Submitted:
11 December 2024
Posted:
11 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
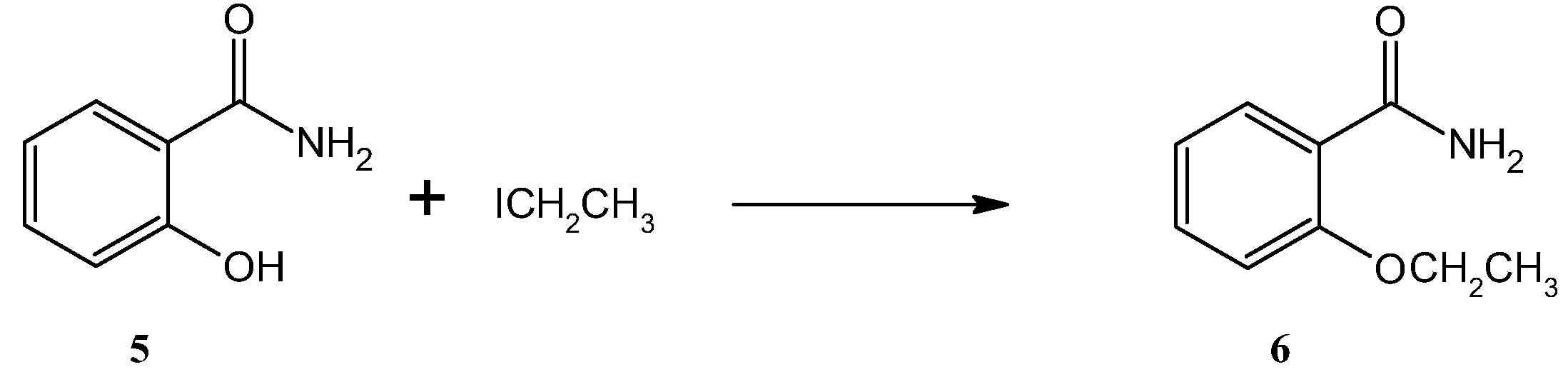
3.1. Conventional Methods of Ethenzamide Synthesis
3.2. Synthesis of Ethenzamide in the Presence of Microwave Radiation, Ultrasound and Mechanochemically
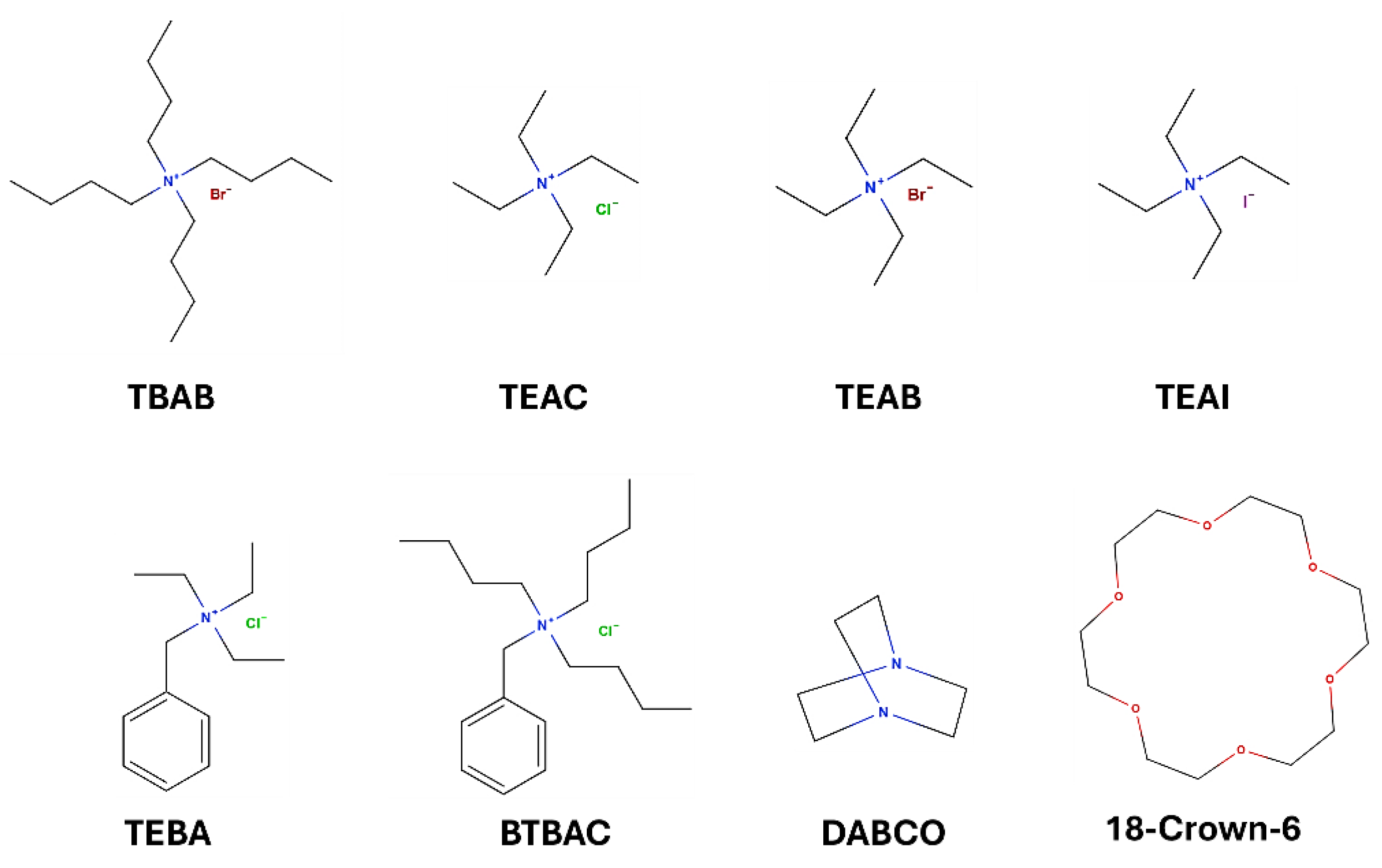
| No | Catalyst PTC | Solvent | Conditions | Time [min] |
Yield [%] |
|---|---|---|---|---|---|
| 1 | TBAB | H2O | K2CO3 | 10 | 84 |
| 2 | TBAB | H2O | - | 10 | 0 |
| 3 | BTBAC | H2O | K2CO3 | 10 | 88 |
| 4 | TEBA | H2O | K2CO3 | 10 | 89 |
| 5 | TEAC | H2O | K2CO3 | 10 | 74 |
| 6 | - | H2O | K2CO3 | 10 | 30 |
4. Synthesis
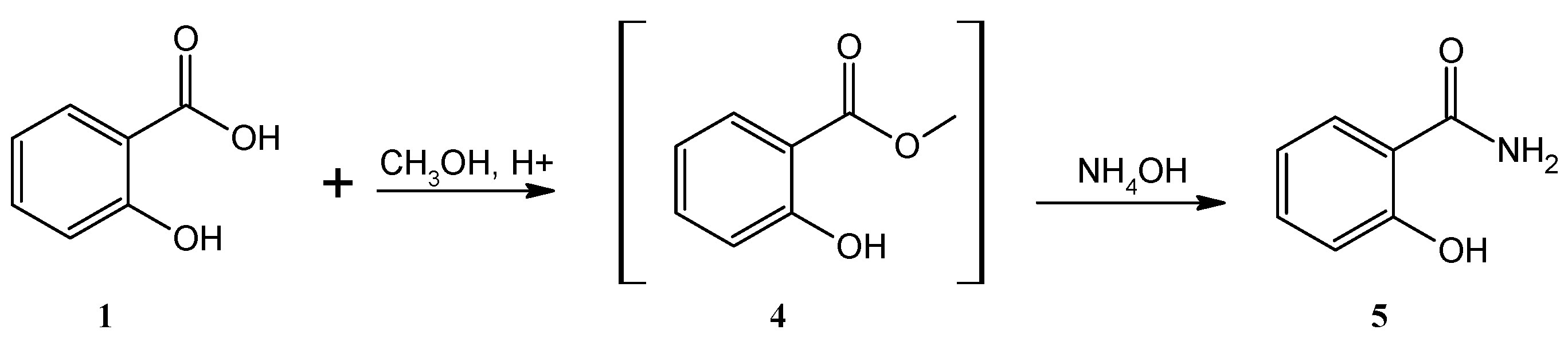
4.1. Synthesis of Salicylamide (5)
4.2. Synthesis of Ethenzamide (6)
4.2.1. Synthesis of Ethenzamide (6) in Conventional Conditions in Ethanol
4.2.2. Synthesis of Ethenzamide (6) in Conventional Conditions in DMF
4.2.3. Synthesis of Ethenzamide (6) in Conventional and Solvent-Free Conditions
4.2.4. Synthesis of Salicylamide (5) in MW Conditions
4.2.5. Synthesis of Salicylamide (5) in US Conditions
4.2.6. Synthesis of Salicylamide (5) in in Mechanochemical Conditions
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Thun, M. Beyond willow bark: Aspirin the prevention of chronic disease. Epidemiology 2000, 11, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Hedner, T.; Everts, B. Clinical history of salicylates in rheumatology and pain. Clin. Rheumatol. 1998, 17, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Mueller, R.L.; Scheidt, S. History of drugs for thrombotic disease. Discovery, development, and directions for the future. Circulation 1994, 89, 432–439. [Google Scholar] [CrossRef]
