Submitted:
11 December 2024
Posted:
11 December 2024
Read the latest preprint version here
Abstract
Cardiovascular disease (CVD), a major global health concern, is characterized by cardiac complications that can lead to death. The commonly used treatments for this condition are synthetic drugs, but these often come with risky side effects. A potential alternative is the use of traditional medicinal plants, such as Amaranthus viridis, which is rich in bioactive compounds. This study aimed to determine the non-toxic concentration of A. viridis ethanolic extract, investigate its cardioprotective effects on zebrafish (Danio rerio) heart rate and cardiac phenotype, qualitatively assess the presence of phytochemicals, and assess its antioxidant activity using DPPH free radical scavenging activity. Zebrafish larvae at 72 hours post-fertilization (hpf) were used to evaluate mortality and optimize dosing. Physio-morphological screening was conducted by pre-treating zebrafish larvae with the extract 4 hours prior to administering a heart failure inducer, verapamil. The maximum non-toxic concentration was found to be 25 µg/mL, as all zebrafish survived after 24 hours. Mortality began at 50 µg/mL, and concentrations from 100 µg/mL to 400 µg/mL resulted in 100% mortality. All tested concentrations of A. viridis leaf extract showed cardioprotective activity in the physio-morphological analysis, with 3.125 µg/mL identified as the lowest effective dose. Phytochemical analysis detected the presence of alkaloids and phenols. Furthermore, A. viridis exhibited antioxidant activity from all tested concentrations. Based on the results, A. viridis exhibited cardioprotective effects against verapamil-induced cardiotoxicity, as evidenced by the recovery of heart rate and cardiac phenotype in the zebrafish model.
Keywords:
1. Introduction
2. Material and Methods
