Submitted:
09 December 2024
Posted:
10 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sample Preparation
2.2.1. Impurity Identification
CEF Peptide Pool
Peptide 20 and 23
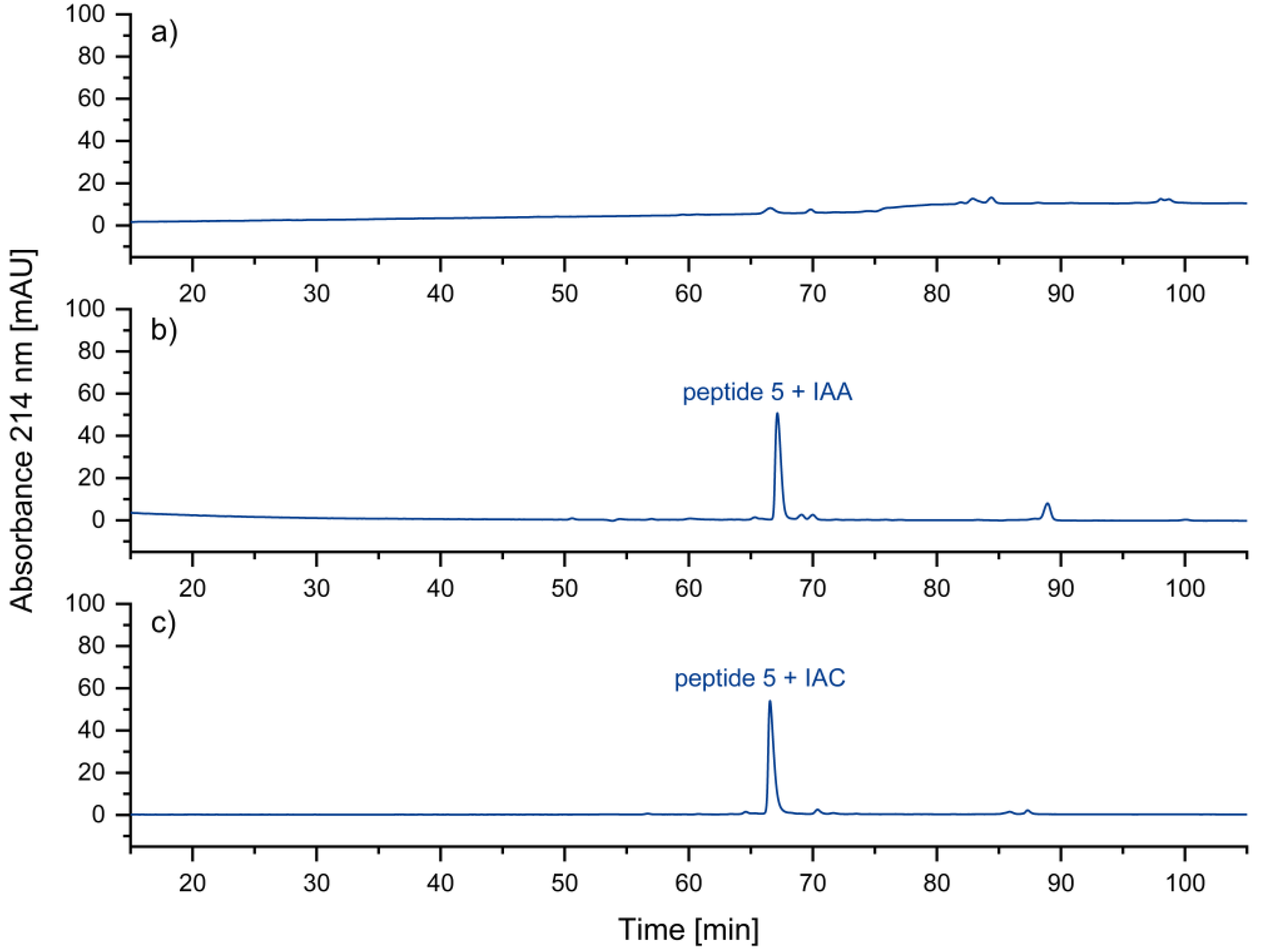
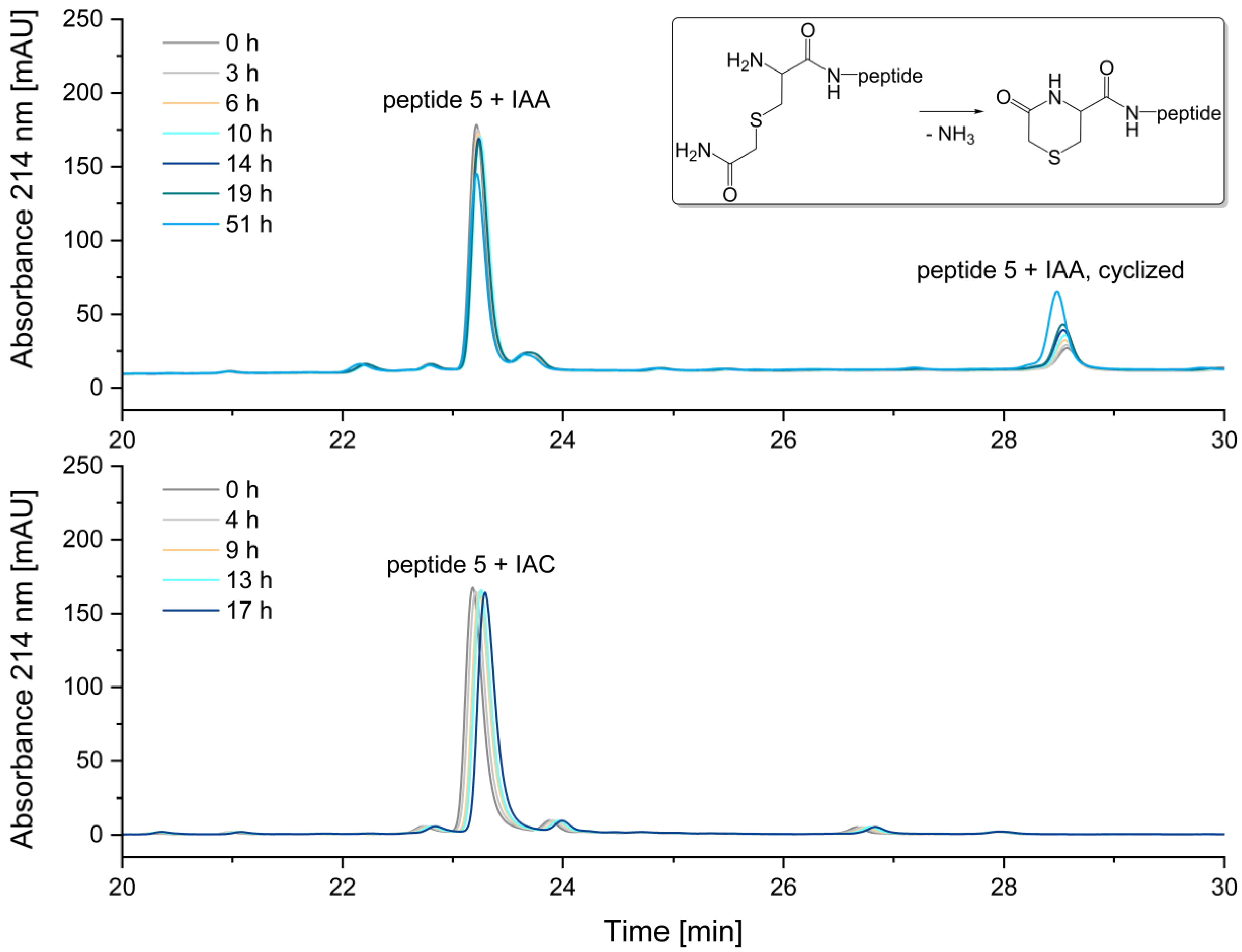
2.2.2. Alkylation of Cysteine-Residues
Reduced Peptide 5 (Figure 8a)
IAA-alkylation of Peptide 5
IAC-Alkylation of Peptide 5
2.3. UHPLC-HRMS Analysis
2.4. Data Processing
3. Results
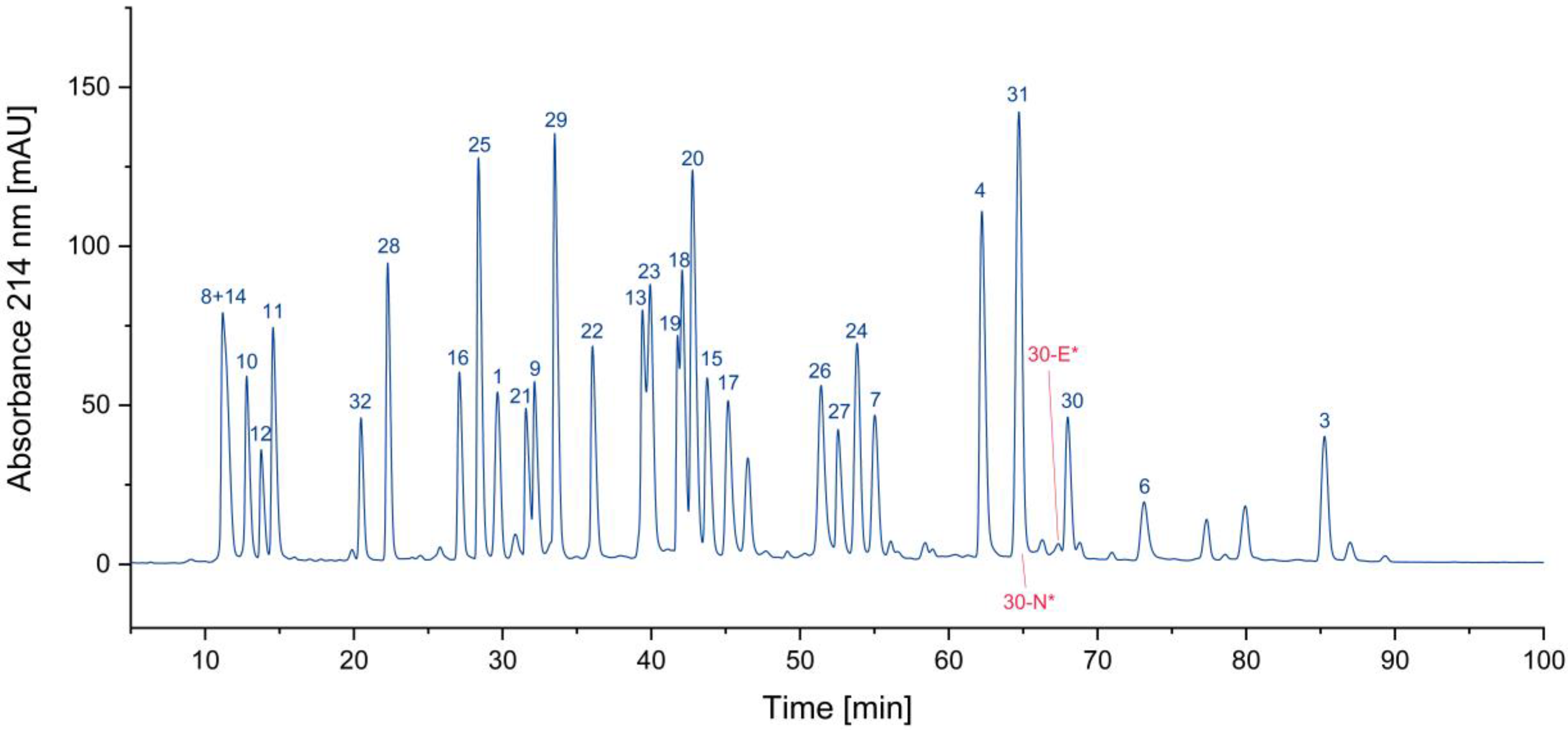
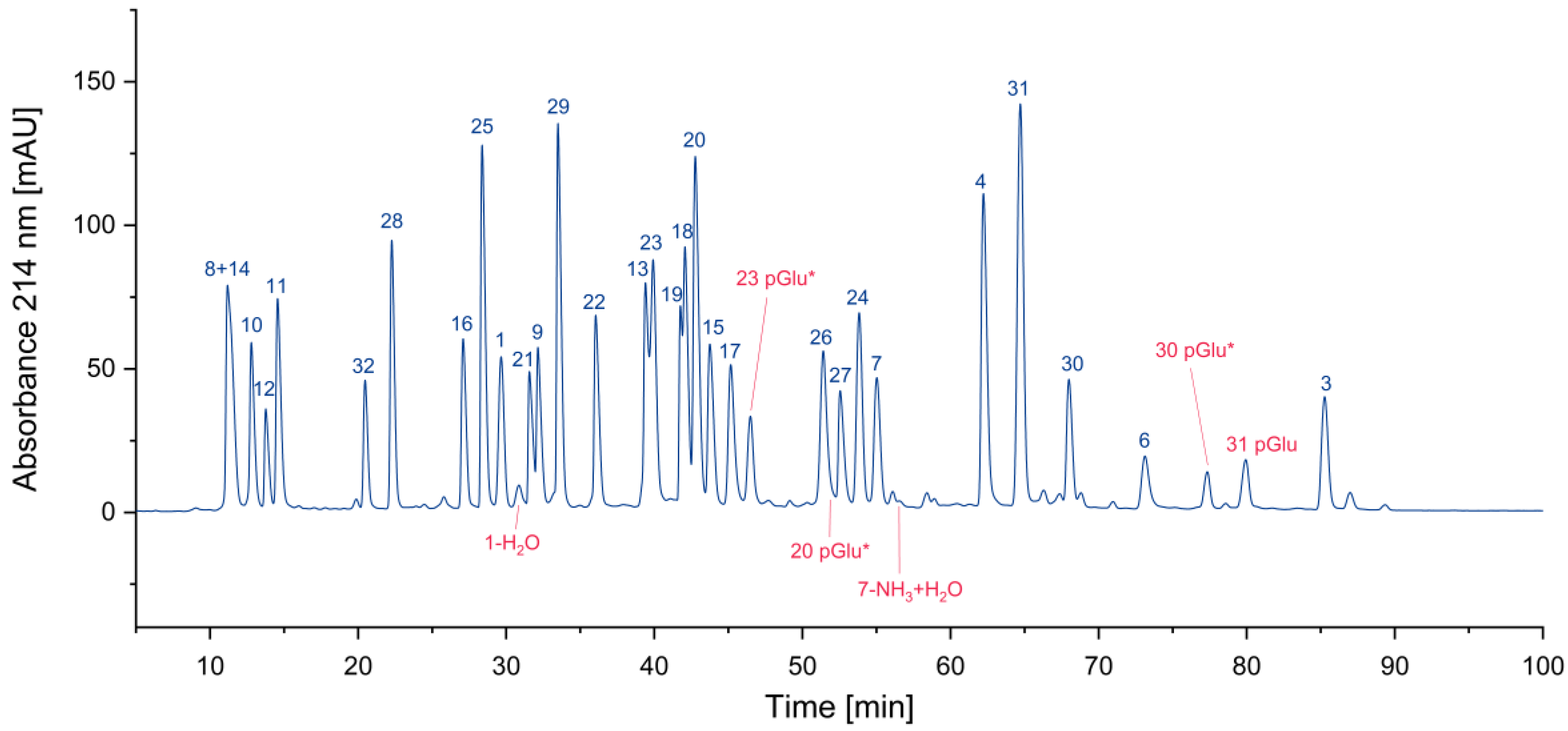
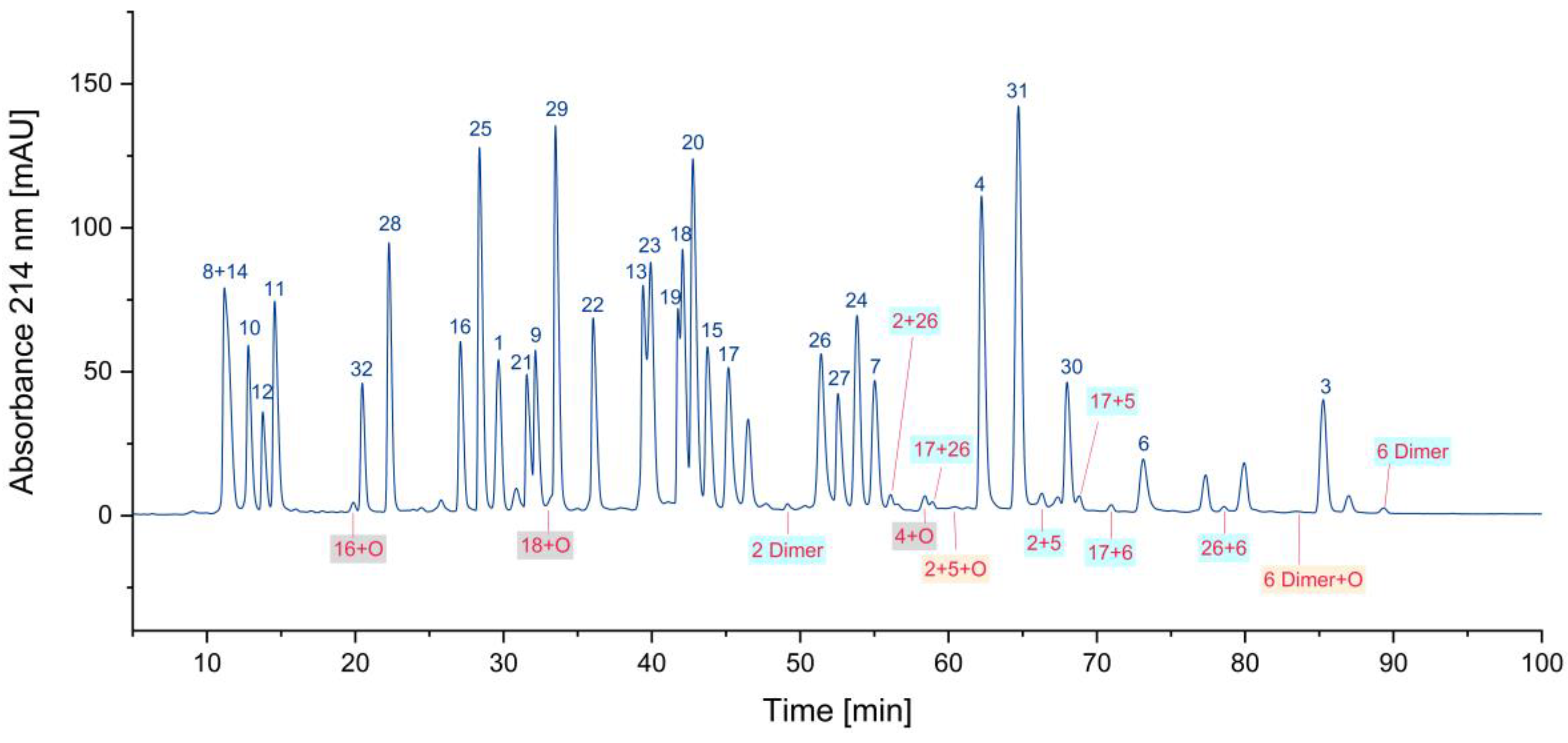
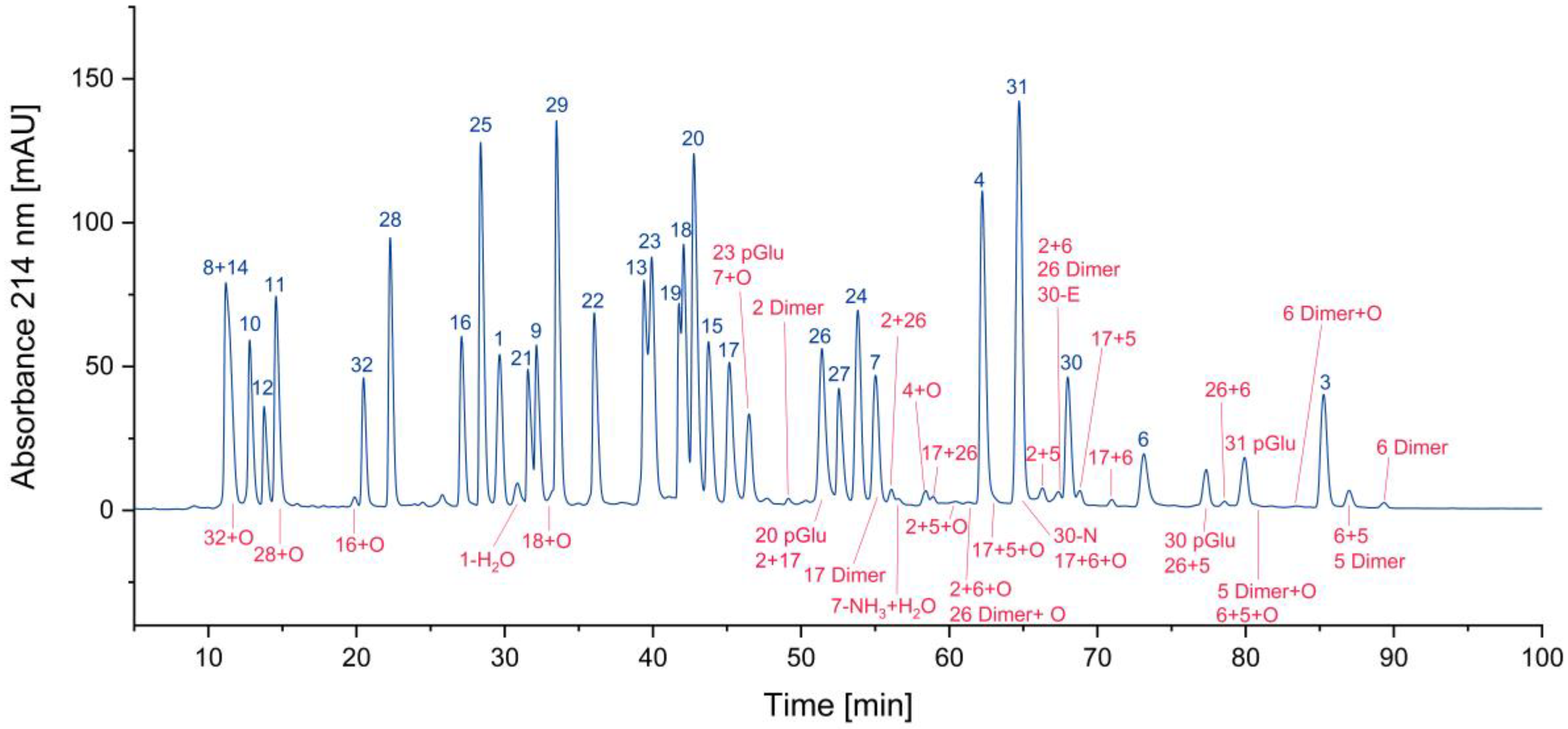
3.1. Impurity Identification
3.1.1. Deletion Peptides
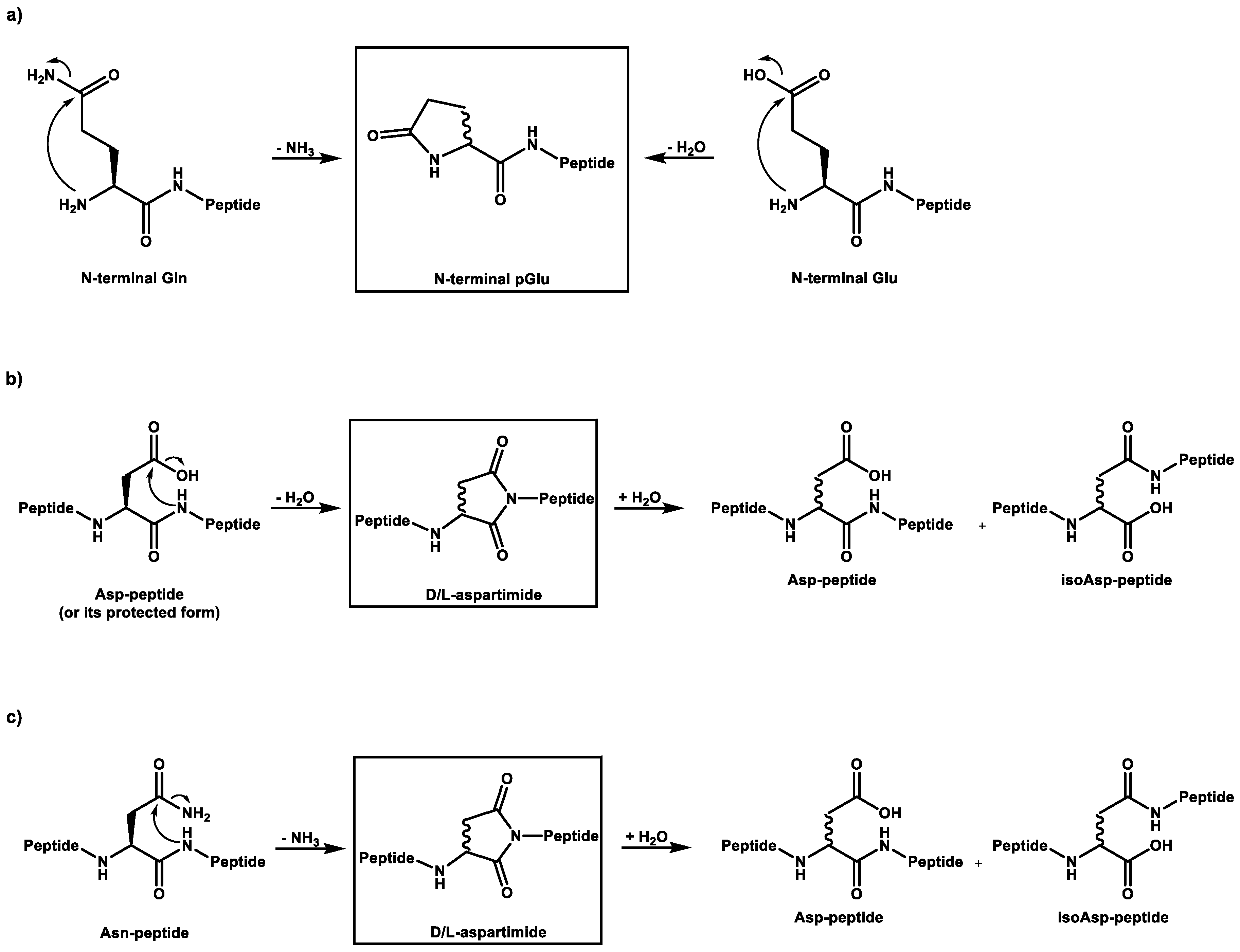
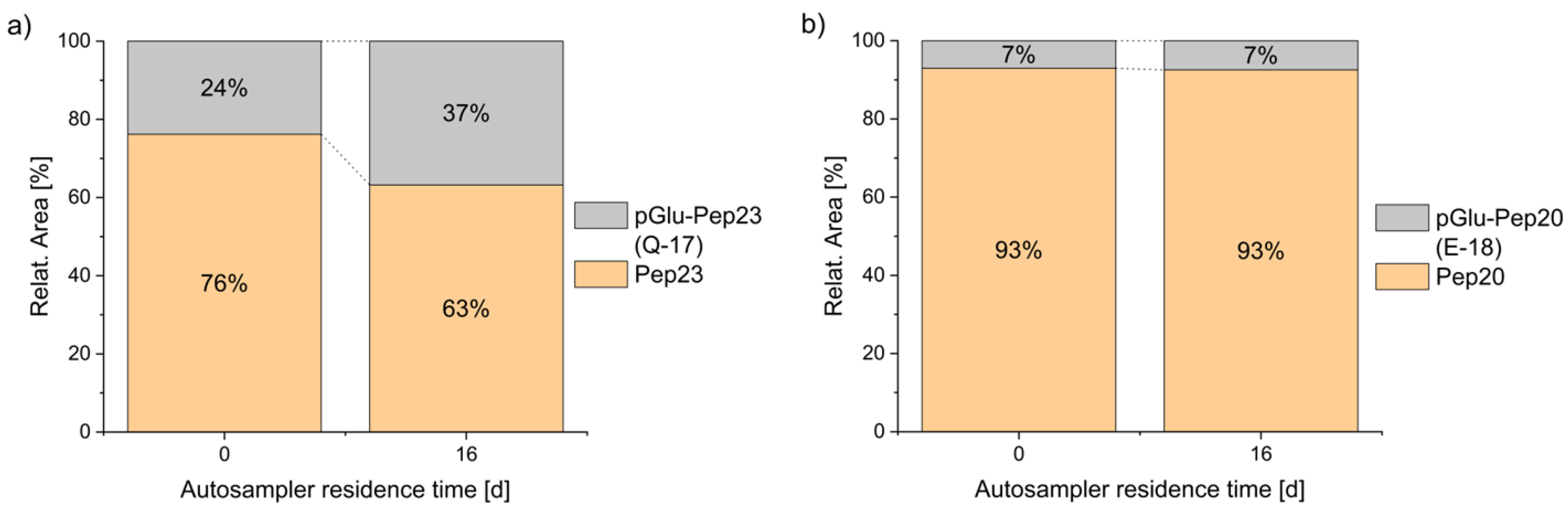
