Submitted:
09 December 2024
Posted:
10 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Limitations and Latest Advents of Molecular Dynamic Simulations in Sampling Conformational Ensembles in IDPs
3. The Emergence of Deep Learning methods in Protein Structure Prediction
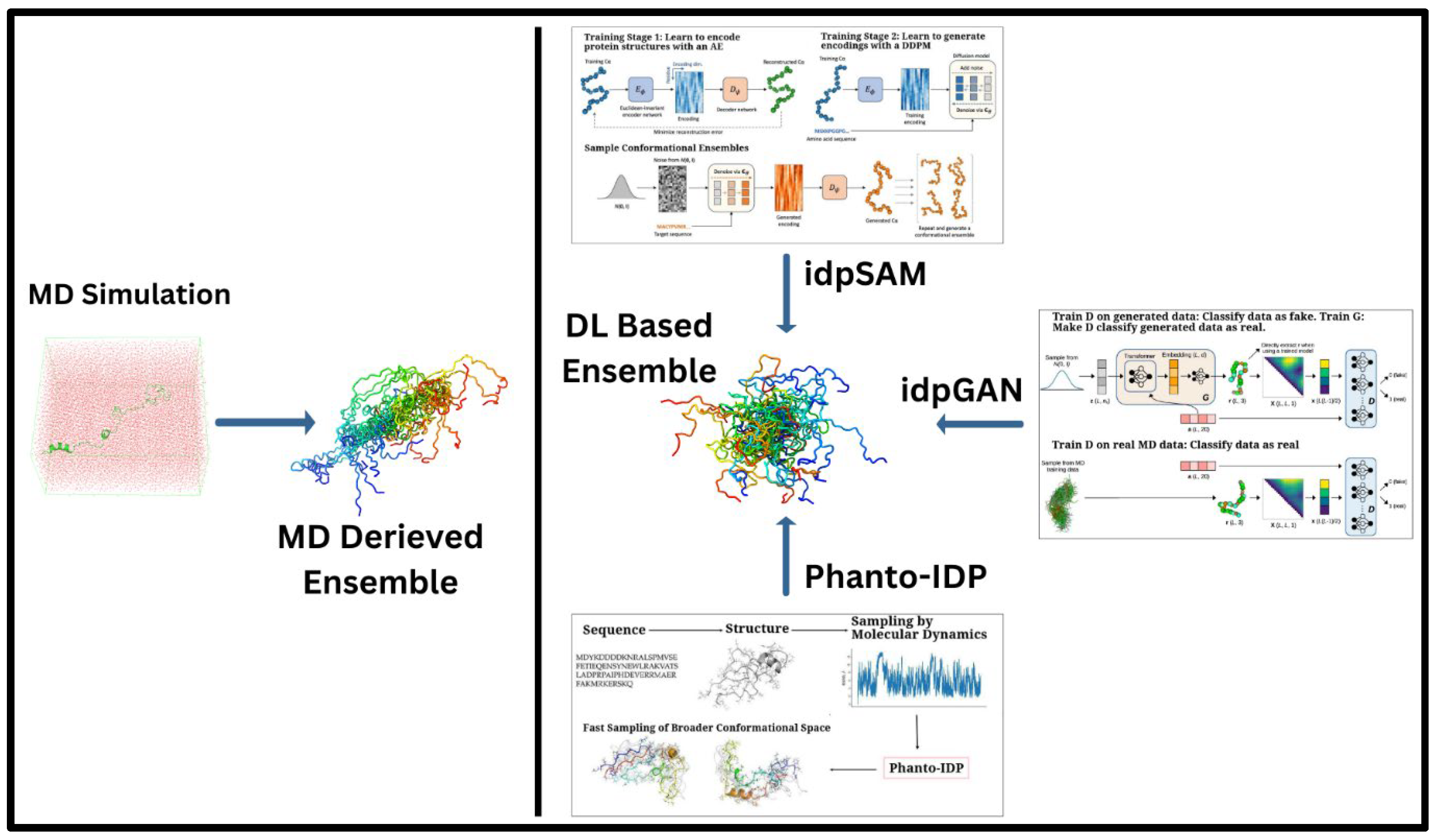
4. Deep Learning Models Employed in the Conformational Sampling of IDPs
4.1. Generative Adversarial Networks
4.2. Variational AutoEncoders
4.3. Transformers (AphaFold Pipelines)
4.4. Diffusion Models
5. Overcoming the Energy Landscape in IDPs
6. Enhanced Conformational Sampling using AI in MD Simulation
7. Comparative Efficiency: DL versus MD
8. Disadvantages of DL Over MD Simulations
9. Applications and Case Studies: Deep Learning in IDP Research
10. Discussion and Future Directions
Funding
Competing Interests
Acknowledgments
Author’s Contribution
Data Availability
References
- Abrams, C. , and Bussi, G. (2014). Enhanced Sampling in Molecular Dynamics Using Metadynamics, Replica-Exchange, and Temperature-Acceleration. Entropy. [CrossRef]
- Abramson, J. , Adler, J., Dunger, J., Evans, R., Green, T., Pritzel, A., et al. (2024). Accurate structure prediction of biomolecular interactions with AlphaFold 3. ( 2024). Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500. [CrossRef]
- Abyzov, A. , Blackledge, M., and Zweckstetter, M. (2022). Conformational Dynamics of Intrinsically Disordered Proteins Regulate Biomolecular Condensate Chemistry. ( 2022). Conformational Dynamics of Intrinsically Disordered Proteins Regulate Biomolecular Condensate Chemistry. Chem Rev 122, 6719–6748. [CrossRef]
- Aftab, A. , Sil, S., Nath, S., Basu, A., and Basu, S. (2024). Intrinsic Disorder and Other Malleable Arsenals of Evolved Protein Multifunctionality. J Mol Evol. [CrossRef]
- Ahmed, S. F. , Alam, Md. S. B., Hassan, M., Rozbu, M. R., Ishtiak, T., Rafa, N., et al. (2023). Deep learning modelling techniques: current progress, applications, advantages, and challenges. Artif Intell Rev, 1361. [Google Scholar] [CrossRef]
- Alzubaidi, L. , Zhang, J., Humaidi, A. J., Al-Dujaili, A., Duan, Y., Al-Shamma, O., et al. (2021). Review of deep learning: concepts, CNN architectures, challenges, applications, future directions. Journal of Big Data. [CrossRef]
- Anstine, D. M. , and Isayev, O. (2023). Generative Models as an Emerging Paradigm in the Chemical Sciences. ( 2023). Generative Models as an Emerging Paradigm in the Chemical Sciences. J. Am. Chem. Soc. 145, 8736–8750. [CrossRef]
- Arai, M. , Suetaka, S., and Ooka, K. (2024). Dynamics and interactions of intrinsically disordered proteins. ( 2024). Dynamics and interactions of intrinsically disordered proteins. Current Opinion in Structural Biology 84, 102734. [CrossRef]
- Aranganathan, A. , Gu, X., Wang, D., Vani, B., and Tiwary, P. (2024). Modeling Boltzmann weighted structural ensembles of proteins using AI based methods. [CrossRef]
- Baek, M. , DiMaio, F., Anishchenko, I., Dauparas, J., Ovchinnikov, S., Lee, G. R., et al. (2021). Accurate prediction of protein structures and interactions using a three-track neural network. Science. [CrossRef]
- Bah, A. , and Forman-Kay, J. D. (2016). Modulation of Intrinsically Disordered Protein Function by Post-translational Modifications. J Biol Chem, 6705. [Google Scholar] [CrossRef]
- Balupuri, A. , Choi, K.-E., and Kang, N. S. (2020). Aggregation Mechanism of Alzheimer’s Amyloid β-Peptide Mediated by α-Strand/α-Sheet Structure. International Journal of Molecular Sciences. [CrossRef]
- Bandyopadhyay, A. , and Basu, S. (2020). Criticality in the conformational phase transition among self-similar groups in intrinsically disordered proteins: Probed by salt-bridge dynamics. Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics, 4047. [Google Scholar] [CrossRef]
- Bhattacharya, S. , and Lin, X. (2019). Recent Advances in Computational Protocols Addressing Intrinsically Disordered Proteins. ( 2019). Recent Advances in Computational Protocols Addressing Intrinsically Disordered Proteins. Biomolecules 9, 146. [CrossRef]