2.1. Collection and Authentication of Plant Material
2.2. Plant Extraction
2.3. Preparation of Egg Water
2.4. Ethics Declaration
2.5. Establishment of Zebrafish Aquarium and Husbandry
2.6. Procurement of Zebrafish
2.7. Breeding and Embryo Isolation
2.8. Preliminary Mortality Test of A. viridis Extracts Against Zebrafish Larvae
2.9. Treatment Protocol for Assessing Cardioprotective Activity
2.10. Heart Rate Assessment
2.11. Scoring on Cardiac Phenotypes
2.12. Disposal of Zebrafish Carcass
2.13. Qualitative Phytochemical Analysis
2.14. Statistical Analyses
3. Results
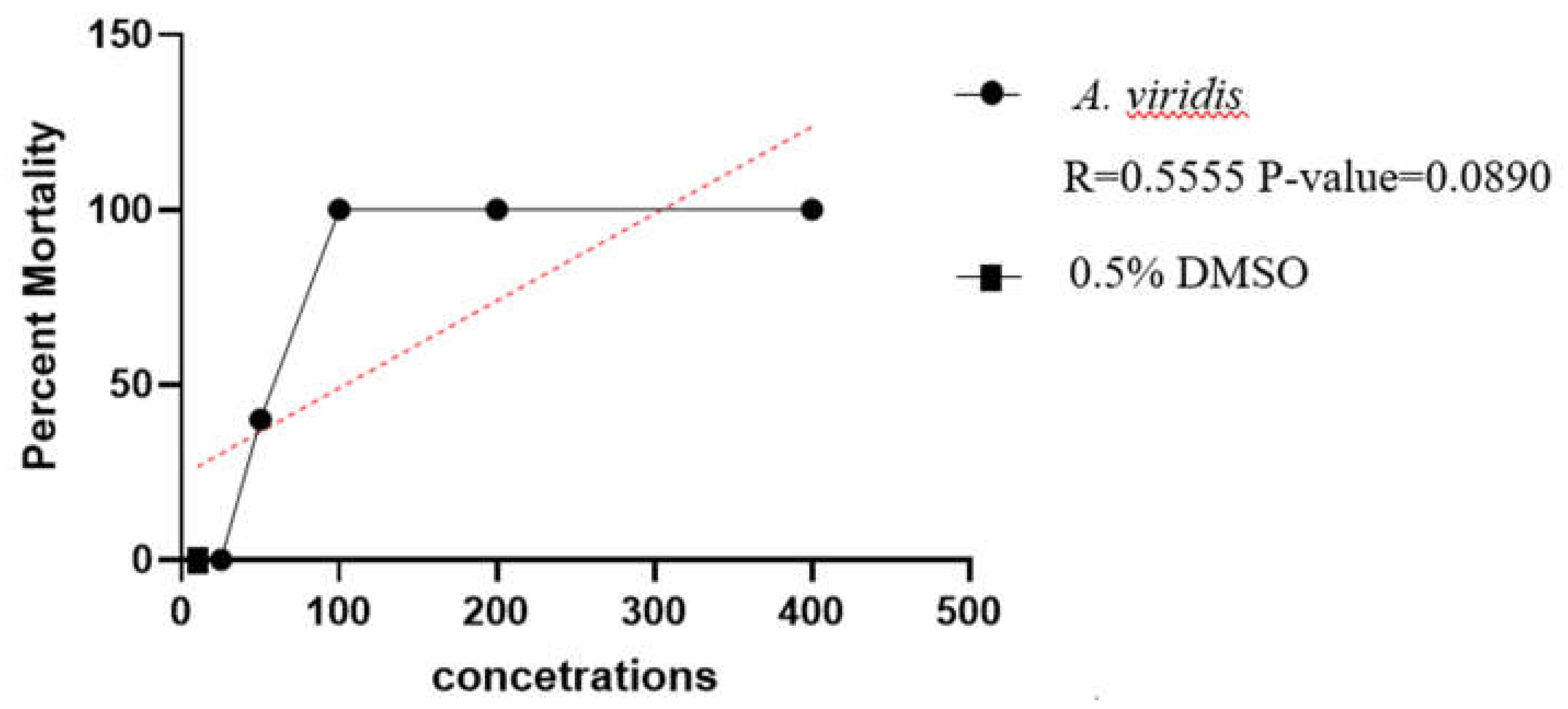
3.1. In Vivo Maximum Non-Toxic Concentration of the Ethanolic Leaf Extracts of A. viridis Against 72hpf Zebrafish
3.2. Heart Rate Response of Verapamil-Induced Zebrafish Larvae Treated with A. viridis Leaf Ethanolic Extracts
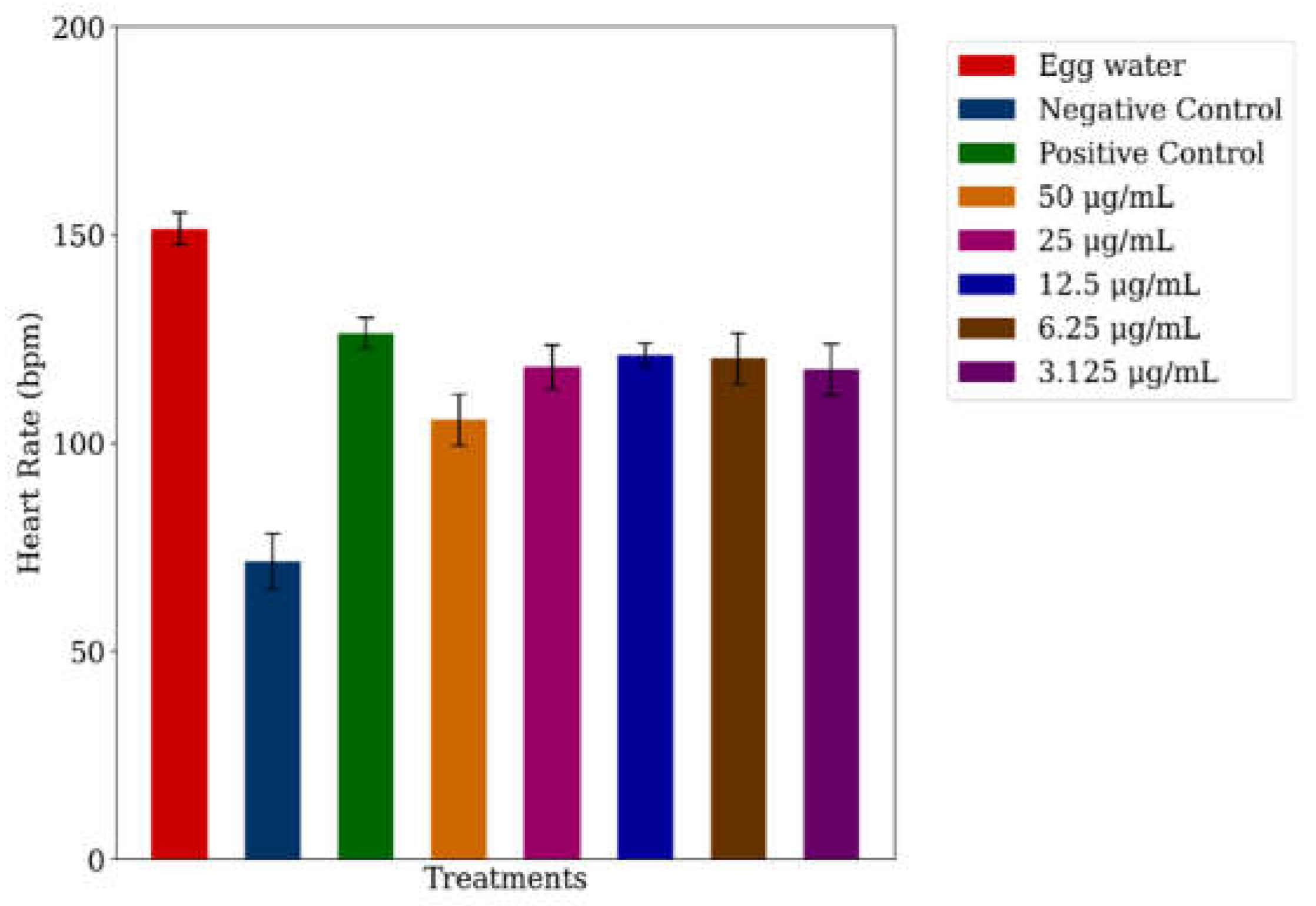
3.3. Cardiac Scoring of the Cardioprotective Response of A. viridis Leaf Ethanolic Extracts on Verpamil-Induced Heart Failure in Zebrafish Larvae
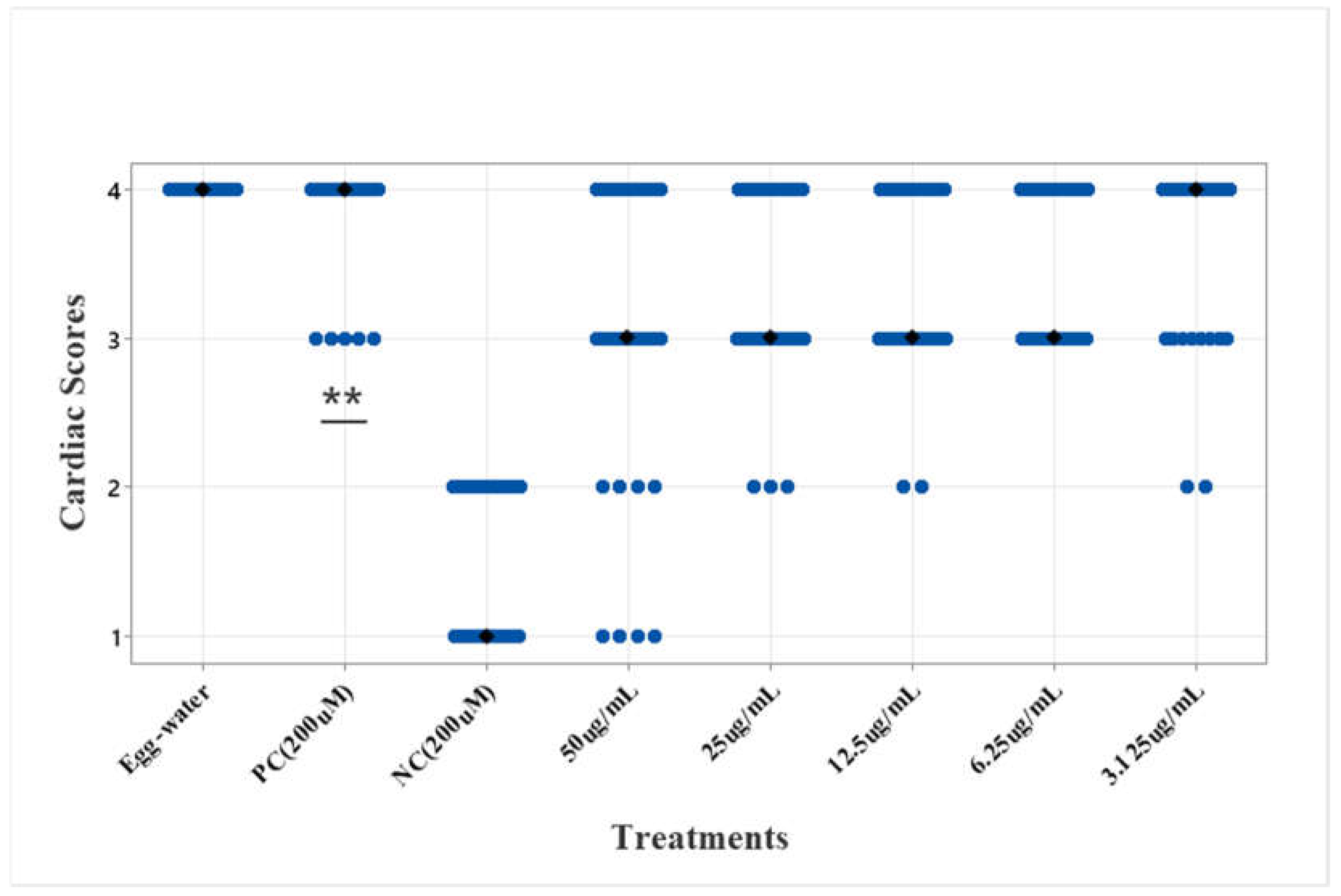

3.4. Phytochemical Composition of A. viridis Crude Leaf Extracts
| Treatments | Concentration | Cardiac functions and phenotypes |
| Egg water | 60 μg/mL | Normal function and no visible abnormality |
| NC - Verapamil | 200 μM | No contraction in the heart; Weak contraction in the atria; No circulation; Pericardial edema |
| PC - Eplerenone + Verapamil | 200 μM | Some showed slow circulation, while most had no visible abnormalities |
| Leaf extract + Verapamil | 50 μg/mL | Venous congestion; No contraction in the heart; Weak contraction in the atria; No circulation; |
| Leaf extract + Verapamil | 25 μg/mL | Some showed slow circulation, while most had no visible abnormalities |
| Leaf extract + Verapamil | 12.5 μg/mL | Some showed slow circulation, while most had no visible abnormalities |