3.1.2. Cyclized Amino Acid Residues and Their Derivatives
3.1.3. Oxidized Peptides
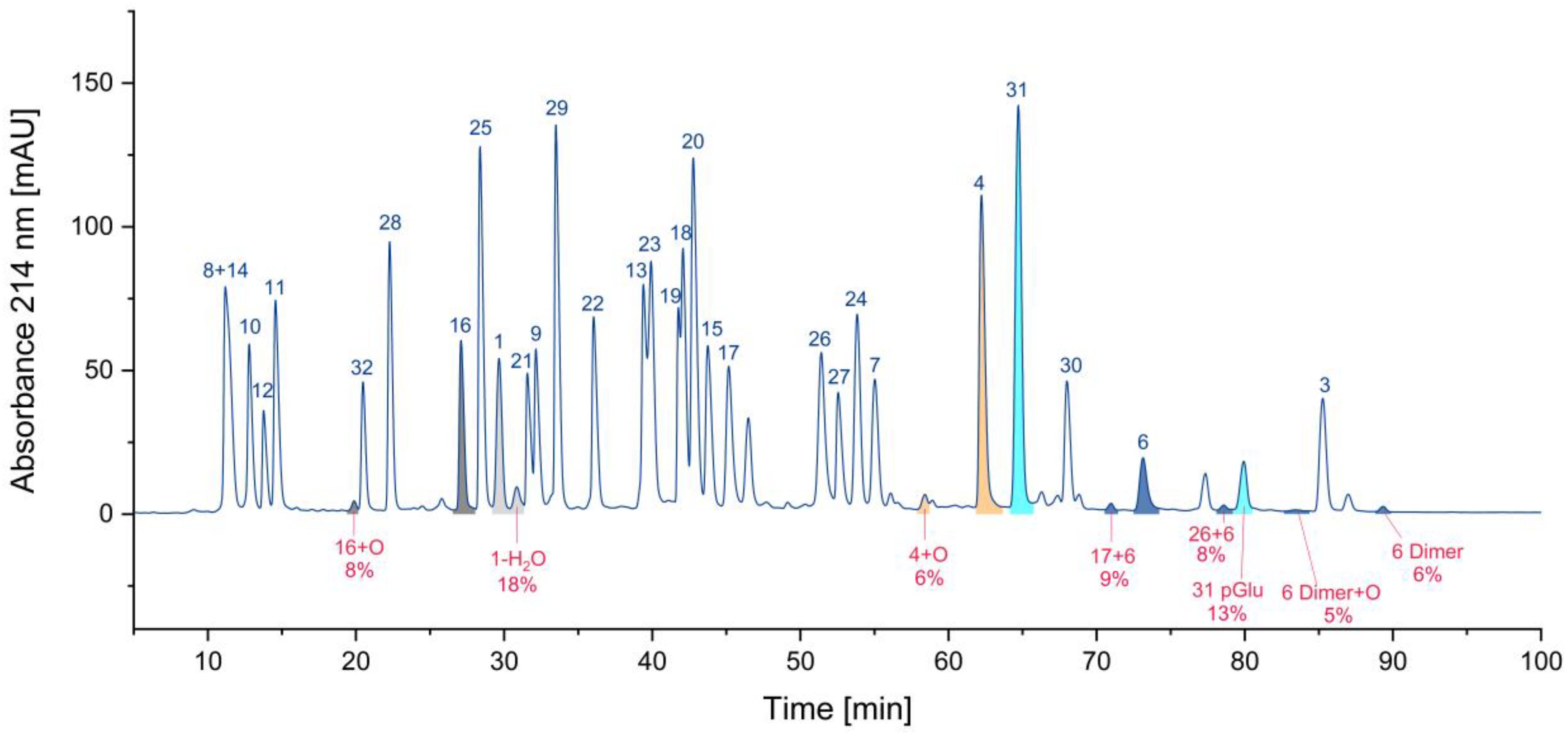
3.1.4. Overview
3.2. Alkylation of Cysteine Residues
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Weller, M.G. The Protocol Gap. Methods and Protocols 2021, 4, 12. [CrossRef]
- Patiny, L.; Borel, A. ChemCalc: A Building Block for Tomorrow's Chemical Infrastructure. J Chem Inf Model 2013, 53, 1223-1228. [CrossRef]
- Currier, J.R.; Kuta, E.G.; Turk, E.; Earhart, L.B.; Loomis-Price, L.; Janetzki, S.; Ferrari, G.; Birx, D.L.; Cox, J.H. A panel of MHC class I restricted viral peptides for use as a quality control for vaccine trial ELISPOT assays. J Immunol Methods 2002, 260, 157-172. [CrossRef]