- Velcheva, E.A.; Stamboliyska, B.A. Structural changes caused by the conversion of 2-hydroxybenzamide (salicylamide) into the oxyanion, J. Mol. Struct. 2008, 875, 264–271. [Google Scholar] [CrossRef]
- Javaid, M.H.; Murray Smith, G.C.; Morrison Barr Martin, N.; Gomez, S.; Ming Lai Loh, V. Jr.; Fan Cockcroft, X.L.; Menear, K.A. PARP inhibitors, Panent WO-2006067472-A1 2024.
- Javaid, M.H.; Morrison Barr Martin, N.; Gomez, S.; Fan Cockcroft, X.L.; Menear, K.A. 2-oxybenzamide derivatives as PARP inhibitors, Patent WO-2007144639-A1 2006.
- Javaid, M.H.; Morrison Barr Martin, N.; Gomez, S.; Fan Cockcroft, X.L.; Menear, K.A. PARP inhibitors, Patent WO-2007144652-A2 2006.
- Lin, C.F.; Yang, J.S.; Chang, C.Y.; Kuo, S.C.; Lee, M.R.; Huang, L.J. ; Synthesis and anticancer activity of benzyloxybenzaldehyde derivatives against HL-60 cells, Bioorg. Med. Chem. 2005, 13, 1537–1544. [Google Scholar] [CrossRef]
- Lee, S.; Yi, Y.K.; Lee, B.H.; Oh, K.S. Studies on Benzofuran-7-carboxamides as Poly(ADP-ribose) Polymerase-1 (PARP-1) Inhibitors. Bulletin of the Korean Chemical Society 2012, 33, 1147–1153. [Google Scholar] [CrossRef]
- Broughton, B.J.; Chaplen, P.; Knowles, P.; Lunt, E.; Marshall, S.M.; Pain, D.L.; Wooldrige, K.R. H. Antiallergic Activity of 2-Phenyl-8-azapurin-6-ones, J. Med. Chem. 1975, 18, 1117–1122. [Google Scholar] [CrossRef]
- Kamei, K.; Maeda, N.; Nomura, K.; Shibata, M.; Katsuragi-Ogino, R.; Koyama, M.; Nakajima, M.; Inoue, T.; Ohno, T.; Tatsuoka, T. ; Synthesis, SAR studies and evaluation of 1,4-benzoxazepine derivatives as selective 5-HT1A receptor agonists with neuroprotective effect: Discovery of Piclozotan, Bioorg. Med. Chem. 2006, 14, 1978–1992. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Wahi, A.K.; Singh, R. Synthesis, computational studies and preliminary pharmacological evaluation of new arylpiperazines as potential antipsychotics, Med. Chem. Res. 2012, 21, 1218–1225. [Google Scholar] [CrossRef]