| Leaf extract + Verapamil | 6.25 μg/mL | Some showed slow circulation, while most had no visible abnormalities |
| Leaf extract + Verapamil | 3.125 μg/mL | Some showed slow circulation, while most had no visible abnormalities |
| A. viridis | |
| Alkaloids | - |
| Flavonoids | + |
| Phenols | - |
| Saponins | + |
3.5. In Vitro Free Radical Scavenging Activity of A. viridis Ethanolic Leaf Extracts
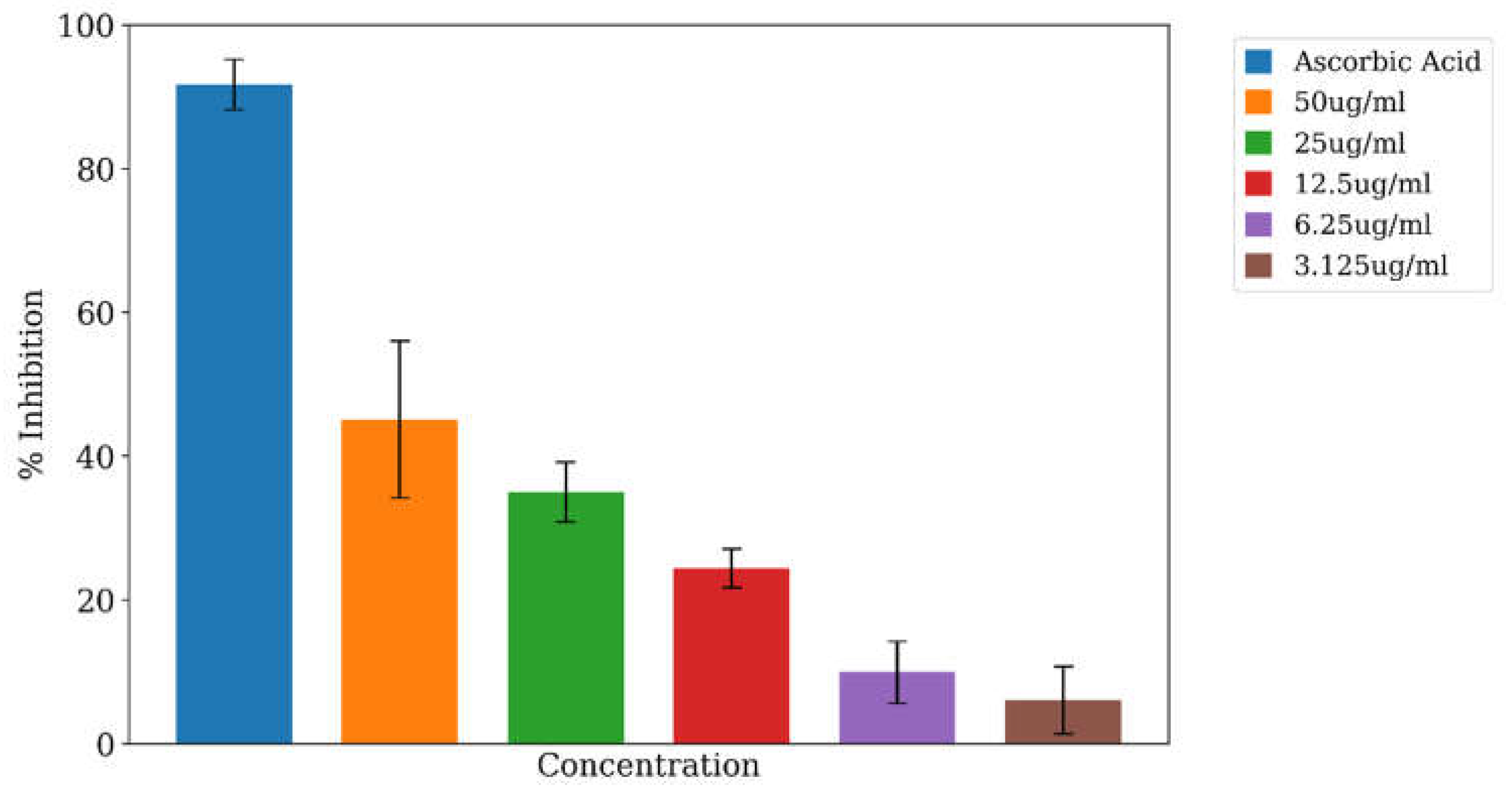
4. Discussion
4.1. Toxicity of A. viridis Ethanolic Leaf Extracts on Zebrafish Larvae
4.2. Cardioprotective Activity of the Ethanolic Leaf Extracts of A. viridis on Verapamil-Induced Zebrafish Larvae
4.3. Phytochemicals Composition of A. viridis Leaf Ethanolic Extracts
4.4. Free Radical Scavenging Activity of A. viridis Ethanolic Leaf Extract
5. Conclusions
6. Recommendation
Author Contributions
Conflicts of Interest
References
- Abdel-Alim, M.E.; Serag, M.S.; Moussa, H.R.; Elgendy, M.A.; Mohesien, M.T.; Salim, N.S. Phytochemical Screening and Antioxidant Potential of Lotus corniculatusand Amaranthus viridis. Egypt. J. Bot. 2023, 63, 665–681. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Hanif, S.; Iftkhar, T. Phytochemical Profiling with Antioxidant and Antimicrobial Screening of Amaranthus viridis L. Leaf and Seed Extracts. Open J. Med Microbiol. 2013, 03, 164–171. [Google Scholar] [CrossRef]
- Aronson, J. K. (Ed.). (2014). Meyler’s Side Effects of Drugs 15E: The International Encyclopedia of Adverse Drug Reactions and Interactions. Newnes.
- Bachheti, R.K.; Worku, L.A.; Gonfa, Y.H.; Zebeaman, M.; Deepti; Pandey, D.P.; Bachheti, A. Prevention and Treatment of Cardiovascular Diseases with Plant Phytochemicals: A Review. Evidence-Based Complement. Altern. Med. 2022, 2022, 1–21. [CrossRef]