- Bosc-Bierne, G.; Ewald, S.; Kreuzer, O.J.; Weller, M.G. Efficient Quality Control of Peptide Pools by UHPLC and Simultaneous UV and HRMS Detection. Separations 2024, 11. [CrossRef]
- Beck, A.; Bussat, M.C.; Klinguer-Hamour, C.; Goetsch, L.; Aubry, J.P.; Champion, T.; Julien, E.; Haeuw, J.F.; Bonnefoy, J.Y.; Corvaia, N. Stability and CTL activity of N-terminal glutamic acid containing peptides. J Pept Res 2001, 57, 528-538. [CrossRef]
- Gazme, B.; Boachie, R.T.; Tsopmo, A.; Udenigwe, C.C. Occurrence, properties and biological significance of pyroglutamyl peptides derived from different food sources. Food Sci Hum Well 2019, 8, 268-274. [CrossRef]
- Behrendt, R.; White, P.; Offer, J. Advances in Fmoc solid-phase peptide synthesis. J Pept Sci 2016, 22, 4-27. [CrossRef]
- Liu, S.; Moulton, K.R.; Auclair, J.R.; Zhou, Z.S. Mildly acidic conditions eliminate deamidation artifact during proteolysis: digestion with endoprotease Glu-C at pH 4.5. Amino Acids 2016, 48, 1059-1067. [CrossRef]
- Zhang, W.J.; Moldovan, I.; Targoni, O.S.; Subbramanian, R.A.; Lehmann, P.V. How much of Virus-Specific CD8 T Cell Reactivity is Detected with a Peptide Pool when Compared to Individual Peptides? Viruses 2012, 4, 2636-2649. [CrossRef]
- Kreuzer, O.J.; Weller, M.G.; Bosc-Bierne, G. Stabilization of N-terminal cysteines in HPLC-HRMS quality control of peptide pools. The Journal of Immunology 2023, 210, 159.109-159.109, doi: . [CrossRef]
- Müller, T.; Winter, D. Systematic Evaluation of Protein Reduction and Alkylation Reveals Massive Unspecific Side Effects by Iodine-containing Reagents. Mol Cell Proteomics 2017, 16, 1173-1187. [CrossRef]
