Introduction
During the last few years much evidence has accrued to show that the action potential as originally described by Hodgkin and Huxley [
1] (HH) is accompanied by a synchronized, coupled soliton pressure pulse in the membrane known as the action the action potential pulse (APPulse) [
2,
3,
4,
5,
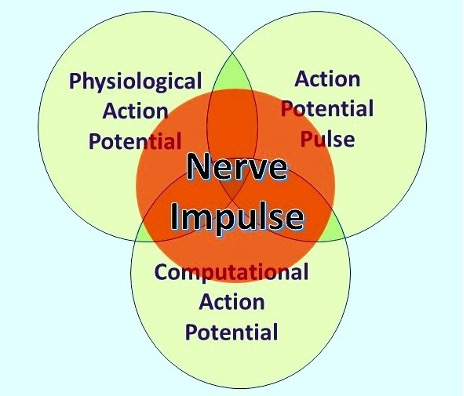
6] Furthermore, action potentials are an ensemble three separate states (
Figure 1): the HH physiological action potential (AP); a non-binary computational element, the computational action potential (CAP) [
4,
6,
7,
8,
9,
10] as described by Winlow and Johnson [
2,
7], and a soliton pulse [
11,
12], which we describe as the action potential pulse (APPulse). The speed of action potential conduction is defined by the exact properties of the membrane through which the pulse travels using the concomitant entropy (released energy) from the physiological action potential. The conduction velocity, and therefore the latency of the AP, is therefore fixed to the properties of the membrane. Collisions between converging APs cancel according to the properties described by HH and the pulse structure. This adaptation and combination of both the HH and Heimburg\Tasaki [11.12] theories is essential to explain the higher and previously unconsidered functioning of neurons such as computation, especially within a brain neural network.
From a physiological point of view, it is clear that synaptic transmission allows the HH action potential to pass from one neuron to another, but the question is, how does the APPulse pass from one cell to the next or is it generated de novo by each cell as it passes through a chain of neurons in the central nervous system. We think the latter suggestion to be unlikely as it would introduce delays in passing information from one cell to another. Another solution would be a physical connection between pre- and postsynaptic neurons and the most likely candidates to provide this are, in our opinion, the astrocytes found at central synapses in the nervous system.
Synapses
Synapses function at a slower rate than action potentials, with latencies typically in milliseconds or up to100ms where inhibitory synapses are involved [
13]. However, they remain stable over the computational limits of the APPulse, i.e. in microseconds. Synapses are inherently plastic, varying latencies with time and activity, making any form of computation error prone if timing is of the same order and there is no feasible opportunity to redact error; in neural networks, quantal frequency synchronisation permits computation within the limits of plasticity and redacts error: this must take place during timing when plasticity is fixed in microseconds or less.
The probable role of astrocytes in physically connecting pre and postsynaptic neurons
Some glial cells function primarily as a scaffold for the physical support for neurons. Astrocytes are the most abundant types of neuroglia and neuroglia in general have been described as “living nerve glue” by Kettenmann and Verkhratsky [
14]. While neurons have been extensively studied over many decades, the glial cells less so, both types of cells co-evolved [
14,
15]. Astrocytes have been shown to form tripartite synapses [
16] throughout the CNS [
15] and are known to interact with neurons by releasing neurotransmitters [
16] often modulating neuronal intracellular calcium release and thus transmitter release from presynaptic terminals [
17].