- Bagher, P.; Segal, S.S. Regulation of blood flow in the microcirculation: role of conducted vasodilation. Acta Physiol. 2010, 202, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, K.; Chopra, C.; Bhardwaj, P.; Dhanjal, D.S.; Singh, R.; Najda, A.; Cruz-Martins, N.; Singh, S.; Sharma, R.; Kuča, K.; et al. Biogenic Metallic Nanoparticles from Seed Extracts: Characteristics, Properties, and Applications. J. Nanomater. 2022, 2022, 1–22. [Google Scholar] [CrossRef]
- Bray, F.; Laversanne, M.; Cao, B.; Varghese, C.; Mikkelsen, B.; Weiderpass, E.; Soerjomataram, I. Comparing cancer and cardiovascular disease trends in 20 middle- or high-income countries 2000–19: A pointer to national trajectories towards achieving Sustainable Development goal target 3.4. Cancer Treat. Rev. 2021, 100, 102290. [Google Scholar] [CrossRef]
- Brittijn, S.A.; Duivesteijn, S.J.; Belmamoune, M.; Bertens, L.F.; Bitter, W.; Debruijn, J.D.; Champagne, D.L.; Cuppen, E.; Flik, G.; Vandenbroucke-Grauls, C.M.; et al. Zebrafish development and regeneration: new tools for biomedical research. Int. J. Dev. Biol. 2009, 53, 835–850. [Google Scholar] [CrossRef]
- Brown, D.R.; Samsa, L.A.; Qian, L.; Liu, J. Advances in the Study of Heart Development and Disease Using Zebrafish. J. Cardiovasc. Dev. Dis. 2016, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Bucciarelli, V.; Caterino, A.L.; Bianco, F.; Caputi, C.G.; Salerni, S.; Sciomer, S.; Maffei, S.; Gallina, S. Depression and cardiovascular disease: The deep blue sea of women's heart. Trends Cardiovasc. Med. 2019, 30, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Chia, N. , Fulcher, J., & Keech, A. (2016). Beta-blocker, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker, nitrate-hydralazine, diuretics, aldosterone antagonist, ivabradine, devices and digoxin (BANDAID 2): an evidence-based mnemonic for the treatment of systolic heart failure. Internal medicine journal, 46(6), 653-662.