- Brosey, C. A. , and Tainer, J. A. (2019). Evolving SAXS versatility: solution X-ray scattering for macromolecular architecture, functional landscapes, and integrative structural biology. Current Opinion in Structural Biology. [CrossRef]
- Brotzakis, Z. F. , Zhang, S., and Vendruscolo, M. (2023). AlphaFold Prediction of Structural Ensembles of Disordered Proteins. 2023.01.19.524720. [CrossRef]
- Brown, B. P. , Stein, R. A., Meiler, J., and Mchaourab, H. S. (2024). Approximating Projections of Conformational Boltzmann Distributions with AlphaFold2 Predictions: Opportunities and Limitations. J. Chem. Theory Comput. 1447. [Google Scholar] [CrossRef]
- Bugge, K. , Brakti, I., Fernandes, C. B., Dreier, J. E., Lundsgaard, J. E., Olsen, J. G., et al. (2020). Interactions by Disorder – A Matter of Context. Front. Mol. Biosci. [CrossRef]
- Chandra, A. , Tünnermann, L., Löfstedt, T., and Gratz, R. (2023). Transformer-based deep learning for predicting protein properties in the life sciences. eLife. [CrossRef]
- Cheng, S. , Zhao, X., Lu, G., Fang, J., Yu, Z., Zheng, T., et al. (2023). FastFold: Reducing AlphaFold Training Time from 11 Days to 67 Hours. [CrossRef]
- Chennakesavalu, S. , and Rotskoff, G. M. (2024). Data-Efficient Generation of Protein Conformational Ensembles with Backbone-to-Side-Chain Transformers. J. Phys. Chem. B, 2123. [Google Scholar] [CrossRef]
- Chien, J.-T. (2019). “Chapter 7 - Deep Neural Network,” in Source Separation and Machine Learning, ed. J.-T. Chien (Academic Press), 259–320. [CrossRef]
- Choi, J.-M. , and Pappu, R. V. (2019). Improvements to the ABSINTH forcefield for proteins based on experimentally derived amino-acid specific backbone conformational statistics. Journal of chemical theory and computation. [CrossRef]
- Choi, S. R. , and Lee, M. (2023). Transformer Architecture and Attention Mechanisms in Genome Data Analysis: A Comprehensive Review. Biology (Basel). [CrossRef]
- Dishman, A. F. , and Volkman, B. F. (2018). Unfolding the Mysteries of Protein Metamorphosis. F. ( 2018). Unfolding the Mysteries of Protein Metamorphosis. ACS Chem Biol 13, 1438–1446. [CrossRef]
- Do, T. N. , Choy, W.-Y., and Karttunen, M. (2014). Accelerating the Conformational Sampling of Intrinsically Disordered Proteins. ( 2014). Accelerating the Conformational Sampling of Intrinsically Disordered Proteins. J. Chem. Theory Comput. 10, 5081–5094. [CrossRef]
- Elofsson, A. (2023). Progress at protein structure prediction, as seen in CASP15. Current Opinion in Structural Biology. [CrossRef]
- Erdős, G. , and Dosztányi, Z. (2024). Deep learning for intrinsically disordered proteins: From improved predictions to deciphering conformational ensembles. Current Opinion in Structural Biology. [CrossRef]
- Evans, R. , Ramisetty, S., Kulkarni, P., and Weninger, K. (2023). Illuminating Intrinsically Disordered Proteins with Integrative Structural Biology. ( 2023). Illuminating Intrinsically Disordered Proteins with Integrative Structural Biology. Biomolecules 13, 124. [CrossRef]
- Fan, J. , Li, Z., Alcaide, E., Ke, G., Huang, H., and Weinan, E. (2024). Accurate Conformation Sampling via Protein Structural Diffusion. 2024.05.20.594916. [CrossRef]
- Ferruz, N. , Heinzinger, M., Akdel, M., Goncearenco, A., Naef, L., and Dallago, C. (2023). From sequence to function through structure: Deep learning for protein design. Computational and Structural Biotechnology Journal. [CrossRef]
- Fischer, A.-L. M. , Tichy, A., Kokot, J., Hoerschinger, V. J., Wild, R. F., Riccabona, J. R., et al. (2024). The Role of Force Fields and Water Models in Protein Folding and Unfolding Dynamics. J. Chem. Theory Comput. 2333. [Google Scholar] [CrossRef]
- Fukuchi, S. , Sakamoto, S., Nobe, Y., Murakami, S. D., Amemiya, T., Hosoda, K., et al. (2012). IDEAL: Intrinsically Disordered proteins with Extensive Annotations and Literature. Nucleic Acids Research. [CrossRef]