- Kowalski, P.; Jaśkowska, J.; Bojarski, A.J.; Duszyńska, B.; Bucki, A.; Kołaczkowski, M. ; Evaluation of 1-Arylpiperazine Derivative of Hydroxybenzamides as 5-HT1A and 5-HT7 Serotonin Receptor Ligands: An Experimental and Molecular Modeling Approach, J. Heterocycl. Chem. 2011, 48, 192–198. [Google Scholar] [CrossRef]
- Kowalski, P.; Jaśkowska, J.; Bojarski, A.J.; Duszyńska, B. ; M. ; The Synthesis of Cyclic and Acyclic Long-chain Arylpiperazine Derivatives of Salicylamide as Serotonin Receptor Ligands, J. Heterocycl. Chem. 2008, 45, 209–214. [Google Scholar] [CrossRef]
- Jaśkowska, J.; Drabczyk, A.K.; Śliwa, P.; Jodłowski, P.; Pindelska, E.; Kułaga, D.; Zaręba, P.; Majka, Z.; Siwek, A.; Wolak, M.; Kołaczkowski, M. Ultrasound assisted one-pot synthesis and preliminary in vitro studies of salicylamide arylpiperazines as dual 5-HT1A/5-HT7 ligands J. Mol. Str. 2023, 1275, 134585. [Google Scholar] [CrossRef]
- Qiao, C.; Gupte, A.; Boshoff, H.I.; Wilson, D.J.; Benett, E.M.; Somu, R.V.; Barry, C.E.; Aldrich, C.C. 5’-O-[(N-Acyl)sulfamoyl]adenosines as Antitubercular Agents that Inhibit MbtA: An Adenylation Enzyme Required for Siderophore Biosynthesis of the Mycobactins, J. Med. Chem. 2007, 50, 6080–6094. [Google Scholar] [CrossRef] [PubMed]
- Żmudzka, E.; Lustyk, K.; Siwek, A.; Wolak, M.; Gałuszka, A.; Jaśkowska, J.; Kołaczkowski, M.; Sapa, J.; Pytka, K. Novel Arylpiperazine Derivatives of Salicylamide with α1-Adrenolytic Properties Showed Antiarrhythmic and Hypotensive Properties in Rats. Int. J. Mol. Sci. 2023, 24, 293. [Google Scholar] [CrossRef]
- Żmudzka, E.; Lustyk, K.; Głuch-Lutwin, M.; Wolak, M.; Jaśkowska, J.; Kołaczkowski, M.; Sapa, J.; Pytka, K. Novel Multimodal Salicylamide Derivative with Antidepressant-like, Anxiolytic-like, Antipsychotic-like, and Anti-Amnesic Activity in Mice. Pharmaceuticals 2023, 16, 175. [Google Scholar] [CrossRef] [PubMed]