- Choy, K.W.; Murugan, D.; Mustafa, M.R. Natural products targeting ER stress pathway for the treatment of cardiovascular diseases. Pharmacol. Res. 2018, 132, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Corpuz, J. C. (2023). Cardiovascular disease in the Philippines: a new public health emergency? Journal of Public Health, fdad175.
- Daniels, M. , Donilon, T., & Bollyky, T. J. (2014). The emerging global health crisis: noncommunicable diseases in low-and middle-income countries. Council on Foreign Relations independent task force report, (72).
- Dlugos, C.A.; Rabin, R.A. Structural and Functional Effects of Developmental Exposure to Ethanol on the Zebrafish Heart. Alcohol. Clin. Exp. Res. 2010, 34, 1013–1021. [Google Scholar] [CrossRef]
- Dubey, A.; Ghosh, N.S.; Singh, R. Zebrafish as An Emerging Model: An Important Testing Platform for Biomedical Science. J. Pharm. Negat. Results 2022, 13, 1–7. [Google Scholar] [CrossRef]
- Dweck, M.R.; Williams, M.C.; Moss, A.J.; Newby, D.E.; Fayad, Z.A. Computed Tomography and Cardiac Magnetic Resonance in Ischemic Heart Disease. Circ. 2016, 68, 2201–2216. [Google Scholar] [CrossRef] [PubMed]
- Eimon, P.M.; Ashkenazi, A. The zebrafish as a model organism for the study of apoptosis. Apoptosis 2009, 15, 331–349. [Google Scholar] [CrossRef] [PubMed]
- Forbes, E.L.; Preston, C.D.; Lokman, P.M. Zebrafish (Danio rerio) and the egg size versus egg number trade off: effects of ration size on fecundity are not mediated by orthologues of the Fec gene. Reprod. Fertil. Dev. 2010, 22, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, H.; Little, W.C. The Cardiac Cycle and the Physiologic Basis of Left Ventricular Contraction, Ejection, Relaxation, and Filling. Hear. Fail. Clin. 2008, 4, 1–11. [Google Scholar] [CrossRef]
- Gandhi, P., Samarth, R. M., & Peter, K. (2020). Bioactive Compounds of Amaranth (Genus Amaranthus). Bioactive Compounds in Underutilized Vegetables and Legumes, 1-37.
- Geetha, R.G.; Ramachandran, S. Recent Advances in the Anti-Inflammatory Activity of Plant-Derived Alkaloid Rhynchophylline in Neurological and Cardiovascular Diseases. Pharmaceutics 2021, 13, 1170. [Google Scholar] [CrossRef]
- Genge, C. E., Lin, E., Lee, L., Sheng, X., Rayani, K., Gunawan, M., & Tibbits, G. F. (2016). The zebrafish heart as a model of mammalian cardiac function. Reviews of Physiology, Biochemistry and Pharmacology, Vol. 171, 99-136.
- Giardoglou, P.; Beis, D. On Zebrafish Disease Models and Matters of the Heart. Biomedicines 2019, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- González-Rosa, J.M. Zebrafish Models of Cardiac Disease: From Fortuitous Mutants to Precision Medicine. Circ. Res. 2022, 130, 1803–1826. [Google Scholar] [CrossRef]
- Haege, E.R.; Huang, H.-C.; Huang, C.-C. Identification of Lactate as a Cardiac Protectant by Inhibiting Inflammation and Cardiac Hypertrophy Using a Zebrafish Acute Heart Failure Model. Pharmaceuticals 2021, 14, 261. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-C.; Monte, A.; Cook, J.M.; Kabir, M.S.; Peterson, K.P. Zebrafish Heart Failure Models for the Evaluation of Chemical Probes and Drugs. ASSAY Drug Dev. Technol. 2013, 11, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Hu, N. , Sedmera, D., Yost, H. J., & Clark, E. B. (2000). Structure and function of the developing zebrafish heart. The Anatomical Record: An Official Publication of the American Association of Anatomists, 260(2), 148-157.
- Iamonico, D.; Hussain, A.N.; Sindhu, A.; Kumar, V.N.S.A.; Shaheen, S.; Munir, M.; Fortini, P. Trying to Understand the Complicated Taxonomy in Amaranthus (Amaranthaceae): Insights on Seeds Micromorphology. Plants 2023, 12, 987. [Google Scholar] [CrossRef]
- Imam, M.U.; Zhang, S.; Ma, J.; Wang, H.; Wang, F. Antioxidants Mediate Both Iron Homeostasis and Oxidative Stress. Nutrients 2017, 9, 671. [Google Scholar] [CrossRef]
- Iqbal, M. J., Hanif, S., Mahmood, Z., Anwar, F., & Jamil, A. (2012). Antioxidant and antimicrobial activities of Chowlai (Amaranthus viridis L.) leaf and seed extracts. J. Med. Plants Res, 6(27), 4450-4455.
- Irion, U., & Nüsslein-Volhard, C. (2022). Developmental genetics with model organisms. Proceedings of the National Academy of Sciences, 119(30), e2122148119.