- Galvelis, R. , and Sugita, Y. (2017). Neural Network and Nearest Neighbor Algorithms for Enhancing Sampling of Molecular Dynamics. ( 2017). Neural Network and Nearest Neighbor Algorithms for Enhancing Sampling of Molecular Dynamics. J. Chem. Theory Comput. 13, 2489–2500. [CrossRef]
- Ghafouri, H. , Lazar, T., Del Conte, A., Tenorio Ku, L. G., PED Consortium, Tompa, P., et al. (2024). PED in 2024: improving the community deposition of structural ensembles for intrinsically disordered proteins. Nucleic Acids Research. [CrossRef]
- Gichoya, J. W. , Thomas, K., Celi, L. A., Safdar, N., Banerjee, I., Banja, J. D., et al. (2023). AI pitfalls and what not to do: mitigating bias in AI. Br J Radiol, 3002. [Google Scholar] [CrossRef]
- Giri, N. , Roy, R. S., and Cheng, J. (2023). Deep learning for reconstructing protein structures from cryo-EM density maps: recent advances and future directions. Curr Opin Struct Biol. [CrossRef]
- Gomes, G.-N. W. , Krzeminski, M., Namini, A., Martin, E. W., Mittag, T., Head-Gordon, T., et al. (2020). Conformational ensembles of an intrinsically disordered protein consistent with NMR, SAXS and single-molecule FRET. J Am Chem Soc, 1571. [Google Scholar] [CrossRef]
- Gong, X. , Zhang, Y., and Chen, J. (2021). Advanced Sampling Methods for Multiscale Simulation of Disordered Proteins and Dynamic Interactions. ( 2021). Advanced Sampling Methods for Multiscale Simulation of Disordered Proteins and Dynamic Interactions. Biomolecules 11, 1416. [CrossRef]
- Gopal, S. M. , Wingbermühle, S., Schnatwinkel, J., Juber, S., Herrmann, C., and Schäfer, L. V. (2021). Conformational Preferences of an Intrinsically Disordered Protein Domain: A Case Study for Modern Force Fields. J. Phys. Chem. B. [CrossRef]
- Guan, X. , Tang, Q.-Y., Ren, W., Chen, M., Wang, W., Wolynes, P. G., et al. (2024). Predicting protein conformational motions using energetic frustration analysis and AlphaFold2. ( 2024). Predicting protein conformational motions using energetic frustration analysis and AlphaFold2. Proceedings of the National Academy of Sciences 121, e2410662121. [CrossRef]
- Gui, J. , Sun, Z., Wen, Y., Tao, D., and Ye, J. (2020). A Review on Generative Adversarial Networks: Algorithms, Theory, and Applications. [CrossRef]
- Gupta, A. , Dey, S., Hicks, A., and Zhou, H.-X. (2022). Artificial intelligence guided conformational mining of intrinsically disordered proteins. ( 2022). Artificial intelligence guided conformational mining of intrinsically disordered proteins. Commun Biol 5, 1–11. [CrossRef]
- Guvench, O. , and MacKerell, A. D. (2008). Comparison of protein force fields for molecular dynamics simulations. D. ( 2008). Comparison of protein force fields for molecular dynamics simulations. Methods Mol Biol 443, 63–88. [CrossRef]
- Han, M. , Xu, J., and Ren, Y. (2017). Sampling conformational space of intrinsically disordered proteins in explicit solvent: Comparison between well-tempered ensemble approach and solute tempering method. J Mol Graph Model. [CrossRef]
- Hatos, A. , Monzon, A. M., Tosatto, S. C. E., Piovesan, D., and Fuxreiter, M. (2022). FuzDB: a new phase in understanding fuzzy interactions. Nucleic Acids Research. [CrossRef]
- Ho, J. , Jain, A., and Abbeel, P. (2020). Denoising Diffusion Probabilistic Models. [CrossRef]
- Hollingsworth, S. A. , and Dror, R. O. (2018). Molecular dynamics simulation for all. O. ( 2018). Molecular dynamics simulation for all. Neuron 99, 1129–1143. [CrossRef]
- Hu, Z. , Sun, T., Chen, W., Nordenskiöld, L., and Lu, L. (2024). Refined Bonded Terms in Coarse-Grained Models for Intrinsically Disordered Proteins Improve Backbone Conformations. J. Phys. Chem. B, 6508. [Google Scholar] [CrossRef]
- Huang, J. , Rauscher, S., Nawrocki, G., Ran, T., Feig, M., de Groot, B. L., et al. (2017). CHARMM36m: an improved force field for folded and intrinsically disordered proteins. Nat Methods. [CrossRef]
