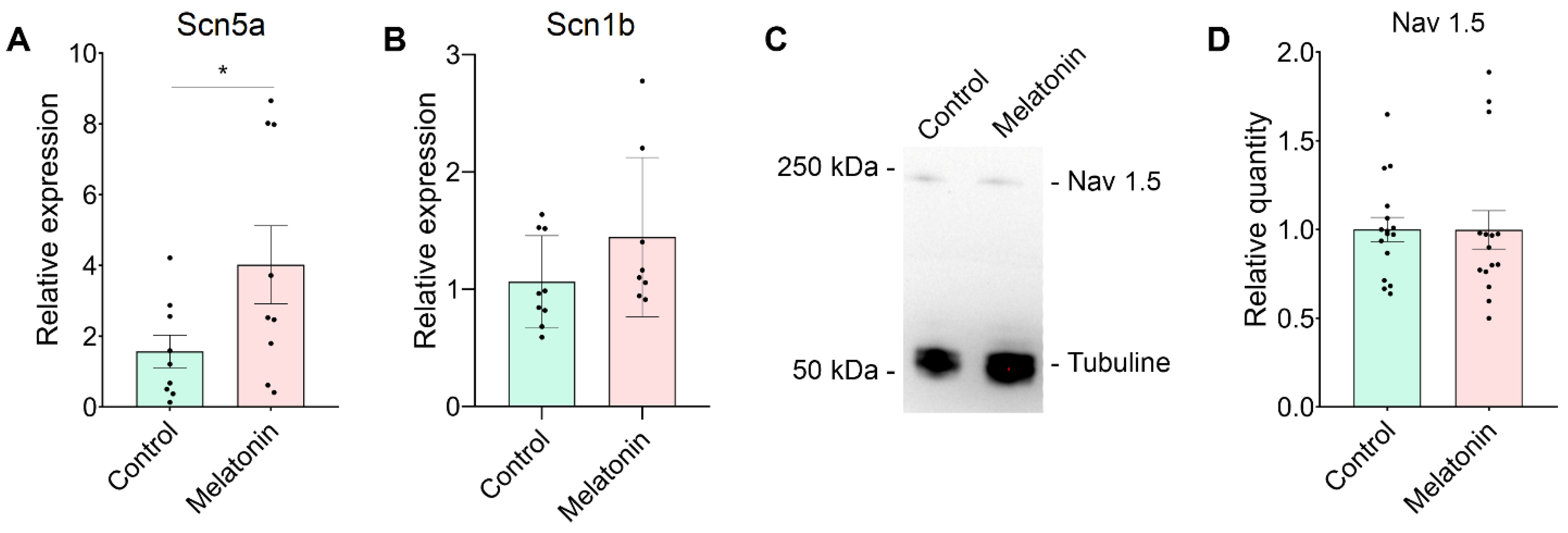
Expression of Scn5a mRNA and NaV1.5 Protein in Neonatal Rat Cardiomyocytes After Melatonin Treatment
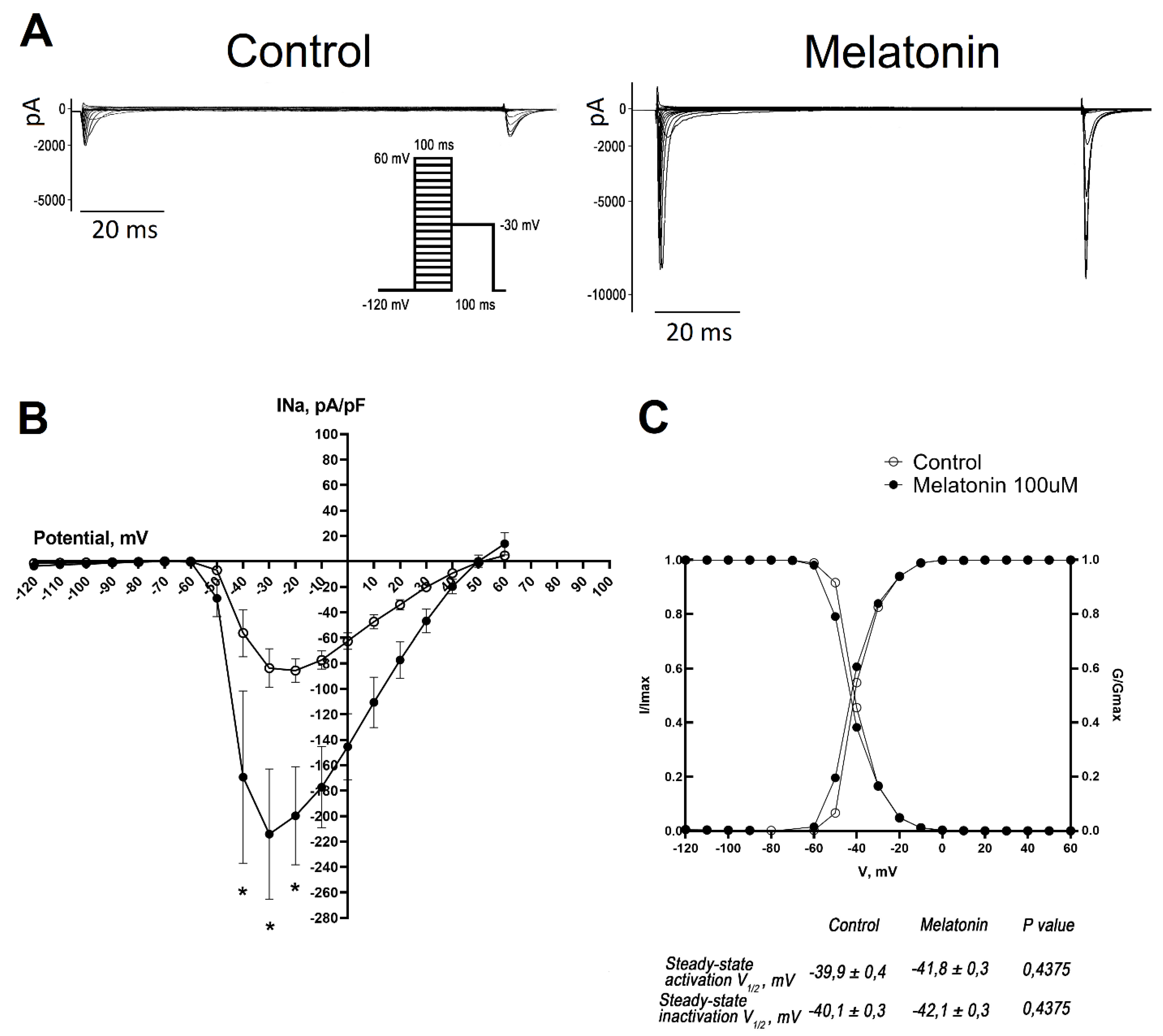
Melatonin Increases the Amplitude of INa Current in Cultured Neonatal Rat Cardiomyocytes
Discussion
Conclusion
Authors’ Contribution
Funding
References
- Holz GG 4th, Rane SG, Dunlap K. GTP-binding proteins mediate transmitter inhibition of voltage-dependent calcium channels. Nature 1986, 319, 670–672. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hille B, Dickson E, Kruse M, Falkenburger B. Dynamic metabolic control of an ion channel. Prog Mol Biol Transl Sci. 2014, 123, 219–247. [Google Scholar] [CrossRef] [PubMed]
- Mattheisen GB, Tsintsadze T, Smith SM. Strong G-Protein-Mediated Inhibition of Sodium Channels. Cell Rep. 2018, 23, 2770–2781. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Durkina AV, Bernikova OG, Gonotkov MA, Mikhaleva NJ, Sedova KA, Malykhina IA, Kuzmin VS, Velegzhaninov IO, Azarov JE. Melatonin treatment improves ventricular conduction via upregulation of Nav1.5 channel proteins and sodium current in the normal rat heart. J Pineal Res. 2022, 73, e12798. [Google Scholar] [CrossRef] [PubMed]
- Sedova KA, Bernikova OG, Cuprova JI, Ivanova AD, Kutaeva GA, Pliss MG, Lopatina EV, Vaykshnorayte MA, Diez ER, Azarov JE. Association Between Antiarrhythmic, Electrophysiological, and Antioxidative Effects of Melatonin in Ischemia/Reperfusion. Int J Mol Sci. 2019, 20, 6331. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tsvetkova AS, Bernikova OG, Mikhaleva NJ, Khramova DS, Ovechkin AO, Demidova MM, Platonov PG, Azarov JE. Melatonin Prevents Early but Not Delayed Ventricular Fibrillation in the Experimental Porcine Model of Acute Ischemia. Int J Mol Sci. 2020, 22, 328. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Durkina AV, Szeiffova Bacova B, Bernikova OG, Gonotkov MA, Sedova KA, Cuprova J, Vaykshnorayte MA, Diez ER, Prado NJ, Azarov JE. Blockade of Melatonin Receptors Abolishes Its Antiarrhythmic Effect and Slows Ventricular Conduction in Rat Hearts. Int J Mol Sci. 2023, 24, 11931. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Han D, Wang Y, Chen J, Zhang J, Yu P, Zhang R, Li S, Tao B, Wang Y, Qiu Y, Xu M, Gao E, Cao F. Activation of melatonin receptor 2 but not melatonin receptor 1 mediates melatonin-conferred cardioprotection against myocardial ischemia/reperfusion injury. J Pineal Res. 2019, 67, e12571. [Google Scholar] [CrossRef] [PubMed]
- Pereira AHM, Cardoso AC, Franchini KG. Isolation, culture, and immunostaining of neonatal rat ventricular myocytes. STAR Protoc. 2021, 2, 100950. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nguyen PD, Hsiao ST, Sivakumaran P, Lim SY, Dilley RJ. Enrichment of neonatal rat cardiomyocytes in primary culture facilitates long-term maintenance of contractility in vitro. Am J Physiol Cell Physiol. 2012, 303, C1220–C1228. [Google Scholar] [CrossRef] [PubMed]
- Li C, Rong X, Qin J, Wang S. An improved two-step method for extraction and purification of primary cardiomyocytes from neonatal mice. J Pharmacol Toxicol Methods. 2020, 104, 106887. [Google Scholar] [CrossRef] [PubMed]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Karmažínová M, Lacinová L. Measurement of cellular excitability by whole cell patch clamp technique. Measurement of cellular excitability by whole cell patch clamp technique. Physiol Res. 2010, 59 (Suppl. S1), S1–S7. [Google Scholar] [CrossRef] [PubMed]
- Brackenbury WJ, Isom LL. Na channel beta subunits: overachievers of the ion channel family. Front Pharmacol. 2011, 2, 53. [Google Scholar]
- Rook MB, Evers MM, Vos MA, Bierhuizen MF. Biology of cardiac sodium channel Nav1.5 expression. Cardiovasc Res. 2012, 93, 12–23. [Google Scholar] [CrossRef]
- Cecon E, Oishi A, Jockers R. Melatonin receptors: molecular pharmacology and signalling in the context of system bias. Br J Pharmacol. 2018, 175, 3263–3280. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tse LH, Cheung ST, Lee S, Wong YH. Real-Time Determination of Intracellular cAMP Reveals Functional Coupling of Gs Protein to the Melatonin MT1 Receptor. Int J Mol Sci. 2024, 25, 2919. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Iqbal SM, Lemmens-Gruber R. Phosphorylation of cardiac voltage-gated sodium channel: Potential players with multiple dimensions. Acta Physiol 2019, 225, e13210. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Palygin OA, Pettus JM, Shibata EF. Regulation of caveolar cardiac sodium current by a single Gsalpha histidine residue. Am J Physiol Heart Circ Physiol. 2008, 294, H1693–H1699. [Google Scholar] [CrossRef] [PubMed]