- Jewhurst, K.; McLaughlin, K.A. Beyond the Mammalian Heart: Fish and Amphibians as a Model for Cardiac Repair and Regeneration. J. Dev. Biol. 2015, 4, 1. [Google Scholar] [CrossRef]
- Jin, L.; Pan, Y.; Li, Q.; Li, J.; Wang, Z. Elabela gene therapy promotes angiogenesis after myocardial infarction. J. Cell. Mol. Med. 2021, 25, 8537–8545. [Google Scholar] [CrossRef] [PubMed]
- Jing, L. , Xi, L., Chen, Y., Chen, Z., Zhang, L. H., Fang, F., & Jiang, L. X. (2013). National survey of doctor-reported secondary preventive treatment for patients with acute coronary syndrome in China. Chinese medical journal, 126(18), 3451-3455.
- Jyotsna, F.; Ahmed, A.; Kumar, K.; Kaur, P.; Chaudhary, M.H.; Kumar, S.; Khan, E.; Khanam, B.; Shah, S.U.; Varrassi, G.; et al. Exploring the Complex Connection Between Diabetes and Cardiovascular Disease: Analyzing Approaches to Mitigate Cardiovascular Risk in Patients With Diabetes. Cureus 2023, 15, e43882. [Google Scholar] [CrossRef] [PubMed]
- Keßler, M.; Just, S.; Rottbauer, W. Ion Flux Dependent and Independent Functions of Ion Channels in the Vertebrate Heart: Lessons Learned from Zebrafish. Stem Cells Int. 2012, 2012, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kirshenblatt-Gimblett, B.; Fernandez, D.G. Culture Ingested: On the Indigenization of Phillipine Food. Gastronomica 2003, 3, 58–71. [Google Scholar] [CrossRef]
- Kossack, M.; Hein, S.; Juergensen, L.; Siragusa, M.; Benz, A.; Katus, H.A.; Most, P.; Hassel, D. Induction of cardiac dysfunction in developing and adult zebrafish by chronic isoproterenol stimulation. J. Mol. Cell. Cardiol. 2017, 108, 95–105. [Google Scholar] [CrossRef]
- Kris-Etherton, P. M. , Petersen, K. S., Velarde, G., Barnard, N. D., Miller, M., Ros, E.,& Freeman, A. M. (2020). Barriers, opportunities, and challenges in addressing disparities in diet-related cardiovascular disease in the United States. Journal of the American Heart Association, 9(7), e014433.
- Krum, H.; Massie, B.; Abraham, W.T.; Dickstein, K.; Kober, L.; McMurray, J.J.; Desai, A.; Gimpelewicz, C.; Kandra, A.; Reimund, B.; et al. Direct renin inhibition in addition to or as an alternative to angiotensin converting enzyme inhibition in patients with chronic systolic heart failure: rationale and design of the Aliskiren Trial to Minimize OutcomeS in Patients with HEart failuRE (ATMOSPHERE) study. Eur. J. Hear. Fail. 2011, 13, 107–114. [Google Scholar] [CrossRef]
- Anuradha; Kumari, M. ; Zinta, G.; Chauhan, R.; Kumar, A.; Singh, S.; Singh, S. Genetic resources and breeding approaches for improvement of amaranth (Amaranthus spp.) and quinoa (Chenopodium quinoa). Front. Nutr. 2023, 10, 1129723. [Google Scholar] [CrossRef]
- Lahera, V.; Goicoechea, M.; De Vinuesa, S.G.; Miana, M.; de las Heras, N.; Cachofeiro, V.; Luño, J. Endothelial dysfunction, oxidative stress and inflammation in atherosclerosis: beneficial effects of statins. Curr. Med. Chem. 2007, 14, 243–248. [Google Scholar] [CrossRef]
- Lee, R.T.; Walsh, K. The Future of Cardiovascular Regenerative Medicine. Circulation 2016, 133, 2618–2625. [Google Scholar] [CrossRef]
- Li, S. , Liu, H., Li, Y., Qin, X., Li, M., Shang, J., & Zhou, M. (2021). Shen-Yuan-Dan capsule attenuates verapamil-induced zebrafish heart failure and exerts antiapoptotic and anti-inflammatory effects via reactive oxygen species–induced NF-κB pathway. Frontiers in Pharmacology, 12, 626515.
- Li, Q.; Wang, P.; Chen, L.; Gao, H.; Wu, L. Acute toxicity and histopathological effects of naproxen in zebrafish (Danio rerio) early life stages. Environ. Sci. Pollut. Res. 2016, 23, 18832–18841. [Google Scholar] [CrossRef]
- Li, J.; Zhu, Y.; Zhao, X.; Zhao, L.; Wang, Y.; Yang, Z. Screening of anti-heart failure active compounds from fangjihuangqi decoction in verapamil-induced zebrafish model by anti-heart failure index approach. Front. Pharmacol. 2022, 13, 999950. [Google Scholar] [CrossRef]
- Low Wang, C. C. , Hess, C. N., Hiatt, W. R., & Goldfine, A. B. (2016). Clinical update: cardiovascular disease in diabetes mellitus: atherosclerotic cardiovascular disease and heart failure in type 2 diabetes mellitus–mechanisms, management, and clinical considerations. Circulation, 133(24), 2459-2502.
- Mahmood, A., Eqan, M., Pervez, S., Javed, R., Ullah, R., Islam, A., & Rafiq, M. (2021). Drugs Resistance in Heart Diseases. Biochemistry of Drug Resistance, 295-334.
- Meid, H. , & S Haddad, P. (2017). The antidiabetic potential of quercetin: underlying mechanisms. Current medicinal chemistry, 24(4), 355-364.
- Minamino, T. (2012). Cardioprotection From Ischemia/Reperfusion Injury–Basic and Translational Research–. Circulation Journal, 76(5), 1074-1082.
- Monteiro, L.M.; Vasques-Nóvoa, F.; Ferreira, L.; Pinto-Do-Ó, P.; Nascimento, D.S. Restoring heart function and electrical integrity: closing the circuit. npj Regen. Med. 2017, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C. , & Cheng-Lai, A. (2013). The polypill: a potential global solution to cardiovascular disease. Cardiology in review, 21(1), 49-54.