- Geoghegan, K.F.; Hoth, L.R.; Tan, D.H.; Borzillerl, K.A.; Withka, J.M.; Boyd, J.G. Cyclization of N-terminal S-carbamoylmethylcysteine causing loss of 17 Da from peptides and extra peaks in peptide maps. J Proteome Res 2002, 1, 181-187. [CrossRef]
- Krokhin, O.V.; Ens, W.; Standing, K.G. Characterizing degradation products of peptides containing N-terminal Cys residues by (off-line high-performance liquid chromatography)/matrix-assisted laser desorption/ionization quadrupole time-of-flight measurements. Rapid Commun Mass Sp 2003, 17, 2528-2534. [CrossRef]
- Reimer, J.; Shamshurin, D.; Harder, M.; Yamchuk, A.; Spicer, V.; Krokhin, O.V. Effect of cyclization of N-terminal glutamine and carbamidomethyl-cysteine (residues) on the chromatographic behavior of peptides in reversed-phase chromatography. J Chromatogr A 2011, 1218, 5101-5107. [CrossRef]
- Neumann, K.; Farnung, J.; Baldauf, S.; Bode, J.W. Prevention of aspartimide formation during peptide synthesis using cyanosulfurylides as carboxylic acid-protecting groups. Nat Commun 2020, 11. [CrossRef]
| Peptide | Sequence | Exact mass [2,3] |
| 1 | VSDGGPNLY | 920.4240 |
| 2 | CTELKLSDY | 1070.4954 |
| 3 | GILGFVFTL | 965.5586 |
| 4 | FMYSDFHFI | 1205.5216 |
| 5 | CLGGLLTMV | 905.4715 |
| 6 | GLCTLVAML | 919.4871 |
| 7 | NLVPMVATV | 942.5208 |
| 8 | KTGGPIYKR | 1018.5924 |
| 9 | RVLSFIKGTK | 1147.7077 |
| 10 | ILRGSVAHK | 979.5927 |
| 11 | RVRAYTYSK | 1142.6196 |
| 12 | RLRAEAQVK | 1069.6356 |
| 13 | SIIPSGPLK | 910.5488 |