- Invernizzi, M. , Krämer, A., Clementi, C., and Noé, F. (2022). Skipping the Replica Exchange Ladder with Normalizing Flows. ( 2022). Skipping the Replica Exchange Ladder with Normalizing Flows. J Phys Chem Lett 13, 11643–11649. [CrossRef]
- Jagtap, A. D. , Kharazmi, E., and Karniadakis, G. E. (2020). Conservative physics-informed neural networks on discrete domains for conservation laws: Applications to forward and inverse problems. Computer Methods in Applied Mechanics and Engineering, 3028. [Google Scholar] [CrossRef]
- Janson, G. , and Feig, M. Transferable deep generative modeling of intrinsically disordered protein conformations. 2024.02.08.57 9522. [CrossRef]
- Janson, G. , Valdes-Garcia, G., Heo, L., and Feig, M. (2023). Direct generation of protein conformational ensembles via machine learning. ( 2023). Direct generation of protein conformational ensembles via machine learning. Nat Commun 14, 774. [CrossRef]
- Jing, B. , Berger, B., and Jaakkola, T. (2024). AlphaFold Meets Flow Matching for Generating Protein Ensembles. [CrossRef]
- Jones, M. S. , Shmilovich, K., and Ferguson, A. L. (2023). DiAMoNDBack: Diffusion-Denoising Autoregressive Model for Non-Deterministic Backmapping of Cα Protein Traces. J. Chem. Theory Comput. 7923. [Google Scholar] [CrossRef]
- Joshi, P. , and Vendruscolo, M. (2015). Druggability of Intrinsically Disordered Proteins. ( 2015). Druggability of Intrinsically Disordered Proteins. Adv Exp Med Biol 870, 383–400. [CrossRef]
- Jumper, J. , Evans, R., Pritzel, A., Green, T., Figurnov, M., Ronneberger, O., et al. (2021). Highly accurate protein structure prediction with AlphaFold. ( 2021). Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589. [CrossRef]
- Kingma, D. P. , and Welling, M. (2022). Auto-Encoding Variational Bayes. [CrossRef]
- Klein, L. , Foong, A. Y. K., Fjelde, T. E., Mlodozeniec, B., Brockschmidt, M., Nowozin, S., et al. (2023a). Timewarp: Transferable Acceleration of Molecular Dynamics by Learning Time-Coarsened Dynamics. [CrossRef]
- Klein, L. , Krämer, A., and Noé, F. (2023b). Equivariant flow matching. [CrossRef]
- Klein, L. , and Noé, F. (2024). Transferable Boltzmann Generators. [CrossRef]
- Koval, A. , Sharif Mansouri, S., and Kanellakis, C. (2023). “Chapter 10 - Machine learning for ARWs,” in Aerial Robotic Workers, eds. G. Nikolakopoulos, S. Sharif Mansouri, and C. Kanellakis (Butterworth-Heinemann), 159–174. [CrossRef]
- Krieger, J. M. , Fusco, G., Lewitzky, M., Simister, P. C., Marchant, J., Camilloni, C., et al. (2014). Conformational Recognition of an Intrinsically Disordered Protein. ( 2014). Conformational Recognition of an Intrinsically Disordered Protein. Biophys J 106, 1771–1779. [CrossRef]
- Kulkarni, P. , Achuthan, S., Bhattacharya, S., Jolly, M. K., Kotnala, S., Leite, V. B. P., et al. (2021). Protein conformational dynamics and phenotypic switching. Biophys Rev, 1138. [Google Scholar] [CrossRef]
- Kulkarni, P. , Leite, V. B. P., Roy, S., Bhattacharyya, S., Mohanty, A., Achuthan, S., et al. (2022). Intrinsically disordered proteins: Ensembles at the limits of Anfinsen’s dogma. Biophys Rev (Melville). [CrossRef]
- Kumar, N. , and Srivastava, R. (2024). Deep learning in structural bioinformatics: current applications and future perspectives. Brief Bioinform. [CrossRef]
- Latham, A. P. , and Zhang, B. (2019). Improving Coarse-Grained Protein Force Fields with Small-Angle X-ray Scattering Data. J. Phys. Chem. B, 1034. [Google Scholar] [CrossRef]
- Lee, H. , Turilli, M., Jha, S., Bhowmik, D., Ma, H., and Ramanathan, A. (2019). DeepDriveMD: Deep-Learning Driven Adaptive Molecular Simulations for Protein Folding., (IEEE Computer Society), 12–19. [CrossRef]
- Li, J. , Beaudoin, C., and Ghosh, S. (2023). Energy-based generative models for target-specific drug discovery. Front. Mol. Med. [CrossRef]
- Li, S. , Li, M., Wang, Y., He, X., Zheng, N., Zhang, J., et al. (2024). Improving AlphaFlow for Efficient Protein Ensembles Generation. [CrossRef]