- Nowbar, A. N. , Gitto, M., Howard, J. P., Francis, D. P., & Al-Lamee, R. (2019). Mortality from ischemic heart disease: Analysis of data from the World Health Organization and coronary artery disease risk factors From NCD Risk Factor Collaboration. Circulation: cardiovascular quality and outcomes, 12(6), e005375.
- Omondi, J. O. (2017). Phenotypic variation in morphology, yield and seed quality in selected accessions of leafy Amaranths (Doctoral dissertation, Maseno University).
- Pearson, T. A. , Mensah, G. A., Alexander, R. W., Anderson, J. L., Cannon III, R. O., Criqui, M. & Vinicor, F. (2003). Markers of inflammation and cardiovascular disease: application to clinical and public health practice: a statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. circulation, 107(3), 499-511.
- Peter, K.; Gandhi, P. Rediscovering the therapeutic potential of Amaranthus species: A review. Egypt. J. Basic Appl. Sci. 2017, 4, 196–205. [Google Scholar] [CrossRef]
- Pichaivel, M. , Dhandayuthapani, D., Porwal, O., Sharma, P. K., & Manickam, D. (2022). Phytochemical And Pharmacological Evaluation Of Hydroalcoholic Extract Of Amaranthus Viridis Linn. Journal of Pharmaceutical Negative Results, 2250-2269.
- Pirdhankar, M. S. , Sadamate, P. S., & Tasgaonkar, D. R. (2023). Cardioprotective Herbal Plants: A Review. International Journal for Research in Applied Science & Engineering Technology, 11(3), 1114-1118.
- Plein, S.; Greenwood, J.P.; Ridgway, J.P.; Cranny, G.; Ball, S.G.; Sivananthan, M.U. Assessment of non–ST-segment elevation acute coronary syndromes with cardiac magnetic resonance imaging. Circ. 2004, 44, 2173–2181. [Google Scholar] [CrossRef]
- Poon, K.L.; Brand, T. The zebrafish model system in cardiovascular research: A tiny fish with mighty prospects. Glob. Cardiol. Sci. Pr. 2013, 2013, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Pruthvi, N. (2010). Phytochemical Studies And Evaluation Of Leaves Of Amaranthus Viridis (Amaranthaceae) For Antidiabetic Activity (Doctoral dissertation, Rajiv Gandhi University of Health Sciences (India).
- Raggi, P.; Genest, J.; Giles, J.T.; Rayner, K.J.; Dwivedi, G.; Beanlands, R.S.; Gupta, M. Role of inflammation in the pathogenesis of atherosclerosis and therapeutic interventions. Atherosclerosis 2018, 276, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Ribas, L.; Piferrer, F. The zebrafish (Danio rerio) as a model organism, with emphasis on applications for finfish aquaculture research. Rev. Aquac. 2013, 6, 209–240. [Google Scholar] [CrossRef]
- Rotariu, D.; Babes, E.E.; Tit, D.M.; Moisi, M.; Bustea, C.; Stoicescu, M.; Radu, A.-F.; Vesa, C.M.; Behl, T.; Bungau, A.F.; et al. Oxidative stress – Complex pathological issues concerning the hallmark of cardiovascular and metabolic disorders. Biomed. Pharmacother. 2022, 152, 113238. [Google Scholar] [CrossRef]
- Salvamani, S. , Gunasekaran, B., Shukor, M. Y., Shaharuddin, N. A., Sabullah, M. K., & Ahmad, S. A. (2016). Anti-HMG-CoA reductase, antioxidant, and anti-inflammatory activities of Amaranthus viridis leaf extract as a potential treatment for hypercholesterolemia. Evidence-Based Complementary and Alternative Medicine, 2016.
- Santoso, F.; Farhan, A.; Castillo, A.L.; Malhotra, N.; Saputra, F.; Kurnia, K.A.; Chen, K.H.-C.; Huang, J.-C.; Chen, J.-R.; Hsiao, C.-D. An Overview of Methods for Cardiac Rhythm Detection in Zebrafish. Biomedicines 2020, 8, 329. [Google Scholar] [CrossRef] [PubMed]
- Sapna, F.; Raveena, F.; Chandio, M.; Bai, K.; Sayyar, M.; Varrassi, G.; Khatri, M.; Kumar, S.; Mohamad, T. Advancements in Heart Failure Management: A Comprehensive Narrative Review of Emerging Therapies. Cureus 2023, 15, e46486. [Google Scholar] [CrossRef]
- Saravanan, G.; Ponmurugan, P. Amaranthus viridis Linn., a common spinach, modulates C-reactive protein, protein profile, ceruloplasmin and glycoprotein in experimental induced myocardial infarcted rats. J. Sci. Food Agric. 2012, 92, 2459–2464. [Google Scholar] [CrossRef] [PubMed]
- Sarker, U.; Oba, S. Nutraceuticals, antioxidant pigments, and phytochemicals in the leaves of Amaranthus spinosus and Amaranthus viridis weedy species. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Schwitter, J.; Wacker, C.M.; Wilke, N.; Al-Saadi, N.; Sauer, E.; Huettle, K.; Schönberg, S.O.; Luchner, A.; Strohm, O.; Ahlstrom, H.; et al. MR-IMPACT II: Magnetic Resonance Imaging for Myocardial Perfusion Assessment in Coronary artery disease Trial: perfusion-cardiac magnetic resonance vs. single-photon emission computed tomography for the detection of coronary artery disease: a comparative multicentre, multivendor trial. Eur. Hear. J. 2012, 34, 775–781. [Google Scholar] [CrossRef]
- Sedmera, D. , Reckova, M., DeAlmeida, A., Sedmerova, M., Biermann, M., Volejnik, J., & Thompson, R. P. (2003). Functional and morphological evidence for a ventricular conduction system in zebrafish and Xenopus hearts. American Journal of Physiology-Heart and Circulatory Physiology, 284(4), H1152-H1160.