| 14 | AVFDRKSDAK | 1135.5986 |
| 15 | IVTDFSVIK | 1020.5856 |
| 16 | ATIGTAMYK | 954.4845 |
| 17 | DYCNVLNKEF | 1243.5543 |
| 18 | LPFDKTTVM | 1050.5420 |
| 19 | RPPIFIRRL | 1166.7400 |
| 20 | ELRSRYWAI | 1192.6353 |
| 21 | RAKFKQLL | 1002.6338 |
| 22 | FLRGRAYGL | 1051.5927 |
| 23 | QAKWRLQTL | 1142.6560 |
| 24 | SDEEEAIVAYTL | 1338.6191 |
| 25 | SRYWAIRTR | 1207.6574 |
| 26 | ASCMGLIY | 856.3823 |
| 27 | RRIYDLIEL | 1189.6819 |
| 28 | YPLHEQHGM | 1110.4917 |
| 29 | IPSINVHHY | 1078.5560 |
| 30 | EENLLDFVRF | 1280.6401 |
| 31 | EFFWDANDIY | 1318.5506 |
| 32 | TPRVTGGGAM | 945.4702 |
| Peptide | Sequence | ∆ mass [Da] | Impurity |
| 1 | VSDGGPNLY | -18 | aspartimide |
| 2 | CTELKLSDY | - | |
| 4 | FMYSDFHFI | - | |
| 7 | NLVPMVATV | +1 | deamidation |