- Lincoff, J. , Haghighatlari, M., Krzeminski, M., Teixeira, J. M. C., Gomes, G.-N. W., Gradinaru, C. C., et al. (2020). Extended experimental inferential structure determination method in determining the structural ensembles of disordered protein states. Commun Chem. [CrossRef]
- Lindorff-Larsen, K. , and Kragelund, B. B. (2021). On the Potential of Machine Learning to Examine the Relationship Between Sequence, Structure, Dynamics and Function of Intrinsically Disordered Proteins. B. ( Dynamics and Function of Intrinsically Disordered Proteins. Journal of Molecular Biology 433, 167196. [CrossRef]
- Liu, Y. , Yang, Z., Yu, Z., Liu, Z., Liu, D., Lin, H., et al. (2023). Generative artificial intelligence and its applications in materials science: Current situation and future perspectives. Journal of Materiomics. [CrossRef]
- Liu, Y. , Yu, Z. 2024.10.04.61 6517. [CrossRef]
- Liu, Z. , and He, K. (2024). A Decade’s Battle on Dataset Bias: Are We There Yet? [CrossRef]
- López, O. A. M. , López, A. M., and Crossa, D. J. (2022). “Overfitting, Model Tuning, and Evaluation of Prediction Performance,” in Multivariate Statistical Machine Learning Methods for Genomic Prediction [Internet], (Springer). [CrossRef]
- Lu, J. , Zhong, B., Zhang, Z., and Tang, J. (2024). Str2Str: A Score-based Framework for Zero-shot Protein Conformation Sampling. [CrossRef]
- Maiti, S. , Singh, A., Maji, T., Saibo, N. V., and De, S. (2024). Experimental methods to study the structure and dynamics of intrinsically disordered regions in proteins. ( 2024). Experimental methods to study the structure and dynamics of intrinsically disordered regions in proteins. Current Research in Structural Biology 7, 100138. [CrossRef]
- Mansoor, S. , Baek, M., Park, H., Lee, G. R., and Baker, D. (2024). Protein Ensemble Generation Through Variational Autoencoder Latent Space Sampling. ( 2024). Protein Ensemble Generation Through Variational Autoencoder Latent Space Sampling. J. Chem. Theory Comput. 20, 2689–2695. [CrossRef]
- Martinelli, A. H. S. , Lopes, F. C., John, E. B. O., Carlini, C. R., and Ligabue-Braun, R. (2019). Modulation of Disordered Proteins with a Focus on Neurodegenerative Diseases and Other Pathologies. ( 2019). Modulation of Disordered Proteins with a Focus on Neurodegenerative Diseases and Other Pathologies. Int J Mol Sci 20, 1322. [CrossRef]
- Mu, J. , Liu, H., Zhang, J., Luo, R., and Chen, H.-F. (2021). Recent Force Field Strategies for Intrinsically Disordered Proteins. ( 2021). Recent Force Field Strategies for Intrinsically Disordered Proteins. J Chem Inf Model 61, 1037–1047. [CrossRef]
- Munappy, A. R. , Bosch, J., Olsson, H. H., Arpteg, A., and Brinne, B. (2022). Data management for production quality deep learning models: Challenges and solutions. Journal of Systems and Software, 1359. [Google Scholar] [CrossRef]
- Mura, C. , Draizen, E. J., and Bourne, P. E. (2018). Structural biology meets data science: does anything change? Current Opinion in Structural Biology. [CrossRef]
- Nikolados, E.-M. , Wongprommoon, A., Aodha, O. M., Cambray, G., and Oyarzún, D. A. (2022). Accuracy and data efficiency in deep learning models of protein expression. Nat Commun. [CrossRef]
- Noé, F. , Olsson, S., Köhler, J., and Wu, H. (2019). Boltzmann generators: Sampling equilibrium states of many-body systems with deep learning. Science, 1147. [Google Scholar] [CrossRef]
- Orosz, F. , and Ovádi, J. (2011). Proteins without 3D structure: definition, detection and beyond. Bioinformatics, 1454. [Google Scholar] [CrossRef]
- Pakhrin, S. C. , Shrestha, B., Adhikari, B., and KC, D. B. (2021). Deep Learning-Based Advances in Protein Structure Prediction. Int J Mol Sci. [CrossRef]
- Patel, Y. , and Tewari, A. (2022). RL Boltzmann Generators for Conformer Generation in Data-Sparse Environments. Available at: http://arxiv.org/abs/2211.10771 (Accessed , 2024). 28 October.
