Submitted:
08 December 2024
Posted:
10 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
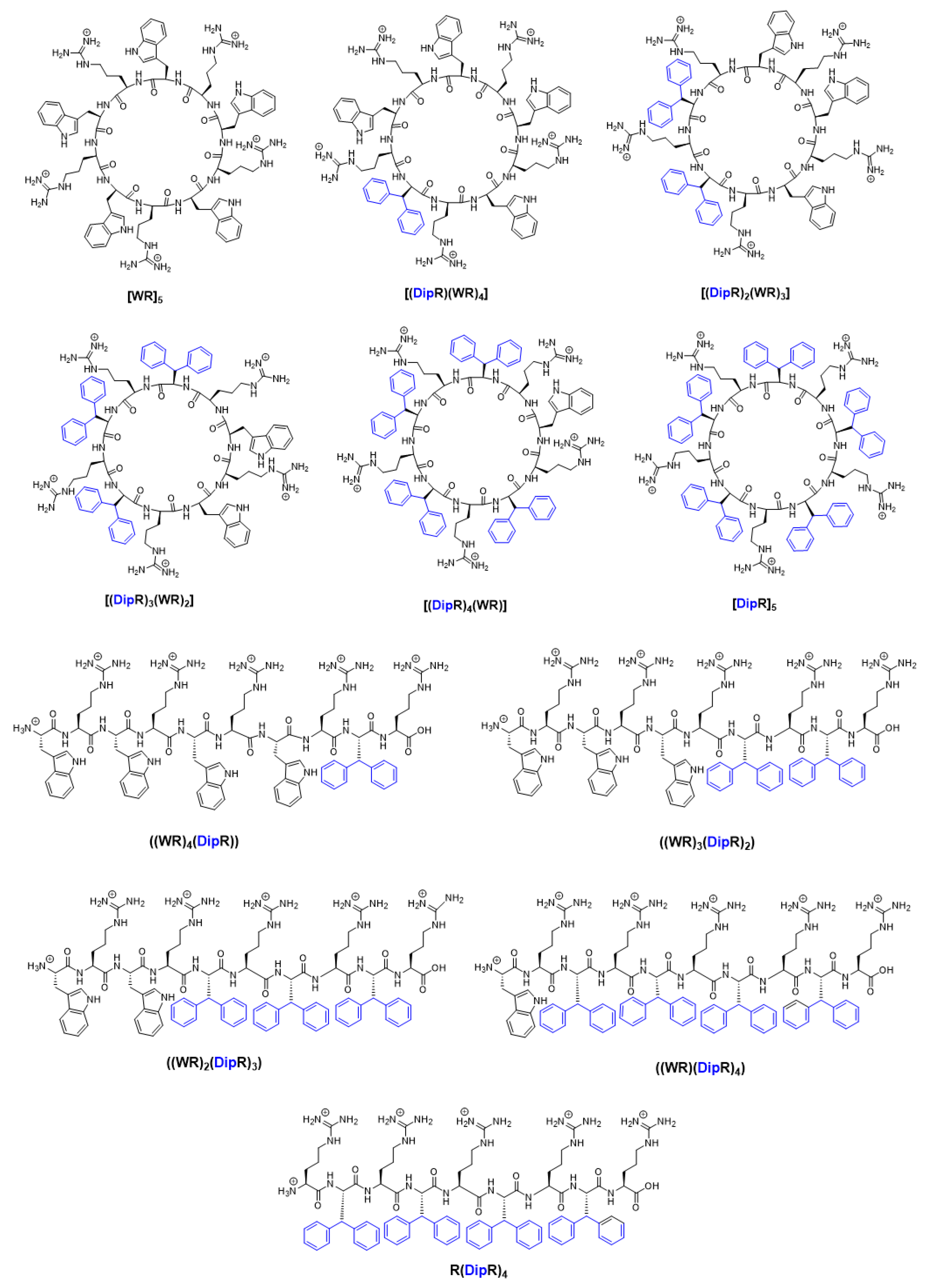
2. Results and Discussion
2.1. Antibacterial Activity
2.2. Antifungal Activity
3. Materials and Methods
3.1. Antibacterial Assays
3.2. Antifungal Assays
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raphael, E.; Riley, L.W. Infections Caused by Antimicrobial Drug-Resistant Saprophytic Gram-Negative Bacteria in the Environment. Front Med (Lausanne) 2017, 4, 183. [CrossRef]
- Zhabiz, G.; Omar, B.; Donald Gene, P. Bacteriophage therapy: a potential solution for the antibiotic resistance crisis. The Journal of Infection in Developing Countries 2014, 8. [CrossRef]
- Hand, K. Antibiotic stewardship. Clin Med (Lond) 2013, 13, 499-503. [CrossRef]
- Control, C.f.D. Biggest Threats and Data. Available online: https://www.cdc.gov/drugresistance/biggest-threats.html (accessed on 01/11/2021).