- Żmudzka, E.; Lustyk, K.; Sałaciak, K.; Siwek, A.; Jaśkowska, J.; Kołaczkowski, M.; Sapa, J.; Pytka, K. Potential Anti-Amnesic Activity of a Novel Multimodal Derivative of Salicylamide, JJGW08, in Mice. Pharmaceuticals 2023, 16, 399. [Google Scholar] [CrossRef] [PubMed]
- García-March, F.J.; García-Domenech, R.; Gálvez, J.; Antón-Fos, G.M.; Julián-Ortiz, J.V.; Giner-Pons, R.; Recio-Iglesias, M.C. Pharmacological Studies of 1-(p-Chlorophenyl)propanol and 2-(1-Hydroxy-3-butenyl)phenol: Two New Non-narcotic Analgesics Designed by Molecular Connectivity. J Pharm Pharmacol. 1997, 1997. 49, 10–15. [Google Scholar] [CrossRef]
- Nikaido, T.; Maruyama, C.; Hamanaka, M.; Yamaguchi, C.; Fujimaru, Y.; Nakanishi, Y.; Asano, T.; Takaoka, A. Ethenzamide Exerts Analgesic Effect at the Spinal Cord via Multiple Mechanisms of Action Including the 5HT2B Receptor Blockade in the Rat Formalin Test. Biol Pharm Bull. 2020, 43, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Darias, V.; Bravo, L.; Abdallah, S.S.; Sánchez Mateo, C.C.; Expósito-orta M., A.; Lissavetsky, J.; Manzanares, J. Synthesis and Preliminary Pharmacological Study of Thiophene Analogues of the Antipyretic and Analgesic Agent Ethenzamide. Archiv der Pharmazie, 1992, 325, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.J.; Guo, H.R. ; Frequent analgesics consumption in migraineurs: Comparison between chronic and episodic migraineurs. J. Headache Pain 2004, 5, 30–35. [Google Scholar] [CrossRef]
- Bertholdt, H.; Michalczyk, D.; Hartl, R.; Lieb, H. Medicament with an analgesic, antipyretic and/or antiphlogistic action and use of 2-ethoxybenzoic acid. Patent US5419915 A 1995.