- Piovesan, D. , Necci, M., Escobedo, N., Monzon, A. M., Hatos, A., Mičetić, I., et al. (2021). MobiDB: intrinsically disordered proteins in 2021. Nucleic Acids Research. [CrossRef]
- Plaxco, K. W. , Simons, K. T., and Baker, D. (1998). Contact order, transition state placement and the refolding rates of single domain proteins1. ( transition state placement and the refolding rates of single domain proteins1. Journal of Molecular Biology 277, 985–994. [CrossRef]
- Prapas, I. , Derakhshan, B., Mahdiraji, A. R., and Markl, V. (2021). Continuous Training and Deployment of Deep Learning Models. ( 2021). Continuous Training and Deployment of Deep Learning Models. Datenbank Spektrum 21, 203–212. [CrossRef]
- Prašnikar, E. , Ljubič, M., Perdih, A., and Borišek, J. (2024). Machine learning heralding a new development phase in molecular dynamics simulations. ( 2024). Machine learning heralding a new development phase in molecular dynamics simulations. Artif Intell Rev 57, 102. [CrossRef]
- Qi, R. , Wei, G., Ma, B., and Nussinov, R. (2018). Replica Exchange Molecular Dynamics: A Practical Application Protocol with Solutions to Common Problems and a Peptide Aggregation and Self-Assembly Example. Methods Mol Biol. [CrossRef]
- Raissi, M. , Perdikaris, P., and Karniadakis, G. E. (2019). Physics-informed neural networks: A deep learning framework for solving forward and inverse problems involving nonlinear partial differential equations. Journal of Computational Physics. [CrossRef]
- Robustelli, P. , Piana, S., and Shaw, D. E. (2018). Developing a molecular dynamics force field for both folded and disordered protein states. E. ( 2018). Developing a molecular dynamics force field for both folded and disordered protein states. Proc Natl Acad Sci U S A 115, E4758–E4766. [CrossRef]
- Roca-Martinez, J. , Lazar, T., Gavalda-Garcia, J., Bickel, D., Pancsa, R., Dixit, B., et al. (2022). Challenges in describing the conformation and dynamics of proteins with ambiguous behavior. Front. Mol. Biosci. [CrossRef]
- Roy, S. , Basu, S., Dasgupta, D., Bhattacharyya, D., and Banerjee, R. (2015). The Unfolding MD Simulations of Cyclophilin: Analyzed by Surface Contact Networks and Their Associated Metrics. PLOS ONE, 4217. [Google Scholar] [CrossRef]
- Roy, S. , Basu, S., Datta, A. K., Bhattacharyya, D., Banerjee, R., and Dasgupta, D. (2014). Equilibrium unfolding of cyclophilin from Leishmania donovani: Characterization of intermediate states. International Journal of Biological Macromolecules. [CrossRef]
- Roy, S. , Ghosh, P., Bandyopadhyay, A., and Basu, S. (2022). Capturing a Crucial ‘Disorder-to-Order Transition’ at the Heart of the Coronavirus Molecular Pathology—Triggered by Highly Persistent, Interchangeable Salt-Bridges. Vaccines. [CrossRef]
- Ruff, K. M. , and Pappu, R. V. (2021). AlphaFold and Implications for Intrinsically Disordered Proteins. V. ( 2021). AlphaFold and Implications for Intrinsically Disordered Proteins. Journal of Molecular Biology 433, 167208. [CrossRef]
- Ruzmetov, T. , Hung, T. I., Jonnalagedda, S. P., Chen, S., Fasihianifard, P., Guo, Z., et al. (2024). Sampling Conformational Ensembles of Highly Dynamic Proteins via Generative Deep Learning. 2024.05.05.592587. [CrossRef]
- Salladini, E. , Jørgensen, M. L. M., Theisen, F. F., and Skriver, K. (2020). Intrinsic Disorder in Plant Transcription Factor Systems: Functional Implications. Int J Mol Sci. [CrossRef]
- Samek, W. , Montavon, G., Vedaldi, A., Hansen, L. K., and Müller, K.-R. eds. (2019). Explainable AI: Interpreting, Explaining and Visualizing Deep Learning. [CrossRef]
- Sarker, I. H. (2021). Deep Learning: A Comprehensive Overview on Techniques, Taxonomy, Applications and Research Directions. SN COMPUT. SCI. [CrossRef]
- Schafer, N. P. , Kim, B. L., Zheng, W., and Wolynes, P. G. (2014). Learning To Fold Proteins Using Energy Landscape Theory. G. ( 2014). Learning To Fold Proteins Using Energy Landscape Theory. Isr J Chem 54, 1311–1337. [CrossRef]
- Schlick, T. , Portillo-Ledesma, S., Myers, C. G., Beljak, L., Chen, J., Dakhel, S., et al. (2021). Biomolecular Modeling and Simulation: A Prospering Multidisciplinary Field. Annual review of biophysics. [CrossRef]
- Schreiner, M. , Winther, O., and Olsson, S. (2023). Implicit Transfer Operator Learning: Multiple Time-Resolution Surrogates for Molecular Dynamics. [CrossRef]
- Scollo, F. , and Rosa, C. L. (2020). Amyloidogenic Intrinsically Disordered Proteins: New Insights into Their Self-Assembly and Their Interaction with Membranes. Life. [CrossRef]
- Sengupta, U. , and Kayed, R. (2022). Amyloid β, Tau, and α-Synuclein aggregates in the pathogenesis, prognosis, and therapeutics for neurodegenerative diseases. Prog Neurobiol, 2270. [Google Scholar] [CrossRef]