- Watson, R. Multidrug resistance responsible for half of deaths from healthcare associated infections in Europe. BMJ 2008, 336, 1266-1267. [CrossRef]
- Control, C.f.D. Methicillin-resistant Staphylococcus aureus (MRSA). Available online: https://www.cdc.gov/mrsa/community/index.html (accessed on 11/15/2020).
- Tong, S.Y.C.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G., Jr. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clinical microbiology reviews 2015, 28, 603-661. [CrossRef]
- Control, C.f.D. Klebsiella pneumoniae in Healthcare Settings. Available online: https://www.cdc.gov/hai/organisms/klebsiella/klebsiella.html (accessed on 11/15/2020).
- Control, C.f.D. Pseudomonas aeruginosa in Healthcare Settings. Available online: https://www.cdc.gov/hai/organisms/pseudomonas.html (accessed on 11/15/2020).
- Allocati, N.; Masulli, M.; Alexeyev, M.F.; Di Ilio, C. Escherichia coli in Europe: an overview. Int J Environ Res Public Health 2013, 10, 6235-6254. [CrossRef]
- Wade, J.J. Enterococcus faecium in hospitals. Eur J Clin Microbiol Infect Dis 1997, 16, 113-119. [CrossRef]
- Gross, P.A.; Harkavy, L.M.; Barden, G.E.; Flower, M.F. The epidemiology of nosocomial enterococcal urinary tract infection. Am J Med Sci 1976, 272, 75-81. [CrossRef]
- Emori, T.G.; Gaynes, R.P. An overview of nosocomial infections, including the role of the microbiology laboratory. Clin Microbiol Rev 1993, 6, 428-442. [CrossRef]
- Tuomanen, E.I. Microbiology and pathogenesis of Streptococcus pneumoniae. Available online: www.uptodate.com (accessed on 11/15/2020).
- Brogden, K.A. Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nature Reviews Microbiology 2005, 3, 238-250. [CrossRef]
- Raheem, N.; Straus, S.K. Mechanisms of action for antimicrobial peptides with antibacterial and antibiofilm functions. Frontiers in microbiology 2019, 10, 2866. [CrossRef]
- Woodburn, K.W.; Jaynes, J.M.; Clemens, L.E. Evaluation of the antimicrobial peptide, RP557, for the broad-spectrum treatment of wound pathogens and biofilm. Frontiers in Microbiology 2019, 10, 1688. [CrossRef]
- Harder, J.; Bartels, J.; Christophers, E.; Schroder, J.M. Isolation and characterization of human beta -defensin-3, a novel human inducible peptide antibiotic. J Biol Chem 2001, 276, 5707-5713. [CrossRef]
- Hancock, R.E.; Lehrer, R. Cationic peptides: a new source of antibiotics. Trends Biotechnol 1998, 16, 82-88. [CrossRef]
- Ong, P.Y.; Ohtake, T.; Brandt, C.; Strickland, I.; Boguniewicz, M.; Ganz, T.; Gallo, R.L.; Leung, D.Y.M. Endogenous Antimicrobial Peptides and Skin Infections in Atopic Dermatitis. New England Journal of Medicine 2002, 347, 1151-1160. [CrossRef]