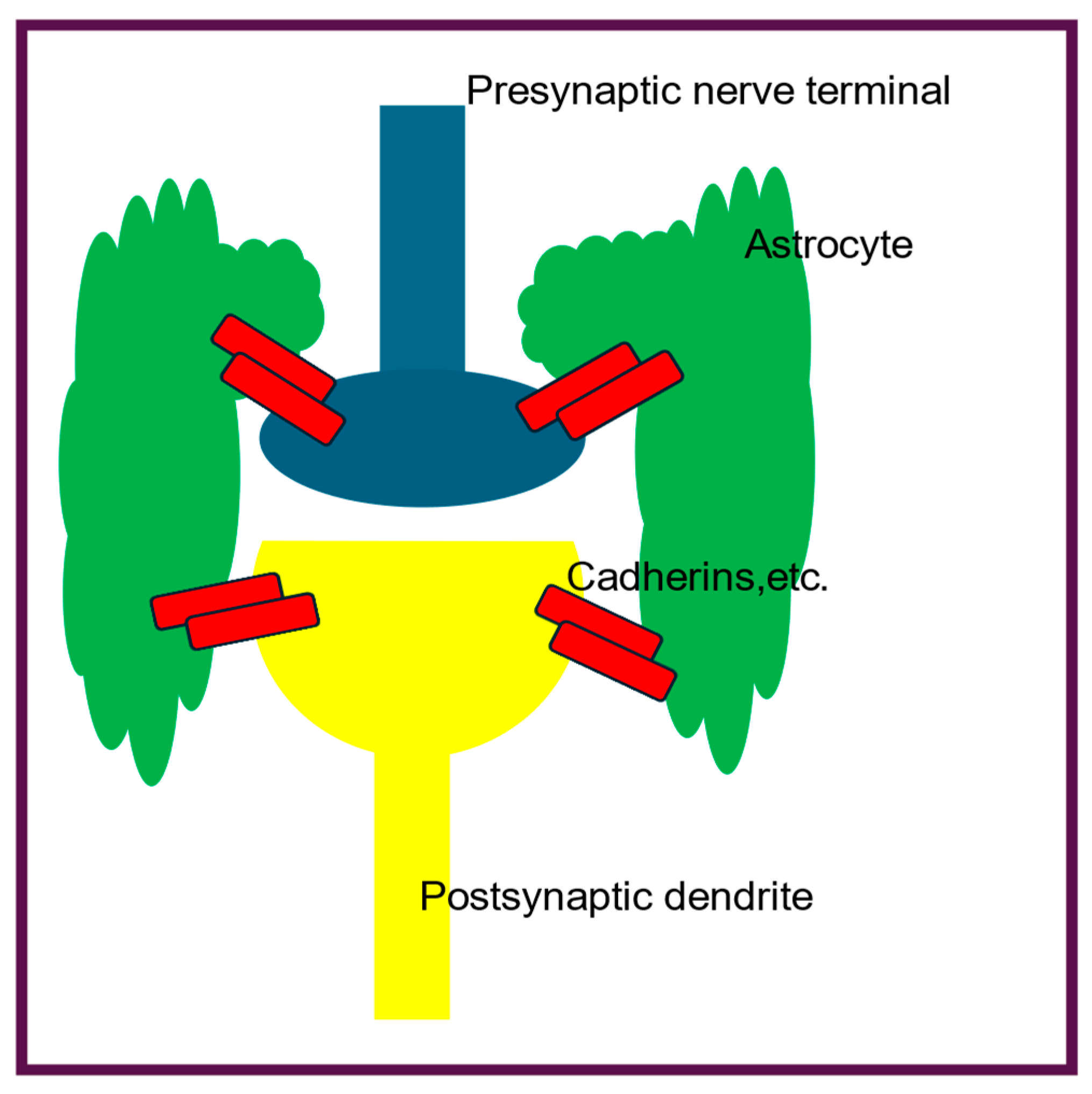
Here we consider the astrocytes found at central synapses where they form specific connections between presynaptic axon terminals and postsynaptic dendrites via synaptic adhesion molecules (SAMS), thus effectively gluing them together and preventing stress damage. Thus they may also have a previously undescribed, additional role to play in the passage of the APPulse at the tripartite synapses, where they physically connect the pre- and post-synaptic neurons (
Figure 2) via cadehrins and nectins. Any physical movement in the presynaptic cell will be physically transmitted to the postsynaptic cell via the astrocyte. If this is the case, it would allow physical transmission of the APPulse from the presynaptic nerve terminal to postsynaptic dendrites.
Cadherins and nectins
Cadherins were first described by Takeichi [
18] and are transmembrane proteins mediating cell-cell adhesion. At synapses they are termed synaptic adhesion molecules and contact pre- and post-synaptic cells at
puncta adherentia junctions (PAJs), thus providing an extracellular skeleton holding the cells tightly together and preventing separation due to physical stress (
Figure 2). Nectins also have similar functions at PAJs [
19]
Discussion
Synapses are an essential part of the process of perception and motor control, in that they have finite control of latencies over the 0.3ms to 100ms slower than parallel processing which in the retina occurs at 10-6s (7). Synapses separate cellularly diverse structures where each neuron, its shape morphology and position histologically has a functional basis. Synapses provide changing latency permitting memory loops to align and form abstract patterns. For example, object representation in the retina and LGN from large object forms a pattern in the visual cortex L1 and L2 changing to detail L5 which are assimilated into the pyramidal neurons which synchronise from L2 to L5 and output accurately final objects in L6 where synchronisation occurs with L1 from parallel output from the LGN. This redistribution of circulating synchronised memory is essential in learning when considering the cerebral cortex and is predetermined by the plasticity inherent in the synapses which permits redistribution according to object representation. The action of astrocytes surrounding and adhering to synapses increases the span of latency timings and changes the neural networks by raising the concentrations and effectiveness of neurotransmitters. The close-joints of cadherins etc and protein structure may also facilitate a direct transmission of impulses by mechanical impulses described by the APPulse, reducing the latencies, similar to gap junctions, and increasing uniform stable transmission. This concurs with our theory that synapses act as changers of latency in an inherently plastic environment. Thus, a brain neural network is by no means fixed in functional form but adjusts according to activity.