- Shamsi, Z. , Cheng, K. J., and Shukla, D. (2018). Reinforcement Learning Based Adaptive Sampling: REAPing Rewards by Exploring Protein Conformational Landscapes. J Phys Chem B, 8395. [Google Scholar] [CrossRef]
- Shaw, D. E. , Deneroff, M. M., Dror, R. O., Kuskin, J. S., Larson, R. H., Salmon, J. K., et al. (2008). Anton, a special-purpose machine for molecular dynamics simulation. Commun. ACM. [CrossRef]
- Shrestha, U. R. , Smith, J. C., and Petridis, L. (2021). Full structural ensembles of intrinsically disordered proteins from unbiased molecular dynamics simulations. ( 2021). Full structural ensembles of intrinsically disordered proteins from unbiased molecular dynamics simulations. Commun Biol 4, 1–8. [CrossRef]
- Sickmeier, M. , Hamilton, J. A., LeGall, T., Vacic, V., Cortese, M. S., Tantos, A., et al. (2007). DisProt: the Database of Disordered Proteins. Nucleic Acids Res. [CrossRef]
- Smyth, M. S. , and Martin, J. H. J. (2000). x Ray crystallography. Mol Pathol, /: Available at: https, 6 December 1186. [Google Scholar]
- Son, A. , Kim, W., Park, J., Lee, W., Lee, Y., Choi, S., et al. (2024). Utilizing Molecular Dynamics Simulations, Machine Learning, Cryo-EM, and NMR Spectroscopy to Predict and Validate Protein Dynamics. Int J Mol Sci. [CrossRef]
- Song, D. , Liu, H., Luo, R., and Chen, H.-F. (2020). Environment-Specific Force Field for Intrinsically Disordered and Ordered Proteins. J Chem Inf Model, 2267. [Google Scholar] [CrossRef]
- Song, D. , Luo, R., and Chen, H.-F. (2017). The IDP-Specific Force Field ff14IDPSFF Improves the Conformer Sampling of Intrinsically Disordered Proteins. J. Chem. Inf. Model. 1178. [Google Scholar] [CrossRef]
- Su, J. , Song, Y., Zhu, Z., Huang, X., Fan, J., Qiao, J., et al. (2024). Cell–cell communication: new insights and clinical implications. Sig Transduct Target Ther. [CrossRef]
- Sullivan, S. S. , and Weinzierl, R. O. J. (2020). Optimization of Molecular Dynamics Simulations of c-MYC1-88—An Intrinsically Disordered System. Life (Basel). [CrossRef]
- Taneja, I. , and Lasker, K. (2024). Machine-learning-based methods to generate conformational ensembles of disordered proteins. Biophysical Journal. [CrossRef]
- Taye, M. M. (2023). Understanding of Machine Learning with Deep Learning: Architectures, Workflow, Applications and Future Directions. Computers. [CrossRef]
- Teixeira, J. M. C. , Liu, Z. H., Namini, A., Li, J., Vernon, R. M., Krzeminski, M., et al. (2022). IDPConformerGenerator: A Flexible Software Suite for Sampling the Conformational Space of Disordered Protein States. J Phys Chem A, 6003. [Google Scholar] [CrossRef]
- Trivedi, R. , and Nagarajaram, H. A. (2022). Intrinsically Disordered Proteins: An Overview. International Journal of Molecular Sciences. [CrossRef]
- Tsafou, K. , Tiwari, P. B., Forman-Kay, J. D., Metallo, S. J., and Toretsky, J. A. (2018). Targeting Intrinsically Disordered Transcription Factors: Changing the Paradigm. Journal of Molecular Biology, 2341. [Google Scholar] [CrossRef]
- Uversky, V. N. (2013). The alphabet of intrinsic disorder. Intrinsically Disord Proteins. [CrossRef]
- Vani, B. P. , Aranganathan, A., Wang, D., and Tiwary, P. (2023). AlphaFold2-RAVE: From Sequence to Boltzmann Ranking. J. Chem. Theory Comput. 4354. [Google Scholar] [CrossRef]
- Vaswani, A. , Shazeer, N. , Parmar, N., Uszkoreit, J., Jones, L., Gomez, A. N., in Proceedings of the 31st International Conference on Neural Information Processing Systems, (Red Hook, NY, USA: Curran Associates Inc.), 6000–6010., et al. (2017). Attention is all you need. [Google Scholar]
- Velankar, S. , Burley, S. K., Kurisu, G., Hoch, J. C., and Markley, J. L. (2021). “The Protein Data Bank Archive,” in Structural Proteomics, ed. R. J. Owens (New York, NY: Springer US), 3–21. [CrossRef]
- Vignesh, U. , Parvathi, R., and Gokul Ram, K. (2024). Ensemble deep learning model for protein secondary structure prediction using NLP metrics and explainable AI. ( 2024). Ensemble deep learning model for protein secondary structure prediction using NLP metrics and explainable AI. Results in Engineering 24, 103435. [CrossRef]
- Wang, B. , and Li, W. (2024). Advances in the Application of Protein Language Modeling for Nucleic Acid Protein Binding Site Prediction. ( 2024). Advances in the Application of Protein Language Modeling for Nucleic Acid Protein Binding Site Prediction. Genes 15, 1090. [CrossRef]
- Wang, J. (Ed.) (2025). “8 - AlphaFold, the successful prediction of three-dimensional protein structures and its impact on structural biology,” in Proteins, (Academic Press), 295–335. [CrossRef]