- Krause, A.; Neitz, S.; Mägert, H.J.; Schulz, A.; Forssmann, W.G.; Schulz-Knappe, P.; Adermann, K. LEAP-1, a novel highly disulfide-bonded human peptide, exhibits antimicrobial activity. FEBS Lett 2000, 480, 147-150. [CrossRef]
- de Paula, V.S.; Razzera, G.; Barreto-Bergter, E.; Almeida, F.C.; Valente, A.P. Portrayal of complex dynamic properties of sugarcane defensin 5 by NMR: multiple motions associated with membrane interaction. Structure 2011, 19, 26-36. [CrossRef]
- Mohammed, E.H.M.; Lohan, S.; Tiwari, R.K.; Parang, K. Amphiphilic cyclic peptide [W4KR5]-Antibiotics combinations as broad-spectrum antimicrobial agents. Eur. J. Med. Chem. 2022, 235, 114278. [CrossRef]
- Salehi, D.; Mozaffari, S.; Zoghebi, K.; Lohan, S.; Mandal, D.; Tiwari, R.K.; Parang, K. Amphiphilic Cell-Penetrating Peptides Containing Natural and Unnatural Amino Acids as Drug Delivery Agents. Cells 2022, 11, 1156. [CrossRef]
- Mozaffari, S.; Salehi, D.; Mahdipoor, P.; Beuttler, R.; Tiwari, R.; Aliabadi, H.M.; Parang, K. Design and application of hybrid cyclic-linear peptide-doxorubicin conjugates as a strategy to overcome doxorubicin resistance and toxicity. Eur J Med Chem 2021, 226, 113836. [CrossRef]
- Riahifard, N.; Mozaffari, S.; Aldakhil, T.; Nunez, F.; Alshammari, Q.; Alshammari, S.; Yamaki, J.; Parang, K.; Tiwari, R.K. Design, Synthesis, and Evaluation of Amphiphilic Cyclic and Linear Peptides Composed of Hydrophobic and Positively-Charged Amino Acids as Antibacterial Agents. Molecules 2018, 23. [CrossRef]
- Oh, D.; Sun, J.; Nasrolahi Shirazi, A.; LaPlante, K.L.; Rowley, D.C.; Parang, K. Antibacterial activities of amphiphilic cyclic cell-penetrating peptides against multidrug-resistant pathogens. Mol Pharm 2014, 11, 3528-3536. [CrossRef]
- Hernandez, J.; Ashley, D.; Cao, R.; Abrahem, R.; Nguyen, T.; To, K.; Yegiazaryan, A.; Akinwale David, A.; Kumar Tiwari, R.; Venketaraman, V. Cyclic peptide [R4W4] in improving the ability of first-line antibiotics to inhibit Mycobacterium tuberculosis inside in vitro human granulomas. Frontiers in Immunology 2020, 11, 1677. [CrossRef]
- El-Mowafi, S.A.; Konshina, A.G.; Mohammed, E.H.M.; Krylov, N.A.; Efremov, R.G.; Parang, K. Structural Analysis and Activity Correlation of Amphiphilic Cyclic Antimicrobial Peptides Derived from the [W(4)R(4)] Scaffold. Molecules 2023, 28. [CrossRef]
- Akinwale, A.D.; Parang, K.; Tiwari, R.K.; Yamaki, J. Mechanistic Study of Antimicrobial Effectiveness of Cyclic Amphipathic Peptide [R(4)W(4)] against Methicillin-Resistant Staphylococcus aureus Clinical Isolates. Antibiotics (Basel) 2024, 13. [CrossRef]