| 12 | RLRAEAQVK | - | |
| 14 | AVFDRKSDAK | - | |
| 15 | IVTDFSVIK | - | |
| 17 | DYCNVLNKEF | - | |
| 18 | LPFDKTTVM | - | |
| 20 | ELRSRYWAI | -18 | pyroglutamate |
| 21 | RAKFKQLL | - | |
| 23 | QAKWRLQTL | -17 | pyroglutamate |
| 24 | SDEEEAIVAYTL | - | |
| 27 | RRIYDLIEL | - | |
| 28 | YPLHEQHGM | - | |
| 29 | IPSINVHHY | - | |
| 30 | EENLLDFVRF | -18 | pyroglutamate |
| 31 | EFFWDANDIY | -18 | pyroglutamate |
|
Peptide No. |
Relative Area (UV) [%] |
| 1 | 82 |
| 1-H2O | 18 |
| 31 | 87 |
| 31 pGlu | 13 |
| 16 | 92 |
| 16+O | 8 |
| 4 | 94 |
| 4+O | 6 |
| 6 | 71 |
| 6 Dimer | 6 |
| 6 Dimer+O | 5 |
| 17+6 | 9 |
| 26+6 | n.a. |
| 2+6+O | n.a. |
| 2+6 | n.a. |
| 17+6+O | n.a. |
| 6+5 | n.a. |
| 6+5+O | n.a. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