We have shown that propagation must occur through mechanical means and the only available candidate is the well-documented mechanical pulse that accompanies the HH AP. This pulse is ubiquitous whether the neuron is spiking or is spikeless, as have been described in the eye and elsewhere [
7]. That the pulse is propagated mechanically is evident from the timing constraints of the demonstrated computation in the eye [
7] and must take place mathematically within 10
-6s: without this timing synchronisation and thus computation, as demonstrated [
7] cannot be reliable and accurate. This is the predicted precision of timing between the start of propagation from the beginning of the soliton to the opening of the ionic gates.
Working hypothesis
The action potential pulse may be mechanically transmitted between neurons by astrocytes connected to pre- and postsynaptic neurons by cadherins and nectins.
Conflict of interest
The authors received no external support for this work and have no conflicts of interest.
References
- Hodgkin AL, Huxley AF. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. 1952; 117, 500-544. [CrossRef]
- Winlow W and Johnson AS. Nerve Impulses Have Three Interdependent Functions: Communication, Modulation, and Computation. Bioelectricity. 2021.161-170. [CrossRef]
- Johnson AS, Winlow W. The soliton and the action potential: primary elements underlying sentience. Frontiers in Physiology. 2018;9:779. [CrossRef]
- Johnson AS, Winlow W. Does the brain function as a quantum phase computer using phase ternary computation? Frontiers in Physiology 2021; 12, article 572041. [CrossRef]
- Winlow W, Johnson, AS. The action potential peak is not suitable for computational modelling and coding in the brain. EC Neurology 2020; 12.4: 46-48.
- Johnson AS, Winlow W. Computing action potentials by phase interference in realistic neural networks”. EC Neurology 2017;5.3: 123-134.
- Johnson AS and Winlow W. (2019). Are Neural Transactions in the Retina Performed by Phase Ternary Computation? Annals of Behavioural Neuroscience, 2(1), 223-236. [CrossRef]
- Johnson AS. The Coupled Action Potential Pulse (APPulse)–Neural Network Efficiency from a Synchronised Oscillating Lipid Pulse Hodgkin Huxley Action Potential. EC Neurology 2015;2: 94-101.
- Winlow W, Fatemi R, Johnson AS. Classical and Non-Classical Neural Communications. OBM Neurobiology 2023; 7(3): 181; [CrossRef]
- Winlow, W. The plastic nature of action potentials. In: The cellular basis of neuronal plasticity – physiology, morphology and biochemistry of molluscan neurons, Ed A.G.M. Bulloch, 1989; 3-27. Manchester University Press, UK.
- Heimburg T, Jackson, AD. On soliton propagation in biomembranes and nerves. Proc Natl Acad Sci USA. 2005;102: 9790–9795. [CrossRef]
- El Hady A, Machta B. Mechanical Surface Waves Accompany Action Potential Propagation. Nature Communications 2015; 6: 6697. [CrossRef]
- Katz B, Miledi R.1965. The measurement of synaptic delay, and the time course of acetylcholine release at the neuromuscular junction. Proc. R. Soc. Lond. B.161483-495. [CrossRef]
- Kettenmann H, Hanisch UK, Noda M, Verkhratsky A. Physiology of microglia. Physiol Rev. 2011 Apr;91(2):461-553. [CrossRef] [PubMed]
- Togashi, H., Sakisaka, T., & Takai, Y. (2009). Cell adhesion molecules in the central nervous system. Cell Adhesion &Amp; Migration, 3(1), 29-35. [CrossRef]
- Araque A, Parpura V, Sanzgirl RP, Haydon PG. Tripartite synapses: glia the unacknowledged partner. TINS, 1999, 208-215.
- Brini M, Calì T, Ottolini D, Carafoli E. Neuronal calcium signaling function and dysfunction. Cell Mol Life Sci. 2014 Aug;71(15):2787-814. [CrossRef] [PubMed] [PubMed Central]
- Takeichi, M. Functional correlation between cell adhesive properties and some cell surface proteins. J Cell Biol. 1977, 75(2 Pt 1):464-74. [CrossRef]
- Mizutani, R., Saiga, R., Yamamoto, Y. et al. Structural diverseness of neurons between brain areas and between cases. Transl Psychiatry 11, 49 (2021). [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).