- Lowe JS, Palygin O, Bhasin N, Hund TJ, Boyden PA, Shibata E, Anderson ME, Mohler PJ. Voltage-gated Nav channel targeting in the heart requires an ankyrin-G dependent cellular pathway. J Cell Biol. 2008, 180, 173–186. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ono K, Fozzard HA, Hanck DA. Mechanism of cAMP-dependent modulation of cardiac sodium channel current kinetics. Circ Res. 1993, 72, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Schubert B, Vandongen AM, Kirsch GE, Brown AM. Inhibition of cardiac Na+ currents by isoproterenol. Am J Physiol. 1990, 258, H977–H982. [Google Scholar] [CrossRef] [PubMed]
- Wang HW, Yang ZF, Zhang Y, Yang JM, Liu YM, Li CZ. Beta-receptor activation increases sodium current in guinea pig heart. Acta Pharmacol Sin. 2009, 30, 1115–1122. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Matsuda JJ, Lee H, Shibata EF. Enhancement of rabbit cardiac sodium channels by beta-adrenergic stimulation. Circ Res. 1992, 70, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Bernas, T. , Seo, J., Wilson, Z. T., Tan, B. H., Deschenes, I., Carter, C., Liu, J., & Tseng, G. N. Persistent PKA activation redistributes NaV1.5 to the cell surface of adult rat ventricular myocytes. J. Gen. Physiol. 2024, 156, e202313436. [Google Scholar] [CrossRef]
- Hallaq H, Wang DW, Kunic JD, George AL Jr, Wells KS, Murray KT. Activation of protein kinase C alters the intracellular distribution and mobility of cardiac Na+ channels. Am J Physiol Heart Circ Physiol. 2012, 302, H782–H789. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wagner S, Dybkova N, Rasenack EC, Jacobshagen C, Fabritz L, Kirchhof P, Maier SK, Zhang T, Hasenfuss G, Brown JH, Bers DM, Maier LS. Ca2+/calmodulin-dependent protein kinase II regulates cardiac Na+ channels. J Clin Invest. 2006, 116, 3127–3138. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liu X, Ren L, Yu S, Li G, He P, Yang Q, Wei X, Thai PN, Wu L, Huo Y. Late sodium current in synergism with Ca2+/calmodulin-dependent protein kinase II contributes to β-adrenergic activation-induced atrial fibrillation. Philos Trans R Soc Lond B Biol Sci. 2023, 378, 20220163. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ballou, L.M.; Lin, R.Z.; Cohen, I.S. Control of Cardiac Repolarization by Phosphoinositide 3-Kinase Signaling to Ion Channels. Circ Res 2015, 116, 127–137. [Google Scholar] [CrossRef]
- Shu, T.; Wu, T.; Pang, M.; Liu, C.; Wang, X.; Wang, J.; Liu, B.; Rong, L. Effects and mechanisms of melatonin on neural differentiation of induced pluripotent stem cells. Biochem Biophys Res Commun 2016, 474, 566–571. [Google Scholar] [CrossRef]
- Wu, H.; Song, C.; Zhang, J.; Zhao, J.; Fu, B.; Mao, T.; Zhang, Y. Melatonin-mediated upregulation of GLUT1 blocks exit from pluripotency by increasing the uptake of oxidized vitamin C in mouse embryonic stem cells. Faseb j 2017, 31, 1731–1743. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, H.H.; Cecon, E.; Nureki, O.; Rivara, S.; Jockers, R. Melatonin receptor structure and signaling. J Pineal Res 2024, 76. [Google Scholar] [CrossRef] [PubMed]
- Turjanski, A.G.; Estrin, D.A.; Rosenstein, R.E.; McCormick, J.E.; Martin, S.R.; Pastore, A.; Biekofsky, R.R.; Martorana, V. NMR and molecular dynamics studies of the interaction of melatonin with calmodulin. Protein Sci 2004, 13, 2925–2938. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Reyes, R.; Quero-Chávez, D.B.; Alarcón-Elizalde, S.; Cercós, M.G.; Trueta, C.; Constantino-Jonapa, L.A.; Oikawa-Sala, J.; Argueta, J.; Cruz-Garduño, R.; Dubocovich, M.L.; et al. Antidepressant Low Doses of Ketamine and Melatonin in Combination Produce Additive Neurogenesis in Human Olfactory Neuronal Precursors. Molecules 2022, 27, 5650. [Google Scholar] [CrossRef]
- Benítez-King, G.; Argueta, J.; Miranda-Riestra, A.; Muñoz-Delgado, J.; Estrada-Reyes, R. Interaction of the Melatonin/Ca<sup>2+</sup>-CaM Complex with Calmodulin Kinase II: Physiological Importance. Mol Pharmacol 2024, 106, 3–12. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