- Mathur, K.C.; Gupta, S.; Khadikar, P.V. Topological modelling of analgesia. Bioorg. Med. Chem. 2003, 11, 1915–1928. [Google Scholar] [CrossRef]
- Fahmy, H.H.; El-Eraky, W. Synthesis and evaluation of the analgesic and antiinflammatory activities of O-substituted salicylamides. Arch Pharm Res. 2001, 24, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Ohnacker, G.; Kottler, A. ; Process for the production of benzoic acid derivatives, USA Patent 2895992, 1959. [Google Scholar]
- Li, S.; Cao, X.; Chen, C.; Ke, S. Novel salicylic acid-oriented thiourea-type receptors as colorimetric chemosensor: Synthesis, characterizations and selective naked-eye recognition properties. Spectrochim Acta A Mol Biomol Spectrosc 2012, 96, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Allais, F.; Brogi, S.; Castro, G.R.; Vidali, V.P. Editorial: Advances in green synthesis for drug discovery. Front. Chem 2023, 11, 1166887. [Google Scholar] [CrossRef] [PubMed]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: New York, 1998. [Google Scholar]
- Sanderson, K. ; Chemistry: It’s not easy being green. Nature. 2011, 469, 18–20. [Google Scholar] [CrossRef]
- Jaśkowska, J.; Drabczyk, A.K.; Michorczyk, P.; Kułaga, D.; Zaręba, P.; Jodłowski, P.; Majka, Z.; Jakubski, J.; Pindelska, E. Mechanochemical Synthesis Method for Drugs Used in the Treatment of CNS Diseases under PTC Conditions. Catalysts 2022, 12, 464. [Google Scholar] [CrossRef]
| No | Catalyst | Solvent | Base | Conditions | Time [h] |
Yield* [%] |
|---|---|---|---|---|---|---|
| 1 | - | ethanol | NaOH | reflux | 3 | 43 |
| 2 | KI | DMF | K2CO3 | reflux | 4 | 60 |
| 3 | TBAB | - | K2CO3 | 20 °C | 192 | 34 |
| 4 | TBAB | - | K2CO3 | 40 °C | 3.5 | 14 |
| 5 | TBAB | - | K2CO3 | 60 °C | 1.5 | 52 |
| 6 | TBAB | - | K2CO3 | 80 °C | 0.25 | 79 |
| 7 | TEBA | - | K2CO3 | 80 °C | 0.25 | 72 |
| 8 | TEAC | - | K2CO3 | 80 °C | 1 | 83 |
| 9 | TEAI | - | K2CO3 | 80 °C | 0.25 | 74 |
| 10 | TEAB | - | K2CO3 | 80 °C | 0.25 | 86 |
| 11 | DABCO | - | K2CO3 | 80 °C | 0.25 | 69 |
| 12 | 18-CROWN-6 | - | K2CO3 | 80 °C | 0.25 | 78 |
| No | Catalyst PTC | Solvent | Conditions | Time [s] |
Temp. [°C] |
Pressure [bar] |
Yield [%] |
|---|---|---|---|---|---|---|---|
| 1 | TBAB | - | K2CO3 | 90 | - | atm. | 92* |
| 2 | TBAB | - | K2CO3/DMF | 60 | - | atm. | 85* |
| 3 | TBAB | glycerin | DIPEA | 120 | 99 | 2.3 | 69 |
| 4 | TBAB | H2O | K2CO3 | 120 | 99 | 3.5 | 80 |
| 5 | TBAB | H2O | - | 120 | 65 | 1.4 | 0 |
| 6 | BTBAC | H2O | K2CO3 | 120 | 93 | 3.2 | 90 |
| 7 | TEBA | H2O | K2CO3 | 120 | 95 | 3.7 | 85 |
| 8 | TEAC | H2O | K2CO3 | 120 | 102 | 4.3 | 90 |
| 9 | - | H2O | K2CO3 | 120 | 120 | 4.2 | 78 |
| No | Catalyst PTC | Solvent | Conditions | Time [min] |
Yield [%] |
|---|---|---|---|---|---|
| 1 | TBAB | - | K2CO3 | 20 | 23 |
| 2 | TEAC | - | K2CO3 | 20 | 17 |
| 3 | BTBAC | - | K2CO3 | 20 | 10 |
| 4 | TEBA | - | K2CO3 | 20 | 52 |
| 5 | TEBA | - | K2CO3 | 60 | 60 |
| 6 | - | - | K2CO3 | 20 | 53 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