- Riahifard, N.; Tavakoli, K.; Yamaki, J.; Parang, K.; Tiwari, R. Synthesis and Evaluation of Antimicrobial Activity of [R₄W₄K]-Levofloxacin and [R₄W₄K]-Levofloxacin-Q Conjugates. Molecules 2017, 22. [CrossRef]
- Lohan, S.; Mandal, D.; Choi, W.; Konshina, A.G.; Tiwari, R.K.; Efremov, R.G.; Maslennikov, I.; Parang, K. Small Amphiphilic Peptides: Activity Against a Broad Range of Drug-Resistant Bacteria and Structural Insight into Membranolytic Properties. J Med Chem 2022, 65, 665-687. [CrossRef]
- Darwish, S.; Mozaffari, S.; Mohammed, E.; Mahdieh, Z.; Tiwari, R.K.; Parang, K. Synthesis and biological evaluation of Bicyclic Peptides containing Arginine and Tryptophan residues. Chemical Biology Letters 2024, 11, 678. [CrossRef]
- Salehi, D.; Mozaffari, S.; Zoghebi, K.; Lohan, S.; Mandal, D.; Tiwari, R.K.; Parang, K. Amphiphilic Cell-Penetrating Peptides Containing Natural and Unnatural Amino Acids as Drug Delivery Agents. Cells 2022, 11. [CrossRef]
| MRSA (ATCC BAA-1556) | S. aureus (ATCC 29213) | E. faecium (ATCC 700221) | S. pneumoniae (ATCC 51938) | E. faecalis (ATCC 29212) | B. subtilis (ATCC-6633) | K. pneumoniae (ATCC BAA-1705) | P. aeruginosa (ATCC 27883) | E. Coli (ATCC 25922) | |
|---|---|---|---|---|---|---|---|---|---|
| MIC (µM)a | |||||||||
| ((DipR)4(WR)) | 3.1 | 3.1 | 1.6 | 0.78 | 1.6 | 12.5 | 25 | 25 | 12.5 |
| ((DipR)3(WR)2) | 3.1 | 3.1 | 3.1 | 0.78 | 3.1 | 3.1 | 25 | 25 | 12.5 |
| ((DipR)2(WR)3) | 3.1 | 3.1 | 3.1 | 0.78 | 6.3 | 1.6 | 12.5 | 12.5 | 6.3 |
| ((DipR)(WR)4) | 6.3 | 6.3 | 3.1 | 0.78 | 12.5 | 3.1 | 12.5 | 25 | 12.5 |
| (DipR)4R | 1.6 | 1.6 | 1.6 | 0.39 | 3.1 | 1.6 | 6.3 | 12.5 | 6.3 |
| [(DipR)4(WR)] | 3.1 | NTb | NT | NT | NT | NT | 25 | 25 | 12.5 |
| [(DipR)3(WR)2] | 3.1 | NT | NT | NT | NT | NT | 12.5 | 25 | 12.5 |
| [(DipR)2(WR)3] | 3.1 | NT | NT | NT | NT | NT | 12.5 | 25 | 12.5 |
| [(DipR)(WR)4] | 3.1 | NT | NT | NT | NT | NT | 12.5 | 25 | 12.5 |
| [DipR]5 | 3.1 | 3.13 | 0.78 | 0.39 | 0.78 | 6.3 | 25 | 25 | 12.5 |
| Daptomycin | 1.2 | 0.62 | 1.2 | 4.9 | 9.87 | 0.30 | NAc | NA | NA |
| Meropenem | 5.2 | 0.50 | 83 | 0.60 | 5.20 | 0.60 | 42 | 2.6 | 2.6 |
|
C. albicans (ATCC 60193) |
C. parapsilosis (ATCC 22019) |
A. fumigatus (Af-293) |
|
|---|---|---|---|
| MIC (µM)a | |||
| ((DipR)4(WR)) | 6.6 | 6.6 | 1.6 |
| ((DipR)3(WR)2) | 3.4 | 6.8 | 1.7 |
| ((DipR)2(WR)3) | 3.5 | 6.9 | 3.5 |
| ((DipR)(WR)4) | 28.2 | 14.1 | 3.5 |
| (DipR)4R | 7.4 | 7.4 | 1.8 |
| [(DipR)4(WR)] | 6.7 | 6.7 | 1.7 |
| [(DipR)3(WR)2] | 6.8 | 6.8 | 1.7 |
| [(DipR)2(WR)3] | 7.0 | 7.0 | 1.7 |
| [(DipR)(WR)4] | 14.3 | 7.1 | 1.8 |
| [DipR]5 | 13.1 | 6.6 | 1.6 |
| Fluconazole | 4.9 | 3.3 | >209 |
| Amphotericin B | 0.42 | 0.42 | 0.84 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