- Shah, S.M.A.; Akram, M.; Riaz, M.; Munir, N.; Rasool, G. Cardioprotective Potential of Plant-Derived Molecules: A Scientific and Medicinal Approach. Dose-Response 2019, 17. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Zuo, Z. Zebrafish (Danio rerio) as an excellent vertebrate model for the development, reproductive, cardiovascular, and neural and ocular development toxicity study of hazardous chemicals. Environ. Sci. Pollut. Res. 2020, 27, 43599–43614. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.M.; Danaei, G.; Farzadfar, F.; Stevens, G.A.; Woodward, M.; Wormser, D.; Kaptoge, S.; Whitlock, G.; Qiao, Q.; Lewington, S.; et al. The Age-Specific Quantitative Effects of Metabolic Risk Factors on Cardiovascular Diseases and Diabetes: A Pooled Analysis. PLOS ONE 2013, 8, e65174. [Google Scholar] [CrossRef] [PubMed]
- Stajer, D.; Bervar, M.; Horvat, M. CARDIOGENIC SHOCK FOLLOWING A SINGLE THERAPEUTIC ORAL DOSE OF VERAPAMIL. Int. J. Clin. Pr. 2001, 55, 69–70. [Google Scholar] [CrossRef]
- Sogorski, A.; Spindler, S.; Wallner, C.; Dadras, M.; Wagner, J.; Behr, B.; Lehnhardt, M.; Kolbenschlag, J. Optimizing remote ischemic conditioning (RIC) of cutaneous microcirculation in humans: Number of cycles and duration of acute effects. J. Plast. Reconstr. Aesthetic Surg. 2020, 74, 819–827. [Google Scholar] [CrossRef]
- Srivastava, D. , & Baldwin, H. S. (2001). Molecular determinants of cardiac development. Moss and Adams’ Heart Disease in Infants, Children, and Adolescents, Hugh D. Allen, Howard P. Gutgesell, et al., 3-23.
- Stevens, C.M.; Rayani, K.; Genge, C.E.; Singh, G.; Liang, B.; Roller, J.M.; Li, C.; Li, A.Y.; Tieleman, D.P.; van Petegem, F.; et al. Characterization of Zebrafish Cardiac and Slow Skeletal Troponin C Paralogs by MD Simulation and ITC. Biophys. J. 2016, 111, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Sullivan-Brown, J.; E Bisher, M.; Burdine, R.D. Embedding, serial sectioning and staining of zebrafish embryos using JB-4 resin. Nat. Protoc. 2010, 6, 46–55. [Google Scholar] [CrossRef]
- Swathy, M.; Saruladha, K. A comparative study of classification and prediction of Cardio-Vascular Diseases (CVD) using Machine Learning and Deep Learning techniques. ICT Express 2021, 8, 109–116. [Google Scholar] [CrossRef]
- Tavares, B.; Lopes, S.S. The Importance of Zebrafish in Biomedical Research. Acta medica Port. 2013, 26, 583–592. [Google Scholar] [CrossRef]
- Timmis, A.; Townsend, N.; Gale, C.P.; Torbica, A.; Lettino, M.; Petersen, S.E.; Mossialos, E.A.; Maggioni, A.P.; Kazakiewicz, D.; May, H.T.; et al. European Society of Cardiology: Cardiovascular Disease Statistics 2019. Eur. Heart J. 2020, 41, 12–85. [Google Scholar] [CrossRef]
- Timmis, A.; Vardas, P.; Townsend, N.; Torbica, A.; Katus, H.; De Smedt, D.; Gale, C.P.; Maggioni, A.P.; Petersen, S.E.; Huculeci, R.; et al. European Society of Cardiology: cardiovascular disease statistics 2021. Eur. Hear. J. 2022, 43, 716–799. [Google Scholar] [CrossRef] [PubMed]
- Ward, E.; Jemal, A.; Cokkinides, V.; Singh, G.K.; Cardinez, C.; Ghafoor, A.; Thun, M. Cancer Disparities by Race/Ethnicity and Socioeconomic Status. CA: A Cancer J. Clin. 2004, 54, 78–93. [Google Scholar] [CrossRef] [PubMed]
- White, R.; Rose, K.; Zon, L. Zebrafish cancer: the state of the art and the path forward. Nat. Rev. Cancer 2013, 13, 624–636. [Google Scholar] [CrossRef]
- Wiciński, M.; Socha, M.; Walczak, M.; Wódkiewicz, E.; Malinowski, B.; Rewerski, S.; Górski, K.; Pawlak-Osińska, K. Beneficial Effects of Resveratrol Administration—Focus on Potential Biochemical Mechanisms in Cardiovascular Conditions. Nutrients 2018, 10, 1813. [Google Scholar] [CrossRef]
- Williams, J.S.; Walker, R.J.; Egede, L.E. Achieving Equity in an Evolving Healthcare System: Opportunities and Challenges. Am. J. Med Sci. 2016, 351, 33–43. [Google Scholar] [CrossRef]
- World Health Organization. (2007). Prevention of cardiovascular disease. Pocket guidelines for assessment and management of cardiovascular risk. Africa: Who/Ish cardiovascular risk prediction charts for the African region. World Health Organization.
- Zhu, X.-Y.; Wu, S.-Q.; Guo, S.-Y.; Yang, H.; Xia, B.; Li, P.; Li, C.-Q. A Zebrafish Heart Failure Model for Assessing Therapeutic Agents. Zebrafish 2018, 15, 243–253. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