- Wang, T. , He, X., Li, M., Li, Y., Bi, R., Wang, Y., et al. (2024a). Ab initio characterization of protein molecular dynamics with AI2BMD. ( 2024a). Ab initio characterization of protein molecular dynamics with AI2BMD. Nature, 1–9. [CrossRef]
- Wang, Y. , Ribeiro, J. M. L., and Tiwary, P. (2019). Past–future information bottleneck for sampling molecular reaction coordinate simultaneously with thermodynamics and kinetics. Nat Commun. [CrossRef]
- Wang, Y. , Wang, L., Shen, Y., Wang, Y., Yuan, H., Wu, Y., et al. (2024b). Protein Conformation Generation via Force-Guided SE(3) Diffusion Models. [CrossRef]
- Williams, J. K. , Yang, X., and Baum, J. (2018). Interactions between the Intrinsically Disordered Proteins β-Synuclein and α-Synuclein. Proteomics, 0010. [Google Scholar] [CrossRef]
- Wodak, S. J. , Vajda, S., Lensink, M. F., Kozakov, D., and Bates, P. A. (2023). Critical assessment of methods for predicting the 3D structure of proteins and protein complexes. A. ( 2023). Critical assessment of methods for predicting the 3D structure of proteins and protein complexes. Annu Rev Biophys 52, 183–206. [CrossRef]
- Wohl, S. , and Zheng, W. (2023). Interpreting Transient Interactions of Intrinsically Disordered Proteins. ( 2023). Interpreting Transient Interactions of Intrinsically Disordered Proteins. J Phys Chem B 127, 2395–2406. [CrossRef]
- Wright, P. E. , and Dyson, H. J. (2015). Intrinsically Disordered Proteins in Cellular Signaling and Regulation. J. ( 2015). Intrinsically Disordered Proteins in Cellular Signaling and Regulation. Nat Rev Mol Cell Biol 16, 18–29. [CrossRef]
- Wu, H. , Wolynes, P. G., and Papoian, G. A. (2018). AWSEM-IDP: A Coarse-Grained Force Field for Intrinsically Disordered Proteins. J. Phys. Chem. B, 1112. [Google Scholar] [CrossRef]
- Yang, L. , Daskalakis, C., and Karniadakis, G. E. (2022). Generative Ensemble Regression: Learning Particle Dynamics from Observations of Ensembles with Physics-informed Deep Generative Models. SIAM J. Sci. Comput. [CrossRef]
- Yang, L. , Zhang, D., and Karniadakis, G. E. (2020). Physics-Informed Generative Adversarial Networks for Stochastic Differential Equations. SIAM J. Sci. Comput. [CrossRef]
- Yang, Z. , Wang, Y., Ni, X., and Yang, S. (2023). DeepDRP: Prediction of intrinsically disordered regions based on integrated view deep learning architecture from transformer-enhanced and protein information. International Journal of Biological Macromolecules, 7390. [Google Scholar] [CrossRef]
- Zhang, D. , Wang, J., and Luo, F. (2024). Directly Denoising Diffusion Models. [CrossRef]
- Zhang, J. , Chen, D., Xia, Y., Huang, Y.-P., Lin, X., Han, X., et al. (2023a). Artificial Intelligence Enhanced Molecular Simulations. ( 2023a). Artificial Intelligence Enhanced Molecular Simulations. J. Chem. Theory Comput. 19, 4338–4350. [CrossRef]
- Zhang, L. , Wang, H., and E, W. (2018). Reinforced dynamics for enhanced sampling in large atomic and molecular systems. ( 2018). Reinforced dynamics for enhanced sampling in large atomic and molecular systems. The Journal of Chemical Physics 148, 124113. [CrossRef]
- Zhang, O. , Haghighatlari, M., Li, J., Liu, Z. H., Namini, A., Teixeira, J. M. C., et al. (2023b). Learning to evolve structural ensembles of unfolded and disordered proteins using experimental solution data. The Journal of Chemical Physics, 4113. [Google Scholar] [CrossRef]
- Zheng, L.-E. , Barethiya, S., Nordquist, E., and Chen, J. (2023). Machine Learning Generation of Dynamic Protein Conformational Ensembles. ( 2023). Machine Learning Generation of Dynamic Protein Conformational Ensembles. Molecules 28, 4047. [CrossRef]
- Zhu, J. , Li, Z., Tong, H., Lu, Z., Zhang, N., Wei, T., et al. (2024a). Phanto-IDP: compact model for precise intrinsically disordered protein backbone generation and enhanced sampling. Briefings in Bioinformatics. [CrossRef]
- Zhu, J. , Li, Z., Zhang, B., Zheng, Z., Zhong, B., Bai, J., et al. (2024b). Precise Generation of Conformational Ensembles for Intrinsically Disordered Proteins Using Fine-tuned Diffusion Models. 2024.05.05.592611. [CrossRef]
- Zhu, J. , Li, Z., Zheng, Z., Zhang, B., Zhong, B., Bai, J., et al. (2024c). Precise Generation of Conformational Ensembles for Intrinsically Disordered Proteins via Fine-tuned Diffusion Models. 2024.05.05.592611. [CrossRef]
- Zhu, J.-J. , Zhang, N.-J., Wei, T., and Chen, H.-F. (2023). Enhancing Conformational Sampling for Intrinsically Disordered and Ordered Proteins by Variational Autoencoder. ( 2023). Enhancing Conformational Sampling for Intrinsically Disordered and Ordered Proteins by Variational Autoencoder. Int J Mol Sci 24, 6896. [CrossRef]
| 1 | In the context of this review, IDPs refer to both completely and partially disordered proteins (IDPs, IDPRs). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



